Vitamin D status during Pregnancy and Aspects of Offspring Health
Abstract
:1. Review
2. Synthesis and Action of 1,25(OH)2D
2.1. Non-calcitropic Actions
3. Physiology
4. The Prevalence of Low Vitamin D Status during Pregnancy
| Low ambient UVR level | High latitude location |
| Winter season | |
| Inadequate sun exposure | Built or indoor environment |
| Extensive clothing cover (e.g., veiled) | |
| Excess sun avoidance – (shade, sunscreen) | |
| Host factors | Dark skin pigmentation |
| Old age | |
| Obesity | |
| Malabsorptive syndromes (e.g., inflammatory bowel disease) |
| Reference | Year | Country | Population | Stage of Pregnancy | Definition | % n/N |
|---|---|---|---|---|---|---|
| [127] | 1997 | Iran | Iran women attending largest Tehran hospital | Delivery | <25 nmol/L | 80% (40/50) |
| [128] | 1997-2001 | USA | African American–90% receiving prenatal vitamins | 4-21 week gestation | <37.5 nmol/L | 44.9% (89/194) |
| [128] | 1997-2001 | USA | White–90% receiving prenatal vitamins | 4-21 week gestation | <37.5 nmol/L | 2% (4/199) |
| [128] | 1997-2001 | USA | African–American | 37-42 weeks | <37.5 nmol/L | 29.2% (54/185) |
| [128] | 1997-2001 | USA | White | 37-42 weeks | <37.5 nmol/L | 5% (10/199) |
| [128] | 1997-2001 | USA | Turkish | 12 week gestation | <25 nmol/L | 83.5% (66/79) |
| [128] | 1997-2001 | USA | Moroccan | 12 week gestation | <25 nmol/L | 81.2% (56/69) |
| [128] | 1997-2001 | USA | Other non-western | 12 week gestation | <25 nmol/L | 59% (62/105) |
| [129] | 1999 | Ireland | Caucasians in Ulster–on supplements (54–55 oN) | 12 weeks | <25 nmol/L | 4.5% (1/22) |
| [129] | 1999 | Ireland | No supplements | 12 weeks | <25 nmol/L | 44.2% (34/77) |
| [129] | 1999 | Ireland | Supplements | 20 weeks | <25 nmol/L | 22.7% (5/22) |
| [129] | 1999 | Ireland | No supplements | 20 weeks | <25 nmol/L | 50.6% (39/77) |
| [129] | 1999 | Ireland | Supplements | 35 weeks | <25 nmol/L | 0% (0/22) |
| [129] | 1999 | Ireland | No supplements | 35 weeks | <25 nmol/L | 20.8% (16/77) |
| [32] | 1999-2000 | Australia | Veiled &/or dark skinned women attending antenatal clinic | During antenatal care, when a routine blood test was ordered | <22.5 nmol/L | 80.5% (66/82) |
| [130] | 1999-2000 | United Arab Emirates | Kuwait | Delivery | <25 nmol/L | 40% (86/214) |
| [131] | 2002 | India | Attendees of Queen Mary’s Hosptal, Lucknow (26.8 oN) Northern India – urban | Delivery–full term, live | <22.5 nmol/L | 84.3% (118/140) |
| [131] | 2002 | India | Attendees of Queen Mary’s Hosptal, Lucknow (26.8 oN) Northern India–urban | Rural | <56.3 nmol/L | 84%(56/67) |
| [132] | 2005-2006 | India | Residents of Barabanki district, 26.8 oN | Second trimester | <50 nmol/L | 74.1% (103/139) |
4.1. Vitamin D Supplementation
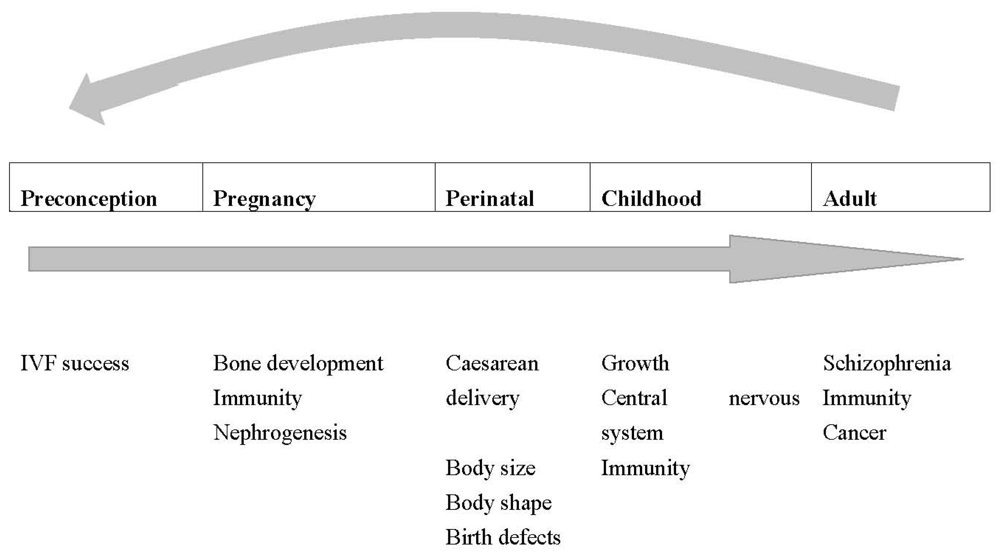
5. Vitamin D Status during Pregnancy and the Effects on Offspring Health over the Life Course
5.1. Fetal Life
5.1.1. Conception
5.1.2. Immunity
5.1.3. Bone formation
5.2. Perinatal Period
5.2.1. Immunomodulation
5.2.2. Mode of Birth
5.2.3. Birth Weight
5.2.4. Birth Defects
5.3. Childhood and Adulthood
5.3.1. Bone Health
5.3.2. Postnatal Infection
5.3.3. Immune diseases
5.3.4. Central nervous system disorders
5.3.5. Other candidate diseases that require further research with regard to the potential role of prenatal vitamin D status.
6. Overview
7. Future Directions
Acknowledgements
References and Notes
- Holick, M.F.; MacLaughlin, J.A.; Clark, M.B.; Holick, S.A.; Potts, J.T., Jr.; Anderson, R.R.; Blank, I.H.; Parrish, J.A.; Elias, P. Photosynthesis of previtamin D3 in human skin and the physiologic consequences. Science 1980, 210, 203–235. [Google Scholar]
- Nowson, C.A.; Margerison, C. Vitamin D intake and vitamin D status of Australians. Med. J. Aust. 2002, 177, 149–152. [Google Scholar]
- Holick, M.F. Sunlight and vitamin D for bone health and prevention of autoimmune diseases, cancers, and cardiovascular disease. Am. J. Clin. Nutr. 2004, 80, 1678S–1688S. [Google Scholar] [PubMed]
- Holick, M.F. Vitamin D status: measurement, interpretation, and clinical application. Ann. Epidemiol. 2009, 19, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Bikle, D. Nonclassic actions of vitamin D. J. Clin. Endocrinol. Metab. 2009, 94, 26–34. [Google Scholar]
- Holick, M.F. McCollum Award Lecture, 1994: vitamin D--new horizons for the 21st century. Am. J. Clin. Nutr. 1994, 60, 619–630. [Google Scholar]
- DeLuca, H.F.; Zierold, C. Mechanisms and functions of vitamin D. Nutr. Rev. 1998, 56 discussion S 54-75, S4–S10. [Google Scholar] [CrossRef] [PubMed]
- Maalouf, N.M. Mechanisms and functions of vitamin D. Curr. Opin. Nephrol. Hypertens. 2008, 17, 408–415. [Google Scholar]
- Chiu, K.C.; Chu, A.; Go, V.L.; Saad, M.F. Hypovitaminosis D is associated with insulin resistance and beta cell dysfunction. Am. J. Clin. Nutr. 2004, 79, 820–825. [Google Scholar]
- Smith, M.A.; McHenry, C.; Oslapas, R.; Hofmann, C.; Hessel, P.; Paloyan, E. Altered TSH levels associated with increased serum 1,25-dihydroxyvitamin D3: a possible link between thyroid and parathyroid disease. Surgery 1989, 106, 987–991. [Google Scholar]
- Achinger, S.G.; Ayus, J.C. The role of vitamin D in left ventricular hypertrophy and cardiac function. Kidney Int. Suppl. 2005, S37–S42. [Google Scholar]
- Szodoray, P.; Nakken, B.; Gaal, J.; Jonsson, R.; Szegedi, A.; Zold, E.; Szegedi, G.; Brun, J.G.; Gesztelyi, R.; Zeher, M.; Bodolay, E. The complex role of vitamin D in autoimmune diseases. Scand. J. Immunol. 2008, 68, 261–269. [Google Scholar]
- Zasloff, M. Fighting infections with vitamin D. Nat. Med. 2006, 12, 388–390. [Google Scholar]
- Liu, N.; Kaplan, A.T.; Low, J.; Nguyen, L.; Liu, G.Y.; Equils, O.; Hewison, M. Vitamin D induces innate antibacterial responses in human trophoblasts via an intracrine pathway. Biol. Reprod. 2009, 80, 398–406. [Google Scholar]
- Deluca, H.F.; Cantorna, M.T. Vitamin D: its role and uses in immunology. Faseb. J. 2001, 15, 2579–2585. [Google Scholar]
- Abe, E.; Miyaura, C.; Sakagami, H.; Takeda, M.; Konno, K.; Yamazaki, T.; Yoshiki, S.; Suda, T. Differentiation of mouse myeloid leukemia cells induced by 1 alpha,25-dihydroxyvitamin D3. Proc. Natl. Acad. Sci. USA 1981, 78, 4990–4994. [Google Scholar]
- Eisman, J.A.; Koga, M.; Sutherland, R.L.; Barkla, D.H.; Tutton, P.J. Dihydroxyvitamin D3 and the regulation of human cancer cell replication. Proc. Soc. Exp. Biol. Med. 1989, 191, 221–226. [Google Scholar]
- Specker, B. Vitamin D requirements during pregnancy. Am. J. Clin. Nutr. 2004, 80, 1740S–1747S. [Google Scholar]
- Perez-Lopez, F.R. Vitamin D: the secosteroid hormone and human reproduction. Gynecol. Endocrinol. 2007, 23, 13–24. [Google Scholar]
- Salle, B.L.; Delvin, E.E.; Lapillonne, A.; Bishop, N.J.; Glorieux, F.H. Perinatal metabolism of vitamin D. Am. J. Clin. Nutr. 2000, 71, 1317S–1324S. [Google Scholar] [PubMed]
- Abrams, S.A. In utero physiology: role in nutrient delivery and fetal development for calcium, phosphorus, and vitamin D. Am. J. Clin. Nutr. 2007, 85, 604S–607S. [Google Scholar] [PubMed]
- Mulligan, M.L.; Felton, S.K.; Riek, A.E.; Bernal-Mizrachi, C. Implications of vitamin D deficiency in pregnancy and lactation. Am. J. Obstet. Gynecol. 2009. epub ahead of print. [Google Scholar]
- Ritchie, L.D.; Fung, E.B.; Halloran, B.P.; Turnlund, J.R.; van Loan, M.D.; Cann, C.E.; King, J.C. A longitudinal study of calcium homeostasis during human pregnancy and lactation and after resumption of menses. Am. J. Clin. Nutr. 1998, 67, 693–701. [Google Scholar]
- Maghbooli, Z.; Hossein-Nezhad, A.; Shafaei, A.R.; Karimi, F.; Madani, F.S.; Larijani, B. Vitamin D status in mothers and their newborns in Iran. BMC Pregnancy Childbirth 2007, 7, 1. [Google Scholar]
- Markestad, T.; Aksnes, L.; Ulstein, M.; Aarskog, D. 25-Hydroxyvitamin D and 1,25-dihydroxyvitamin D of D2 and D3 origin in maternal and umbilical cord serum after vitamin D2 supplementation in human pregnancy. Am. J. Clin. Nutr. 1984, 40, 1057–1063. [Google Scholar] [PubMed]
- Zehnder, D.; Evans, K.N.; Kilby, M.D.; Bulmer, J.N.; Innes, B.A.; Stewart, P.M.; Hewison, M. The ontogeny of 25-hydroxyvitamin D(3) 1alpha-hydroxylase expression in human placenta and decidua. Am. J. Pathol. 2002, 161, 105–114. [Google Scholar]
- Evans, K.N.; Bulmer, J.N.; Kilby, M.D.; Hewison, M. Vitamin D and placental-decidual function. J. Soc. Gynecol. Investig. 2004, 11, 263–271. [Google Scholar]
- Holick, M.F. Environmental factors that influence the cutaneous production of vitamin D. Am. J. Clin. Nutr. 1995, 61, 638S–645S. [Google Scholar]
- Matsuoka, L.Y.; Wortsman, J.; Haddad, J.G.; Hollis, B.W. Skin types and epidermal photosynthesis of vitamin D3. J. Am. Acad. Dermatol. 1990, 23, 525–526. [Google Scholar]
- Matsuoka, L.Y.; Wortsman, J.; Hanifan, N.; Holick, M.F. Chronic sunscreen use decreases circulating concentrations of 25-hydroxyvitamin D. A preliminary study. Arch. Dermatol. 1988, 124, 1802–1804. [Google Scholar] [PubMed]
- van der Meer, I.M.; Karamali, N.S.; Boeke, A.J.; Lips, P.; Middelkoop, B.J.; Verhoeven, I.; Wuister, J.D. High prevalence of vitamin D deficiency in pregnant non-Western women in The Hague, Netherlands. Am. J. Clin. Nutr. 2006, 84, 350–353. [Google Scholar]
- Grover, S.R.; Morley, R. Vitamin D deficiency in veiled or dark-skinned pregnant women. Med. J. Aust. 2001, 175, 251–252. [Google Scholar]
- Hollis, B.W.; Wagner, C.L. Assessment of dietary vitamin D requirements during pregnancy and lactation. Am. J. Clin. Nutr. 2004, 79, 717–726. [Google Scholar]
- Bischoff-Ferrari, H.A.; Kiel, D.P.; Dawson-Hughes, B.; Orav, J.E.; Li, R.; Spiegelman, D.; Dietrich, T.; Willett, W.C. Dietary calcium and serum 25-hydroxyvitamin D status in relation to BMD among U.S. adults. J. Bone Miner Res. 2009, 24, 935–942. [Google Scholar] [CrossRef] [PubMed]
- Lapillonne, A. Vitamin D deficiency during pregnancy may impair maternal and fetal outcomes. Med. Hypotheses 2009, 74, 71–75. [Google Scholar]
- Farrant, H.J.; Krishnaveni, G.V.; Hill, J.C.; Boucher, B.J.; Fisher, D.J.; Noonan, K.; Osmond, C.; Veena, S.R.; Fall, C.H. Vitamin D insufficiency is common in Indian mothers but is not associated with gestational diabetes or variation in newborn size. Eur. J. Clin. Nutr. 2009, 63, 646–652. [Google Scholar]
- Mallet, E.; Gugi, B.; Brunelle, P.; Henocq, A.; Basuyau, J.P.; Lemeur, H. Vitamin D supplementation in pregnancy: a controlled trial of two methods. Obstet. Gynecol. 1986, 68, 300–304. [Google Scholar]
- Datta, S.; Alfaham, M.; Davies, D.P.; Dunstan, F.; Woodhead, S.; Evans, J.; Richards, B. Vitamin D deficiency in pregnant women from a non-European ethnic minority population--an interventional study. BJOG 2002, 109, 905–908. [Google Scholar] [Green Version]
- Hollis, B.W.; Wagner, C.L. Vitamin D deficiency during pregnancy: an ongoing epidemic. Am. J. Clin. Nutr. 2006, 84, 273. [Google Scholar] [Green Version]
- Hollis, B.W.; Wagner, C.L. Nutritional vitamin D status during pregnancy: reasons for concern. CMAJ 2006, 174, 1287–1290. [Google Scholar] [Green Version]
- Gillie, O. Vitamin D ‘may cut premature birth risk and protect newborn babies’. The Times. 10 Oct 2009. Available online: http://www.timesonline.co.uk/tol/news/uk/scotland/article6868729.ece (accessed on 2 February 2010).
- Vieth, R. Vitamin D supplementation, 25-hydroxyvitamin D concentrations, and safety. Am. J. Clin. Nutr. 1999, 69, 842–856. [Google Scholar] [PubMed][Green Version]
- Vieth, R.; Chan, P.C.; MacFarlane, G.D. Efficacy and safety of vitamin D3 intake exceeding the lowest observed adverse effect level. Am. J. Clin. Nutr. 2001, 73, 288–294. [Google Scholar] [Green Version]
- Vieth, R. Vitamin D toxicity, policy, and science. J. Bone Miner Res. 2007, 22, V64–V68. [Google Scholar] [Green Version]
- Effectiveness and Safety of vitamin D in relation to bone health. In Report no. 07-E013; Agency for Healthcare Research and Quality: Maryland, USA, 2007; pp. 119–142.
- Halloran, B.P.; DeLuca, H.F. Effect of vitamin D deficiency on fertility and reproductive capacity in the female rat. J. Nutr. 1980, 110, 1573–1580. [Google Scholar] [Green Version]
- Kinuta, K.; Tanaka, H.; Moriwake, T.; Aya, K.; Kato, S.; Seino, Y. Vitamin D is an important factor in estrogen biosynthesis of both female and male gonads. Endocrinology 2000, 141, 1317–1324. [Google Scholar] [Green Version]
- Johnson, L.E.; DeLuca, H.F. Vitamin D receptor null mutant mice fed high levels of calcium are fertile. J. Nutr. 2001, 131, 1787–1791. [Google Scholar] [Green Version]
- Ozkan, S.; Jindal, S.; Greenseid, K.; Shu, J.; Zeitlian, G.; Hickmon, C.; Pal, L. Replete vitamin D stores predict reproductive success following in vitro fertilization. Fertil Steril 2009. epub ahead of print. [Google Scholar]
- Evans, K. N.; Bulmer, J.N.; Kilby, M.D.; Hewison, M. Vitamin D and placental-decidual function. J. Soc. Gynecol. Investig. 2004, 11, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Novakovic, B.; Sibson, M.; Ng, H.K.; Manuelpillai, U.; Rakyan, V.; Down, T.; Beck, S.; Fournier, T.; Evain-Brion, D.; Dimitriadis, E.; Craig, J.M.; Morley, R.; Saffery, R. Placenta-specific methylation of the vitamin D 24-hydroxylase gene: implications for feedback autoregulation of active vitamin D levels at the fetomaternal interface. J. Biol. Chem. 2009, 284, 14838–14848. [Google Scholar]
- Correale, J.; Ysrraelit, M.C.; Gaitan, M.I. Immunomodulatory effects of Vitamin D in multiple sclerosis. Brain 2009, 132, 1146–1160. [Google Scholar]
- Smith, S.E.; Li, J.; Garbett, K.; Mirnics, K.; Patterson, P.H. Maternal immune activation alters fetal brain development through interleukin-6. J. Neurosci. 2007, 27, 10695–10702. [Google Scholar]
- Kovacs, C.S. Vitamin D in pregnancy and lactation: maternal, fetal, and neonatal outcomes from human and animal studies. Am. J. Clin. Nutr. 2008, 88, 520S–528S. [Google Scholar] [PubMed]
- 22nd Marabou Symposium: the changing faces of vitamin D. Nutr. Rev. 2008, 66, S195–212. [CrossRef] [PubMed]
- Mahon, P.; Harvey, N.; Crozier, S.; Inskip, H.; Robinson, S.; Arden, N.; Swaminathan, R.; Cooper, C.; Godfrey, K. Low Maternal Vitamin D Status and Fetal Bone Development. J. Bone Miner Res. 2009, 25, 14–19. [Google Scholar]
- Zittermann, A.; Dembinski, J.; Stehle, P. Low vitamin D status is associated with low cord blood levels of the immunosuppressive cytokine interleukin-10. Pediatr. Allergy Immunol. 2004, 15, 242–246. [Google Scholar]
- Pichler, J.; Gerstmayr, M.; Szepfalusi, Z.; Urbanek, R.; Peterlik, M.; Willheim, M. 1 alpha,25(OH)2D3 inhibits not only Th1 but also Th2 differentiation in human cord blood T cells. Pediatr. Res. 2002, 52, 12–18. [Google Scholar]
- Merewood, A.; Mehta, S.D.; Chen, T.C.; Bauchner, H.; Holick, M.F. Association between vitamin D deficiency and primary cesarean section. J. Clin. Endocrinol. Metab. 2009, 94, 940–945. [Google Scholar]
- Morley, R.; Carlin, J.B.; Pasco, J.A.; Wark, J.D. Maternal 25-hydroxyvitamin D and parathyroid hormone concentrations and offspring birth size. J. Clin. Endocrinol. Metab. 2006, 91, 906–912. [Google Scholar]
- Mannion, C.A.; Gray-Donald, K.; Koski, K.G. Association of low intake of milk and vitamin D during pregnancy with decreased birth weight. CMAJ 2006, 174, 1273–1277. [Google Scholar]
- Gale, C.R.; Robinson, S.M.; Harvey, N.C.; Javaid, M.K.; Jiang, B.; Martyn, C.N.; Godfrey, K.M.; Cooper, C. Maternal vitamin D status during pregnancy and child outcomes. Eur. J. Clin. Nutr. 2008, 62, 68–77. [Google Scholar]
- Scholl, T.O.; Chen, X. Vitamin D intake during pregnancy: association with maternal characteristics and infant birth weight. Early Hum. Dev. 2009, 85, 231–234. [Google Scholar]
- Weiler, H.; Fitzpatrick-Wong, S.; Veitch, R.; Kovacs, H.; Schellenberg, J.; McCloy, U.; Yuen, C.K. Vitamin D deficiency and whole-body and femur bone mass relative to weight in healthy newborns. CMAJ 2005, 172, 757–761. [Google Scholar]
- Zmuda, J.M.; Cauley, J.A.; Ferrell, R.E. Molecular epidemiology of vitamin D receptor gene variants. Epidemiol. Rev. 2000, 22, 203–217. [Google Scholar]
- Morley, R.; Carlin, J.B.; Pasco, J.A.; Wark, J.D.; Ponsonby, A.L. Maternal 25-hydroxyvitamin D concentration and offspring birth size: effect modification by infant VDR genotype. Eur. J. Clin. Nutr. 2009, 63, 802–804. [Google Scholar]
- Hollis, B.W. Randomized controlled trials to determine the safety of vitamin D supplementation during pregnancy and lactation (Session XV Skin and Reproduction). In 14th Workshop on Vitamin D; Brugge, Belgium, 2009. [Google Scholar]
- Maka, N.; Makrakis, J.; Parkington, H.C.; Tare, M.; Morley, R.; Black, M.J. Vitamin D deficiency during pregnancy and lactation stimulates nephrogenesis in rat offspring. Pediatr. Nephrol. 2008, 23, 55–61. [Google Scholar]
- Morris, G.S.; Zhou, Q.; Hegsted, M.; Keenan, M.J. Maternal consumption of a low vitamin D diet retards metabolic and contractile development in the neonatal rat heart. J. Mol. Cell Cardiol. 1995, 27, 1245–1250. [Google Scholar]
- Eyles, D.; Brown, J.; Mackay-Sim, A.; McGrath, J.; Feron, F. Vitamin D3 and brain development. Neuroscience 2003, 118, 641–653. [Google Scholar]
- Ko, P.; Burkert, R.; McGrath, J.; Eyles, D. Maternal vitamin D3 deprivation and the regulation of apoptosis and cell cycle during rat brain development. Brain Res. Dev. Brain Res. 2004, 153, 61–68. [Google Scholar]
- Feron, F.; Burne, T.H.; Brown, J.; Smith, E.; McGrath, J.J.; Mackay-Sim, A.; Eyles, D.W. Developmental Vitamin D3 deficiency alters the adult rat brain. Brain Res. Bull. 2005, 65, 141–148. [Google Scholar]
- Norman, P.; Moss, I.; Sian, M.; Gosling, M.; Powell, J. Maternal and postnatal vitamin D ingestion influences rat aortic structure, function and elastin content. Cardiovasc. Res. 2002, 55, 369–374. [Google Scholar]
- Toda, T.; Ito, M.; Toda, Y.; Smith, T.; Kummerow, F. Angiotoxicity in swine of a moderate excess of dietary vitamin D3. Food Chem. Toxicol. 1985, 23, 585–592. [Google Scholar]
- Toda, T.; Toda, Y.; Kummerow, F.A. Coronary arterial lesions in piglets from sows fed moderate excesses of vitamin D. Tohoku J. Exp. Med. 1985, 145, 303–310. [Google Scholar]
- Pasco, J.A.; Wark, J.D.; Carlin, J.B.; Ponsonby, A.L.; Vuillermin, P.J.; Morley, R. Maternal vitamin D in pregnancy may influence not only offspring bone mass but other aspects of musculoskeletal health and adiposity. Med. Hypotheses 2008, 71, 266–269. [Google Scholar]
- Cooper, C.; Javaid, K.; Westlake, S.; Harvey, N.; Dennison, E. Developmental origins of osteoporotic fracture: the role of maternal vitamin D insufficiency. J. Nutr. 2005, 135, 2728S–2734S. [Google Scholar]
- Javaid, M.K.; Crozier, S.R.; Harvey, N.C.; Gale, C.R.; Dennison, E.M.; Boucher, B.J.; Arden, N.K.; Godfrey, K.M.; Cooper, C. Maternal vitamin D status during pregnancy and childhood bone mass at age 9 years: a longitudinal study. Lancet 2006, 367, 36–43. [Google Scholar]
- Karatekin, G.; Kaya, A.; Salihoglu, O.; Balci, H.; Nuhoglu, A. Association of subclinical vitamin D deficiency in newborns with acute lower respiratory infection and their mothers. Eur. J. Clin. Nutr. 2009, 63, 473–477. [Google Scholar]
- Roth, D.E.; Jones, A.B.; Prosser, C.; Robinson, J.L.; Vohra, S. Vitamin D status is not associated with the risk of hospitalization for acute bronchiolitis in early childhood. Eur. J. Clin. Nutr. 2009, 63, 297–299. [Google Scholar]
- Mehta, S.; Hunter, D.J.; Mugusi, F.M.; Spiegelman, D.; Manji, K.P.; Giovannucci, E.L.; Hertzmark, E.; Msamanga, G.I.; Fawzi, W.W. Perinatal Outcomes, Including Mother-to-Child Transmission of HIV, and Child Mortality and Their Association with Maternal Vitamin D Status in Tanzania. J. Infect. Dis. 2009, 200, 1022–1030. [Google Scholar] [PubMed]
- van Ranst, M.; Joossens, M.; Joossens, S.; van Steen, K.; Pierik, M.; Vermeire, S.; Rutgeerts, P. Crohn's disease and month of birth. Inflamm Bowel. Dis. 2005, 11, 597–599. [Google Scholar]
- Rothwell, P.M.; Gutnikov, S.A.; McKinney, P.A.; Schober, E.; Ionescu-Tirgoviste, C.; Neu, A. Seasonality of birth in children with diabetes in Europe: multicentre cohort study. European Diabetes Study Group. BMJ 1999, 319, 887–888. [Google Scholar] [PubMed]
- Willis, J.A.; Scott, R.S.; Darlow, B.A.; Lewy, H.; Ashkenazi, I.; Laron, Z. Seasonality of birth and onset of clinical disease in children and adolescents (0-19 years) with type 1 diabetes mellitus in Canterbury, New Zealand. J. Pediatr. Endocrinol. Metab. 2002, 15, 645–647. [Google Scholar]
- Samuelsson, U.; Johansson, C.; Ludvigsson, J. Month of birth and risk of developing insulin dependent diabetes in south east Sweden. Arch. Dis. Child. 1999, 81, 143–146. [Google Scholar]
- Laron, Z.; Lewy, H.; Wilderman, I.; Casu, A.; Willis, J.; Redondo, M. J.; Libman, I.; White, N.; Craig, M. Seasonality of month of birth of children and adolescents with type 1 diabetes mellitus in homogenous and heterogeneous populations. Isr. Med. Assoc. J. 2005, 7, 381–384. [Google Scholar]
- Badenhoop, K.; Kahles, H.; Seidl, C.; Kordonouri, O.; Lopez, E.R.; Walter, M.; Rosinger, S.; Ziegler, A.; Bohm, B.O. MHC-environment interactions leading to type 1 diabetes: feasibility of an analysis of HLA DR-DQ alleles in relation to manifestation periods and dates of birth. Diabetes Obes. Metab. 2009, 11, 88–91. [Google Scholar]
- Fronczak, C.M.; Baron, A.E.; Chase, H.P.; Ross, C.; Brady, H.L.; Hoffman, M.; Eisenbarth, G.S.; Rewers, M.; Norris, J.M. In utero dietary exposures and risk of islet autoimmunity in children. Diabetes Care 2003, 26, 3237–3242. [Google Scholar]
- Stene, L.C.; Ulriksen, J.; Magnus, P.; Joner, G. Use of cod liver oil during pregnancy associated with lower risk of Type I diabetes in the offspring. Diabetologia 2000, 43, 1093–1098. [Google Scholar]
- Hypponen, E.; Laara, E.; Reunanen, A.; Jarvelin, M.R.; Virtanen, S.M. Intake of vitamin D and risk of type 1 diabetes: a birth-cohort study. Lancet 2001, 358, 1500–1503. [Google Scholar]
- Zipitis, C.S.; Akobeng, A.K. Vitamin D supplementation in early childhood and risk of type 1 diabetes: a systematic review and meta-analysis. Arch. Dis. Child. 2008, 93, 512–517. [Google Scholar]
- Devereux, G.; Litonjua, A.A.; Turner, S.W.; Craig, L.C.; McNeill, G.; Martindale, S.; Helms, P.J.; Seaton, A.; Weiss, S. T. Maternal vitamin D intake during pregnancy and early childhood wheezing. Am. J. Clin. Nutr. 2007, 85, 853–859. [Google Scholar]
- Camargo, C.A., Jr.; Rifas-Shiman, S.L.; Litonjua, A.A.; Rich-Edwards, J.W.; Weiss, S.T.; Gold, D.R.; Kleinman, K.; Gillman, M.W. Maternal intake of vitamin D during pregnancy and risk of recurrent wheeze in children at 3 y of age. Am. J. Clin. Nutr. 2007, 85, 788–795. [Google Scholar]
- Staples, J.A.; Ponsonby, A.L.; Lim, L.L.; McMichael, A.J. Ecologic analysis of some immune-related disorders, including type 1 diabetes, in Australia: latitude, regional ultraviolet radiation, and disease prevalence. Environ. Health Perspect. 2003, 111, 518–523. [Google Scholar] [PubMed]
- Wjst, M.; Dold, S. Genes, factor X, and allergens: what causes allergic diseases? Allergy 1999, 54, 757–759. [Google Scholar] [CrossRef] [PubMed]
- Zanolin, M.E.; Pattaro, C.; Corsico, A.; Bugiani, M.; Carrozzi, L.; Casali, L.; Dallari, R.; Ferrari, M.; Marinoni, A.; Migliore, E.; Olivieri, M.; Pirina, P.; Verlato, G.; Villani, S.; Marco, R. The role of climate on the geographic variability of asthma, allergic rhinitis and respiratory symptoms: results from the Italian study of asthma in young adults. Allergy 2004, 59, 306–314. [Google Scholar]
- Sole, D.; Wandalsen, G.F.; Camelo-Nunes, I.C.; Naspitz, C.K. Prevalence of symptoms of asthma, rhinitis, and atopic eczema among Brazilian children and adolescents identified by the International Study of Asthma and Allergies in Childhood (ISAAC) - Phase 3. Pediatr. (Rio J.) 2006, 82, 341–346. [Google Scholar]
- Wion, D.; MacGrogan, D.; Neveu, I.; Jehan, F.; Houlgatte, R.; Brachet, P. 1,25-Dihydroxyvitamin D3 is a potent inducer of nerve growth factor synthesis. J. Neurosci. Res. 1991, 28, 110–114. [Google Scholar]
- Almeras, L.; Eyles, D.; Benech, P.; Laffite, D.; Villard, C.; Patatian, A.; Boucraut, J.; Mackay-Sim, A.; McGrath, J.; Feron, F. Developmental vitamin D deficiency alters brain protein expression in the adult rat: implications for neuropsychiatric disorders. Proteomics 2007, 7, 769–780. [Google Scholar]
- Eyles, D.W.; Smith, S.; Kinobe, R.; Hewison, M.; McGrath, J.J. Distribution of the vitamin D receptor and 1 alpha-hydroxylase in human brain. J. Chem. Neuroanat. 2005, 29, 21–30. [Google Scholar]
- McGrath, J.J.; Welham, J.L. Season of birth and schizophrenia: a systematic review and meta-analysis of data from the Southern Hemisphere. Schizophr. Res. 1999, 35, 237–242. [Google Scholar]
- Torrey, E.F.; Miller, J.; Rawlings, R.; Yolken, R.H. Seasonality of births in schizophrenia and bipolar disorder: a review of the literature. Schizophr. Res. 1997, 28, 1–38. [Google Scholar]
- Bhugra, D.; Leff, J.; Mallett, R.; Der, G.; Corridan, B.; Rudge, S. Incidence and outcome of schizophrenia in whites, African-Caribbeans and Asians in London. Psychol. Med. 1997, 27, 791–798. [Google Scholar]
- McGrath, J.; Selten, J.P.; Chant, D. Long-term trends in sunshine duration and its association with schizophrenia birth rates and age at first registration--data from Australia and the Netherlands. Schizophr. Res. 2002, 54, 199–212. [Google Scholar]
- Becker, A.; Eyles, D.W.; McGrath, J.J.; Grecksch, G. Transient prenatal vitamin D deficiency is associated with subtle alterations in learning and memory functions in adult rats. Behav. Brain Res. 2005, 161, 306–312. [Google Scholar]
- McGrath, J.; Saari, K.; Hakko, H.; Jokelainen, J.; Jones, P.; Jarvelin, M. R.; Chant, D.; Isohanni, M. Vitamin D supplementation during the first year of life and risk of schizophrenia: a Finnish birth cohort study. Schizophr. Res. 2004, 67, 237–245. [Google Scholar]
- van der Mei, I.A.; Ponsonby, A.L.; Dwyer, T.; Blizzard, L.; Simmons, R.; Taylor, B.V.; Butzkueven, H.; Kilpatrick, T. Past exposure to sun, skin phenotype, and risk of multiple sclerosis: case-control study. BMJ 2003, 327, 316. [Google Scholar] [PubMed]
- Munger, K.L.; Levin, L.I.; Hollis, B.W.; Howard, N.S.; Ascherio, A. Serum 25-hydroxyvitamin D levels and risk of multiple sclerosis. JAMA 2006, 296, 2832–2838. [Google Scholar]
- Willer, C.J.; Dyment, D.A.; Sadovnick, A.D.; Rothwell, P.M.; Murray, T.J.; Ebers, G.C. Timing of birth and risk of multiple sclerosis: population based study. BMJ 2005, 330, 120. [Google Scholar]
- Staples, J.; Ponsonby, A.-L.; Lim, L. Prenatal ultraviolet radiation exposure, month of birth and subsequent risk of multiple sclerosis: a longitudinal analysis. BMJ 2010. In press.
- Ebers, G.C.; Sadovnick, A.D.; Dyment, D.A.; Yee, I.M.; Willer, C.J.; Risch, N. Parent-of-origin effect in multiple sclerosis: observations in half-siblings. Lancet 2004, 363, 1773–1774. [Google Scholar]
- Hoppenbrouwers, I.A.; Liu, F.; Aulchenko, Y.S.; Ebers, G.C.; Oostra, B.A.; van Duijn, C.M.; Hintzen, R.Q. Maternal transmission of multiple sclerosis in a Dutch population. Arch. Neurol. 2008, 65, 345–348. [Google Scholar]
- Sayers, A.; Tilling, K.; Boucher, B.J.; Noonan, K.; Tobias, J.H. Predicting ambient ultraviolet from routine meteorological data; its potential use as an instrumental variable for vitamin D status in pregnancy in a longitudinal birth cohort in the UK. Int. J. Epidemiol. 2009, 38, 1681–1688. [Google Scholar] [CrossRef] [PubMed]
- Brenner, A.V.; Linet, M.S.; Shapiro, W.R.; Selker, R.G.; Fine, H.A.; Black, P.M.; Inskip, P.D. Season of birth and risk of brain tumors in adults. Neurology 2004, 63, 276–281. [Google Scholar]
- Ko, P.; Eyles, D.; Burne, T.; Mackay-Sim, A.; McGrath, J.J. Season of birth and risk of brain tumors in adults. Neurology 2005, 64, 1317. [Google Scholar]
- Procopio, M.; Marriott, P.K. Seasonality of birth in epilepsy: a Danish study. Acta Neurol. Scand. 1998, 98, 297–301. [Google Scholar]
- Newschaffer, C.J.; Croen, L.A.; Daniels, J.; Giarelli, E.; Grether, J.K.; Levy, S.E.; Mandell, D.S.; Miller, L.A.; Pinto-Martin, J.; Reaven, J.; Reynolds, A.M.; Rice, C.E.; Schendel, D.; Windham, G.C. The epidemiology of autism spectrum disorders. Annu. Rev. Public Health 2007, 28, 235–258. [Google Scholar]
- Stevens, M.C.; Fein, D.H.; Waterhouse, L.H. Season of birth effects in autism. J. Clin. Exp. Neuropsychol. 2000, 22, 399–407. [Google Scholar]
- Levitan, R.D.; Masellis, M.; Lam, R.W.; Kaplan, A.S.; Davis, C.; Tharmalingam, S.; Mackenzie, B.; Basile, V.S.; Kennedy, J.L. A birth-season/DRD4 gene interaction predicts weight gain and obesity in women with seasonal affective disorder: A seasonal thrifty phenotype hypothesis. Neuropsychopharmacology 2006, 31, 2498–2503. [Google Scholar] [CrossRef] [PubMed]
- Phillips, D.I.; Young, J.B. Birth weight, climate at birth and the risk of obesity in adult life. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 281–287. [Google Scholar]
- Li, H.; Stampfer, M.J.; Hollis, J.B.; Mucci, L.A.; Gaziano, J.M.; Hunter, D.; Giovannucci, E.L.; Ma, J. A prospective study of plasma vitamin D metabolites, vitamin D receptor polymorphisms, and prostate cancer. PLoS Med. 2007, 4, e103. [Google Scholar]
- Giovannucci, E. The epidemiology of vitamin D and cancer incidence and mortality: a review. Cancer Causes Control 2005, 16, 83–95. [Google Scholar]
- Konety, B.R.; Nangia, A.K.; Nguyen, T.S.; Thomas, A.; Getzenberg, R.H. Effect of prenatal vitamin D (calcitriol) exposure on the growth and development of the prostate. Prostate 1999, 41, 181–189. [Google Scholar]
- Mahomed, K.; Gulmezoglu, A.M. Vitamin D supplementation in pregnancy. Cochrane Database Syst. Rev. 2000, CD000228. [Google Scholar]
- De Stavola, B.L.; Nitsch, D.; dos Santos Silva, I.; McCormack, V.; Hardy, R.; Mann, V.; Cole, T.J.; Morton, S.; Leon, D.A. Statistical issues in life course epidemiology. Am. J. Epidemiol. 2006, 163, 84–96. [Google Scholar]
- Eyles, D.; Anderson, C.; Ko, P.; Jones, A.; Thomas, A.; Burne, T.; Mortensen, P.B.; Norgaard-Pedersen, B.; Hougaard, D.M.; McGrath, J. A sensitive LC/MS/MS assay of 25OH vitamin D3 and 25OH vitamin D2 in dried blood spots. Clin. Chim. Acta 2009, 403, 145–151. [Google Scholar]
- Bassir, M.; Laborie, S.; Lapillonne, A.; Claris, O.; Chappuis, M.C.; Salle, B.L. Vitamin D deficiency in Iranian mothers and their neonates: a pilot study. Acta Paediatr. 2001, 90, 577–579. [Google Scholar]
- Bodnar, L.M.; Simhan, H.N.; Powers, R.W.; Frank, M.P.; Cooperstein, E.; Roberts, J.M. High prevalence of vitamin D insufficiency in black and white pregnant women residing in the northern United States and their neonates. J. Nutr. 2007, 137, 447–452. [Google Scholar]
- Holmes, V.A.; Barnes, M.S.; Alexander, H.D.; McFaul, P.; Wallace, J.M. Vitamin D deficiency and insufficiency in pregnant women: a longitudinal study. Br. J. Nutr. 2009, 102, 876–881. [Google Scholar]
- Molla, A.M.; Al Badawi, M.; Hammoud, M.S.; Molla, A.M.; Shukkur, M.; Thalib, L.; Eliwa, M.S. Vitamin D status of mothers and their neonates in Kuwait. Pediatr. Int. 2005, 47, 649–652. [Google Scholar]
- Sachan, A.; Gupta, R.; Das, V.; Agarwal, A.; Awasthi, P.K.; Bhatia, V. High prevalence of vitamin D deficiency among pregnant women and their newborns in northern India. Am. J. Clin. Nutr. 2005, 81, 1060–1064. [Google Scholar]
- Sahu, M.; Bhatia, V.; Aggarwal, A.; Rawat, V.; Saxena, P.; Pandey, A.; Das, V. Vitamin D deficiency in rural girls and pregnant women despite abundant sunshine in northern India. Clin. Endocrinol. (Oxf) 2009, 70, 680–684. [Google Scholar] [CrossRef] [PubMed]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ponsonby, A.-L.; Lucas, R.M.; Lewis, S.; Halliday, J. Vitamin D status during Pregnancy and Aspects of Offspring Health. Nutrients 2010, 2, 389-407. https://doi.org/10.3390/nu2030389
Ponsonby A-L, Lucas RM, Lewis S, Halliday J. Vitamin D status during Pregnancy and Aspects of Offspring Health. Nutrients. 2010; 2(3):389-407. https://doi.org/10.3390/nu2030389
Chicago/Turabian StylePonsonby, Anne-Louise, Robyn M. Lucas, Sharon Lewis, and Jane Halliday. 2010. "Vitamin D status during Pregnancy and Aspects of Offspring Health" Nutrients 2, no. 3: 389-407. https://doi.org/10.3390/nu2030389
APA StylePonsonby, A.-L., Lucas, R. M., Lewis, S., & Halliday, J. (2010). Vitamin D status during Pregnancy and Aspects of Offspring Health. Nutrients, 2(3), 389-407. https://doi.org/10.3390/nu2030389





