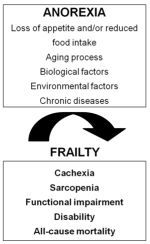
Anorexia of Aging: A Modifiable Risk Factor for Frailty
Abstract
:1. Introduction
2. Risk Factors for Anorexia of Aging
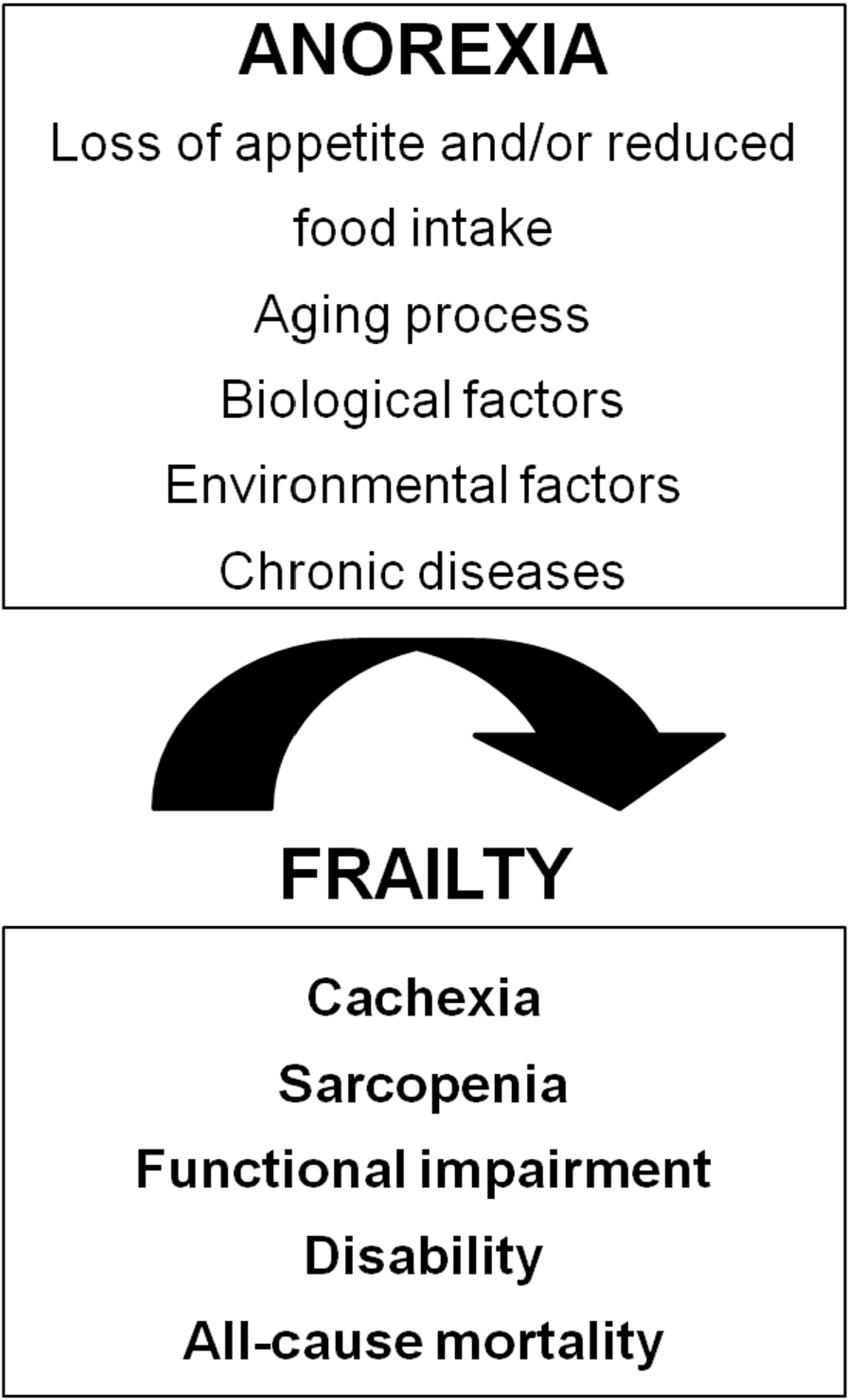
3. Consequences of Anorexia of Aging
4. Correlation among Anorexia, Sarcopenia and Frailty Syndrome
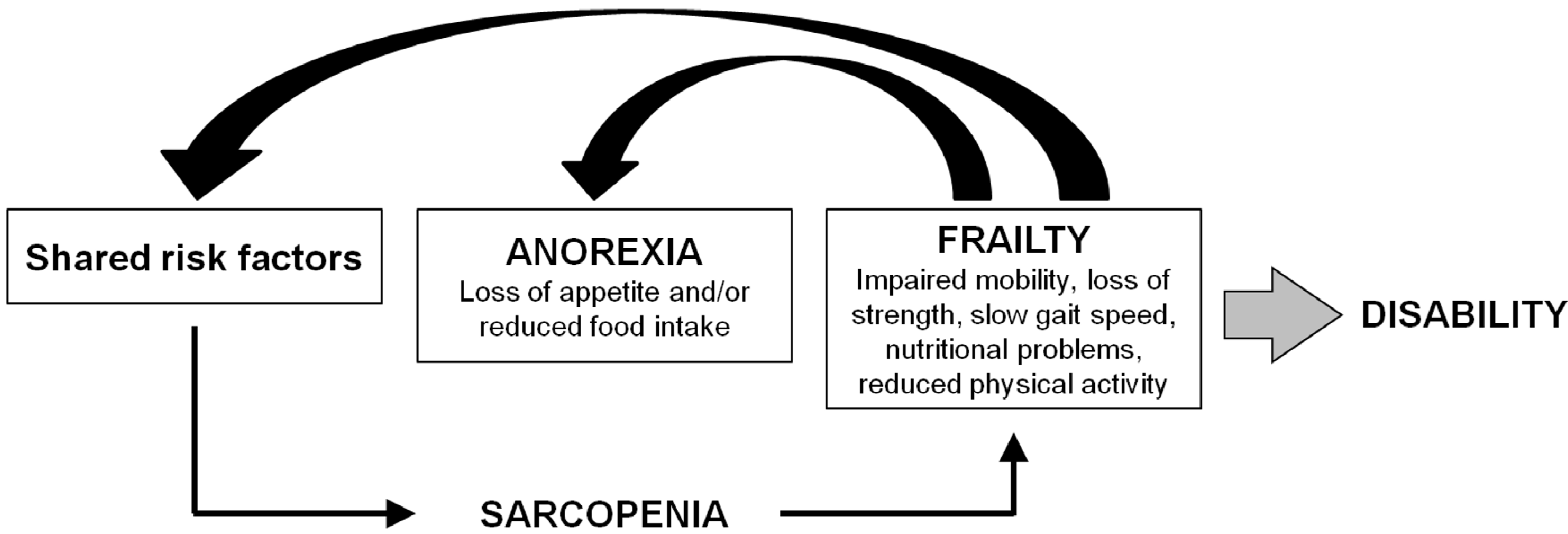
5. Conclusions
Conflicts of interest
References
- MacIntosh, C.; Morley, J.E.; Chapman, I.M. The anorexia of aging. Nutrition 2000, 16, 983–995. [Google Scholar] [CrossRef]
- Donini, L.M.; Savina, C.; Piredda, M.; Cucinotta, D.; Fiorito, A.; Inelmen, E.M.; Sergi, G.; Domiguez, L.J.; Barbagallo, M.; Cannella, C. Senile anorexia in acute-ward and rehabilitation settings. J. Nutr. Health Aging 2008, 12, 511–517. [Google Scholar] [CrossRef]
- Donini, L.M.; Dominguez, L.J.; Barbagallo, M.; Savina, C.; Castellaneta, E.; Cucinotta, D.; Fiorito, A.; Inelmen, E.M.; Sergi, G.; Enzi, G.; et al. Senile anorexia in different geriatric settings in Italy. J. Nutr. Health Aging 2011, 15, 775–781. [Google Scholar] [CrossRef]
- Morley, J.E. Anorexia of aging: A true geriatric syndrome. J. Nutr. Health Aging 2012, 16, 422–425. [Google Scholar] [CrossRef]
- Di Francesco, V.; Barazzoni, R.; Bissoli, L.; Fantin, F.; Rizzotti, P.; Residori, L.; Antonioli, A.; Graziani, M.S.; Zanetti, M.; Bosello, O.; et al. The quantity of meal fat influences the profile of postprandial hormones as well as hunger sensation in healthy elderly people. J. Am. Med. Dir. Assoc. 2010, 11, 188–193. [Google Scholar] [CrossRef]
- Horwitz, B.A.; Blanton, C.A.; McDonald, R.B. Physiologic determinants of the anorexia of aging: Insights from animal studies. Annu. Rev. Nutr. 2002, 22, 417–438. [Google Scholar] [CrossRef]
- Conte, C.; Cascino, A.; Bartali, B.; Donini, L.M.; Rossi-Fanelli, F.; Laviano, A. Anorexia of aging. Curr. Nutr. Food Sci. 2009, 5, 9–12. [Google Scholar] [CrossRef]
- Morley, J.E.; Anker, S.D.; Evans, W.J. Cachexia and aging: An update based on the Fourth International Cachexia Meeting. J. Nutr. Health Aging 2009, 13, 47–55. [Google Scholar] [CrossRef]
- Serra-Prat, M.; Palomera, E.; Clave, P.; Puig-Domingo, M. Effect of age and frailty on ghrelin and cholecystokinin responses to a meal test. Am. J. Clin. Nutr. 2009, 89, 1410–1417. [Google Scholar] [CrossRef]
- Laviano, A.; Meguid, M.M.; Inui, A.; Muscaritoli, M.; Rossi-Fanelli, F. Theraphy insight: Cancer anorexia-cachexia syndrome when all you can eat is yourself. Nat. Clin. Pract. Oncol. 2005, 2, 158–165. [Google Scholar]
- Toffanello, E.D.; Inelmen, E.M.; Imoscopi, A.; Perissinotto, E.; Coin, A.; Miotto, F.; Donini, L.M.; Cucinotta, D.; Barbagallo, M.; Manzato, E.; et al. Taste loss in hospitalized multimorbid elderly subjects. Clin. Interv. Aging 2013, 8, 167–174. [Google Scholar]
- Lee, J.S.; Kritchevsky, S.B.; Tylavsky, F.; Harris, T.B.; Ayonayon, H.N.; Newman, A.B. Factors associated with impaired appetite in well-functioning community-dwelling older adults. J. Nutr. Elder 2006, 26, 27–43. [Google Scholar] [CrossRef]
- Clarkston, W.K.; Pantano, M.M.; Morley, J.E.; Horowitz, M.; Littlefield, J.M.; Burton, F.R. Evidence for the anorexia of aging: Gastrointestinal transit and hunger in healthy elderly vs. young adults. Am. J. Physiol. 1997, 272, 243–248. [Google Scholar]
- Parker, B.A.; Chapman, I.M. Food intake and ageing-the role of the gut. Mech. Ageing Dev. 2004, 125, 859–866. [Google Scholar] [CrossRef]
- Grassi, M.; Petraccia, L.; Mennuni, G.; Fontana, M.; Scarno, A.; Sabetta, S.; Fraioli, A. Changes, functional disorders, and diseases in the gastrointestinal tract of elderly. Nutr. Hosp. 2011, 26, 659–668. [Google Scholar]
- Laugier, R.; Bernard, J.P.; Berthezene, P.; Dupuy, P. Changes in pancreatic exocrine secretion with age: Pancreatic exocrine secretion does decrease in the elderly. Digestion 1991, 50, 202–211. [Google Scholar] [CrossRef]
- Baumgartner, R.N.; Waters, D.L.; Morley, J.E.; Patrick, P.; Montoya, G.D.; Garry, P.J. Age-Related changes in sex hormones affect the sex difference in serum leptin independently of changes in body fat. Metabolism 1999, 48, 378–384. [Google Scholar] [CrossRef]
- Cabrera, M.A.; Mesas, A.E.; Garcia, A.R.; de Andrade, S.M. Malnutrition and depression among community-dwelling elderly people. J. Am. Med. Dir. Assoc. 2007, 8, 582–584. [Google Scholar] [CrossRef]
- Donini, L.M.; Poggiogalle, E.; Piredda, M.; Pinto, A.; Barbagallo, M.; Cucinotta, D.; Sergi, G. Anorexia and eating patterns in the elderly. PLoS One 2013, 8, e63539. [Google Scholar]
- Morley, J.E. Anorexia of aging: Physiologic and pathologic. Am. J. Clin. Nutr. 1997, 66, 760–773. [Google Scholar]
- Morley, J.E.; Thomas, D.R. Anorexia and aging: Pathophysiology. Nutrition 1999, 15, 499–503. [Google Scholar] [CrossRef]
- Landi, F.; Lattanzio, F.; Dell’Aquila, G.; Eusebi, P.; Gasperini, B.; Liperoti, R.; Belluigi, A.; Bernabei, R.; Cherubini, A. Prevalence and potentially reversible factors associated with anorexia among older nursing home residents. Results from the ULISSE project. J. Am. Med. Dir. Assoc. 2013, 14, 119–124. [Google Scholar] [CrossRef]
- Salva, A.; Coll-Planas, L.; Bruce, S.; de Groot, L.; Andrieu, S.; Abellan, G.; Vellas, B.; Andrieu, S.; Bartorelli, L.; Berner, Y.N.; et al. Nutritional assessment of residents in long-term care facilities (LTCFs): Recommendations of the task force on nutrition and ageing of the IAGG European region and the IANA. J. Nutr. Health Aging 2009, 13, 475–483. [Google Scholar] [CrossRef]
- Chapman, I.M.; MacIntosh, C.G.; Morley, J.E.; Horowitz, M. The anorexia of ageing. Biogerontology 2002, 3, 67–71. [Google Scholar] [CrossRef]
- Muscaritoli, M.; Anker, S.D.; Argilés, J.; Aversa, Z.; Bauer, J.M.; Biolo, G.; Boirie, Y.; Bosaeus, I.; Cederholm, T.; Costelli, P.; et al. Consensus definition of sarcopenia, cachexia and pre-cachexia: Joint document elaborated by Special Interest Group [SIG] “cachiexia-anorexia in chronic wasting diseases” and “nutrition in geriatrics”. Clin. Nutr. 2010, 29, 154–159. [Google Scholar] [CrossRef]
- Landi, F.; Liperoti, R.; Russo, A.; Giovannini, S.; Tosato, M.; Barillaro, C.; Capoluongo, E.; Bernabei, R.; Onder, G. Association of anorexia with sarcopenia in a community-dwelling elderly population: Results from il Sirente study. Eur. J. Nutr. 2012, 52, 1261–1268. [Google Scholar]
- Morley, J.E. Anorexia, sarcopenia and aging. Nutrition 2001, 17, 660–663. [Google Scholar] [CrossRef]
- Landi, F.; Laviano, A.; Cruz-Jentoft, A. The anorexia of aging: Is it a geriatric syndrome? J. Am. Med. Dir. Assoc. 2010, 11, 153–156. [Google Scholar] [CrossRef]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, 146–156. [Google Scholar] [CrossRef]
- Rieu, I.; Balage, M.; Sornet, C.; Giraudet, C.; Pujos, E.; Grizard, J.; Mosoni, L.; Dardevet, D. Leucine supplementation improve muscle protein synthesis in erderly men independently of hyperaminoacidaemia. J. Physiol. 2006, 575, 305–315. [Google Scholar] [CrossRef]
- Dreyer, H.C.; Drummond, M.J.; Pennings, B.; Fujita, S.; Glynn, E.L.; Chinkes, D.L.; Dhanani, S.; Volpi, E.; Rasmussen, B.B. Leucine enriched essential amino acid and carbohydrate ingestion following resistance exercise enhances mTOR signaling and protein synthesis in human muscle. Am. J. Physiol. Endocrionol. Metab. 2008, 294, 392–400. [Google Scholar]
- Calvani, R.; Miccheli, A.; Landi, F.; Bossola, M.; Cesari, M.; Leeuwenburgh, C.; Sieber, C.C.; Bernabei, R.; Marzetti, E. Current nutritional recommendations and novel dietary strategies to manage sarcopenia. J. Frailty Aging 2013, 2, 38–53. [Google Scholar]
- Gaffney-Stomberg, E.; Insogna, K.L.; Rodriguez, N.R.; Kerstetter, J.E. Increasing dietary protein requirements in elderly people for optimal muscle and bone health. J. Am. Geriatr. Soc. 2009, 57, 1073–1079. [Google Scholar] [CrossRef]
- Morley, J.E.; Argiles, J.M.; Evans, W.J.; Bhasin, S.; Cella, D.; Deutz, N.E.; Doehner, W.; Fearon, K.C.; Ferrucci, L.; Hellerstein, M.K.; et al. Nutritional recommendations for the management of sarcopenia. J. Am. Med. Dir. Assoc. 2010, 11, 391–396. [Google Scholar] [CrossRef]
- Wassner, S.J.; Li, J.B.; Sperduto, A.; Norman, M.E. Vitamin D deficiency, hypocalcemia and increate skeletal muscle degradation in rats. J. Clin. Investig. 1983, 72, 102–112. [Google Scholar] [CrossRef]
- Scott, D.; Blizzard, L.; Fell, J.; Ding, C.; Winzenberg, T.; Jones, G. A prospective study of the associations between 25-hydroxy-vitamin D, sarcopenia progression and physical activity in older adults. Clin. Endocrinol. 2010, 73, 581–587. [Google Scholar] [CrossRef]
- Montero-Odasso, M.; Duque, G. Vitamin D in the aging musculo-skeletal system: An authentic strenght preserving hormone. Mol. Aspects Med. 2005, 26, 203–219. [Google Scholar] [CrossRef]
- Landi, F.; Russo, A.; Liperoti, R.; Tosato, M.; Barillaro, C.; Pahor, M.; Bernabei, R.; Onder, G. Anorexia, Physical function, and incident disability among the frail elderly population: Results from the ilSirente Study. J. Am. Med. Dir. Assoc. 2010, 11, 153–156. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Martone, A.M.; Onder, G.; Vetrano, D.L.; Ortolani, E.; Tosato, M.; Marzetti, E.; Landi, F. Anorexia of Aging: A Modifiable Risk Factor for Frailty. Nutrients 2013, 5, 4126-4133. https://doi.org/10.3390/nu5104126
Martone AM, Onder G, Vetrano DL, Ortolani E, Tosato M, Marzetti E, Landi F. Anorexia of Aging: A Modifiable Risk Factor for Frailty. Nutrients. 2013; 5(10):4126-4133. https://doi.org/10.3390/nu5104126
Chicago/Turabian StyleMartone, Anna Maria, Graziano Onder, Davide Liborio Vetrano, Elena Ortolani, Matteo Tosato, Emanuele Marzetti, and Francesco Landi. 2013. "Anorexia of Aging: A Modifiable Risk Factor for Frailty" Nutrients 5, no. 10: 4126-4133. https://doi.org/10.3390/nu5104126
APA StyleMartone, A. M., Onder, G., Vetrano, D. L., Ortolani, E., Tosato, M., Marzetti, E., & Landi, F. (2013). Anorexia of Aging: A Modifiable Risk Factor for Frailty. Nutrients, 5(10), 4126-4133. https://doi.org/10.3390/nu5104126