Benefits of Docosahexaenoic Acid, Folic Acid, Vitamin D and Iodine on Foetal and Infant Brain Development and Function Following Maternal Supplementation during Pregnancy and Lactation
Abstract
:1. Introduction
1.1. DHA
1.1.1. Importance of Fatty Acids in Brain Development and Function
1.1.2. Maternal Nutrition: During Preconception, Gestation and Lactation
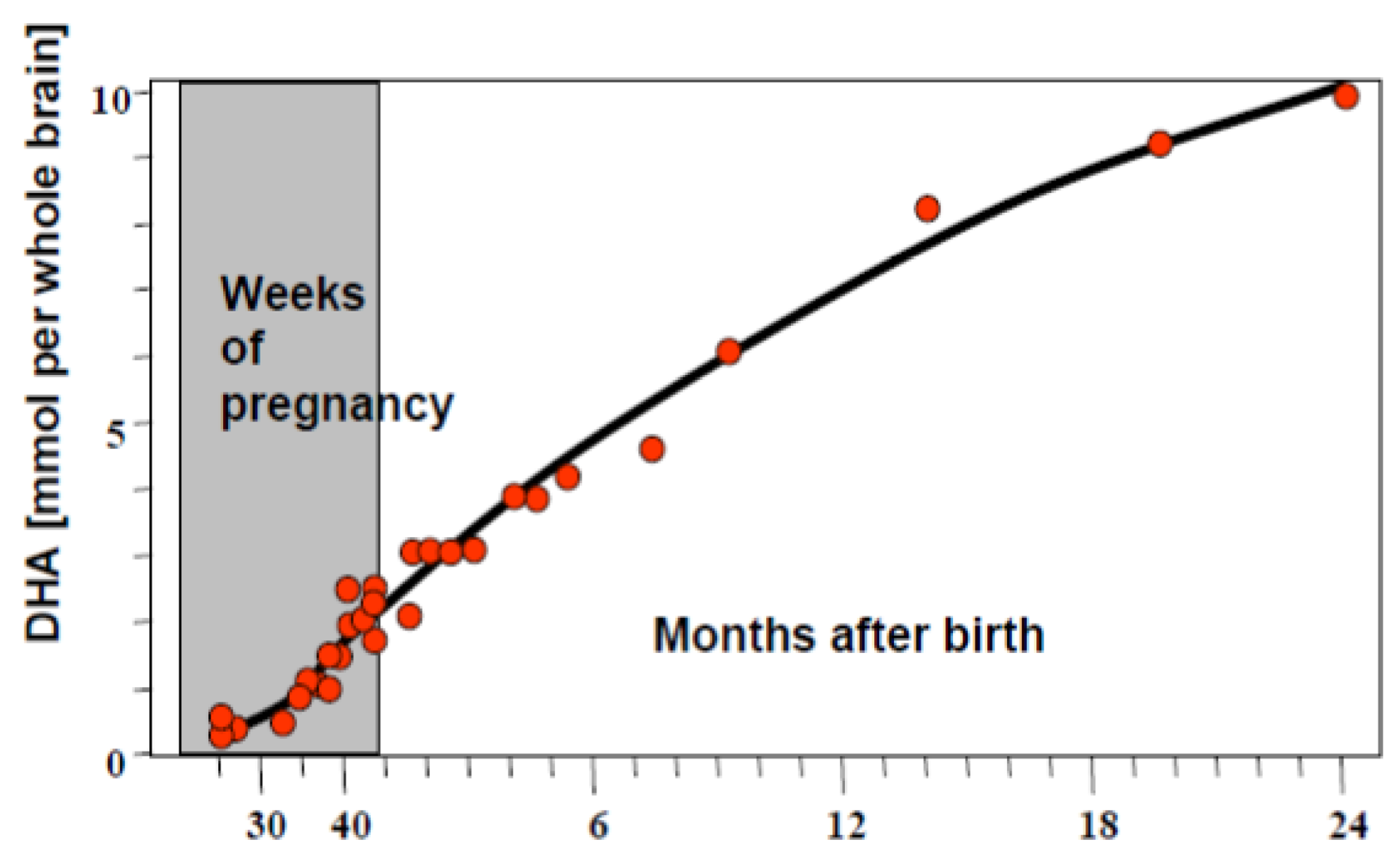
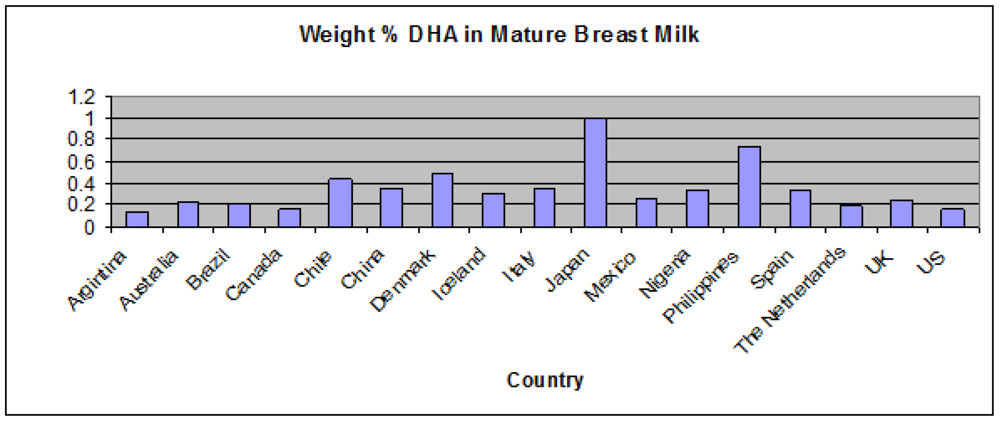
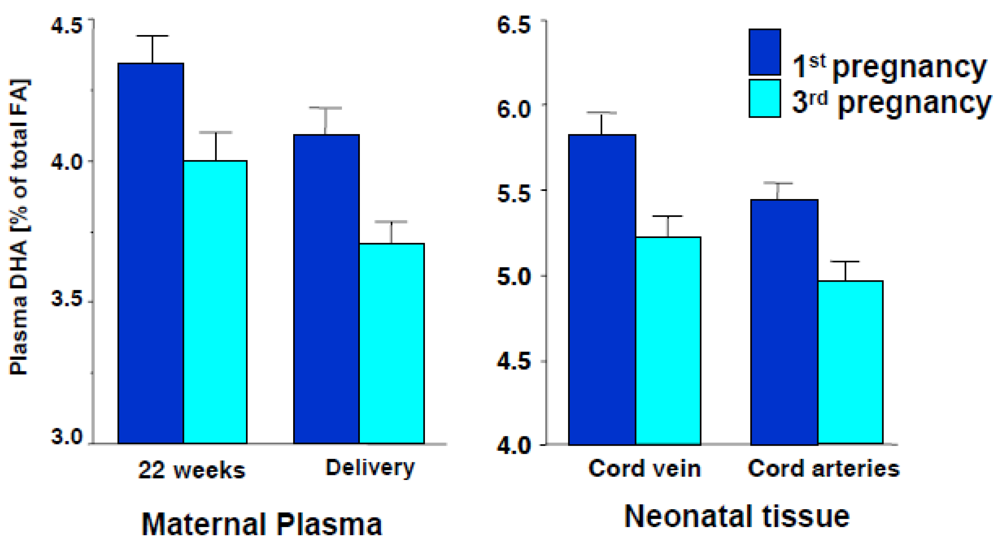
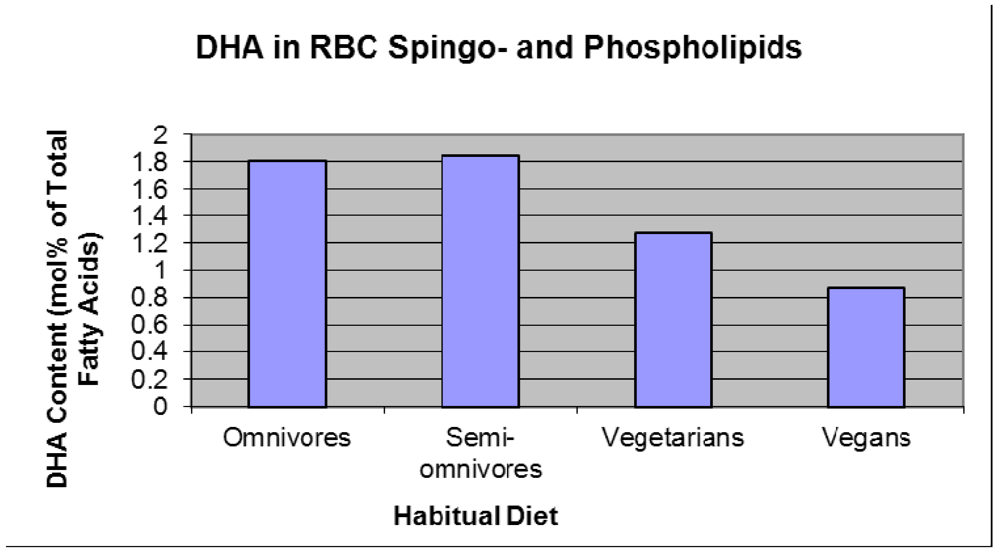
1.1.3. Infant Supplementation Studies
1.2. Vitamin D
- Build strong bones—vitamin D ensures foetal supply of calcium for strong bones [45] including those of the skull. Severe hypocalcaemic is associated with high risk of brain damage [50]. vitamin D insufficiency has been associated with reduction in bone mineral content of the offspring [51] and perinatal growth restriction [52].
- Maintain pregnancy—the circulating concentration of maternal active vitamin D rises in the first trimester and doubles by the end of the third trimester [53]. The early rise is believed to be necessary to enable immunological adaptation by the mother that is required to maintain normal pregnancy [53]. These vitamin D induced immunological changes in the mother prevent miscarriage [45,53].
1.3. Folic Acid
- Those not taking the recommended quantity of folic acid supplement;
- Those on restricted diets (chronic dieters);
- Those with lower socio-economic status;
- Those with limited or uncertain availability of nutritionally adequate and safe food.
1.4. Iodine
- An increase in maternal T4 concentration to maintain her normal thyroid hormone levels while transferring additional thyroid hormone to the foetus early in the first trimester (before the foetal thyroid is functioning) [66];
- Iodine transfer to the foetus, particularly towards the end of pregnancy [66];
- An increase in iodine urinary excretion [66].
2. Evidence of the Potential Benefits of Maternal DHA Supplementation for Foetal/Infant Brain Health
2.1. Effects of Maternal DHA Supplementation on Maternal DHA Status
2.2. Effects of Maternal DHA Supplementation on Foetal/Infant DHA Status
2.3. Benefits to the Fetus/Infant/Child
2.3.1. Observational Studies

- DHA status at birth is significantly and positively related to movement quality [100] and reduced problem behaviour at 7 years of age [101] visual acuity at age 6 months [102] and performance on behavioral tests including the Digit Span Forward Test and California Verbal Learning Test—Children’s Version at 11.3 years of age. Children with higher cord DHA, that is exposure to a higher amount of DHA during pregnancy, responded faster when making decisions that relied on memory than those with lower cord DHA. In addition, children with higher current DHA, that is higher routine intake of DHA, also responded faster when making decisions that relied on memory, than those with lower current DHA [103].
- Among children who were breast-fed for less than 6 months, maternal fish intake of greater than 2–3 times/week during pregnancy is associated with better scores on the McCarthy Scales of Children’s Abilities for verbal, perceptual-performance, quantitative, general cognitive, memory, and motor skills [105].
- Higher maternal plasma DHA during pregnancy is associated with more mature neonatal sleep-state, suggesting greater central nervous system maturity [106].
- Higher maternal DHA status at birth is associated with enhanced attention functioning during the second year of life [107].
2.3.2. Intervention Trials
2.3.2.1. During Pregnancy
- DHA reduces the incidence of premature delivery, increases birth weight, and gestation and may be useful to prolong gestational duration in some high-risk pregnancies [110].
- Fish oil supplementation increases breast milk EPA and DHA content up to 6 weeks postpartum and these higher amounts are directly correlated with better Griffith’s developmental scores including hand and eye co-ordination in the infant at 1 year of age [94].
- Higher foetal DHA exposure due to maternal supplementation results in better neurological outcome at 5.5 years of age [115]. The odds of children with maximal neurological optimality scores increases with every unit increment in cord blood DHA at delivery.
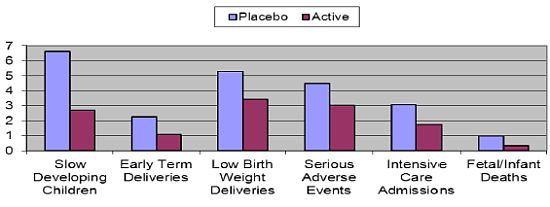
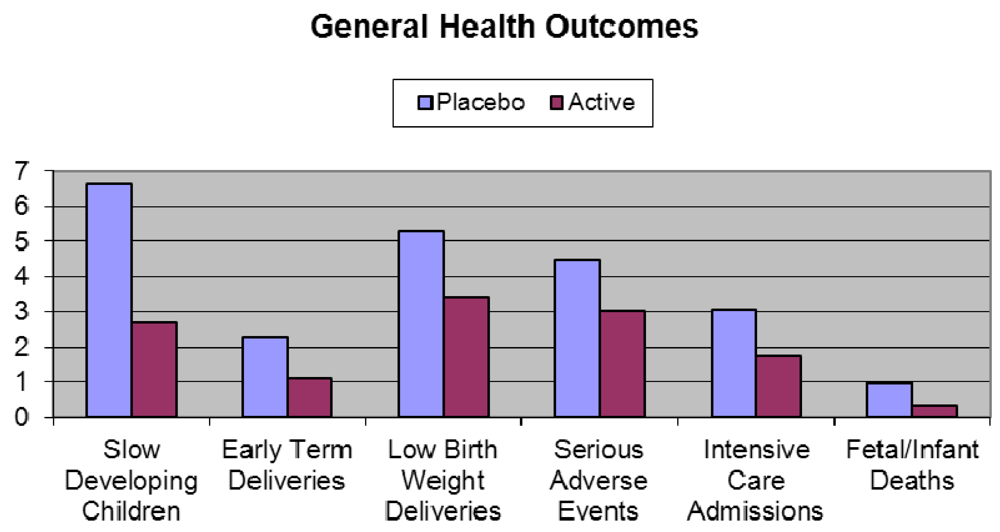
- The largest clinical study ever providing DHA to pregnant women was aptly named the DOMInO trial (DHA to Optimize Mother Infant Outcome) (Figure 6) [116]. The multicentered, randomised, double-blind, placebo-controlled clinical trial, conducted in 5 Australian maternity hospitals and supported by a grant from the Australian National Health and Medical Research Council included 2399 women with gestation of less than 21 weeks during singleton pregnancies and 726 of their infants. From twenty weeks until birth, the women took either three capsules providing 800 mg/day of DHA and 100 mg/day of EPA or three 500 mg/day vegetable oil capsules without DHA that matched the fatty acid composition of the average Australian diet. Cognitive and language development in the infants was assessed by the Bayley Scales of Infant and Toddler Development, Third Edition at 18 months of age. The primary outcome of cognitive and language development of infants in the DHA group did not differ from those in the control. However, major benefits were seen in disadvantaged slow developing children (those with an IQ of less than 85) where in all infants 6.64% in the placebo group were classed as “slow developers” compared to only 2.71% in the DHA group—a reduction of almost 60%. In boys, the reduction was even greater at 64%. Based on Australia’s birth rate this would represent 10,000 children per year no longer being classed as slow developers. For general health outcomes, DHA significantly reduced the incidence of low birth weight babies by 35% and the number of very early pre-term deliveries by more than 50% compared to the control. This represents a major public health benefit, in countries such as Australia for example, where there would be more than 3000 fewer preterm births per year if women were supplemented with DHA during pregnancy. Pre-term delivery and low birth weight are two of the major risk factors for ill health and poor mental development in children. Thirty-three percent less infants in the DHA group required admission to intensive care; there were two thirds less infant deaths in the DHA group and one third less infants in the DHA group experienced a serious adverse event relative to control. These findings were all highly significant and illustrate much better general health of the infants whose mothers were given DHA. There was no difference between groups for maternal hemorrhage, antenatal hospitalization, nose bleeds, vaginal blood loss, constipation, nausea or vomiting at 28 or 36 weeks gestation. However, more women in the DHA group reported eructations than the control group.
2.3.2.2. During Lactation
2.3.2.3. During Pregnancy and Lactation
3. Evidence of the Potential Benefits of Maternal Vitamin D Supplementation for Foetal/Infant Brain Health
3.1. Epidemiological Evidence
3.1.1. Studies Reporting Maternal Vitamin D Deficiency
- In a study completed by a coalition of scientists formed to optimize vitamin D fortification in the northern European countries, the average dietary intake of vitamin D in young women was only around 80 IU (2 μg) per day [144]. This falls short of even the most modest dietary recommendations.
- A cross-sectional study in Iran included 147 pregnant women whose serum status of vitamin D, A, and E were assessed at 5–9 months of pregnancy. The prevalence of vitamin D deficiency was 95.8% [133].
- The prevalence of vitamin D deficiency was determined in a diverse group of 559 women in South Carolina, USA at latitude 32°N. Mean age was 25.0 ± 5.4 (range 14–43) years; African American (48%), Hispanic (38%), Caucasian/Other (14%). Mean gestational age was 18.5 ± 8.4 (median 14.6, range 6.4–39.6) weeks. Vitamin D status was defined as 25(OH)D < 50 nmol/L deficiency; <80 nmol/L insufficiency. Forty-eight percent were vitamin D deficient, an additional 37% insufficient. The greatest degree was in the African American women (68% deficient; 94% insufficient) [125].
- Despite abundant sunshine and latitude consistent with year-long vitamin D synthesis, 65.5% of a largely low-risk antenatal population in rural Victoria, Australia had insufficient vitamin D. Over 5.0% of women had vitamin D levels that pose a significant neonatal and adult health risk [135].
- A cross-sectional study including 50 women in labour with a singleton term pregnancy in Pakistan measured vitamin D status in maternal blood before delivery and cord blood at delivery. Vitamin D sufficiency was noted in 11 (22%), insufficiency in 16 (32%), and deficiency in 23 (46%) of the 50 participants whereas sufficiency and deficiency, respectively, were noted in 6 (12%) and 44 (88%) of the newborns. There was a positive correlation between the vitamin D levels in maternal and cord blood (r = 0.03; p < 0.003). Maternal vitamin D levels were significantly affected by sunlight exposure (p < 0.007) and quality of diet (p < 0.01). The authors concluded that vitamin D deficiency is high among pregnant urban Pakistani women and their newborns and is a public health problem that needs urgent attention [137].
3.1.2. Bone Health
- Doctors in Leicester City, UK reported that a significant number of south Asian mothers visiting their clinic had vitamin D deficiency at the end of pregnancy. A substantial number of their offspring had infantile and adolescent rickets including some with extremely severe bony deformities. In addition, there was an increase in late (5–10 days of age) and late-late (2–12 weeks of age) neonatal hypocalcaemia presenting predominantly with seizures, demonstrating the involvement of vitamin D in brain function [50].
3.1.3. Pregnancy Maintenance
- A cohort study of 23,423 nulliparous pregnant women taking part in the Norwegian Mother and Child Cohort Study found a 27% reduction in risk of preeclampsia in women taking vitamin D supplements relative to those who did not take supplements [145]. However, because vitamin D intake is highly correlated with the intake of long chain n-3 fatty acids in the Norwegian diet, the authors cautioned that further research is needed to disentangle the separate effects of these nutrients.
3.1.4. Brain Development
- Vitamin D’s nuclear hormone receptor regulates gene expression and nervous system development [54]. There is evidence that vitamin D during pregnancy is involved in foetal brain development and that maternal vitamin D deficiency during pregnancy can alter the structure and function of the brain resulting in life long behavioural changes in the offspring [146,147,148].
- ○ A pilot case-control study assessing the association between third trimester maternal serum 25(OH)D and the risk of schizophrenia included 26 cases and 51 controls. The results showed that 25(OH)D concentration varied by season and were lower in African American women as predicted. Within the African American mothers, a subgroup with markedly lower levels of 25(OH)D had a non-significant increase in schizophrenia [149].
- ○ A larger case-control study included 424 cases and matched control (sex and age) from the Danish Psychiatric Central Register. There was a significant seasonal variation in 25(OH)D and significantly lower 25(OH)D in the offspring of migrants. The risk of schizophrenia was significantly associated with neonatal 25(OH)D. Those with the lowest concentration had an increased risk of schizophrenia although the exposure risk was nonlinear (i.e., higher than normal 25(OH)D was also associated with schizophrenia). Shifting all subjects to the optimal concentration of 25(OH)D could potentially avert 43.6% of schizophrenic cases in this group of patients. The authors concluded that prenatal vitamin D supplements in women at risk of deficiency could reduce the risk of schizophrenia in their offspring [149]. However, one should consider the complex nature of vitamin D effects since either deficiency or excess may be harmful [53].
3.2. Intervention Trials
3.2.1. Studies Showing Correlation between Maternal and Foetal Vitamin D Status
- Five hundred and six pregnant women were given 400 IU (10μg) of vitamin D per day from about the 12th week of pregnancy until delivery [150]. A control group of 633 pregnant women was given a placebo. Maternal vitamin D was measured at the 24th and 34th weeks of pregnancy and at delivery and infant vitamin D was measured in umbilical blood at birth and on the sixth day following birth. Plasma concentrations of 25(OH)D, which showed a seasonal variation, was higher in mothers and infants in the treated group. Cord-blood 25(OH)D correlated with maternal values at delivery. A defect of dental enamel was found in a high proportion of infants (many of whom had suffered from hypocalcaemia) born to the control women. These results suggest that vitamin D supplementation during pregnancy would be beneficial for mothers, whose intake from diet and skin synthesis is appreciably less than 500 IU of vitamin D daily.
3.2.2. Bone Health
- A prospective partially randomised study of vitamin D supplementation during pregnancy included Indian subjects (known to be vitamin D deficient) randomised in the second trimester to receive either one oral dose of 1500 μg vitamin D (group 1, n = 48) or two doses of 3000 μg vitamin D each in the second and third trimesters (group 2, n = 49) [151]. A control group included 43 non-supplemented mother-infant pairs under “usual care”. Median maternal 25(OH)D at term was higher in group 2 (58.7, interquartile range (IQR) 38.4–89.4 nmol/L) vs. group 1 (26.2, IQR 17.7–57.7 nmol/L) and Control group (39.2, IQR 21.2–73.4 nmol/L) (p = 0.000). Birth weight, length and head circumference were greater and the anterior fontanelle (soft spot on the head) was smaller in groups 1 and 2 (3.08 and 3.03 kg, 50.0 and 50.1 cm, 34.5 and 34.4 cm, 2.6 and 2.5 cm, respectively) vs. Control (2.77 kg, 49.4, 33.6, 3.3 cm; p = 0.000 for length, head circumference and fontanelle and p = 0.003 for weight). These differences were still evident at 9 months.
4. Evidence of the Potential Benefits of Maternal Folic Acid Supplementation for Foetal/Infant Brain Health
5. Evidence of the Potential Benefits of Maternal Iodine Supplementation for Foetal/Infant Brain Health
5.1. Epidemiological Evidence
5.1.1. Iodine Deficiency—Girls of Child Bearing Age
- A cross-sectional survey of iodine status systematically assessed in schoolgirls aged 14–15 years attending secondary school in nine UK centres included 810 participants provided 737 urine samples [162]. Data for dietary habits and iodine status were available for 664 participants. Urinary iodine measurements indicative of mild iodine deficiency were present in 51% (n = 379) of participants, moderate deficiency in 16% (n = 120), and severe deficiency in 1% (n = 8). Prevalence of iodine deficiency was highest in Belfast (85%, n = 135). Tap water iodine concentrations were low or undetectable and were not positively associated with urinary iodine concentrations. There were independent associations between low urinary iodine excretion and sampling in summer (p < 0·0001), UK geographical location (p < 0·0001), low intake of milk (p = 0·03), and high intake of eggs (p = 0·02). These results suggest that the UK population is iodine deficient. Since developing foetuses are the most susceptible to adverse effects of iodine deficiency and even mild perturbations of maternal and foetal thyroid function have an effect on neurodevelopment, these findings are of potential major public health importance. This study draws attention to an urgent need for a comprehensive investigation of UK iodine status and implementation of evidence-based recommendations for iodine supplementation [154].
5.1.2. Iodine Deficiency—Pregnant Women
- A 2005 study including urban and rural sites from each of the 11 Chinese provinces concluded that effective iodised salt program has brought iodine sufficiency to most of China, but pregnant women in some areas may still risk deficiency and need further supplements [155].
- A 2008 State-wide survey in Rajasthan, an Indian State where the sale of non-iodised salt for human consumption was banned in 1992 reported that 41.9% of the households used salt containing insufficient levels of iodine, 23.0% used non-iodised salt and the median urine iodine concentration was 127 μg/L in pregnant women indicating iodine deficiency. These results indicate that household salt iodine content at its current mandated concentration does not supply sufficient iodine for pregnancy requirements [156].
- A national, Kyrgyzstan population-representative survey during autumn 2007 collected household salt and urine samples of school-age children and pregnant women for quantitative iodine measurements and measured maternal thyroid volume. Even though universal salt iodization was re-mandated in 2001 and 97.9% of salt samples were iodised, 39.5% had > or = 15 mg iodine/kg. The median urinary iodine concentration of pregnant women was only 111 μg/L and their thyroid volume increased with the duration of pregnancy. The iodine consumption among pregnant women from iodised salt did not assure their dietary requirements [157].
- A 2004 study to determine the prevalence of reduced iodine intake by measuring urinary iodide concentrations in pregnant and non-pregnant women from the north east of England included 227 women at 15 weeks gestation and 227 non-pregnant age matched controls. 3.5% of pregnant women had evidence of iodine deficiency, and 40% were borderline deficient [154].
- A case-control study completed in Surrey, UK included 100 women at 12 weeks gestation and 57 women of childbearing age as a control. Based on urine analysis, the pregnant women were mild to moderately iodine deficient. Seventy-five percent of participants took a nutritional supplement but only 42% took a supplement containing iodine. Significantly lower iodine levels were found in those who did not consume milk daily [163].
5.1.3. Iodine Deficiency—Pregnant or Lactating Women
- A 2007 review of cross-sectional and prospective studies to describe the iodine nutrition of pregnant and lactating women in Hong Kong, where intake is of borderline sufficiency revealed an increase in the urinary iodine concentration as pregnancy advances. A significant percentage of women had a sub-normal serum thyroid hormone concentration at full term. Although iodine is concentrated by the mammary gland, 19% of all mothers had low iodine concentrations in their breast milk. The moderate correlation between the concentrations of iodine in breast milk and urine suggests that an adequate maternal urinary iodine concentration cannot reliably indicate that an infant is getting enough iodine in breast milk. Therefore, some breast-fed infants may still be at risk of low iodine intake, and additional iodine supplements, other than salt iodisation, would be warranted in this population [158].
- A cross-sectional study conducted in Iran between 1996 and 1998 in 403 pregnant women and a study of 100 lactating women conducted in 2003 included evaluated thyroid size, and both urinary and breast milk iodine concentrations. When data were combined for the cities of Ilam, Isfahan and Tehran, where women have an adequate or more than adequate median urinary iodine concentration, 51% of pregnant women had a urinary iodine concentration less than that recommended during pregnancy. The mean urinary iodine concentration in lactating women was 250 μg/L, and 16% of women had a urinary iodine concentration <100 μg/L. Grade 1 goitre was present in 8% of lactating women, and another 8% had grade 2 goitre [159].
- A study including 433 pregnant and 95 non-pregnant women in Tayside, Scotland, mean gestational age at recruitment of 11.5 weeks measured urinary iodine and a range of thyroid hormones. Even though iodised salt was available in the area, only 30% of women consumed it and the iodine intake of these women had not increased to meet the higher requirements of pregnancy (~250 µg/day). Indeed, the urinary iodine was the same in pregnant and non-pregnant women. Approximately 40% of the pregnant women from this area of the UK had urinary iodine excretion below those corresponding to half the recommended intake [160]. The ensuing failure to increase their T4 during the 1st trimester of pregnancy may well have adverse effects on the progeny’s neurodevelopment [77].
5.1.4. Studies Showing Maternal Thyroid Deficiency Impacts Brain Development of Her Child
- Serum samples collected from 25,216 pregnant women between January 1987 and March 1990 were tested for thyrotropin to recruit 47 women with serum thyrotropin concentration at or above the 99.7th percentile of the values for all the pregnant women, 15 women with values between the 98th and 99.6th percentiles, inclusive, in combination with low thyroxine levels, and 124 matched women with normal values. Their seven-to-nine-year-old children, none of whom had hypothyroidism as newborns, underwent 15 tests relating to intelligence, attention, language, reading ability, school performance, and visual-motor performance. The children of the 62 women with high serum thyrotropin concentrations performed slightly less well on all 15 tests. Their full-scale IQ scores on the Wechsler Intelligence Scale for Children, third edition, averaged 4 points lower than those of the children of the 124 matched control women (p = 0.06); 15% had scores of 85 or less, as compared with 5% of the matched control children. Of the 62 women with thyroid deficiency, 48 were not treated for the condition during the pregnancy under study. The full-scale IQ scores of their children averaged 7 points lower than those of the 124 matched control children (p = 0.005); 19% had scores of 85 or less. Eleven years after the pregnancy under study, 64% of the untreated women and 4% of the matched control women had confirmed hypothyroidism. Although this study did not include testing for iodine status during pregnancy, it does show that undiagnosed hypothyroidism in pregnant women may adversely affect their offspring [161].
5.1.5. Studies Showing Maternal Iodine Status Impacts Brain Development of Her Child
5.1.5.1. Severe Deficiency
- A meta-analysis of 37 studies including 12,291 sixteen year olds born and raised in China before and after iodine food fortification compared to those living in naturally iodine sufficient locations (IS) with those in severely iodine deficient (ID) areas, or children in ID areas born before and after the introduction of iodine supplementation. IQ was measured using Binet or Raven Scales. There was a 12.45, 12.3 and 4.8 increase in IQ points respectively, for the children living in IS communities compared with:
- ○ Those living in ID areas with no iodine supplementation;
- ○ With inadequate iodine supplementation;
- ○ Or children who had received iodine during their mothers’ pregnancy and after birth.
5.1.5.2. Mild to Moderate Deficiency
- Iodine status was investigated in 1,000 women of the Avon Longitudinal Study of Parents and Children (ALSPAC) cohort who were recruited in the 1990s. Iodine concentration (and creatinine to adjust for urine volume) was measured in urine samples from pregnant women of median gestational age 13 weeks [83]. Women were grouped as iodine-deficient or sufficient according to WHO criteria. The relationships between maternal iodine status and child’s IQ at age 8 (Wechsler Intelligence Scale for Children), reading ability at age 9 (Neale Analysis of Reading Ability), and Key Stage 2 scores at age 11 were analysed using logistic regression. The group was mildly-to-moderately iodine deficient and 61% of women were classed as iodine deficient when using the creatinine-adjusted data. The children of women deficient in iodine were more likely to have a total IQ score below the 25th percentile (unadjusted OR = 1.42, 95% CI 1.05–1.94) after adjusting for mother’s parenting score, home score, family adversity during pregnancy, life-event score, dietary intake of n-3 fatty acids and iron, gender, ethnicity, maternal age, smoking, alcohol intake, parity, breastfeeding, partner at birth, parental education, housing status, crowding and use of iron, fish oil and vitamin/mineral supplements. The level of maternal iodine appeared sufficient to affect brain development in the offspring as shown by:
- ○ Significantly lower total IQ at age 8;
- ○ Significantly lower reading accuracy at age 9;
- ○ Poorer school performance at age 11, including significantly poorer in mathematics.
5.2. Intervention Trials
6. Safe Intake Recommendations
6.1. DHA
- Child Health Foundation;
- Diabetic Pregnancy Study Group;
- European Association of Perinatal Medicine;
- European Society for Clinical Nutrition and Metabolism;
- European Society for Pediatric Gastroenterology;
- Hepatology and Nutrition;
- Committee on Nutrition;
- International Federation of Placenta Associations;
- International Society for the Study of Fatty Acids and Lipids.
- Fat, as a proportion of total energy needs should be the same in pregnant and lactating women as recommended for the normal population.
- Pregnant and lactating women require at least 200 mg of DHA per day. Numerous populations studies throughout westernized countries have confirmed that our intake is much lower than this, with a mean of about 150 mg per day [169].
- Maternal intake of fish, fish oils or omega-3 LC-PUFAs result in a slightly longer duration of gestation, a somewhat higher birth weight and a reduced risk of early preterm delivery.
- The foetus and neonate must receive sufficient LC-PUFA to support optimal visual and cognitive development.
- Breast feeding is endorsed as the preferred method of feeding to supply LC-PUFAs to the growing infant for the first 6 months of life. Dietary LC-PUFA supply should also continue after that time, but currently there is insufficient data to provide specific recommendations.
- Dietary intakes up to 1 g DHA/day or 2.7 g EPA + DHA/day have been used in clinical trials without occurrence of significant adverse effects.
6.2. Vitamin D
| Agency/Government | Recommendation |
|---|---|
| UK—for the elderly, pregnant & lactating women [173] | Dietary Reference Values 10 µg/day (400 IU/day) |
| UK Department of Health 2007—for pregnant and lactating women [45] | 10 µg/day (400 IU/day) |
| UK National Institute of Health and Clinical Excellence Guideline Review Panel 2007 [45] | All women should be informed about the importance for their own and their baby’s health of maintaining adequate vitamin D stores during pregnancy and whilst breast feeding and may choose to take 400 IU/day. |
| Canadian Paediatric Society [45] | 50 μg/day (2000 IU/day) throughout pregnancy |
| Federal Department of Health Canada [45] | 5 μg/day (200 IU/day) for pregnant and breast-feeding women |
| European Commission [45] | 10 μg/day (400 IU/day) during pregnancy |
| World Health Organisation 2004 [45] | 5 μg/day (200 IU/day) during pregnancy |
| The Institute of Medicine US 2010 [172,174] | 15 μg/day (600 IU/day) in pregnant and lactating women |
| US Endocrine Task Force on Vitamin D 2011 [174] | 37.5–50 μg/day (1500–2000 IU) in pregnant and lactating women with vitamin D deficiency. |
6.3. Folic Acid
| Age (Years) | Females (μg/day) | During Pregnancy (μg/day) | During Lactation (μg/day) |
|---|---|---|---|
| 9–13 | 600 | N/A | N/A |
| 14–18 | 600 | 800 | 800 |
| >19 | 1000 | 1000 | 1000 |
6.4. Iodine
| Agency/Government | Recommendation |
|---|---|
| The Australian National Health and Medical Research Council [188] | 220 μg/day for pregnant women and 270 μg/day for breast feeding women |
| New Zealand Ministry of Health [188] | 220 μg/day for pregnant women and 270 μg/day for breast feeding women |
| US Food and Nutrition Board of the Institute of Medicine [66,189] | 220 μg/day for pregnant women and 290 μg/day for lactating women |
| The American Thyroid Association [165] | 150 μg/day during pregnancy [67] and lactation, and that vitamins for prenatal use or use during pregnancy should be enriched with 150 μg/day of iodine |
7. Conclusions
Acknowledgments
Conflict of Interest
References
- Innis, S.M. Perinatal biochemistry and physiology of long chain polyunsaturated fatty acids. J. Pediatr. 2003, 143, 81–88. [Google Scholar] [CrossRef]
- Martinez, M. Developmental profiles of polyunsaturated fatty acids in the brain of normal infants and patients with peroxisomal diseases: Severe deficiency of docosahexaenoic acid in Zellweger’s and pseudo-Zellweger’s syndromes. World Rev. Nutr. Diet. 1991, 66, 87–102. [Google Scholar]
- Gerster, H. Can adults adequately convert alpha-linolenic acid (18:3n-3) to eicosapentaenoic acid (20:5n-3) and docosahexaenoic acid (22:6n-3). Int. J. Vitam. Nutr. Res. 1998, 68, 159–173. [Google Scholar]
- Hussein, N.; Ah-Sing, E.; Wilkinson, P.; Leach, C.; Griffin, B.A.; Millwood, D.J. Long chain conversion of [13C] linoleic acid and alpha-linolenic acid in response to marked changes in their dietary intake in men. J. Lipid Res. 2005, 46, 269–680. [Google Scholar]
- Plourde, M.; Cunnane, S.C. Extremely limited synthesis of long chain polyunsaturates in adults: Implications for their dietary essentiality and use as supplements. Appl. Physiol. Nutr. Metab. 2007, 2, 619–634. [Google Scholar] [CrossRef]
- McCann, J.C.; Ames, B.N. Is docosahexaenoic acid, an n-3 long-chain polyunsaturated fatty acid, required for development of normal brain function? An overview of evidence from cognitive and behavioral tests in humans and animals. Am. J. Clin. Nutr. 2005, 82, 281–295. [Google Scholar]
- Rapoport, S.I.; Chang, M.C.; Spector, A.A. Delivery and turnover of plasma-derived essential PUFAs in mammalian brain. J. Lipid Res. 2001, 42, 678–685. [Google Scholar]
- Wainwright, P.E. Dietary essential fatty acids and brain function: A developmental perspective on mechanisms. Proc. Nutr. Soc. 2002, 61, 61–69. [Google Scholar] [CrossRef]
- Lukiw, W.; Cui, J.G.; Marcheselli, V.L.; Bodker, M.; Botkjaer, A.; Gotlinger, K.; Serhan, C.N.; Bazan, N.G. A role for docosahexaenoic acid-derived neuroprotectin D1 in neural cell survival and Alzheimer disease. J. Clin. Invest. 2005, 115, 2774–2783. [Google Scholar]
- Calon, F.; Lim, G.P.; Yang, F.; Morihara, T.; Teter, B.; Ubeda, O.; Rostaing, P.; Triller, A.; Salem, N.; Ashe, K.H.; Frautschy, S.A.; Cole, G.M. Docosahexaenoic acid protects from dendritic pathology in an Azheimer’s disease mouse model. Neuron 2004, 43, 633–645. [Google Scholar]
- Calon, F.; Lim, G.P.; Morihara, T.; Yang, F.; Ubeda, O.; Salem, N.; Frautschy, S.A.; Cole, G.M. Dietary n-3 polyunsaturated fatty acid depletion activates caspases and decreases NMDA receptors in the brain of a transgenic mouse model of Alzheimer’s disease. Eur. J. Neurosci. 2005, 22, 617–626. [Google Scholar] [CrossRef]
- Hashimoto, M.; Tanabe, Y.; Fujii, Y.; Kikuta, T.; Shibata, H.; Shido, O. Chronic administration of docosahexaenoic acid ameliorates the impairment of spatial cognition learning ability in amyloid beta-infused rats. J. Nutr. 2005, 135, 549–555. [Google Scholar]
- Gamoh, S.; Hashimoto, M.; Hossain, S.; Masumura, S. Chronic administration of docosahexaenoic acid improves the performance of radial arm maze task in aged rats. Clin. Exp. Pharmacol. 2001, 28, 266–270. [Google Scholar] [CrossRef]
- Otto, S.J.; van Houwelingen, A.C.; Badart-Smook, A.; Hornstra, G. Changes in the maternal essential fatty acid profile during early pregnancy and the relation of the profile to diet. Am. J. Clin. Nutr. 2001, 73, 302–307. [Google Scholar]
- Otto, S.J.; van Houwelingen, A.C.; Badart-Smook, A.; Hornstra, G. Comparison of the peripartum and postpartum phospholipid PUFA profiles of lactating and non-lactating women. Am. J. Clin. Nutr. 2001, 73, 1074–1079. [Google Scholar]
- Hornstra, G. LCPUFA in Maternal Nutrition; DSM Nutritional Products Ltd.: Basel, Switzerland, 2004; p. 11. [Google Scholar]
- Makrides, M.; Gibson, R. LC-PUFA requirements during pregnancy and lactation. Am. J. Clin. Nutr. 2000, 71, 307S–311S. [Google Scholar]
- Helland, I.B.; Saugstad, O.D.; Smith, L.; Saarem, K.; Solvoll, K.; Ganes, T.; Drevon, C.A. Similar effects on infants of n-3 and n-6 fatty acids supplementation to pregnant and lactating women. Pediatrics 2001, 108. [Google Scholar]
- De la Presa Owens, S.; Lopez-Sabater, M.D.; Rivero-Urgell, M. Fatty acid composition of human milk in Spain. J. Pediatr. Gastroenterol. Nutr. 1996, 22, 180–185. [Google Scholar] [CrossRef]
- Huisman, M.; van Beusekom, C.M.; Lantins, C.I.; Nijeboer, H.J.; Muskiet, F.A.; Boersma, E.R. Triglycerides, fatty acids, sterols, mono- and disaccharides and sugar alcohols in human milk and current types of infant formula milk. Eur. J. Clin. Nutr. 1995, 50, 255–260. [Google Scholar]
- Marangoni, F.; Agostani, C.; Lammardo, A.M.; Bonvissuto, M.; Giovannini, M.; Galli, C.; Riva, E. Polyunsaturated fatty acids in maternal plasma and in breast milk. Prostaglandins Leukot. Essent. Fatty Acids 2002, 66, 535–540. [Google Scholar] [CrossRef]
- Marin, M.C.; Sanjurjo, A.; Rodrigo, M.A.; de Alaniz, M.J.T. Long-chain polyunsaturated fatty acids in breast milk in La Plata, Argentina: Relationship with maternal nutrition status. Prostaglandins Leukot. Essent. Fatty Acids 2005, 73, 355–360. [Google Scholar] [CrossRef]
- Ogunleye, A.; Fakoya, A.T.; Niizeki, S.; Tojo, H.; Sasajima, I.; Kobayashi, M.; Tateishi, S.; Yamaguchi, K. Fatty acid composition of breast milk from Nigerian and Japanese women. J. Nutr. Sci. Vitaminol. 1991, 37, 435–442. [Google Scholar] [CrossRef]
- Olafsdottir, A.S.; Thorsdottir, I.; Wagner, K.H.; Elmadfa, I. Polyunsaturated fatty acids in the diet and breast milk of lactating Icelandic women with traditional fish and cod liver consumption. Ann. Nutr. Metab. 2006, 50, 270–276. [Google Scholar] [CrossRef]
- Torres, A.G.; Ney, J.G.; Meneses, F.; Trugo, N.M.F. Polyunsaturated fatty acids and conjugated linoleic acid isomers in breast milk are associated with plasma non-esterified and erythrocyte membrane fatty acid composition in lactating women. Br. J. Nutr. 2006, 95, 517–524. [Google Scholar] [CrossRef]
- Yuhas, R.; Pramuk, K.; Lien, E.L. Human milk fatty acid composition from nine countries varies most in DHA. Lipids 2006, 41, 851–858. [Google Scholar] [CrossRef]
- Strucinska, M. Vegetarian diets of breastfeeding women in the light of dietary recommendations. Rocz. Panstw. Zakl. Hig. 2002, 53, 65–79. [Google Scholar]
- Singer, P. Lower levels of omega-3 fatty acids in vegetarians and vegans. KIM Komplemet. Integr. Med. Artztezeitschrift Naturheilverfahren 2008, 49, 16–23. [Google Scholar]
- Kornsteiner, M.; Singer, I.; Elmadfa, I. Very low n-3 long chain polyunsaturated fatty acid status in Austrian vegetarians and vegans. Ann. Nutr. Metab. 2008, 52, 37–47. [Google Scholar] [CrossRef]
- Al, M.D.; van Houwelingen, A.C.; Hornstra, G. Relationship between birth order and the maternal and neonatal DHA status. Eur. J. Clin. Nutr. 1997, 51, 548–553. [Google Scholar]
- Monique, D.M.; Adriana, A.; Houwelingena, C.V.; Magritha, M.H.P. Essential fatty acids, pregnancy and pregnancy outcome. Chapter 2 biochemical EFA status of mothers and their neonates after normal pregnancy. Early Hum. Dev. 1990, 24, 239–248. [Google Scholar] [CrossRef]
- Reynolds, A. Breast feeding and brain development. Pediatr. Clin. North Am. 2001, 48, 159–171. [Google Scholar] [CrossRef]
- Lucas, A.; Morley, R.; Isaacs, E. Nutrition and mental development. Nutr. Rev. 2001, 59, S32–S33. [Google Scholar]
- Anderson, J.W.; Johnson, B.M.; Remley, D.T. Breast feeding and cognitive development: A meta-analysis. Am. J. Clin. Nutr. 1999, 70, 525–535. [Google Scholar]
- Jain, A.; Concato, J.; Leventhal, J.M. How good is the evidence linking breastfeeding and intelligence? Pediatrics 2002, 109, 1044–1053. [Google Scholar]
- Simmer, K. Long chain polyunsaturated fatty acid supplementation in preterm infants. Cochrane Database Syst. Rev. 2000. [Google Scholar]
- Drover, J.R.; Hoffman, D.R.; Castañeda, Y.S.; Morale, S.E.; Garfield, S.; Wheaton, D.H.; Birch, E.E. Cognitive function in 18-month-old term infants of the DIAMOND study: A randomised, controlled clinical trial with multiple dietary levels of docosahexaenoic acid. Early Hum. Dev. 2011, 87, 223–230. [Google Scholar] [CrossRef]
- Minns, L.M.; Kerling, E.H.; Neely, M.R.; Sullivan, D.K.; Wampler, J.L.; Harris, C.L.; Berseth, C.L.; Carlson, S.E. Toddler formula supplemented with docosahexaenoic acid (DHA) improves DHA status and respiratory health in a randomised, double-blind, controlled trial of US children less than 3 years of age. Prostagland Leukot. Essent. Fatty Acids 2010, 82, 287–293. [Google Scholar] [CrossRef]
- Ryan, A.S.; Astwood, J.D.; Gautier, S.; Kuratko, C.N.; Nelson, E.B.; Salem, N. Effects of long-chain polyunsaturated fatty acid supplementation on neurodevelopment in childhood: A review of human studies. Prostagland Leukot. Essent. Fatty Acids 2010, 82, 305–314. [Google Scholar]
- Birch, E.E.; Carlson, S.E.; Hoffman, D.R.; Fitzgerald-Gustafson, K.M.; Fu, V.L.; Drover, J.R.; Castañeda, Y.S.; Minns, L.; Wheaton, D.K.; Mundy, D.; Marunycz, J.; Diersen-Schade, D.A. The DIAMOND (DHA Intake And Measurement Of Neural Development) Study: A double-masked, controlled clinical trial of the maturation of infant visual acuity as a function of the dietary level of docosahexaenoic acid. Am. J. Clin. Nutr. 2010, 91, 848–859. [Google Scholar] [CrossRef]
- Hoffman, D.R.; Boettcher, J.A.; Diersen-Schade, D.A. Toward optimizing vision and cognition in term infants by dietary docosahexaenoic and arachidonic acid supplementation: A review of randomised controlled trials. Prostaglandins Leukot. Essent. Fatty Acids 2009, 81, 151–158. [Google Scholar] [CrossRef]
- Makrides, M.; Gibson, R.A.; McPhee, A.J.; Collins, C.T.; Davis, P.G.; Doyle, L.W.; Simmer, K.; Colditz, P.B.; Morris, S.; Smithers, L.G.; Willson, K.; Ryan, P. Neurodevelopmental outcomes of preterm infants fed high-dose docosahexaenoic acid: A randomised controlled trial. JAMA 2009, 301, 175–182. [Google Scholar]
- Fleith, M.; Clandinin, M.T. Dietary PUFA for preterm and term infants: Review of clinical studies. Crit. Rev. Food Sci. Nutr. 2005, 45, 205–229. [Google Scholar] [CrossRef]
- Hyppönen, E.; Power, C. Hypovitaminosis D in British adults at age 45 y: Nationwide cohort study of dietary and lifestyle predictors. Am. J. Clin. Nutr. 2007, 85, 860–868. [Google Scholar]
- Hyppönen, E.; Boucher, B.J. Avoidance of vitamin D deficiency in pregnancy in the United Kingdom: The case for a unified approach in National policy. Br. J. Nutr. 2010, 104, 309–314. [Google Scholar] [CrossRef]
- Calvo, M.S.; Whiting, S.J.; Barton, C.N. Vitamin D intake: A global perspective of current status. J. Nutr. 2005, 135, 310–316. [Google Scholar]
- Vieth, R.; Bischoff-Ferrari, H.; Boucher, B.J.; Dawson-Hughes, B.; Garland, C.F.; Heaney, R.P.; Holick, M.F.; Hollis, B.W.; Lamberg-Allardt, C.; McGrath, J.J.; et al. The urgent need to recommend an intake of vitamin D that is effective. Am. J. Clin. Nutr. 2007, 85, 649–650. [Google Scholar]
- Lips, P. Vitamin D status and nutrition in Europe and Asia. J. Steroid. Biochem Mol. Biol. 2007, 103, 620–625. [Google Scholar] [CrossRef]
- Liu, N.Q.; Hewison, M. Vitamin D, the placenta and pregnancy. Arch. Biochem. Biophys. 2011. Available online: http://pubget.com/paper/22155151 (accessed on 22 March 2012).
- Shenoy, S.D.; Swift, P.; Cody, D.; Iqbal, J. Maternal vitamin D deficiency, refractory neonatal hypocalcaemia, and nutritional rickets. Arch. Dis. Child. 2005, 90, 437–438. [Google Scholar]
- Javaid, M.K.; Crozier, S.R.; Harvey, N.C.; Gale, C.R.; Dennison, E.M.; Boucher, B.J.; Arden, N.K.; Godfrey, K.M.; Cooper, C. Princess Anne Hospital Study Group. Maternal vitamin D status during pregnancy and childhood bone mass at age 9 years: A longitudinal study. Lancet 2006, 367, 36–43. [Google Scholar]
- Brooke, O.G.; Brown, I.R.; Bone, C.D.; Carter, N.D.; Cleeve, H.J.; Maxwell, J.D.; Robinson, V.P.; Winder, S.M. Vitamin D supplements in pregnant Asian women: effects on calcium status and foetal growth. Br. Med. J. 1980, 280, 751–754. [Google Scholar]
- Hyppönen, E. Preventing vitamin D deficiency in pregnancy: Importance for the mother and child. Ann. Nutr. Metab. 2011, 59, 28–31. [Google Scholar] [CrossRef]
- Mackay-Sim, A.; Féron, F.; Eyles, D.; Burne, T.; McGrath, J. Schizophrenia, vitamin D, and brain development. Int. Rev. Neurobiol. 2004, 59, 351–380. [Google Scholar] [CrossRef]
- Sørensen, I.M.; Joner, G.; Jenum, P.A.; Eskild, A.; Torjesen, P.A.; Stene, L.C. Maternal serum levels of 25-hydroxy-vitamin D during pregnancy and risk of type 1 diabetes in the offspring. Diabetes 2012, 61, 175–178. [Google Scholar]
- Morales, E.; Romieu, I.; Guerra, S.; Ballester, F.; Rebagliato, M.; Vioque, J.; Tardón, A.; Rodriguez Delhi, C.; Arranz, L.; Torrent, M.; Espada, M.; Basterrechea, M.; Sunyer, J. INMA Project. Maternal vitamin D status in pregnancy and risk of lower respiratory tract infections, wheezing, and asthma in offspring. Epidemiology 2012, 23, 64–71. [Google Scholar]
- Antony, A.C. In utero physiology: Role of folic acid in nutrient delivery and foetal development. Am. J. Clin. Nutr. 2007, 85, 598S–603S. [Google Scholar]
- Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin and Choline. Available online: http://www.nap.edu/catalog.php?record_id=6015#toc (accessed on 23 March 2012).
- Power, E.M. Determinants of healthy eating among low-income Canadians. Can. J. Public Health 2005, 96, S37–S42. [Google Scholar]
- Ortega, R.M.; López-Sobaler, A.M.; Andrés, P.; Rodriguez-Rodriguez, E.; Aparicio, A.; Bermejo, L.M.; López-Plaza, B. Changes in folate status in overweight/obese women following two different weight control programmes based on increased consumption of vegetables or fortified breakfast cereals. Br. J. Nutr. 2006, 96, 712–718. [Google Scholar]
- Sherwood, K.L.; Houghton, L.A.; Tarasuk, V.; O’Connor, D.L. One-third of pregnant and lactating women may not be meeting their folate requirements from diet alone based on mandated levels of folic acid fortification. J. Nutr. 2006, 136, 2820–2826. [Google Scholar]
- Kirkpatrick, S.I.; Tarasuk, V. Food insecurity is associated with nutrient inadequacies among Canadian adults and adolescents. J. Nutr. 2008, 138, 604–612. [Google Scholar]
- Prenatal Nutrition Guidelines for Health Professionals. Available online: http://www.hc-sc.gc.ca/fn-an/alt_formats/hpfb-dgpsa/pdf/pubs/folate-eng.pdf (accessed on 23 March 2012).
- Folate/Folic Acid Questions & Answers, New Zealand Ministry of Health. Available online: http://www.health.govt.nz/our-work/preventative-health-wellness/nutrition/folate-folic-acid (accessed on 23 March 2012).
- Stockley, L.; Lund, V. Use of folic acid supplements, particularly by low-income and young women: A series of systematic reviews to inform public health policy in the UK. Public Health Nutr. 2008, 11, 807–821. [Google Scholar]
- Zimmerman, M.M. Iodine deficiency. Endocr. Rev. 2009, 30, 376–408. [Google Scholar] [CrossRef]
- The Public Health Committee of the American Thyroid Association. Iodine Supplementation for pregnancy and lactation—United States and Canada. Recommendations of the American Thyroid Association. Thyroid 2006, 16, 949–951. [CrossRef]
- Zimmerman, M.B. Iodine deficiency in pregnancy and the effects of maternal iodine supplementation on the offspring: A review. Am. J. Clin. Nutr. 2009, 89, 668S–672S. [Google Scholar] [CrossRef]
- Glinoer, D. The regulation of thyroid function during normal pregnancy: Importance of the iodine nutrition status. Best Pract. Res. Clin. Endocrinol. Metab. 2004, 18, 133–152. [Google Scholar] [CrossRef]
- Smyth, P.P. Dietary iodine intakes in pregnancy. Ir. Med. J. 2006, 99, 103. [Google Scholar]
- Zimmermann, M.B. The adverse effects of mild-to-moderate iodine deficiency during pregnancy and childhood: A review. Thyroid 2007, 17, 829–835. [Google Scholar] [CrossRef]
- Iodine Supplementation for Pregnant and Breastfeeding Women. NHMRC Public Statement, January 2010. Available online: http://www.nhmrc.gov.au/_files_nhmrc/publications/attachments/new45_statement.pdf (accessed on 23 March 2012).
- International Council for Control of Iodine Deficiency Disorders. The Network for the Sustained Elimination of Iodine Deficiency, Global Scorecard 2008. Available online: http://www.iccidd.org/pages/global-scorecard.php (accessed on 24 March 2012).
- Andersson, M.; Karumbunathan, V.; Zimmerman, M. Global iodine status in 2011 and trends over the past decade. J. Nutr. 2012, 142, 744–750. [Google Scholar] [CrossRef]
- Henderson, L.; Irving, K.; Gregory, J.; Bates, C.J.; Prentice, A.; Perks, J.; Swan, G.; Farron, M. The National Diet & Nutrition Survey: Adults Aged 19 to 64 Years. Vitamin and Mineral Intake and Urinary Analytes. 2003, 3. Available online: http://www.food.gov.uk/multimedia/pdfs/ndnsv3.pdf (accessed on 24 March 2012).
- Vanderpump, M.P.; Lazarus, J.H.; Smyth, P.P.; Laurberg, P.; Holder, R.L.; Boelaert, K.; Franklyn, J.A. British Thyroid Association UK Iodine Survey Group. Iodine status of UK schoolgirls: A cross-sectional survey. Lancet 2011, 377, 2007–2012. [Google Scholar]
- Bath, S.C.; Button, S.; Rayman, M.P. Iodine concentration of organic and conventional milk: Implications for iodine intake. Br. J. Nutr. 2011, 5, 1–6. [Google Scholar]
- Bath, S.; Button, S.; Rayman, M.P. Iodised salt availability in the United Kingdom. Proc. Nutr. Soc. 2011, 70, E117. [Google Scholar]
- Morreale de Escobar, G.; Jesús Obregón, M.; Escobar del Rey, F. Iodine deficiency and brain development in the first half of pregnancy. Public Health Nutr. 2007, 10, 1554–1570. [Google Scholar]
- Aghini Lombardi, F.A.; Pinchera, A.; Antonangeli, L.; Rago, T.; Chiovato, L.; Bargagna, S.; Bertucelli, B.; Ferretti, G.; Sbrana, B.; Marcheschi, M. Mild iodine deficiency during fetal/neonatal life and neuropsychological impairment in Tuscany. J. Endocrinol. Invest. 1995, 18, 57–62. [Google Scholar]
- Vermiglio, F.; Lo Presti, V.P.; Moleti, M.; Sidoti, M.; Tortorella, G.; Scaffidi, G.; Castagna, M.G.; Mattina, F.; Viola, M.A.; CrisÃ, A.; Artemisia, A.; Trimarchi, F. Attention deficit and hyperactivity disorders in the offspring of mothers exposed to mild-moderate iodine deficiency: A possible novel iodine deficiency disorder in developed countries. J. Clin. Endocrinol. Metab. 2004, 89, 6054–6060. [Google Scholar]
- Choudhury, N.; Gorman, K.S. Sub-clinical prenatal iodine deficiency negatively affects infant development in Northern China. J. Nutr. 2003, 133, 3162–3165. [Google Scholar]
- Bath, S. Maternal iodine status during pregnancy and the impact on cognitive outcomes in the offspring. Lancet 2012, in press.. [Google Scholar]
- Zimmerman, M.B. The role of iodine in human growth and development. Semin. Cell Dev. Biol. 2011, 22, 645–652. [Google Scholar]
- Hawkes, J.S.; Bryan, D.L.; Makrides, M.; Neumann, M.A.; Gibson, R.A. A randomised trial of supplementation with DHA rich tuna oil and its effects on the human milk cytokines interleuin 1B, interleukin 6 and tumor necrosis factor. Am. J. Clin. Nutr. 2002, 75, 754–760. [Google Scholar]
- Krauss-Etschmann, S.; Shadid, R.; Campoy, C.; Hoster, E.; Demmelmair, H.; Jiménez, M.; Gil, A.; Rivero, M.; Veszprémi, B.; Decsi, T.; Koletzko, B.V. Nutrition and Health Lifestyle (NUHEAL) Study Group. Effects of fish-oil and folate supplementation of pregnant women on maternal and foetal plasma concentrations of docosahexaenoic acid and eicosapentaenoic acid: A European randomised multicenter trial. Am. J. Clin. Nutr. 2007, 85, 1392–1400. [Google Scholar]
- Furuhjelm, C.; Warstedt, K.; Larsson, J.; Fredriksson, M.; Böttcher, M.F.; Fälth-Magnusson, K.; Duchén, K. Fish oil supplementation in pregnancy and lactation may decrease the risk of infant allergy. Acta Paediatr. 2009, 98, 1461–1467. [Google Scholar] [CrossRef]
- Innis, S.M.; Friesen, R.W. Essential n-3 fatty acids in pregnant women and early visual acuity maturation in term infants. Am. J. Clin. Nutr. 2008, 87, 548–557. [Google Scholar]
- Van Houwelingen, A.C.; Sørensen, J.D.; Hornstra, G.; Simonis, M.M.; Boris, J.; Olsen, S.F.; Secher, N.J. Essential fatty acid status in neonates after fish-oil supplementation during late pregnancy. Br. J. Nutr. 1995, 74, 723–731. [Google Scholar] [CrossRef]
- Dunstan, J.A.; Mori, T.A.; Barden, A.; Beilin, L.J.; Holt, P.G.; Calder, P.C.; Taylor, A.L.; Prescott, S.L. Effects of n-3 polyunsaturated fatty acid supplementation in pregnancy on maternal and foetal erythrocyte fatty acid composition. Eur. J. Clin. Nutr. 2004, 58, 429–437. [Google Scholar] [CrossRef]
- Velzing Aarts, F.V.; van der Klis, F.R.; van der Dijs, F.P.; van Beusekom, C.M.; Landman, H.; Capello, J.J.; Muskiet, F.A. Effect of three low dose fish oil supplements, administered during pregnancy, on neonatal long-chain polyunsaturated fatty acid stats at birth. Prostaglandins Leukot. Essent. Fatty Acids 2001, 65, 51–57. [Google Scholar] [CrossRef]
- Gibson, R.A.; Neumann, M.A.; Makrides, M. Effect of increasing breast milk docosahexaenoic acid on plasma and erythrocyte phospholipid fatty acids and neural indices of exclusively breast fed infants. Eur. J. Clin. Nutr. 1997, 51, 578–584. [Google Scholar]
- Furuhjelm, C.; Warstedt, K.; Fagerås, M.; Fälth-Magnusson, K.; Larsson, J.; Fredriksson, M. Allergic disease in infants up to 2 years of age in relation to plasma omega-3 fatty acids and maternal fish oil supplementation in pregnancy and lactation. Pediatr. Allergy Immunol. 2011, 22, 505–514. [Google Scholar] [CrossRef]
- Dunstan, J.A.; Mitoulas, L.R.; Dixon, G.; Doherty, D.A.; Hartmann, P.E.; Simmer, K.; Prescott, S.L. The effects of fish oil supplementation in pregnancy on breast milk fatty acid composition over the course of lactation: A randomised controlled trial. Pediatr. Res. 2007, 62, 689–694. [Google Scholar] [CrossRef]
- Jensen, C.L.; Maude, M.; Anderson, R.E.; Heird, W.C. Effect of docosahexaenoic acid supplementation of lactating women on the fatty acid composition of breast milk lipids and maternal and infant plasma phospholipids. Am. J. Clin. Nutr. 2000, 71, 292S–299S. [Google Scholar]
- Dunstan, J.; Simmer, K.; Dixon, G.; Prescott, S. Cognitive assessment of children at age 2.5 years after maternal fish oil supplementation in pregnancy: A randomised controlled trial. Arch. Dis. Child. Fetal Neonatal Ed. 2008, 93, F45–F50. [Google Scholar] [CrossRef]
- Helland, I.B.; Saugstad, O.D.; Saarem, K.; Van Houwelingen, A.C.; Nylander, G.; Drevon, C.A. Supplementation of n-3 fatty acids during pregnancy and lactation reduces maternal plasma lipid levels and provides DHA to the infants. J. Matern. Fetal Neonatal Med. 2006, 19, 397–406. [Google Scholar] [CrossRef]
- Hibbeln, J.R.; Davis, J.M.; Steer, C.; Emmett, P.; Rogers, I.; Williams, C.; Golding, J. Maternal seafood consumption in pregnancy and neurodevelopmental outcomes in childhood (ALSPAC study): An observational cohort study. Lancet 2007, 369, 578–585. [Google Scholar]
- Crawford, M.A.; Doyle, W.; Drury, P.; Lennon, A.; Costeloe, K.; Leighfield, M. n-6 and n-3 Fatty acids during early human development. J. Intern. Med. Suppl. 1989, 225, 159–169. [Google Scholar]
- Bakker, E.C.; Hornstra, G.; Blanco, C.E.; Vles, J.S. Relationship between long-chain polyunsaturated fatty acids at birth and motor function at 7 years of age. Eur. J. Clin. Nutr. 2009, 63, 499–504. [Google Scholar] [CrossRef]
- Krabbendam, L.; Bakker, E.; Hornstra, G.; van Os, J. Relationship between DHA status at birth and child problem behaviour at 7 years of age. Prostaglandins Leukot. Essent. Fatty Acids 2007, 76, 29–34. [Google Scholar] [CrossRef]
- Jacobson, J.L.; Jacobson, S.W.; Muckle, G.; Kaplan-Estrin, M.; Ayotte, P.; Dewailly, E. Beneficial effects of a polyunsaturated fatty acid on infant development evidence from the Inuit of Arctic Quebec. J. Pediatr. 2008, 152, 356–364. [Google Scholar] [CrossRef]
- Boucher, O.; Burden, M.J.; Muckle, G.; Saint-Amour, D.; Ayotte, P.; Dewailly, E.; Nelson, C.A.; Jacobson, S.W.; Jacobson, J.L. Neurophysiologic and neurobehavioral evidence of beneficial effects of prenatal omega-3 fatty acid intake on memory function at school age. Am. J. Clin. Nutr. 2011, 93, 1025–1037. [Google Scholar]
- Jacques, C.; Levy, E.; Muckle, G.; Jacobson, S.W.; Bastien, C.; Dewailly, E.; Ayotte, P.; Jacobson, J.L.; Saint-Amour, D. Long-term effects of prenatal omega-3 fatty acid intake on visual function in school-age children. J. Pediatr. 2001, 158, 83–90. [Google Scholar]
- Mendez, M.A.; Torrent, M.; Julvez, J.; Ribas-Fitó, N.; Kogevinas, M.; Sunyer, J. Maternal fish and other seafood intakes during pregnancy and child neurodevelopment at age 4 years. Public Health Nutr. 2009, 12, 1702–1710. [Google Scholar] [CrossRef]
- Cheruku, S.R.; Montgomery-Downs, H.E.; Farkas, S.L.; Thoman, E.B.; Lammi-Keefe, C.J. Higher maternal plasma DHA during pregnancy is associated with more mature neonatal sleep-state patterning. Amer. J. Clin. Nutr. 2002, 76, 608–613. [Google Scholar]
- Kannass, K.N.; Colombo, J.; Carlson, S.E. Maternal DHA levels and toddler free-play attention. Dev. Neuropsychol. 2009, 34, 159–174. [Google Scholar] [CrossRef]
- Szajewska, H.; Horvath, A.; Koletzko, B. Effect of n-3 long-chain polyunsaturated fatty acid supplementation of women with low-risk pregnancies on pregnancy outcomes and growth measures at birth: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2006, 83, 1337–1344. [Google Scholar]
- Makrides, M.; Duley, L.; Olsen, S.F. Marine oil, and other prostaglandin precursor, supplementation for pregnancy uncomplicated by pre-eclampsia or intrauterine growth restriction. Cochrane Database Syst. Rev. 2006. [Google Scholar]
- Allen, K.G.; Harris, M.A. The role of n-3 fatty acids in gestation and parturition. Exp. Biol. Med. 2001, 226, 498–506. [Google Scholar]
- Ramakrishnan, U.; Stein, A.D.; Parra-Cabrera, S.; Wang, M.; Imhoff-Kunsch, B.; Juárez-Márquez, S.; Rivera, J.; Martorell, R. Effects of docosahexaenoic acid supplementation during pregnancy on gestational age and size at birth: Randomized, double-blind, placebo-controlled trial in Mexico. Food Nutr. Bull. 2010, 31, S108–S116. [Google Scholar]
- Stein, A.D.; Wang, M.; Martorell, R.; Neufeld, L.M.; Flores-Ayala, R.; Rivera, J.A.; Ramakrishnan, U. Growth to age 18 months following prenatal supplementation with docosahexaenoic acid differs by maternal gravidity in Mexico. J. Nutr. 2011, 141, 316–320. [Google Scholar] [CrossRef]
- Columbo, J.; Kannass, K.N.; Shaddy, D.J.; Kundurthi, S.; Maikranz, J.M.; Anderson, C.J.; Blaga, O.M.; Carlson, S.E. Maternal DHA and the development of attention in infancy and toddlerhood. Child. Dev. 2004, 75, 1254–1267. [Google Scholar]
- Judge, M.; Harel, O.; Lammi-Keefe, C.J. Maternal consumption of DHA containing functional food during pregnancy: Benefit for infant performance on problem-solving but not on recognition memory tasks at age 9 months. Am. J. Clin. Nutr. 2007, 85, 1572–1577. [Google Scholar]
- Escolano-Margarit, M.V.; Ramos, R.; Beyer, J.; Csábi, G.; Parrilla-Roure, M.; Cruz, F.; Perez-Garcia, M.; Hadders-Algra, M.; Gil, A.; Decsi, T.; Koletzko, B.V.; Campoy, C. Prenatal DHA status and neurological outcome in children at age 5.5 years are positively associated. J. Nutr. 2011, 141, 1216–1223. [Google Scholar] [CrossRef]
- Makrides, M.; Gibson, R.; McPhee, A.; Yelland, L.; Quinlivan, J.; Ryan, P. OMInO Investigative Team. Effect of DHA supplementation during pregnancy on maternal depression and neurodevelopment of young children. JAMA 2010, 304, 1675–1683. [Google Scholar]
- Palmer, D.J.; Sullivan, T.; Gold, M.S.; Prescott, S.L.; Heddle, R.; Gibson, R.A.; Makrides, M. Effect of n-3 long chain polyunsaturated fatty acid supplementation in pregnancy on infants’ allergies in first year of life: Randomised controlled trial. BMJ. 2012, 344. Available online: http://www.bmj.com/content/344/bmj.e184 (accessed on 2 April 2012).
- Jensen, C.L.; Voigt, R.G.; Prager, T.C.; Zou, Y.L.; Fraley, J.K.; Rozelle, J.C.; Turcich, M.R.; Llorente, A.M.; Anderson, R.E.; Heird, W.C. Effects of maternal docosahexaenoic acid intake on visual function and neurodevelopment in breastfed term infants. Am. J. Clin. Nutr. 2005, 82, 125–132. [Google Scholar] [Green Version]
- Jensen, C.L.; Voigt, R.G.; Llorente, A.M.; Peters, S.U.; Prager, T.C.; Zou, Y.L.; Rozelle, J.C.; Turcich, M.R.; Fraley, J.K.; Anderson, R.E.; Heird, W.C. Effects of early maternal docosahexaenoic acid intake on neuropsychological status and visual acuity at five years of age of breast-fed term infants. J. Pediatr. 2010, 157, 900–905. [Google Scholar] [CrossRef]
- Helland, I.B.; Smith, L.; Saarem, K.; Saugstad, O.D.; Drevon, C.A. Maternal supplementation with very long chain n-3 fatty acids during pregnancy and lactation augments children’s IQ at 4 years of age. Pediatrics 2003, 111, e39–e44. [Google Scholar]
- Helland, I.B.; Smith, L.; Blomén, B.; Saarem, K.; Saugstad, O.D.; Drevo, C.A. Effect of supplementing pregnant and lactating mothers with n-3 very-long-chain fatty acids on children’s IQ and body mass index at 7 years of age. Pediatrics 2008, 122, e472–e479. [Google Scholar] [CrossRef]
- Bodnar, L.M.; Catov, J.M.; Simhan, H.N.; Holick, M.F.; Powers, R.W.; Roberts, J.M. Maternal vitamin D deficiency increases the risk of preeclampsia. J. Clin. Endocr. Metab. 2007, 92, 3517–3522. [Google Scholar]
- Holmes, V.A.; Barnes, M.S.; Alexander, H.D.; McFaul, P.; Wallace, J.M. Vitamin D deficiency and insufficiency in pregnant women: A longitudinal study. Br. J. Nutr. 2009, 102, 876–881. [Google Scholar] [CrossRef]
- O'Riordan, M.N.; Kiely, M.; Higgins, J.R.; Cashman, K.D. Prevalence of suboptimal vitamin D status during pregnancy. Ir. Med. J. 2008, 101, 240–243. [Google Scholar]
- Hamilton, S.A.; McNeil, R.; Hollis, B.W.; Davis, D.J.; Winkler, J.; Cook, C.; Warner, G.; Bivens, B.; McShane, P.; Wagner, C.L. Profound vitamin D deficiency in a diverse group of women during pregnancy living in a sun-rich environment at Latitude 32°N. Int. J. Endocrinol. 2010, 2010, p. 917428. Available online: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3004407/?tool=pubmed (accessed on 2 Apirl 2012).
- Johnson, D.D.; Wagner, C.L.; Hulsey, T.C.; McNeil, R.B.; Ebeling, M.; Hollis, B.W. Vitamin D deficiency and insufficiency is common during pregnancy. Am. J. Perinatol. 2011, 28, 7–12. [Google Scholar]
- Davis, L.M.; Chang, S.C.; Mancini, J.; Nathanson, M.S.; Witter, F.R.; O’Brien, K.O. Vitamin D insufficiency is prevalent among pregnant African American adolescents. J. Pediatr. Adolesc. Gynecol. 2010, 23, 45–52. [Google Scholar] [CrossRef]
- Alfaham, M.; Woodhead, S.; Pask, G.; Davies, D. Vitamin D deficiency: A concern in pregnant Asian women. Br. J. Nutr. 1995, 73, 881–887. [Google Scholar] [CrossRef]
- Grover, S.R.; Morley, R. Vitamin D deficiency in veiled or dark-skinned pregnant women. Med. J. Aust. 2001, 175, 251–252. [Google Scholar]
- Van der Meer, I.M.; Karamali, N.S.; Boeke, A.J.; Lips, P.; Middelkoop, B.J.; Verhoeven, I.; Wuister, J.D. High prevalence of vitamin D deficiency in pregnant non-Western women in The Hague, Netherlands. Am. J. Clin. Nutr. 2006, 84, 350–353. [Google Scholar]
- Cavalier, E.; Delanaye, P.; Morreale, A.; Carlisi, A.; Mourad, I.; Chapelle, J.P.; Emonts, P. Vitamin D deficiency in recently pregnant women. Rev. Med. Liege 2008, 63, 87–91. [Google Scholar]
- Kazemi, A.; Sharifi, F.; Jafari, N.; Mousavinasab, N. High prevalence of vitamin D deficiency among pregnant women and their newborns in an Iranian population. J. Womens Health 2009, 18, 835–839. [Google Scholar] [CrossRef]
- Asemi, Z.; Taghizadeh, M.; Sarahroodi, S.; Jazayeri, S.; Tabasi, Z.; Seyyedi, F. Assessment of the relationship of vitamin D with serum antioxidant vitamins E and A and their deficiencies in Iranian pregnant women. Saudi Med. J. 2010, 31, 1119–1123. [Google Scholar]
- Sachan, A.; Gupta, R.; Das, V.; Agarwal, A.; Awasthi, P.K.; Bhatia, V. High prevalence of vitamin D deficiency among pregnant women and their newborns in northern India. Am. J. Clin. Nutr. 2005, 81, 1060–1064. [Google Scholar]
- Teale, G.R.; Cunningham, C.E. Vitamin D deficiency is common among pregnant women in rural Victoria. Aust. N. Z. J. Obstet. Gynaecol. 2010, 50, 259–261. [Google Scholar] [CrossRef]
- Madar, A.A.; Stene, L.C.; Meyer, H.E. Vitamin D status among immigrant mothers from Pakistan, Turkey and Somalia and their infants attending child health clinics in Norway. Br. J. Nutr. 2009, 101, 1052–1058. [Google Scholar] [CrossRef]
- Karim, S.A.; Nusrat, U.; Aziz, S. Vitamin D deficiency in pregnant women and their newborns as seen at a tertiary-care center in Karachi, Pakistan. Int. J. Gynaecol. Obstet. 2011, 112, 59–62. [Google Scholar]
- Al Kalbani, M.; Elshafie, O.; Rawahi, M.; Al-Mamari, A.; Al-Zakwani, A.; Woodhouse, N. Vitamin D status in pregnant omanis: A disturbingly high proportion of patients with low vitamin D stores. Sultan Qaboos Univ. Med. J. 2011, 11, 52–55. [Google Scholar]
- Nicolaidou, P.; Hatzistamatiou, Z.; Papadopoulou, A.; Kaleyias, J.; Floropoulou, E.; Lagona, E.; Tsagris, V.; Costalos, C.; Antsaklis, A. Low vitamin D status in mother-newborn pairs in Greece. Calcif. Tissue Int. 2006, 78, 337–342. [Google Scholar] [CrossRef]
- Sloka, S.; Stokes, J.; Randell, E.; Newhook, L.A. Seasonal variation of maternal serum vitamin D in Newfoundland and Labrador. J. Obstet. Gynaecol. Can. 2009, 31, 313–321. [Google Scholar]
- Dror, D.K. Vitamin D status during pregnancy: Maternal, foetal, and postnatal outcomes. Curr. Opin. Obstet. Gynecol. 2011, 23, 422–426. [Google Scholar] [CrossRef]
- Butte, N.F.; Lopez-Alarcon, M.G.; Garza, C. Nutrient Adequacy of Exclusive Breastfeeding of the Term Infant during the First Six Months of Life; World Health Organization: Geneva, Switerland, 2002. Available online: http://whqlibdoc.who.int/publications/9241562110.pdf (accessed on 2 Apirl 2012).
- Derbyshire, E.; Davies, G.J.; Costarelli, V.; Dettmar, P.W. Habitual micronutrient intake during and after pregnancy in Caucasian Londoners. Matern. Child. Nutr. 2009, 5, 1–9. [Google Scholar]
- Tylavsky, F.A.; Cheng, S.; Lyytikäinen, A.; Viljakainen, H.; Lamberg-Allardt, C. Strategies to improve vitamin D status in northern European children: Exploring the merits of vitamin D fortification and supplementation. J. Nutr. 2006, 136, 1130–1134. [Google Scholar]
- Haugen, M.; Brantsaeter, A.L.; Trogstad, L.; Alexander, J.; Roth, C.; Magnus, P.; Meltzer, H.M. Vitamin D supplementation and reduced risk of preeclampsia in nulliparous women. Epidemiology 2009, 20, 720–726. [Google Scholar]
- Eyles, D.; Brown, J.; Mackay-Sim, A.; McGrath, J.; Feron, F. Vitamin D3 and brain development. Neuroscience 2003, 118, 641–653. [Google Scholar] [CrossRef]
- Eyles, D.W.; Feron, F.; Cui, X.; Kesby, J.P.; Harms, L.H.; Ko, P.; McGrath, J.J.; Burne, T.H. Developmental vitamin D deficiency causes abnormal brain development. Psychoneuroendocrinology 2009, 34, S247–S257. [Google Scholar]
- O’Loan, J.; Eyles, D.W.; Kesby, J.; Ko, P.; McGrath, J.J.; Burne, T.H. Vitamin D deficiency during various stages of pregnancy in the rat; its impact on development and behaviour in adult offspring. Psychoneuroendocrinology 2007, 32, 227–234. [Google Scholar]
- McGrath, J.J.; Burne, T.H.; Féron, F.; Mackay-Sim, A.; Eyles, D.W. Developmental vitamin D deficiency and risk of schizophrenia: A 10-year update. Schizophr. Bull. 2010, 36, 1073–1078. [Google Scholar] [CrossRef]
- Cockburn, F.; Belton, N.R.; Purvis, R.J.; Giles, M.M.; Brown, J.K.; Turner, T.L.; Wilkinson, E.M.; Forfar, J.O.; Barrie, W.J.; McKay, G.S.; Pocock, S.J. Maternal vitamin D intake and mineral metabolism in mothers and their newborn infants. Br. Med. J. 1980, 281, 11–14. [Google Scholar]
- Kalra, P.; Das, V.; Agarwal, A.; Kumar, M.; Ramesh, V.; Bhatia, E.; Gupta, S.; Singh, S.; Saxena, P.; Bhatia, V. Effect of vitamin D supplementation during pregnancy on neonatal mineral homeostasis and anthropometry of the newborn and infant. Br. J. Nutr. 2012, 3, 1–7. [Google Scholar]
- De-Regil, L.M.; Fernández-Gaxiola, A.C.; Dowswell, T.; Peña-Rosas, J.P. Effects and safety of periconceptional folate supplementation for preventing birth defects. Cochrane Database Syst. Rev. 2010. Available online: http://onlinelibrary.wiley.com/doi/10.1002/14651858.CD007950.pub2/otherversions (accessed on 2 Apirl 2012).
- Wolff, T.; Witkop, C.T.; Miller, T.; Syed, S.B. U.S. Preventive Services Task Force. Folic acid supplementation for the prevention of neural tube defects: an update of the evidence for the US Preventive Services Task Force. Ann. Intern. Med. 2009, 150, 632–639. [Google Scholar]
- Kibirige, M.S.; Hutchison, S.; Owen, C.J.; Delves, H.T. Prevalence of maternal dietary iodine insufficiency in the north east of England: Implications for the fetus. Arch. Dis. Child. Fetal Neonatal Ed. 2004, 89, F436–F439. [Google Scholar] [CrossRef]
- Yan, Y.Q.; Chen, Z.P.; Yang, X.M.; Liu, H.; Zhang, J.X.; Zhong, W.; Yao, W.; Zhao, J.K.; Zhang, Z.Z.; Hua, J.L.; et al. Attention to the hiding iodine deficiency in pregnant and lactating women after universal salt iodization: A multi-community study in China. J. Endocrinol. Invest. 2005, 28, 547–553. [Google Scholar]
- Ategbo, E.A.; Sankar, R.; Schultink, W.; van der Haar, F.; Pandav, C.S. An assessment of progress toward universal salt iodization in Rajasthan, India, using iodine nutrition indicators in school-aged children and pregnant women from the same households. Asia Pac. J. Clin. Nutr. 2008, 17, 56–62. [Google Scholar]
- Sultanalieva, R.B.; Mamutova, S.; van der Haar, F. The current salt iodization strategy in Kyrgyzstan ensures sufficient iodine nutrition among school-age children but not pregnant women. Public Health Nutr. 2010, 13, 623–630. [Google Scholar] [CrossRef]
- Kung, A.W. Iodine nutrition of pregnant and lactating women in Hong Kong, where intake is of borderline sufficiency. Public Health Nutr. 2007, 10, 1600–1601. [Google Scholar]
- Azizi, F. Iodine nutrition in pregnancy and lactation in Iran. Public Health Nutr. 2007, 10, 1596–1599. [Google Scholar]
- Barnett, C.; Visser, T.; Williams, F.; Toor, H.; Duran, S.; Presas, M.; Morreale de Escobar, G.; Hume, R. Inadequate iodine intake of 40% of pregnant women from a region in Scotland. J. Endocrino. Invest. 2002, 25, 90. [Google Scholar]
- Haddow, J.E.; Palomaki, G.E.; Allan, W.C.; Williams, J.R.; Knight, G.J.; Gagnon, J.; O’Heir, C.E.; Mitchell, M.L.; Hermos, R.J.; Waisbren, S.E.; Faix, J.D.; Klein, R.Z. Maternal thyroid deficiency during pregnancy and subsequent neuropsychological development of the child. New Engl. J. Med. 1999, 341, 549–555. [Google Scholar]
- Bath, S.; Rayman, M.P. Iodine deficiency in UK schoolgirls. Lancet 2011, 5, 1623–1624. [Google Scholar]
- Bath, S.; Wright, J.; Taylor, A.; Walter, A.; Rayman, M.P. Iodine deficiency in pregnant women living in the South-East of the UK. Proc. Nutr. Soc. 2010, 69, E483. [Google Scholar]
- Qian, M.; Wang, D.; Watkins, W.E.; Gebski, V.; Yan, Y.Q.; Li, M.; Chen, Z.P. The effects of iodine on intelligence in children: A meta-analysis of studies conducted in China. Asia Pac. J. Clin. Nutr. 2005, 14, 32–42. [Google Scholar]
- Velasco, I.; Carreira, M.; Santiago, P.; Muela, J.A.; García-Fuentes, E.; Sánchez-Muñoz, B.; Garriga, M.J.; González-Fernández, M.C.; Rodríguez, A.; Caballero, F.F.; Machado, A.; González-Romero, S.; Anarte, M.T.; Soriguer, F. Effect of iodine prophylaxis during pregnancy on neurocognitive development of children during the first two years of life. J. Clin. Endocrinol. Metab. 2009, 94, 3234–3241. [Google Scholar]
- Pharoah, P.O.; Connolly, K.J. Effects of maternal iodine supplementation during pregnancy. Arch. Dis. Child. 1991, 66, 145–147. [Google Scholar] [CrossRef]
- Koletzko, B.; Cetin, I.; Brenna, J.T. Perinatal Lipid Working Group. Dietary fat intakes for pregnant and lactating women. Br. J. Nutr. 2007, 98, 873–877. [Google Scholar]
- Koletzko, B.; Lien, E.; Agostoni, C.; Böhles, H.; Campoy, C.; Cetin, I.; Decsi, T.; Dudenhausen, J.W.; Dupont, C.; Forsyth, S.; et al. The roles of long-chain polyunsaturated fatty acids in pregnancy, lactation and infancy: Review of current knowledge and consensus recommendations. J. Perinat. Med. 2008, 36, 5–14. [Google Scholar] [CrossRef]
- Morse, N.L. A meta-analysis of blood fatty acids in people with learning disorders with particular interest in arachidonic acid. Prostaglandins Leukot. Essent. Fatty Acids 2009, 81, 373–389. [Google Scholar] [CrossRef]
- Holub, B.J. Clinical Nutrition: Omega-3 fatty acids in cardiovascular care. CMAJ 2002, 166, 608–615. [Google Scholar]
- Hollis, B.W.; Wagner, C.L. Vitamin D requirements during lactation: High-dose maternal supplementation as therapy to prevent hypovitaminosis D for both the mother and the nursing infant. Am. J. Clin. Nutr. 2004, 80, 175S–178S. [Google Scholar]
- Bischoff-Ferrari, H.A. Vitamin D—Role in Pregnancy and early childhood. Ann. Nutr. Metab. 2001, 59, 17–21. [Google Scholar] [CrossRef]
- NHS Choices: Vitamins and Minerals—Vitamin D. What Does the Department of Health Recommend? Available online: http://www.nhs.uk/Conditions/vitamins-minerals/Pages/Vitamin-D.aspx (accessed on 3 April 2012).
- Institute of Medicine. Dietary Reference Ranges for Calcium and Vitamin D, 2010. Available online: http://www.iom.edu/Reports/2010/Dietary-Reference-Intakes-for-Calcium-and-vitamin-D.aspx (accessed on 3 April 2012).
- De-Regil, L.M.; Palacios, C.; Ansary, A.; Kulier, R.; Peña-Rosas, J.P. Vitamin D supplementation for women during pregnancy. Cochrane Database Syst. Rev. 2012. Available online: http://onlinelibrary.wiley.com/doi/10.1002/14651858.CD008873.pub2/abstract (accessed on 3 April 2012).
- Hathcock, J.N.; Shao, A.; Vieth, R.; Heaney, R. Risk assessment for vitamin D. Am. J. Clin. Nutr. 2007, 85, 6–18. [Google Scholar]
- Heaney, R.P. Vitamin D: Criteria for safety and efficacy. Nutr. Rev. 2008, 66, S178–S181. [Google Scholar] [CrossRef]
- Vieth, R. Vitamin D supplementation, 25-hydroxyvitamin D concentrations, and safety. Am. J. Clin. Nutr. 1999, 69, 842–856. [Google Scholar]
- Hollis, B.W.; Johnson, D.; Hulsey, T.C.; Ebeling, M.; Wagner, C.L. Vitamin D supplementation during pregnancy: Double-blind, randomized clinical trial of safety and effectiveness. J. Bone Miner. Res. 2011, 26, 2341–2357. [Google Scholar] [CrossRef]
- National Institute for Health and Clinical Excellence. Quick Reference Guide: Antenatal Care, Routine Care for the Healthy Pregnant Woman, March 2008. Available online: http://www.nice.org.uk/nicemedia/pdf/CG062NICEguideline.pdf (accessed on 3 April 2012).
- Dietary Supplements Fact Sheet: Folate. Available online: http://ods.od.nih.gov/factsheets/folate (accessedon 3 April 2012).
- Department of Health and Aging, Australian Government: Australian Guide to Healthy Eating. Available online: http://www.health.gov.au/internet/healthyactive/publishing.nsf/content/pregnant-women (accessed on 3 April 2012).
- EFSA Meeting Summary Report: Folic Acid: An Update on Scientific Developments. Available online: http://www.efsa.europa.eu/en/supporting/doc/2e.pdf (accessed on 3 Apirl 2012).
- Obstetrical and Gynaecological Society of Malaysia. For Patients: The Well Woman—A Guide to Women’s Health. Available online: http://www.ogsm.org.my/planning_for_baby.php (accessed on 3 April 2012).
- Hathcock, J.N. Vitamins and minerals: Efficacy and safety. Am. J. Clin. Nutr. 1997, 66, 427–437. [Google Scholar]
- Herbert, V. Folic Acid. In Nutrition in Health and Disease; Shils, M., Olson, J., Shike, M., Ross, A.C., Eds.; Williams & Wilkins: Baltimore, MD, USA, 1999. [Google Scholar]
- International Council for Control of Iodine Deficiency DisordersUnited Nations Children’s FundWorld Health OrganizationAssessment of Iodine Deficiency Disorders and Monitoring Their Elimination, 2nd edWHO: Geneva, Switzerland, 2007.
- National Health and Medical Research Council and New Zealand Ministry of Health. Nutrient Reference Values for Australia and New Zealand including Recommended Dietary Intakes. Available online: http://www.nhmrc.gov.au (accessed on 3 April 2012).
- Food and Nutrition Board 2002 Dietary reference intakes for Vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium and zinc. National Academy of Sciences: Washington, DC, USA. Available online: http://www.nap.edu/books/0309072794/html/ (accessed on 3 April 2012).
- EU Scientific Committee on Food Safety. Opinion of the Scientific Committee on Food on the Tolerable Upper Intake Level of Iodine. Available online: http://ec.europa.eu/food/fs/sc/scf/out146_en.pdf (accessed on 3 April 2012).
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Morse, N.L. Benefits of Docosahexaenoic Acid, Folic Acid, Vitamin D and Iodine on Foetal and Infant Brain Development and Function Following Maternal Supplementation during Pregnancy and Lactation. Nutrients 2012, 4, 799-840. https://doi.org/10.3390/nu4070799
Morse NL. Benefits of Docosahexaenoic Acid, Folic Acid, Vitamin D and Iodine on Foetal and Infant Brain Development and Function Following Maternal Supplementation during Pregnancy and Lactation. Nutrients. 2012; 4(7):799-840. https://doi.org/10.3390/nu4070799
Chicago/Turabian StyleMorse, Nancy L. 2012. "Benefits of Docosahexaenoic Acid, Folic Acid, Vitamin D and Iodine on Foetal and Infant Brain Development and Function Following Maternal Supplementation during Pregnancy and Lactation" Nutrients 4, no. 7: 799-840. https://doi.org/10.3390/nu4070799
APA StyleMorse, N. L. (2012). Benefits of Docosahexaenoic Acid, Folic Acid, Vitamin D and Iodine on Foetal and Infant Brain Development and Function Following Maternal Supplementation during Pregnancy and Lactation. Nutrients, 4(7), 799-840. https://doi.org/10.3390/nu4070799