Gut Microbiota in Patients with Prediabetes
Abstract
1. Introduction
2. Methods
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tabák, A.G.; Herder, C.; Rathmann, W.; Brunner, E.J.; Kivimäki, M. Prediabetes: A high-risk state for diabetes development. Lancet 2012, 379, 2279–2290. [Google Scholar] [CrossRef] [PubMed]
- Williams, R.; Karuranga, S.; Malanda, B.; Saeedi, P.; Basit, A.; Besançon, S.; Bommer, C.; Esteghamati, A.; Ogurtsova, K.; Zhang, P.; et al. Global and regional estimates and projections of diabetes-related health expenditure: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res. Clin. Pract. 2020, 162, 108072. [Google Scholar] [CrossRef] [PubMed]
- Tuomilehto, J.; Lindström, J.; Eriksson, J.G.; Valle, T.T.; Hämäläinen, H.; Ilanne-Parikka, P.; Keinänen-Kiukaanniemi, S.; Laakso, M.; Louheranta, A.; Rastas, M.; et al. Finnish Diabetes Prevention Study Group. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N. Engl. J. Med. 2001, 344, 1343–1350. [Google Scholar] [CrossRef] [PubMed]
- Knowler, W.C.; Barrett-Connor, E.; Fowler, S.E.; Hamman, R.F.; Lachin, J.M.; Walker, E.A.; Nathan, D.M.; Diabetes Prevention Program Research Group. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N. Engl. J. Med. 2002, 346, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Carlsson, L.M.; Peltonen, M.; Ahlin, S.; Anveden, Å.; Bouchard, C.; Carlsson, B.; Jacobson, P.; Lönroth, H.; Maglio, C.; Näslund, I.; et al. Bariatric surgery and prevention of type 2 diabetes in Swedish obese subjects. N. Engl. J. Med. 2012, 367, 695–704. [Google Scholar] [CrossRef] [PubMed]
- Gurung, M.; Li, Z.; You, H.; Rodrigues, R.; Jump, D.B.; Morgun, A.; Shulzhenko, N. Role of gut microbiota in type 2 diabetes pathophysiology. EBiomedicine 2020, 51, 102590. [Google Scholar] [CrossRef] [PubMed]
- Gomes, A.C.; Bueno, A.A.; de Souza, R.G.; Mota, J.F. Gut microbiota, probiotics and diabetes. Nutr. J. 2014, 13, 60. [Google Scholar] [CrossRef]
- Snelson, M.; de Pasquale, C.; Ekinci, E.I.; Coughlan, M.T. Gut microbiome, prebiotics, intestinal permeability and diabetes complications. Best Pract. Res. Clin. Endocrinol. Metab. 2021, 35, 101507. [Google Scholar] [CrossRef] [PubMed]
- Tahapary, D.; Fatya, A.; Kurniawan, F.; Marcella, C.; Rinaldi, I.; Tarigan, T.; Harbuwono, D.S.; Yunir, E.; Soewondo, P.; Purnamasari, D. Increased intestinal-fatty acid binding protein in obesity-associated type 2 diabetes mellitus. PLoS ONE 2023, 18, e0279915. [Google Scholar] [CrossRef]
- Allam-Ndoul, B.; Castonguay-Paradis, S.; Veilleux, A. Gut Microbiota and Intestinal Trans-Epithelial Permeability. Int. J. Mol. Sci. 2020, 21, 6402. [Google Scholar] [CrossRef]
- Erdem, B.; Kaya, Y.; Kıran, T.R.; Yılmaz, S. An Association Between the Intestinal Permeability Biomarker Zonulin and the Development of Diabetic Retinopathy in Type II Diabetes Mellitus. Turk. J. Ophthalmol. 2023, 53, 91–96. [Google Scholar] [CrossRef]
- Tian, H.; Ge, X.; Nie, Y.; Yang, L.; Ding, C.; McFarland, L.V.; Zhang, X.; Chen, Q.; Gong, J.; Li, N. Fecal microbiota transplantation in patients with slow-transit constipation: A randomized, clinical trial. PLoS ONE 2017, 12, e0171308. [Google Scholar] [CrossRef] [PubMed]
- Vermeire, S.; Joossens, M.; Verbeke, K.; Wang, J.; Machiels, K.; Sabino, J.; Ferrante, M.; Van Assche, G.; Rutgeerts, P.; Raes, J. Donor Species Richness Determines Faecal Microbiota Transplantation Success in Inflammatory Bowel Disease. J. Crohn’s Colitis 2015, 10, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Kimura, I.; Ozawa, K.; Inoue, D.; Imamura, T.; Kimura, K.; Maeda, T.; Terasawa, K.; Kashihara, D.; Hirano, K.; Tani, T.; et al. The gut microbiota suppresses insulin-mediated fat accumulation via the short-chain fatty acid receptor GPR43. Nat. Commun. 2013, 4, 1829. [Google Scholar] [CrossRef] [PubMed]
- Qiu, B.; Liang, J.; Li, C. Effects of fecal microbiota transplantation in metabolic syndrome: A meta-analysis of randomized controlled trials. PLoS ONE 2023, 18, e0288718. [Google Scholar] [CrossRef] [PubMed]
- Apekey, T.A.; Maynard, M.J.; Kittana, M.; Kunutsor, S.K. Comparison of the Effectiveness of Low Carbohydrate Versus Low Fat Diets, in Type 2 Diabetes: Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2022, 14, 391. [Google Scholar] [CrossRef] [PubMed]
- Heiman, M.L.; Greenway, F.L. A healthy gastrointestinal microbiome is dependent on dietary diversity. Mol. Metab. 2016, 5, 317–320. [Google Scholar] [CrossRef]
- Odamaki, T.; Kato, K.; Sugahara, H.; Hashikura, N.; Takahashi, S.; Xiao, J.Z.; Abe, F.; Osawa, R. Age-related changes in gut microbiota composition from newborn to centenarian: A cross-sectional study. BMC Microbiol. 2016, 16, 90. [Google Scholar] [CrossRef]
- Rogers, M.A.M.; Aronoff, D.M. The influence of non-steroidal anti-inflammatory drugs on the gut microbiome. Clin. Microbiol. Infect. 2016, 22, e171–e179. [Google Scholar] [CrossRef]
- Zhang, N.; Ju, Z.; Zuo, T. Time for food: The impact of diet on gut microbiota and human health. Nutrition 2018, 51–52, 80–85. [Google Scholar] [CrossRef]
- Makki, K.; Deehan, E.C.; Walter, J.; Bäckhed, F. The impact of dietary fiber on gut microbiota in host health and disease. Cell Host Microbe 2018, 23, 705–715. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Tremaroli, V.; Schmidt, C.; Lundqvist, A.; Olsson, L.M.; Krämer, M.; Gummesson, A.; Perkins, R.; Bergström, G.; Bäckhed, F. The gut microbiota in prediabetes and diabetes: A population-based cross-sectional study. Cell Metab. 2020, 32, 379–390.e373. [Google Scholar] [CrossRef] [PubMed]
- Gaike, A.H.; Paul, D.; Bhute, S.S.; Dhotre, D.P.; Pande, P.; Upadhyaya, S.; Reddy, Y.; Sampath, R.; Ghosh, D.; Chandraprabha, D.; et al. The gut microbial diversity of newly diagnosed diabetics but not of prediabetics is significantly different from that of healthy nondiabetics. mSystems 2020, 5, 10–1128. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, A.; Lee, C.Y.; Lee, Y.C.; Liu, C.L.; Chen, H.K.; Li, Y.H.; Lai, L.C.; Tsai, M.H.; Ni, Y.H.; Chiu, H.M.; et al. Twnbiome: A public database of the healthy Taiwanese gut microbiome. BMC Bioinform. 2023, 24, 20231214. [Google Scholar] [CrossRef] [PubMed]
- Bui, T.P.N.; Mannerås-Holm, L.; Puschmann, R.; Wu, H.; Troise, A.D.; Nijsse, B.; Boeren, S.; Bäckhed, F.; Fiedler, D.; deVos, W.M. Conversion of dietary inositol into propionate and acetate by commensal anaerostipes associates with host health. Nat. Commun. 2021, 12, 4798. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.; Li, Y.; Cai, Z.; Li, S.; Zhu, J.; Zhang, F.; Liang, S.; Zhang, W.; Guan, Y.; Shen, D.; et al. A Metagenome-Wide Association Study of Gut Microbiota in Type 2 Diabetes. Nature 2012, 490, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.R.; Varela-Nieto, I. Diabetes and the role of inositol-containing lipids in insulin signaling. Mol. Med. 1999, 5, 505–514. [Google Scholar] [CrossRef]
- Shettigar, K.; Murali, T.S. Virulence factors and clonal diversity of Staphylococcus aureus in colonization and wound infection with emphasis on diabetic foot infection. Eur. J. Clin. Microbiol. Infect. Dis. 2020, 39, 2235–2246. [Google Scholar] [CrossRef]
- Allin, K.H.; Tremaroli, V.; Caesar, R.; Jensen, B.A.H.; Damgaard, M.T.F.; Bahl, M.I.; Licht, T.R.; Hansen, T.H.; Nielsen, T.; Dantoft, T.M.; et al. Aberrant intestinal microbiota in individuals with prediabetes. Diabetologia 2018, 61, 810–820. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, M.; Wang, S.; Han, R.; Cao, Y.; Hua, W.; Mao, Y.; Zhang, X.; Pang, X.; Wei, C.; et al. Interactions between gut microbiota, host genetics and diet relevant to development of metabolic syndromes in mice. ISME J. 2010, 4, 232–241. [Google Scholar] [CrossRef]
- Yang, J.; Yu, J. The association of diet, gut microbiota and colorectal cancer: What we eat may imply what we get. Protein Cell 2018, 9, 474–487. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Hui, H.; Cai, D. Differences in Fecal Bifidobacterium species Between Patients with Type 2 Diabetes and Healthy Individuals. Nan Fang Yi Ke Da Xue Xue Bao 2012, 32, 531–533, 564. [Google Scholar] [PubMed]
- Watanabe, M.; Sianoya, A.; Mishima, R.; Therdtatha, P.; Rodriguez, A.; Ramos, D.C.; Lee, Y.K.; Dalmacio, L.M.; Nakayama, J. Gut microbiome status of urban and rural Filipino adults in relation to diet and metabolic disorders. FEMS Microbiol. Lett. 2021, 368, fnab149. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Silva, C.; Kashani, A.; Hansen, T.H.; Pinna, N.K.; Anjana, R.M.; Dutta, A.; Saxena, S.; Støy, J.; Kampmann, U.; Nielsen, T.; et al. Trans-ethnic gut microbiota signatures of type 2 diabetes in Denmark and India. Genome Med. 2021, 13, 20210303. [Google Scholar] [CrossRef] [PubMed]
- Ganesan, K.; Chung, S.K.; Vanamala, J.; Xu, B. Causal relationship between diet-induced gut microbiota changes and diabetes: A novel strategy to transplant Faecalibacterium prausnitzii in preventing diabetes. Int. J. Mol. Sci. 2018, 19, 3720. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.D.; Qi, Q.; Wang, Z.; Usyk, M.; Sotres-Alvarez, D.; Mattei, J.; Tamez, M.; Gellman, M.D.; Daviglus, M.; Hu, F.B.; et al. The gut microbiome modifies the association between a Mediterranean diet and diabetes in USA Hispanic/Latino population. J. Clin. Endocrinol. Metab. 2022, 107, e924–e934. [Google Scholar] [CrossRef] [PubMed]
- Baothman, O.A.; Zamzami, M.A.; Taher, I.; Abubaker, J.; Abu-Farha, M. The role of gut microbiota in the development of obesity and diabetes. Lipids Health Dis. 2016, 15, 108. [Google Scholar] [CrossRef] [PubMed]
- Vrieze, A.; Van Nood, E.; Holleman, F.; Salojärvi, J.; Kootte, R.S.; Bartelsman, J.F.; Dallinga-Thie, G.M.; Ackermans, M.T.; Serlie, M.J.; Oozeer, R.; et al. Transfer of intestinal microbiota from lean donors increases insulin sensitivity in individuals with metabolic syndrome. Gastroenterology 2012, 143, e917. [Google Scholar] [CrossRef] [PubMed]
- Bhute, S.S.; Suryavanshi, M.V.; Joshi, S.M.; Yajnik, C.S.; Shouche, Y.S.; Ghaskadbi, S.S. Gut microbial diversity assessment of indian type-2-diabetics reveals alterations in eubacteria, archaea, and eukaryotes. Front. Microbiol. 2017, 8, 214. [Google Scholar] [CrossRef]
- Jia, L.; Shan, K.; Pan, L.L.; Feng, N.; Lv, Z.; Sun, Y.; Li, J.; Wu, C.; Zhang, H.; Chen, W.; et al. Clostridium butyricum CGMCC0313.1 protects against autoimmune diabetes by modulating intestinal immune homeostasis and inducing pancreatic regulatory T cells. Front. Immunol. 2017, 8, 20171019. [Google Scholar] [CrossRef]
- Cohen, T.S.; Takahashi, V.; Bonnell, J.; Tovchigrechko, A.; Chaerkady, R.; Yu, W.; Jones-Nelson, O.; Lee, Y.; Raja, R.; Hess, S.; et al. Staphylococcus aureus drives expansion of low-density neutrophils in diabetic mice. J. Clin. Investig. 2019, 129, 2133–2144. [Google Scholar] [CrossRef] [PubMed]
- Fox, T.E.; Han, X.; Kelly, S.; Merrill, A.H.; Martin, R.E.; Anderson, R.E.; Gardner, T.W.; Kester, M. Diabetes alters sphingolipid metabolism in the retina: A potential mechanism of cell death in diabetic retinopathy. Diabetes 2006, 55, 3573–3580. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.; DeCoffe, D.; Molcan, E.; Gibson, D.L. Diet-induced dysbiosis of the intestinal microbiota and the effects on immunity and disease. Nutrients 2012, 4, 1095–1119. [Google Scholar] [CrossRef] [PubMed]
- Hippe, B.; Remely, M.; Aumueller, E.; Pointner, A.; Magnet, U.; Haslberger, A.G. Faecalibacterium prausnitzii phylotypes in type two diabetic, obese, and lean control subjects. Benef. Microbes 2016, 7, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Potrykus, M.; Czaja-Stolc, S.; Stankiewicz, M.; Kaska, Ł.; Małgorzewicz, S. Intestinal Microbiota as a Contributor to Chronic Inflammation and Its Potential Modifications. Nutrients 2021, 13, 3839. [Google Scholar] [CrossRef] [PubMed]
- Schwabe, R.F.; Jobin, C. The microbiome and cancer. Nat. Rev. Cancer 2013, 13, 800–812. [Google Scholar] [CrossRef]
- Kort, R.; Notting, F.; Pirovano, W.; Sybesma, W. The butyrate-producing and spore-forming bacterial genus coprococcus as a potential biomarker for neurological disorders. Gut. Microbiome 2023, 2023, e16. [Google Scholar] [CrossRef]
- Di Vincenzo, F.; Del Gaudio, A.; Petito, V.; Lopetuso, L.R.; Scaldaferri, F. Gut microbiota, intestinal permeability, and systemic inflammation: A narrative review. Intern. Emerg. Med. 2024, 19, 275–293. [Google Scholar] [CrossRef]

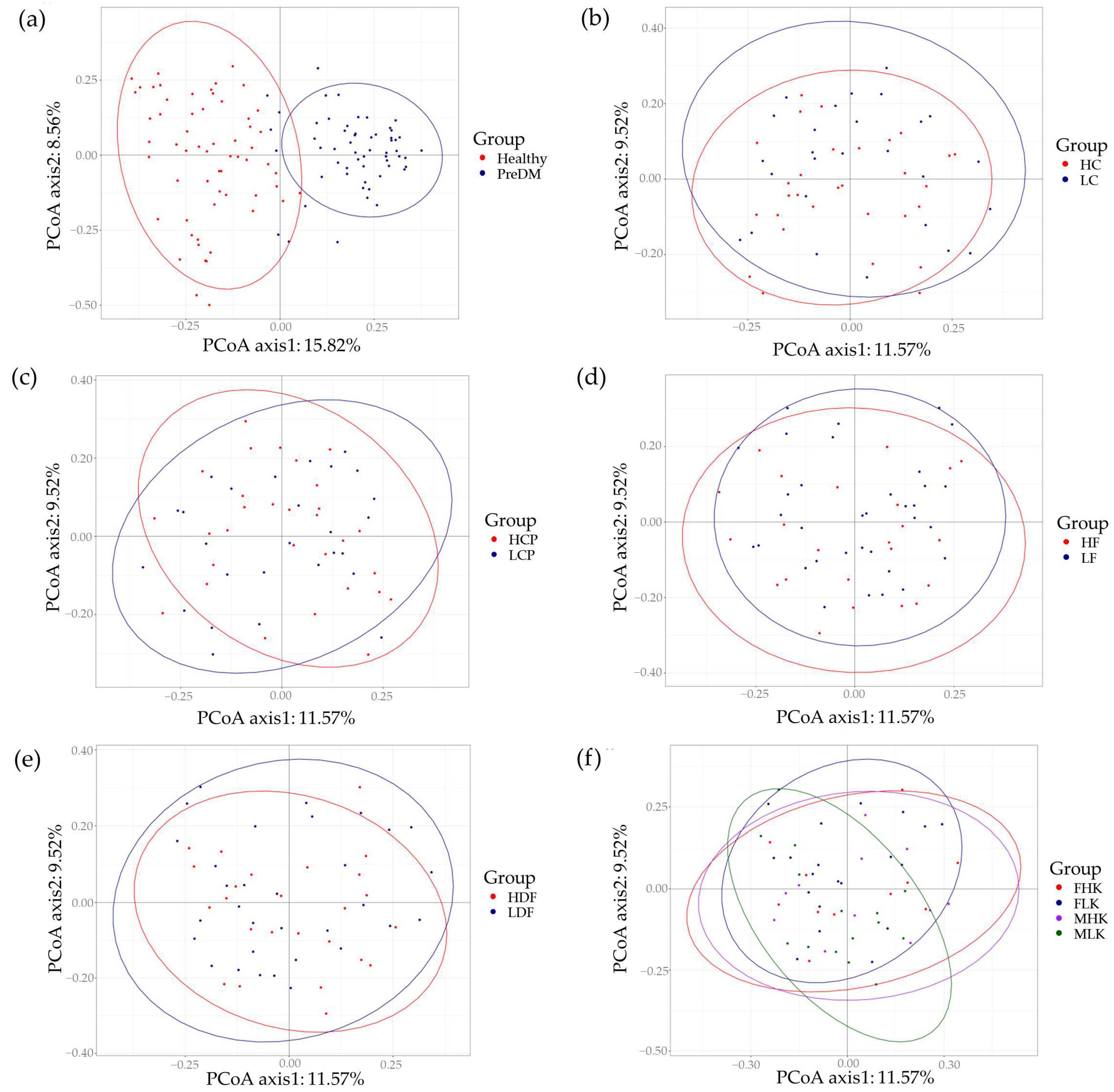
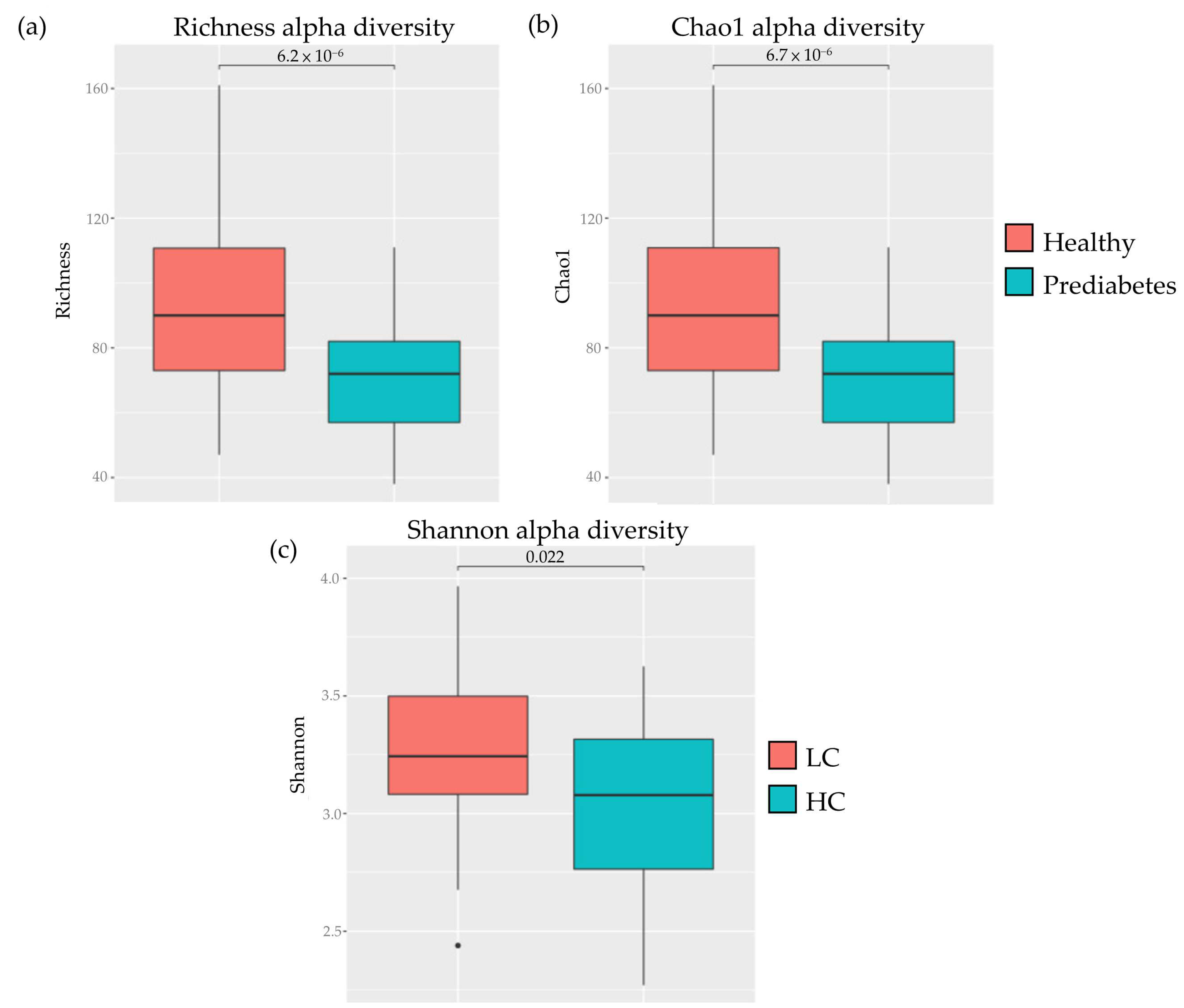
| Category | Male | Female | Total |
|---|---|---|---|
| Healthy | 25 | 35 | 60 |
| Prediabetes | 26 | 31 | 57 |
| Prediabetes group | |||
| Carbohydrate group | |||
| LC | 14 | 13 | 27 |
| HC | 12 | 18 | 30 |
| Crude Protein group | |||
| LCP | 10 | 17 | 27 |
| HCP | 16 | 14 | 30 |
| Crude Fat group | |||
| LF | 14 | 20 | 34 |
| HF | 12 | 11 | 33 |
| Dietary Fiber group | |||
| LDF | 15 | 17 | 32 |
| HDF | 11 | 14 | 25 |
| Calories group | |||
| FHK | 0 | 11 | 11 |
| FLK | 0 | 20 | 20 |
| MHK | 11 | 0 | 11 |
| MLK | 15 | 0 | 15 |
| Healthy vs. Prediabetes Group | |||||
| Healthy | |||||
| Bacterial genera | Group | LDA score | p-value | Fold change (healthy/prediabetes) * | Reference |
| Bacteroides | Healthy | 4.70 | 6.92793 × 10−8 | 0.300 | [25] |
| Phocaeicola | Healthy | 4.55 | 8.30783 × 10−8 | 0.367 | |
| Alistipes | Healthy | 4.29 | 4.65658 × 10−8 | 0.074 | |
| Phascolarctobacterium | Healthy | 4.28 | 1.14771 × 10−7 | 0.121 | [26] |
| Parabacteroides | Healthy | 3.87 | 9.24002 × 10−11 | 0.190 | [27] |
| Paraprevotella | Healthy | 3.55 | 0.001328657 | 0.133 | [28] |
| Sutterella | Healthy | 3.25 | 1.29326 × 10−5 | 0.206 | [29] |
| Ruthenibacterium | Healthy | 3.19 | 2.69626 × 10−9 | 0.078 | [30] |
| Marseillibacter | Healthy | 3.01 | 6.86676 × 10−8 | 0.049 | [31] |
| Prediabetes | |||||
| Bacterial genera | Group | LDA score | p-value | Fold change (healthy/prediabetes) * | Reference |
| Blautia | Prediabetes | 4.75 | 2.94217 × 10−20 | 8.423 | [29,32] |
| Faecalibacterium | Prediabetes | 4.34 | 4.1797 × 10−7 | 2.335 | [33,34] |
| Bifidobacterium | Prediabetes | 4.32 | 3.62082 × 10−8 | 4.187 | [35] |
| Collinsella | Prediabetes | 4.22 | 1.76318 × 10−7 | 8.219 | |
| Fusicatenibacter | Prediabetes | 4.19 | 3.71865 × 10−10 | 9.128 | [36] |
| Streptococcus | Prediabetes | 4.11 | 1.00677 × 10−9 | 11.946 | [33,37] |
| Clostridium | Prediabetes | 4.01 | 1.40866 × 10−6 | 5.096 | [38] |
| Lachnoclostridium | Prediabetes | 3.94 | 0.001795935 | 1.985 | |
| Anaerostipes | Prediabetes | 3.92 | 3.32433 × 10−9 | 3.972 | [39] |
| Mediterraneibacter | Prediabetes | 3.89 | 2.233 × 10−7 | 5.735 | [40] |
| Butyricicoccus | Prediabetes | 3.57 | 4.19266 × 10−8 | 3.015 | [41] |
| Dorea | Prediabetes | 3.49 | 4.89717 × 10−11 | 6.086 | [37,42] |
| Clostridioides | Prediabetes | 3.18 | 8.49243 × 10−6 | 7.105 | |
| Eggerthella | Prediabetes | 3.02 | 1.11996 × 10−8 | 9.801 | [43] |
| Carbohydrate group | |||||
| Bacterial genera | Group | LDA score | p-value | Fold change (HC/LC) * | |
| Coprococcus | LC | 3.73 | 0.00920058 | 0.414 | [44] |
| Group | Kegg ID | Kegg Pathway Name | Healthy/Prediabetes * | p-Value |
|---|---|---|---|---|
| healthy/prediabetes | ko04975 | Fat digestion and absorption | 6.36 | 6.312 × 10−5 |
| ko00062 | Fatty acid elongation | 0.17 | 4.219 × 10−14 | |
| ko00563 | GPI-anchor biosynthesis | 64.97 | 1.528 × 10−2 | |
| ko00604 | Glycosphingolipid biosynthesis—ganglio series | 2.04 | 9.552 × 10−10 | |
| ko00072 | Synthesis and degradation of ketone bodies | 0.45 | 7.304 × 10−12 | |
| Kegg ID | Kegg pathway name | HC/LC * | p-value | |
| HC/LC | ko04975 | Fat digestion and absorption | 0.39 | 2.033 × 10−2 |
| Kegg ID | Kegg pathway name | HCP/LCP * | p-value | |
| HCP/LCP | ko04975 | Fat digestion and absorption | 2.31 | 4.899 × 10−2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, W.-L.; Chen, Y.-E.; Tseng, H.-T.; Cheng, C.-F.; Wu, J.-H.; Hou, Y.-C. Gut Microbiota in Patients with Prediabetes. Nutrients 2024, 16, 1105. https://doi.org/10.3390/nu16081105
Chang W-L, Chen Y-E, Tseng H-T, Cheng C-F, Wu J-H, Hou Y-C. Gut Microbiota in Patients with Prediabetes. Nutrients. 2024; 16(8):1105. https://doi.org/10.3390/nu16081105
Chicago/Turabian StyleChang, Wei-Lin, Yu-En Chen, Hsiang-Tung Tseng, Ching-Feng Cheng, Jing-Hui Wu, and Yi-Cheng Hou. 2024. "Gut Microbiota in Patients with Prediabetes" Nutrients 16, no. 8: 1105. https://doi.org/10.3390/nu16081105
APA StyleChang, W.-L., Chen, Y.-E., Tseng, H.-T., Cheng, C.-F., Wu, J.-H., & Hou, Y.-C. (2024). Gut Microbiota in Patients with Prediabetes. Nutrients, 16(8), 1105. https://doi.org/10.3390/nu16081105









