Synthetic or Food-Derived Vitamin C—Are They Equally Bioavailable?
Abstract
:1. Introduction
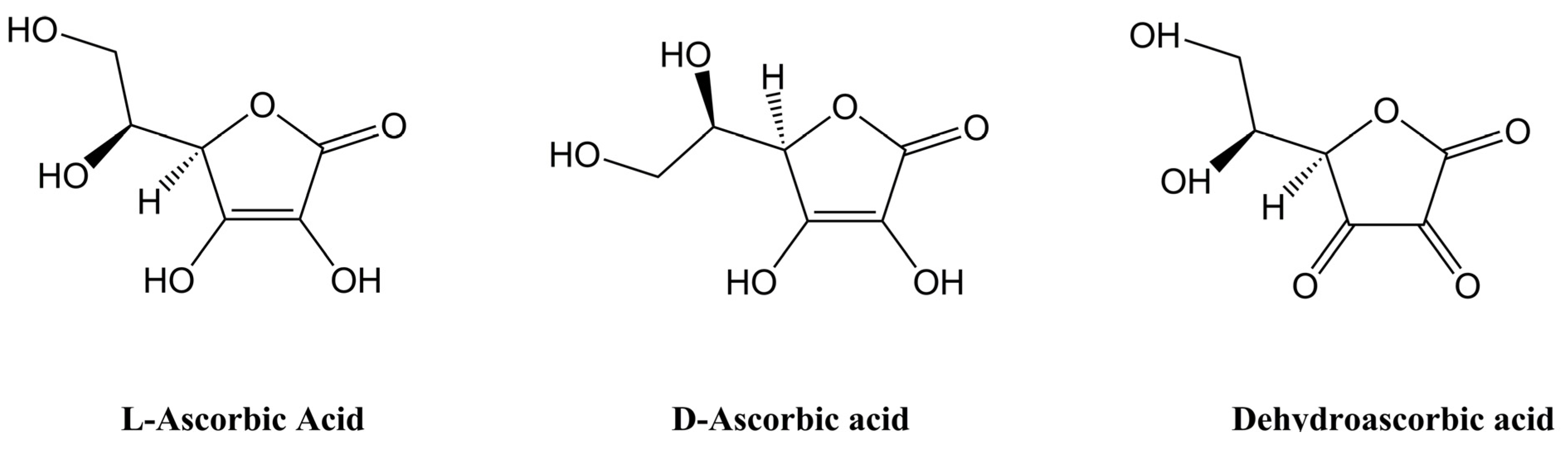
2. Vitamin C Bioavailability Studies Using Animal Models
3. Steady State Bioavailability Studies in Humans
4. Pharmacokinetic Bioavailability Studies in Humans
| Animal Model | Intervention | Study Design | Vitamin C Analysis | Bioavailability Findings: Natural vs. Synthetic Vitamin C | Bioavailability Summary: Natural vs. Synthetic Vitamin C | Reference |
|---|---|---|---|---|---|---|
| Gulo−/− mice | 0.5–5 mg/day vitamin C solution | 4 weeks intervention | HPLC-ECD | Kiwifruit ↑ serum, leukocyte, heart, liver, and kidney, but not brain vitamin C | Enhanced uptake in 5/6 pools | [68] |
| Kiwifruit gel | ||||||
| CD rats | 60 mg/kg vitamin C gavage | Single dose; 4 h sampling | HPLC-ECD | Quercetin ↓ plasma vitamin C (at 4 h) | Decreased uptake in 1/1 pool | [54] |
| 15 mg/kg quercetin | ||||||
| Guinea pigs | 50 mg vitamin C solution | Single dose; 4 h sampling | Fluorometric (NQSA) | Citrus fruit media ↑ plasma AUC | Enhanced uptake in 1/1 pool | [65] |
| Citrus fruit media | ||||||
| 1 mg/kg vitamin C (low vitamin C diet) | 26 days intervention | Colorimetric (DCPIP) | Orange peel extract ↑ adrenal, spleen and leukocyte, but not brain vitamin C; hesperidin ↑ adrenal and leukocyte, but not spleen vitamin C | Enhanced uptake in 3/4 pools | [61] | |
| 50 mg/kg orange peel extract | ||||||
| 50 mg/kg hesperidin | ||||||
| 5 mg/kg vitamin C solution | 23 days intervention | Colorimetric (DCPIP) | Black current juice ↑ adrenal and spleen vitamin C; acerola cherry juice comparable adrenal and spleen vitamin C | Enhanced uptake in 2/2 organs | [64] | |
| Black current juice | ||||||
| Acerola cherry juice | ||||||
| 0, 5 and 10 mg/kg vitamin C | 3 weeks intervention | Colorimetric (DNPH) | Rutin ↑ adrenal, but not liver vitamin C | Enhanced uptake in 1/2 pools | [62] | |
| 50 mg rutin | ||||||
| 4 mg/kg vitamin C (low vitamin C diet) | 22 days intervention | Colorimetric (DNPH) | Rutin ↑ adrenal, but not liver or whole blood vitamin C of adequate animals | Enhanced uptake in 1/3 pools | [63] | |
| 10 mg rutin tablet | ||||||
| 18 mg/kg vitamin C (adequate vitamin C diet) | ||||||
| 10 mg rutin tablet | ||||||
| Basic diet | 23 days intervention | Colorimetric (DCPIP) | Vitamin C + catechin ↑ liver, spleen, kidney, and adrenal vitamin C | Enhanced uptake in 4/4 organs | [42] | |
| 1 mg/animal catechin | ||||||
| 10 mg/animal vitamin C | ||||||
| Vitamin C + catechin | ||||||
| 0.5 mg/day vitamin C solution | 20 days intervention | Colorimetric (DCPIP) | Lemon juice comparable plasma and adrenal vitamin C | Comparable uptake in 2/2 pools | [67] | |
| 1 mL lemon juice |
| Subjects | Intervention | Study Design | Vitamin C Analysis | Bioavailability Findings: Natural vs. Synthetic Vitamin C | Bioavailability Summary: Natural vs. Synthetic Vitamin C | Reference |
|---|---|---|---|---|---|---|
| 36 non-smoking males 18–35 years | 50 mg/day vitamin C tablet | 6 weeks of supplementation; Parallel design | HPLC-ECD | Kiwifruit comparable plasma, urine, semen, mononuclear cell, neutrophil and muscle tissue vitamin C | Comparable uptake in 6/6 pools | [77] |
| Gold kiwifruit (50 mg vitamin C) | ||||||
| 11 non-smoking women 21–39 years | 69 mg/day vitamin C capsule | 2 weeks of supplementation; Crossover design (2 week washout) | Colorimetric (DNPH) | Orange juice comparable plasma vitamin C | Comparable uptake in 1/1 pool | [76] |
| Orange juice (66 mg vitamin C) | ||||||
| 68 non-smoking males 30–59 years | 108 mg/day vitamin C tablet | 4 weeks of supplementation; Crossover design (4 week washout) | Colorimetric (DNPH) | Orange pieces/juice or cooked broccoli comparable plasma vitamin C; raw broccoli ↓ plasma vitamin C | Comparable uptake in 1/1 pool | [35] |
| Orange—pieces or juice | ||||||
| Broccoli—cooked or raw | ||||||
| 14 men and women | 75 mg/day vitamin C | Sequential design | Colorimetric | Papayas and guava juice comparable plasma and urinary vitamin C | Comparable uptake in 2/2 pools | [72] |
| Papayas (75 mg/day vitamin C) | ||||||
| Guava juice (75 mg/day vitamin C) | ||||||
| 4 healthy young subjects | 75 mg/day vitamin C tablets | Pre-study saturation; Sequential design | Colorimetric | Raw cabbage and tomato juice comparable plasma and urinary vitamin C | Comparable uptake in 2/2 pools | [73] |
| Raw cabbage (75 mg/day vitamin C) | ||||||
| Tomato juice (75 mg/day vitamin C) | ||||||
| 7 college women | 40 mg/day vitamin C solution | Pre-study saturation; Sequential design | Colorimetric (DCPIP) | Raspberries comparable blood and urinary vitamin C | Comparable uptake in 2/2 pools | [74] |
| Red raspberries (40 mg/day vitamin C) | ||||||
| 12 young adults | 100 mg/day vitamin C | Sequential design | Colorimetric (DCPIP) | Orange juice comparable urinary vitamin C | Comparable uptake in 1/1 pool | [75] |
| Orange juice (100 mg/day vitamin C) |
| Subjects | Intervention | Study Design | Vitamin C Analysis | Plasma Uptake | Urinary Excretion | Reference |
|---|---|---|---|---|---|---|
| 9 non-smoking males 18–35 years | 200 mg vitamin C tablet | 8 h sampling; Crossover design (3 week washout) | HPLC-ECD | Kiwifruit comparable plasma vitamin C and AUC | Kiwifruit ↑ urinary vitamin C and AUC (relative to creatinine) | [89] |
| Gold kiwifruit (200 mg vitamin C) | ||||||
| 5 non-smoking males 22–27 years | 50 mg vitamin C solution; | 8 h sampling; Crossover design (4 week washout) | HPLC-ECD | Mashed potatoes ↓ plasma vitamin C (at 1 to 2.5 h); potato chips ↓ AUC | Mashed potatoes ↓ urinary vitamin C (at 3 h) | [84] |
| 282 g mashed potato (50 mg vitamin C) | ||||||
| 87 g potato chips (50 mg vitamin C) | ||||||
| Placebo | ||||||
| 5 non-smoking males 22–26 years | 50–500 mg vitamin C solution | 6 h sampling; Crossover design | HPLC-ECD | Acerola juice comparable plasma vitamin C and AUC | Acerola juice ↓ urinary vitamin C (at 1, 2 and 5 h) | [85] |
| 100 mL acerola juice (50 mg vitamin C) | ||||||
| 12 males 20–35 years | 284 mg vitamin C drink | 4.5 h sampling; Crossover design (1 week washout) | Colorimetric (TPTZ) | Orange juice comparable bioavailability (AUC/concentration) | ND | [86] |
| 590 mL orange juice (68 mg vitamin C) | ||||||
| Placebo (milk) | ||||||
| 7 non-smoking females | 150 mg vitamin C solution | 8 h sampling; Crossover design (2 week washout) | HPLC-UV | Orange juice comparable plasma vitamin C | ND | [87] |
| 300 mL orange juice (150 mg vitamin C) | ||||||
| Placebo | ||||||
| 7 non-smokers 26–59 years | 30 mg vitamin C solution | 4 h sampling; Crossover design (3–4 week washout) | Fluorometric (phenylene diamine) | Grape juice ↓ plasma vitamin C (at 16 to 28 min) | ND | [34] |
| 200 mL red grape juice (30 mg vitamin C) | ||||||
| 9 healthy subjects 19–41 years | 500 mg vitamin C tablet | 1 g/day vitamin C for 2 weeks pre-study; 8 h sampling; Crossover design (1 week washout) | Colorimetric (DNPH) | Bioflavonoids comparable AUC | Bioflavonoids comparable 24 h vitamin C excretion | [83] |
| Mixed bioflavonoids | ||||||
| Placebo | ||||||
| 12 non-smoking subjects 18–41 years | 500 mg vitamin C solution | Subgroup had 1 g/day vitamin C for 2 weeks pre-study; 8 h sampling; Crossover design (1 week washout) | Fluorometric | Citrus extract ↑ AUC | Citrus extract ↓ 24 h vitamin C excretion in non-saturated subjects and ↑ 24 h vitamin C excretion in saturated subjects | [81] |
| 2 g citrus extract | ||||||
| Placebo | ||||||
| 5 men 21–25 years | 500 mg vitamin C solution | 100 mg/day vitamin C for 1 month pre-study; 8 h sampling; Crossover design (1 week washout) | Colorimetric (Indophenol dye) | ND | Blackcurrant juice slight ↑ 8 h vitamin C excretion in saturated subjects | [82] |
| 500 mg vitamin C in blackcurrant juice | ||||||
| 15 normal subjects (4 smokers) 20–42 years | 70 mg/h vitamin C solution | Intestinal perfusion; Tandem design | Colorimetric (DNPH) | Orange juice comparable intestinal absorption | ND | [88] |
| Orange juice | ||||||
| 12 men (6 smokers) 23–44 years | 75 mg vitamin C solution | Pre- and post-saturation with 1 mg/day vitamin C; 2–24 h sampling; Crossover design (1 day washout) | Colorimetric | Orange juice and rutin ↓ plasma vitamin C (at 2 h) | Orange juice slight ↑ 24 h vitamin C excretion | [71] |
| 400 mg rutin | ||||||
| Orange juice (75 mg vitamin C) |
5. Vitamin C Bioavailability from Different Tablet Formulations
6. Conclusions
Conflicts of Interest
Acknowledgments
References
- Carr, A.C.; Frei, B. Toward a new recommended dietary allowance for vitamin C based on antioxidant and health effects in humans. Am. J. Clin. Nutr. 1999, 69, 1086–1107. [Google Scholar]
- Englard, S.; Seifter, S. The biochemical functions of ascorbic acid. Annu. Rev. Nutr. 1986, 6, 365–406. [Google Scholar]
- Ozer, A.; Bruick, R.K. Non-heme dioxygenases: Cellular sensors and regulators jelly rolled into one? Nat. Chem. Biol. 2007, 3, 144–153. [Google Scholar] [CrossRef]
- Monfort, A.; Wutz, A. Breathing-in epigenetic change with vitamin C. EMBO Rep. 2013, 14, 337–346. [Google Scholar]
- Hornig, D. Distribution of ascorbic acid, metabolites and analogues in man and animals. Ann. N. Y. Acad. Sci. 1975, 258, 103–118. [Google Scholar]
- Omaye, S.T.; Schaus, E.E.; Kutnink, M.A.; Hawkes, W.C. Measurement of vitamin C in blood components by high-performance liquid chromatography. Implication in assessing vitamin C status. Ann. N. Y. Acad. Sci. 1987, 498, 389–401. [Google Scholar]
- Tsao, C.S. An Overview of Ascorbic Acid Chemistry and Biochemistry. In Vitamin C in Health and Disease; Packer, L., Fuchs, J., Eds.; Marcel Dekker: New York, NY, USA, 1997; pp. 25–58. [Google Scholar]
- Nishikimi, M.; Fukuyama, R.; Minoshima, S.; Shimizu, N.; Yagi, K. Cloning and chromosomal mapping of the human nonfunctional gene for l-gulono-γ-lactone oxidase, the enzyme for l-ascorbic acid biosynthesis missing in man. J. Biol. Chem. 1994, 269, 13685–13688. [Google Scholar]
- Krebs, H.A. The Sheffield Experiment on the vitamin C requirement of human adults. Proc. Nutr. Soc. 1953, 12, 237–246. [Google Scholar]
- Sauberlich, H.E. A History of Scurvy and Vitamin C. In Vitamin C in Health and Disease; Packer, L., Fuchs, J., Eds.; Marcel Dekker: New York, NY, USA, 1997; pp. 1–24. [Google Scholar]
- Michels, A.J.; Hagen, T.M.; Frei, B. Human genetic variation influences vitamin C homeostasis by altering vitamin C transport and antioxidant enzyme function. Annu. Rev. Nutr. 2013, 33, 45–70. [Google Scholar]
- Tsukaguchi, H.; Tokui, T.; Mackenzie, B.; Berger, U.V.; Chen, X.Z.; Wang, Y.; Brubaker, R.F.; Hediger, M.A. A family of mammalian Na+-dependent l-ascorbic acid transporters. Nature 1999, 399, 70–75. [Google Scholar]
- Savini, I.; Rossi, A.; Pierro, C.; Avigliano, L.; Catani, M.V. SVCT1 and SVCT2: Key proteins for vitamin C uptake. Amino Acids 2008, 34, 347–355. [Google Scholar]
- Rumsey, S.C.; Welch, R.W.; Garraffo, H.M.; Ge, P.; Lu, S.F.; Crossman, A.T.; Kirk, K.L.; Levine, M. Specificity of ascorbate analogs for ascorbate transport. Synthesis and detection of [(125)I]6-deoxy-6-iodo-l-ascorbic acid and characterization of its ascorbate-specific transport properties. J. Biol. Chem. 1999, 274, 23215–23222. [Google Scholar]
- Goldman, H.M.; Gould, B.S.; Munro, H.N. The antiscorbutic action of l-ascorbic acid and d-isoascorbic acid (erythorbic acid) in the guinea pig. Am. J. Clin. Nutr. 1981, 34, 24–33. [Google Scholar]
- Hughes, R.E.; Hurley, R.J. The uptake of d-araboascorbic acid (d-isoascorbic acid) by guinea-pig tissues. Br. J. Nutr. 1969, 23, 211–216. [Google Scholar]
- Levine, M. Fruits and vegetables: There is no substitute. Am. J. Clin. Nutr. 1996, 64, 381–382. [Google Scholar]
- Sauberlich, H.E.; Tamura, T.; Craig, C.B.; Freeberg, L.E.; Liu, T. Effects of erythorbic acid on vitamin C metabolism in young women. Am. J. Clin. Nutr. 1996, 64, 336–346. [Google Scholar]
- Corpe, C.P.; Eck, P.; Wang, J.; Al-Hasani, H.; Levine, M. Intestinal dehydroascorbic acid (DHA) transport mediated by the facilitative sugar transporters, GLUT2 and GLUT8. J. Biol. Chem. 2013, 288, 9092–9101. [Google Scholar]
- Rumsey, S.C.; Kwon, O.; Xu, G.W.; Burant, C.F.; Simpson, I.; Levine, M. Glucose transporter isoforms GLUT1 and GLUT3 transport dehydroascorbic acid. J. Biol. Chem. 1997, 272, 18982–18989. [Google Scholar]
- Vera, J.C.; Rivas, C.I.; Fischbarg, J.; Golde, D.W. Mammalian facilitative hexose transporters mediate the transport of dehydroascorbic acid. Nature 1993, 364, 79–82. [Google Scholar]
- Washko, P.W.; Wang, Y.; Levine, M. Ascorbic acid recycling in human neutrophils. J. Biol. Chem. 1993, 268, 15531–15535. [Google Scholar]
- Corti, A.; Casini, A.F.; Pompella, A. Cellular pathways for transport and efflux of ascorbate and dehydroascorbate. Arch. Biochem. Biophys. 2010, 500, 107–115. [Google Scholar]
- Nishiyama, I.; Yamashita, Y.; Yamanaka, M.; Shimohashi, A.; Fukuda, T.; Oota, T. Varietal difference in vitamin C content in the fruit of kiwifruit and other actinidia species. J. Agric. Food Chem. 2004, 52, 5472–5475. [Google Scholar]
- Dhariwal, K.R.; Hartzell, W.O.; Levine, M. Ascorbic acid and dehydroascorbic acid measurements in human plasma and serum. Am. J. Clin. Nutr. 1991, 54, 712–716. [Google Scholar]
- Ogiri, Y.; Sun, F.; Hayami, S.; Fujimura, A.; Yamamoto, K.; Yaita, M.; Kojo, S. Very low vitamin C activity of orally administered l-dehydroascorbic acid. J. Agric. Food Chem. 2002, 50, 227–229. [Google Scholar]
- Welch, R.W.; Wang, Y.; Crossman, A., Jr.; Park, J.B.; Kirk, K.L.; Levine, M. Accumulation of vitamin C (ascorbate) and its oxidized metabolite dehydroascorbic acid occurs by separate mechanisms. J. Biol. Chem. 1995, 270, 12584–12592. [Google Scholar]
- Wang, Y.; Russo, T.A.; Kwon, O.; Chanock, S.; Rumsey, S.C.; Levine, M. Ascorbate recycling in human neutrophils: Induction by bacteria. Proc. Natl. Acad. Sci. USA 1997, 94, 13816–13819. [Google Scholar]
- Packer, J.E.; Slater, T.F.; Willson, R.L. Direct observation of a free radical interaction between vitamin E and vitamin C. Nature 1979, 278, 737–738. [Google Scholar]
- Tanaka, K.; Hashimoto, T.; Tokumaru, S.; Iguchi, H.; Kojo, S. Interactions between vitamin C and vitamin E are observed in tissues of inherently scorbutic rats. J. Nutr. 1997, 127, 2060–2064. [Google Scholar]
- Beck, K.; Conlon, C.A.; Kruger, R.; Coad, J.; Stonehouse, W. Gold kiwifruit consumed with an iron-fortified breakfast cereal meal improves iron status in women with low iron stores: A 16-week randomised controlled trial. Br. J. Nutr. 2011, 105, 101–109. [Google Scholar]
- Hallberg, L.; Brune, M.; Rossander, L. Effect of ascorbic acid on iron absorption from different types of meals. Studies with ascorbic-acid-rich foods and synthetic ascorbic acid given in different amounts with different meals. Hum. Nutr. Appl. Nutr. 1986, 40, 97–113. [Google Scholar]
- Scheers, N.M.; Sandberg, A.S. Iron regulates the uptake of ascorbic acid and the expression of sodium-dependent vitamin C transporter 1 (SVCT1) in human intestinal Caco-2 cells. Br. J. Nutr. 2011, 105, 1734–1740. [Google Scholar]
- Bates, C.J.; Jones, K.S.; Bluck, L.J. Stable isotope-labelled vitamin C as a probe for vitamin C absorption by human subjects. Br. J. Nutr. 2004, 91, 699–705. [Google Scholar]
- Mangels, A.R.; Block, G.; Frey, C.M.; Patterson, B.H.; Taylor, P.R.; Norkus, E.P.; Levander, O.A. The bioavailability to humans of ascorbic acid from oranges, orange juice and cooked broccoli is similar to that of synthetic ascorbic acid. J. Nutr. 1993, 123, 1054–1061. [Google Scholar]
- Keltz, F.R.; Kies, C.; Fox, H.M. Urinary ascorbic acid excretion in the human as affected by dietary fiber and zinc. Am. J. Clin. Nutr. 1978, 31, 1167–1171. [Google Scholar]
- Rusznyak, S.; Szent-Gyorgyi, A. Vitamin P: Flavonols as vitamins. Nature 1936, 138, 27. [Google Scholar]
- Bentsath, A.; Rusznyak, S.; Szent-Gyorgyi, A. Vitamin nature of flavones. Nature 1936, 138, 798. [Google Scholar]
- Bentsath, A.; Rusznyak, S.; Szent-Gyorgyi, A. Vitamin P. Nature 1937, 139, 326–327. [Google Scholar]
- Rusznyak, S.; Benko, A. Experimental vitamin P deficiency. Science 1941, 94, 25. [Google Scholar]
- Zilva, S.S. Vitamin P. Biochem. J. 1937, 31, 915–919. [Google Scholar]
- Cotereau, H.; Gabe, M.; Géro, E.; Parrot, J.-L. Influence of vitamin P (vitamin C2) upon the amount of ascorbic acid in the organs of the guinea pig. Nature 1948, 161, 557. [Google Scholar]
- Scarborough, H. Deficiency of vitamin C and vitamin P in man. Lancet 1940, 236, 644–647. [Google Scholar]
- Scarborough, H. Vitamin P. Biochem. J. 1939, 33, 1400–1407. [Google Scholar]
- Elmby, A.; Warburg, E. The inadequacy of synthetic ascorbic acid as an antiscorbutic agent. Lancet 1937, 230, 1363–1365. [Google Scholar]
- Ivanov, V.; Carr, A.C.; Frei, B. Red wine antioxidants bind to human lipoproteins and protect them from metal ion-dependent and -independent oxidation. J. Agric. Food Chem. 2001, 49, 4442–4449. [Google Scholar]
- Bors, W.; Michel, C.; Saran, M. Flavonoid antioxidants: rate constants for reactions with oxygen radicals. Methods Enzymol. 1994, 234, 420–429. [Google Scholar] [CrossRef]
- Beker, B.Y.; Sonmezoglu, I.; Imer, F.; Apak, R. Protection of ascorbic acid from copper(II)-catalyzed oxidative degradation in the presence of flavonoids: Quercetin, catechin and morin. Int. J. Food Sci. Nutr. 2011, 62, 504–512. [Google Scholar]
- Clemetson, C.A.; Andersen, L. Plant polyphenols as antioxidants for ascorbic acid. Ann. N. Y. Acad Sci. 1966, 136, 341–376. [Google Scholar] [CrossRef]
- Harper, K.A.; Morton, A.D.; Rolfe, E.J. The phenolic compounds of blackcurrant juice and their protective effect on ascorbic acid III. The mechansim of ascorbic acid oxidation and its inhibition by flavonoids. Int. J. Food Sci. Technol. 1969, 4, 255–267. [Google Scholar]
- Clegg, K.M.; Morton, A.D. The phenolic compounds of blackcurrant juice and their protective effect on ascorbic acid II. The stability of ascorbic acid in model systems containing some of the phenolic compounds associated with blackcurrant juice. Int. J. Food Sci. Technol. 1968, 3, 277–284. [Google Scholar]
- Carr, A.; Frei, B. Does vitamin C act as a pro-oxidant under physiological conditions? FASEB J. 1999, 13, 1007–1024. [Google Scholar]
- Lotito, S.B.; Frei, B. Consumption of flavonoid-rich foods and increased plasma antioxidant capacity in humans: Cause, consequence, or epiphenomenon? Free Radi. Biol. Med. 2006, 41, 1727–1746. [Google Scholar]
- Song, J.; Kwon, O.; Chen, S.; Daruwala, R.; Eck, P.; Park, J.B.; Levine, M. Flavonoid inhibition of sodium-dependent vitamin C transporter 1 (SVCT1) and glucose transporter isoform 2 (GLUT2), intestinal transporters for vitamin C and Glucose. J. Biol. Chem. 2002, 277, 15252–15260. [Google Scholar]
- Park, J.B.; Levine, M. Intracellular accumulation of ascorbic acid is inhibited by flavonoids via blocking of dehydroascorbic acid and ascorbic acid uptakes in HL-60, U937 and Jurkat cells. J. Nutr. 2000, 130, 1297–1302. [Google Scholar]
- Corpe, C.P.; Lee, J.H.; Kwon, O.; Eck, P.; Narayanan, J.; Kirk, K.L.; Levine, M. 6-Bromo-6-deoxy-l-ascorbic acid: An ascorbate analog specific for Na+-dependent vitamin C transporter but not glucose transporter pathways. J. Biol. Chem. 2005, 280, 5211–5220. [Google Scholar]
- Washko, P.W.; Welch, R.W.; Dhariwal, K.R.; Wang, Y.; Levine, M. Ascorbic acid and dehydroascorbic acid analyses in biological samples. Anal. Biochem. 1992, 204, 1–14. [Google Scholar]
- Mizushima, Y.; Harauchi, T.; Yoshizaki, T.; Makino, S. A rat mutant unable to synthesize vitamin C. Experientia 1984, 40, 359–361. [Google Scholar]
- Maeda, N.; Hagihara, H.; Nakata, Y.; Hiller, S.; Wilder, J.; Reddick, R. Aortic wall damage in mice unable to synthesize ascorbic acid. Proc. Natl. Acad. Sci. USA 2000, 97, 841–846. [Google Scholar]
- Mohan, S.; Kapoor, A.; Singgih, A.; Zhang, Z.; Taylor, T.; Yu, H.; Chadwick, R.B.; Chung, Y.S.; Donahue, L.R.; Rosen, C.; et al. Spontaneous fractures in the mouse mutant sfx are caused by deletion of the gulonolactone oxidase gene, causing vitamin C deficiency. J. Bone Miner. Res. 2005, 20, 1597–1610. [Google Scholar]
- Wilson, H.K.; Price-Jones, C.; Hughes, R.E. The influence of an extract of orange peel on the growth and ascorbic acid metabolism of young guinea-pigs. J. Sci. Food Agric. 1976, 27, 661–666. [Google Scholar]
- Douglass, C.D.; Kamp, G.H. The effect of orally administered rutin on the adrenal ascorbic acid level in guinea pigs. J. Nutr. 1959, 67, 531–536. [Google Scholar]
- Papageorge, E.; Mitchell, G.L., Jr. The effect of oral administration of rutin on blood, liver and adrenal ascorbic acid and on liver and adrenal cholesterol in guinea pigs. J. Nutr. 1949, 37, 531–540. [Google Scholar]
- Hughes, R.E.; Hurley, R.J.; Jones, P.R. The retention of ascorbic acid by guinea-pig tissues. Br. J. Nutr. 1971, 26, 433–438. [Google Scholar] [CrossRef]
- Vinson, J.A.; Bose, P. Comparative bioavailability of synthetic and natural vitamin C in guinea pigs. Nutr. Rep. Int. 1983, 27, 875–879. [Google Scholar]
- Ambrose, A.M.; De, E.F. The value of rutin and quercetin in scurvy. J. Nutr. 1949, 38, 305–317. [Google Scholar]
- Todhunter, E.N.; Robbins, R.C.; Ivey, G.; Brewer, W. A comparison of the utilization by guinea pigs of equivalent amounts of ascorbic acid (vitamin C) in lemon juice and in crystalline form. J. Nutr. 1940, 19, 113–120. [Google Scholar]
- Vissers, M.C.M.; Bozonet, S.M.; Pearson, J.F.; Braithwaite, L.J. Dietary ascorbate affects steady state tissue levels in vitamin C-deficient mice: Tissue deficiency after sub-optimal intake and superior bioavailability from a food source (kiwifruit). Am. J. Clin. Nutr. 2011, 93, 292–301. [Google Scholar]
- Latocha, P.; Krupa, T.; Wolosiak, R.; Worobiej, E.; Wilczak, J. Antioxidant activity and chemical difference in fruit of different Actinidia sp. Int J. Food Sci. Nutr. 2010, 61, 381–394. [Google Scholar]
- Fiorentino, A.; D’Abrosca, B.; Pacifico, S.; Mastellone, C.; Scognamiglio, M.; Monaco, P. Identification and assessment of antioxidant capacity of phytochemicals from kiwi fruits. J. Agric. Food Chem. 2009, 57, 4148–4155. [Google Scholar]
- Pelletier, O.; Keith, M.O. Bioavailability of synthetic and natural ascorbic acid. J. Am. Diet. Assoc. 1974, 64, 271–275. [Google Scholar]
- Hartzler, E.R. The availability of ascorbic acid in papayas and guavas. J. Nutr. 1945, 30, 355–365. [Google Scholar]
- Clayton, M.M.; Borden, R.A. The availability for human nutrition of the vitamin C in raw cabbage and home-canned tomato juice. J. Nutr. 1943, 25, 349–369. [Google Scholar]
- Todhunter, E.N.; Fatzer, A.S. A comparison of the utilization by college women of equivalent amounts of ascorbic acid (vitamin C) in red raspberries and in crystalline form. J. Nutr. 1940, 19, 121–130. [Google Scholar]
- Hawley, E.E.; Stephens, D.J.; Anderson, G. The excretion of vitamin C in normal individuals following a comparable quantitative administration in the form of orange juice, cevitamic acid by mouth and cevitamic acid intravenously. J. Nutr. 1936, 11, 135–145. [Google Scholar]
- Johnston, C.S.; Dancho, C.L.; Strong, G.M. Orange juice ingestion and supplemental vitamin C are equally effective at reducing plasma lipid peroxidation in healthy adult women. J. Am. Coll. Nutr. 2003, 22, 519–523. [Google Scholar]
- Carr, A.C.; Bozonet, S.M.; Pullar, J.M.; Simcock, J.W.; Vissers, M.C. A randomised steady-state bioavailability study of synthetic and natural (kiwifruit-derived) vitamin C. Nutrients 2013, 5, 3684–3695. [Google Scholar]
- Levine, M.; Conry-Cantilena, C.; Wang, Y.; Welch, R.W.; Washko, P.W.; Dhariwal, K.R.; Park, J.B.; Lazarev, A.; Graumlich, J.F.; King, J.; et al. Vitamin C pharmacokinetics in healthy volunteers: Evidence for a recommended dietary allowance. Proc. Natl. Acad. Sci. USA 1996, 93, 3704–3709. [Google Scholar]
- Block, G.; Mangels, A.R.; Patterson, B.H.; Levander, O.A.; Norkus, E.P.; Taylor, P.R. Body weight and prior depletion affect plasma ascorbate levels attained on identical vitamin C intake: A controlled-diet study. J. Am. Coll. Nutr. 1999, 18, 628–637. [Google Scholar]
- Carr, A.C.; Bozonet, S.M.; Pullar, J.M.; Simcock, J.W.; Vissers, M.C. Human skeletal muscle ascorbate is highly responsive to changes in vitamin C intake and plasma concentrations. Am. J. Clin. Nutr. 2013, 97, 800–807. [Google Scholar]
- Vinson, J.A.; Bose, P. Comparative bioavailability to humans of ascorbic acid alone or in a citrus extract. Am. J. Clin. Nutr. 1988, 48, 601–604. [Google Scholar]
- Jones, E.; Hughes, R.E. The influence of bioflavonoids on the absorption of vitamin C. IRCS J. Med. Sci. 1984, 12, 320. [Google Scholar]
- Johnston, C.S.; Luo, B. Comparison of the absorption and excretion of three commercially available sources of vitamin C. J. Am. Diet. Assoc. 1994, 94, 779–781. [Google Scholar]
- Kondo, Y.; Higashi, C.; Iwama, M.; Ishihara, K.; Handa, S.; Mugita, H.; Maruyama, N.; Koga, H.; Ishigami, A. Bioavailability of vitamin C from mashed potatoes and potato chips after oral administration in healthy Japanese men. Br. J. Nutr. 2012, 107, 885–892. [Google Scholar]
- Uchida, E.; Kondo, Y.; Amano, A.; Aizawa, S.; Hanamura, T.; Aoki, H.; Nagamine, K.; Koizumi, T.; Maruyama, N.; Ishigami, A. Absorption and excretion of ascorbic acid alone and in acerola (Malpighia emarginata) juice: Comparison in healthy japanese subjects. Biol. Pharm. Bull. 2011, 34, 1744–1747. [Google Scholar]
- Carter, B.; Monsivais, P.; Drewnowski, A. Absorption of folic acid and ascorbic acid from nutrient comparable beverages. J. Food Sci. 2010, 75, H289–H293. [Google Scholar]
- Guarnieri, S.; Riso, P.; Porrini, M. Orange juice vs vitamin C: Effect on hydrogen peroxide-induced DNA damage in mononuclear blood cells. Br. J. Nutr. 2007, 97, 639–643. [Google Scholar]
- Nelson, E.W.; Streiff, R.R.; Cerda, J.J. Comparative bioavailability of folate and vitamin C from a synthetic and a natural source. Am. J. Clin. Nutr. 1975, 28, 1014–1019. [Google Scholar]
- Carr, A.C.; Bozonet, S.M.; Vissers, M.C.M. A randomised cross-over pharmacokinetic bioavailabiltiy study of synthetic versus kiwifruit-derived vitamin C. Nutrients 2013, 5, 3684–3695. [Google Scholar]
- Hathcock, J.N.; Azzi, A.; Blumberg, J.; Bray, T.; Dickinson, A.; Frei, B.; Jialal, I.; Johnston, C.S.; Kelly, F.J.; Kraemer, K.; et al. Vitamins E and C are safe across a broad range of intakes. Am. J. Clin. Nutr. 2005, 81, 736–745. [Google Scholar]
- Yung, S.; Mayersohn, M.; Robinson, J.B. Ascorbic acid absorption in humans: A comparison among several dosage forms. J. Pharm. Sci. 1982, 71, 282–285. [Google Scholar]
- Bhagavan, H.N.; Wolkoff, B.I. Correlation between the disintegration time and the bioavailability of vitamin C tablets. Pharm. Res. 1993, 10, 239–242. [Google Scholar]
- Sacharin, R.; Taylor, T.; Chasseaud, L.F. Blood levels and bioavailability of ascorbic acid after administration of a sustained-release formulation to humans. Int. J. Vitam. Nutr. Res. 1977, 47, 68–74. [Google Scholar]
- Nyyssonen, K.; Poulsen, H.E.; Hayn, M.; Agerbo, P.; Porkkala-Sarataho, E.; Kaikkonen, J.; Salonen, R.; Salonen, J.T. Effect of supplementation of smoking men with plain or slow release ascorbic acid on lipoprotein oxidation. Eur. J. Clin. Nutr. 1997, 51, 154–163. [Google Scholar]
- Bush, M.J.; Verlangieri, A.J. An acute study on the relative gastro-intestinal absorption of a novel form of calcium ascorbate. Res. Commun. Chem. Pathol. Pharmacol. 1987, 57, 137–140. [Google Scholar]
- Verlangieri, A.J.; Fay, M.J.; Bannon, A.W. Comparison of the anti-scorbutic activity of l-ascorbic acid and Ester C in the non-ascorbate synthesizing Osteogenic Disorder Shionogi (ODS) rat. Life Sci. 1991, 48, 2275–2281. [Google Scholar]
- Gruenwald, J.; Graubaum, H.J.; Busch, R.; Bentley, C. Safety and tolerance of ester-C compared with regular ascorbic acid. Adv. Ther. 2006, 23, 171–178. [Google Scholar]
- Knight, A. Systematic reviews of animal experiments demonstrate poor human clinical and toxicological utility. Altern. Lab. Anim. 2007, 35, 641–659. [Google Scholar]
- Seok, J.; Warren, H.S.; Cuenca, A.G.; Mindrinos, M.N.; Baker, H.V.; Xu, W.; Richards, D.R.; McDonald-Smith, G.P.; Gao, H.; Hennessy, L.; et al. Genomic responses in mouse models poorly mimic human inflammatory diseases. Proc. Natl. Acad. Sci. USA 2013, 110, 3507–3512. [Google Scholar]
- He, F.J.; Nowson, C.A.; MacGregor, G.A. Fruit and vegetable consumption and stroke: Meta-analysis of cohort studies. Lancet 2006, 367, 320–326. [Google Scholar]
- Dauchet, L.; Amouyel, P.; Hercberg, S.; Dallongeville, J. Fruit and vegetable consumption and risk of coronary heart disease: A meta-analysis of cohort studies. J. Nutr. 2006, 136, 2588–2593. [Google Scholar]
- Steinmetz, K.A.; Potter, J.D. Vegetables, fruit, and cancer prevention: A review. J. Am. Diet. Assoc. 1996, 96, 1027–1039. [Google Scholar]
- Riboli, E.; Norat, T. Epidemiologic evidence of the protective effect of fruit and vegetables on cancer risk. Am. J. Clin. Nutr. 2003, 78, 559S–569S. [Google Scholar]
- Block, G.; Norkus, E.; Hudes, M.; Mandel, S.; Helzlsouer, K. Which plasma antioxidants are most related to fruit and vegetable consumption? Am. J. Epidemiol. 2001, 154, 1113–1118. [Google Scholar]
- Mente, A.; de Koning, L.; Shannon, H.S.; Anand, S.S. A systematic review of the evidence supporting a causal link between dietary factors and coronary heart disease. Arch. Intern. Med. 2009, 169, 659–669. [Google Scholar]
- Ye, Z.; Song, H. Antioxidant vitamins intake and the risk of coronary heart disease: Meta-analysis of cohort studies. Eur. J. Cardiovasc. Prev. Rehabil. 2008, 15, 26–34. [Google Scholar]
- Knekt, P.; Ritz, J.; Pereira, M.A.; O’Reilly, E.J.; Augustsson, K.; Fraser, G.E.; Goldbourt, U.; Heitmann, B.L.; Hallmans, G.; Liu, S.; et al. Antioxidant vitamins and coronary heart disease risk: A pooled analysis of 9 cohorts. Am. J. Clin. Nutr. 2004, 80, 1508–1520. [Google Scholar]
- Lawlor, D.A.; Davey Smith, G.; Kundu, D.; Bruckdorfer, K.R.; Ebrahim, S. Those confounded vitamins: What can we learn from the differences between observational versus randomised trial evidence? Lancet 2004, 363, 1724–1727. [Google Scholar]
- Maserejian, N.N.; Giovannucci, E.L.; McVary, K.T.; McKinlay, J.B. Dietary, but not supplemental, intakes of carotenoids and vitamin C are associated with decreased odds of lower urinary tract symptoms in men. J. Nutr. 2011, 141, 267–273. [Google Scholar]
- Osganian, S.K.; Stampfer, M.J.; Rimm, E.; Spiegelman, D.; Hu, F.B.; Manson, J.E.; Willett, W.C. Vitamin C and risk of coronary heart disease in women. J. Am. Coll. Cardiol. 2003, 42, 246–252. [Google Scholar]
- Agarwal, M.; Mehta, P.K.; Dwyer, J.H.; Dwyer, K.M.; Shircore, A.M.; Nordstrom, C.K.; Sun, P.; Paul-Labrador, M.; Yang, Y.; Merz, C.N. Differing relations to early atherosclerosis between vitamin C from supplements vs. food in the Los Angeles atherosclerosis Study: A prospective cohort study. Open Cardiovasc. Med. J. 2012, 6, 113–121. [Google Scholar]
- Henriquez-Sanchez, P.; Sanchez-Villegas, A.; Doreste-Alonso, J.; Ortiz-Andrellucchi, A.; Pfrimer, K.; Serra-Majem, L. Dietary assessment methods for micronutrient intake: A systematic review on vitamins. Br. J. Nutr. 2009, 102, S10–S37. [Google Scholar]
- Thompson, F.E.; Byers, T. Dietary assessment resource manual. J. Nutr. 1994, 124, 2245S–2317S. [Google Scholar]
- Dehghan, M.; Akhtar-Danesh, N.; McMillan, C.R.; Thabane, L. Is plasma vitamin C an appropriate biomarker of vitamin C intake? A systematic review and meta-analysis. Nutr. J. 2007, 6, 41. [Google Scholar]
- Lykkesfeldt, J.; Poulsen, H.E. Is vitamin C supplementation beneficial? Lessons learned from randomised controlled trials. Br. J. Nutr. 2010, 103, 1251–1259. [Google Scholar]
- Carr, A.C.; Pullar, J.M.; Moran, S.; Vissers, M.C.M. Bioavailability of vitamin C from kiwifruit in non-smoking males: Determination of ‘healthy’ and ‘optimal’ intakes. J. Nutr. Sci. 2012, 1, e14. [Google Scholar]
- Franz, W.L.; Heyl, H.L.; Sands, G.W. Blood ascorbic acid level in bioflavonoid and ascorbic acid therapy of common cold. J. Am. Med. Assoc. 1956, 162, 1224–1226. [Google Scholar]
- Arminio, J.J.; Johnston, J.H.; Tebrock, H.E. Usefulness of bioflavonoids and ascorbic acid in treatment of common cold. J. Am. Med. Assoc. 1956, 162, 1227–1233. [Google Scholar]
- Baird, I.M.; Hughes, R.E.; Wilson, H.K.; Davies, J.E.; Howard, A.N. The effects of ascorbic acid and flavonoids on the occurrence of symptoms normally associated with the common cold. Am. J. Clin. Nutr. 1979, 32, 1686–1690. [Google Scholar]
- Stevenson, D.E.; Hurst, R.D. Polyphenolic phytochemicals—Just antioxidants or much more? Cell. Mol. Life Sci. 2007, 64, 2900–2916. [Google Scholar]
- Gao, Y.; Li, W.; Jia, L.; Li, B.; Chen, Y.C.; Tu, Y. Enhancement of (−)-epigallocatechin-3-gallate and theaflavin-3-3′-digallate induced apoptosis by ascorbic acid in human lung adenocarcinoma SPC-A-1 cells and esophageal carcinoma Eca-109 cells via MAPK pathways. Biochem. Biophys. Res. Commun. 2013, 438, 370–374. [Google Scholar]
- Calero, C.I.; Beltran Gonzalez, A.N.; Gasulla, J.; Alvarez, S.; Evelson, P.; Calvo, D.J. Quercetin antagonism of GAB receptors is prevented by ascorbic acid through a redox-independent mechanism. Eur. J. Pharmacol. 2013. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Carr, A.C.; Vissers, M.C.M. Synthetic or Food-Derived Vitamin C—Are They Equally Bioavailable? Nutrients 2013, 5, 4284-4304. https://doi.org/10.3390/nu5114284
Carr AC, Vissers MCM. Synthetic or Food-Derived Vitamin C—Are They Equally Bioavailable? Nutrients. 2013; 5(11):4284-4304. https://doi.org/10.3390/nu5114284
Chicago/Turabian StyleCarr, Anitra C., and Margreet C. M. Vissers. 2013. "Synthetic or Food-Derived Vitamin C—Are They Equally Bioavailable?" Nutrients 5, no. 11: 4284-4304. https://doi.org/10.3390/nu5114284
APA StyleCarr, A. C., & Vissers, M. C. M. (2013). Synthetic or Food-Derived Vitamin C—Are They Equally Bioavailable? Nutrients, 5(11), 4284-4304. https://doi.org/10.3390/nu5114284






