Steroidal Compounds in Commercial Parenteral Lipid Emulsions
Abstract
:1. Introduction
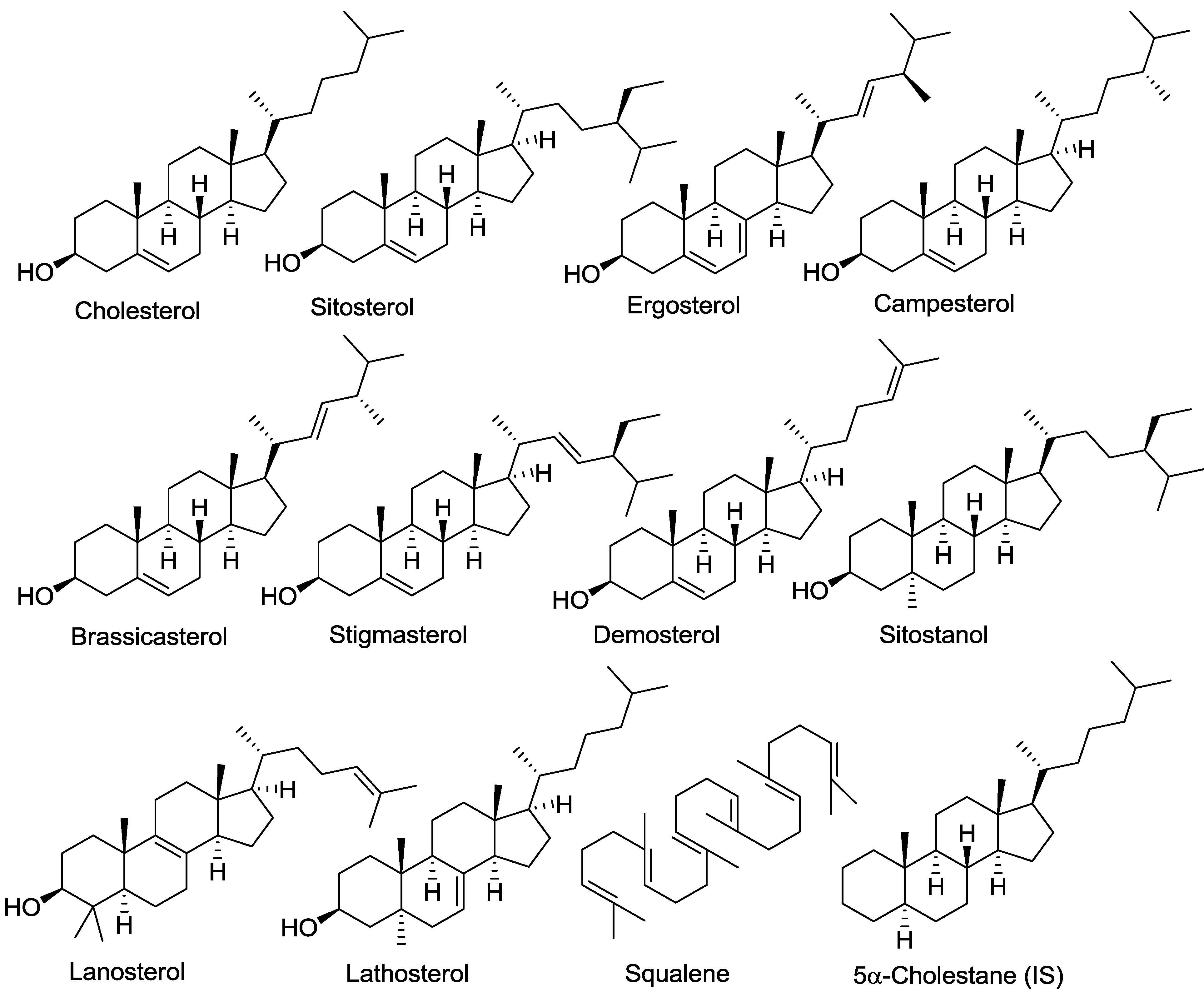
2. Experimental Section
2.1. Chemicals and Reagents
2.2. Instrumentation
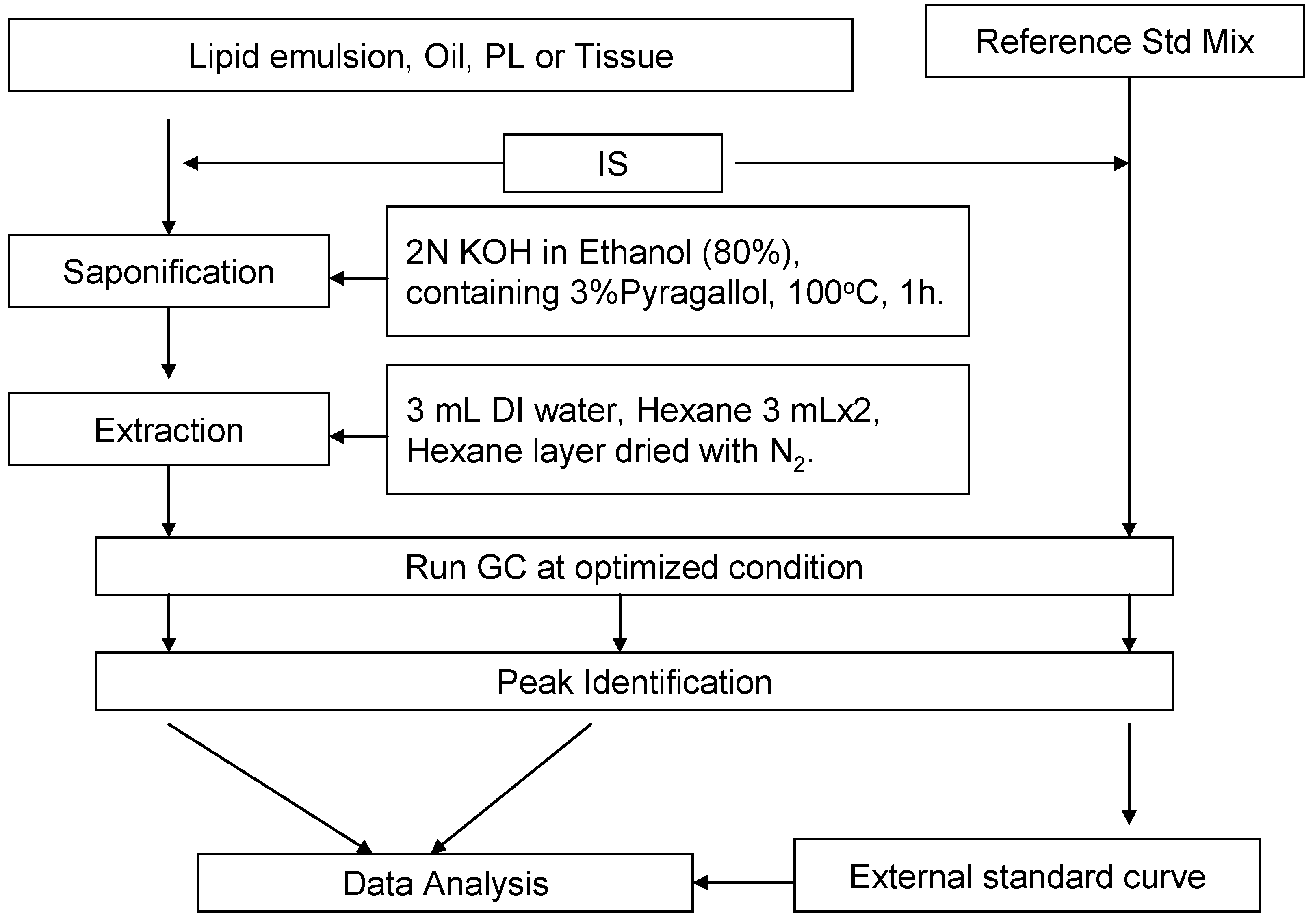
2.3. Saponification and Extraction
2.4. External Standard Curve and Equations
| Sterols | LOD (µg/mL) | Linear Range (µg/mL) |
|---|---|---|
| Squalene | 0.2 | 0.2–100 |
| Cholesterol | 0.4 | 0.4–500 |
| Desmosterol | 0.7 | 0.7–100 |
| Brassicasterol | 0.4 | 0.4–50 |
| Lathosterol | 0.7 | 0.7–100 |
| Ergosterol | 0.7 | 0.7–100 |
| Campesterol | 0.4 | 0.4–100 |
| Stigmasterol | 0.7 | 0.7–200 |
| β-Sitosterol | 0.9 | 0.9–500 |
| β-Sitostanol | 0.4 | 0.4–200 |
| Lanosterol | 0.2 | 0.2–100 |
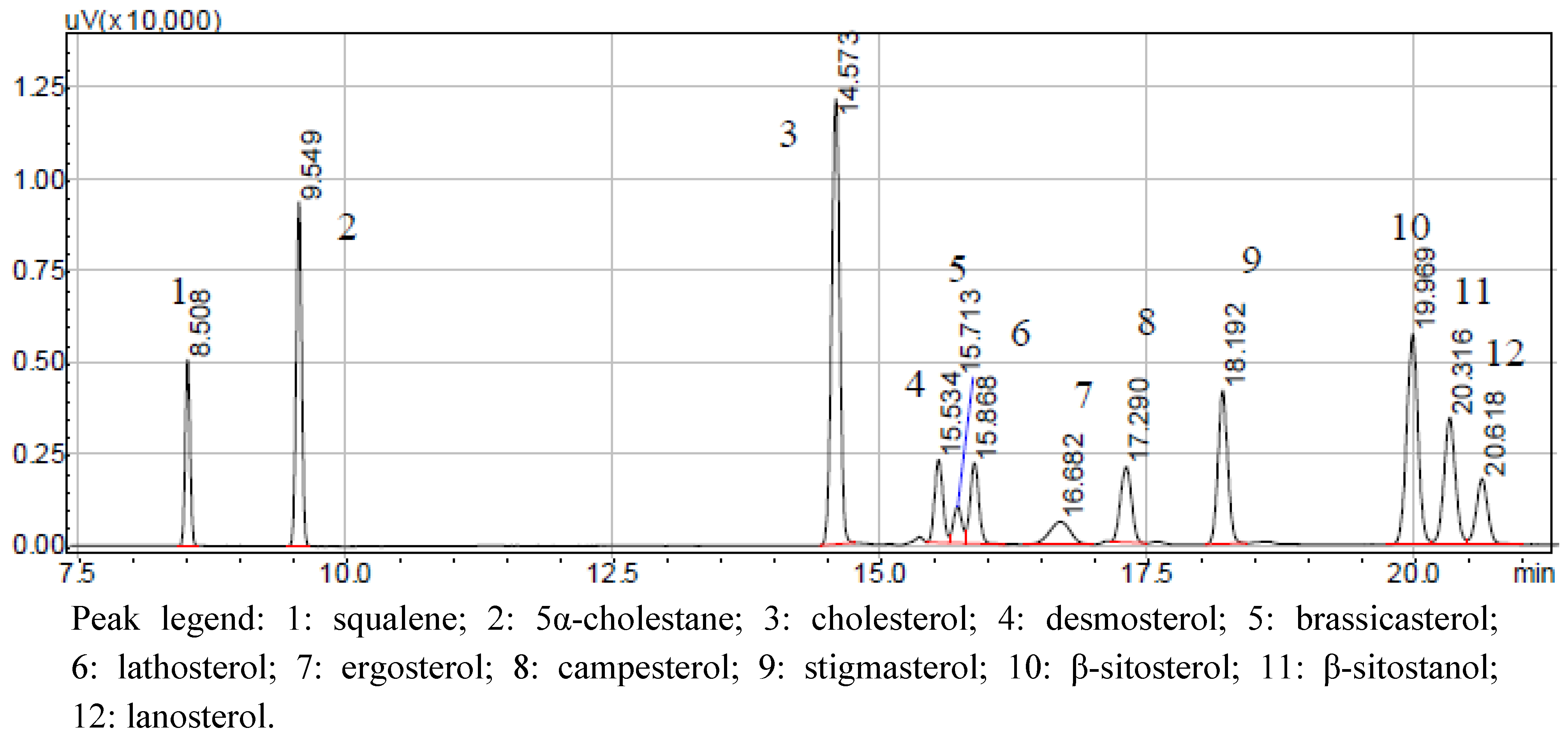
3. Results
3.1. Recovery of the Extraction and Saponification
| Sterol | Std. Alone | Std. Spiked in Olive Oil | Std. Spiked in Liver Homogenate | Std. Spiked in Intralipid® |
|---|---|---|---|---|
| IS | 100.0 ± 4.6 | 100.0 ± 4.4 | 100.0 ± 3.3 | 100.0 ± 2.8 |
| Squalene | 96.8 ± 2.1 | 95.6 ± 2.6 | 98.5 ± 1.9 | 101.3 ± 2.2 |
| Cholesterol | 102.7 ± 1.0 | 94.8 ± 4.0 | 99.9 ± 5.5 | 103.2 ± 1.4 |
| Desmosterol | 101.6 ± 2.4 | 98.3 ± 2.5 | 102.1 ± 2.9 | 98.3 ± 1.7 |
| Brassicasterol | 103.0 ± 1.6 | 94.9 ± 1.3 | 96.5 ± 0.7 | 103.1 ± 1.3 |
| Lathosterol | 97.5 ± 3.0 | 96.5 ± 2.4 | 99.2 ± 2.8 | 97.9 ± 2.6 |
| Ergosterol | 99.9 ± 0.9 | 94.1 ± 2.0 | 100.1 ± 0.5 | 100.0 ± 1.9 |
| Campesterol | 102.3 ± 1.2 | 94.0 ± 3.0 | 99.0 ± 8.6 | 101.7 ± 1.9 |
| Stigmasterol | 106.4 ± 1.1 | 99.5 ± 2.1 | 101.1 ± 1.0 | 100.7 ± 1.5 |
| β-Sitosterol | 103.1 ± 1.3 | 97.4 ± 3.7 | 97.1 ± 1.0 | 99.8 ± 1.4 |
| β-Sitostanol | 94.3 ± 2.8 | 93.22 ± 2.7 | 98.5 ± 1.3 | 95.6 ± 0.7 |
| Lanosterol | 98.2 ± 0.9 | 96.6 ± 2.8 | 100.5 ± 1.2 | 97.7 ± 1.7 |
3.2. Sensitivity of the Method
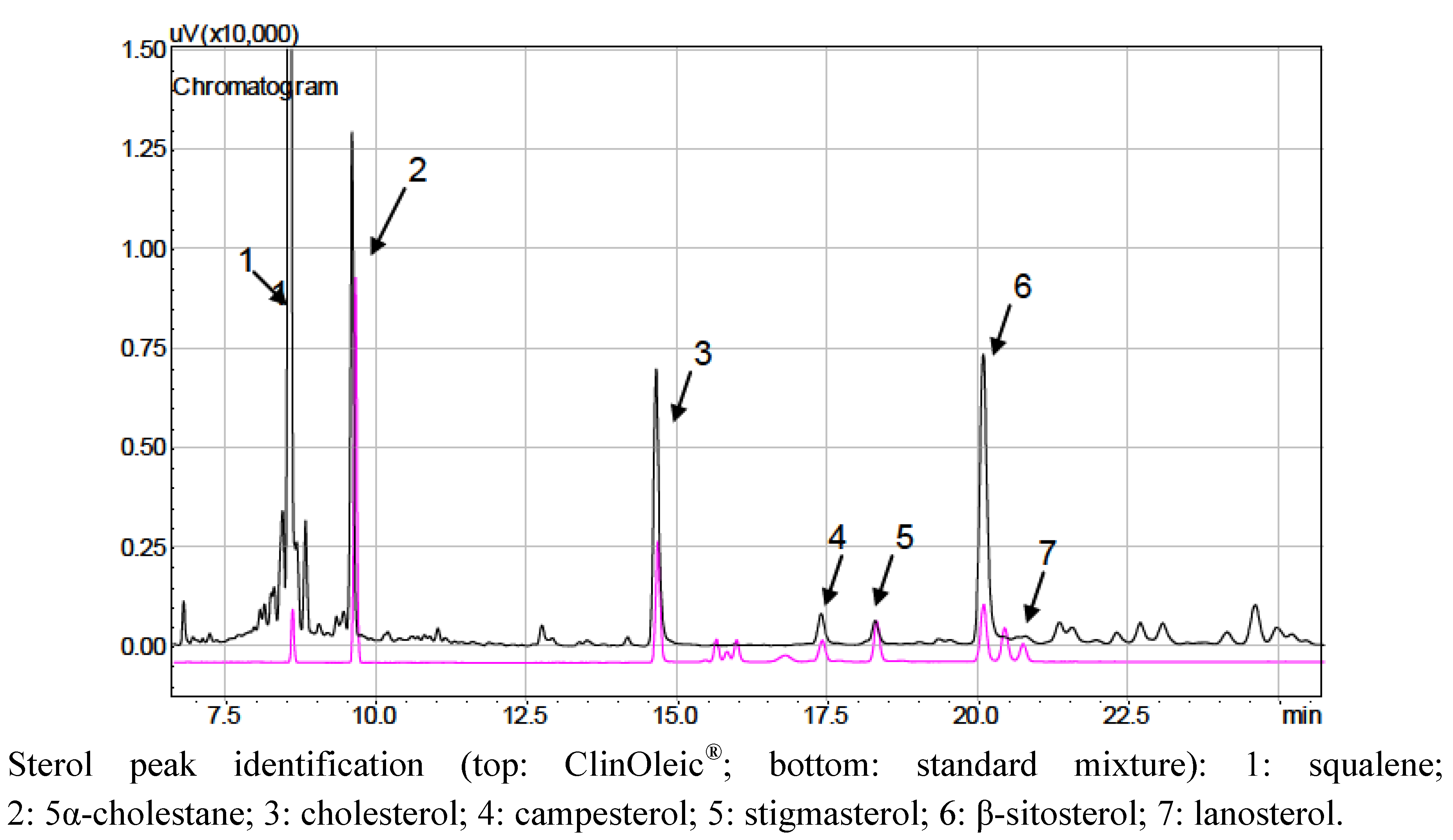
3.3. Sterol Composition of Lipid Emulsions
| Emulsions | Manufacturer | Lot No. | Major component |
|---|---|---|---|
| Soybean oil-based | |||
| Intralipid® | Fresenius Kabi (DE) | 10BK7082 | Soybean oil 20 g/100 mL and 1.2 g |
| Ivelip® | Baxter Healthcare Corporation (BE) | 08K25A92 | Soybean oil 20 g/100 mL and 1.2 g of egg yolk phospholipids |
| Lipofundin® N | B. Braun (DE) | 9173A184 | Soybean oil 20 g/100 mL and 1.2 g of egg yolk phospholipids |
| Liposyn® III | Hospira, Inc. (US) | 70913DW | Soybean oil 20 g/100mL and 1.2 g of egg yolk phospholipids |
| Liposyn® II | Hospira, Inc. (US) | 74906DW | Mixture of soybean oil (50%) and safflower oil (50%) 20 g/100 mL, and 1.2 g of egg yolk phospholipids |
| Medium- & Long-chain fatty acid-based | |||
| Lipofundin® MCT | B. Braun (DE) | 8494A181 | Mixture of soybean oil (50%) and medium-chain structured triglycerides (50%), 20 g/100mL, and 1.2 g of egg yolk phospholipids |
| Structolipid® | Fresenius Kabi (DE) | 10CD2533 | Interesterified mixture of equimolar amounts of long chain triglycerides and medium chain triglycerides, corresponding to 64% (w/w) and 36% (w/w), respectively. |
| Olive oil-based | |||
| ClinOleic® | Baxter Healthcare Corporation (FR) | 09D09A91 | Mixture of olive oil (approximately 80%) and soybean oil (approximately 20%), 20 g/100 mL, and 1.2 g of egg yolk phospholipids |
| Soybean Oil-Based | Medium- & Long-Chain Fatty Acid-Based | Olive Oil-Based | ||||||
|---|---|---|---|---|---|---|---|---|
| Sterols | Intralipid® | Ivelip® | Lipofundin® N | Liposyn® III | Liposyn® II | Lipofundin® MCT | Structolipid® | ClinOleic® |
| Squalene | 7.43 ± 0.10 | 15.87 ± 0.26 | 9.18 ± 0.26 | 8.91 ± 0.41 | 6.17 ± 0.10 | 6.08 ± 0.12 | 4.52 ± 0.02 | 387.45 ± 2.30 |
| Cholesterol | 274.08 ± 3.55 | 74.92 ± 1.33 | 217.46 ± 2.40 | 63.57 ± 2.85 | 88.99 ± 0.49 | 219.39 ± 2.93 | 280.43 ± 0.51 | 109.70 ± 0.38 |
| Desmosterol | ND | ND | ND | ND | ND | ND | ND | ND |
| Brassicasterol | ND | ND | ND | ND | ND | ND | ND | ND |
| Lathosterol | ND | ND | ND | ND | ND | ND | ND | ND |
| Ergosterol | ND | ND | ND | ND | ND | ND | ND | ND |
| Campesterol | 55.35 ± 0.48 | 51.59 ± 1.19 | 85.85 ± 0.97 | 94.26 ± 4.30 | 68.12 ± 0.60 | 30.87 ± 0.62 | 43.96 ± 0.39 | 13.33 ± 0.12 |
| Stigmasterol | 65.05 ± 0.52 | 59.41 ± 1.25 | 96.50 ± 0.88 | 93.41 ± 3.67 | 60.93 ± 0.65 | 46.00 ± 0.95 | 48.80 ± 0.31 | 12.15 ± 0.04 |
| β-Sitosterol | 302.64 ± 2.00 | 277.04 ± 5.61 | 420.32 ± 4.73 | 390.65 ± 18.68 | 339.78 ± 2.62 | 191.63 ± 3.18 | 240.04 ± 0.61 | 240.59 ± 2.08 |
| β-Sitostanol | 7.68 ± 0.25 | 6.88 ± 0.15 | 10.06 ± 0.39 | 8.27 ± 0.39 | 10.45 ± 0.30 | 5.67 ± 0.12 | 6.77 ± 0.25 | 4.57 ± 0.16 |
| Lanosterol | 8.35 ± 2.48 | 7.49 ± 0.34 | 9.13 ± 0.39 | 11.78 ± 0.62 | 8.50 ± 0.34 | 3.96 ± 0.22 | 6.28 ± 0.08 | 3.74 ± 0.20 |
| Total sterols | 713.15 ± 9.27 | 477.33 ± 9.87 | 839.31 ± 9.75 | 661.93 ± 30.50 | 576.77 ± 5.00 | 497.52 ± 8.02 | 626.28 ± 2.15 | 384.08 ± 2.98 |
| Total phytosterols | 439.07 ± 5.72 | 402.41 ± 8.55 | 621.85 ± 7.36 | 598.37 ± 27.65 | 487.77 ± 4.51 | 278.14 ± 5.09 | 345.85 ± 1.64 | 274.38 ± 2.60 |
| Phytosterols (mg) per 100 g of lipid | 220 ± 2.86 | 201 ± 4.25 | 311 ± 3.68 | 299 ± 13.82 | 249 ± 2.13 | 139 ± 2.55 | 173 ± 0.82 | 137 ± 1.30 |
4. Discussion
5. Conclusion
Acknowledgements
References
- Cober, M.P.; Teitelbaum, D.H. Prevention of parenteral nutrition-associated liver disease: Lipid minimization. Curr. Opin. Organ Trasplant. 2010, 15, 330–333. [Google Scholar] [CrossRef]
- Kelly, D.A. Preventing parenteral nutrition liver disease. Early Hum. Dev. 2010, 86, 683–687. [Google Scholar] [CrossRef]
- Lloyd, D.A.J.; Gabe, S.M. Managing liver dysfunction in parenteral nutrition. Proc. Nutr. Soc. 2007, 66, 530–538. [Google Scholar] [CrossRef]
- Clayton, P.T.; Whitfield, P.; Iyer, K. The role of phytosterols in the pathogenesis of liver complications of pediatric parenteral nutrition. Nutrition 1998, 14, 158–164. [Google Scholar]
- Cavicchi, M.; Beau, P.; Crenn, P.; Degott, C.; Messing, B. Prevalence of liver disease and contributing factors in patients receiving home parenteral nutrition for permanent intestinal failure. Ann. Intern. Med. 2000, 132, 525–532. [Google Scholar]
- Kurvinen, A.; Nissinen, M.J.; Gylling, H.; Miettinen, T.A.; Lampela, H.; Koivusalo, A.I.; Rintala, R.J.; Pakarinen, M.P. Effects of long-term parenteral nutrition on serum lipids, plant sterols, cholesterol metabolism, and liver histology in pediatric intestinal failure. J. Pediatr. Gasteroenterol. Nutr. 2011, 53, 440–446. [Google Scholar]
- Kono, H.; Fujii, H.; Asakawa, M.; Yamamoto, M.; Matsuda, M.; Maki, A.; Matsumoto, Y. Protective effects of medium-chain triglycerides on the liver and gut in rats administered endotoxin. Ann. Surg. 2003, 237, 246–255. [Google Scholar]
- Klaassen, C.D.; Aleksunes, L.M. Xenobiotic, bile acid, and cholesterol transporters: Function and regulation. Pharmacol. Rev. 2010, 62, 1–96. [Google Scholar] [CrossRef]
- Ohvo-Rekila, H.; Ramstedt, B.; Leppimaki, P.; Slotte, J.P. Cholesterol interactions with phospholipids in membranes. Prog. Lipid Res. 2002, 41, 66–97. [Google Scholar] [CrossRef]
- Ikonen, E. Cellular cholesterol trafficking and compartmentalization. Nat. Rev. Mol. Cell Biol. 2008, 9, 125–138. [Google Scholar] [CrossRef]
- Tabas, I. Consequences of cellular cholesterol accumulation: Basic concepts and physiological implications. J. Clin. Invest. 2002, 110, 905–911. [Google Scholar]
- Law, M.R.; Wald, N.J.; Rudnicka, A.R. Quantifying effect of statins on low density lipoprotein cholesterol, ischaemic heart disease, and stroke: Systematic review and meta-analysis. Br. Med. J. 2003, 326, 1423. [Google Scholar] [CrossRef]
- Raffai, R.L.; Weisgraber, K.H. Cholesterol: From heart attacks to Alzheimer’s disease. J. Lipid Res. 2003, 44, 1423–1430. [Google Scholar] [CrossRef]
- Heinemann, T.; Axtmann, G.; von Bergmann, K. Comparison of intestinal absorption of cholesterol with different plant sterols in man. Eur. J. Clin. Invest. 1993, 23, 827–831. [Google Scholar] [CrossRef]
- Berge, K.E.; Tian, H.; Graf, G.A.; Yu, L.; Grishin, N.V.; Schultz, J.; Kwiterovich, P.; Shan, B.; Barnes, R.; Hobbs, H.H. Accumulation of dietary cholesterol in sitosterolemia caused by mutations in adjacent ABC transporters. Science 2000, 290, 1771–1775. [Google Scholar]
- Lee, M.H.; Lu, K.; Hazard, S.; Yu, H.; Shulenin, S.; Hidaka, H.; Kojima, H.; Allikmets, R.; Sakuma, N.; Pegoraro, R.; et al. Identification of a gene, ABCG5, important in the regulation of dietary cholesterol absorption. Nat. Genet. 2001, 27, 79–83. [Google Scholar]
- Lu, K.; Lee, M.H.; Hazard, S.; Brooks-Wilson, A.; Hidaka, H.; Kojima, H.; Ose, L.; Stalenhoef, A.F.; Mietinnen, T.; Bjorkhem, I.; et al. Two genes that map to the STSL locus cause sitosterolemia: Genomic structure and spectrum of mutations involving sterolin-1 and sterolin-2, encoded by ABCG5 and ABCG8, respectively. Am. J. Hum. Genet. 2001, 69, 278–290. [Google Scholar] [CrossRef]
- Moruisi, K.G.; Oosthuizen, W.; Opperman, A.M. Phytosterols/stanols lower cholesterol concentrations in familial hypercholesterolemic subjects: A systematic review with meta-analysis. J. Am. Coll. Nutr. 2006, 25, 41–48. [Google Scholar]
- Jones, P.J.; MacDougall, D.E.; Ntanios, F.; Vanstone, C.A. Dietary phytosterols as cholesterol-lowering agents in humans. Can. J. Physiol. Pharmacol. 1997, 75, 217–227. [Google Scholar] [CrossRef]
- Ostlund, R.E., Jr.; Racette, S.B.; Okeke, A.; Stenson, W.F. Phytosterols that are naturally present in commercial corn oil significantly reduce cholesterol absorption in humans. Am. J. Clin. Nutr. 2002, 75, 1000–1004. [Google Scholar]
- Ellegard, L.; Sunesson, A.; Bosaeus, I. High serum phytosterol levels in short bowel patients on parenteral nutrition support. Clin. Nutr. 2005, 24, 415–420. [Google Scholar]
- Llop, J.M.; Virgili, N.; Moreno-Villares, J.M.; Garcia-Peris, P.; Serrano, T.; Forga, M.; Solanich, J.; Pita, A.M. Phytosterolemia in parenteral nutrition patients: Implications for liver disease development. Nutrition 2008, 24, 1145–1152. [Google Scholar] [CrossRef]
- Clayton, P.T.; Bowron, A.; Mills, K.A.; Massoud, A.; Casteels, M.; Milla, P.J. Phytosterolemia in children with parenteral nutrition-associated cholestatic liver disease. Gastroenterology 1993, 105, 1806–1813. [Google Scholar]
- Itoh, T.; Tamura, T.; Matsumoto, T. Sterol composition of 19 vegetable oils. J. Am. Chem. Soc. 1973, 50, 122–125. [Google Scholar] [CrossRef]
- Phillips, K.M.; Puggio, D.M.; Toivo, J.I.; Simpkins, A.H. Free and esterified sterol composition of edible oils and fats. J. Food Comp. Anal. 2002, 15, 123–142. [Google Scholar] [CrossRef]
- Demacker, P.N.; Vos-Janssen, H.E.; Hijmans, A.G.; van’t Laar, A.; Jansen, A.P. Measurement of high-density lipoprotein cholesterol in serum: Comparison of six isolation methods combined with enzymic cholesterol analysis. Clin. Chem. 1980, 26, 1780–1786. [Google Scholar]
- Fromm, H.; Amin, P.; Klein, H.; Kupke, I. Use of a simple enzymatic assay for cholesterol analysis in human bile. J. Lipid Res. 1980, 21, 259–261. [Google Scholar]
- Bachman, K.C.; Lin, J.H.; Wilcox, C.J. Sensitive colorimetric determination of cholesterol in dairy products. J. Assoc. Off. Anal. Chem. 1976, 59, 1146–1149. [Google Scholar]
- Ballesteros, E.; Gallego, M.; Valcarcel, M. Gas chromatographic determination of cholesterol and tocopherols in edible oils and fats with automatic removal of interfering triglycerides. J. Chromatogr. A 1996, 719, 221–227. [Google Scholar] [CrossRef]
- Phillips, K.M.; Ruggio, D.M.; Bailey, J.A. Precise quantitative determination of phytosterols, stanols, and cholesterol metabolites in human serum by capillary gas-liquid chromatography. J. Chromatogr. B Biomed. Sci. Appl. 1999, 732, 17–29. [Google Scholar] [CrossRef]
- Guardiola, F.; Codony, R.; Rafecas, M.; Boatella, J. Selective gas chromatographic determination of cholesterol in eggs. J. Am. Chem. Soc. 1994, 71, 867–871. [Google Scholar] [CrossRef]
- Ishikawa, T.T.; MacGee, J.; Morrison, J.A.; Glueck, C.J. Quantitative analysis of cholesterol in 5 to 20 microliter of plasma. J. Lipid Res. 1974, 15, 286–291. [Google Scholar]
- Goh, E.H.; Colles, S.M.; Otte, K.D. HPLC analysis of desmosterol, 7-dehydrocholesterol, and cholesterol. Lipids 1989, 24, 652–655. [Google Scholar] [CrossRef]
- Cayuela, J.M.; Garrido, M.D.; Banon, S.J.; Ros, J.M. Simultaneous HPLC analysis of alpha-tocopherol and cholesterol in fresh pig meat. J. Agric. Food Chem. 2003, 51, 1120–1124. [Google Scholar] [CrossRef]
- Canabate-Diaz, B.; Segura Carretero, A.; Fernandez-Gutierrez, A.; Belmonte Vega, A.; Garrido Frenich, A.; Martinez Vidal, J.L.; Duran Martos, J. Separation and determination of sterols in olive oils by HPLC-MS. Food Chem. 2007, 102, 593–598. [Google Scholar]
- Volin, P. Analysis of steroidal lipids by gas and liquid chromatography. J. Chromatogr. A 2001, 935, 125–140. [Google Scholar] [CrossRef]
- Abidi, S.L. Chromatographic analysis of plant sterols in foods and vegetable oils. J. Chromatogr. A 2001, 935, 173–201. [Google Scholar] [CrossRef]
- Osman, H.; Chin, Y.K. Comparitive sensitivities of cholesterol analysis using GC, HPLC an spectrophotometric method. Malays. J. Anal. Sci. 2006, 10, 205–210. [Google Scholar]
- Forchielli, M.L.; Bersani, G.; Tala, S.; Grossi, G.; Puggioli, C.; Masi, M. The spectrum of plant and animal sterols in different oil-derived intravenous emulsions. Lipids 2010, 45, 63–71. [Google Scholar] [CrossRef]
- Bohac, C.E.; Rhee, K.S.; Cross, H.R.; Ono, K. Assessment of methodologies for calorimetric cholesterol assay of meats. J. Food Sci. 1988, 53, 1642–1644. [Google Scholar] [CrossRef]
- Institute of Medicine Cholesterol, Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids; The National Academies Press: Washington, DC, USA, 2005; pp. 542–588.
- Whitfield, P.D.; Clayton, P.T.; Muller, D.P.R. Effect of intravenous lipid emulsions on hepatic cholesterol metabolism. J. Pediatr. Gastroenterol. Nutr. 2000, 30, 538–546. [Google Scholar] [CrossRef]
- Nguyen, L.B.; Shefer, S.; Salen, G.; Tint, S.G.; Batta, A.K. Competitive inhibition of hepatic sterol 27-hydroxylase by sitosterol. Decreased activity in sitosterolemia. Proc. Assoc. Am. Phys. 1998, 110, 32–39. [Google Scholar]
- Shefer, S.; Salen, G.; Nguyen, L.; Batta, A.K.; Packin, V.; Tint, G.S.; Hauser, S. Competitive inhibition of bile acid synthesis by endogenous cholestanol and sitosterol in sitosterolaemia with xanthomatosis. J. Clin. Invest. 1988, 82, 1833–1839. [Google Scholar] [CrossRef]
- Boberg, K.M.; Akerlund, J.E.; Bjorkhem, I. Effect of sitosterol on the rate-limiting enzymes in cholesterol synthesis and degradation. Lipids 1989, 24, 9–12. [Google Scholar] [CrossRef]
- Iyer, K.R.; Spitz, L.; Clayton, P. New insight into mechansims of parenteral nutrition-associated cholestasis: Role of plant sterols. J. Pediatr. Surg. 1998, 33, 1–6. [Google Scholar] [CrossRef]
- Carter, B.A.; Taylor, O.A.; Prendergast, D.R.; Zimmerman, T.L.; von Furstenberg, R.; Moore, D.D.; Karpen, S.J. Stigmasterol, a soy lipid-derived phytosterol, is an antagonist of the bile acid nuclear receptor FXR. Pediatr. Res. 2007, 62, 301–306. [Google Scholar] [CrossRef]
- Hallikainen, M.; Huikko, L.; Kontra, K.; Nissinen, M.; Piironen, V.; Miettinen, T.; Gylling, H. Effect of parenteral serum plant sterols on liver enzymes and cholesterol metabolism in a patient with short bowel syndrome. Nutr. Clin. Pract. 2008, 23, 429–435. [Google Scholar] [CrossRef]
- Puder, M.; Valim, C.; Meisel, J.A.; Le, H.D.; de Meijer, V.E.; Robinson, E.M.; Zhou, J.; Duggan, C.; Gura, K.M. Parenteral fish oil improves outcomes in patients with parenteral nutrition-associated liver injury. Ann. Surg. 2009, 250, 395–402. [Google Scholar]
- Ramirez-Torres, A.; Gabas, C.; Barranquero, C. Squalene: Current Knowledge and Potential Therapeutic Uses; Nova Biomedical Books: New York, NY, USA, 2011; pp. 1–73. [Google Scholar]
- Nes, D.W. Isopentenoids and Other Natural Products: Evolution and Function; American Chemical Society: Washington, DC, USA, 1994. [Google Scholar]
- Kritchevsky, D.; Moyer, A.W.; Tesar, W.C.; Logan, J.B.; Brown, R.A.; Richmond, G. Squalene feeding in experimental atherosclerosis. Circ. Res. 1954, 2, 340–343. [Google Scholar] [CrossRef]
- Chan, P.; Tomlinson, B.; Lee, C.B.; Lee, Y.S. Effectiveness and safety of low-dose pravastatin and squalene, alone and in combination, in elderly patients with hypercholesterolemia. J. Clin. Pharmacol. 1996, 36, 422–427. [Google Scholar]
- Strandberg, T.E.; Tilvis, R.S.; Miettinen, T.A. Metabolic variables of cholesterol during squalene feeding in humans: Comparison with cholestyramine treatment. J. Lipid Res. 1990, 31, 1637–1643. [Google Scholar]
- Strandberg, T.E.; Tilvis, R.S.; Miettinen, T.A. Variations of hepatic cholesterol precursors during altered flows of endogenous and exogenous squalene in the rat. Biochim. Biophys. Acta 1989, 1001, 150–156. [Google Scholar]
- Newmark, H.L. Squalene, olive oil, and cancer risk: A review and hypothesis. Cancer Epidemiol. Biomark. Prev. 1997, 6, 1101–1103. [Google Scholar]
- Waterman, E.; Lockwood, B. Active components and clinical applications of olive oil. Altern. Med. Rev. 2007, 12, 331–342. [Google Scholar]
- Newmark, H.L. Squalene, olive oil, and cancer risk. Review and hypothesis. Ann. N. Y. Acad. Sci. 1999, 889, 193–203. [Google Scholar] [CrossRef]
- Sporn, M.B.; Suh, N. Chemoprevention of cancer. Carcinogenesis 2000, 21, 525–530. [Google Scholar] [CrossRef]
- Matthews, J. Sharks still intrigue cancer researchers. J. Natl. Cancer Inst. 1992, 84, 1000–1002. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xu, Z.; Harvey, K.A.; Pavlina, T.; Dutot, G.; Hise, M.; Zaloga, G.P.; Siddiqui, R.A. Steroidal Compounds in Commercial Parenteral Lipid Emulsions. Nutrients 2012, 4, 904-921. https://doi.org/10.3390/nu4080904
Xu Z, Harvey KA, Pavlina T, Dutot G, Hise M, Zaloga GP, Siddiqui RA. Steroidal Compounds in Commercial Parenteral Lipid Emulsions. Nutrients. 2012; 4(8):904-921. https://doi.org/10.3390/nu4080904
Chicago/Turabian StyleXu, Zhidong, Kevin A. Harvey, Thomas Pavlina, Guy Dutot, Mary Hise, Gary P. Zaloga, and Rafat A. Siddiqui. 2012. "Steroidal Compounds in Commercial Parenteral Lipid Emulsions" Nutrients 4, no. 8: 904-921. https://doi.org/10.3390/nu4080904
APA StyleXu, Z., Harvey, K. A., Pavlina, T., Dutot, G., Hise, M., Zaloga, G. P., & Siddiqui, R. A. (2012). Steroidal Compounds in Commercial Parenteral Lipid Emulsions. Nutrients, 4(8), 904-921. https://doi.org/10.3390/nu4080904




