Diet Diversity and Adherence to a Mediterranean Diet Pattern in Pregnancy Is Protective Against the Development of Early-Childhood Atopic Dermatitis
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Definitions
Food Frequency Questionnaire (FFQ)
2.3. Scores and Indices
2.3.1. Mediterranean Diet Score During Pregnancy
2.3.2. Maternal Diet Index (MDI)
2.3.3. Plant-Based Diet Index (PDI)
2.3.4. Atopic Dermatitis
2.4. Statistical Analysis
2.5. Pattern Analysis
3. Results
3.1. Demographic Information
3.2. Maternal Diet
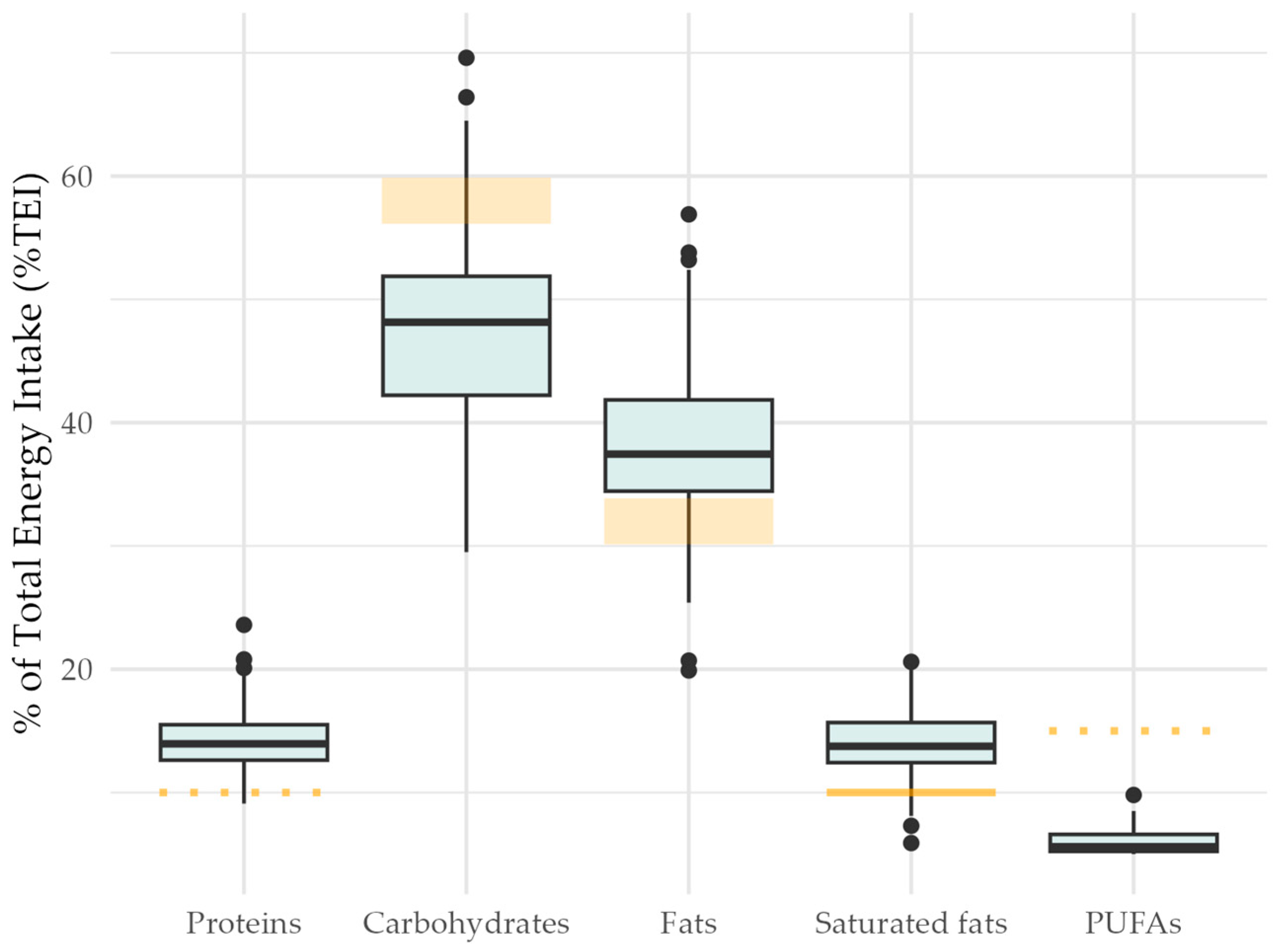
3.2.1. Macronutrients
3.2.2. Micronutrients, Minerals, and Fiber
3.3. AD in the Child
3.4. Association of Maternal Diet with AD in the Child
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hadi, H.A.; Tarmizi, A.I.; Khalid, K.A.; Gajdács, M.; Aslam, A.; Jamshed, S. The Epidemiology and Global Burden of Atopic Dermatitis: A Narrative Review. Life 2021, 11, 936. [Google Scholar] [CrossRef]
- Roduit, C.; Frei, R.; Depner, M.; Karvonen, A.M.; Renz, H.; Braun-Fahrländer, C.; Schmausser-Hechfellner, E.; Pekkanen, J.; Riedler, J.; Dalphin, J.-C.; et al. Phenotypes of Atopic Dermatitis Depending on the Timing of Onset and Progression in Childhood. JAMA Pediatr. 2017, 171, 655–662. [Google Scholar] [CrossRef]
- Brough, H.A.; Lanser, B.J.; Sindher, S.B.; Teng, J.M.C.; Leung, D.Y.M.; Venter, C.; Chan, S.M.; Santos, A.F.; Bahnson, H.T.; Guttman-Yassky, E.; et al. Early intervention and prevention of allergic diseases. Allergy 2022, 77, 416–441. [Google Scholar] [CrossRef]
- Berdyshev, E.; Kim, J.; Kim, B.E.; Goleva, E.; Lyubchenko, T.; Bronova, I.; Bronoff, A.S.; Xiao, O.; Kim, J.; Kim, S.; et al. Stratum corneum lipid and cytokine biomarkers at age 2 months predict the future onset of atopic dermatitis. J. Allergy Clin. Immunol. 2023, 151, 1307–1316. [Google Scholar] [CrossRef]
- Acevedo, N.; Alashkar Alhamwe, B.; Caraballo, L.; Ding, M.; Ferrante, A.; Garn, H.; Garssen, J.; Hii, C.S.; Irvine, J.; Llinás-Caballero, K.; et al. Perinatal and Early-Life Nutrition, Epigenetics, and Allergy. Nutrients 2021, 13, 724. [Google Scholar] [CrossRef]
- Selma-Royo, M.; García-Mantrana, I.; Calatayud, M.; Parra-Llorca, A.; Martínez-Costa, C.; Collado, M.C. Maternal diet during pregnancy and intestinal markers are associated with early gut microbiota. Eur. J. Nutr. 2021, 60, 1429–1442. [Google Scholar] [CrossRef]
- Parker, E.L.; Silverstein, R.B.; Mysorekar, I.U. Bacteria make T cell memories in utero. Cell 2021, 184, 3356–3357. [Google Scholar] [CrossRef]
- Cheng, C.; Wei, H.; Xu, C.; Xie, X.; Jiang, S.; Peng, J. Maternal Soluble Fiber Diet during Pregnancy Changes the Intestinal Microbiota, Improves Growth Performance, and Reduces Intestinal Permeability in Piglets. Appl. Environ. Microbiol. 2018, 84, e01047–e01118. [Google Scholar] [CrossRef]
- Olarini, A.; Ernst, M.; Gürdeniz, G.; Kim, M.; Brustad, N.; Bønnelykke, K.; Cohen, A.; Hougaard, D.; Lasky-Su, J.; Bisgaard, H.; et al. Vertical Transfer of Metabolites Detectable from Newborn’s Dried Blood Spot Samples Using UPLC-MS: A Chemometric Study. Metabolites 2022, 12, 94. [Google Scholar] [CrossRef]
- Brustad, N.; Olarini, A.; Kim, M.; Chen, L.; Ali, M.; Wang, T.; Cohen, A.S.; Ernst, M.; Hougaard, D.; Schoos, A.-M.; et al. Diet-associated vertically transferred metabolites and risk of asthma, allergy, eczema, and infections in early childhood. Pediatr. Allergy Immunol. 2023, 34, e13917. [Google Scholar] [CrossRef]
- Bodén, S.; Lindam, A.; Domellöf, M.; Venter, C.; West, C.E. Diet diversity in pregnancy and early allergic manifestations in the offspring. Clin. Exp. Allergy 2023, 53, 963–968. [Google Scholar] [CrossRef]
- Venter, C.; Palumbo, M.P.; Glueck, D.H.; Sauder, K.A.; O’Mahony, L.; Fleischer, D.M.; Ben-Abdallah, M.; Ringham, B.M.; Dabelea, D. The maternal diet index in pregnancy is associated with offspring allergic diseases: The Healthy Start study. Allergy 2022, 77, 162–172. [Google Scholar] [CrossRef]
- Zulyniak, M.A.; de Souza, R.J.; Shaikh, M.; Ramasundarahettige, C.; Tam, K.; Williams, N.; Desai, D.; Lefebvre, D.L.; Gupta, M.; Subbarao, P.; et al. Ethnic differences in maternal diet in pregnancy and infant eczema. PLoS ONE 2020, 15, e0232170. [Google Scholar] [CrossRef]
- Chen, L.-W.; Lyons, B.; Navarro, P.; Shivappa, N.; Mehegan, J.; Murrin, C.M.; Hébert, J.R.; Kelleher, C.C.; Phillips, C.M. Maternal dietary inflammatory potential and quality are associated with offspring asthma risk over 10-year follow-up: The Lifeways Cross-Generation Cohort Study. Am. J. Clin. Nutr. 2020, 111, 440–447. [Google Scholar] [CrossRef]
- Hanson, C.; Rifas-Shiman, S.L.; Shivappa, N.; Wirth, M.D.; Hebert, J.R.; Gold, D.; Camargo, C.A.; Sen, S.; Sordillo, J.E.; Oken, E.; et al. Associations of Prenatal Dietary Inflammatory Potential with Childhood Respiratory Outcomes in Project Viva. J. Allergy Clin. Immunol. Pract. 2020, 8, 945–952.e4. [Google Scholar] [CrossRef]
- Chatzi, L.; Torrent, M.; Romieu, I.; Garcia-Esteban, R.; Ferrer, C.; Vioque, J.; Kogevinas, M.; Sunyer, J. Mediterranean diet in pregnancy is protective for wheeze and atopy in childhood. Thorax 2008, 63, 507–513. [Google Scholar] [CrossRef]
- Nakano, K.; Kuraoka, S.; Oda, M.; Ohba, T.; Mitsubuchi, H.; Nakamura, K.; Katoh, T. The Japan Environment and Children’s Study Jecs Group, null Relationship between the Mediterranean Diet Score in Pregnancy and the Incidence of Asthma at 4 Years of Age: The Japan Environment and Children’s Study. Nutrients 2023, 15, 1772. [Google Scholar] [CrossRef]
- Saito, K.; Yokoyama, T.; Miyake, Y.; Sasaki, S.; Tanaka, T.; Ohya, Y.; Hirota, Y. Maternal meat and fat consumption during pregnancy and suspected atopic eczema in Japanese infants aged 3-4 months: The Osaka Maternal and Child Health Study. Pediatr. Allergy Immunol. 2010, 21, 38–46. [Google Scholar] [CrossRef]
- Castro-Rodriguez, J.A.; Ramirez-Hernandez, M.; Padilla, O.; Pacheco-Gonzalez, R.M.; Pérez-Fernández, V.; Garcia-Marcos, L. Effect of foods and Mediterranean diet during pregnancy and first years of life on wheezing, rhinitis and dermatitis in preschoolers. Allergol. Immunopathol. 2016, 44, 400–409. [Google Scholar] [CrossRef]
- Brzozowska, A.; Podlecka, D.; Jankowska, A.; Król, A.; Kaleta, D.; Trafalska, E.; Nowakowska-Świrta, E.; Kałużny, P.; Hanke, W.; Bal-Gierańczyk, K.; et al. Maternal diet during pregnancy and risk of allergic diseases in children up to 7–9 years old from Polish Mother and Child Cohort study. Environ. Res. 2022, 208, 112682. [Google Scholar] [CrossRef]
- Li, X.; Xiao, Z.; Li, C.; Chen, Q.; Jia, L. Maternal dietary patterns during pregnancy and the risk of infantile eczema during the first year of life: A cohort study in northeast China. BMC Public Health 2023, 23, 1641. [Google Scholar] [CrossRef] [PubMed]
- Chatzi, L.; Garcia, R.; Roumeliotaki, T.; Basterrechea, M.; Begiristain, H.; Iñiguez, C.; Vioque, J.; Kogevinas, M.; Sunyer, J.; INMA study group; et al. Mediterranean diet adherence during pregnancy and risk of wheeze and eczema in the first year of life: INMA (Spain) and RHEA (Greece) mother-child cohort studies. Br. J. Nutr. 2013, 110, 2058–2068. [Google Scholar] [CrossRef]
- Lange, N.E.; Rifas-Shiman, S.L.; Camargo, C.A.; Gold, D.R.; Gillman, M.W.; Litonjua, A.A. Maternal dietary pattern during pregnancy is not associated with recurrent wheeze in children. J. Allergy Clin. Immunol. 2010, 126, 250–255.e4. [Google Scholar] [CrossRef]
- Miyake, Y.; Okubo, H.; Sasaki, S.; Tanaka, K.; Hirota, Y. Maternal dietary patterns during pregnancy and risk of wheeze and eczema in Japanese infants aged 16-24 months: The Osaka Maternal and Child Health Study. Pediatr. Allergy Immunol. 2011, 22, 734–741. [Google Scholar] [CrossRef]
- Shaheen, S.O.; Northstone, K.; Newson, R.B.; Emmett, P.M.; Sherriff, A.; Henderson, A.J. Dietary patterns in pregnancy and respiratory and atopic outcomes in childhood. Thorax 2009, 64, 411–417. [Google Scholar] [CrossRef]
- Sivula, E.; Puharinen, H.; Hantunen, S.; Keski-Nisula, L.; Backman, K. Maternal dietary indexes are not linked to early childhood wheezing or atopic eczema. Pediatr. Allergy Immunol. 2024, 35, e14099. [Google Scholar] [CrossRef]
- Sasaki, M.; Sundberg, M.; Frei, R.; Ferstl, R.; Heye, K.N.; Willems, E.P.; Akdis, C.A.; Lauener, R.; CK-CARE Study Group; Roduit, C. Electrical impedance spectroscopy detects skin barrier dysfunction in childhood atopic dermatitis. Allergy 2024, 79, 142–152. [Google Scholar] [CrossRef]
- Sasaki, M.; Schwab, C.; Garcia, A.R.; Li, Q.; Ferstl, R.; Bersuch, E.; Akdis, C.A.; Lauener, R.; CK-CARE study group; Frei, R.; et al. The abundance of Ruminococcus bromii is associated with faecal butyrate levels and atopic dermatitis in infancy. Allergy 2022, 77, 3629–3640. [Google Scholar] [CrossRef]
- Appert, O.; Garcia, A.R.; Frei, R.; Roduit, C.; Constancias, F.; Neuzil-Bunesova, V.; Ferstl, R.; Zhang, J.; Akdis, C.; Lauener, R.; et al. Initial butyrate producers during infant gut microbiota development are endospore formers. Environ. Microbiol. 2020, 22, 3909–3921. [Google Scholar] [CrossRef]
- Morabia, A.; Bernstein, M.; Kumanyika, S.; Sorenson, A.; Mabiala, I.; Prodolliet, B.; Rolfo, I.; Luong, B.L. Development and validation of a semi-quantitative food questionnaire based on a population survey. Soz. Praventivmed. 1994, 39, 345–369. [Google Scholar] [CrossRef]
- Abreu, D.; Cardoso, I.; Gaspoz, J.-M.; Guessous, I.; Marques-Vidal, P. Trends in dietary intake in Switzerland, 1999 to 2009. Public Health Nutr. 2014, 17, 479–485. [Google Scholar] [CrossRef]
- Satija, A.; Bhupathiraju, S.N.; Spiegelman, D.; Chiuve, S.E.; Manson, J.E.; Willett, W.; Rexrode, K.M.; Rimm, E.B.; Hu, F.B. Healthful and Unhealthful Plant-Based Diets and the Risk of Coronary Heart Disease in U.S. Adults. J. Am. Coll. Cardiol. 2017, 70, 411–422. [Google Scholar] [CrossRef]
- Bédard, A.; Northstone, K.; Henderson, A.J.; Shaheen, S.O. Mediterranean diet during pregnancy and childhood respiratory and atopic outcomes: Birth cohort study. Eur. Respir. J. 2020, 55, 1901215. [Google Scholar] [CrossRef]
- Abrignani, V.; Salvo, A.; Pacinella, G.; Tuttolomondo, A. The Mediterranean Diet, Its Microbiome Connections, and Cardiovascular Health: A Narrative Review. Int. J. Mol. Sci. 2024, 25, 4942. [Google Scholar] [CrossRef]
- Amati, F.; Hassounah, S.; Swaka, A. The Impact of Mediterranean Dietary Patterns During Pregnancy on Maternal and Offspring Health. Nutrients 2019, 11, 1098. [Google Scholar] [CrossRef]
- Mazzocchi, A.; Leone, L.; Agostoni, C.; Pali-Schöll, I. The Secrets of the Mediterranean Diet. Does [Only] Olive Oil Matter? Nutrients 2019, 11, 2941. [Google Scholar] [CrossRef]
- Plöger, S.; Stumpff, F.; Penner, G.B.; Schulzke, J.-D.; Gäbel, G.; Martens, H.; Shen, Z.; Günzel, D.; Aschenbach, J.R. Microbial butyrate and its role for barrier function in the gastrointestinal tract. Ann. N. Y. Acad. Sci. 2012, 1258, 52–59. [Google Scholar] [CrossRef]
- Du, Y.; He, C.; An, Y.; Huang, Y.; Zhang, H.; Fu, W.; Wang, M.; Shan, Z.; Xie, J.; Yang, Y.; et al. The Role of Short Chain Fatty Acids in Inflammation and Body Health. Int. J. Mol. Sci. 2024, 25, 7379. [Google Scholar] [CrossRef]
- Trompette, A.; Pernot, J.; Perdijk, O.; Alqahtani, R.A.A.; Domingo, J.S.; Camacho-Munoz, D.; Wong, N.C.; Kendall, A.C.; Wiederkehr, A.; Nicod, L.P.; et al. Gut-derived short-chain fatty acids modulate skin barrier integrity by promoting keratinocyte metabolism and differentiation. Mucosal Immunol. 2022, 15, 908–926. [Google Scholar] [CrossRef]
- Schwarz, A.; Bruhs, A.; Schwarz, T. The Short-Chain Fatty Acid Sodium Butyrate Functions as a Regulator of the Skin Immune System. J. Investig. Dermatol. 2017, 137, 855–864. [Google Scholar] [CrossRef]
- Mann, E.R.; Lam, Y.K.; Uhlig, H.H. Short-chain fatty acids: Linking diet, the microbiome and immunity. Nat. Rev. Immunol. 2024, 24, 577–595. [Google Scholar] [CrossRef] [PubMed]
- Venter, C.; Pickett-Nairne, K.; Leung, D.; Fleischer, D.; O’Mahony, L.; Glueck, D.H.; Dabelea, D. Maternal allergy-preventive diet index, offspring infant diet diversity, and childhood allergic diseases. Allergy 2024, 79, 3475–3488. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Gao, Y.; Chen, W.; Hu, Q.; He, Z.; Wang, X.; Li, D.; Lin, R. Dietary variety relates to gut microbiota diversity and abundance in humans. Eur. J. Nutr. 2022, 61, 3915–3928. [Google Scholar] [CrossRef] [PubMed]
- Roduit, C.; Frei, R.; Depner, M.; Schaub, B.; Loss, G.; Genuneit, J.; Pfefferle, P.; Hyvärinen, A.; Karvonen, A.M.; Riedler, J.; et al. Increased food diversity in the first year of life is inversely associated with allergic diseases. J. Allergy Clin. Immunol. 2014, 133, 1056–1064.e7. [Google Scholar] [CrossRef]
- Chu, D.M.; Antony, K.M.; Ma, J.; Prince, A.L.; Showalter, L.; Moller, M.; Aagaard, K.M. The early infant gut microbiome varies in association with a maternal high-fat diet. Genome Med. 2016, 8, 77. [Google Scholar] [CrossRef]
- Lundgren, S.N.; Madan, J.C.; Emond, J.A.; Morrison, H.G.; Christensen, B.C.; Karagas, M.R.; Hoen, A.G. Maternal diet during pregnancy is related with the infant stool microbiome in a delivery mode-dependent manner. Microbiome 2018, 6, 109. [Google Scholar] [CrossRef]
- Hoskinson, C.; Dai, D.L.Y.; Del Bel, K.L.; Becker, A.B.; Moraes, T.J.; Mandhane, P.J.; Finlay, B.B.; Simons, E.; Kozyrskyj, A.L.; Azad, M.B.; et al. Delayed gut microbiota maturation in the first year of life is a hallmark of pediatric allergic disease. Nat. Commun. 2023, 14, 4785. [Google Scholar] [CrossRef]
- Jacobs, D.R.; Gross, M.D.; Tapsell, L.C. Food synergy: An operational concept for understanding nutrition. Am. J. Clin. Nutr. 2009, 89, 1543S–1548S. [Google Scholar] [CrossRef]
- Netting, M.J.; Middleton, P.F.; Makrides, M. Does maternal diet during pregnancy and lactation affect outcomes in offspring? A systematic review of food-based approaches. Nutrition 2014, 30, 1225–1241. [Google Scholar] [CrossRef]
- Venter, C.; Agostoni, C.; Arshad, S.H.; Ben-Abdallah, M.; Toit, G.D.; Fleischer, D.M.; Greenhawt, M.; Glueck, D.H.; Groetch, M.; Lunjani, N.; et al. Dietary factors during pregnancy and atopic outcomes in childhood: A systematic review from the European Academy of Allergy and Clinical Immunology. Pediatr. Allergy Immunol. 2020, 31, 889–912. [Google Scholar] [CrossRef]
- Méndez, L.; Medina, I. Polyphenols and Fish Oils for Improving Metabolic Health: A Revision of the Recent Evidence for Their Combined Nutraceutical Effects. Molecules 2021, 26, 2438. [Google Scholar] [CrossRef] [PubMed]
- Schneider, E.; Balasubramanian, R.; Ferri, A.; Cotter, P.D.; Clarke, G.; Cryan, J.F. Fibre & fermented foods: Differential effects on the microbiota-gut-brain axis. Proc. Nutr. Soc. 2024, 1–16. [Google Scholar] [CrossRef]
- Paparo, L.; di Costanzo, M.; di Scala, C.; Cosenza, L.; Leone, L.; Nocerino, R.; Canani, R.B. The Influence of Early Life Nutrition on Epigenetic Regulatory Mechanisms of the Immune System. Nutrients 2014, 6, 4706–4719. [Google Scholar] [CrossRef] [PubMed]
- Hogenkamp, A.; Ehlers, A.; Garssen, J.; Willemsen, L.E.M. Allergy Modulation by N-3 Long Chain Polyunsaturated Fatty Acids and Fat Soluble Nutrients of the Mediterranean Diet. Front. Pharmacol. 2020, 11, 1244. [Google Scholar] [CrossRef]
- Venter, C.; Palumbo, M.P.; Sauder, K.A.; Glueck, D.H.; O’Mahony, L.; Yang, I.; Davidson, E.J.; Brough, H.A.; Holloway, J.W.; Fleischer, D.M.; et al. Associations between child filaggrin mutations and maternal diet with the development of allergic diseases in children. Pediatr. Allergy Immunol. 2022, 33, e13753. [Google Scholar] [CrossRef]
- Coppola, S.; Paparo, L.; Bedogni, G.; Nocerino, R.; Costabile, D.; Cuomo, M.; Chiariotti, L.; Carucci, L.; Agangi, A.; Napolitano, M.; et al. Effects of Mediterranean diet during pregnancy on the onset of overweight or obesity in the offspring: A randomized trial. Int. J. Obes. 2025, 49, 101–108. [Google Scholar] [CrossRef]

| Overall n = 116 | Controls | AD 2 Years | ||
|---|---|---|---|---|
| n (%), Mean (sd) | 89 (76.7%) | 27 (23.3%) | p-Value (Healthy vs. AD) | |
| Female, n (%) | 56 (48.3) | 45 (50.6) | 11 (40.7) | 0.500 |
| Antibiotic therapy during pregnancy, n (%) | 12 (10.3) | 4 (4.5) | 8 (29.6) | 0.001 |
| Antibiotic therapy in child up to 4 months, n (%) | 7 (6.0) | 4 (4.5) | 3 (11.1) | 0.351 |
| Antibiotic therapy in child up to 1 year, n (%) | 17 (14.7) | 11 (12.4) | 6 (22.2) | 0.338 |
| Cesarean section, n (%) | 15 (12.9) | 12 (13.5) | 3 (11.1) | 1.000 |
| Mother avoided food (during pregnancy), n (%) | 6 (5.2) | 5 (5.6) | 1 (3.7) | 1.000 |
| Mother avoided food at 4 months, n (%) | 11 (9.8) | 7 (8.0) | 4 (16.0) | 0.426 |
| Child breastfed at any timepoint, n (%) | 110 (95.7) | 86 (96.6) | 24 (92.3) | 0.686 |
| Weaning, (months), mean (sd) | 7 (2.9) | 7 (2.8) | 8 (3.1) | 0.178 |
| Introduction of solids (wks), mean (sd) | 19.8 (2.9) | 19.7 (3.0) | 20.0 (2.6) | 0.668 |
| Any siblings, n (%) | 41 (35.3) | 35 (39.3) | 6 (22.2) | 0.162 |
| Pet (cat, dog, bird, rodent), n (%) | 22 (19.0) | 16(18.0) | 6 (22.2) | 0.832 |
| Mother and father atopic disease, n (%) | 28 (24.1) | 17 (19.1) | 11 (40.7) | 0.041 |
| Atopic Dermatitis Until 2 Years | |||
|---|---|---|---|
| n = 116 | n = 27 (23.3%) | ||
| Mean (SD), Median (Q1, Q3), n (%) | OR (95% CI) | Adjusted p-Value | |
| Maternal diet | |||
| Macronutrients in %TEI | |||
| Total protein | 13.95 [12.67, 15.50] | 0.60 (0.04–9.31) | 0.714 |
| Vegetal-to-animal-protein ratio | 0.51 [0.38, 0.70] | 0.96 (0.16–4.53) | 0.958 |
| Total carbohydrates | 48.15 [42.15, 52.40] | 0.69 (0.04–13.66) | 0.805 |
| Total fats | 37.40 [34.20, 42.00] | 1.43 (0.09–25.88) | 0.803 |
| Saturated fat (SFA) | 14.03 (2.83) | 1.04 (0.88–1.23) | 0.655 |
| Monounsaturated fat (MUFA) | 15.81 (3.72) | 1.03 (0.89–1.18) | 0.696 |
| Polyunsaturated fat (PUFA) | 4.90 [4.20, 5.60] | 0.93 (0.61–1.38) | 0.738 |
| Fiber and Micronutrients | |||
| Total fiber, gr | 15.13 [10.64, 20.35] | 1.36 (0.32–6.12) | 0.678 |
| Iron, mg | 9.65 (3.44) | 3.25 (0.16–83.36) | 0.458 |
| Retinol | 338.42 (225.83) | 1.00 (1.00–1.01) | 0.220 |
| Vitamin D | 1.79 (1.24) | 1.14 (0.54–2.42) | 0.733 |
| Daily multivitamins | 74 (63.8) | 2.47 (0.84–8.56) | 0.121 |
| Sweetened drinks, mL | 70.54 [25.00, 210.27] | 0.95 (0.73–1.25) | 0.698 |
| Water consumption, mL | 771.15 (325.90) | 1.01 (0.66–1.81) | 0.967 |
| Food groups | |||
| Fruit portions/day | 1.43 [0.91, 2.68] | 1.12 (0.34–3.48) | 0.849 |
| Vegetable portions/day | 1.48 [1.01, 2.13] | 0.79 (0.17–3.62) | 0.766 |
| Any nut and soy consumption | 23 (19.8) | 1.96 (0.59–6.20) | 0.255 |
| Quantity of red meat, g/day | 41.07 [16.63, 58.08] | 1.10 (0.73–1.77) | 0.668 |
| Quantity of white meat, g/day | 27.68 [13.79, 40.45] | 0.67 (0.41–1.07) | 0.094 |
| Fish, g/day | 13.39 [5.36, 25.11] | 0.80 (0.55–1.18) | 0.206 |
| Dairy products, g/day * | 2.43 [1.67, 3.34] | 0.55 (0.10–3.17) | 0.502 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Heye, K.N.; Bogl, L.H.; Sasaki, M.; Frei, R.; Breunig, A.; Bühler, N.; Kahlert, C.R.; Goekkaya, M.; Traidl-Hoffmann, C.; Lauener, R.; et al. Diet Diversity and Adherence to a Mediterranean Diet Pattern in Pregnancy Is Protective Against the Development of Early-Childhood Atopic Dermatitis. Nutrients 2025, 17, 2243. https://doi.org/10.3390/nu17132243
Heye KN, Bogl LH, Sasaki M, Frei R, Breunig A, Bühler N, Kahlert CR, Goekkaya M, Traidl-Hoffmann C, Lauener R, et al. Diet Diversity and Adherence to a Mediterranean Diet Pattern in Pregnancy Is Protective Against the Development of Early-Childhood Atopic Dermatitis. Nutrients. 2025; 17(13):2243. https://doi.org/10.3390/nu17132243
Chicago/Turabian StyleHeye, Kristina Nadine, Leonie Helen Bogl, Mari Sasaki, Remo Frei, Anna Breunig, Neeta Bühler, Christian Raphael Kahlert, Mehmet Goekkaya, Claudia Traidl-Hoffmann, Roger Lauener, and et al. 2025. "Diet Diversity and Adherence to a Mediterranean Diet Pattern in Pregnancy Is Protective Against the Development of Early-Childhood Atopic Dermatitis" Nutrients 17, no. 13: 2243. https://doi.org/10.3390/nu17132243
APA StyleHeye, K. N., Bogl, L. H., Sasaki, M., Frei, R., Breunig, A., Bühler, N., Kahlert, C. R., Goekkaya, M., Traidl-Hoffmann, C., Lauener, R., CK-CARE study group, & Roduit, C. (2025). Diet Diversity and Adherence to a Mediterranean Diet Pattern in Pregnancy Is Protective Against the Development of Early-Childhood Atopic Dermatitis. Nutrients, 17(13), 2243. https://doi.org/10.3390/nu17132243







