Do Herbal Supplements and Probiotics Complement Antibiotics and Diet in the Management of SIBO? A Randomized Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Data Collection
2.2.1. SIBO Diagnosis
2.2.2. Complementary Analytical Tests
2.2.3. Measurement of Covariates
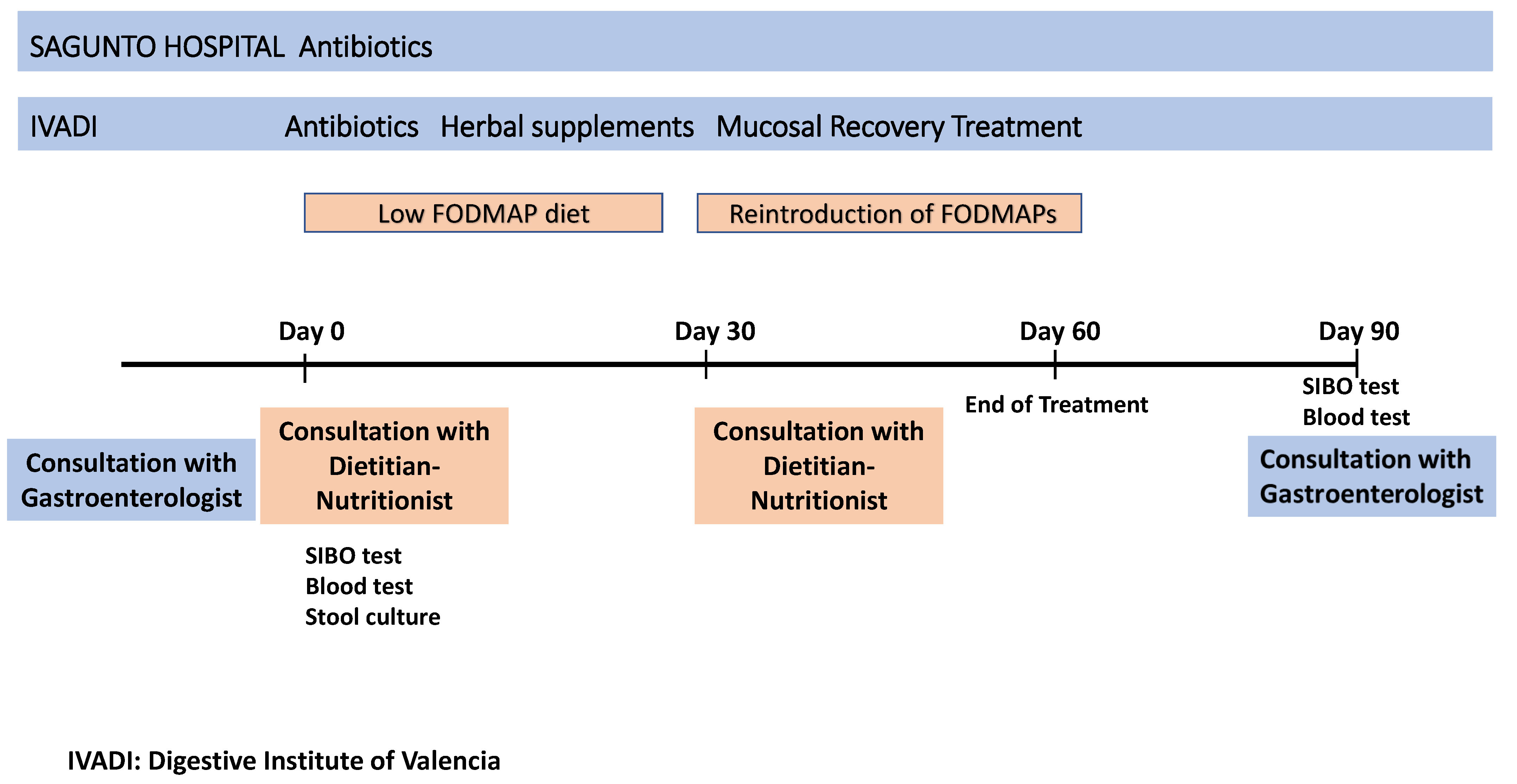
2.2.4. Procedure
2.2.5. Treatment
- -
- H2-SIBO: rifaximin 200 mg (2-2-2) for 10 days. After this, two commercial herbal preparations were recommended: Oleocaps 2 (Ingredients: rapeseed oil, gelatin, glycerin, essential oils (Origanum vulgare, Cinnamomum cassia, Citrus limon, Mentha piperita), extract rich in tocopherol) (Pranarom, Barcelona, Spain) 1-1-1, and berberine (Nutilab, Benaguasil, Valencia, Spain) 1-1-1, both for 20 days until completing the month. In patients with polypharmacy, wormwood (Nutri Holistic, Huelva, Spain) 2-2-2 was used instead of berberine. For gastritis, wormwood (Nutri Holistic) and berberine (Nutilab) were used as herbal supplements.
- -
- CH4-SIBO: rifaximin 200 mg (2-2-2) for 10 days and neomycin 500 mg (1-0-1). If this caused symptoms, the dose was reduced to one tablet per day. During these 10 days, the patients took the probiotic Ultra Levura (Saccharomyces boulardii, 250 mg per day) (Laboratorios Salvat, SA., Esplugues de Llobregat, Spain). Subsequently, two commercial herbal preparations were recommended: Oleocaps 2 (Pranarom) 1-1-1 and wormwood (Nutri Holistic) 2-2-2. For gastritis, wormwood (Nutri Holistic) 2-2-2 and black cumin oil (Sura Vitasan, Renteria, Guipuzkoa, Spain) 1-1-1 were used.
- -
- H2-SIBO: Probiotic Proinflam (Soria Natural, Garray, Soria, Spain) 1-0-0, L-glutamine (VByotics, Natural Probiotics, SL., Barcelona, Spain) 5 g/2 times daily between meals, and partially hydrolyzed guar gum (Max Fibra, Deiters, Badalona, Barcelona, Spain) 5 g daily, together with one of the meals.
- -
- CH4-SIBO: Probiotic Proinflam (Soria Natural) 1-0-0 and L-glutamine (VByotics) 5 g/two times daily between meals.
- -
- H2-SIBO: rifaximin 200 mg (2-2-2) for 10 days.
- -
- CH4-SIBO: rifaximin 200 mg (2-2-2) for 10 days and neomycin 500 mg (1-0-1). If this caused symptoms, the dose was reduced to one tablet per day.
2.3. Statistical Analysis
2.4. Ethical Considerations
3. Results
4. Discussion
4.1. Strengths and Limitations
4.2. Implications for Clinical Practice and Research
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Weiss, G.A.; Hennet, T. Mechanisms and consequences of intestinal dysbiosis. Cell Mol. Life Sci. 2017, 74, 2959–2977. [Google Scholar] [CrossRef] [PubMed]
- Bamba, S.; Imai, T.; Sasaki, M.; Ohno, M.; Yoshida, S.; Nishida, A.; Takahashi, K.; Inatomi, O.; Andoh, A. Altered gut microbiota in patients with small intestinal bacterial overgrowth. J. Gastroenterol. Hepatol. 2022, 38, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, R.; Ma, J.; Tang, S.; Li, Y.; Li, Y.; Wan, J. Mucosa-Associated Microbial Profile Is Altered in Small Intestinal Bacterial Overgrowth. Front. Microbiol. 2021, 12, 710940. [Google Scholar] [CrossRef] [PubMed]
- Bushyhead, D.; Quigley, E.M. Small Intestinal Bacterial Overgrowth-Pathophysiology and Its Implications for Definition and Management. Gastroenterology 2022, 163, 593–607. [Google Scholar] [CrossRef] [PubMed]
- Oana, K.; Shimizu, K.; Takada, T.; Makino, H.; Yamazaki, M.; Katto, M.; Ando, M.; Kurakawa, T.; Oishi, K. Manipulating the growth environment through co-culture to enhance stress tolerance and viability of probiotic strains in the gastrointestinal tract. Appl. Environ. Microbiol. 2023, 89, e0150223. [Google Scholar] [CrossRef] [PubMed]
- Sroka, N.; Rydzewska-Rosołowska, A.; Kakareko, K.; Rosołowski, M.; Głowińska, I.; Hryszko, T. Show Me What You Have Inside—The Complex Interplay between SIBO and Multiple Medical Conditions—A Systematic Review. Nutrients 2022, 15, 90. [Google Scholar] [CrossRef] [PubMed]
- Fujimori, S. What are the effects of proton pump inhibitors on the small intestine? World J. Gastroenterol. 2015, 21, 6817–6819. [Google Scholar] [CrossRef] [PubMed]
- Achufusi, T.G.O.; Sharma, A.; Zamora, E.A.; Manocha, D. Small Intestinal Bacterial Overgrowth: Comprehensive Review of Diagnosis, Prevention, and Treatment Methods. Cureus 2020, 12, e8860. [Google Scholar] [CrossRef] [PubMed]
- Losurdo, G.; Leandro, G.; Ierardi, E.; Perri, F.; Barone, M.; Principi, M.; Di Leo, A. Breath Tests for the Non-invasive Diagnosis of Small Intestinal Bacterial Overgrowth: A Systematic Review with Meta-analysis. J. Neurogastroenterol. Motil. 2020, 26, 16–28. [Google Scholar] [CrossRef]
- Pimentel, M.; Saad, R.J.; Long, M.D.; Rao, S.S.C. ACG Clinical Guideline: Small Intestinal Bacterial Overgrowth. Am. J. Gastroenterol. 2020, 115, 165–178. [Google Scholar] [CrossRef]
- Ghoshal, U.C.; Sachdeva, S.; Ghoshal, U.; Misra, A.; Puri, A.S.; Pratap, N.; Shah, A.; Rahman, M.M.; Gwee, K.A.; Tan, V.P.Y.; et al. Asian-Pacific consensus on small intestinal bacterial overgrowth in gastrointestinal disorders: An initiative of the Indian Neurogastroenterology and Motility Association. Indian J. Gastroenterol. 2022, 41, 483–507. [Google Scholar] [CrossRef]
- Barkin, J.A.; Keihanian, T.; Barkin, J.S.; Antequera, C.M.; Moshiree, B. Preferential usage of rifaximin for the treatment of hydrogen-positive smallintestinal bacterial overgrowth. Rev. Gastroenterol. Peru. 2019, 39, 111–115. [Google Scholar]
- Wang, J.; Zhang, L.; Hou, X. Efficacy of rifaximin in treating with small intestine bacterial overgrowth: A systematic review and meta-analysis. Expert Rev. Gastroenterol. Hepatol. 2021, 15, 1385–1399. [Google Scholar] [CrossRef]
- Chedid, V.; Dhalla, S.; Clarke, J.O.; Roland, B.C.; Dunbar, K.B.; Koh, J.; Justino, E.; Tomakin, E.; Mullin, G.E. Herbal therapy is equivalent to rifaximin for the treatment of small intestinal bacterial overgrowth. Glob. Adv. Health Med. 2014, 3, 16–24. [Google Scholar] [CrossRef]
- Guo, H.; Lu, S.; Zhang, J.; Chen, C.; Du, Y.; Wang, K.; Duan, L. Berberine and rifaximin effects on small intestinal bacterial overgrowth: Study protocol for an investigator-initiated, double-arm, open-label, randomized clinical trial (BRIEF-SIBO study). Front. Pharmacol. 2023, 14, 1121435. [Google Scholar] [CrossRef]
- Ren, X.; Di, Z.; Zhang, Z.; Fu, B.; Wang, Y.; Huang, C.; Du, Y. Chinese herbal medicine for the treatment of small intestinal bacterial overgrowth (SIBO): A protocol for systematic review and meta-analysis. Medicine 2020, 99, e23737. [Google Scholar] [CrossRef]
- Zhong, C.; Qu, C.; Wang, B.; Liang, S.; Zeng, B. Probiotics for Preventing and Treating Small Intestinal Bacterial Overgrowth: A Meta-Analysis and Systematic Review of Current Evidence. J. Clin. Gastroenterol. 2017, 51, 300–311. [Google Scholar] [CrossRef]
- Perna, S.; Alalwan, T.A.; Alaali, Z.; Alnashaba, T.; Gasparri, C.; Infantino, V.; Hammad, L.; Riva, A.; Petrangolini, G.; Allegrini, P.; et al. The Role of Glutamine in the Complex Interaction between Gut Microbiota and Health: A Narrative Review. Int. J. Mol. Sci. 2019, 20, 5232. [Google Scholar] [CrossRef]
- Zhou, Q.; Verne, M.L.; Fields, J.Z.; Lefante, J.J.; Basra, S.; Salameh, H.; Verne, G.N. Randomised placebo-controlled trial of dietary glutamine supplements for postinfectious irritable bowel syndrome. Gut 2018, 68, 996–1002. [Google Scholar] [CrossRef]
- Rastgoo, S.; Ebrahimi-Daryani, N.; Agah, S.; Karimi, S.; Taher, M.; Rashidkhani, B.; Hejazi, E.; Mohseni, F.; Ahmadzadeh, M.; Sadeghi, A.; et al. Glutamine Supplementation Enhances the Effects of a Low FODMAP Diet in Irritable Bowel Syndrome Management. Front. Nutr. 2021, 8, 746703. [Google Scholar] [CrossRef]
- Yasukawa, Z.; Inoue, R.; Ozeki, M.; Okubo, T.; Takagi, T.; Honda, A.; Naito, Y. Effect of Repeated Consumption of Partially Hydrolyzed Guar Gum on Fecal Characteristics and Gut Microbiota: A Randomized, Double-Blind, Placebo-Controlled, and Parallel-Group Clinical Trial. Nutrients 2019, 11, 2170. [Google Scholar] [CrossRef] [PubMed]
- Souza, C.; Rocha, R.; Cotrim, H.P. Diet and intestinal bacterial overgrowth: Is there evidence? World J. Clin. Cases 2022, 10, 4713–4716. [Google Scholar] [CrossRef]
- Morariu, I.-D.; Avasilcai, L.; Vieriu, M.; Lupu, V.V.; Morariu, B.-A.; Lupu, A.; Morariu, P.-C.; Pop, O.-L.; Starcea, I.M.; Trandafir, L. Effects of a Low-FODMAP Diet on Irritable Bowel Syndrome in Both Children and Adults—A Narrative Review. Nutrients 2023, 15, 2295. [Google Scholar] [CrossRef] [PubMed]
- Lauritano, E.C.; Gabrielli, M.; Scarpellini, E.; Lupascu, A.; Novi, M.; Sottili, S.; Vitale, G.; Cesario, V.; Serricchio, M.; Cammarota, G.; et al. Small intestinal bacterial overgrowth recurrence after antibiotic therapy. Am. J. Gastroenterol. 2008, 103, 2031–2035. [Google Scholar] [CrossRef] [PubMed]
- Rezaie, A.; Buresi, M.; Lembo, A.; Lin, H.; McCallum, R.; Rao, S.; Schmulson, M.; Valdovinos, M.; Zakko, S.; Pimentel, M. Hydrogen and Methane-Based Breath Testing in Gastrointestinal Disorders: The North American Consensus. Am. J. Gastroenterol. 2017, 112, 775–784. [Google Scholar] [CrossRef]
- Monash University Fodmap App. Revisado Noviembre 2023. Available online: https://www.monashfodmap.com/ibs-central/i-have-ibs/starting-the-low-fodmap-diet/ (accessed on 6 September 2021).
- Madrid, A.M.; Defilippi, C.; Defilippi, C.; Slimming, J.; Quera, R. Sobrecrecimiento bacteriano en trastornos funcionales del intestino [Small intestinal bacterial overgrowth in patients with functional gastrointestinal diseases]. Rev. Médica De Chile 2007, 135, 1245–1252. (In Spanish) [Google Scholar]
- Plauzolles, A.; Uras, S.; Pénaranda, G.; Bonnet, M.; Dukan, P.; Retornaz, F.; Halfon, P. Small Intestinal Bacterial Overgrowths and Intestinal Methanogen Overgrowths Breath Testing in a Real-Life French Cohort. Clin. Transl. Gastroenterol. 2022, 14, e00556. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Kim, J.J.-W.; Zhang, Y.; Du, L.; Dai, N. Prevalence and predictors of small intestinal bacterial overgrowth in irritable bowel syndrome: A systematic review and meta-analysis. J. Gastroenterol. 2018, 53, 807–818. [Google Scholar] [CrossRef]
- Quigley, E.M.; Murray, J.A.; Pimentel, M. AGA Clinical Practice Update on Small Intestinal Bacterial Overgrowth: Expert Review. Gastroenterology 2020, 159, 1526–1532. [Google Scholar] [CrossRef]
- Ponziani, F.R.; Zocco, M.A.; D’aversa, F.; Pompili, M.; Gasbarrini, A. Eubiotic properties of rifaximin: Disruption of the traditional concepts in gut microbiota modulation. World J. Gastroenterol. 2017, 23, 4491–4499. [Google Scholar] [CrossRef]
- Ghoshal, U.C.; Nehra, A.; Mathur, A.; Rai, S. A meta-analysis on small intestinal bacterial overgrowth in patients with different subtypes of irritable bowel syndrome. J. Gastroenterol. Hepatol. 2019, 35, 922–931. [Google Scholar] [CrossRef] [PubMed]
- Gatta, L.; Scarpignato, C. Systematic review with meta-analysis: Rifaximin is effective and safe for the treatment of small intestine bacterial overgrowth. Aliment. Pharmacol. Ther. 2017, 45, 604–616. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.; Talley, N.J.; Jones, M.; Kendall, B.J.; Koloski, N.; Walker, M.M.; Morrison, M.; Holtmann, G.J. Small Intestinal Bacterial Overgrowth in Irritable Bowel Syndrome: A Systematic Review and Meta-Analysis of Case-Control Studies. Am. J. Gastroenterol. 2020, 115, 190–201. [Google Scholar] [CrossRef] [PubMed]
- Scarpellini, E.; Gabrielli, M.; Lauritano, C.E.; Lupascu, A.; Merra, G.; Cammarota, G.; Cazzato, I.A.; Gasbarrini, G.; Gasbarrini, A. High dosage rifaximin for the treatment of small intestinal bacterial overgrowth. Aliment. Pharmacol. Ther. 2007, 25, 781–786. [Google Scholar] [CrossRef] [PubMed]
- Pimentel, M.; Chang, C.; Chua, K.S.; Mirocha, J.; DiBaise, J.; Rao, S.; Amichai, M. Antibiotic treatment of constipation-predominant irritable bowel syndrome. Dig. Dis. Sci. 2014, 59, 1278–1285. [Google Scholar] [CrossRef] [PubMed]
- Mitten, E.; Goldin, A.; Hanifi, J.; Chan, W.W. Recent probiotic use is independently associated with methane-positive breath test for small intestinal bacterial overgrowth. Am. J. Gastroenterol. 2018, 113, S660. [Google Scholar] [CrossRef]
- Saffouri, G.B.; Shields-Cutler, R.R.; Chen, J.; Yang, Y.; Lekatz, H.R.; Hale, V.L.; Cho, J.M.; Battaglioli, E.J.; Bhattarai, Y.; Thompson, K.J.; et al. Small intestinal microbial dysbiosis underlies symptoms associated with functional gastrointestinal disorders. Nat. Commun. 2019, 10, 2012. [Google Scholar] [CrossRef] [PubMed]
- Fernández, L.M.B.; Man, F.; Lasa, J.S. Impact of Saccharomyces boulardii CNCM I-745 on Bacterial Overgrowth and Composition of Intestinal Microbiota in Diarrhea-Predominant Irritable Bowel Syndrome Patients: Results of a Randomized Pilot Study. Dig. Dis. 2023, 41, 798–809. [Google Scholar] [CrossRef]
- Efremova, I.; Maslennikov, R.; Poluektova, E.; Vasilieva, E.; Zharikov, Y.; Suslov, A.; Letyagina, Y.; Kozlov, E.; Levshina, A.; Ivashkin, V. Epidemiology of small intestinal bacterial overgrowth. World J. Gastroenterol. 2023, 29, 3400–3421. [Google Scholar] [CrossRef]
- Leite, G.; Morales, W.; Weitsman, S.; Celly, S.; Parodi, G.; Mathur, R.; Barlow, G.M.; Sedighi, R.; Millan, M.J.V.; Rezaie, A.; et al. The duodenal microbiome is altered in small intestinal bacterial overgrowth. PLoS ONE 2020, 15, e0234906. [Google Scholar] [CrossRef]
- Tan, N.; Gwee, K.A.; Tack, J.; Zhang, M.; Li, Y.; Chen, M.; Xiao, Y. Herbal medicine in the treatment of functional gastrointestinal disorders: A systematic review with meta-analysis. J. Gastroenterol. Hepatol. 2019, 35, 544–556. [Google Scholar] [CrossRef] [PubMed]
- Jun, H.; Ko, S.-J.; Kim, K.; Kim, J.; Park, J.-W. An Overview of Systematic Reviews of Herbal Medicine for Irritable Bowel Syndrome. Front. Pharmacol. 2022, 13, 894122. [Google Scholar] [CrossRef] [PubMed]
- Nickles, M.A.; Hasan, A.; Shakhbazova, A.; Wright, S.; Chambers, C.J.; Sivamani, R.K. Alternative Treatment Approaches to Small Intestinal Bacterial Overgrowth: A Systematic Review. J. Altern. Complement. Med. 2021, 27, 108–119. [Google Scholar] [CrossRef] [PubMed]

| Variable | Total (n = 179) Mean ± SD; n (%) | H2-SIBO (n = 56; 31.3%) Mean ± SD; n (%) | CH4-SIBO (n = 123; 69.7%) Mean ± SD; n (%) | p-Value |
|---|---|---|---|---|
| Age (years) | 45.7 ± 16.2 | 46.4 ± 15.5 | 45.4 ± 16.5 | 0.347 |
| Women | 148 (82.7) | 46 (82.1) | 102 (82.9) | 0.898 |
| BMI (kg/m2) | 23.9 ± 4.3 | 24.1 ± 4.0 | 23.9 ± 4.4 | 0.218 |
| H2 (ppm) * | 34.3 ± 31.6 | 45.6 ± 28.3 | 29.2 ± 31.8 | <0.001 |
| CH4 (ppm) ** | 29.6 ± 25.4 | 8.8 ± 11.7 | 39.1 ± 24.3 | <0.001 |
| Smokers | 21 (11.7) | 5 (8.9) | 16 (13.0) | 0.432 |
| Chronically ill | 40 (22.3) | 11 (19.6) | 29 (23.6) | 0.597 |
| H2-SIBO H2 (ppm) Mean ± SD | CH4-SIBO CH4 (ppm) Mean ± SD | |||||
|---|---|---|---|---|---|---|
| Control n = 24 | Intervention n = 32 | p-Value | Control n = 35 | Intervention n = 88 | p-Value | |
| Diagnosis | 47.0 ± 30.3 | 44.6 ± 27.2 | 0.377 | 37.6 ± 24.1 | 39.7 ± 24.5 | 0.340 |
| 3 months | 31.9 ± 34.0 | 38.3 ± 33.7 | 0.242 | 28.0 ± 32.8 | 25.7 ± 31.9 | 0.480 |
| Decrease | 15.1 ± 38.7 | 6.3 ± 31.6 | 0.175 | 11.7 ± 27.0 | 14.0 ± 28.4 | 0.338 |
| p-value i vs. f | 0.184 | 0.006 | - | <0.001 | 0.005 | - |
| Variable | H2-SIBO n = 56; n (%) | CH4-SIBO n = 123; n (%) | p * Value |
|---|---|---|---|
| Normalized gases | |||
| Total G | 19 (33.9) | 55 (44.7) | 0.174 |
| CG | 9 (37.5) | 14 (40.0) | 0.847 |
| IG | 10 (31.3) | 41 (46.6) | 0.133 |
| p-value CG vs IG | 0.625 | 0.507 | |
| Normalized clinical manifestations | |||
| Total G | 40 (71.4) | 90 (73.2) | 0.808 |
| CG | 17 (70.8) | 21 (60.0) | 0.393 |
| IG | 23 (71.9) | 69 (78.4) | 0.454 |
| p-value CG vs. IG | 0.932 | 0.038 |
| Variable | Normalized H2-SIBO n = 19; 33.9% | p-Value N vs. NN | Normalized CH4-SIBO n = 55; 44.7% | p-Value N vs. NN |
|---|---|---|---|---|
| Age (years) | 48.8 ± 14.1 | 0.092 | 48.1 ± 16.5 | 0.023 |
| Women | 14 (30.4) | 0.236 | 47 (46.1) | 0.503 |
| BMI (kg/m2) | 25.4 ± 5.0 | 0.402 | 23.4 ± 3.9 | 0.174 |
| H2 (ppm) | 37.6 ± 24.0 | 0.010 | 32.5 ± 38.5 | 0.101 |
| CH4 (ppm) | 6.5 ± 4.0 | 0.053 | 44.1 ± 26.2 | 0.004 |
| Smokers | 1 (20.0) | 0.491 | 8 (50.0) | 0.649 |
| Chronically ill | 5 (45.5) | 0.395 | 11 (37.9) | 0.401 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Redondo-Cuevas, L.; Belloch, L.; Martín-Carbonell, V.; Nicolás, A.; Alexandra, I.; Sanchis, L.; Ynfante, M.; Colmenares, M.; Mora, M.; Liebana, A.R.; et al. Do Herbal Supplements and Probiotics Complement Antibiotics and Diet in the Management of SIBO? A Randomized Clinical Trial. Nutrients 2024, 16, 1083. https://doi.org/10.3390/nu16071083
Redondo-Cuevas L, Belloch L, Martín-Carbonell V, Nicolás A, Alexandra I, Sanchis L, Ynfante M, Colmenares M, Mora M, Liebana AR, et al. Do Herbal Supplements and Probiotics Complement Antibiotics and Diet in the Management of SIBO? A Randomized Clinical Trial. Nutrients. 2024; 16(7):1083. https://doi.org/10.3390/nu16071083
Chicago/Turabian StyleRedondo-Cuevas, Lucia, Lucia Belloch, Vanesa Martín-Carbonell, Angela Nicolás, Iulia Alexandra, Laura Sanchis, Marina Ynfante, Michel Colmenares, María Mora, Ana Reyes Liebana, and et al. 2024. "Do Herbal Supplements and Probiotics Complement Antibiotics and Diet in the Management of SIBO? A Randomized Clinical Trial" Nutrients 16, no. 7: 1083. https://doi.org/10.3390/nu16071083
APA StyleRedondo-Cuevas, L., Belloch, L., Martín-Carbonell, V., Nicolás, A., Alexandra, I., Sanchis, L., Ynfante, M., Colmenares, M., Mora, M., Liebana, A. R., Antequera, B., Grau, F., Molés, J. R., Cuesta, R., Díaz, S., Sancho, N., Tomás, H., Gonzalvo, J., Jaén, M., ... Cortés-Rizo, X. (2024). Do Herbal Supplements and Probiotics Complement Antibiotics and Diet in the Management of SIBO? A Randomized Clinical Trial. Nutrients, 16(7), 1083. https://doi.org/10.3390/nu16071083






