The Antioxidant Power of a Diet May Improve the Quality of Life of Young Women with Acne Vulgaris
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Data Collection
2.2. Antioxidant Power of a Diet
2.3. Statistical Analysis
3. Results
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chen, H.; Zhang, T.C.; Yin, X.L.; Man, J.Y.; Yang, X.R.; Lu, M. Magnitude and temporal trend of acne vulgaris burden in 204 countries and territories from 1990 to 2019: An analysis from the Global Burden of Disease Study 2019. Br. J. Dermatol. 2022, 186, 673–683. [Google Scholar] [CrossRef] [PubMed]
- Heng, A.H.S.; Chew, F.T. Systematic review of the epidemiology of acne vulgaris. Sci. Rep. 2020, 10, 5754. [Google Scholar] [CrossRef] [PubMed]
- Richard, M.A.; Paul, C.; Nijsten, T.; Gisondi, P.; Salavastru, C.; Taieb, C.; Trakatelli, M.; Puig, L.; Stratigos, A. EADV burden of skin diseases project team. Prevalence of most common skin diseases in Europe: A population-based study. J. Eur. Acad. Dermatol. Venereol. 2022, 36, 1088–1096. [Google Scholar] [CrossRef]
- Zaenglein, A.L. Acne vulgaris. N. Engl. J. Med. 2018, 379, 1343–1352. [Google Scholar] [CrossRef]
- Jankowska, B.; Zujko, M.E. The effectiveness of pyruvic acid peeling in improving the quality of life of patients with acne vulgaris. J. Clin. Med. 2023, 12, 3592. [Google Scholar] [CrossRef] [PubMed]
- Morshed, A.S.M.; Noor, T.; Uddin Ahmed, M.A.; Mili, F.S.; Ikram, S.; Rahman, M.; Ahmed, S.; Uddin, M.B. Understanding the impact of acne vulgaris and associated psychological distress on self-esteem and quality of life via regression modeling with CADI, DLQI, and WHOQoL. Sci. Rep. 2023, 13, 21084. [Google Scholar] [CrossRef] [PubMed]
- Heng, A.H.S.; Say, Y.H.; Sio, Y.Y.; Ng, Y.T.; Chew, F.T. Epidemiological risk factors associated with acne vulgaris presentation, severity, and scarring in a Singapore Chinese population: A cross-sectional study. Dermatology 2022, 238, 226–235. [Google Scholar] [CrossRef]
- Baldwin, H.; Tan, J. Effects of diet on acne and its response to treatment. Am. J. Clin. Dermatol. 2021, 22, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Penso, L.; Touvier, M.; Deschasaux, M.; Szabo de Edelenyi, F.; Hercberg, S.; Ezzedine, K.; Sbidian, E. Association between adult acne and dietary behaviors: Findings from the NutriNet-Santé prospective cohort study. JAMA Dermatol. 2020, 156, 854–862. [Google Scholar] [CrossRef]
- Meixiong, J.; Ricco, C.; Vasavda, C.; Ho, B.K. Diet and acne: A systematic review. JAAD Int. 2022, 7, 95–112. [Google Scholar] [CrossRef]
- Łożyńska, K.; Głąbska, D. Association between nutritional behaviours and acne-related quality of life in a population of Polish male adolescents. Nutrients 2022, 14, 2677. [Google Scholar] [CrossRef] [PubMed]
- Bungau, A.F.; Radu, A.F.; Bungau, S.G.; Vesa, C.M.; Tit, D.M.; Endres, L.M. Oxidative stress and metabolic syndrome in acne vulgaris: Pathogenetic connections and potential role of dietary supplements and phytochemicals. Biomed. Pharmacother. 2023, 164, 115003. [Google Scholar] [CrossRef]
- Zujko, M.E.; Waśkiewicz, A.; Drygas, W.; Cicha-Mikołajczyk, A.; Zujko, K.; Szcześniewska, D.; Kozakiewicz, K.; Witkowska, A.M. Dietary habits and dietary antioxidant intake are related to socioeconomic status in Polish adults: A nationwide study. Nutrients 2020, 12, 518. [Google Scholar] [CrossRef]
- Cyuńczyk, M.; Zujko, M.E.; Jamiołkowski, J.; Zujko, K.; Łapińska, M.; Zalewska, M.; Kondraciuk, M.; Witkowska, A.M.; Kamiński, K.A. Dietary total antioxidant capacity is inversely associated with prediabetes and insulin resistance in Bialystok PLUS population. Antioxidants 2022, 11, 283. [Google Scholar] [CrossRef]
- Zujko, M.E.; Rożniata, M.; Zujko, K. Individual diet modification reduces the metabolic syndrome in patients before pharmacological treatment. Nutrients 2021, 13, 2102. [Google Scholar] [CrossRef]
- Podgórska, A.; Puścion-Jakubik, A.; Markiewicz-Żukowska, R.; Gromkowska-Kępka, K.J.; Socha, K. Acne vulgaris and intake of selected dietary nutrients—A summary of information. Healthcare 2021, 9, 668. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; An, F.; Wei, X.; Hong, M.; Lu, Y. Comparative effects of Schisandrin A, B, and C on acne-related inflammation. Inflammation 2017, 40, 2163–2172. [Google Scholar] [CrossRef] [PubMed]
- Doshi, A.; Zaheer, A.; Stiller, M.J. A comparison of current acne grading systems and proposal of a novel system. Int. J. Dermatol. 1997, 36, 416–418. [Google Scholar] [CrossRef]
- Zaenglein, A.L.; Pathy, A.L.; Schlosser, B.J.; Alikhan, A.; Baldwin, H.E.; Berson, D.S.; Bowe, W.P.; Graber, E.M.; Harper, J.C.; Kang, S.; et al. Guidelines of care for the management of acne vulgaris. J. Am. Acad. Dermatol. 2016, 74, 945–973.e33. [Google Scholar] [CrossRef]
- Biernat, E.; Stupnicki, R.; Gajewski, A.K. Międzynarodowy Kwestionariusz Aktywności Fizycznej (IPAQ)—Wersja Polska [International Physical Activity Questionnaire (IPAQ)—Polish Version]. Wychow. Fiz. Sport [Phys. Educ. Sport] 2007, 51, 47–54. (In Polish) [Google Scholar]
- Motley, R.J.; Finlay, A.Y. Practical use of a disability index in the routine management of acne. Clin. Exp. Dermatol. 1992, 17, 1–3. [Google Scholar] [CrossRef]
- Abdelrazik, Y.T.; Ali, F.M.; Salek, M.S.; Finlay, A.Y. Clinical experience and psychometric properties of the Cardiff Acne Disability Index (CADI). Br. J. Dermatol. 2021, 185, 711–724. [Google Scholar] [CrossRef]
- Finlay, A.Y.; Khan, G.K. Dermatology Life Quality Index (DLQI)—A simple practical measure for routine clinical use. Clin. Exp. Dermatol. 1994, 19, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Pavot, W.; Diener, E. Review of the satisfaction with life scale. Psychol. Assess. 1993, 5, 164–172. [Google Scholar] [CrossRef]
- Beck, A.T.; Ward, C.H.; Mendelson, M.; Mock, J.; Erbaugh, J. An inventory for measuring depression. Arch. Gen. Psychiatry 1961, 4, 561–571. [Google Scholar] [CrossRef] [PubMed]
- Zujko-Kowalska, K.; Masłowska, J.; Knaś-Dawidziuk, M.; Hamulka, J.; Zujko, M.E. Dietary antioxidants may support cosmetic treatment in patients with rosacea. Antioxidants 2024, 13, 381. [Google Scholar] [CrossRef]
- Jarosz, M.; Rychlik, E.; Stoś, K.; Charzewska, J. Normy Żywienia dla Populacji Polski i ich Zastosowanie [Nutrition Standards for the Polish Population and Their Application]; National Institute of Public Health-National Institute of Hygiene: Warsaw, Poland, 2020.
- Gupta, A.; Sharma, Y.K.; Dash, K.N.; Chaudhari, N.D.; Jethani, S. Quality of life in acne vulgaris: Relationship to clinical severity and demographic data. Indian J. Dermatol. Venereol. Leprol. 2016, 82, 292–297. [Google Scholar] [CrossRef] [PubMed]
- Dabash, D.; Salahat, H.; Awawdeh, S.; Hamadani, F.; Khraim, H.; Koni, A.A.; Zyoud, S.H. Prevalence of acne and its impact on quality of life and practices regarding self-treatment among medical students. Sci. Rep. 2024, 14, 4351. [Google Scholar] [CrossRef]
- Witkam, W.C.A.M.; Dal Belo, S.E.; Pourhamidi, S.; Raynaud, E.; Moreau, M.; Aguilar, L.; Jansen, P.W.; Nijsten, T.E.C.; Pardo, L.M. The epidemiology of acne vulgaris in a multiethnic adolescent population from Rotterdam, the Netherlands: A cross-sectional study. J. Am. Acad. Dermatol. 2024, 90, 552–560. [Google Scholar] [CrossRef]
- Capitanio, B.; Sinagra, J.L.; Ottaviani, M.; Bordignon, V.; Amantea, A.; Picardo, M. Acne and smoking. Dermato-Endocrinology 2009, 1, 129–135. [Google Scholar] [CrossRef]
- Pelle, E.; Miranda, E.P.; Fthenakis, C.; Mammone, T.; Marenus, K.; Maes, D. Cigarette smoke-induced lipid peroxidation in human skin and its inhibition by topically applied antioxidants. Skin. Pharmacol. Appl. Skin Physiol. 2002, 15, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Say, Y.H.; Heng, A.H.S.; Reginald, K.; Wong, Y.R.; The, K.F.; Rawanan Shah, S.M.; Sio, Y.Y.; Ng, Y.T.; Matta, S.A.; Pang, S.L.; et al. Modifiable and non-modifiable epidemiological risk factors for acne, acne severity and acne scarring among Malaysian Chinese: A cross-sectional study. BMC Public Health 2021, 21, 601. [Google Scholar] [CrossRef]
- Dreno, B.; Shourick, J.; Kerob, D.; Bouloc, A.; Taïeb, C. The role of exposome in acne: Results from an international patient survey. J. Eur. Acad. Dermatol. Venereol. 2020, 34, 1057–1064. [Google Scholar] [CrossRef] [PubMed]
- Ryosuke, O.; Yoshie, S.; Hiromi, A. The association between activity levels and skin moisturizing function in adults. Dermatol. Rep. 2021, 13, 8811. [Google Scholar]
- Kucharska, A.; Szmurło, A.; Sińska, B. Significance of diet in treated and untreated acne vulgaris. Adv. Dermatol. Allergol. 2016, 33, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Kowalska, H.; Sysa-Jędrzejowska, A.; Woźniacka, A. Role of diet in the aetiopathogenesis of acne. Dermatol. Rev. 2018, 105, 51–62. [Google Scholar]
- Ilaria, P.; Ersilia, T.; Nicoletta, B.; Federica, T.; Andrea, V.; Nevena, S.; Concetta, P. The role of the Western diet on atopic dermatitis: Our experience and review of the current literature. Nutrients 2023, 15, 3896. [Google Scholar] [CrossRef] [PubMed]
- Manzel, A.; Muller, D.N.; Hafler, D.A.; Erdman, S.E.; Linker, R.A.; Kleinewietfeld, M. Role of “Western diet” in inflammatory autoimmune diseases. Curr. Allergy Asthma Rep. 2014, 14, 404. [Google Scholar] [CrossRef]
- Zinöcker, M.K.; Lindseth, I.A. The Western diet–microbiome-host interaction and its role in metabolic disease. Nutrients 2018, 10, 365. [Google Scholar] [CrossRef]
- Skroza, N.; Tolino, E.; Semyonov, L.; Proietti, I.; Bernardini, N.; Nicolucci, F.; La Viola, G.; Del Prete, G.; Saulle, R.; Potenza, C.; et al. Mediterranean diet and familial dysmetabolism as factors influencing the development of acne. Scand. J. Public Health 2012, 40, 466–474. [Google Scholar] [CrossRef]
- Ah-Thiane, L.; Nguyen, J.M.; Khammari, A.; Dréno, B. Lifestyle habits and impact of the Mediterranean diet on facial acne severity in French women: A case-control study. Int. J. Women’s Dermatol. 2022, 8, e017. [Google Scholar] [CrossRef] [PubMed]
- Bertolani, M.; Rodighiero, E.; Saleri, R.; Pedrazzi, G.; Bertoli, S.; Leone, A.; Feliciani, C.; Lotti, T.; Satolli, F. The influence of Mediterranean diet in acne pathogenesis and the correlation with insulin-like growth factor-1 serum levels: Implications and results. Dermatol. Rep. 2021, 14, 9143. [Google Scholar] [CrossRef] [PubMed]
- Barrea, L.; Donnarumma, M.; Cacciapuoti, S.; Muscogiuri, G.; De Gregorio, L.; Blasio, C.; Savastano, S.; Colao, A.; Fabbrocini, G. Phase angle and Mediterranean diet in patients with acne: Two easy tools for assessing the clinical severity of disease. J. Transl. Med. 2021, 19, 171. [Google Scholar] [CrossRef] [PubMed]
- Taha, S.; Shakhshir, M.; Zyoud, S.H. Acne vulgaris and adherence to the Mediterranean diet among university students: A case-control study. J. Health Popul. Nutr. 2024, 43, 41. [Google Scholar] [CrossRef] [PubMed]
- El-Akawi, Z.; Abdel-Latif, N.; Abdul-Razzak, K. Does the plasma level of vitamins A and E affect acne condition? Clin. Exp. Dermatol. 2006, 31, 430–434. [Google Scholar] [CrossRef]
- Bowe, W.P.; Joshi, S.S.; Shalita, A.R. Diet and acne. J. Am. Acad. Dermatol. 2010, 63, 124–141. [Google Scholar] [CrossRef] [PubMed]
- Yee, B.E.; Richards, P.; Sui, J.Y.; Marsch, A.F. Serum zinc levels and efficacy of zinc treatment in acne vulgaris: A systematic review and meta-analysis. Dermatol. Ther. 2020, 33, e14252. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Takahashi, A.; Kojima, M.; Akimoto, N.; Yano, M.; Ito, A. A citrus polymethoxy flavonoid, nobiletin inhibits sebum production and sebocyte proliferation, and augments sebum excretion in hamsters. J. Investig. Dermatol. 2007, 127, 2740–2748. [Google Scholar] [CrossRef] [PubMed]
- Taylor, E.J.; Yu, Y.; Champer, J.; Kim, J. Resveratrol demonstrates antimicrobial effects against Propionibacterium acnes in vitro. Dermatol. Ther. 2014, 4, 249–257. [Google Scholar] [CrossRef]
- Telang, P.S. Vitamin C in dermatology. Indian Dermatol. Online J. 2013, 4, 143–146. [Google Scholar] [CrossRef]
- Arican, O.; Kurutas, E.B.; Sasmaz, S. Oxidative stress in patients with acne vulgaris. Mediat. Inflamm. 2005, 2005, 380–384. [Google Scholar] [CrossRef] [PubMed]
- Popa, G.L.; Mitran, C.I.; Mitran, M.I.; Tampa, M.; Matei, C.; Popa, M.I.; Georgescu, S.R. Markers of oxidative stress in patients with acne: A literature review. Life 2023, 13, 1433. [Google Scholar] [CrossRef] [PubMed]
- Wong, A.; Zhang, B.; Jiang, M.; Gong, E.; Zhang, Y.; Lee, S.W. Oxidative stress in acne vulgaris. J. Clin. Dermatol. Ther. 2016, 3, 020. [Google Scholar] [CrossRef] [PubMed]
- Melnik, B.C. Linking diet to acne metabolomics, inflammation, and comedogenesis: An update. Clin. Cosmet. Investig. Dermatol. 2015, 8, 371–388. [Google Scholar] [CrossRef] [PubMed]

| Tests Assessing the QoL | Points | Outcomes |
|---|---|---|
| CADI [21,22] (Cardiff Acne Disability Index) | 0 | No impairment |
| 1–5 | Mild impairment | |
| 6–10 | Moderate impairment | |
| 11–15 | Severe impairment | |
| DLQI [23] (Dermatology Life Quality Index) | 0–1 | No effect |
| 2–5 | Small effect | |
| 6–10 | Moderate effect | |
| 11–20 | Very large effect | |
| 21–30 | Extremely large effect | |
| SWLS [24] (Satisfaction With Life Scale) | 5–17 | Low satisfaction |
| 18–23 | Average satisfaction | |
| 24–35 | High satisfaction | |
| BDI [25] (Beck Depression Inventory) | 0–13 | No depression |
| 14–19 | Mild depression | |
| 20–28 | Moderate depression | |
| 29–63 | Severe depression |
| Dietary Components | Score 1 | Score 0 |
|---|---|---|
| DTAC/1000 kcal [mmol] | ≥Me | <Me |
| DP/1000 kcal [mg] | ≥Me | <Me |
| DPH/1000 kcal | ≥Me | <Me |
| DL/1000 kcal | ≥Me | <Me |
| β-carotene/1000 kcal [µg] | ≥Me | <Me |
| Vitamin E [mg] | ≥90% AI (≥7.2) | <90% AI (<7.2) |
| Vitamin C [mg] | ≥90% RDA (≥67.5) | <90% RDA (<67.5) |
| Iron [mg] | ≥90% RDA (≥16.2) | <90% RDA (<16.2) |
| Zinc [mg] | ≥90% RDA (≥7.2) | <90% RDA (<7.2) |
| Copper [mg] | ≥90% RDA (≥0.81) | <90% RDA (<0.81) |
| Manganese [mg] | ≥90% AI (≥1.62) | <90% AI (<1.62) |
| Selenium [µg] | ≥90% RDA (≥49.5) | <90% RDA (<49.5) |
| Overall score | Minimum 12 | Maximum 0 |
| Variable | Women |
|---|---|
| Age (year), X ± SD (range) | 23.65 ± 6.24 (18–35) |
| BMI (kg/m2), X ± SD (range) | 24.35 ± 6.22 |
| BMI, N (%) | |
| Underweight (<18.5 kg/m2) | 18 (10.91) |
| Normal (18.5–24.9 kg/m2) | 124 (75.15) |
| Overweight (25.0–29.9 kg/m2) | 23 (13.94) |
| Level of education, N (%) | |
| Higher | 48 (29.09) |
| Middle | 107 (64.85) |
| Bellow middle | 10 (6.06) |
| Working status, N (%) | |
| Working | 37 (22.42) |
| Learning | 126 (76.36) |
| Not working and learning | 2 (1.22) |
| Marital status, N (%) | |
| Married/cohabiting | 20 (12.12) |
| Single | 145 (87.88) |
| Physical activity, N (%) | |
| High level | 3 (1.81) |
| Moderate level | 90 (54.55) |
| Low level | 72 (43.64) |
| Smoking, N (%) | 43 (26.06) |
| Alcohol intake, N (%) | |
| More than once a week | 27 (16.36) |
| Once a week | 42 (25.45) |
| Occasionally or never | 96 (58.19) |
| Acne duration (years), N (%) | |
| <2 | 33 (20.00) |
| 2–5 | 87 (52.73) |
| >5 | 45 (27.27) |
| Acne severity, N (%) | |
| Mild | 61 (36.97) |
| Moderate | 89 (53.94) |
| Severe | 15 (9.09) |
| QoL Scores | Results |
|---|---|
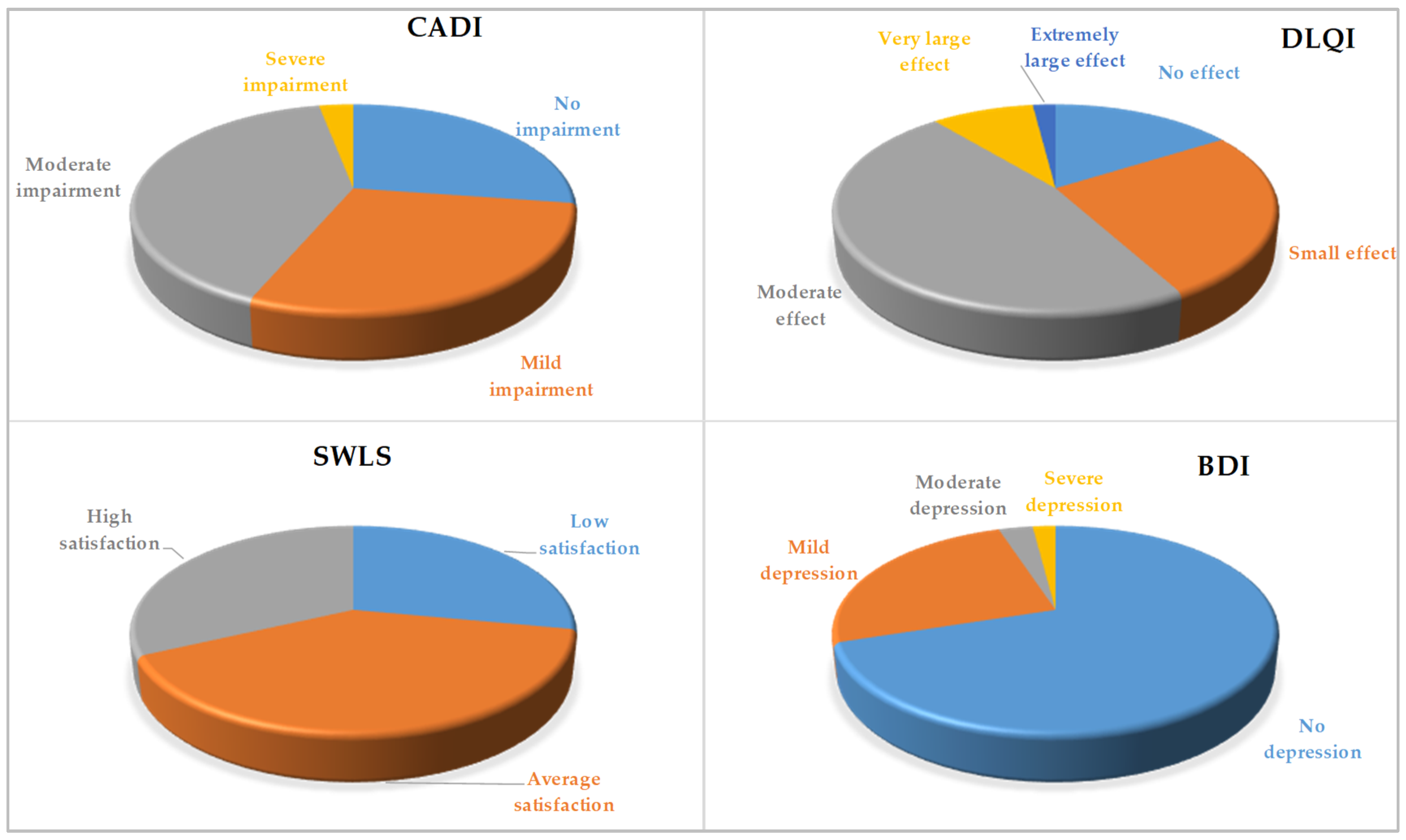
| CADI, X ± SD | 8.14 ± 4.25 |
| No impairment, N (%) | 45 (27.27) |
| Mild impairment, N (%) | 48 (29.09) |
| Moderate impairment, N (%) | 67 (40.61) |
| Severe impairment, N (%) | 5 (3.03) |
| DLQI, X ± SD | 9.52 ± 4.36 |
| No effect, N (%) | 26 (15.76) |
| Small effect, N (%) | 43 (26.06) |
| Moderate effect, N (%) | 78 (47.27) |
| Very large effect, N (%) | 15 (9.09) |
| Extremely large effect, N (%) | 3 (1.82) |
| SWLS, X ± SD | 20.32 ± 4.56 |
| Low satisfaction, N (%) | 46 (27.88) |
| Average satisfaction, N (%) | 66 (40.00) |
| High satisfaction, N (%) | 53 (32.12) |
| BDI, X ± SD | 9.75 ± 6.31 |
| No depression, N (%) | 115 (69.70) |
| Mild depression, N (%) | 42 (25.45) |
| Moderate depression, N (%) | 5 (3.03) |
| Severe depression, N (%) | 3 (1.82) |
| No. | Variables | X ± SD | Range | ≥Me, N (%) |
|---|---|---|---|---|
| Energy of diet (kcal/d.) | 1625.7 ± 583.4 | 577.4–4556.6 | - | |
| 1. | DTAC (mmol/d.) | 10.8 ± 7.2 | 4.5–34.8 | - |
| DTAC/1000 kcal | 6.5 ± 4.1 | - | 39 (23.6) | |
| 2. | DP (mg/d.) | 1715.7 ± 583.2 | 543.2–3455.4 | - |
| DP/1000 kcal | 993.8 ± 312.5 | - | 45 (27.3) | |
| 3. | DPH (mg/d.) | 223.5 ± 194.3 | 22.4–623.5 | - |
| DPH/1000 kcal | 137.2 ± 119.5 | - | 51 (30.9) | |
| 4. | DL (µg/d.) | 784.7 ± 545.2 | 27.8–1458.4 | - |
| DL/1000 kcal | 483.1 ± 335.4 | - | 49 (29.7) | |
| 5. | β-carotene (µg/d.) | 3462.4 ± 2345.2 | 135.2–6556.7 | - |
| β-carotene/1000 kcal | 2130.5 ± 1376.5 | - | 87 (52.7) | |
| Variables | X ± SD | Range | ≥90% RDA, N (%) | |
| 6. | Vitamin C (mg/d.) | 56.5 ± 36.2 | 5.2–267.5 | 42 (25.5) |
| 7. | Zinc (mg/d.) | 4.8 ± 3.5 | 1.8–21.4 | 53 (32.1) |
| 8. | Iron (mg/d.) | 7.4 ± 5.5 | 4.2–28.9 | 56 (33.9) |
| 9. | Copper (mg/d.) | 0.7 ± 0.4 | 0.2–3.5 | 68 (41.2) |
| 10. | Selenium (µg/d.) | 33.5 ± 19.3 | 11.5–154.3 | 70 (42.4) |
| Variables | X ± SD | Range | ≥90% AI, N (%) | |
| 11. | Vitamin E (mg/d.) | 6.2 ± 4.48 | 1.65–28.4 | 51 (30.9) |
| 12. | Manganese (mg/d.) | 1.4 ± 1.2 | 0.4–8.5 | 72 (43.6) |
| DAQI, X ± SD (range) | 8.2 ± 4.8 (3–12) | |||
| DAQI | p-Value | |||
|---|---|---|---|---|
| T1 (1–4) | T2 (5–8) | T3 (9–12) | ||
| Number of participants | 42 | 85 | 38 | |
| Age (years), X ± SD | 22.25 ± 6.12 | 23.27 ± 6.85 | 24.62 ± 6.24 | 0.011 |
| BMI (kg/m2) | 25.45 ± 5.43 | 24.11 ± 5.47 | 22.67 ± 4.23 | 0.009 |
| Level of education, N (%) | ||||
| Higher | 15 (35.71) | 20 (23.53) | 13 (34.21) | 0.545 |
| Middle | 22 (52.38) | 60 (70.59) | 25 (65.79) | |
| Bellow middle | 5 (11.91) | 5 (5.88) | 0 (0.00) | |
| Working status, N (%) | ||||
| Working | 10 (23.81) | 15 (17.65) | 12 (31.58) | 0.684 |
| Learning | 32 (76.19) | 68 (80.00) | 26 (68.42) | |
| Not working and learning | 0 (0.00) | 2 (2.35) | 0 (0.00) | |
| Marital status, N (%) | ||||
| Married/cohabiting | 5 (11.90) | 10 (11.76) | 5 (13.16) | 0.828 |
| Single | 37 (88.10) | 75 (88.24) | 33 (86.84) | |
| Energy of diet (kcal/d.), X ± SD | 1545.2 ± 534.1 | 1694.2 ± 545.8 | 1843.2 ± 634.5 | <0.001 |
| Physical activity, N (%) | ||||
| High | 0 (0.00) | 0 (0.00) | 3 (7.89) | <0.001 |
| Moderate | 17 (40.48) | 47 (55.29) | 26 (68.43) | |
| Low | 25 (59.52) | 38 (44.71) | 9 (23.68) | |
| Smoking, N (%) | 20 (47.62) | 18 (21.18) | 5 (13.16) | <0.001 |
| Alcohol intake, N (%) | ||||
| More than once a week | 7 (16.67) | 14 (16.47) | 6 (15.79) | 0.576 |
| Once a week | 11 (26.19) | 21 (24.71) | 10 (26.32) | |
| Occasionally | 24 (57.14) | 50 (58.82) | 22 (57.89) | |
| Acne severity, N (%) | ||||
| Mild | 15 (35.71) | 31 (36.47) | 15 (39.47) | 0.024 |
| Moderate | 22 (52.38) | 47 (55.29) | 20 (52.63) | |
| Severe | 5 (11.91) | 7 (8.24) | 3 (7.90) | |
| DAQI | |||
|---|---|---|---|
| T1 | T2 | T3 | |
| CADI | 9.72 ± 5.28 | 8.43 ± 4.24 | 7.12 ± 4.81 |
| p < 0.001 | |||
| CADI > 5 points | |||
| Model 1 [OR (95% CI)] 1 | 1 | 0.64 (0.52–1.93) | 0.59 (0.44–0.95) * |
| Model 2 [OR (95% CI)] 2 | 1 | 0.67 (0.57–1.85) | 0.62 (0.45–0.97) * |
| Model 3 [OR (95% CI)] 3 | 1 | 0.68 (0.55–1.77) | 0.70 (0.42–0.98) * |
| DLQI | 10.75 ± 6.12 | 9.29 ± 5.67 | 7.94 ± 4.83 |
| p < 0.001 | |||
| DLQI > 5 points | |||
| Model 1 [OR (95% CI)] 1 | 1 | 0.64 (0.54–1.15) | 0.61 (0.45–0.96) * |
| Model 2 [OR (95% CI)] 2 | 1 | 0.65 (0.53–1.23) | 0.63 (0.42–0.95) * |
| Model 3 [OR (95% CI)] 3 | 1 | 0.67 (0.55–1.26) | 0.68 (0.46–0.96) * |
| SWLS | 18.24 ± 5.38 | 21.2 ± 4.93 | 23.45 ± 5.83 |
| p < 0.001 | |||
| SWLS < 18 points | |||
| Model 1 [OR (95% CI)] 1 | 1 | 0.83 (0.57–1.42) | 0.94 (0.65–1.69) |
| Model 2 [OR (95% CI)] 2 | 1 | 0.89 (0.49–1.57) | 0.92 (0.62–1.72) |
| Model 3 [OR (95% CI)] 3 | 1 | 0.96 (0.64–1.68) | 0.95 (0.74–1.77) |
| BDI | 7.42 ± 4.35 | 10.21 ± 5.17 | 13.12 ± 5.85 |
| p < 0.001 | |||
| BDI > 13 points | |||
| Model 1 [OR (95% CI)] 1 | 1 | 0.59 (0.45–0.95) * | 0.61 (0.45–0.96) * |
| Model 2 [OR (95% CI)] 2 | 1 | 0.54 (0.46–1.13) | 0.63 (0.41–0.95) * |
| Model 3 [OR (95% CI)] 3 | 1 | 0.52 (0.48–1.15) | 0.67 (0.42–0.97) * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zujko-Kowalska, K.; Jankowska, B.; Zujko, M.E. The Antioxidant Power of a Diet May Improve the Quality of Life of Young Women with Acne Vulgaris. Nutrients 2024, 16, 1270. https://doi.org/10.3390/nu16091270
Zujko-Kowalska K, Jankowska B, Zujko ME. The Antioxidant Power of a Diet May Improve the Quality of Life of Young Women with Acne Vulgaris. Nutrients. 2024; 16(9):1270. https://doi.org/10.3390/nu16091270
Chicago/Turabian StyleZujko-Kowalska, Kinga, Beata Jankowska, and Małgorzata Elżbieta Zujko. 2024. "The Antioxidant Power of a Diet May Improve the Quality of Life of Young Women with Acne Vulgaris" Nutrients 16, no. 9: 1270. https://doi.org/10.3390/nu16091270
APA StyleZujko-Kowalska, K., Jankowska, B., & Zujko, M. E. (2024). The Antioxidant Power of a Diet May Improve the Quality of Life of Young Women with Acne Vulgaris. Nutrients, 16(9), 1270. https://doi.org/10.3390/nu16091270







