Flavonoids Derived from Opuntia ficus-indica Fruit Alleviate Renal Injury in Diabetic Nephropathy Mice by Altering Gut Microbiota and Promoting the Production of SCFAs
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of OFI-F
2.2. Animal Models
2.3. Drugs and Chemicals
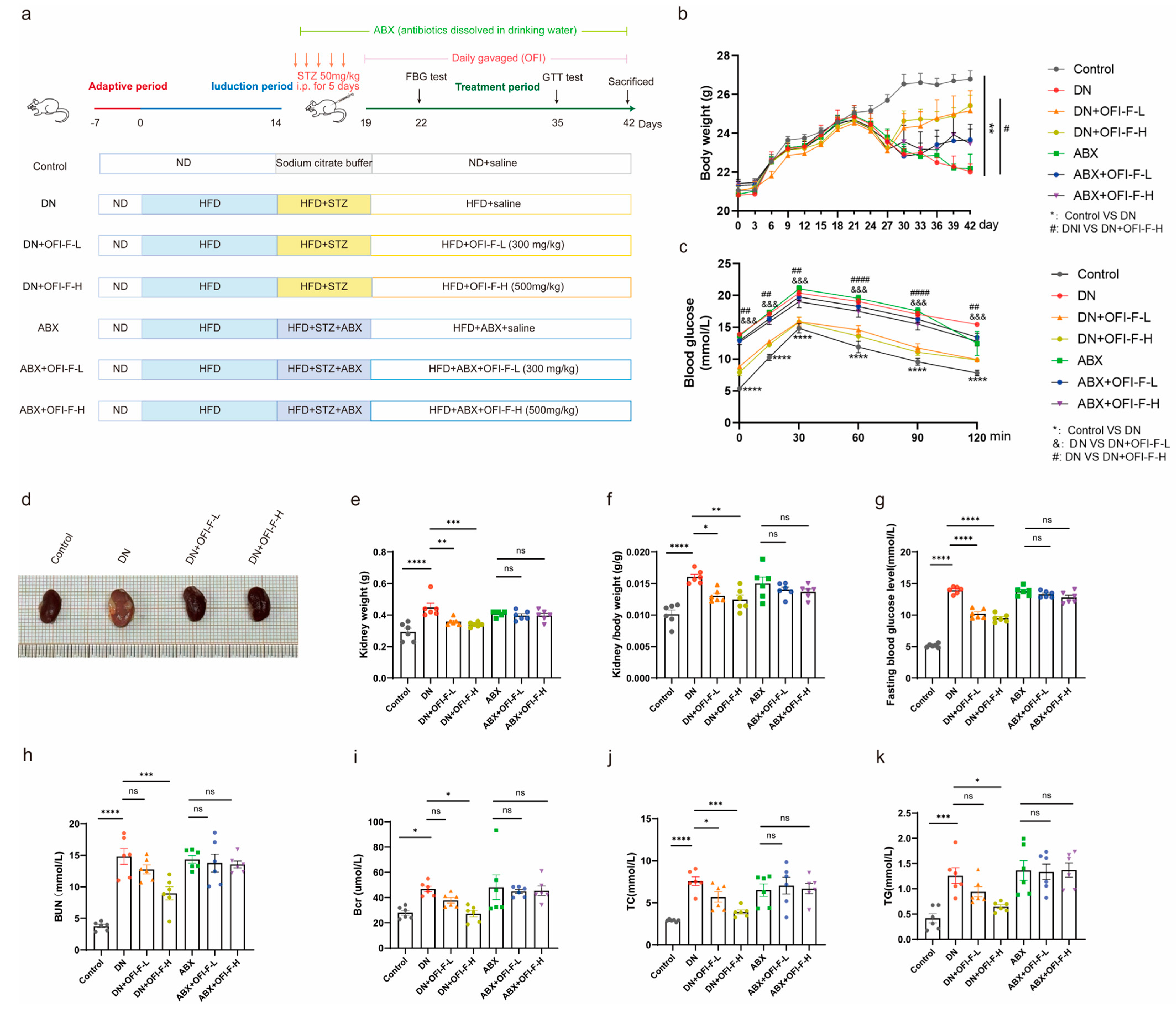
2.4. Experimental Design
2.5. Induction of DN in Mice Model
2.6. Genomics DNA Graxtraction and 16S rRNA Gene Pyrosequencing
2.7. SCFAs Measurement
2.8. Cell Culture
2.9. CCK8 (Cell Counting Kit-8) Assay
2.10. Fasting Blood Sugar and Glucose Tolerance Test (GTT)
2.11. Measurement of Bcr, BUN, TC, and TG
2.12. Measurement of MDA, CAT, SOD, and GSH
2.13. Measurement of IL-6 and TNFα
2.14. Quantitative Real-Time PCR
2.15. Western Blotting Analysis
2.16. Histology Assessment and Immunohistochemical Analysis
2.17. FITC Dextran Gut Permeability
2.18. Statistical Assessment
3. Results
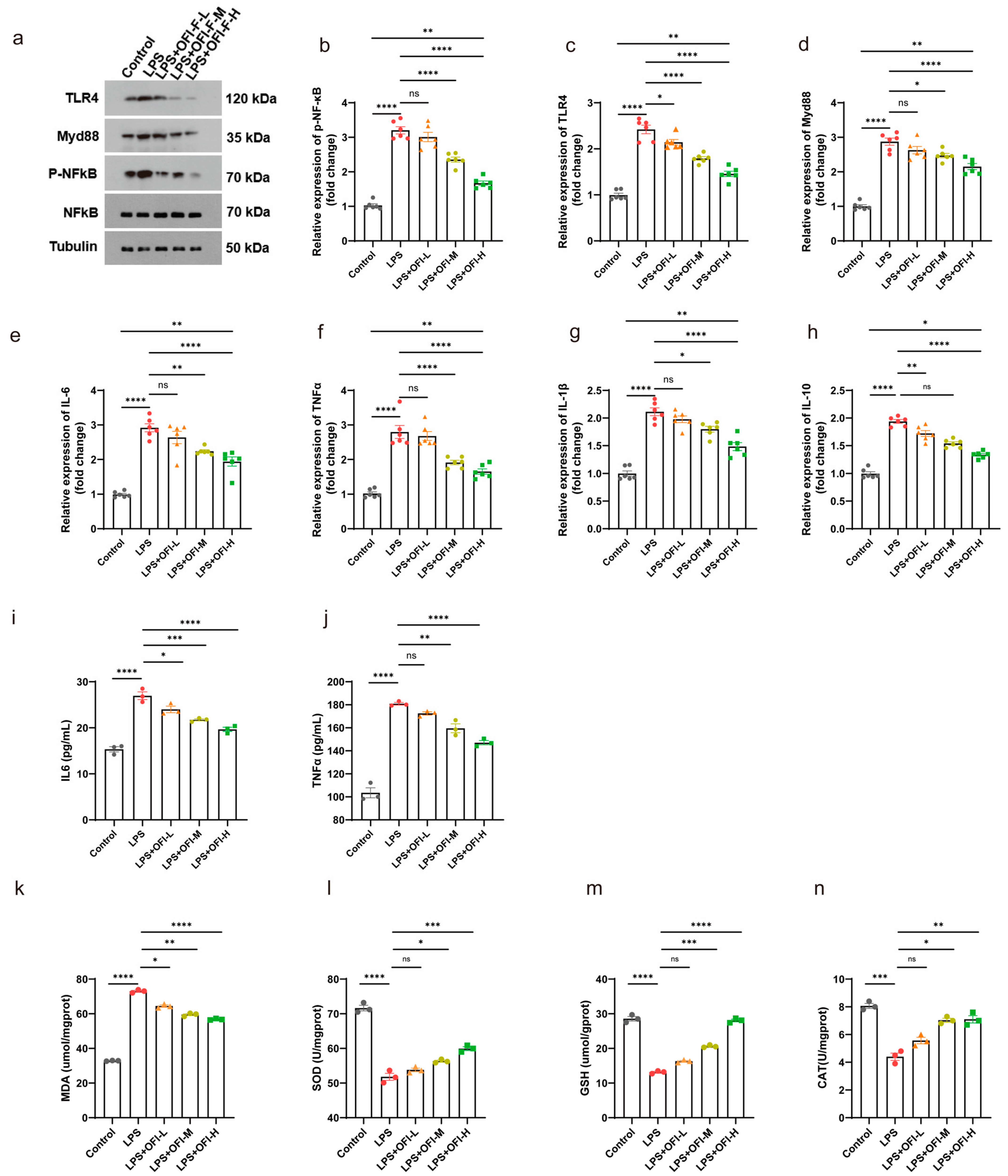
3.1. OFI-F Alleviated LPS-Induced Inflammation
3.2. OFI-F Improved the Severity of DN in Mice
3.3. OFI-F Effectively Alleviated Renal Injuries in DN Mice
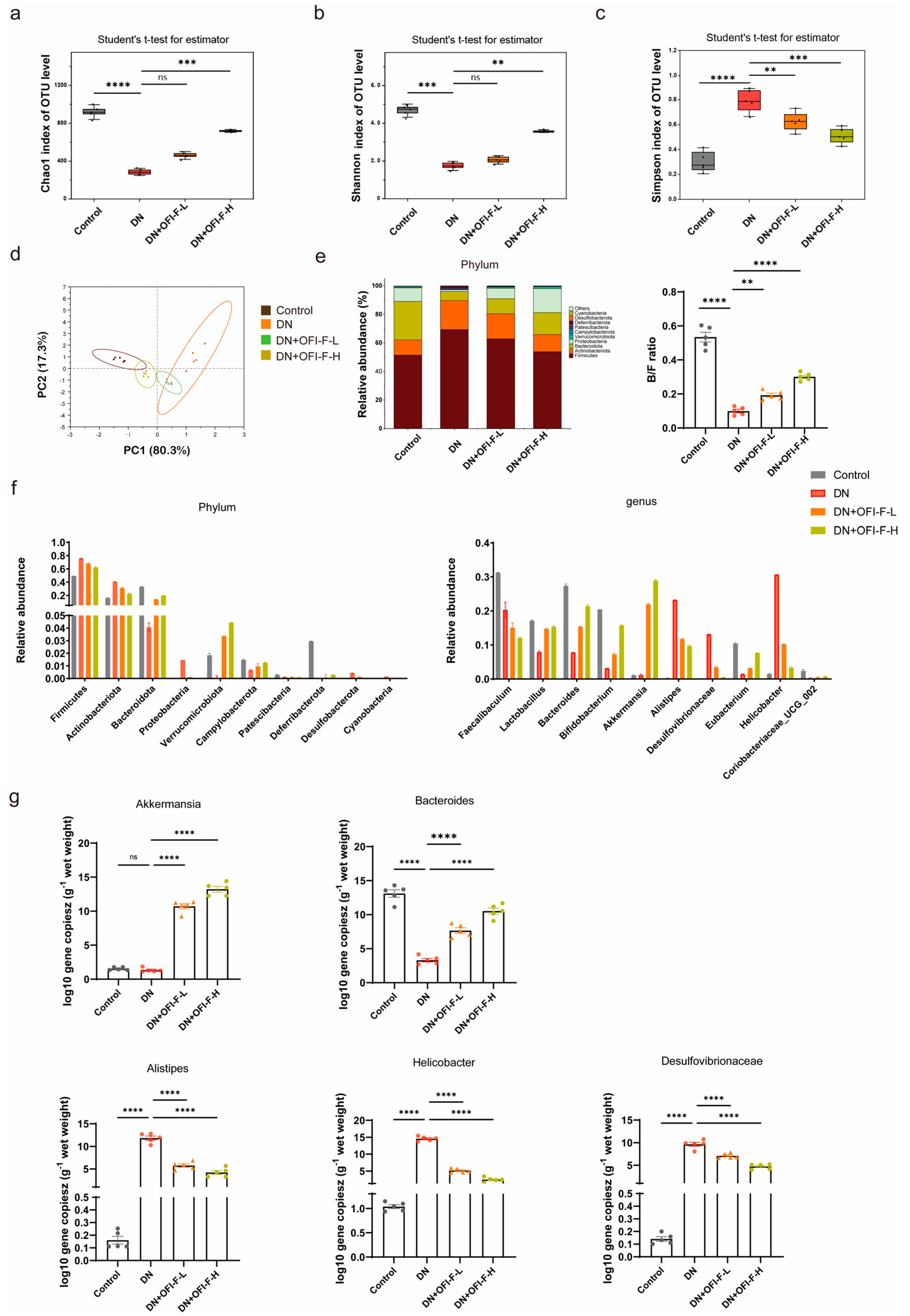
3.4. OFI-F Improved Gut Microbiota Dysbiosis in STZ/HFD-Induced DN Mice
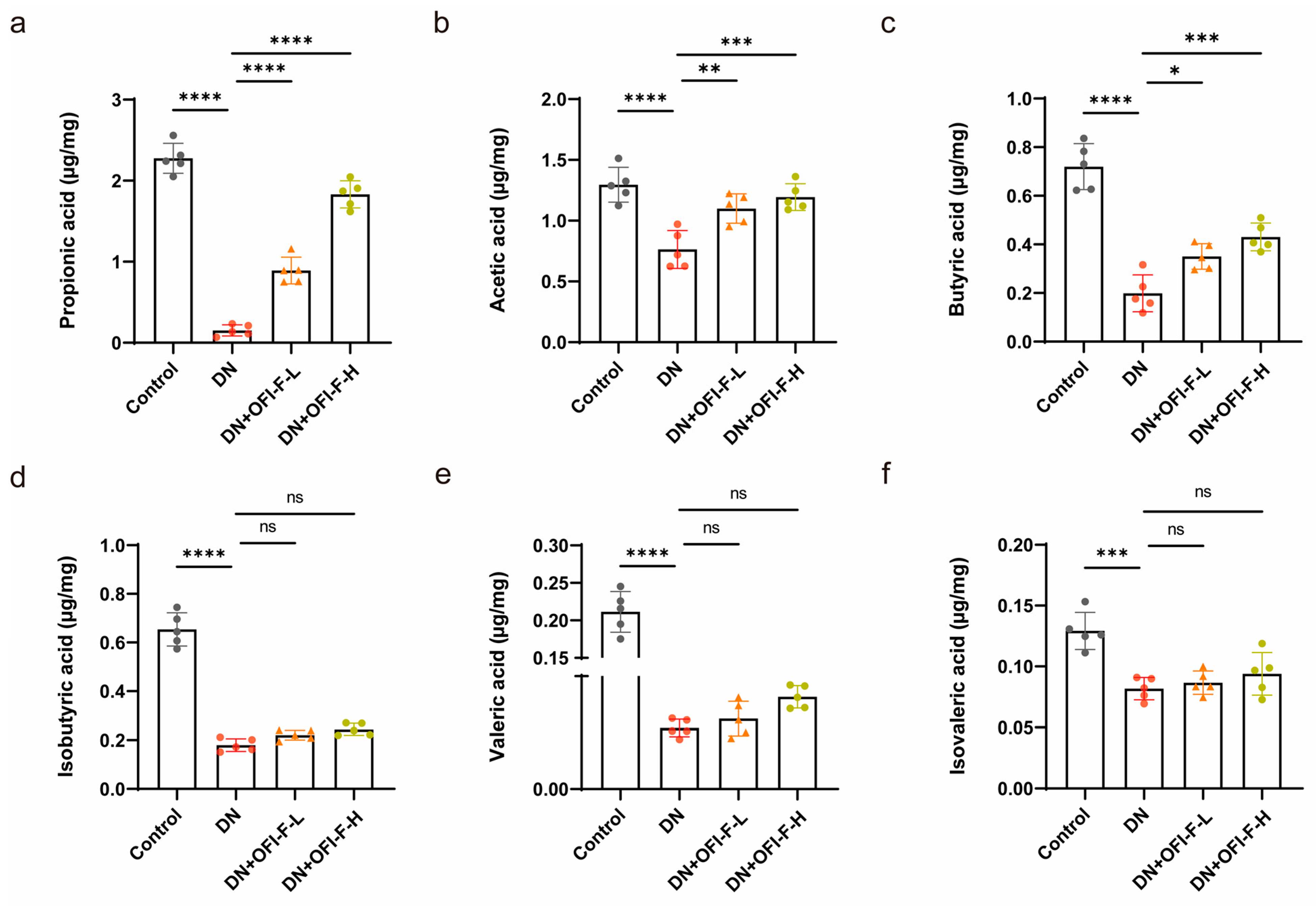
3.5. OFI-F Treatment Modulated Fecal SCFA Levels in DN Mice
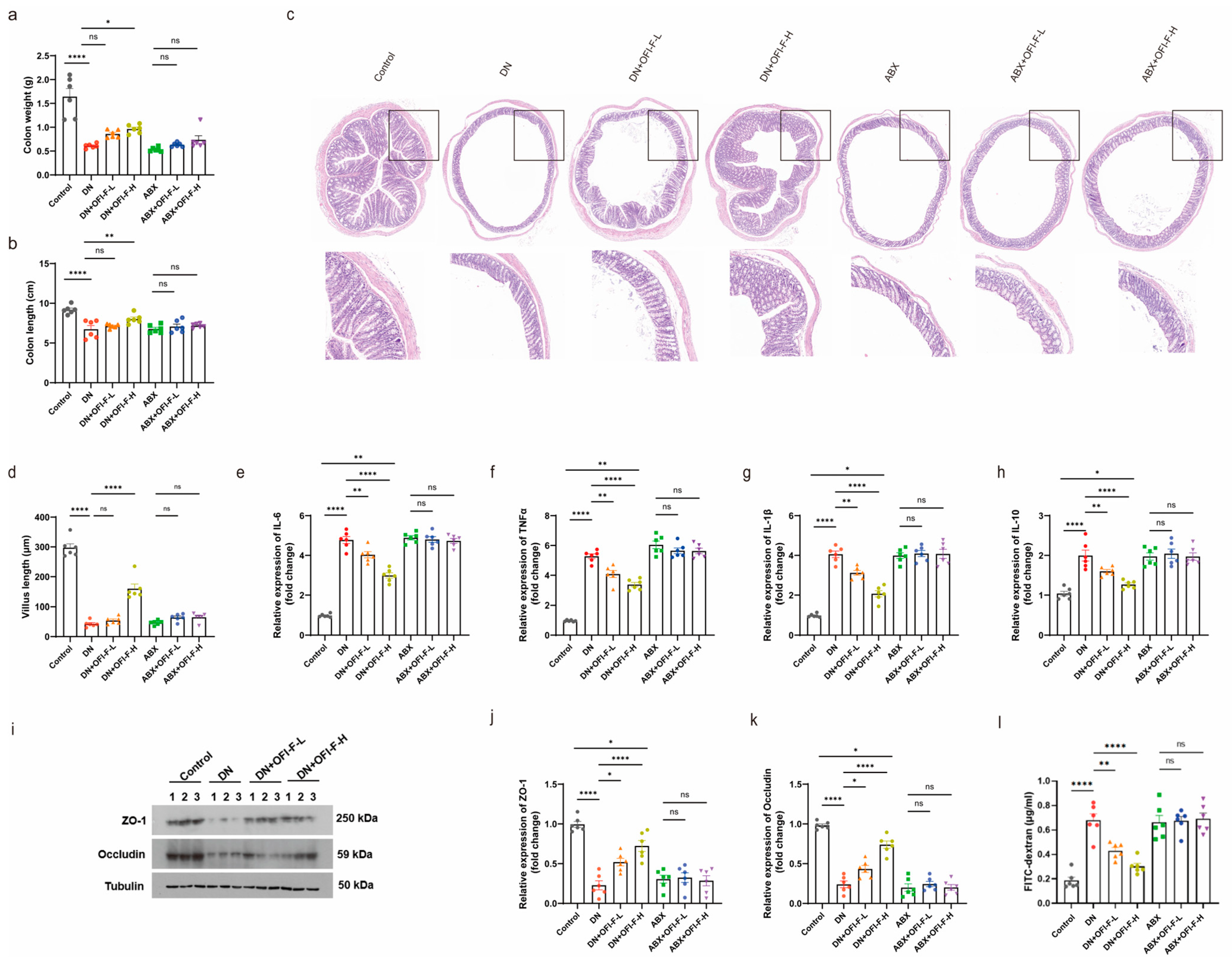
3.6. OFI-F Restored Intestinal Inflammation and Repaired Damaged Intestinal Barrier in DN Mice
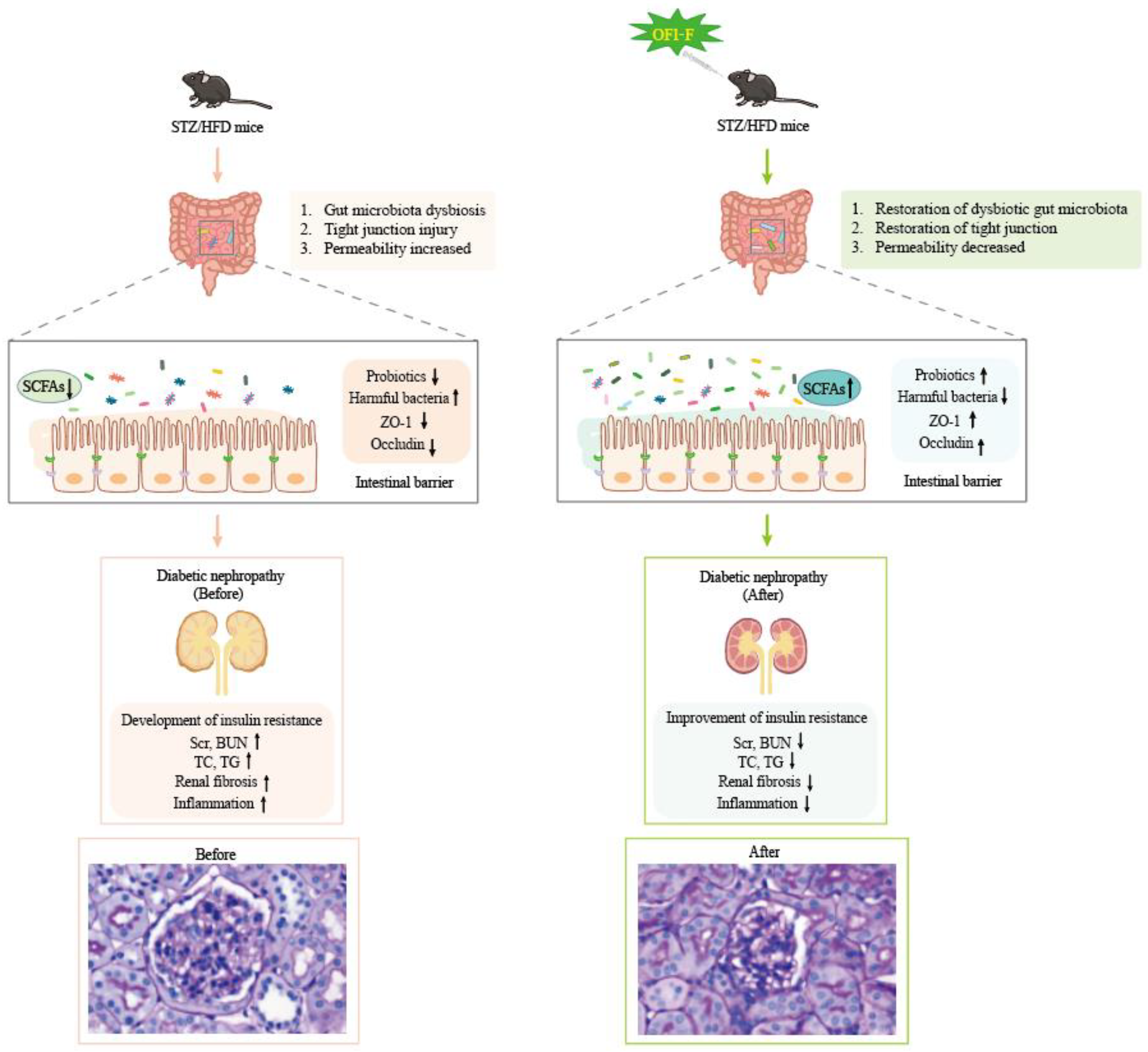
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Samsu, N. Diabetic nephropathy: Challenges in pathogenesis, diagnosis, and treatment. BioMed Res. Int. 2021, 2021, 1497449. [Google Scholar] [CrossRef] [PubMed]
- Jha, R.; Lopez-Trevino, S.; Kankanamalage, H.R.; Jha, J.C. Diabetes and renal complications: An overview on pathophysiology, biomarkers and therapeutic interventions. Biomedicines 2024, 12, 1098. [Google Scholar] [CrossRef]
- Fujii, M.; Ohno, Y.; Ikeda, A.; Godai, K.; Li, Y.; Nakamura, Y.; Yabe, D.; Tsushita, K.; Kashihara, N.; Kamide, K.; et al. Current status of the rapid decline in renal function due to diabetes mellitus and its associated factors: Analysis using the national database of health checkups in Japan. Hypertens. Res. 2023, 46, 1075–1089. [Google Scholar] [CrossRef] [PubMed]
- Natesan, V.; Kim, S.J. Diabetic nephropathy—A review of risk factors, progression, mechanism, and dietary management. Biomol. Ther. 2021, 29, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Yang, M.; Lang, Y.; Lu, S.; Kong, Z.; Gao, Y.; Shen, N.; Zhang, D.; Lv, Z. Mitochondrial metabolic reprogramming in diabetic kidney disease. Cell Death Dis. 2024, 15, 442. [Google Scholar] [CrossRef]
- Wang, Y.; Jin, M.; Cheng, C.K.; Li, Q. Tubular injury in diabetic kidney disease: Molecular mechanisms and potential therapeutic perspectives. Front. Endocrinol. 2023, 14, 1238927. [Google Scholar] [CrossRef]
- Fernandez-Fernandez, B.; Sarafidis, P.; Kanbay, M.; Navarro-González, J.F.; Soler, M.J.; Górriz, J.L.; Ortiz, A. SGLT2 inhibitors for non-diabetic kidney disease: Drugs to treat CKD that also improve glycaemia. Clin. Kidney J. 2020, 13, 728–733. [Google Scholar] [CrossRef]
- Yamazaki, T.; Mimura, I.; Tanaka, T.; Nangaku, M. Treatment of diabetic kidney disease: Current and future. Diabetes Metab. J. 2021, 45, 11–26. [Google Scholar] [CrossRef]
- Li, Y.; Liu, Y.; Liu, S.; Gao, M.; Wang, W.; Chen, K.; Huang, L.; Liu, Y. Diabetic vascular diseases: Molecular mechanisms and therapeutic strategies. Signal Transduct. Target. Ther. 2023, 8, 152. [Google Scholar] [CrossRef]
- Yu, R.; Shi, D.; Ru, Q.; Chen, Q.; Shen, J. Flavonoids from Camellia oleifera flower ameliorate type 2 diabetes mellitus by regulating the p53 pathway. Fitoterapia 2024, 179, 106267. [Google Scholar] [CrossRef]
- Sharma, B.R.; Jaiswal, S.; Ravindra, P. Modulation of gut microbiota by bioactive compounds for prevention and management of type 2 diabetes. Biomed. Pharmacother. 2022, 152, 113148. [Google Scholar] [CrossRef]
- Lin, K.; Zhu, L.; Yang, L. Gut and obesity/metabolic disease: Focus on microbiota metabolites. MedComm 2022, 3, e171. [Google Scholar] [CrossRef] [PubMed]
- Cheng, G.; Liu, Y.; Guo, R.; Wang, H.; Zhang, W.; Wang, Y. Molecular mechanisms of gut microbiota in diabetic nephropathy. Diabetes Res. Clin. Pract. 2024, 213, 111726. [Google Scholar] [CrossRef] [PubMed]
- Martins, M.; Ribeiro, M.H.; Almeida, C.M.M. Physicochemical, nutritional, and medicinal properties of Opuntia ficus-indica (L.) Mill. and its main agro-industrial use: A review. Plants 2023, 12, 1512. [Google Scholar] [CrossRef]
- Alqudah, S.M.; Hailat, M.; Zakaraya, Z.; Abu Dayah, A.A.; Abu Assab, M.; Alarman, S.M.; Awad, R.M.; Hamad, M.F.; Vicaș, L.G.; Abu Dayyih, W. Impact of Opuntia ficus-indica Juice and Empagliflozin on Glycemic Control in Rats. Curr. Issues Mol. Biol. 2024, 46, 12343–12353. [Google Scholar] [CrossRef]
- Abbas, E.Y.; Ezzat, M.I.; El Hefnawy, H.M.; Abdel-Sattar, E. An overview and update on the chemical composition and potential health benefits of Opuntia ficus-indica (L.) Miller. J. Food Biochem. 2022, 46, e14310. [Google Scholar] [CrossRef]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and other phenolic compounds from medicinal plants for pharmaceutical and medical aspects: An overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Jiang, Y.; Song, J.; Wang, S.; Lu, J.; Wei, F.; Li, X. Urinary exosomes exacerbates diabetic kidney disease by promoting NLRP3 inflammasome activation via the microRNA-516b-5p/SIRT3/AMPK pathway. Am. J. Physiol. Endocrinol. Metab. 2025. [Google Scholar] [CrossRef] [PubMed]
- Osmic-Husni, A.; Hukic, F.; Saric, M. Comparison of Jaffe Method and Enzymatic Method at Measuring Serum Creatinine Level, Creatinine Clearance and Estimated Glomerular Filtration Rate. Mater. Socio Medica 2023, 35, 113–117. [Google Scholar] [CrossRef]
- Davis, J.; Raisis, A.L.; Miller, D.W.; Rossi, G. Validation of a commercial magnetic bead-based multiplex assay for 5 novel biomarkers of acute kidney injury in canine serum. J. Vet. Diagn. Investig. 2020, 32, 656–663. [Google Scholar] [CrossRef]
- Gao, A.; Zou, J.; Zeng, T.; Qin, M.; Tang, X.; Yi, T.; Song, G.; Zhong, J.; Zeng, Y.; Zhou, W.; et al. IGF2BP3/ESM1/KLF10/BECN1 positive feedback loop: A novel therapeutic target in ovarian cancer via lipid metabolism reprogramming. Cell Death Dis. 2025, 16, 308. [Google Scholar] [CrossRef]
- Zhu, C.; Hu, W.; Wu, H.; Hu, X. No evident dose-response relationship between cellular ROS level and its cytotoxicity—A paradoxical issue in ROS-based cancer therapy. Sci. Rep. 2014, 4, 5029. [Google Scholar] [CrossRef]
- Zhang, M.; Feng, L.; Gu, J.; Ma, L.; Qin, D.; Wu, C.; Jia, X. The attenuation of Moutan Cortex on oxidative stress for renal injury in AGEs-induced mesangial cell dysfunction and streptozotocin-induced diabetic nephropathy rats. Oxid. Med. Cell. Longev. 2014, 2014, 463815. [Google Scholar] [CrossRef]
- Loh, K.P.; Qi, J.; Tan, B.K.H.; Liu, X.H.; Wei, B.G.; Zhu, Y.Z. Leonurine protects middle cerebral artery occluded rats through antioxidant effect and regulation of mitochondrial function. Stroke 2010, 41, 2661–2668. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.-Q.; Shen, M.; Zhu, C.-C.; Yu, F.-X.; Liu, Z.-Q.; Ally, N.; Sun, S.-C.; Li, K.; Liu, H.-L. 3-Nitropropionic acid induces ovarian oxidative stress and impairs follicle in mouse. PLoS ONE 2014, 9, e86589. [Google Scholar] [CrossRef]
- Liu, S.-H.; Ma, K.; Xu, X.-R.; Xu, B. A single dose of carbon monoxide intraperitoneal administration protects rat intestine from injury induced by lipopolysaccharide. Cell Stress Chaperones 2010, 15, 717–727. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Wang, W.; Yu, P.; Xi, Z.; Xu, L.; Li, X.; He, N. Comparison of taurine, GABA, Glu, and Asp as scavengers of malondialdehyde in vitro and in vivo. Nanoscale Res. Lett. 2013, 8, 190. [Google Scholar] [CrossRef] [PubMed]
- Chenxu, G.; Minxuan, X.; Yuting, Q.; Tingting, G.; Jinxiao, L.; Mingxing, W.; Sujun, W.; Yongjie, M.; Deshuai, L.; Qiang, L.; et al. iRhom2 loss alleviates renal injury in long-term PM2.5-exposed mice by suppression of inflammation and oxidative stress. Redox Biol. 2018, 19, 147–157. [Google Scholar] [CrossRef]
- Zhang, Q.; Jiang, Y.; Qin, Y.; Liu, J.; Xie, Y.; Zhang, L.; Li, K.; Wang, X.; Liu, G. Linoleic Acid Alleviates Lipopolysaccharide Induced Acute Liver Injury via Activation of Nrf2. Physiol. Res. 2024, 73, 381–391. [Google Scholar] [CrossRef]
- Tan, W.; Wang, Y.; Dai, H.; Deng, J.; Wu, Z.; Lin, L.; Yang, J. Potential Therapeutic Strategies for Renal Fibrosis: Cordyceps and Related Products. Front. Pharmacol. 2022, 13, 932172. [Google Scholar] [CrossRef]
- Thomas, M.C.; Brownlee, M.; Susztak, K.; Sharma, K.; Jandeleit-Dahm, K.A.; Zoungas, S.; Rossing, P.; Groop, P.H.; Cooper, M.E. Diabetic kidney disease. Nat. Rev. Dis. Primers 2015, 1, 15018. [Google Scholar] [CrossRef] [PubMed]
- Ueki, K.; Sasako, T.; Okazaki, Y.; Miyake, K.; Nangaku, M.; Ohashi, Y.; Noda, M.; Kadowaki, T. Multifactorial intervention has a significant effect on diabetic kidney disease in patients with type 2 diabetes. Kidney Int. 2021, 99, 256–266. [Google Scholar] [CrossRef]
- Zhang, Z.-T.; Qi, Y.; Chen, P.; Chen, L.; Jiang, Y.; Fan, Z.; Guan, H.; Bai, L.; Liu, J.; Zhao, D.; et al. Dang-Gui-Bu-Xue decoction against diabetic nephropathy via modulating the carbonyl compounds metabolic profile and AGEs/RAGE pathway. Phytomedicine 2024, 135, 156104. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.-C.; Tang, S.-Q.; Liu, Y.-T.; Li, A.-M.; Zhan, M.; Yang, M.; Song, N.; Zhang, W.; Wu, X.-Q.; Peng, C.-H.; et al. AMPK agonist alleviate renal tubulointerstitial fibrosis via activating mitophagy in high fat and streptozotocin induced diabetic mice. Cell Death Dis. 2021, 12, 925. [Google Scholar] [CrossRef] [PubMed]
- de Luca, C.; Olefsky, J.M. Inflammation and insulin resistance. FEBS Lett. 2008, 582, 97–105. [Google Scholar] [CrossRef]
- de Queiroz, J.C.F.; Alonso-Vale, M.I.; Curi, R.; Lima, F.B. Control of adipogenesis by fatty acids. Arq. Bras. Endocrinol. Metab. 2009, 53, 582–594. [Google Scholar]
- Vazquez-Carrera, M. Unraveling the Effects of PPARbeta/delta on Insulin Resistance and Cardiovascular Disease. Trends Endocrinol. Metab. 2016, 27, 319–334. [Google Scholar] [CrossRef]
- Yu, J.-X.; Chen, X.; Zang, S.-G.; Wu, Y.-Y.; Wu, L.-P.; Xuan, S.-H. Gut microbiota microbial metabolites in diabetic nephropathy patients: Far to go. Front. Cell. Infect. Microbiol. 2024, 14, 1359432. [Google Scholar] [CrossRef]
- Radjabzadeh, D.; Boer, C.G.; Beth, S.A.; van der Wal, P.; Jong, J.C.K.-D.; Jansen, M.A.E.; Konstantinov, S.R.; Peppelenbosch, M.P.; Hays, J.P.; Jaddoe, V.W.V.; et al. Diversity, compositional and functional differences between gut microbiota of children and adults. Sci. Rep. 2020, 10, 1040. [Google Scholar] [CrossRef]
- Zhao, H.; Yang, C.-E.; Liu, T.; Zhang, M.-X.; Niu, Y.; Wang, M.; Yu, J. The roles of gut microbiota and its metabolites in diabetic nephropathy. Front. Microbiol. 2023, 14, 1207132. [Google Scholar] [CrossRef]
- Li, Y.; Su, X.; Gao, Y.; Lv, C.; Gao, Z.; Liu, Y.; Wang, Y.; Li, S.; Wang, Z. The potential role of the gut microbiota in modulating renal function in experimental diabetic nephropathy murine models established in same environment. Biochim. Et Biophys. Acta-Mol. Basis Dis. 2020, 1866, 165764. [Google Scholar] [CrossRef]
- Lu, X.; Ma, J.; Li, R. Alterations of gut microbiota in biopsy-proven diabetic nephropathy and a long history of diabetes without kidney damage. Sci. Rep. 2023, 13, 12150. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Hu, J.; Geng, F.; Nie, S. Bacteroides utilization for dietary polysaccharides and their beneficial effects on gut health. Food Sci. Hum. Wellness 2022, 11, 1101–1110. [Google Scholar] [CrossRef]
- Zhou, W.; Wu, W.-H.; Si, Z.-L.; Liu, H.-L.; Wang, H.; Jiang, H.; Liu, Y.-F.; Alolga, R.N.; Chen, C.; Liu, S.-J.; et al. The gut microbe bacteroides fragilis ameliorates renal fibrosis in mice. Nat. Commun. 2022, 13, 6081. [Google Scholar] [CrossRef]
- Gao, Y.; Yang, R.; Guo, L.; Wang, Y.; Liu, W.J.; Ai, S.; Woon, T.H.; Wang, Z.; Zhai, Y.; Peng, L. Qing-Re-Xiao-Zheng formula modulates gut microbiota and inhibits inflammation in mice with diabetic kidney disease. Front. Med. 2021, 8, 719950. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Yang, H.; Wu, P.; Yang, S.; Xue, W.; Xu, B.; Zhang, S.; Tang, B.; Xu, D. Akkermansia muciniphila: A promising probiotic against inflammation and metabolic disorders. Virulence 2024, 15, 2375555. [Google Scholar] [CrossRef]
- Pei, T.; Hu, R.; Wang, F.; Yang, S.; Feng, H.; Li, Q.; Zhang, J.; Yan, S.; Ju, L.; He, Z.; et al. Akkermansia muciniphila ameliorates chronic kidney disease interstitial fibrosis via the gut-renal axis. Microb. Pathog. 2023, 174, 105891. [Google Scholar] [CrossRef]
- Xu, Y.; Duan, J.; Wang, D.; Liu, J.; Chen, X.; Qin, X.Y.; Yu, W. Akkermansia muciniphila alleviates persistent inflammation, immunosuppression, and catabolism syndrome in mice. Metabolites 2023, 13, 194. [Google Scholar] [CrossRef]
- Rodrigues, V.F.; Elias-Oliveira, J.; Pereira, Í.S.; Pereira, J.A.; Barbosa, S.C.; Machado, M.S.G.; Carlos, D. Akkermansia muciniphila and gut immune system: A good friendship that attenuates inflammatory bowel disease, obesity, and diabetes. Front. Immunol. 2022, 13, 934695. [Google Scholar] [CrossRef]
- Fu, J.; Li, G.; Li, X.; Song, S.; Cheng, L.; Rui, B.; Jiang, L. Gut commensal alistipes as a potential pathogenic factor in colorectal cancer. Discov. Oncol. 2024, 15, 473. [Google Scholar] [CrossRef]
- Parker, B.J.; Wearsch, P.A.; Veloo, A.C.M.; Rodriguez-Palacios, A. The genus Alistipes: Gut bacteria with emerging implications to inflammation, cancer, and mental health. Front. Immunol. 2020, 11, 906. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Q.-Y.; Wang, R.-L.; Wu, H.-J.; Kuang, W.-B.; Meng, W.-W.; Cheng, Z. Effect of helicobacter Pylori infection on glucose metabolism, lipid metabolism and inflammatory cytokines in nonalcoholic fatty liver disease patients. J. Multidiscip. Healthc. 2024, 17, 1127–1135. [Google Scholar] [CrossRef] [PubMed]
- Su, L.; Hong, Z.; Zhou, T.; Jian, Y.; Xu, M.; Zhang, X.; Zhu, X.; Wang, J. Health improvements of type 2 diabetic patients through diet and diet plus fecal microbiota transplantation. Sci. Rep. 2022, 12, 1152. [Google Scholar] [CrossRef] [PubMed]
- Corte-Iglesias, V.; Saiz, M.L.; Andrade-Lopez, A.C.; Salazar, N.; Bernet, C.R.; Martin-Martin, C.; Borra, J.M.; Lozano, J.-J.; Aransay, A.M.; Diaz-Corte, C.; et al. Propionate and butyrate counteract renal damage and progression to chronic kidney disease. Nephrol. Dial. Transplant. 2024, 40, 133–150. [Google Scholar] [CrossRef] [PubMed]
- Lohia, S.; Vlahou, A.; Zoidakis, J. Microbiome in chronic kidney disease (CKD): An omics perspective. Toxins 2022, 14, 176. [Google Scholar] [CrossRef]
- Zhu, W.; Liu, Y.; Lan, Y.; Li, X.; Luo, L.; Duan, X.; Lei, M.; Liu, G.; Yang, Z.; Mai, X.; et al. Dietary vinegar prevents kidney stone recurrence via epigenetic regulations. EBioMedicine 2019, 45, 231–250. [Google Scholar] [CrossRef]
- Hashimoto, T.; Shibata, K.; Honda, K.; Nobe, K. Acetic acid treatment causes renal inflammation and chronic kidney disease in mice. J. Pharmacol. Sci. 2021, 146, 160–168. [Google Scholar] [CrossRef]
- Gustafsson, A.; Olofsson, H.; Grass, J.N. Multiorgan failure after ingestion of acetic acid. Clin. Toxicol. 2022, 60, 1379–1380. [Google Scholar] [CrossRef]
- Chen, W.-J.; Chen, Y.-T.; Ko, J.-L.; Chen, J.-Y.; Zheng, J.-Y.; Liao, J.-W.; Ou, C.-C. Butyrate modulates gut microbiota and anti-inflammatory response in attenuating cisplatin-induced kidney injury. Biomed. Pharmacother. 2024, 181, 117689. [Google Scholar] [CrossRef]
- Coccia, C.; Bonomi, F.; Cricchio, A.L.; Russo, E.; Peretti, S.; Bandini, G.; Lepri, G.; Bartoli, F.; Moggi-Pignone, A.; Guiducci, S.; et al. The potential role of butyrate in the pathogenesis and treatment of autoimmune rheumatic diseases. Biomedicines 2024, 12, 1760. [Google Scholar] [CrossRef]
- Wang, X.; Lin, B.; Nie, L.; Li, P. microRNA-20b contributes to high glucose-induced podocyte apoptosis by targeting SIRT7. Molec. Med. Rep. 2017, 16, 5667–5674. [Google Scholar] [CrossRef] [PubMed]
- Carollo, C.; Sorce, A.; Cirafici, E.; Mulè, G.; Caimi, G. Sirtuins and Resveratrol in Cardiorenal Diseases: A Narrative Review of Mechanisms and Therapeutic Potential. Nutrients 2025, 17, 1212. [Google Scholar] [CrossRef] [PubMed]

| Gene Name | Forward | Reverse |
|---|---|---|
| TNF-α | 5′-CATCTTCTCAAAATTCGAGTGACAA-3′ | 5′-TGGGAGTAGACAAGGTACAACCC-3′ |
| IL-6 | 5′- AACGATGATGCACTTGCAGA -3′ | 5′- GAGCATTGGAAATTGGGGTA -3′ |
| IL-1β | 5’-GATCCACACTCTCCAGCTGCA-3’ | 5’-CAACCAACAAGTGATATTCTCCATG-3’ |
| GADPH | 5′-TGGCAAAGTGGAGATTGTTGC-3′ | 5′-AAGATGGTGATGGGCTTCCCG-3′ |
| IL-10 | 5’-ACCTGCTCCACTGCCTTGCT-3’ | 5’-GGTTGCCAAGCCTTATCGGA-3’ |
| TLR4 | 5′-ATGGCATGGCTTACACCACC-3′ | 5′- GAGGCCAATTTTGTCTCCACA-3′ |
| MYD88 | 5′-TCATGTTCTCCATACCCTTGGT-3′ | 5′-AAACTGCGAGTGGGGTCAG-3′ |
| NF-κB | 5′-ATGGCAGACGATGATCCCTAC-3′ | 5′-TGTTGACAGTGGTATTTCTGGTG-3′ |
| COL1A1 | 5′-GAGAACCAGCAGAGCCA-3′ | 5′-GAACAAGGTGACAGAGGCATA-3′ |
| YAP | 5′-CTGGGTGCCGTTTCTCCTG-3′ | 5′-CCATGTTGTTGTCTGATCGTTG-3′ |
| TGF-β | 5′-CCGCAACAACGCCATCTA-3′ | 5′-ACCAAGGTAACGCCAGGAAT-3′ |
| ZO-1 | 5′-AGCTCATAGTTCAACACAGCCTCCAG-3′ | 5′-TTCTTCCACAGCTGAAGGACTCACAG-3′ |
| Occludin | 5′-GGACCCTGACCACTATGAAACAGACTAC-3′ | 5′-ATAGGTGGATATTCCCTGACCCAGTC-3′ |
| Bacteria Name | Forward | Reverse |
|---|---|---|
| Bacteroidota | 5′-TCCTACGGGAGGCAGCAGT-3′ | 5′-CAATCGGAGTTCTTCGTG-3′ |
| Desulfobacterota | 5′-GTCCACGCCGCCTATGGGGTGCAGCAGTG-3′ | 5′-GTCCATTGCCGGACTACAAGGGTATCTAATCCTGTTTG-3′ |
| Akkermansia | 5′-CAGCACGTGAAGGTGGGGAC-3′ | 5′-CCTTGCGGTTGGCTTCAGAT-3′ |
| Helicobacter | 5′-GCCTGGGGAGTTTATGGAGAGTTTGATCCTGGC-3′ | 5′-ATCCTCCGTAAAGGAGGTGATCCAGCCG-3′ |
| Alistipes | 5′-GCGTGGGGAGTACGGAGGATTCAAGCGTTATCC-3′ | 5′-ATCCTCCGTACTCCCCACGCTTTCGTGC-3′ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liao, H.; Zhao, Y.; Liang, Y.; Zou, K. Flavonoids Derived from Opuntia ficus-indica Fruit Alleviate Renal Injury in Diabetic Nephropathy Mice by Altering Gut Microbiota and Promoting the Production of SCFAs. Nutrients 2025, 17, 1800. https://doi.org/10.3390/nu17111800
Liao H, Zhao Y, Liang Y, Zou K. Flavonoids Derived from Opuntia ficus-indica Fruit Alleviate Renal Injury in Diabetic Nephropathy Mice by Altering Gut Microbiota and Promoting the Production of SCFAs. Nutrients. 2025; 17(11):1800. https://doi.org/10.3390/nu17111800
Chicago/Turabian StyleLiao, Haiping, Yunyi Zhao, Yongheng Liang, and Kang Zou. 2025. "Flavonoids Derived from Opuntia ficus-indica Fruit Alleviate Renal Injury in Diabetic Nephropathy Mice by Altering Gut Microbiota and Promoting the Production of SCFAs" Nutrients 17, no. 11: 1800. https://doi.org/10.3390/nu17111800
APA StyleLiao, H., Zhao, Y., Liang, Y., & Zou, K. (2025). Flavonoids Derived from Opuntia ficus-indica Fruit Alleviate Renal Injury in Diabetic Nephropathy Mice by Altering Gut Microbiota and Promoting the Production of SCFAs. Nutrients, 17(11), 1800. https://doi.org/10.3390/nu17111800





