Vegan and Plant-Based Diets in the Management of Metabolic Syndrome: A Narrative Review from Anti-Inflammatory and Antithrombotic Perspectives
Highlights
- Metabolic syndrome (MetS) is not only a metabolic disorder but also a driver of thrombosis and inflammation, increasing the risk of CVD and T2DM.
- Vegan diets may play a role in reducing the burden of MetS by improving insulin sensitivity, lipid profiles, blood pressure, and inflammatory markers.
- Additional benefits on the gut microbiota and endothelial health further enhance their therapeutic potential.
Abstract
1. Introduction
2. Metabolic Syndrome
3. The Effect of Type 2 Diabetes and Hyperglycemia on Thrombosis and Inflammation
4. The Effect of Obesity on Thrombosis and Inflammation
5. The Effect of Hypertension on Thrombosis and Inflammation
6. Dietary Interventions: Plant-Based and Vegan Diets in MetS Management
7. The Effect of a Vegan Diet on Gut Microbiota and Metabolic Syndrome
8. Current Research Limitations and Future Directions
9. Discussion
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| MetS | Metabolic syndrome |
| CVDs | Cardiovascular diseases |
| CRP | C-reactive protein |
| hs-CRP | High-sensitivity C-reactive protein |
| IR | Insulin resistance |
| MPV | Mean platelet volume |
| FBS | Fasting blood glucose |
| vWF | von Willebrand factor |
| T2DM | Type 2 diabetes |
| PAI-1 | Plasminogen activator inhibitor-1 |
| ICAM-1 | Intracellular adhesion molecule 1 |
| VCAM-1 | Vascular cell adhesion molecule |
| IL-1RA | IL-1 receptor antagonist |
| IAPP | Islet amyloid polypeptide |
| BMI | Body mass index |
| MCP-1 | Protein monocyte chemokine-1 |
| ROS | Reactive oxygen species |
| PP2A | Protein phosphatase 2A |
| Treg | Regulatory T cells |
| PKC | Protein kinase C |
| AGEs | Advanced glycation end products |
| TLR4 | Toll-like receptor 4 |
| LPS | Lipopolysaccharide |
| HIF-1 | Hypoxia-inducible factor 1 |
| tPA | Tissue plasminogen activator |
| uPA | Urokinase-type plasminogen activator |
| GLUT1 | Glucose transporter 1 |
| Th2 | T helper type 2 |
| PGI2 | Prostacyclin |
| PI3K | Phosphatidylinositol 3-kinase |
| MAPK | Mitogen-activated protein kinase |
| IRS | Insulin receptor substrates |
| BH4 | Tetrahydrobiopterin |
| DAG | Diacylglycerol |
| RBP4 | Retinol binding protein 4 |
| NF-Κb | Nuclear factor kappa B |
| TF | Tissue factor |
| AT1 | Angiotensin type 1 receptor |
| Ang | Angiotensin |
| IKK | IκB kinase |
| JNK | c-Jun N-terminal kinase |
| GP | Glycoprotein |
| MC4R | Melanocortin 4 receptor |
| TCF7L2 | Transcription factor 7-like 2 |
| ADIPOQ | Adiponectin gene |
| GWAS | Genome-wide association studies |
| FBG | Fasting blood glucose |
| MCP1 | Monocyte chemokine 1 |
| DDAH | Dimethylarginine dimethylaminohydrolase |
| ADMA | Asymmetric dimethylarginine |
| NET | Neutrophil extracellular trap |
| SOD | Superoxide dismutase |
| CAT | Catalase |
| GPx | Peroxidase |
| ET-1 | Endothelin-1 |
| PTP1B | Protein-tyrosine phosphatase 1B |
| TFPI | Tissue factor pathway inhibitor |
| LDL-C | Low-density lipoprotein cholesterol |
| VEGF | Vascular endothelial growth factor |
| PDGF-β | Platelet-derived growth factor-β |
| PUFAs | Poly unsaturated fatty acids |
| SCFA | Short-chain fatty acid |
| TMAO | Trimethylamine-N-oxide |
| ALA | α-linolenic acid |
| CAP1 | Cyclase-associated protein 1 |
| HMGB1 | High-mobility group box 1 protein |
References
- Masenga, S.K.; Kabwe, L.S.; Chakulya, M.; Kirabo, A. Mechanisms of oxidative stress in metabolic syndrome. Int. J. Mol. Sci. 2023, 24, 7898. [Google Scholar] [CrossRef]
- Samson, S.L.; Garber, A.J. Metabolic syndrome. Endocrinol. Metab. Clin. N. Am. 2014, 43, 1–23. [Google Scholar] [CrossRef]
- Cho, Y.; Lee, S.Y. Useful Biomarkers of Metabolic Syndrome. Int. J. Environ. Res. Public Health 2022, 19, 15003. [Google Scholar] [CrossRef]
- Kg, A. Metabolic syndrome-a new world-wide definition. A consensus statement from the International Diabetes Federation. Diabet Med. 2006, 23, 469–480. [Google Scholar]
- Lemieux, I.; Després, J.P. Metabolic Syndrome: Past, Present and Future. Nutrients 2020, 12, 3501. [Google Scholar] [CrossRef]
- Huang, P.L. A comprehensive definition for metabolic syndrome. Dis. Model. Mech. 2009, 2, 231–237. [Google Scholar] [CrossRef]
- Sharebiani, H.; Keramat, S.; Chavoshan, A.; Fazeli, B.; Stanek, A. The influence of antioxidants on oxidative stress-induced vascular aging in obesity. Antioxidants 2023, 12, 1295. [Google Scholar] [CrossRef]
- Keramat, S.; Sharebiani, H.; Patel, M.; Fazeli, B.; Stanek, A. The Potential Role of Antioxidants in the Treatment of Peripheral Arterial Disease: A Systematic Review. Antioxidants 2022, 11, 2126. [Google Scholar] [CrossRef]
- Rohla, M.; Weiss, T.W. Metabolic syndrome, inflammation and atherothrombosis. Hamostaseologie 2013, 33, 283–294. [Google Scholar] [CrossRef]
- Wu, Y.; Dong, Y.; Duan, S.; Zhu, D.; Deng, L. Metabolic Syndrome, Inflammation, and Cancer. Mediators Inflamm. 2017, 2017, 8259356. [Google Scholar] [CrossRef]
- Grandl, G.; Wolfrum, C. Hemostasis, endothelial stress, inflammation, and the metabolic syndrome. Semin. Immunopathol. 2018, 40, 215–224. [Google Scholar] [CrossRef]
- Dentali, F.; Romualdi, E.; Ageno, W. The metabolic syndrome and the risk of thrombosis. Haematologica 2007, 92, 297–299. [Google Scholar] [CrossRef]
- Alessi, M.C.; Juhan-Vague, I. Metabolic syndrome, haemostasis and thrombosis. Thromb. Haemost. 2008, 99, 995–1000. [Google Scholar] [CrossRef] [PubMed]
- Franchini, M.; Targher, G.; Montagnana, M.; Lippi, G. The metabolic syndrome and the risk of arterial and venous thrombosis. Thromb. Res. 2008, 122, 727–735. [Google Scholar] [CrossRef]
- Samad, F.; Ruf, W. Inflammation, obesity, and thrombosis. Blood 2013, 122, 3415–3422. [Google Scholar] [CrossRef] [PubMed]
- Stewart, L.K.; Kline, J.A. Metabolic syndrome increases risk of venous thromboembolism recurrence after acute deep vein thrombosis. Blood Adv. 2020, 4, 127–135. [Google Scholar] [CrossRef]
- Satija, A.; Hu, F.B. Plant-based diets and cardiovascular health. Trends Cardiovasc. Med. 2018, 28, 437–441. [Google Scholar] [CrossRef]
- Pieters, M.; Swanepoel, A.C. The effect of plant-based diets on thrombotic risk factors. Pol. Arch. Intern. Med. 2021, 131, 16123. [Google Scholar] [CrossRef] [PubMed]
- Zuelch, M.L. The Power of Plants: The Role of Phytonutrients in Vascular Health. UC Davis Electron. Theses Diss. 2023, 1–404. [Google Scholar]
- Li, D. Chemistry behind Vegetarianism. J. Agric. Food Chem. 2011, 59, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Satija, A.; Bhupathiraju, S.N.; Rimm, E.B.; Spiegelman, D.; Chiuve, S.E.; Borgi, L.; Willett, W.C.; Manson, J.E.; Sun, Q.; Hu, F.B. Plant-based dietary patterns and incidence of type 2 diabetes in US men and women: Results from three prospective cohort studies. PLoS Med. 2016, 13, e1002039. [Google Scholar] [CrossRef] [PubMed]
- De Biase, S.G.; Fernandes, S.F.C.; Gianini, R.J.; Duarte, J.L.G. Vegetarian diet and cholesterol and triglycerides levels. Arq. Bras. Cardiol. 2007, 88, 35–39. [Google Scholar] [CrossRef]
- Moore, E.; Patanwala, I.; Jafari, A.; Davies, I.G.; Kirwan, R.P.; Newson, L.; Mazidi, M.; Lane, K.E. A systematic review and meta-analysis of randomized controlled trials to evaluate plant-based omega-3 polyunsaturated fatty acids in nonalcoholic fatty liver disease patient biomarkers and parameters. Nutr. Rev. 2024, 82, 143–165. [Google Scholar] [CrossRef]
- Ilari, S.; Proietti, S.; Milani, F.; Vitiello, L.; Muscoli, C.; Russo, P.; Bonassi, S. Dietary patterns, oxidative stress, and early inflammation: A systematic review and meta-analysis comparing mediterranean, vegan, and vegetarian diets. Nutrients 2025, 17, 548. [Google Scholar] [CrossRef]
- Marrone, G.; Guerriero, C.; Palazzetti, D.; Lido, P.; Marolla, A.; Di Daniele, F.; Noce, A. Vegan Diet Health Benefits in Metabolic Syndrome. Nutrients 2021, 13, 817. [Google Scholar] [CrossRef]
- Mennen, L.I.; Witteman, J.C.; Den Breeijen, J.H.; Schouten, E.G.; de Jong, P.T.; Hofman, A.; Grobbee, D.E. The association of dietary fat and fiber with coagulation factor VII in the elderly: The Rotterdam Study. Am. J. Clin. Nutr. 1997, 65, 732–736. [Google Scholar] [CrossRef]
- Paduchová, Z.; Katrenčíková, B.; Vaváková, M.; Laubertová, L.; Nagyová, Z.; Garaiova, I.; Ďuračková, Z.; Trebatická, J. The Effect of Omega-3 Fatty Acids on Thromboxane, Brain-Derived Neurotrophic Factor, Homocysteine, and Vitamin D in Depressive Children and Adolescents: Randomized Controlled Trial. Nutrients 2021, 13, 1095. [Google Scholar] [CrossRef]
- Li, D.; Sinclair, A.; Mann, N.; Turner, A.; Ball, M.; Kelly, F.; Abedin, L.; Wilson, A. The association of diet and thrombotic risk factors in healthy male vegetarians and meat-eaters. Eur. J. Clin. Nutr. 1999, 53, 612–619. [Google Scholar] [CrossRef]
- Goldman, D.M.; Nagra, M.; Warbeck, C.; Winings, E.; Landry, M.J. Plant-Based Diets: Nutritional Considerations and Public Health Implications. In Handbook of Public Health Nutrition: International, National, and Regional Perspectives; Preedy, V.R., Patel, V.B., Eds.; Springer Nature: Cham, Switzerland, 2025; pp. 1–27. [Google Scholar]
- Pawlak, R.; Parrott, S.J.; Raj, S.; Cullum-Dugan, D.; Lucus, D. How prevalent is vitamin B12 deficiency among vegetarians? Nutr. Rev. 2013, 71, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Stabler, S.P. Clinical practice. Vitamin B12 deficiency. N. Engl. J. Med. 2013, 368, 149–160. [Google Scholar] [CrossRef] [PubMed]
- Rizos, E.C.; Ntzani, E.E.; Bika, E.; Kostapanos, M.S.; Elisaf, M.S. Association between omega-3 fatty acid supplementation and risk of major cardiovascular disease events: A systematic review and meta-analysis. Jama 2012, 308, 1024–1033. [Google Scholar] [CrossRef] [PubMed]
- Harrison, S.; Couture, P.; Lamarche, B. Diet Quality, Saturated Fat and Metabolic Syndrome. Nutrients 2020, 12, 3232. [Google Scholar] [CrossRef] [PubMed]
- Swarup, S.; Ahmed, I.; Grigorova, Y.; Zeltser, R. Metabolic syndrome. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2024. [Google Scholar]
- Saklayen, M.G. The global epidemic of the metabolic syndrome. Curr. Hypertens. Rep. 2018, 20, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Li, X.; Adams, H.; Kubena, K.; Guo, S. Etiology of metabolic syndrome and dietary intervention. Int. J. Mol. Sci. 2018, 20, 128. [Google Scholar] [CrossRef]
- Kilpeläinen, T.O.; Zillikens, M.C.; Stančákova, A.; Finucane, F.M.; Ried, J.S.; Langenberg, C.; Zhang, W.; Beckmann, J.S.; Luan, J.; Vandenput, L.; et al. Genetic variation near IRS1 associates with reduced adiposity and an impaired metabolic profile. Nat. Genet. 2011, 43, 753–760. [Google Scholar] [CrossRef]
- Ferguson-Smith, A.C.; Patti, M.-E. You are what your dad ate. Cell Metab. 2011, 13, 115–117. [Google Scholar] [CrossRef]
- Giannopoulos, C.K.; Tzima, I.G.; Tentolouris, N.K.; Vasileiadis, I.A. Common pathogenetic pathways of non-alcoholic fatty liver disease and type 2 diabetes mellitus. Curr. Diabetes Rev. 2023, 19, 96–114. [Google Scholar] [CrossRef]
- Esser, N.; Legrand-Poels, S.; Piette, J.; Scheen, A.J.; Paquot, N. Inflammation as a link between obesity, metabolic syndrome and type 2 diabetes. Diabetes Res. Clin. Pract. 2014, 105, 141–150. [Google Scholar] [CrossRef]
- Mohamed, S.M.; Shalaby, M.A.; El-Shiekh, R.A.; El-Banna, H.A.; Emam, S.R.; Bakr, A.F. Metabolic syndrome: Risk factors, diagnosis, pathogenesis, and management with natural approaches. Food Chem. Adv. 2023, 3, 100335. [Google Scholar] [CrossRef]
- Gluvic, Z.; Zaric, B.; Resanovic, I.; Obradovic, M.; Mitrovic, A.; Radak, D.; Isenovic, E.R. Link between metabolic syndrome and insulin resistance. Curr. Vasc. Pharmacol. 2017, 15, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Kaur, R.; Kaur, M.; Singh, J. Endothelial dysfunction and platelet hyperactivity in type 2 diabetes mellitus: Molecular insights and therapeutic strategies. Cardiovasc. Diabetol. 2018, 17, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Knebel, S.M.; Sprague, R.S.; Stephenson, A.H. Prostacyclin receptor expression on platelets of humans with type 2 diabetes is inversely correlated with hemoglobin A1c levels. Prostaglandins Other Lipid Mediat. 2015, 116, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Kelem, A.; Adane, T.; Shiferaw, E. Insulin resistance-induced platelet hyperactivity and a potential biomarker role of platelet parameters: A narrative review. Diabetes Metab. Syndr. Obes. 2023, 16, 2843–2853. [Google Scholar] [CrossRef] [PubMed]
- Saluja, M.; Swami, Y.K.; Meena, S. Study of Impact of Glycemic Status (HbA1c) on Platelet Activity measured by Mean Platelet Volume & Vascular Complications in Diabetics. J. Assoc. Physicians India 2019, 67, 26–29. [Google Scholar]
- Shimodaira, M.; Niwa, T.; Nakajima, K.; Kobayashi, M.; Hanyu, N.; Nakayama, T. Correlation between mean platelet volume and fasting plasma glucose levels in prediabetic and normoglycemic individuals. Cardiovasc. Diabetol. 2013, 12, 14. [Google Scholar] [CrossRef]
- Cassano, V.; Armentaro, G.; Iembo, D.; Miceli, S.; Fiorentino, T.V.; Succurro, E.; Perticone, M.; Arturi, F.; Hribal, M.L.; Montalcini, T. Mean platelet volume (MPV) as new marker of diabetic macrovascular complications in patients with different glucose homeostasis: Platelets in cardiovascular risk. Cardiovasc. Diabetol. 2024, 23, 89. [Google Scholar] [CrossRef]
- Batten, L.; Sathyapalan, T.; Palmer, T.M. Molecular Mechanisms Linking Diabetes with Increased Risk of Thrombosis. Int. J. Mol. Sci. 2023, 24, 17465. [Google Scholar] [CrossRef]
- Chung, A.W.; Jurasz, P.; Hollenberg, M.D.; Radomski, M.W. Mechanisms of action of proteinase-activated receptor agonists on human platelets. Br. J. Pharmacol. 2002, 135, 1123. [Google Scholar] [CrossRef]
- Ferreiro, J.L.; Gómez-Hospital, J.A.; Angiolillo, D.J. Platelet abnormalities in diabetes mellitus. Diabetes Vasc. Dis. Res. 2010, 7, 251–259. [Google Scholar] [CrossRef]
- Undas, A.; Ariëns, R.A. Fibrin clot structure and function: A role in the pathophysiology of arterial and venous thromboembolic diseases. Arterioscler. Thromb. Vasc. Biol. 2011, 31, e88–e99. [Google Scholar] [CrossRef]
- Zhang, J.; Cao, J.; Zhang, Y.; Li, H.; Zhang, H.; Huo, Y.; Li, J.; Liu, X.; Wang, X.; Qin, X. Baseline plasma zinc and risk of first stroke in hypertensive patients: A nested case-control study. Stroke 2019, 50, 3255–3258. [Google Scholar] [CrossRef]
- Sobczak, A.I.; Stewart, A.J. Coagulatory defects in type-1 and type-2 diabetes. Int. J. Mol. Sci. 2019, 20, 6345. [Google Scholar] [CrossRef]
- Sobczak, A.I.; Stefanowicz, F.; Pitt, S.J.; Ajjan, R.A.; Stewart, A.J. Total plasma magnesium, zinc, copper and selenium concentrations in type-I and type-II diabetes. Biometals 2019, 32, 123–138. [Google Scholar] [CrossRef]
- Li, Z.; Wang, C.; Li, L.; Shao, M.; Wang, L.; Lv, X.; Gao, C.; Niu, H.; Li, B. The study on the correlation between six kinds of mineral elements and diabetes. Biol. Trace Elem. Res. 2018, 183, 226–232. [Google Scholar] [CrossRef]
- Grüngreiff, K.; Gottstein, T.; Reinhold, D. Zinc deficiency—An independent risk factor in the pathogenesis of haemorrhagic stroke? Nutrients 2020, 12, 3548. [Google Scholar] [CrossRef]
- Ding, P.-F.; Zhang, H.-S.; Wang, J.; Gao, Y.-Y.; Mao, J.-N.; Hang, C.-H.; Li, W. Insulin resistance in ischemic stroke: Mechanisms and therapeutic approaches. Front. Endocrinol. 2022, 13, 1092431. [Google Scholar] [CrossRef] [PubMed]
- Tousoulis, D.; Simopoulou, C.; Papageorgiou, N.; Oikonomou, E.; Hatzis, G.; Siasos, G.; Tsiamis, E.; Stefanadis, C. Endothelial dysfunction in conduit arteries and in microcirculation. Novel therapeutic approaches. Pharmacol. Ther. 2014, 144, 253–267. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.-S.; Schulman, I.H.; Raij, L. Vascular inflammation, insulin resistance, and endothelial dysfunction in salt-sensitive hypertension: Role of nuclear factor kappa B activation. J. Hypertens. 2010, 28, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Ormazabal, V.; Nair, S.; Elfeky, O.; Aguayo, C.; Salomon, C.; Zuñiga, F.A. Association between insulin resistance and the development of cardiovascular disease. Cardiovasc. Diabetol. 2018, 17, 1–14. [Google Scholar] [CrossRef]
- Cusi, K.; Maezono, K.; Osman, A.; Pendergrass, M.; Patti, M.E.; Pratipanawatr, T.; DeFronzo, R.A.; Kahn, C.R.; Mandarino, L.J. Insulin resistance differentially affects the PI 3-kinase–and MAP kinase–mediated signaling in human muscle. J. Clin. Investig. 2000, 105, 311–320. [Google Scholar] [CrossRef]
- Schultze, S.M.; Hemmings, B.A.; Niessen, M.; Tschopp, O. PI3K/AKT, MAPK and AMPK signalling: Protein kinases in glucose homeostasis. Expert. Rev. Mol. Med. 2012, 14, e1. [Google Scholar] [CrossRef]
- Gutiérrez, A.; Contreras, C.; Sánchez, A.; Prieto, D. Role of phosphatidylinositol 3-kinase (PI3K), mitogen-activated protein kinase (MAPK), and protein kinase C (PKC) in calcium signaling pathways linked to the α1-adrenoceptor in resistance arteries. Front. Physiol. 2019, 10, 55. [Google Scholar] [CrossRef]
- Tangvarasittichai, S. Oxidative stress, insulin resistance, dyslipidemia and type 2 diabetes mellitus. World J. Diabetes 2015, 6, 456. [Google Scholar] [CrossRef] [PubMed]
- Mohammadian, K.; Fakhar, F.; Keramat, S.; Stanek, A. The Role of antioxidants in the treatment of metabolic dysfunction-associated fatty liver disease: A systematic review. Antioxidants 2024, 13, 797. [Google Scholar] [CrossRef]
- Potenza, M.A.; Gagliardi, S.; Nacci, C.; Carratu, M.R.; Montagnani, M. Endothelial dysfunction in diabetes: From mechanisms to therapeutic targets. Curr. Med. Chem. 2009, 16, 94–112. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, A.M.; Yan, S.D.; Wautier, J.-L.; Stern, D. Activation of receptor for advanced glycation end products: A mechanism for chronic vascular dysfunction in diabetic vasculopathy and atherosclerosis. Circ. Res. 1999, 84, 489–497. [Google Scholar] [CrossRef]
- Wautier, M.-P.; Chappey, O.; Corda, S.; Stern, D.M.; Schmidt, A.M.; Wautier, J.-L. Activation of NADPH oxidase by AGE links oxidant stress to altered gene expression via RAGE. Am. J. Physiol.-Endocrinol. Metab. 2001, 280, E685–E694. [Google Scholar] [CrossRef] [PubMed]
- Donath, M.Y.; Shoelson, S.E. Type 2 diabetes as an inflammatory disease. Nat. Rev. Immunol. 2011, 11, 98–107. [Google Scholar] [CrossRef]
- Masters, S.L.; Dunne, A.; Subramanian, S.L.; Hull, R.L.; Tannahill, G.M.; Sharp, F.A.; Becker, C.; Franchi, L.; Yoshihara, E.; Chen, Z. Activation of the NLRP3 inflammasome by islet amyloid polypeptide provides a mechanism for enhanced IL-1β in type 2 diabetes. Nat. Immunol. 2010, 11, 897–904. [Google Scholar] [CrossRef]
- Fuchs, T.A.; Brill, A.; Duerschmied, D.; Schatzberg, D.; Monestier, M.; Myers Jr, D.D.; Wrobleski, S.K.; Wakefield, T.W.; Hartwig, J.H.; Wagner, D.D. Extracellular DNA traps promote thrombosis. Proc. Natl. Acad. Sci. USA 2010, 107, 15880–15885. [Google Scholar] [CrossRef]
- Asghar, A.; Sheikh, N. Role of immune cells in obesity induced low grade inflammation and insulin resistance. Cell. Immunol. 2017, 315, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Han, M.S.; Jung, D.Y.; Morel, C.; Lakhani, S.A.; Kim, J.K.; Flavell, R.A.; Davis, R.J. JNK expression by macrophages promotes obesity-induced insulin resistance and inflammation. Science 2013, 339, 218–222. [Google Scholar] [CrossRef]
- Kanda, H.; Tateya, S.; Tamori, Y.; Kotani, K.; Hiasa, K.; Kitazawa, R.; Kitazawa, S.; Miyachi, H.; Maeda, S.; Egashira, K. MCP-1 contributes to macrophage infiltration into adipose tissue, insulin resistance, and hepatic steatosis in obesity. J. Clin. Investig. 2006, 116, 1494–1505. [Google Scholar] [CrossRef]
- Pan, J.; Xia, L.; McEver, R.P. Comparison of promoters for the murine and human P-selectin genes suggests species-specific and conserved mechanisms for transcriptional regulation in endothelial cells. J. Biol. Chem. 1998, 273, 10058–10067. [Google Scholar] [CrossRef] [PubMed]
- Blokhin, I.O.; Lentz, S.R. Mechanisms of thrombosis in obesity. Curr. Opin. Hematol. 2013, 20, 437–444. [Google Scholar] [CrossRef]
- Molofsky, A.B.; Nussbaum, J.C.; Liang, H.-E.; Van Dyken, S.J.; Cheng, L.E.; Mohapatra, A.; Chawla, A.; Locksley, R.M. Innate lymphoid type 2 cells sustain visceral adipose tissue eosinophils and alternatively activated macrophages. J. Exp. Med. 2013, 210, 535–549. [Google Scholar] [CrossRef]
- Lee, M.-W.; Odegaard, J.I.; Mukundan, L.; Qiu, Y.; Molofsky, A.B.; Nussbaum, J.C.; Yun, K.; Locksley, R.M.; Chawla, A. Activated type 2 innate lymphoid cells regulate beige fat biogenesis. Cell 2015, 160, 74–87. [Google Scholar] [CrossRef]
- Karczewski, J.; Śledzińska, E.; Baturo, A.; Jończyk, I.; Maleszko, A.; Samborski, P.; Begier-Krasińska, B.; Dobrowolska, A. Obesity and inflammation. Eur. Cytokine Netw. 2018, 29, 83–94. [Google Scholar] [CrossRef]
- Wu, H.; Ghosh, S.; Perrard, X.D.; Feng, L.; Garcia, G.E.; Perrard, J.L.; Sweeney, J.F.; Peterson, L.E.; Chan, L.; Smith, C.W. T-cell accumulation and regulated on activation, normal T cell expressed and secreted upregulation in adipose tissue in obesity. Circulation 2007, 115, 1029–1038. [Google Scholar] [CrossRef]
- Feuerer, M.; Herrero, L.; Cipolletta, D.; Naaz, A.; Wong, J.; Nayer, A.; Lee, J.; Goldfine, A.; Benoist, C.; Shoelson, S. Fat Treg cells: A liaison between the immune and metabolic systems. Nat. Med. 2009, 15, 930. [Google Scholar] [CrossRef] [PubMed]
- Levi, M.; van der Poll, T.; ten Cate, H. Tissue factor in infection and severe inflammation. In Seminars in Thrombosis and Hemostasis; Thieme Medical Publishers, Inc.: New York, NY, USA, 2006; pp. 033–039. [Google Scholar]
- Bladbjerg, E.M.; Stolberg, C.R.; Juhl, C.B. Effects of obesity surgery on blood coagulation and fibrinolysis: A literature review. Thromb. Haemost. 2020, 120, 579–591. [Google Scholar] [CrossRef] [PubMed]
- Patel, V.B. Molecular Nutrition: Carbohydrates; Academic Press: Cambridge, MA, USA, 2019. [Google Scholar]
- Pérez-Pérez, A.; Sánchez-Jiménez, F.; Vilariño-García, T.; Sánchez-Margalet, V. Role of leptin in inflammation and vice versa. Int. J. Mol. Sci. 2020, 21, 5887. [Google Scholar] [CrossRef]
- Tanaka, T.; Narazaki, M.; Kishimoto, T. IL-6 in inflammation, immunity, and disease. Cold Spring Harb. Perspect. Biol. 2014, 6, a016295. [Google Scholar] [CrossRef]
- Alzamil, H. Elevated serum TNF-α is related to obesity in type 2 diabetes mellitus and is associated with glycemic control and insulin resistance. J. Obes. 2020, 2020, 5076858. [Google Scholar] [CrossRef]
- Hirosumi, J.; Tuncman, G.; Chang, L.; Görgün, C.Z.; Uysal, K.T.; Maeda, K.; Karin, M.; Hotamisligil, G.S. A central role for JNK in obesity and insulin resistance. Nature 2002, 420, 333–336. [Google Scholar] [CrossRef] [PubMed]
- Summers, S.A.; Garza, L.A.; Zhou, H.; Birnbaum, M.J. Regulation of insulin-stimulated glucose transporter GLUT4 translocation and Akt kinase activity by ceramide. Mol. Cell. Biol. 1998, 18, 5457–5464. [Google Scholar] [CrossRef]
- Holland, W.L.; Summers, S.A. Sphingolipids, insulin resistance, and metabolic disease: New insights from in vivo manipulation of sphingolipid metabolism. Endocr. Rev. 2008, 29, 381–402. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Zhang, S.; Yang, C.; Li, Q.; Wang, S.; Xu, X.; Hao, J.; Li, C. A novel PTP1B inhibitor-phosphate of polymannuronic acid ameliorates insulin resistance by regulating IRS-1/Akt signaling. Int. J. Mol. Sci. 2021, 22, 12693. [Google Scholar] [CrossRef]
- Fakhar, F.; Mohammadian, K.; Keramat, S.; Stanek, A. The Potential Role of Dietary Polyphenols in the Prevention and Treatment of Acute Leukemia. Nutrients 2024, 16, 4100. [Google Scholar] [CrossRef]
- Ojaroodi, A.F.; Jafarnezhad, F.; Eskandari, Z.; Keramat, S.; Stanek, A. Recent Updates and Advances in the Association Between Vitamin D Deficiency and Risk of Thrombotic Disease. Nutrients 2024, 17, 90. [Google Scholar] [CrossRef]
- Jakubiak, G.K.; Osadnik, K.; Lejawa, M.; Osadnik, T.; Goławski, M.; Lewandowski, P.; Pawlas, N. Obesity and insulin resistance is the component of the metabolic syndrome most strongly associated with oxidative stress. Antioxidants 2021, 11, 79. [Google Scholar] [CrossRef]
- Abad-Jiménez, Z.; López-Domènech, S.; Gómez-Abril, S.Á.; Periañez-Gómez, D.; de Marañón, A.M.; Bañuls, C.; Morillas, C.; Víctor, V.M.; Rocha, M. Effect of roux-en-Y bariatric bypass surgery on subclinical atherosclerosis and oxidative stress markers in leukocytes of obese patients: A one-year follow-up study. Antioxidants 2020, 9, 734. [Google Scholar] [CrossRef]
- Naomi, R.; Teoh, S.H.; Embong, H.; Balan, S.S.; Othman, F.; Bahari, H.; Yazid, M.D. The role of oxidative stress and inflammation in obesity and its impact on cognitive impairments—A narrative review. Antioxidants 2023, 12, 1071. [Google Scholar] [CrossRef]
- Henning, R.J. Obesity and obesity-induced inflammatory disease contribute to atherosclerosis: A review of the pathophysiology and treatment of obesity. Am. J. Cardiovasc. Dis. 2021, 11, 504. [Google Scholar] [PubMed]
- Motie, M.; Evangelista, L.S.; Horwich, T.; Lombardo, D.; Zaldivar, F.; Hamilton, M.; Fonarow, G.C. Association between inflammatory biomarkers and adiposity in obese patients with heart failure and metabolic syndrome. Exp. Ther. Med. 2014, 8, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Damms-Machado, A.; Louis, S.; Schnitzer, A.; Volynets, V.; Rings, A.; Basrai, M.; Bischoff, S.C. Gut permeability is related to body weight, fatty liver disease, and insulin resistance in obese individuals undergoing weight reduction. Am. J. Clin. Nutr. 2017, 105, 127–135. [Google Scholar] [CrossRef]
- Everard, A.; Belzer, C.; Geurts, L.; Ouwerkerk, J.P.; Druart, C.; Bindels, L.B.; Guiot, Y.; Derrien, M.; Muccioli, G.G.; Delzenne, N.M. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc. Natl. Acad. Sci. USA 2013, 110, 9066–9071. [Google Scholar] [CrossRef]
- Tang, W.W.; Hazen, S.L. The gut microbiome and its role in cardiovascular diseases. Circulation 2017, 135, 1008–1010. [Google Scholar] [CrossRef] [PubMed]
- Jayashree, B.; Bibin, Y.; Prabhu, D.; Shanthirani, C.; Gokulakrishnan, K.; Lakshmi, B.; Mohan, V.; Balasubramanyam, M. Increased circulatory levels of lipopolysaccharide (LPS) and zonulin signify novel biomarkers of proinflammation in patients with type 2 diabetes. Mol. Cell. Biochem. 2014, 388, 203–210. [Google Scholar] [CrossRef]
- Ząbczyk, M.; Kruk, A.; Natorska, J.; Undas, A. Low-grade endotoxemia in acute pulmonary embolism: Links with prothrombotic plasma fibrin clot phenotype. Thromb. Res. 2023, 232, 70–76. [Google Scholar] [CrossRef]
- Frazier, T.H.; DiBaise, J.K.; McClain, C.J. Gut microbiota, intestinal permeability, obesity-induced inflammation, and liver injury. J. Parenter. Enter. Nutr. 2011, 35, 14S–20S. [Google Scholar] [CrossRef]
- Ye, J.; Gao, Z.; Yin, J.; He, Q. Hypoxia is a potential risk factor for chronic inflammation and adiponectin reduction in adipose tissue of ob/ob and dietary obese mice. Am. J. Physiol.-Endocrinol. Metab. 2007, 293, E1118–E1128. [Google Scholar] [CrossRef]
- De Pergola, G.; Pannacciulli, N. Coagulation and fibrinolysis abnormalities in obesity. J. Endocrinol. Investig. 2002, 25, 899–904. [Google Scholar] [CrossRef]
- Kornblith, L.Z.; Howard, B.; Kunitake, R.; Redick, B.; Nelson, M.; Cohen, M.J.; Callcut, R. Obesity and clotting: Body mass index independently contributes to hypercoagulability after injury. J. Trauma. Acute Care Surg. 2015, 78, 30–38. [Google Scholar] [CrossRef]
- Tadic, M.; Ivanovic, B.; Celic, V.; Cuspidi, C. Are the metabolic syndrome, blood pressure pattern, and their interaction responsible for the right ventricular remodeling? Blood Press. Monit. 2013, 18, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Faramawi, M.F.; Delongchamp, R.; Said, Q.; Jadhav, S.; Abouelenien, S. Metabolic syndrome is associated with visit-to-visit systolic blood pressure variability in the US adults. Hypertens. Res. 2014, 37, 875–879. [Google Scholar] [CrossRef] [PubMed]
- Senchenkova, E.Y.; Russell, J.; Almeida-Paula, L.D.; Harding, J.W.; Granger, D.N. Angiotensin II–mediated microvascular thrombosis. Hypertension 2010, 56, 1089–1095. [Google Scholar] [CrossRef]
- Dielis, A.W.; Smid, M.; Spronk, H.M.; Hamulyak, K.; Kroon, A.A.; ten Cate, H.; de Leeuw, P.W. The prothrombotic paradox of hypertension: Role of the renin-angiotensin and kallikrein-kinin systems. Hypertension 2005, 46, 1236–1242. [Google Scholar] [CrossRef]
- Verma, K.; Pant, M.; Paliwal, S.; Dwivedi, J.; Sharma, S. An insight on multicentric signaling of angiotensin II in cardiovascular system: A recent update. Front. Pharmacol. 2021, 12, 734917. [Google Scholar] [CrossRef]
- Ikhapoh, I.A.; Pelham, C.J.; Agrawal, D.K. Synergistic effect of angiotensin II on vascular endothelial growth factor-A-mediated differentiation of bone marrow-derived mesenchymal stem cells into endothelial cells. Stem Cell Res. Ther. 2015, 6, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.S.; Shin, J.H. Association between D-dimer and long-term mortality in patients with acute severe hypertension visiting the emergency department. Clin. Hypertens. 2023, 29, 16. [Google Scholar] [CrossRef]
- Eldour, A.A.A.; Khalafallah, T.O.; Noja, H.M.; Saad, E.S.M.; Elsayid, M.; Babker, A.M.A.A.A. Fibrinogen Levels in Hypertensive and Normotensive: A Cross-Sectional Study from El-Obied City, Sudan. J. Biosci. Med. 2016, 4, 28–32. [Google Scholar] [CrossRef][Green Version]
- Osman, S.S.A.; Ali, A.E.; Muddathir, A.R.M. Measurements of Plasma Fibrinogen Level, Total Cholesterol and Triglyceride Levels in Hypertensive Sudanese Patients. Am. J. Med. Med. Sci. 2014, 4, 161–165. [Google Scholar]
- Dörffel, Y.; Lätsch, C.; Stuhlmüller, B.; Schreiber, S.; Scholze, S.; Burmester, G.R.; Scholze, J. Preactivated peripheral blood monocytes in patients with essential hypertension. Hypertension 1999, 34, 113–117. [Google Scholar] [CrossRef]
- Wiśniewska, K.; Okręglicka, K.M.; Nitsch-Osuch, A.; Oczkowski, M. Plant-based diets and metabolic syndrome components: The questions that still need to be answered—A narrative review. Nutrients 2024, 16, 165. [Google Scholar] [CrossRef]
- Ivanova, S.; Delattre, C.; Karcheva-Bahchevanska, D.; Benbasat, N.; Nalbantova, V.; Ivanov, K. Plant-based diet as a strategy for weight control. Foods 2021, 10, 3052. [Google Scholar] [CrossRef]
- Sabaté, J.; Wien, M. A perspective on vegetarian dietary patterns and risk of metabolic syndrome. Br. J. Nutr. 2015, 113, S136–S143. [Google Scholar] [CrossRef]
- Kahleova, H.; Fleeman, R.; Hlozkova, A.; Holubkov, R.; Barnard, N.D. A plant-based diet in overweight individuals in a 16-week randomized clinical trial: Metabolic benefits of plant protein. Nutr. Diabetes 2018, 8, 58. [Google Scholar] [CrossRef] [PubMed]
- Dinu, M.; Abbate, R.; Gensini, G.F.; Casini, A.; Sofi, F. Vegetarian, vegan diets and multiple health outcomes: A systematic review with meta-analysis of observational studies. Crit. Rev. Food Sci. Nutr. 2017, 57, 3640–3649. [Google Scholar] [CrossRef] [PubMed]
- Kahleova, H.; Levin, S.; Barnard, N. Cardio-Metabolic Benefits of Plant-Based Diets. Nutrients 2017, 9, 848. [Google Scholar] [CrossRef]
- Turner-McGrievy, G.M.; Davidson, C.R.; Wingard, E.E.; Wilcox, S.; Frongillo, E.A. Comparative effectiveness of plant-based diets for weight loss: A randomized controlled trial of five different diets. Nutrition 2015, 31, 350–358. [Google Scholar] [CrossRef]
- Huang, R.Y.; Huang, C.C.; Hu, F.B.; Chavarro, J.E. Vegetarian Diets and Weight Reduction: A Meta-Analysis of Randomized Controlled Trials. J. Gen. Intern. Med. 2016, 31, 109–116. [Google Scholar] [CrossRef]
- Anderson, J.W.; Baird, P.; Davis, R.H., Jr.; Ferreri, S.; Knudtson, M.; Koraym, A.; Waters, V.; Williams, C.L. Health benefits of dietary fiber. Nutr. Rev. 2009, 67, 188–205. [Google Scholar] [CrossRef]
- Howarth, N.C.; Saltzman, E.; Roberts, S.B. Dietary fiber and weight regulation. Nutr. Rev. 2001, 59, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Hotta, K.; Funahashi, T.; Arita, Y.; Takahashi, M.; Matsuda, M.; Okamoto, Y.; Iwahashi, H.; Kuriyama, H.; Ouchi, N.; Maeda, K.; et al. Plasma concentrations of a novel, adipose-specific protein, adiponectin, in type 2 diabetic patients. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 1595–1599. [Google Scholar] [CrossRef] [PubMed]
- Kuchta, A.; Lebiedzińska, A.; Fijałkowski, M.; Gałąska, R.; Kreft, E.; Totoń, M.; Czaja, K.; Kozłowska, A.; Ćwiklińska, A.; Kortas-Stempak, B.; et al. Impact of plant-based diet on lipid risk factors for atherosclerosis. Cardiol. J. 2016, 23, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Trautwein, E.A.; McKay, S. The Role of Specific Components of a Plant-Based Diet in Management of Dyslipidemia and the Impact on Cardiovascular Risk. Nutrients 2020, 12, 2671. [Google Scholar] [CrossRef]
- Yokoyama, Y.; Nishimura, K.; Barnard, N.D.; Takegami, M.; Watanabe, M.; Sekikawa, A.; Okamura, T.; Miyamoto, Y. Vegetarian diets and blood pressure: A meta-analysis. JAMA Intern. Med. 2014, 174, 577–587. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Chaimani, A.; Schwedhelm, C.; Toledo, E.; Pünsch, M.; Hoffmann, G.; Boeing, H. Comparative effects of different dietary approaches on blood pressure in hypertensive and pre-hypertensive patients: A systematic review and network meta-analysis. Crit. Rev. Food Sci. Nutr. 2019, 59, 2674–2687. [Google Scholar] [CrossRef]
- Dayi, T.; Ozgoren, M. Effects of the Mediterranean diet on the components of metabolic syndrome. J. Prev. Med. Hyg. 2022, 63, E56. [Google Scholar]
- Rehman, K.; Haider, K.; Jabeen, K.; Akash, M.S.H. Current perspectives of oleic acid: Regulation of molecular pathways in mitochondrial and endothelial functioning against insulin resistance and diabetes. Rev. Endocr. Metab. Disord. 2020, 21, 631–643. [Google Scholar] [CrossRef]
- Massaro, M.; Scoditti, E.; Carluccio, M.A.; Calabriso, N.; Santarpino, G.; Verri, T.; De Caterina, R. Effects of olive oil on blood pressure: Epidemiological, clinical, and mechanistic evidence. Nutrients 2020, 12, 1548. [Google Scholar] [CrossRef] [PubMed]
- Siervo, M.; Lara, J.; Ogbonmwan, I.; Mathers, J.C. Inorganic nitrate and beetroot juice supplementation reduces blood pressure in adults: A systematic review and meta-analysis. J. Nutr. 2013, 143, 818–826. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xie, Y.; Berglund, E.D.; Coate, K.C.; He, T.T.; Katafuchi, T.; Xiao, G.; Potthoff, M.J.; Wei, W.; Wan, Y.; et al. The starvation hormone, fibroblast growth factor-21, extends lifespan in mice. elife 2012, 1, e00065. [Google Scholar] [CrossRef]
- D’Innocenzo, S.; Biagi, C.; Lanari, M. Obesity and the Mediterranean diet: A review of evidence of the role and sustainability of the Mediterranean diet. Nutrients 2019, 11, 1306. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Martínez-González, M.A.; Tong, T.Y.; Forouhi, N.G.; Khandelwal, S.; Prabhakaran, D.; Mozaffarian, D.; de Lorgeril, M. Definitions and potential health benefits of the Mediterranean diet: Views from experts around the world. BMC Med. 2014, 12, 112. [Google Scholar] [CrossRef]
- de Souza, P.A.L.; Marcadenti, A.; Portal, V.L. Effects of olive oil phenolic compounds on inflammation in the prevention and treatment of coronary artery disease. Nutrients 2017, 9, 1087. [Google Scholar] [CrossRef]
- Alam, M.A.; Subhan, N.; Rahman, M.M.; Uddin, S.J.; Reza, H.M.; Sarker, S.D. Effect of citrus flavonoids, naringin and naringenin, on metabolic syndrome and their mechanisms of action. Adv. Nutr. 2014, 5, 404–417. [Google Scholar] [CrossRef]
- Pérez-Jiménez, J.; Neveu, V.; Vos, F.; Scalbert, A. Systematic analysis of the content of 502 polyphenols in 452 foods and beverages: An application of the phenol-explorer database. J. Agric. Food Chem. 2010, 58, 4959–4969. [Google Scholar] [CrossRef]
- Pan, M.H.; Lai, C.S.; Dushenkov, S.; Ho, C.T. Modulation of inflammatory genes by natural dietary bioactive compounds. J. Agric. Food Chem. 2009, 57, 4467–4477. [Google Scholar] [CrossRef]
- Uribarri, J.; Woodruff, S.; Goodman, S.; Cai, W.; Chen, X.; Pyzik, R.; Yong, A.; Striker, G.E.; Vlassara, H. Advanced glycation end products in foods and a practical guide to their reduction in the diet. J. Am. Diet. Assoc. 2010, 110, 911–916.e12. [Google Scholar] [CrossRef]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Habauzit, V.; Morand, C. Evidence for a protective effect of polyphenols-containing foods on cardiovascular health: An update for clinicians. Ther. Adv. Chronic Dis. 2012, 3, 87–106. [Google Scholar] [CrossRef]
- Kersten, S. Integrated physiology and systems biology of PPARα. Mol. Metab. 2014, 3, 354–371. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; McKenzie, C.; Potamitis, M.; Thorburn, A.N.; Mackay, C.R.; Macia, L. The role of short-chain fatty acids in health and disease. Adv. Immunol. 2014, 121, 91–119. [Google Scholar] [PubMed]
- Zhang, L.; Chen, T.; Yin, Y.; Zhang, C.Y.; Zhang, Y.L. Dietary microRNA-A Novel Functional Component of Food. Adv. Nutr. 2019, 10, 711–721. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Bogensberger, B.; Benčič, A.; Knüppel, S.; Boeing, H.; Hoffmann, G. Effects of oils and solid fats on blood lipids: A systematic review and network meta-analysis. J. Lipid Res. 2018, 59, 1771–1782. [Google Scholar] [CrossRef]
- Gylling, H.; Plat, J.; Turley, S.; Ginsberg, H.N.; Ellegård, L.; Jessup, W.; Jones, P.J.; Lütjohann, D.; Maerz, W.; Masana, L.; et al. Plant sterols and plant stanols in the management of dyslipidaemia and prevention of cardiovascular disease. Atherosclerosis 2014, 232, 346–360. [Google Scholar] [CrossRef]
- Trautwein, E.A.; Vermeer, M.A.; Hiemstra, H.; Ras, R.T. LDL-Cholesterol Lowering of Plant Sterols and Stanols—Which Factors Influence Their Efficacy? Nutrients 2018, 10, 1262. [Google Scholar] [CrossRef]
- Vergeer, M.; Holleboom, A.G.; Kastelein, J.J.P.; Kuivenhoven, J.A. The HDL hypothesis: Does high-density lipoprotein protect from atherosclerosis? J. Lipid Res. 2010, 51, 2058–2073. [Google Scholar] [CrossRef]
- Han, M.; Guan, L.; Ren, Y.; Zhao, Y.; Liu, D.; Zhang, D.; Liu, L.; Liu, F.; Chen, X.; Cheng, C.; et al. Dietary iron intake and risk of death due to cardiovascular diseases: A systematic review and dose-response meta-analysis of prospective cohort studies. Asia Pac. J. Clin. Nutr. 2020, 29, 309–321. [Google Scholar]
- Clarke, R.; Peden, J.F.; Hopewell, J.C.; Kyriakou, T.; Goel, A.; Heath, S.C.; Parish, S.; Barlera, S.; Franzosi, M.G.; Rust, S.; et al. Genetic variants associated with Lp(a) lipoprotein level and coronary disease. N. Engl. J. Med. 2009, 361, 2518–2528. [Google Scholar] [CrossRef]
- Jenkins, D.J.; Kendall, C.W.; Marchie, A.; Jenkins, A.L.; Augustin, L.S.; Ludwig, D.S.; Barnard, N.D.; Anderson, J.W. Type 2 diabetes and the vegetarian diet. Am. J. Clin. Nutr. 2003, 78, 610s–616s. [Google Scholar] [CrossRef]
- McMacken, M.; Shah, S. A plant-based diet for the prevention and treatment of type 2 diabetes. J. Geriatr. Cardiol. 2017, 14, 342–354. [Google Scholar]
- Barnard, N.D.; Cohen, J.; Jenkins, D.J.; Turner-McGrievy, G.; Gloede, L.; Jaster, B.; Seidl, K.; Green, A.A.; Talpers, S. A low-fat vegan diet improves glycemic control and cardiovascular risk factors in a randomized clinical trial in individuals with type 2 diabetes. Diabetes Care 2006, 29, 1777–1783. [Google Scholar] [CrossRef] [PubMed]
- Anastasios, T.; Triece, T.; Isaura, A.-B. Effectiveness of plant-based diets in promoting well-being in the management of type 2 diabetes: A systematic review. BMJ Open Diabetes Res. Care 2018, 6, e000534. [Google Scholar] [CrossRef] [PubMed]
- Evert, A.B.; Boucher, J.L.; Cypress, M.; Dunbar, S.A.; Franz, M.J.; Mayer-Davis, E.J.; Neumiller, J.J.; Nwankwo, R.; Verdi, C.L.; Urbanski, P.; et al. Nutrition therapy recommendations for the management of adults with diabetes. Diabetes Care 2013, 36, 3821–3842. [Google Scholar] [CrossRef] [PubMed]
- Craig, W.J.; Mangels, A.R. Position of the American Dietetic Association: Vegetarian diets. J. Am. Diet. Assoc. 2009, 109, 1266–1282. [Google Scholar] [CrossRef]
- Rizzo, G.; Laganà, A.S.; Rapisarda, A.M.; La Ferrera, G.M.; Buscema, M.; Rossetti, P.; Nigro, A.; Muscia, V.; Valenti, G.; Sapia, F.; et al. Vitamin B12 among Vegetarians: Status, Assessment and Supplementation. Nutrients 2016, 8, 767. [Google Scholar] [CrossRef]
- Cicero, A.F.; Baggioni, A. Berberine and Its Role in Chronic Disease. Adv. Exp. Med. Biol. 2016, 928, 27–45. [Google Scholar]
- Marco, M.L.; Heeney, D.; Binda, S.; Cifelli, C.J.; Cotter, P.D.; Foligné, B.; Gänzle, M.; Kort, R.; Pasin, G.; Pihlanto, A.; et al. Health benefits of fermented foods: Microbiota and beyond. Curr. Opin. Biotechnol. 2017, 44, 94–102. [Google Scholar] [CrossRef]
- Longo, V.D.; Panda, S. Fasting, Circadian Rhythms, and Time-Restricted Feeding in Healthy Lifespan. Cell Metab. 2016, 23, 1048–1059. [Google Scholar] [CrossRef]
- Benatar, J.R.; Stewart, R.A.H. Cardiometabolic risk factors in vegans; A meta-analysis of observational studies. PLoS ONE 2018, 13, e0209086. [Google Scholar] [CrossRef]
- Vajdi, M.; Karimi, A.; Tousi, A.Z.; Hosseini, B.; Nikniaz, Z.; Farhangi, M.A. Association between plant-based diets and metabolic syndrome in obese adults from Iran: A cross-sectional study. BMC Endocr. Disord. 2023, 23, 109. [Google Scholar] [CrossRef]
- Chen, P.; Zhao, Y.; Chen, Y. A vegan diet improves insulin resistance in individuals with obesity: A systematic review and meta-analysis. Diabetol. Metab. Syndr. 2022, 14, 114. [Google Scholar] [CrossRef] [PubMed]
- Dybvik, J.S.; Svendsen, M.; Aune, D. Vegetarian and vegan diets and the risk of cardiovascular disease, ischemic heart disease and stroke: A systematic review and meta-analysis of prospective cohort studies. Eur. J. Nutr. 2023, 62, 51–69. [Google Scholar] [CrossRef] [PubMed]
- Famodu, A.; Osilesi, O.; Makinde, Y.; Osonuga, O.; Fakoya, T.; Ogunyemi, E.; Egbenehkhuere, I. The influence of a vegetarian diet on haemostatic risk factors for cardiovascular disease in Africans. Thromb. Res. 1999, 95, 31–36. [Google Scholar] [CrossRef]
- Yamagata, K.; Yamori, Y. Potential effects of soy isoflavones on the prevention of metabolic syndrome. Molecules 2021, 26, 5863. [Google Scholar] [CrossRef]
- Marini, H.R. Mediterranean diet and soy isoflavones for integrated management of the menopausal metabolic syndrome. Nutrients 2022, 14, 1550. [Google Scholar] [CrossRef] [PubMed]
- de Piano, A.; Masquio, D.C.; Dâmaso, A.R. The effects of soy products and isoflavones in metabolic syndrome and nonalcoholic fatty liver disease. Bioact. Food Diet. Interv. Diabetes 2019, 121–136. [Google Scholar]
- Huang, C.; Pang, D.; Luo, Q.; Chen, X.; Gao, Q.; Shi, L.; Liu, W.; Zou, Y.; Li, L.; Chen, Z. Soy isoflavones regulate lipid metabolism through an AKT/mTORC1 pathway in diet-induced obesity (DIO) male rats. Molecules 2016, 21, 586. [Google Scholar] [CrossRef] [PubMed]
- Jiang, G.; Li, L.; Fan, J.; Zhang, B.; Oso, A.; Xiao, C.; Yin, Y. Dietary soy isoflavones differentially regulate expression of the lipid-metabolic genes in different white adipose tissues of the female Bama mini-pigs. Biochem. Biophys. Res. Commun. 2015, 461, 159–164. [Google Scholar] [CrossRef]
- Chalvon-Demersay, T.; Azzout-Marniche, D.; Arfsten, J.; Egli, L.; Gaudichon, C.; Karagounis, L.G.; Tomé, D. A systematic review of the effects of plant compared with animal protein sources on features of metabolic syndrome. J. Nutr. 2017, 147, 281–292. [Google Scholar] [CrossRef]
- Neshatbini Tehrani, A.; Hatami, B.; Daftari, G.; Hekmatdoost, A.; Yari, Z.; Salehpour, A.; Hosseini, S.A.; Helli, B. The effect of soy isoflavones supplementation on metabolic status in patients with non-alcoholic fatty liver disease: A randomized placebo controlled clinical trial. BMC Public. Health 2024, 24, 1362. [Google Scholar] [CrossRef] [PubMed]
- Hernáez, Á.; Castañer, O.; Tresserra-Rimbau, A.; Pintó, X.; Fitó, M.; Casas, R.; Martínez-González, M.Á.; Corella, D.; Salas-Salvadó, J.; Lapetra, J. Mediterranean diet and atherothrombosis biomarkers: A randomized controlled trial. Mol. Nutr. Food Res. 2020, 64, 2000350. [Google Scholar] [CrossRef]
- Turner-McGrievy, G.M.; Wirth, M.D.; Shivappa, N.; Wingard, E.E.; Fayad, R.; Wilcox, S.; Frongillo, E.A.; Hébert, J.R. Randomization to plant-based dietary approaches leads to larger short-term improvements in Dietary Inflammatory Index scores and macronutrient intake compared with diets that contain meat. Nutr. Res. 2015, 35, 97–106. [Google Scholar] [CrossRef]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef] [PubMed]
- De Filippo, C.; Cavalieri, D.; Di Paola, M.; Ramazzotti, M.; Poullet, J.B.; Massart, S.; Collini, S.; Pieraccini, G.; Lionetti, P. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl. Acad. Sci. USA 2010, 107, 14691–14696. [Google Scholar] [CrossRef]
- Sonnenburg, E.D.; Smits, S.A.; Tikhonov, M.; Higginbottom, S.K.; Wingreen, N.S.; Sonnenburg, J.L. Diet-induced extinctions in the gut microbiota compound over generations. Nature 2016, 529, 212–215. [Google Scholar] [CrossRef]
- Wu, G.D.; Chen, J.; Hoffmann, C.; Bittinger, K.; Chen, Y.Y.; Keilbaugh, S.A.; Bewtra, M.; Knights, D.; Walters, W.A.; Knight, R.; et al. Linking long-term dietary patterns with gut microbial enterotypes. Science 2011, 334, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Koh, A.; De Vadder, F.; Kovatcheva-Datchary, P.; Bäckhed, F. From Dietary Fiber to Host Physiology: Short-Chain Fatty Acids as Key Bacterial Metabolites. Cell 2016, 165, 1332–1345. [Google Scholar] [CrossRef]
- McDonald, D.; Hyde, E.; Debelius, J.W.; Morton, J.T.; Gonzalez, A.; Ackermann, G.; Aksenov, A.A.; Behsaz, B.; Brennan, C.; Chen, Y.; et al. American Gut: An Open Platform for Citizen Science Microbiome Research. mSystems 2018, 3, e00031-18. [Google Scholar] [CrossRef] [PubMed]
- Devkota, S.; Wang, Y.; Musch, M.W.; Leone, V.; Fehlner-Peach, H.; Nadimpalli, A.; Antonopoulos, D.A.; Jabri, B.; Chang, E.B. Dietary-fat-induced taurocholic acid promotes pathobiont expansion and colitis in Il10−/− mice. Nature 2012, 487, 104–108. [Google Scholar] [CrossRef]
- Cani, P.D.; Amar, J.; Iglesias, M.A.; Poggi, M.; Knauf, C.; Bastelica, D.; Neyrinck, A.M.; Fava, F.; Tuohy, K.M.; Chabo, C.; et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 2007, 56, 1761–1772. [Google Scholar] [CrossRef]
- Craig, W.J. Health effects of vegan diets. Am. J. Clin. Nutr. 2009, 89, 1627s–1633s. [Google Scholar] [CrossRef]
- Kastorini, C.-M.; Milionis, H.J.; Esposito, K.; Giugliano, D.; Goudevenos, J.A.; Panagiotakos, D.B. The effect of Mediterranean diet on metabolic syndrome and its components: A meta-analysis of 50 studies and 534,906 individuals. J. Am. Coll. Cardiol. 2011, 57, 1299–1313. [Google Scholar] [CrossRef]
- Tylutka, A.; Morawin, B.; Walas, Ł.; Michałek, M.; Gwara, A.; Zembron-Lacny, A. Assessment of metabolic syndrome predictors in relation to inflammation and visceral fat tissue in older adults. Sci. Rep. 2023, 13, 89. [Google Scholar] [CrossRef]
- Kraja, A.T.; Chasman, D.I.; North, K.E.; Reiner, A.P.; Yanek, L.R.; Kilpeläinen, T.O.; Smith, J.A.; Dehghan, A.; Dupuis, J.; Johnson, A.D. Pleiotropic genes for metabolic syndrome and inflammation. Mol. Genet. Metab. 2014, 112, 317–338. [Google Scholar] [CrossRef]
- Alturky, S.; Ashfaq, Y.; Elhance, A.; Barney, M.; Wadiwala, I.; Hunter, A.K.; Nguyen, K.P. Association of post-thrombotic syndrome with metabolic syndrome and inflammation-a systematic review. Front. Immunol. 2025, 16, 1519534. [Google Scholar] [CrossRef] [PubMed]
- Jeitler, M.; Storz, M.A.; Steckhan, N.; Matthiae, D.; Dressler, J.; Hanslian, E.; Koppold, D.A.; Kandil, F.I.; Michalsen, A.; Kessler, C.S. Knowledge, attitudes and application of critical nutrient supplementation in vegan diets among healthcare professionals—Survey results from a medical congress on plant-based nutrition. Foods 2022, 11, 4033. [Google Scholar] [CrossRef] [PubMed]
- Richter, M.; Boeing, H.; Grünewald-Funk, D.; Heseker, H.; Kroke, A.; Leschik-Bonnet, E.; Oberritter, H.; Strohm, D.; Watzl, B. Vegan diet. Position of the German nutrition society (DGE). Ernahr. Umsch. 2016, 63, 92–102. [Google Scholar]
- Wójtowicz, J.; Sajkiewicz, I.; Rudnicka, K.; Miga-Orczykowska, N.; Lemieszek, P.; Jasiuk, I.; Pustelniak, M.; Krukar, K.; Łukaszewska, E.; Kister, K. The Importance of Supplementation in Vegan and Vegetarian Diets for Adults. J. Educ. Health Sport 2024, 70, 55299. [Google Scholar] [CrossRef]
- Weikert, C.; Trefflich, I.; Menzel, J.; Obeid, R.; Longree, A.; Dierkes, J.; Meyer, K.; Herter-Aeberli, I.; Mai, K.; Stangl, G.I. Vitamin and mineral status in a vegan diet. Dtsch. Ärzteblatt Int. 2020, 117, 575. [Google Scholar] [CrossRef]
- Benham, A.J.; Gallegos, D.; Hanna, K.L.; Hannan-Jones, M.T. Vitamin B12 supplementation adequacy in Australian Vegan Study participants. Nutrients 2022, 14, 4781. [Google Scholar] [CrossRef] [PubMed]
- Selinger, E.; Kühn, T.; Procházková, M.; Anděl, M.; Gojda, J. Vitamin B12 deficiency is prevalent among Czech vegans who do not use vitamin B12 supplements. Nutrients 2019, 11, 3019. [Google Scholar] [CrossRef] [PubMed]
- Fuhrman, J.; Ferreri, D.M. Fueling the vegetarian (vegan) athlete. Curr. Sports Med. Rep. 2010, 9, 233–241. [Google Scholar] [CrossRef]
- Groufh-Jacobsen, S.; Hess, S.Y.; Aakre, I.; Folven Gjengedal, E.L.; Blandhoel Pettersen, K.; Henjum, S. Vegans, vegetarians and pescatarians are at risk of iodine deficiency in Norway. Nutrients 2020, 12, 3555. [Google Scholar] [CrossRef]
- Khodarahmi, M.; Seyedhosseini, H.; Askari, G. Effect of Low-Carbohydrate Diets on C-Reactive Protein Level in Adults: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Food Sci. Nutr. 2025, 13, e70566. [Google Scholar] [CrossRef]
- Takacs, B.; Kalea, A.Z.; Borrion, A. Menu Dilemmas: An Integrated Assessment of the Nutritional Quality, Environmental Impact, and Cost of Vegan, Vegetarian, and Meat-Based Versions of Meals. Nutrients 2025, 17, 1569. [Google Scholar] [CrossRef]
- Kahleova, H.; Sutton, M.; Maracine, C.; Nichols, D.; Monsivais, P.; Holubkov, R.; Barnard, N.D. Vegan diet and food costs among adults with overweight: A secondary analysis of a randomized clinical trial. JAMA Netw. Open 2023, 6, e2332106. [Google Scholar] [CrossRef]
- Wilson, M.J. A Cost Effectiveness Analysis Of The Nutritious Eating with Soul Study; University of South Carolina: Columbia, SC, USA, 2021. [Google Scholar]
- Pais, D.F.; Marques, A.C.; Fuinhas, J.A. The cost of healthier and more sustainable food choices: Do plant-based consumers spend more on food? Agric. Food Econ. 2022, 10, 18. [Google Scholar] [CrossRef] [PubMed]
- Bernhart, J.A.; Turner-McGrievy, G.M.; DuBois, K.E.; Sentman, C.; Rudisill, C.; Okpara, N.; Wilcox, S.; Clemons, B.D. Community Health Worker Implementation and Cost Analysis of a Plant-Based Nutrition Program. J. Nutr. Educ. Behav. 2024, 56, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.J.; Jones, P.J.; Abdullah, M.M.; Lamarche, B.; Faulkner, D.; Patel, D.; Sahye-Pudaruth, S.; Paquette, M.; Bashyam, B.; Pichika, S.C. Low-carbohydrate vegan diets in diabetes for weight loss and sustainability: A randomized controlled trial. Am. J. Clin. Nutr. 2022, 116, 1240–1250. [Google Scholar] [CrossRef] [PubMed]
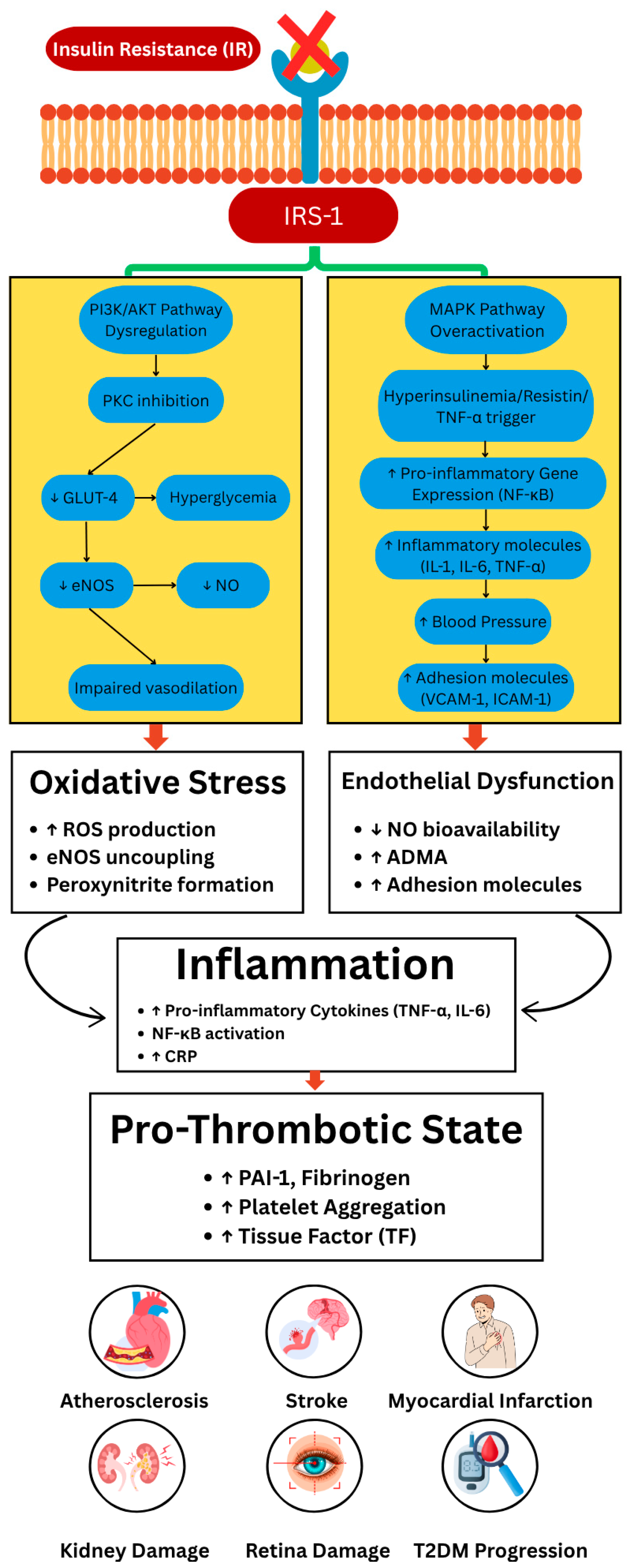
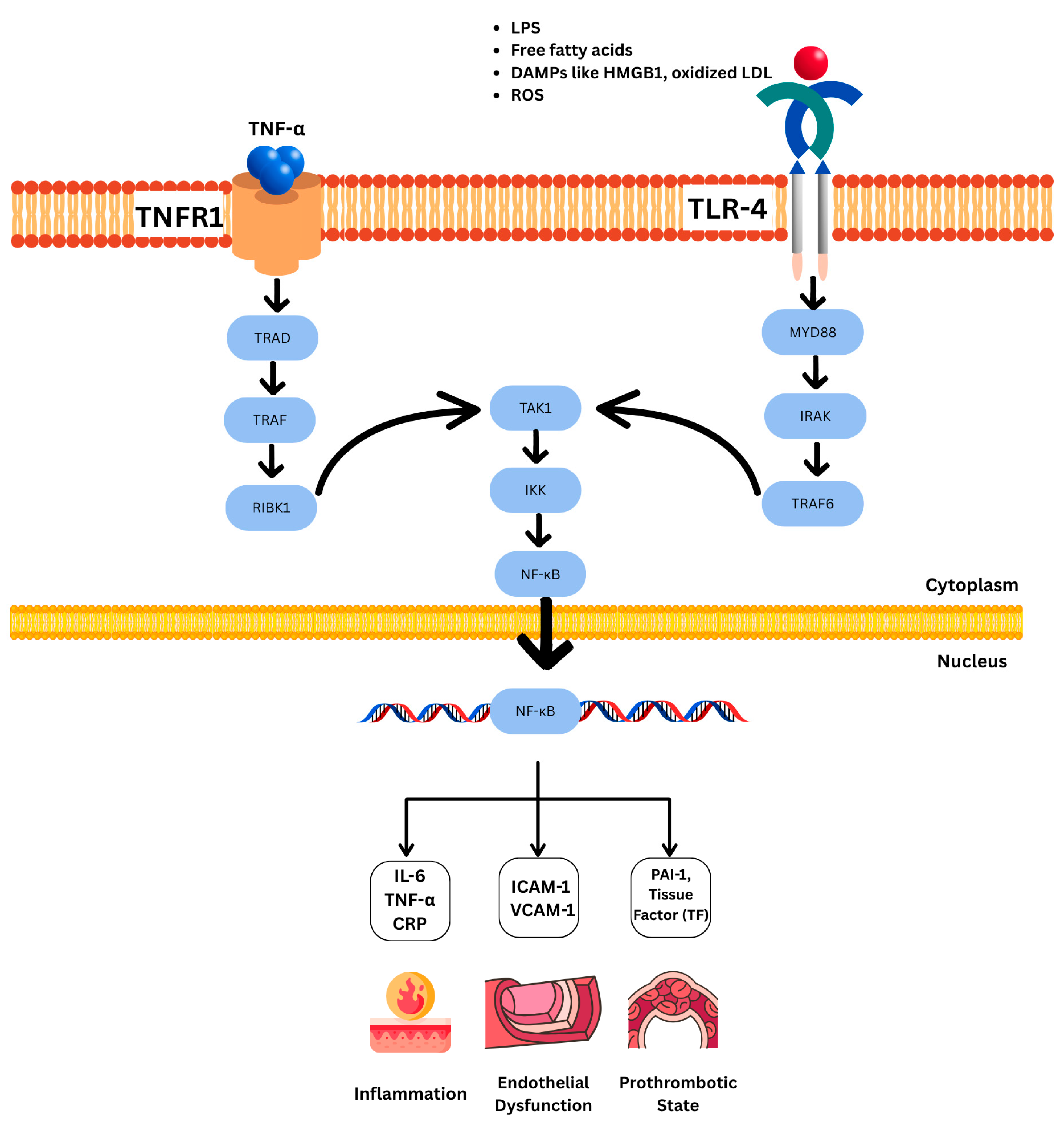
| Marker | Changes in MetS | Impact | |
|---|---|---|---|
| Coagulation Factors | Fibrinogen | Increased levels | Enhances blood viscosity and promotes clot formation |
| Factor VII | Higher activity | Accelerates thrombin generation, contributing to coagulation | |
| Factor VIII | Elevated levels | Promotes the clotting cascade and thrombin generation | |
| Platelet Activity | Enhanced aggregation | Increases thrombus formation and vascular occlusion | |
| Fibrinolysis Factors | PAI-1 | Elevated expression | Inhibits plasminogen activators, reducing fibrinolysis and increasing clot persistence |
| tPA (Tissue Plasminogen Activator) | Reduced activity | Impaired fibrin breakdown, leading to reduced fibrinolysis | |
| D-dimer | Increased concentration | Indicates heightened thrombotic activity and fibrin degradation |
| Factor | Contribution to Inflammation | Contribution to Thrombosis |
|---|---|---|
| Obesity | Increases pro-inflammatory cytokines such as IL-6 and TNF-α | Enhances PAI-1 production, impairing fibrinolysis |
| Accumulation of macrophages in adipose tissue | Raises levels of fibrinogen and coagulation factors | |
| Hypoxia in adipose tissue triggers inflammatory responses | Promotes platelet aggregation through leptin | |
| Gut dysbiosis amplifies local inflammation | ||
| Hypertension | Endothelial activation and oxidative stress | Elevates fibrinogen levels |
| Increased activity of NF-kB pathways | Raises PAI-1 expression, reducing clot breakdown | |
| Not directly addressed | Induces D-dimer elevation, reflecting higher thrombotic activity | |
| Dyslipidemia | Oxidative modification of LDL triggers inflammatory cascades | Enhances platelet hyperactivity |
| Accelerates atherosclerotic plaque formation | Increases coagulation via elevated clotting factors | |
| Insulin Resistance | Chronic low-grade inflammation due to macrophage activation | Promotes hypercoagulability via increased fibrinogen and prothrombotic factors |
| Reduces endothelial nitric oxide, impairing vascular relaxation | Alters platelet reactivity, increasing aggregation risk | |
| Enhances ROS production | Not directly addressed |
| Marker | Omnivorous Diet | Vegan Diet |
|---|---|---|
| Blood Glucose | Higher fasting glucose levels due to refined carbohydrates and saturated fats | Lower fasting glucose levels; improved insulin sensitivity from high fiber intake |
| LDL Cholesterol | Elevated LDL levels from saturated fats and cholesterol in animal products | Reduced LDL levels due to the absence of animal fats and the inclusion of phytosterols |
| HDL Cholesterol | Moderate HDL levels, influenced by dietary fats | Slightly lower HDL levels, but balanced by improved overall lipid profile |
| Triglycerides | Higher triglycerides linked to processed foods and simple sugars | Lower triglycerides due to high fiber and unsaturated fats |
| Inflammatory Markers | Elevated CRP and IL-6 levels from pro-inflammatory animal fats | Reduced CRP and IL-6 levels due to anti-inflammatory phytonutrients and omega-3s |
| Nutrient | Levels in a Vegan Diet | Levels in an Omnivorous Diet | Possible Solutions for Vegans |
|---|---|---|---|
| Vitamin B12 | Low, as it is primarily found in animal-derived products | Sufficient, due to sources such as meat, eggs, and dairy | Fortified foods (e.g., cereals and plant-based milk) and B12 supplements |
| Iron | Non-heme iron (less bioavailable) | Heme iron (more bioavailable from red meat) | Include iron-rich plant foods (e.g., lentils, tofu, and spinach) with vitamin C to enhance absorption |
| Omega-3 | Low levels of long-chain omega-3 fatty acids (EPA and DHA) | Higher levels from fish and seafood | Consume plant sources (e.g., flaxseeds, chia seeds, and walnuts) or algae-based omega-3 supplements |
| Zinc | Moderate but less bioavailable from plant sources | Sufficient, as animal products are rich in bioavailable zinc | Include zinc-rich foods (e.g., beans, nuts, and seeds) and consider zinc supplements if needed |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jafarnezhad, F.; Nazarzadeh, A.; Bazavar, H.; Keramat, S.; Ryszkiel, I.; Stanek, A. Vegan and Plant-Based Diets in the Management of Metabolic Syndrome: A Narrative Review from Anti-Inflammatory and Antithrombotic Perspectives. Nutrients 2025, 17, 2656. https://doi.org/10.3390/nu17162656
Jafarnezhad F, Nazarzadeh A, Bazavar H, Keramat S, Ryszkiel I, Stanek A. Vegan and Plant-Based Diets in the Management of Metabolic Syndrome: A Narrative Review from Anti-Inflammatory and Antithrombotic Perspectives. Nutrients. 2025; 17(16):2656. https://doi.org/10.3390/nu17162656
Chicago/Turabian StyleJafarnezhad, Fatemeh, Ata Nazarzadeh, Haniyeh Bazavar, Shayan Keramat, Ireneusz Ryszkiel, and Agata Stanek. 2025. "Vegan and Plant-Based Diets in the Management of Metabolic Syndrome: A Narrative Review from Anti-Inflammatory and Antithrombotic Perspectives" Nutrients 17, no. 16: 2656. https://doi.org/10.3390/nu17162656
APA StyleJafarnezhad, F., Nazarzadeh, A., Bazavar, H., Keramat, S., Ryszkiel, I., & Stanek, A. (2025). Vegan and Plant-Based Diets in the Management of Metabolic Syndrome: A Narrative Review from Anti-Inflammatory and Antithrombotic Perspectives. Nutrients, 17(16), 2656. https://doi.org/10.3390/nu17162656








