Nonclassical Vitamin D Actions
Abstract
1. Introduction
2. Vitamin D Metabolism
| Stage | 25-hydroxyvitamin D (nmol/l) | Clinical/biochemical alterations |
|---|---|---|
| Deficiency | <25 | Rickets, osteomalacia, myopathy, calcium malabsorption, severe hyperparathyroidism, low calcitriol concentrations, impaired immune and cardiac function?, death |
| Insufficiency | 25 to 49. | Reduced bone mineral density, impaired muscle function, low intestinal calcium absorption rates, elevated PTH levels, slightly reduced calcitriol levels |
| Hypovitaminosis D/suboptimal supply | 50 to 74.9 | Low bodily stores of vitamin D, slightly elevated PTH levels |
| Adequacy | 75 to 372 | No disturbances of vitamin D-dependent functions |
| Intoxication | >372 | Intestinal calcium hyperabsorption, hypercalcemia, soft tissue calcification, death |
3. Worldwide Vitamin D Status
4. Diseases Associated with Nonclassical Vitamin D Actions
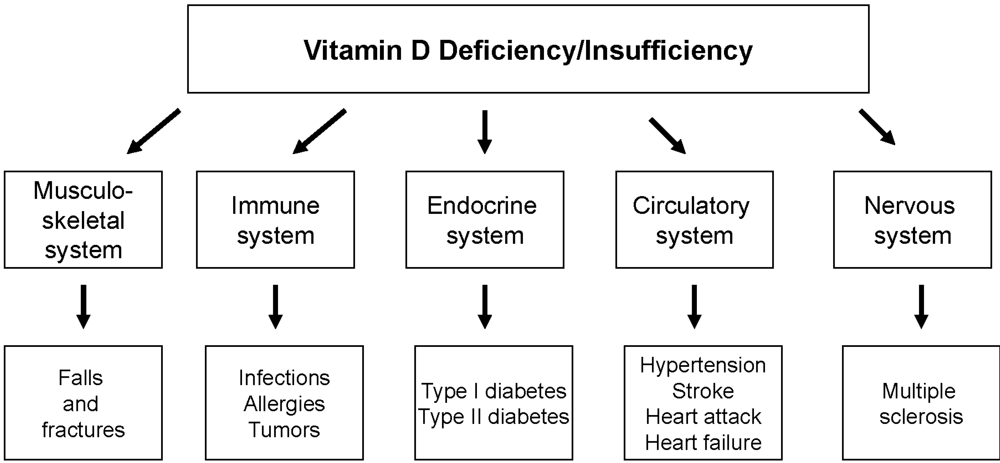
4.1. Vitamin D and Muscle Strengthening
4.2. Infections
4.3. Allergies
4.4. Cancer
4.5. Diabetes Mellitus
4.6. Cardiovascular Disease
| Study | Design | Number of individuals | Comparator | Odds/hazard ratio or Relative risk (95% CI) |
|---|---|---|---|---|
| Fatal stroke | ||||
| Pilz et al. 2009 [63] | Prospective cohort study with coronary angiography | 3258 | Per z value of 25(OH)D | OR 0.58 (0.43 to 0.78) |
| Cardiovascular | ||||
| morbidity | ||||
| Wang et al. 2008 [64] | Prospective observational study | 1739 | 25(OH)D > 37.5 nmol/L versus < 25 nmol/L | HR 0.55 (0.32 to 0.97) |
| Myocardial | ||||
| infarction | ||||
| Giovannucci et al. 2008 [65] | Nested case control study | 1354 | 25(OH)D > 75 nmol/L versus < 37.5 nmol/L | RR 0.48 (0.28 to 0.81) |
| Cardiovascular | ||||
| mortality | ||||
| Dobnig et al. 2008 [66] | Prospective cohort study with coronary angiography | 3258 | Median 25(OH)D 70 nmol/L versus 19 nmol/L | HR 0.45 (0.32 to 0.64) |
| Pilz et al. 2009 [67] | Prospective observational study in individuals 50-75 years | 614 | Three highest versus lowest 25 (OH)D quartile | HR 0.19 (0.07 to 0.50) |
| Ginde et al. 2009 [68] | Prospective observational study in individuals > 65 years. | 3408 | 25(OH)D > 100 nmol/L versus < 25 nmol/L | HR 0.42 (0.21 to 0.86) |
4.7. Multiple Sclerosis
5. Mortality
| Study | Design | Number of individuals | Comparator | Hazard ratio or relative risk (95% CI) |
|---|---|---|---|---|
| Autier and Gandini, 2007 [76] | Meta-analysis of 18 vitamin D supplementation studies | 57,311 | Supplemented versus unsupplemented | RR 0.93 (0.87 to 0.99) |
| Dobnig et al. 2008 [66] | Prospective cohort study with coronary angiography | 3,258 | Median 25(OH)D 70 nmol/L versus 19 nmol/L | HR 0.48 (0.37 to 0.63) |
| Kuroda et al. 2009 [77] | Prospective observational study in postmenopausal women | 1,232 | ≥ 50 nmol/L versus < 50 nmol/L | HR 0.46 (0.27 to 0.79) |
| Ng et al. 2008 [78] | Prospective cohort study in patients with colorectal cancer | 304 | Mean 41 nmol/L versus 100 nmol/L | HR 0.52 (0.29 to 0.94) |
| Ginde et al. 2009 [68] | Prospective observational study in individuals > 65 years. | 3,408 | 25(OH)D > 100 nmol/L versus < 25 nmol/L | HR 0.55 (0.34to 0.88) |
| Pilz et al. 2009 [67] | Prospective observational study In individuals 50-75 years | 614 | Three highest quartiles versus lowest quartile | HR 0.51 (0.28 to 0.93) |
6. Conclusions
References
- Bischoff-Ferrari, H.A.; Willett, W.C.; Wong, J.B.; Stuck, A.E.; Staehelin, H.B.; Orav, E.J.; Thoma, A.; Kiel, D.P.; Henschkowski, J. Prevention of nonvertebral fractures with oral vitamin D and dose dependency: a meta-analysis of randomized controlled trials. Arch. Intern. Med. 2009, 169, 551–561. [Google Scholar]
- McKenna, M.J. Differences in vitamin D status between countries in young adults and the elderly. Am. J. Med. 1992, 93, 69–77. [Google Scholar]
- Zittermann, A. Vitamin D in preventive medicine - are we ignoring the evidence. Br. J. Nutr. 2003, 89, 552–572. [Google Scholar] [CrossRef] [PubMed]
- Vieth, R. Vitamin D and cancer mini-symposium: the risk of additional vitamin D. Ann. Epidemiol. 2009, 19, 441–445. [Google Scholar]
- Patel, M.; Dimaano, R.; Li-Ng, M.; Talwar, S.A.; Mikhail, M.; Pollack, S.; Yeh, J.K. Vitamin D intake to attain a desired serum 25-hydroxyvitamin D concentration. Am. J. Clin. Nutr. 2008, 87, 1952–1958. [Google Scholar]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar]
- Holick, M.F. McCollum Award Lecture, Vitamin D: New horizons for the 21st centur. Am. J. Clin. Nutr. 1994, 60, 619–630. [Google Scholar]
- Clemens, T.L.; Adams, J.S.; Henderson, S.L.; Holick, M.F. Increased skin pigment reduces the capacity of skin to synthesise vitamin D3. Lancet 1982, 1, 74–76. [Google Scholar]
- Krause, R.; Bohring, M.; Hopfenmüller, W.; Holick, M.F.; Sharma, A.M. Ultraviolet B and blood pressure. Lancet 1998, 352, 709–710. [Google Scholar]
- Stamp, T.C.; Haddad, J.G.; Twigg, C.A. Comparison of oral 25-hydroxycholecalciferol, vitamin D, and ultraviolet light as determinants of circulating 25-hydroxyvitamin D. Lancet 1977, 1, 1341–1343. [Google Scholar] [PubMed]
- Zittermann, A.; Frisch, S.; Berthold, H.K.; Götting, C.; Kuhn, J.; Kleesiek, K.; Stehle, P.; Koertke, H.; Koerfer, R. Vitamin D supplementation enhances the beneficial effects of weight loss on cardiovascular risk markers. Am. J. Clin. Nutr. 2009, 89, 1321–1327. [Google Scholar]
- Zittermann, A.; Koerfer, R. Protective and toxic effects of vitamin D on vascular calcification: clinical implications. Mol. Aspects Med. 2008, 29, 423–432. [Google Scholar] [CrossRef] [PubMed]
- Antoniucci, D.M.; Yamashita, T.; Portaloe, A.A. Dietary phosphorus regulates serum fibroblast growth factor-23 concentrations in healthy men. J. Clin. Endocrinol. Metab. 2006, 91, 3144–3149. [Google Scholar]
- Bouillon, R.; Carmeliet, G.; Verlinden, L.; van Etten, E.; Verstuyf, A.; Luderer, H.F.; Lieben, L.; Mathieu, C.; Demay, M. Vitamin D and human health: lessons from vitamin D receptor null mice. Endocr. Rev. 2008, 29, 726–776. [Google Scholar] [CrossRef] [PubMed]
- Hewison, M.; Burke, F.; Evans, K.N.; Lammas, D.A. Extra-renal 25-hydroxyvitamin D3-1alpha-hydroxylase in human health and disease. J. Steroid Biochem. Mol. Biol. 2007, 103, 316–321. [Google Scholar]
- Zittermann, A.; Tenderich, G.; Koerfer, R. Vitamin D and the adaptive immune system with special emphasis to allergic reactions and allograft rejection. Inflamm. Allergy Drug Targets 2009, 8, 161–168. [Google Scholar]
- Gallieni, M.; Kamimura, S.; Ahmed, A.; Bravo, E.; Delmez, J.; Slatopolsky, E.; Dusso, A. Kinetics of monocyte 1 alpha-hydroxylase in renal failure. Am. J. Physiol. 1995, 268, F746–F753. [Google Scholar]
- Zittermann, A.; Gummert, J.F.; Boergermann, J. Vitamin D deficiency and mortality. Curr. Opin. Nutr. Metab. Care 2009, 2, 634–639. [Google Scholar]
- Mithal, A.; Wahl, D.A.; Bonjour, J.P.; Burckhardt, P.; Dawson-Hughes, B.; Eisman, J.A.; El-Hajj Fuleihan, G.; Josse, R.G.; Lips, P.; Morales-Torres, J. IOF Committee of Scientific Advisors. (CSA) Nutrition Working Group. Global vitamin D status and determinants of hypovitaminosis D. Osteoporos Int. 2009, 20, 1807–1820. [Google Scholar] [CrossRef] [PubMed]
- Moreira-Pfrimer, L.D.; Pedrosa, M.A.; Teixeira, L.; Lazaretti-Castro, M. Treatment of vitamin D deficiency increases lower limb muscle strength in institutionalized older people independently of regular physical activity: a randomized double-blind controlled trial. Ann. Nutr. Metab. 2009, 54, 291–300. [Google Scholar]
- Bischoff-Ferrari, H.A.; Dawson-Hughes, B.; Staehelin, H.B.; Orav, J.E.; Stuck, A.E.; Theiler, R.; Wong, J.B.; Egli, A.; Kiel, D.P.; Henschkowski, J. Fall prevention with supplemental and active forms of vitamin D: a meta-analysis of randomised controlled trials. BMJ. 2009, 339, b3692. Oct 1. [Google Scholar] [PubMed]
- Ronge, H.E. Increase of physical effectiveness by systematic ultraviolet radiation. Strahlentherapie 1952, 88, 563–566, [article in German]. [Google Scholar] [PubMed]
- Schauber, J.; Dorschner, R.A.; Coda, A.B.; Büchau, A.S.; Liu, P.T.; Kiken, D.; Helfrich, Y.R.; Kang, S.; Elalieh, H.Z.; Steinmeyer, A.; Zügel, U.; Bikle, D.D.; Modlin, R.L.; Gallo, R.L. Injury enhances TLR2 function and antimicrobial peptide expression through a vitamin D-dependent mechanism. J. Clin. Invest. 2007, 117, 803–811. [Google Scholar]
- Nnoaham, K.E.; Clarke, A. Low serum vitamin D levels and tuberculosis: a systematic review and meta-analysis. Int. J. Epidemiol. 2008, 37, 113–119. [Google Scholar]
- Cannell, J.J.; Vieth, R.; Umhau, J.C.; Holick, M.F.; Grant, W.B.; Madronich, S.; Garland, C.F.; Giovannucci, E. Epidemic influenza and vitamin D. Epidemiol. Infect. 2006, 134, 1129–1140. [Google Scholar]
- Urashima, M.; Segawa, T.; Okazaki, M.; Kurihara, M.; Wada, Y.; Ida, H. Randomized trial of vitamin D supplementation to prevent seasonal influenza A in schoolchildren. Am. J. Clin. Nutr. 2010. Epub ahead of print.
- Laaksi, I.; Ruohola, J.P.; Tuohimaa, P.; Auvinen, A.; Haataja, R.; Pihlajamäki, H.; Ylikomi, T. An association of serum vitamin D concentrations < 40 nmol/L with acute respiratory tract infection in young Finnish men. Am. J. Clin. Nutr. 2007, 86, 714–717. [Google Scholar]
- Ginde, A.A.; Mansbach, J.M.; Camargo, C.A. Association between serum 25-hydroxyvitamin D level and upper respiratory tract infection in the Third National Health and Nutrition Examination Survey. Arch. Intern. Med. 2009, 169, 384–390. [Google Scholar]
- Avenell, A.; Cook, J.A.; Maclennan, G.S.; Macpherson, G.C. Vitamin D supplementation to prevent infections: a sub-study of a randomised placebo-controlled trial in older people (RECORD trial, ISRCTN 51647438). Age Ageing 2007, 36, 574–577. [Google Scholar]
- Aloia, J.F.; Talwar, S.A.; Pollack, S.; Yeh, J. A randomized controlled trial of vitamin D3 supplementation in African American women. Arch. Intern. Med. 2005, 165, 1618–1623. [Google Scholar]
- Li-Ng, M.; Aloia, J.F.; Pollack, S.; Cunha, B.A.; Mikhail, M.; Yeh, J.; Berbari, N. A randomized controlled trial of vitamin D3 supplementation for the prevention of symptomatic upper respiratory tract infections. Epidemiol. Infect. 2009, 137, 1396–1404. [Google Scholar]
- Yamshchikov, A.V.; Desai, N.S.; Blumberg, H.M.; Ziegler, T.R.; Tangpricha, V. Vitamin D for treatment and prevention of infectious diseases: a systematic review of randomized controlled trials. Endocr. Pract. 2009, 15, 438–449. [Google Scholar]
- Misawa, Y.; Baba, A.; Ito, S.; Tanaka, M.; Shiohara, M. Vitamin D(3) induces expression of human cathelicidin antimicrobial peptide 18 in newborns. Int. J. Hematol. 2009. Nov 28. [Epub ahead of print]. [Google Scholar]
- Wjst, M. Another explanation for the low allergy rate in the rural Alpine foothills. Clin. Mol. Allergy 2005, 3, 7. [Google Scholar]
- Von Mutius, E. Influences in allergy: epidemiology and the environment. J. Allergy Clin. Immunol. 2004, 113, 373–379. [Google Scholar]
- Gale, C.R.; Robinson, S.M.; Harvey, N.C.; Javaid, M.K.; Jiang, B.; Martyn, C.N.; Godfrey, K.M.; Cooper, C. the Princess Anne Hospital Study Group. Maternal vitamin D status during pregnancy and childhood outcomes. Eur. J. Clin. Nutr 2008, 62, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Hyppönen, E.; Sovio, U.; Wjst, M.; Patel, S.; Pekkanen, J.; Hartikainen, A.L.; Järvelinb, M.R. Infant vitamin d supplementation and allergic conditions in adulthood: northern Finland birth cohort 1966. Ann. N. Y. Acad. Sci. 2004, 1037, 84–95. [Google Scholar]
- Bäck, O.; Blomquist, H.K.; Hernell, O.; Stenberg, B. Does vitamin D intake during infancy promote the development of atopic allergy. Acta Derm. Venereol. 2009, 89, 28–32. [Google Scholar] [PubMed]
- Wjst, M.; Hyppönen, E. Vitamin D serum levels and allergic rhinitis. Allergy 2007, 62, 1085–1086. [Google Scholar]
- Zittermann, A.; Dembinski, J.; Stehle, P. Low vitamin D status is associated with low cord blood levels of the immunosuppressive cytokine interleukin 10. Pediatr. Allergy Immu. 2004, 15, 242–246. [Google Scholar]
- Camargo, C.A.; Rifas-Shiman, S.L.; Litonjua, A.A.; Rich-Edwards, J.W.; Weiss, S.T.; Gold, D.R.; Kleinman, K.; Gillman, M.W. Maternal intake of vitamin D during pregnancy and risk of recurrent wheeze in children at 3 y of age. Am. J. Clin. Nutr. 2007, 85, 788–795. [Google Scholar]
- Camargo, C.A.; Clark, S.; Kaplan, M.S.; Lieberman, P.; Wood, R.A. Regional differences in EpiPen prescriptions in the United States: the potential role of vitamin D. J. Allergy Clin. Immunol. 2007, 120, 131–136. [Google Scholar]
- Oren, E.; Banerji, A.; Camergo, C.A. Vitamin D and atopic disoerdes in an obese population screened for vitamin D deficiency. J. Allergy Immunol. 2008, 121, 533–534. [Google Scholar]
- Erkkola, M.; Kaila, M.; Nwaru, B.I.; Kronberg-Kippilä, C.; Ahonen, S.; Nevalainen, J.; Veijola, R.; Pekkanen, J.; Ilonen, J.; Simell, O.; Knip, M.; Virtanen, S.M. Maternal vitamin D intake during pregnancy is inversely associated with asthma and allergic rhinitis in 5-year-old children. Clin. Exp. Allergy 2009, 39, 875–882. [Google Scholar]
- Sidbury, R.; Sullivan, A.F.; Thadhani, R.I.; Camargo, C.A. Randomized trial of vitamin D supplementation for winter-related atopic dermatitis in Boston: a pilot study. Br. J. Dermatol. 2008, 159, 245–247. [Google Scholar]
- Schleithoff, S.S.; Zittermann, A.; Tenderich, G.; Berthold, H.K.; Stehle, P.; Koerfer, R. Vitamin D Supplementation Improves Cytokine Profile In Patients With Congestive Heart Failure: A Double-Blind, Randomized, Placebo-Controlled Trial. Am. J. Clin. Nutr. 2006, 83, 754–759. [Google Scholar] [PubMed]
- Hyppönen, E.; Berry, D.J.; Wjst, M.; Power, C. Serum 25-hydroxyvitamin D and IgE - a significant but nonlinear relationship. Allergy 2009, 64, 613–620. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization, International Agency for Research on Cancer. Vitamin D and cancer. IARC Working Group Reports; WHO: Geneva, Switzerland, 2008; 5, p. 148.
- Lappe, J.M.; Travers-Gustafson, D.; Davies, K.M.; Recker, R.R.; Heaney, R.P. Vitamin D and calcium supplementation reduces cancer risk: results of a randomized trial. Am. J. Clin. Nutr. 2007, 85, 1586–1591. [Google Scholar] [PubMed]
- Gorham, E.D.; Mohr, S.B.; Garland, F.C.; Garland, C.F. Vitamin D for Cancer Prevention and Survival. Clin. Rev. Bone Miner Metab. 2009, 7, 159–175. [Google Scholar]
- Garland, C.F.; Gorham, E.D.; Mohr, S.B.; Garland, F.C. Vitamin D for cancer prevention: global perspective. Ann. Epidemiol. 2009, 19, 468–483. [Google Scholar]
- Tuohimaa, P.; Tenkanen, L.; Ahonen, M.; Lumme, S.; Jellum, E.; Hallmans, G.; Stattin, P.; Harvei, S.; Hakulinen, T.; Luostarinen, T.; Dillner, J.; Lehtinen, M.; Hakama, M. Both high and low levels of blood vitamin D are associated with a higher prostate cancer risk: a longitudinal, nested case-control study in the Nordic countries. Int. J. Cancer 2004, 108, 104–108. [Google Scholar] [CrossRef] [PubMed]
- Zittermann, A.; Grant, W.B. 25-hydroxyvitamin D levels and all-cause mortality. Arch. Intern. Med. 2009, 169, 1075–1076. [Google Scholar]
- Mohr, S.B.; Garland, C.F.; Gorham, E.D.; Garland, F.C. The association between ultraviolet B irradiance, vitamin D status and incidence rates of type 1 diabetes in 51 regions worldwid. Diabetologia 2008, 51, 1391–1398. [Google Scholar]
- Hyppönen, E.; Läärä, E.; Reunanen, A.; Järvelin, M.R.; Virtanen, S.M. Intake of vitamin D and risk of type 1 diabetes: a birth-cohort study. Lancet 2001, 358, 1500–1503. [Google Scholar]
- Zipitis, C.S.; Akobeng, A.K. Vitamin D supplementation in early childhood and risk of type 1 diabetes: a systematic review and meta-analysis. Arch. Dis. Child. 2008, 93, 512–517. [Google Scholar]
- Li, X.; Liao, L.; Yan, X.; Huang, G.; Lin, J.; Lei, M.; Wang, X.; Zhou, Z. Protective effects of 1-alpha-hydroxyvitamin D3 on residual beta-cell function in patients with adult-onset latent autoimmune diabetes (LADA). Diabetes Metab. Res. Rev. 2009, 25, 411–416. [Google Scholar]
- Pittas, A.G.; Lau, J.; Hu, F.B.; Dawson-Hughes, B. The role of vitamin D and calcium in type 2 diabetes. A systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 2007, 92, 2017–2029. [Google Scholar] [CrossRef] [PubMed]
- Jorde R, Figenschau, Y. Supplementation with cholecalciferol does not improve glycemic control in diabtetic subjects with normal serum 25-hydroxyvitamin D levels. Eur. J. Nutr. 2009, 48, 349–354. [Google Scholar] [CrossRef] [PubMed]
- von Hurst, P.R.; Stonehouse, W.; Coad, J. Vitamin D supplementation reduces insulin resistance in South Asian women living in New Zealand who are insulin resistant and vitamin D deficient - a randomised, placebo-controlled trial. Br. J. Nutr. 2009, 1–7, [Epub ahead of print]. [Google Scholar] [PubMed][Green Version]
- Zittermann, A. Koerfer R.Vitamin D in the prevention and treatment of coronary heart disease. Curr. Opin. Clin. Nutr. Metab. Care. 2008, 11, 752–757. [Google Scholar] [CrossRef] [PubMed]
- Witham, M.D.; Nadir, M.A.; Struthers, A.D. Effect of vitamin D on blood pressure: a systematic review and meta-analysis. J. Hypertens. 2009, 27, 1948–1954. [Google Scholar]
- Pilz, S.; Dobnig, H.; Fischer, J.E.; Wellnitz, B.; Seelhorst, U.; Boehm, B.O.; März, W. Low vitamin D levels predict stroke in patients referred to coronary angiography. Stroke 2008, 39, 2611–2613. [Google Scholar]
- Wang, T.J.; Pencina, M.J.; Booth, S.L.; Jacques, P.F.; Ingelsson, E.; Lanier, K.; Benjamin, E.J.; D'Agostino, R.B.; Wolf, M.; Vasan, R.S. Vitamin D deficiency and risk of cardiovascular disease. Circulation 2008, 117, 503–511. [Google Scholar]
- Giovannucci, E.; Liu, Y.; Hollis, B.W.; Rimm, E.B. 25-hydroxyvitamin D and risk of myocardial infarction in men: a prospective study. Arch. Intern. Med. 2008, 168, 1174–1180. [Google Scholar]
- Dobnig, H.; Pilz, S.; Scharnagl, H.; Renner, W.; Seelhorst, U.; Wellnitz, B.; Kinkeldei, J.; Boehm, B.O.; Weihrauch, G.; Maerz, W. Independent association of low serum 25-hydroxyvitamin D and 1,25-dihydroxyvitamin D levels with all-cause and cardiovascular mortality. Arch. Intern. Med. 2008, 168, 1340–1349. [Google Scholar]
- Pilz, S.; Dobnig, H.; Nijpels, G.; Heine, R.J.; Stehouwer, C.D.; Snijder, M.B.; van Dam, R.M.; Dekker, J.M. Vitamin D and mortality in older men and women. Clin. Endocrinol. 2009, 71, 666–672. [Google Scholar]
- Ginde, A.A.; Scragg, R.; Schwartz, R.; Camargo, C.A. Prospective Study of Serum 25-Hydroxyvitamin D Level, Cardiovascular Disease Mortality, and All-Cause Mortality in Older U.S. Adults. J. Am. Geriatr. Soc. 2009, 57, 1595–1603. [Google Scholar] [CrossRef] [PubMed]
- LaCroix, A.Z.; Kotchen, J.; Anderson, G.; Brzyski, R.; Cauley, J.A.; Cummings, S.R.; Gass, M.; Johnson, K.C.; Ko, M.; Larson, J.; Manson, J.E.; Stefanick, M.L.; Wactawski-Wende, J. Calcium plus vitamin D supplementation and mortality in postmenopausal women: the Women's Health Initiative calcium-vitamin D randomized controlled trial. J. Gerontol. A Biol. Sci. Med. Sci. 2009, 64, 559–567. [Google Scholar]
- Wang, L.; Manson, J.E.; Song, Y.; Sesso, H.D. Systematic review: Vitamin D and calcium supplementation in prevention of cardiovascular events. Ann. ntern. Med. 2010, 152, 315–323. [Google Scholar]
- Zittermann, A.; Frisch, S.; Berthold, H.K.; Götting, C.; Kuhn, J.; Kleesiek, K. ; Stehle, P.; Koertke, H; Koerfer, R. Vitamin D supplementation enhances the beneficial effects of weight loss on cardiovascular risk markers. Am. J. Clin. Nutr. 2009, 89, 1321–1327. [Google Scholar] [CrossRef] [PubMed]
- Chaudhuri, A. Why we should offer routine vitamin D supplementation in pregnancy and childhood to prevent multiple sclerosis. Med. Hypotheses. 2005, 64, 608–618. [Google Scholar]
- Embry, A.F.; Snowdon, L.R.; Vieth, R. Vitamin D and seasonal fluctuations of gadolinium-enhancing magnetic resonance imaging lesions in multiple sclerosis. Ann. Neurol. 2000, 48, 271–272. [Google Scholar]
- Munger, K.L.; Levin, L.I.; Hollis, B.W.; Howard, N.S.; Ascherio, A. Serum 25-hydroxyvitamin D levels and risk of multiple sclerosis. JAMA 2006, 296, 2832–2838. [Google Scholar]
- Ramagopalan, S.V.; Maugeri, N.J.; Handunnetthi, L.; Lincoln, M.R.; Orton, S.M.; Dyment, D.A.; Deluca, G.C.; Herrera, B.M.; Chao, M.J.; Sadovnick, A.D.; Ebers, G.C.; Knight, J.C. Expression of the multiple sclerosis-associated MHC class II Allele HLA-DRB1*1501 is regulated by vitamin D. PLoS Genet. 2009, 5, e1000369. [Google Scholar]
- Autier, P.; Gandini, S. Vitamin D supplementation and total mortality: a meta-analysis of randomized controlled trials. Arch. Intern. Med. 2007, 167, 1730–1737. [Google Scholar]
- Kuroda, T.; Shiraki, M.; Tanaka, S.; Ohta, H. Contributions of 25-hydroxyvitamin D, co-morbidities and bone mass to mortality in Japanese postmenopausal women. Bone 2009, 44, 168–172. [Google Scholar]
- Ng, K.; Meyerhardt, J.A.; Wu, K.; Feskanich, D.; Hollis, B.W.; Giovannucci, E.L.; Fuchs, C.S. Circulating 25-hydroxyvitamin d levels and survival in patients with colorectal cancer. J. Clin. Oncol. 2008, 26, 2984–2991. [Google Scholar]
- Wang, A.Y.; Lam, C.W.; Sanderson, J.E.; Wang, M.; Chan, I.H.; Lui, S.F.; Sea, M.M.; Woo, J. Serum 25-hydroxyvitamin D status and cardiovascular outcomes in chronic peritoneal dialysis patients: a 3-y prospective cohort study. Am. J. Clin. Nutr. 2008, 87, 1631–1638. [Google Scholar] [PubMed]
- Heist, R.S.; Zhou, W.; Wang, Z. Liu, G.; Neuberg, D.; Su, L.; Asomaning, K.; Hollis, B.W.; Lynch, T.J.; Wain, J.C.; Giovannucci, E.; Christiani, D.C. Circulating 25-hydroxyvitamin D, VDR polymorphisms, and survival in advanced non-small-cell lung cancer. J. Clin. Oncol. 2008, 26, 5596–5602. [Google Scholar] [PubMed]
- Jorde, R.; Sneve, M.; Torjesen, P.; Figenschau, Y. No improvement in cardiovascular risk factors in overweight and obese subjects after supplementation with vitamin D for 1 year. J. Intern. Med. 2009. [epub ahead of print]. [Google Scholar]
- Holmes, V.A.; Barnes, M.S.; Alexander, H.D.; McFaul, P.; Wallace, J.M. Vitamin D deficiency and insufficiency in pregnant women: a longitudinal study. Br. J. Nutr. 2009, 102, 876–881. [Google Scholar] [CrossRef] [PubMed]
- Standing Committee of European Doctors. Vitamin D nutritional policy in Europe. Available online: http://cpme.dyndns.org:591/adopted/2009/CPME_AD_Brd_241009_179_final_EN.pdf (assessed on 24 March 2010).
- Zittermann, A.; Koerfer, R. Protective and toxic effects of vitamin D on vascular calcification: Clinical implications. Mol. Aspects Med. 2008, 29, 423–432. [Google Scholar] [CrossRef] [PubMed]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zittermann, A.; Gummert, J.F. Nonclassical Vitamin D Actions. Nutrients 2010, 2, 408-425. https://doi.org/10.3390/nu2040408
Zittermann A, Gummert JF. Nonclassical Vitamin D Actions. Nutrients. 2010; 2(4):408-425. https://doi.org/10.3390/nu2040408
Chicago/Turabian StyleZittermann, Armin, and Jan F. Gummert. 2010. "Nonclassical Vitamin D Actions" Nutrients 2, no. 4: 408-425. https://doi.org/10.3390/nu2040408
APA StyleZittermann, A., & Gummert, J. F. (2010). Nonclassical Vitamin D Actions. Nutrients, 2(4), 408-425. https://doi.org/10.3390/nu2040408




