Targeting Wnt/β-Catenin Pathway by Flavonoids: Implication for Cancer Therapeutics
Abstract
1. Introduction
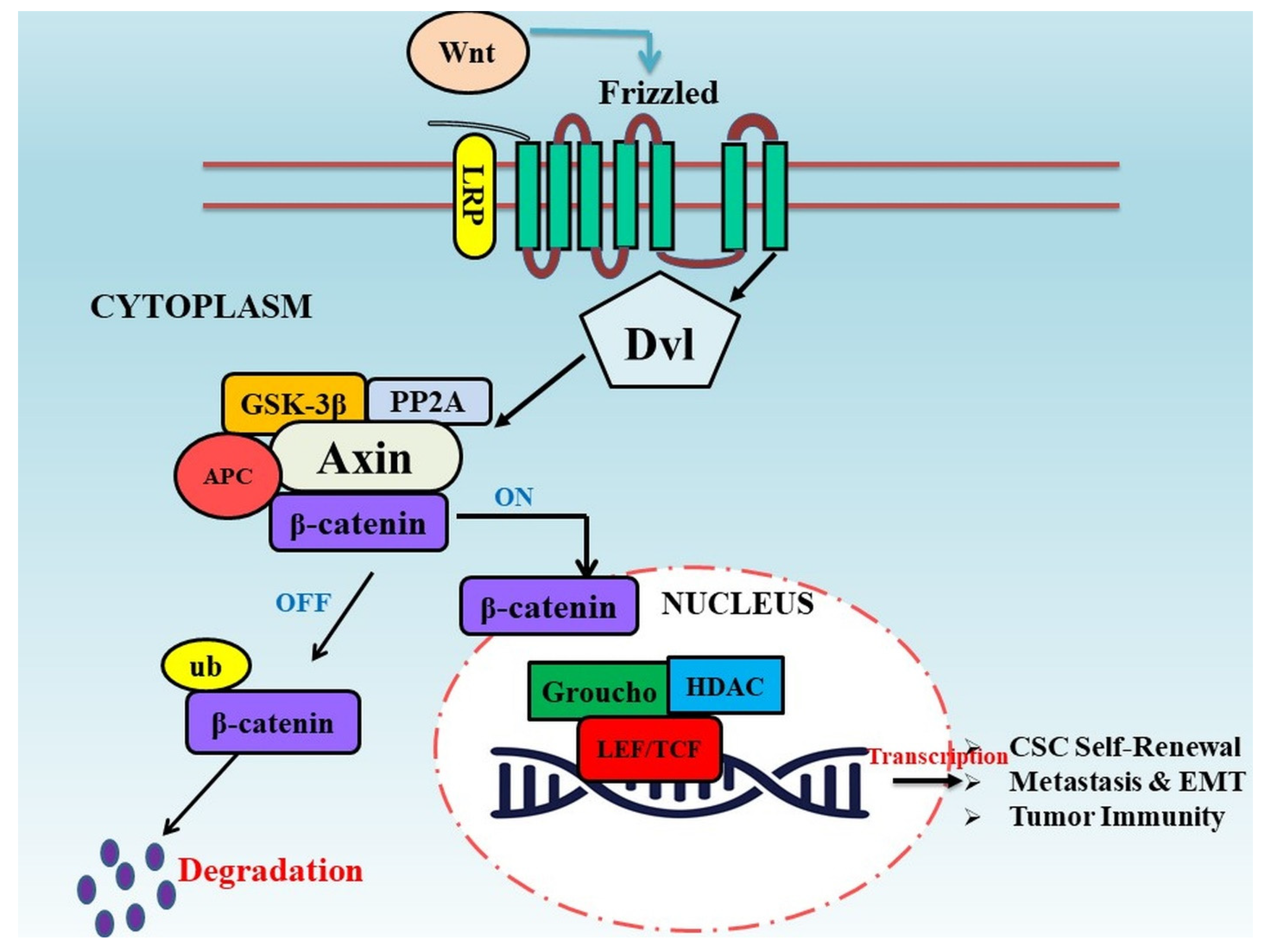
2. Wnt/β-Catenin Signaling Pathway in Tumor Progression
2.1. Correlation of Wnt Signaling in Cancer Stem Cells
2.2. Wnt Signaling and Metastasis
2.3. Wnt Signaling in Tumor Immunity
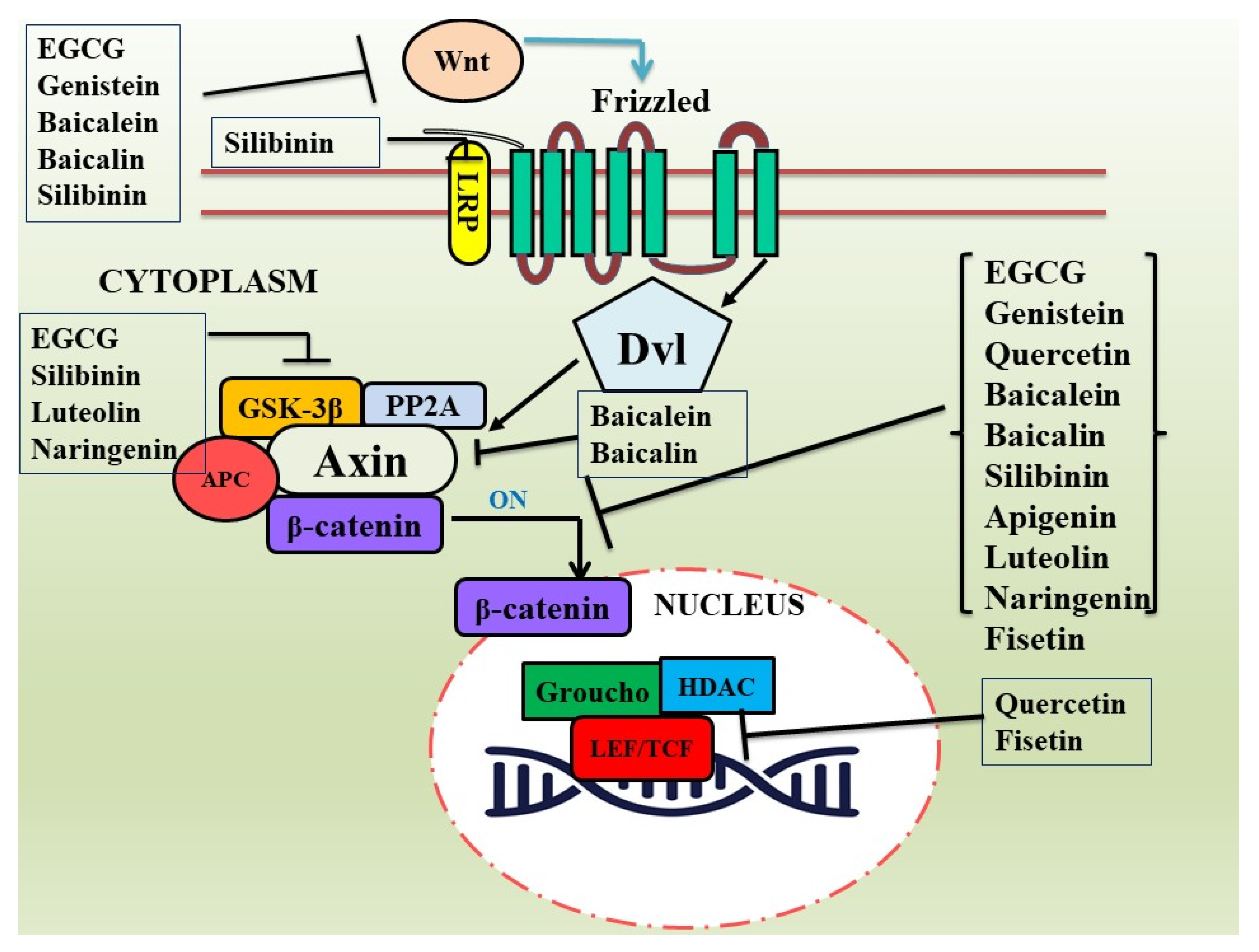
3. Flavonoids as Modulators of Dysregulated Wnt/β-Catenin Pathway in Cancer
3.1. Epigallocatechin-3-Gallate (EGCG)
3.2. Genistein
3.3. Quercetin
3.4. Baicalein and Baicalin
3.5. Silibinin
3.6. Apigenin
3.7. Luteolin
3.8. Miscellaneous
| Compound | Cancer Type | Cancer Model | Target of Wnt/β-Catenin Signaling Pathway | References |
|---|---|---|---|---|
| EGCG (C22H18O11) | Gastric cancer | SGC-7901 | p-β-catenin and p-GSK3β | [97] |
| Breast cancer | MDA-MB-231 | GSK-3β, Wnt, and c-myc | [99] | |
| Colon cancer | DLD-1 and SW480 | GSK-3β, β-catenin, and c-myc | [100] | |
| Lung cancer | A549 and H1299 | GSK-3β and β-catenin | [101] | |
| Colon cancer | SW480 and HCT116 | β-catenin, cyclin D1, and c-myc | [102] | |
| Colon cancer | HT-29 | GSK3-α and-β, β-catenin | [105] | |
| Skin cancer | A431 and SCC13 | Β-catenin, GSK-3β, and casein kinase1α | [107] | |
| Neuroendocrine tumor | PC12 | β-catenin and Wnt-3a | [108] | |
| Osteosarcoma | MG63, 143B, and SaoS2 | GSK-3β and β-catenin | [109] | |
| Genistein (C15H10O5) | Colon pre-neoplasia | Sprague Dawley rats | Wnt5a, Sfrp1, Sfrp2, and Sfrp5 | [116] |
| Colon cancer | Sprague Dawley rats | Sfrp2, Sfrp5, and Wnt5a | [117] | |
| Colon cancer | HT-29 | β-catenin | [118] | |
| Colon cancer | DLD-1 | β-catenin | [119] | |
| Colon cancer | SW1116 | WNT5a | [120] | |
| Renal cell carcinoma | Human sample (43) | sFRP1, Dkk2, and Smad4 | [121] | |
| Renal cancer | A-498 | sFRP1, Dkk2, and Smad4 | [121] | |
| Acute leukemia | U937 and Jurkat | c-myc and β-catenin | [122] | |
| Quercetin (C15H10O7) | Teratocarcinoma | NT2/D1 | β-catenin | [124] |
| Mammary cancer | 4T1 | β-catenin | [125] | |
| Colon cancer | SW480 | β-catenin/Tcf | [126,127] | |
| Prostate cancer | PC-3 | cyclin D1 and β-catenin | [128] | |
| Baicalein (C15H10O5) | Osteosarcoma | 143 B, MG63, and U2OS | β-catenin | [130,131] |
| Osteosarcoma | MG63 | β-catenin | [132] | |
| Osteosarcoma | MG-63 | β-catenin, c-myc, cyclinD1, and survivin | [133] | |
| Cervical cancer | HeLa, CaSki, C-33A, MS751, SN12C, and KBV1 | c-myc and β-catenin | [134] | |
| Leukemia | Jurkat cells | c-Myc, cyclin D1, Axin2, and β-catenin | [135] | |
| Breast cancer | MDA-MB-231 | Wnt1 and β-catenin | [136] | |
| Baicalin (C21H18O11) | Osteosarcoma | HOS, MG63, U2OS, and 143B | β-catenin | [137] |
| Breast cancer | MDA-MB-231 and BALB/c mice | β-catenin | [138] | |
| Silibinin (C25H22O10) | Colon cancer | SW480 and HCT116; athymic (nu/nu) male nude mice | c-Myc, cyclin D1, and β-catenin | [141] |
| Colon cancer | A/J mice | β-catenin and pGSK-3β | [142] | |
| Colon cancer | male Wistar rats | β-catenin | [143,144] | |
| Prostate cancer | PC-3 and C4–2B | LRP6 and Wnt3A | [145] | |
| Prostate cancer | PC-3, DU-145 | LRP6 and Wnt3A | [146] | |
| Breast cancer | MDA-MB-231 and T-47D | LRP6 and Wnt3A | [146] | |
| Renal cell carcinoma | 786-O and ACHN; BALB/c male nude mice | Wnt3a, GSK3β, and β-catenin | [148] | |
| Apigenin (C15H10O5) | Colon cancer | SW480 and HCT15; C57BL/6 mice | β-catenin | [154] |
| Hepatocellular carcinoma | SMMC-7721 and HepG2 | β-catenin | [155] | |
| Osteosarcoma | U2OS and MG63 | β-catenin | [156] | |
| Prostate cancer | C57BL/TGN TRAMP mice, DU145 | c-Myc, cyclin D1, and β-catenin | [157] | |
| Luteolin (C15H10O6) | Colon cancer | HCT-15 | GSK-3β, β-catenin, and c-myc | [159] |
| Colon cancer | Azoxymethane (AOM)-induced mouse | β-catenin, GSK-3β, and cyclin D1 | [160] | |
| Breast cancer | MDA-MB-231, BT5-49; Female nude mice | β-catenin | [161] | |
| Prostate cancer | PC-3, DU145 | GSK-3β, cyclin D1, and c-myc | [162] | |
| Naringenin (C15H12O5) | Gastric cancer | AGS | GSK-3β and β-catenin | [164] |
| Hepatocellular carcinoma | Huh7 and Hep3B | GSK-3β and β-catenin | [165] | |
| Fisetin (C15H10O6) | Colon cancer | HCT116 and HT29 | TCF4 and β-catenin | [166] |
| Melanoma | Mel 928, WM35, and 451Lu | β-catenin | [167] |
4. Limitations Associated with Natural Products as Anticancer Therapeutics
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mattiuzzi, C.; Lippi, G. Current cancer epidemiology. J. Epidemiol. Glob. Health 2019, 9, 217. [Google Scholar] [CrossRef] [PubMed]
- Hayat, R.; Manzoor, M.; Hussain, A. Wnt signaling pathway: A comprehensive review. Cell Biol. Int. 2022, 46, 863–877. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.H.; Cao, G.; Wu, X.Q.; Vaziri, N.D.; Zhao, Y.Y. Wnt signaling pathway in aging-related tissue fibrosis and therapies. Ageing Res. Rev. 2020, 60, 101063. [Google Scholar] [CrossRef] [PubMed]
- Azbazdar, Y.; Karabicici, M.; Erdal, E.; Ozhan, G. Regulation of Wnt signaling pathways at the plasma membrane and their misregulation in cancer. Front. Cell Dev. Biol. 2021, 9, 631623. [Google Scholar] [CrossRef] [PubMed]
- Taheri, N.; Choi, E.L.; Nguyen, V.T.T.; Chandra, A.; Hayashi, Y. Wnt Signaling in the Gastrointestinal Tract in Health and Disease. Physiologia 2023, 3, 86–97. [Google Scholar] [CrossRef]
- Yousefi, F.; Shabaninejad, Z.; Vakili, S.; Derakhshan, M.; Movahedpour, A.; Dabiri, H.; Hamblin, M.R. TGF-β and WNT signaling pathways in cardiac fibrosis: Non-coding RNAs come into focus. Cell Commun. Signal. 2020, 18, 87. [Google Scholar] [CrossRef]
- O'Brien, S.; Chidiac, R.; Angers, S. Modulation of Wnt-β-catenin signaling with antibodies: Therapeutic opportunities and challenges. Trends Pharmacol Sci. 2023. [Google Scholar] [CrossRef]
- Dutta, S.; Mahalanobish, S.; Saha, S.; Ghosh, S.; Sil, P.C. Natural products: An upcoming therapeutic approach to cancer. Food Chem. Toxicol. 2019, 128, 240–255. [Google Scholar] [CrossRef]
- Majolo, F.; Delwing, L.K.D.O.B.; Marmitt, D.J.; Bustamante-Filho, I.C.; Goettert, M.I. Medicinal plants and bioactive natural compounds for cancer treatment: Important advances for drug discovery. Phytochem. Lett. 2019, 31, 196–207. [Google Scholar] [CrossRef]
- De Cicco, P.; Catani, M.V.; Gasperi, V.; Sibilano, M.; Quaglietta, M.; Savini, I. Nutrition and breast cancer: A literature review on prevention, treatment and recurrence. Nutrients 2019, 11, 1514. [Google Scholar] [CrossRef]
- Ullah, A.; Munir, S.; Badshah, S.L.; Khan, N.; Ghani, L.; Poulson, B.G.; Jaremko, M. Important flavonoids and their role as a therapeutic agent. Molecules 2020, 25, 5243. [Google Scholar] [CrossRef] [PubMed]
- Abou Baker, D.H. An ethnopharmacological review on the therapeutical properties of flavonoids and their mechanisms of actions: A comprehensive review based on up to date knowledge. Toxicol. Rep. 2022, 9, 445–469. [Google Scholar] [CrossRef] [PubMed]
- Bourais, I.; Elmarrkechy, S.; Taha, D.; Mourabit, Y.; Bouyahya, A.; El Yadini, M.; Iba, N. A Review on Medicinal Uses, Nutritional Value, and Antimicrobial, Antioxidant, Anti-Inflammatory, Antidiabetic, and Anticancer Potential Related to Bioactive Compounds of J. regia. Food Rev. Int. 2022, 1–51. [Google Scholar] [CrossRef]
- Khan, W.A. Therapeutic Potential of Natural Pharmacological Agents: Flavonoids against Various Diseases. Scholast. J. Nat. Med. Educ. 2022, 1, 39–53. [Google Scholar]
- Khan, A.U.; Dagur, H.S.; Khan, M.; Malik, N.; Alam, M.; Mushtaque, M. Therapeutic role of flavonoids and flavones in cancer prevention: Current trends and future perspectives. Eur. J. Med. Chem. Rep. 2021, 3, 100010. [Google Scholar] [CrossRef]
- Slika, H.; Mansour, H.; Wehbe, N.; Nasser, S.A.; Iratni, R.; Nasrallah, G.; Eid, A.H. Therapeutic potential of flavonoids in cancer: ROS-mediated mechanisms. Biomed. Pharmacother. 2022, 146, 112442. [Google Scholar] [CrossRef]
- Wang, X.; Chen, B.; Xu, D.; Li, Z.; Liu, H.; Huang, Z.; Yao, H. Molecular mechanism and pharmacokinetics of flavonoids in the treatment of resistant EGF receptor-mutated non-small-cell lung cancer: A narrative review. Br. J. Pharmacol. 2021, 178, 1388–1406. [Google Scholar] [CrossRef]
- Tavsan, Z.; Kayali, H.A. Flavonoids showed anticancer effects on the ovarian cancer cells: Involvement of reactive oxygen species, apoptosis, cell cycle and invasion. Biomed. Pharmacother. 2019, 116, 109004. [Google Scholar] [CrossRef]
- Yu, F.; Yu, C.; Li, F.; Zuo, Y.; Wang, Y.; Yao, L.; Ye, L. Wnt/β-catenin signaling in cancers and targeted therapies. Signal Transduct. Target. Ther. 2021, 6, 307. [Google Scholar] [CrossRef]
- Rubinstein, M.R.; Baik, J.E.; Lagana, S.M.; Han, R.P.; Raab, W.J.; Sahoo, D.; Han, Y.W. Fusobacterium nucleatum promotes colorectal cancer by inducing Wnt/β-catenin modulator Annexin A1. EMBO Rep. 2019, 20, e47638. [Google Scholar] [CrossRef]
- Zhang, X.; Li, C.; Wu, Y.; Cui, P. The research progress of Wnt/β-catenin signaling pathway in colorectal cancer. Clin. Res. Hepatol. Gastroenterol. 2023, 47, 102086. [Google Scholar] [CrossRef] [PubMed]
- Krishnamurthy, N.; Kurzrock, R. Targeting the Wnt/beta-catenin pathway in cancer: Update on effectors and inhibitors. Cancer Treat. Rev. 2018, 62, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Ghanghas, P.; Rana, C.; Sanyal, S.N. Role of GSK-3β in regulation of canonical Wnt/β-catenin signaling and PI3-K/Akt oncogenic pathway in colon cancer. Cancer Investig. 2017, 35, 473–483. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Pan, W. GSK3: A multifaceted kinase in Wnt signaling. Trends Biochem. Sci. 2010, 35, 161–168. [Google Scholar] [CrossRef]
- MacDonald, B.T.; Tamai, K.; He, X. Wnt/β-catenin signaling: Components, mechanisms, and diseases. Dev. Cell 2009, 17, 9–26. [Google Scholar] [CrossRef]
- Nusse, R.; Clevers, H. Wnt/β-catenin signaling, disease, and emerging therapeutic modalities. Cell 2017, 169, 985–999. [Google Scholar] [CrossRef]
- Chatterjee, A.; Paul, S.; Bisht, B.; Bhattacharya, S.; Sivasubramaniam, S.; Paul, M.K. Advances in targeting the WNT/β-catenin signaling pathway in cancer. Drug Discov. Today 2022, 27, 82–101. [Google Scholar] [CrossRef]
- Dzobo, K.; Thomford, N.E.; Senthebane, D.A. Targeting the Versatile Wnt/β-Catenin Pathway in Cancer Biology and Therapeutics: From Concept to Actionable Strategy. Omics A J. Integr. Biol. 2019, 23, 517–538. [Google Scholar] [CrossRef]
- Pelullo, M.; Zema, S.; Nardozza, F.; Checquolo, S.; Screpanti, I.; Bellavia, D. Wnt, Notch, and TGF-β Pathways Impinge on Hedgehog Signaling Complexity: An Open Window on Cancer. Front. Genet. 2019, 10, 711. [Google Scholar] [CrossRef]
- Brechbiel, J.; Miller-Moslin, K.; Adjei, A.A. Crosstalk between hedgehog and other signaling pathways as a basis for combination therapies in cancer. Cancer Treat. Rev. 2014, 40, 750–759. [Google Scholar] [CrossRef]
- Kumar, V.; Vashishta, M.; Kong, L.; Wu, X.; Lu, J.J.; Guha, C.; Dwarakanath, B.S. The Role of Notch, Hedgehog, and Wnt Signaling Pathways in the Resistance of Tumors to Anticancer Therapies. Front. Cell Dev. Biol. 2021, 9, 650772. [Google Scholar] [CrossRef] [PubMed]
- Sedgwick, A.E.; D’Souza-Schorey, C. Wnt signaling in cell motility and invasion: Drawing parallels between development and cancer. Cancers 2016, 8, 80. [Google Scholar] [CrossRef] [PubMed]
- Hu, T.; Li, C. Convergence between Wnt-β-catenin and EGFR signaling in cancer. Mol. Cancer 2010, 9, 236. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Jho, E.H. Cross-talk between Wnt/β-catenin and Hippo signaling pathways: A brief review. BMB Rep. 2014, 47, 540. [Google Scholar] [CrossRef] [PubMed]
- Kriz, V.; Korinek, V. Wnt, RSPO and Hippo signaling in the intestine and intestinal stem cells. Genes 2018, 9, 20. [Google Scholar] [CrossRef] [PubMed]
- Tomar, V.S.; Patil, V.; Somasundaram, K. Temozolomide induces activation of Wnt/β-catenin signaling in glioma cells via PI3K/Akt pathway: Implications in glioma therapy. Cell Biol. Toxicol. 2020, 36, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Duda, P.; Akula, S.M.; Abrams, S.L.; Steelman, L.S.; Martelli, A.M.; Cocco, L.; McCubrey, J.A. Targeting GSK3 and associated signaling pathways involved in cancer. Cells 2020, 9, 1110. [Google Scholar] [CrossRef] [PubMed]
- Caspi, M.; Wittenstein, A.; Kazelnik, M.; Shor-Nareznoy, Y.; Rosin-Arbesfeld, R. Therapeutic targeting of the oncogenic Wnt signaling pathway for treating colorectal cancer and other colonic disorders. Adv. Drug Deliv. Rev. 2021, 169, 118–136. [Google Scholar] [CrossRef]
- Koushyar, S.; Powell, A.G.; Vincan, E.; Phesse, T.J. Targeting Wnt signaling for the treatment of gastric cancer. Int. J. Mol. Sci. 2020, 21, 3927. [Google Scholar] [CrossRef]
- Xu, X.; Zhang, M.; Xu, F.; Jiang, S. Wnt signaling in breast cancer: Biological mechanisms, challenges and opportunities. Mol. Cancer 2020, 19, 165. [Google Scholar] [CrossRef]
- Park, H.B.; Kim, J.W.; Baek, K.H. Regulation of Wnt signaling through ubiquitination and deubiquitination in cancers. Int. J. Mol. Sci. 2020, 21, 3904. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Vega, F.; Mina, M.; Armenia, J.; Chatila, W.K.; Luna, A.; La, K.C.; Marra, M.A. Oncogenic signaling pathways in the cancer genome atlas. Cell 2018, 173, 321–337. [Google Scholar] [CrossRef] [PubMed]
- Kleeman, S.O.; Leedham, S.J. Not all Wnt activation is equal: Ligand-dependent versus ligand-independent Wnt activation in colorectal cancer. Cancers 2020, 12, 3355. [Google Scholar] [CrossRef] [PubMed]
- Polakis, P. The many ways of Wnt in cancer. Curr. Opin. Genet. Dev. 2007, 17, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Bugter, J.M.; Fenderico, N.; Maurice, M.M. Mutations and mechanisms of WNT pathway tumour suppressors in cancer. Nat. Rev. Cancer 2021, 21, 5–21. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.S.; Park, J.I. Wnt signaling in cancer: Therapeutic targeting of Wnt signaling beyond β-catenin and the destruction complex. Exp. Mol. Med. 2020, 52, 183–191. [Google Scholar] [CrossRef]
- Harb, J.; Lin, P.J.; Hao, J. Recent development of Wnt signaling pathway inhibitors for cancer therapeutics. Curr. Oncol. Rep. 2019, 21, 12. [Google Scholar] [CrossRef]
- Yang, Y.; Li, X.; Wang, T.; Guo, Q.; Xi, T.; Zheng, L. Emerging agents that target signaling pathways in cancer stem cells. J. Hematol. Oncol. 2020, 13, 1–18. [Google Scholar] [CrossRef]
- Reya, T.; Clevers, H. Wnt signaling in stem cells and cancer. Nature 2005, 434, 843–850. [Google Scholar] [CrossRef]
- Klaus, A.; Birchmeier, W. Wnt signaling and its impact on development and cancer. Nat. Rev. Cancer 2008, 8, 387–398. [Google Scholar] [CrossRef]
- Abrahamsson, A.E.; Geron, I.; Gotlib, J.; Dao KH, T.; Barroga, C.F.; Newton, I.G.; Jamieson, C.H. Glycogen synthase kinase 3β missplicing contributes to leukemia stem cell generation. Proc. Natl. Acad. Sci. USA 2009, 106, 3925–3929. [Google Scholar] [CrossRef] [PubMed]
- Riether, C.; Schürch, C.M.; Flury, C.; Hinterbrandner, M.; Drück, L.; Huguenin, A.L.; Ochsenbein, A.F. Tyrosine kinase inhibitor–induced CD70 expression mediates drug resistance in leukemia stem cells by activating Wnt signaling. Sci. Transl. Med. 2015, 7, 298ra119. [Google Scholar] [CrossRef] [PubMed]
- Schürch, C.; Riether, C.; Matter, M.S.; Tzankov, A.; Ochsenbein, A.F. CD27 signaling on chronic myelogenous leukemia stem cells activates Wnt target genes and promotes disease progression. J. Clin. Investig. 2012, 122, 624–638. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Krivtsov, A.V.; Sinha, A.U.; North, T.E.; Goessling, W.; Feng, Z.; Armstrong, S.A. The Wnt/β-catenin pathway is required for the development of leukemia stem cells in AML. Science 2010, 327, 1650–1653. [Google Scholar] [CrossRef]
- Giambra, V.; Jenkins, C.E.; Lam, S.H.; Hoofd, C.; Belmonte, M.; Wang, X.; Weng, A.P. Leukemia stem cells in T-ALL require active Hif1α and Wnt signaling. Blood J. Am. Soc. Hematol. 2015, 125, 3917–3927. [Google Scholar] [CrossRef]
- Pirozzi, G.; Tirino, V.; Camerlingo, R.; La Rocca, A.; Martucci, N.; Scognamiglio, G.; Franco, R.; Cantile, M.; Normanno, N.; Rocco, G. Prognostic value of cancer stem cells, epithelial-mesenchymal transition and circulating tumor cells in lung cancer. Oncol. Rep. 2013, 29, 1763–1768. [Google Scholar] [CrossRef]
- Zhang, Z.; Xu, Y. FZD7 accelerates hepatic metastases in pancreatic cancer by strengthening EMT and stemness associated with TGF-β/SMAD3 signaling. Mol. Med. 2022, 28, 82. [Google Scholar] [CrossRef]
- Bai, Y.; Sha, J.; Kanno, T. The role of carcinogenesis-related biomarkers in the Wnt pathway and their effects on epithelial–mesenchymal transition (EMT) in oral squamous cell carcinoma. Cancers 2020, 12, 555. [Google Scholar] [CrossRef]
- Huels, D.J.; Ridgway, R.A.; Radulescu, S.; Leushacke, M.; Campbell, A.D.; Biswas, S.; Sansom, O.J. E-cadherin can limit the transforming properties of activating β-catenin mutations. EMBO J. 2015, 34, 2321–2333. [Google Scholar] [CrossRef]
- Conacci-Sorrell, M.; Simcha, I.; Ben-Yedidia, T.; Blechman, J.; Savagner, P.; Ben-Ze’ev, A. Autoregulation of E-cadherin expression by cadherin–cadherin interactions: The roles of β-catenin signaling, Slug, and MAPK. J. Cell Biol. 2003, 163, 847–857. [Google Scholar] [CrossRef]
- Vargas, D.A.; Sun, M.; Sadykov, K.; Kukuruzinska, M.A.; Zaman, M.H. The Integrated Role of Wnt/β-Catenin, N-Glycosylation, and E-Cadherin-Mediated Adhesion in Network Dynamics. PLoS Comput. Biol. 2016, 12, e1005007. [Google Scholar] [CrossRef] [PubMed]
- Brabletz, T.; Hlubek, F.; Spaderna, S.; Schmalhofer, O.; Hiendlmeyer, E.; Jung, A.; Kirchner, T. Invasion and metastasis in colorectal cancer: Epithelial-mesenchymal transition, mesenchymal-epithelial transition, stem cells and β-catenin. Cells Tissues Organs 2005, 179, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Elia, I.; Doglioni, G.; Fendt, S.M. Metabolic hallmarks of metastasis formation. Trends Cell Biol. 2018, 28, 673–684. [Google Scholar] [CrossRef]
- Doglioni, G.; Parik, S.; Fendt, S.M. Interactions in the (pre) metastatic niche support metastasis formation. Front. Oncol. 2019, 9, 219. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Xing, T.; Yang, Z.; Dudek, R.; Lu, Q.; Chen, Y.H. Epithelial mesenchymal transition in embryonic development, tissue repair and cancer: A comprehensive overview. J. Clin. Med. 2017, 7, 1. [Google Scholar] [CrossRef]
- Kalluri, R. EMT: When epithelial cells decide to become mesenchymal-like cells. J. Clin. Investig. 2009, 119, 1417–1419. [Google Scholar] [CrossRef]
- Pastushenko, I.; Blanpain, C. EMT Transition States during Tumor Progression and Metastasis. Trends Cell Biol. 2019, 29, 212–226. [Google Scholar] [CrossRef]
- Wu, Z.Q.; Li, X.Y.; Hu, C.Y.; Ford, M.; Kleer, C.G.; Weiss, S.J. Canonical Wnt signaling regulates Slug activity and links epithelial-mesenchymal transition with epigenetic Breast Cancer 1, Early Onset (BRCA1) repression. Proc. Natl. Acad. Sci. USA 2012, 109, 16654–16659. [Google Scholar] [CrossRef]
- Wang, Y.; Bu, F.; Royer, C.; Serres, S.; Larkin, J.R.; Soto, M.S.; Sibson, N.R.; Salter, V.; Fritzsche, F.; Turnquist, C.; et al. ASPP2 controls epithelial plasticity and inhibits metastasis through β-catenin-dependent regulation of ZEB1. Nat. Cell Biol. 2014, 16, 1092–1104. [Google Scholar] [CrossRef] [PubMed]
- Tenbaum, S.P.; Ordóñez-Morán, P.; Puig, I.; Chicote, I.; Arqués, O.; Landolfi, S.; Fernández, Y.; Herance, J.R.; Gispert, J.D.; Mendizabal, L.; et al. β-catenin confers resistance to PI3K and AKT inhibitors and subverts FOXO3a to promote metastasis in colon cancer. Nat. Med. 2012, 18, 892–901. [Google Scholar] [CrossRef]
- Gujral, T.S.; Chan, M.; Peshkin, L.; Sorger, P.K.; Kirschner, M.W.; MacBeath, G. A noncanonical Frizzled2 pathway regulates epithelial-mesenchymal transition and metastasis. Cell 2014, 159, 844–856. [Google Scholar] [CrossRef] [PubMed]
- Kahlert, C.; Kalluri, R. Exosomes in tumor microenvironment influence cancer progression and metastasis. J. Mol. Med. 2013, 91, 431–437. [Google Scholar] [CrossRef] [PubMed]
- Chairoungdua, A.; Smith, D.L.; Pochard, P.; Hull, M.; Caplan, M.J. Exosome release of β-catenin: A novel mechanism that antagonizes Wnt signaling. J. Cell Biol. 2010, 190, 1079–1091. [Google Scholar] [CrossRef] [PubMed]
- Luga, V.; Zhang, L.; Viloria-Petit, A.M.; Ogunjimi, A.A.; Inanlou, M.R.; Chiu, E.; Buchanan, M.; Hosein, A.N.; Basik, M.; Wrana, J.L. Exosomes mediate stromal mobilization of autocrine Wnt-PCP signaling in breast cancer cell migration. Cell 2012, 151, 1542–1556. [Google Scholar] [CrossRef] [PubMed]
- Maheswaran, S.; Haber, D.A. Circulating tumor cells: A window into cancer biology and metastasis. Curr. Opin. Genet. Dev. 2010, 20, 96–99. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Ting, D.T.; Stott, S.L.; Wittner, B.S.; Ozsolak, F.; Paul, S.; Ciciliano, J.C.; Smas, M.E.; Winokur, D.; Gilman, A.J.; et al. RNA sequencing of pancreatic circulating tumour cells implicates WNT signaling in metastasis. Nature 2012, 487, 510–513. [Google Scholar] [CrossRef]
- Miyamoto, D.T.; Zheng, Y.; Wittner, B.S.; Lee, R.J.; Zhu, H.; Broderick, K.T.; Desai, R.; Fox, D.B.; Brannigan, B.W.; Trautwein, J.; et al. RNA-Seq of single prostate CTCs implicates noncanonical Wnt signaling in antiandrogen resistance. Science 2015, 349, 1351–1356. [Google Scholar] [CrossRef]
- Pai, S.G.; Carneiro, B.A.; Mota, J.M.; Costa, R.; Leite, C.A.; Barroso-Sousa, R.; Kaplan, J.B.; Chae, Y.K.; Giles, F.J. Wnt/beta-catenin pathway: Modulating anticancer immune response. J. Hematol. Oncol. 2017, 10, 101. [Google Scholar] [CrossRef]
- Zhou, Y.; Xu, J.; Luo, H.; Meng, X.; Chen, M.; Zhu, D. Wnt signaling pathway in cancer immunotherapy. Cancer Lett. 2022, 525, 84–96. [Google Scholar] [CrossRef]
- Spranger, S.; Bao, R.; Gajewski, T.F. Melanoma-intrinsic β-catenin signaling prevents anti-tumour immunity. Nature 2015, 523, 231–235. [Google Scholar] [CrossRef]
- da Silva, J.L.; Dos Santos, A.L.S.; Nunes, N.C.C.; de Moraes Lino da Silva, F.; Ferreira, C.G.M.; de Melo, A.C. Cancer immunotherapy: The art of targeting the tumor immune microenvironment. Cancer Chemother. Pharmacol. 2019, 84, 227–240. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Tian, T.; Kalland, K.H.; Ke, X.; Qu, Y. Targeting Wnt/β-Catenin Signaling for Cancer Immunotherapy. Trends Pharmacol. Sci. 2018, 39, 648–658. [Google Scholar] [CrossRef] [PubMed]
- Suryawanshi, A.; Hussein, M.S.; Prasad, P.D.; Manicassamy, S. Wnt Signaling Cascade in Dendritic Cells and Regulation of Anti-tumor Immunity. Front. Immunol. 2020, 11, 122. [Google Scholar] [CrossRef]
- Galluzzi, L.; Spranger, S.; Fuchs, E.; López-Soto, A. WNT Signaling in Cancer Immunosurveillance. Trends Cell Biol. 2019, 29, 44–65. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Alam, A.; Pant, R.; Chattopadhyay, S. Wnt Signaling and Its Significance Within the Tumor Microenvironment: Novel Therapeutic Insights. Front. Immunol. 2019, 10, 2872. [Google Scholar] [CrossRef] [PubMed]
- Shanmugavadivu, A.; Balagangadharan, K.; Selvamurugan, N. Angiogenic and osteogenic effects of flavonoids in bone regeneration. Biotechnol. Bioeng. 2022, 119, 2313–2330. [Google Scholar] [CrossRef]
- Guo, X.F.; Ruan, Y.; Li, Z.H.; Li, D. Flavonoid subclasses and type 2 diabetes mellitus risk: A meta-analysis of prospective cohort studies. Crit. Rev. Food Sci. Nutr. 2019, 59, 2850–2862. [Google Scholar] [CrossRef]
- Kapoor, B.; Gulati, M.; Gupta, R.; Singh, S.K.; Gupta, M.; Nabi, A.; Chawla, P.A. A review on plant flavonoids as potential anticancer agents. Curr. Org. Chem. 2021, 25, 737–747. [Google Scholar]
- Bhosale, P.B.; Ha, S.E.; Vetrivel, P.; Kim, H.H.; Kim, S.M.; Kim, G.S. Functions of polyphenols and its anticancer properties in biomedical research: A narrative review. Transl. Cancer Res. 2020, 9, 7619. [Google Scholar] [CrossRef]
- Tuli, H.S.; Garg, V.K.; Bhushan, S.; Uttam, V.; Sharma, U.; Jain, A.; Sethi, G. Natural flavonoids exhibit potent anticancer activity by targeting microRNAs in cancer: A signature step hinting towards clinical perfection. Transl. Oncol. 2023, 27, 101596. [Google Scholar] [CrossRef]
- Kumar, P.; Dixit, J.; Saini, R.; Verma, P.; Mishra, A.K.; NathTiwari, K. Potential of Flavonoids as Anticancer Drugs. In Phytopharmaceuticals: Potential Therapeutic Applications; John Wiley & Sons: Hoboken, NJ, USA, 2021; pp. 135–159. [Google Scholar]
- Amado, N.G.; Predes, D.; Moreno, M.M.; Carvalho, I.O.; Mendes, F.A.; Abreu, J.G. Flavonoids and Wnt/β-catenin signaling: Potential role in colorectal cancer therapies. Int. J. Mol. Sci. 2014, 15, 12094–12106. [Google Scholar] [CrossRef] [PubMed]
- Boozari, M.; Ebrahimi, S.N.; Soltani, S.; Tayarani-Najaran, Z.; Emami, S.A.; Asili, J.; Iranshahi, M. Absolute configuration and anti-cancer effect of prenylated flavonoids and flavonostilbenes from Sophora pachycarpa: Possible involvement of Wnt signaling pathway. Bioorganic Chem. 2019, 85, 498–504. [Google Scholar] [CrossRef] [PubMed]
- Manandhar, S.; Kabekkodu, S.P.; Pai, K.S.R. Aberrant canonical Wnt signaling: Phytochemical based modulation. Phytomedicine 2020, 76, 153243. [Google Scholar] [CrossRef] [PubMed]
- Gustafson, C.T.; Mamo, T.; Maran, A.; Yaszemski, M.J. Molecular strategies for modulating Wnt signaling. Front. Biosci.-Landmark 2017, 22, 137–156. [Google Scholar] [CrossRef] [PubMed]
- Gan, R.Y.; Li, H.B.; Sui, Z.Q.; Corke, H. Absorption, metabolism, anti-cancer effect and molecular targets of epigallocatechin gallate (EGCG): An updated review. Crit. Rev. Food Sci. Nutr. 2018, 58, 924–941. [Google Scholar] [CrossRef]
- Yang, C.; Du, W.; Yang, D. Inhibition of green tea polyphenol EGCG((-)-epigallocatechin-3-gallate) on the proliferation of gastric cancer cells by suppressing canonical wnt/β-catenin signaling pathway. Int. J. Food Sci. Nutr. 2016, 67, 818–827. [Google Scholar] [CrossRef]
- Liu, L.; Lai, C.Q.; Nie, L.; Ordovas, J.; Band, M.; Moser, L.; Meydani, M. The modulation of endothelial cell gene expression by green tea polyphenol-EGCG. Mol. Nutr. Food Res. 2008, 52, 1182–1192. [Google Scholar] [CrossRef]
- Kim, J.; Zhang, X.; Rieger-Christ, K.M.; Summerhayes, I.C.; Wazer, D.E.; Paulson, K.E.; Yee, A.S. Suppression of Wnt signaling by the green tea compound (–)-epigallocatechin 3-gallate (EGCG) in invasive breast cancer cells: Requirement of the transcriptional repressor HBP1. J. Biol. Chem. 2006, 281, 10865–10875. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, X.Q.; Zhang, Q.; Zhu, J.Y.; Li, Y.; Xie, C.F.; Han, H.Y. (−)-Epigallocatechin-3-gallate inhibits colorectal cancer stem cells by suppressing Wnt/β-catenin pathway. Nutrients 2017, 9, 572. [Google Scholar] [CrossRef]
- Zhu, J.; Jiang, Y.; Yang, X.; Wang, S.; Xie, C.; Li, X.; Li, Y.; Chen, Y.; Wang, X.; Meng, Y.; et al. Wnt/β-catenin pathway mediates (-)-Epigallocatechin-3-gallate (EGCG) inhibition of lung cancer stem cells. Biochem. Biophys. Res. Commun. 2017, 482, 15–21. [Google Scholar] [CrossRef]
- Oh, S.; Gwak, J.; Park, S.; Yang, C.S. Green tea polyphenol EGCG suppresses W nt/β-catenin signaling by promoting GSK-3β-and PP2A-independent β-catenin phosphorylation/degradation. Biofactors 2014, 40, 586–595. [Google Scholar] [CrossRef] [PubMed]
- Dashwood, W.M.; Orner, G.A.; Dashwood, R.H. Inhibition of β-catenin/Tcf activity by white tea, green tea, and epigallocatechin-3-gallate (EGCG): Minor contribution of H2O2 at physiologically relevant EGCG concentrations. Biochem. Biophys. Res. Commun. 2002, 296, 584–588. [Google Scholar] [CrossRef] [PubMed]
- Gao ZH, I.; Xu, Z.; Hung, M.S.; Lin, Y.C.; Wang, T.; Gong MI, N.; You, L. Promoter demethylation of WIF-1 by epigallocatechin-3-gallate in lung cancer cells. Anticancer Res. 2009, 29, 2025–2030. [Google Scholar]
- Pahlke, G.; Ngiewih, Y.; Kern, M.; Jakobs, S.; Marko, D.; Eisenbrand, G. Impact of quercetin and EGCG on key elements of the Wnt pathway in human colon carcinoma cells. J. Agric. Food Chem. 2006, 54, 7075–7082. [Google Scholar] [CrossRef]
- Mount, J.G.; Muzylak, M.; Allen, S.; Althnaian, T.; McGonnell, I.M.; Price, J.S. Evidence that the canonical Wnt signalling pathway regulates deer antler regeneration. In Developmental Dynamics; American Association of Anatomy: Rockville, MD, USA, 2006; Volume 235, pp. 1390–1399. [Google Scholar]
- Singh, T.; Katiyar, S.K. Green tea polyphenol,(−)-epigallocatechin-3-gallate, induces toxicity in human skin cancer cells by targeting β-catenin signaling. Toxicol. Appl. Pharmacol. 2013, 273, 418–424. [Google Scholar] [CrossRef]
- He, C.; Zhou, J.; Wang, D.; Wang, R.; Wu, M.; Dong, T. PLAGL1 Gene Demethylation Induced by Epigallocatechin Gallate Promotes Pheochromocytoma Cell Apoptosis Via Wnt/β-catenin Signaling Pathway. Asian Pac. J. Cancer Prev. 2022, 23, 2119–2125. [Google Scholar] [CrossRef]
- Dong, C.; Wang, Z.; Shen, P.; Chen, Y.; Wang, J.; Wang, H. Epigallocatechin-3-gallate suppresses the growth of human osteosarcoma by inhibiting the Wnt/β-catenin signaling pathway. Bioengineered 2022, 13, 8490–8502. [Google Scholar] [CrossRef]
- Goh, Y.X.; Jalil, J.; Lam, K.W.; Husain, K.; Premakumar, C.M. Genistein: A review on its anti-inflammatory properties. Front. Pharmacol. 2022, 13, 820969. [Google Scholar] [CrossRef]
- Spagnuolo, C.; Russo, G.L.; Orhan, I.E.; Habtemariam, S.; Daglia, M.; Sureda, A.; Nabavi, S.M. Genistein and cancer: Current status, challenges, and future directions. Adv. Nutr. 2015, 6, 408–419. [Google Scholar] [CrossRef]
- Park, C.H.; Hahm, E.R.; Lee, J.H.; Jung, K.C.; Yang, C.H. Inhibition of β-catenin-mediated transactivation by flavanone in AGS gastric cancer cells. Biochem. Biophys. Res. Commun. 2005, 331, 1222–1228. [Google Scholar] [CrossRef]
- Sarkar, F.H.; Li, Y.; Wang, Z.; Kong, D. Cellular signaling perturbation by natural products. Cell. Signal. 2009, 21, 1541–1547. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, Z.; Kong, D.; Li, R.; Sarkar, S.H.; Sarkar, F.H. Regulation of Akt/FOXO3a/GSK-3β/AR signaling network by isoflavone in prostate cancer cells. J. Biol. Chem. 2008, 283, 27707–27716. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Simmen, R.C. Soy isoflavone genistein upregulates epithelial adhesion molecule E-cadherin expression and attenuates β-catenin signaling in mammary epithelial cells. Carcinogenesis 2009, 30, 331–339. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, Q.; Zhou, D.; Chen, H. Genistein, a soya isoflavone, prevents azoxymethane-induced up-regulation of WNT/β-catenin signaling and reduces colon pre-neoplasia in rats. Br. J. Nutr. 2013, 109, 33–42. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, Q.; Chen, H. DNA methylation and histone modifications of Wnt genes by genistein during colon cancer development. Carcinogenesis 2013, 34, 1756–1763. [Google Scholar] [CrossRef]
- Lepri, S.R.; Zanelatto, L.C.; da Silva, P.B.; Sartori, D.; Ribeiro, L.R.; Mantovani, M.S. Effects of genistein and daidzein on cell proliferation kinetics in HT29 colon cancer cells: The expression of CTNNBIP1 (β-catenin), APC (adenomatous polyposis coli) and BIRC5 (survivin). Hum. Cell 2014, 27, 78–84. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, H. Genistein attenuates WNT signaling by up-regulating sFRP2 in a human colon cancer cell line. Exp. Biol. Med. 2011, 236, 714–722. [Google Scholar] [CrossRef]
- Wang, Z.; Chen, H. Genistein increases gene expression by demethylation of WNT5a promoter in colon cancer cell line SW1116. Anticancer Res. 2010, 30, 4537–4545. [Google Scholar]
- Hirata, H.; Ueno, K.; Nakajima, K.; Tabatabai, Z.L.; Hinoda, Y.; Ishii, N.; Dahiya, R. Genistein downregulates onco-miR-1260b and inhibits Wnt-signaling in renal cancer cells. Br. J. Cancer 2013, 108, 2070–2078. [Google Scholar] [CrossRef]
- Zhou, H.R.; Shen, J.Z.; Fu, H.Y.; Zhang, F. Genistein-induced anticancer effects on acute leukemia cells involve the regulation of Wnt signaling pathway through H4K20me1 rather than DNA demethylation. Curr. Med. Sci. 2021, 41, 869–879. [Google Scholar] [CrossRef]
- Tang, S.M.; Deng, X.T.; Zhou, J.; Li, Q.P.; Ge, X.X.; Miao, L. Pharmacological basis and new insights of quercetin action in respect to its anti-cancer effects. Biomed. Pharmacother. 2020, 121, 109604. [Google Scholar] [CrossRef] [PubMed]
- Mojsin, M.; Vicentic, J.M.; Schwirtlich, M.; Topalovic, V.; Stevanovic, M. Quercetin reduces pluripotency, migration and adhesion of human teratocarcinoma cell line NT2/D1 by inhibiting Wnt/β-catenin signaling. Food Funct. 2014, 5, 2564–2573. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Seo, E.M.; Sharma, A.R.; Ganbold, B.; Park, J.; Sharma, G.; Nam, J.S. Regulation of Wnt signaling activity for growth suppression induced by quercetin in 4T1 murine mammary cancer cells. Int. J. Oncol. 2013, 43, 1319–1325. [Google Scholar] [CrossRef] [PubMed]
- Shan, B.E.; Wang, M.X.; Li, R.Q. Quercetin inhibit human SW480 colon cancer growth in association with inhibition of cyclin D1 and survivin expression through Wnt/β-catenin signaling pathway. Cancer Investig. 2009, 27, 604–612. [Google Scholar] [CrossRef] [PubMed]
- Park, C.H.; Chang, J.Y.; Hahm, E.R.; Park, S.; Kim, H.K.; Yang, C.H. Quercetin, a potent inhibitor against beta-catenin/Tcf signaling in SW480 colon cancer cells. Biochem. Biophys. Res. Commun. 2005, 328, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Baruah, M.M.; Khandwekar, A.P.; Sharma, N. Quercetin modulates Wnt signaling components in prostate cancer cell line by inhibiting cell viability, migration, and metastases. Tumour Biol. J. Int. Soc. Oncodevelopmental Biol. Med. 2016, 37, 14025–14034. [Google Scholar] [CrossRef] [PubMed]
- Farooqi, A.A.; Kapanova, G.; Kalmakhanov, S.; Tanbayeva, G.; Zhakipbekov, K.S.; Rakhmetova, V.S.; Syzdykbayev, M.K. Regulation of Cell Signaling Pathways and Non-Coding RNAs by Baicalein in Different Cancers. Int. J. Mol. Sci. 2022, 23, 8377. [Google Scholar] [CrossRef]
- Yaylagül, E.Ö.; Ülger, C. The effect of baicalein on Wnt/ß-catenin pathway and miR-25 expression in Saos-2 osteosarcoma cell line. Turk. J. Med. Sci. 2020, 50, 1168–1179. [Google Scholar] [CrossRef]
- Zhang, F.W.; Peng, L.Y.; Shi, C.J.; Li, J.C.; Pang, F.X.; Fu, W.M.; Zhang, J.F. Baicalein mediates the anti-tumor activity in Osteosarcoma through lncRNA-NEF driven Wnt/β-catenin signaling regulatory axis. J. Orthop. Transl. 2022, 33, 132–141. [Google Scholar] [CrossRef]
- He, N.; Zhang, Z. Baicalein suppresses the viability of MG-63 osteosarcoma cells through inhibiting c-MYC expression via Wnt signaling pathway. Mol. Cell. Biochem. 2015, 405, 187–196. [Google Scholar] [CrossRef]
- Dai, G.; Zheng, D.; Wang, Q.; Yang, J.; Liu, G.; Song, Q.; Guo, W. Baicalein inhibits progression of osteosarcoma cells through inactivation of the Wnt/β-catenin signaling pathway. Oncotarget 2017, 8, 86098. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.; Xia, J.; Yang, H.; Li, Y.; Liu, S.; Cao, Y.; Tang, L.; Yu, X. Baicalein blocked cervical carcinoma cell proliferation by targeting CCND1 via Wnt/β-catenin signaling pathway. Artif. Cells Nanomed. Biotechnol. 2019, 47, 2729–2736. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Liu, S.; Chen, J.; He, L.; Meng, X.; Liu, S. Baicalein suppresses the proliferation of acute T-lymphoblastic leukemia Jurkat cells by inhibiting the Wnt/β-catenin signaling. Ann. Hematol. 2016, 95, 1787–1793. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Yan, W.; Dai, Z.; Gao, X.; Ma, Y.; Xu, Q.; Zhang, S. Baicalein suppresses metastasis of breast cancer cells by inhibiting EMT via downregulation of SATB1 and Wnt/β-catenin pathway. Drug Des. Dev. Ther. 2016, 10, 1419–1441. [Google Scholar] [CrossRef]
- Pang, H.; Wu, T.; Peng, Z.; Tan, Q.; Peng, X.; Zhan, Z.; Song, L.; Wei, B. Baicalin induces apoptosis and autophagy in human osteosarcoma cells by increasing ROS to inhibit PI3K/Akt/mTOR, ERK1/2 and β-catenin signaling pathways. J. Bone Oncol. 2022, 33, 100415. [Google Scholar] [CrossRef]
- Zhou, T.; Zhang, A.; Kuang, G.; Gong, X.; Jiang, R.; Lin, D.; Li, J.; Li, H.; Zhang, X.; Wan, J.; et al. Baicalin inhibits the metastasis of highly aggressive breast cancer cells by reversing epithelial-to-mesenchymal transition by targeting β-catenin signaling. Oncol. Rep. 2017, 38, 3599–3607. [Google Scholar] [CrossRef]
- Wing Ying Cheung, C.; Gibbons, N.; Wayne Johnson, D.; Lawrence Nicol, D. Silibinin-a promising new treatment for cancer. Anti-Cancer Agents Med. Chem. 2010, 10, 186–195. [Google Scholar] [CrossRef]
- Tuli, H.S.; Mittal, S.; Aggarwal, D.; Parashar, G.; Parashar, N.C.; Upadhyay, S.K.; Sharma, A.K. Path of Silibinin from diet to medicine: A dietary polyphenolic flavonoid having potential anti-cancer therapeutic significance. In Seminars in Cancer Biology; Academic Press: Cambridge, MA, USA, 2021; Volume 73, pp. 196–218. [Google Scholar]
- Kaur, M.; Velmurugan, B.; Tyagi, A.; Agarwal, C.; Singh, R.P.; Agarwal, R. Silibinin suppresses growth of human colorectal carcinoma SW480 cells in culture and xenograft through down-regulation of β-catenin-dependent signaling. Neoplasia 2010, 12, 415–424. [Google Scholar] [CrossRef]
- Ravichandran, K.; Velmurugan, B.; Gu, M.; Singh, R.P.; Agarwal, R. Inhibitory Effect of Silibinin against Azoxymethane-Induced Colon Tumorigenesis in A/J MiceSilibinin Suppresses Colon Tumorigenesis in Mice. Clin. Cancer Res. 2010, 16, 4595–4606. [Google Scholar] [CrossRef]
- Sangeetha, N.; Aranganathan, S.; Panneerselvam, J.; Shanthi, P.; Rama, G.; Nalini, N. Oral supplementation of silibinin prevents colon carcinogenesis in a long term preclinical model. Eur. J. Pharmacol. 2010, 643, 93–100. [Google Scholar] [CrossRef]
- Sangeetha, N.; Viswanathan, P.; Balasubramanian, T.; Nalini, N. Colon cancer chemopreventive efficacy of silibinin through perturbation of xenobiotic metabolizing enzymes in experimental rats. Eur. J. Pharmacol. 2012, 674, 430–438. [Google Scholar] [CrossRef] [PubMed]
- Deep, G.; Gangar, S.C.; Agarwal, C.; Agarwal, R. Role of E-cadherin in antimigratory and antiinvasive efficacy of silibinin in prostate cancer cells. Cancer Prev. Res. 2011, 4, 1222–1232. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Lin, C.; King, T.D.; Chen, H.; Reynolds, R.C.; Li, Y. Silibinin inhibits Wnt/β-catenin signaling by suppressing Wnt co-receptor LRP6 expression in human prostate and breast cancer cells. Cell. Signal. 2012, 24, 2291–2296. [Google Scholar] [CrossRef] [PubMed]
- Rajamanickam, S.; Velmurugan, B.; Kaur, M.; Singh, R.P.; Agarwal, R. Chemoprevention of intestinal tumorigenesis in APCmin/+ mice by silibinin. Cancer Res. 2010, 70, 2368–2378. [Google Scholar] [CrossRef]
- Fan, Y.; Hou, T.; Dan, W.; Liu, T.; Luan, J.; Liu, B.; Li, L.; Zeng, J. Silibinin inhibits epithelial-mesenchymal transition of renal cell carcinoma through autophagy-dependent Wnt/β-catenin signaling. Int. J. Mol. Med. 2020, 45, 1341–1350. [Google Scholar] [CrossRef]
- Pandey, P.; Khan, F.; Upadhyay, T.K. Deciphering the modulatory role of apigenin targeting oncogenic pathways in human cancers. In Chemical Biology & Drug Design; Wiley: NewYork, NY, USA, 2023. [Google Scholar] [CrossRef]
- Javed, Z.; Sadia, H.; Iqbal, M.J.; Shamas, S.; Malik, K.; Ahmed, R.; Raza, S.; Butnariu, M.; Cruz-Martins, N.; Sharifi-Rad, J. Apigenin role as cell-signaling pathways modulator: Implications in cancer prevention and treatment. Cancer Cell Int. 2021, 21, 189. [Google Scholar] [CrossRef]
- Lin, C.M.; Chen, H.H.; Lin, C.A.; Wu, H.C.; Sheu, J.J.; Chen, H.J. Apigenin-induced lysosomal degradation of β-catenin in Wnt/β-catenin signaling. Sci. Rep. 2017, 7, 372. [Google Scholar] [CrossRef]
- Yan, X.; Qi, M.; Li, P.; Zhan, Y.; Shao, H. Apigenin in cancer therapy: Anti-cancer effects and mechanisms of action. Cell Biosci. 2017, 7, 50. [Google Scholar] [CrossRef]
- Švehlíková, V.; Wang, S.; Jakubíková, J.; Williamson, G.; Mithen, R.; Bao, Y. Interactions between sulforaphane and apigenin in the induction of UGT1A1 and GSTA1 in CaCo-2 cells. Carcinogenesis 2004, 25, 1629–1637. [Google Scholar] [CrossRef]
- Xu, M.; Wang, S.; Song, Y.U.; Yao, J.; Huang, K.; Zhu, X. Apigenin suppresses colorectal cancer cell proliferation, migration and invasion via inhibition of the Wnt/β-catenin signaling pathway. Oncol. Lett. 2016, 11, 3075–3080. [Google Scholar] [CrossRef]
- Pan, F.F.; Zheng, Y.B.; Shi, C.J.; Zhang, F.W.; Zhang, J.F.; Fu, W.M. H19-Wnt/β-catenin regulatory axis mediates the suppressive effects of apigenin on tumor growth in hepatocellular carcinoma. Eur. J. Pharmacol. 2021, 893, 173810. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Li, L.; Lv, L.; Chen, D.; Shen, L.; Xie, Z. Apigenin inhibits the proliferation and invasion of osteosarcoma cells by suppressing the Wnt/β-catenin signaling pathway. Oncol. Rep. 2015, 34, 1035–1041. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.; MacLennan, G.T.; Flask, C.A.; Fu, P.; Mishra, A.; Resnick, M.I.; Gupta, S. Blockade of β-catenin signaling by plant flavonoid apigenin suppresses prostate carcinogenesis in TRAMP mice. Cancer Res. 2007, 67, 6925–6935. [Google Scholar] [CrossRef] [PubMed]
- Imran, M.; Rauf, A.; Abu-Izneid, T.; Nadeem, M.; Shariati, M.A.; Khan, I.A.; Mubarak, M.S. Luteolin, a flavonoid, as an anticancer agent: A review. Biomed. Pharmacother. 2019, 112, 108612. [Google Scholar] [CrossRef] [PubMed]
- Pandurangan, A.K.; Dharmalingam, P.; Sadagopan, S.K.; Ramar, M.; Munusamy, A.; Ganapasam, S. Luteolin induces growth arrest in colon cancer cells through involvement of Wnt/β-catenin/GSK-3β signaling. J. Environ. Pathol. Toxicol. Oncol. 2013, 32, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Ashokkumar, P.; Sudhandiran, G. Luteolin inhibits cell proliferation during Azoxymethane-induced experimental colon carcinogenesis via Wnt/ β-catenin pathway. Investig. New Drugs 2011, 29, 273–284. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.; Kuang, G.; Wan, J.; Zhang, X.; Li, H.; Gong, X.; Li, H. Luteolin suppresses the metastasis of triple-negative breast cancer by reversing epithelial-to-mesenchymal transition via downregulation of β-catenin expression. Oncol. Rep. 2017, 37, 895–902. [Google Scholar] [CrossRef]
- Han, K.; Lang, T.; Zhang, Z.; Zhang, Y.; Sun, Y.; Shen, Z.; Min, D. Luteolin attenuates Wnt signaling via upregulation of FZD6 to suppress prostate cancer stemness revealed by comparative proteomics. Sci. Rep. 2018, 8, 8537. [Google Scholar] [CrossRef]
- Motallebi, M.; Bhia, M.; Rajani, H.F.; Bhia, I.; Tabarraei, H.; Mohammadkhani, N.; Pereira-Silva, M.; Kasaii, M.S.; Nouri-Majd, S.; Mueller, A.L.; et al. Naringenin: A potential flavonoid phytochemical for cancer therapy. Life Sci. 2022, 305, 120752. [Google Scholar] [CrossRef]
- Lee, J.H.; Park, C.H.; Jung, K.C.; Rhee, H.S.; Yang, C.H. Negative regulation of β-catenin/Tcf signaling by naringenin in AGS gastric cancer cell. Biochem. Biophys. Res. Commun. 2005, 335, 771–776. [Google Scholar] [CrossRef]
- Kang, Q.; Gong, J.; Wang, M.; Wang, Q.; Chen, F.; Cheng, K.W. 6-C-(E-Phenylethenyl) naringenin attenuates the stemness of hepatocellular carcinoma cells by suppressing Wnt/β-catenin signaling. J. Agric. Food Chem. 2019, 67, 13939–13947. [Google Scholar] [CrossRef] [PubMed]
- Syed, D.N.; Afaq, F.; Maddodi, N.; Johnson, J.J.; Sarfaraz, S.; Ahmad, A.; Mukhtar, H. Inhibition of human melanoma cell growth by the dietary flavonoid fisetin is associated with disruption of Wnt/β-catenin signaling and decreased Mitf levels. J. Investig. Dermatol. 2011, 131, 1291–1299. [Google Scholar] [CrossRef] [PubMed]
- Suh, Y.; Afaq, F.; Johnson, J.J.; Mukhtar, H. A plant flavonoid fisetin induces apoptosis in colon cancer cells by inhibition of COX2 and Wnt/EGFR/NF-κB-signaling pathways. Carcinogenesis 2009, 30, 300–307. [Google Scholar] [CrossRef] [PubMed]
- Xiong, G.; Wu, Z.; Yi, J.; Fu, L.; Yang, Z.; Hsieh, C.; Yin, M.; Zeng, X.; Wu, C.; Lu, A.; et al. ADMETlab 2.0: An integrated online platform for accurate and comprehensive predictions of ADMET properties. Nucleic Acids Res. 2021, 49, W5–W14. [Google Scholar] [CrossRef]
| Compound | Properties | Parameters | Values |
|---|---|---|---|
| EGCG | Absorption | Caco-2 permeability | −6.306 |
| HIA | 0.274 | ||
| Distribution | PPB | 91.15% | |
| BBB penetration | 0.019 | ||
| Metabolism | CYP1A2 inhibitor | 0.095 | |
| CYP2C19 inhibitor | 0.124 | ||
| CYP2C9 inhibitor | 0.174 | ||
| CYP2D6 inhibitor | 0.037 | ||
| CYP3A4 inhibitor | 0.142 | ||
| Excretion | CL | 17.081 mL/min/kg | |
| T ½ | 0.87 | ||
| Toxicity | H-HT | 0.092 | |
| DILI | 0.172 | ||
| FDAMDD | 0.17 | ||
| Respiratory toxicity | 0.085 | ||
| Genistein | Absorption | Caco-2 permeability | −4.764 |
| HIA | 0.01 | ||
| Distribution | PPB | 97.55% | |
| BBB penetration | 0.02 | ||
| Metabolism | CYP1A2 inhibitor | 0.981 | |
| CYP2C19 inhibitor | 0.59 | ||
| CYP2C9 inhibitor | 0.615 | ||
| CYP2D6 inhibitor | 0.863 | ||
| CYP3A4 inhibitor | 0.716 | ||
| Excretion | CL | 7.8444 mL/min/kg | |
| T ½ | 0.876 | ||
| Toxicity | H-HT | 0.092 | |
| DILI | 0.51 | ||
| FDAMDD | 0.201 | ||
| Respiratory toxicity | 0.087 | ||
| Quercetin | Absorption | Caco-2 permeability | −5.204 |
| HIA | 0.014 | ||
| Distribution | PPB | 95.49% | |
| BBB penetration | 0.008 | ||
| Metabolism | CYP1A2 inhibitor | 0.943 | |
| CYP2C19 inhibitor | 0.053 | ||
| CYP2C9 inhibitor | 0.598 | ||
| CYP2D6 inhibitor | 0.411 | ||
| CYP3A4 inhibitor | 0.348 | ||
| Excretion | CL | 8.284 | |
| T ½ | 0.929 | ||
| Toxicity | H-HT | 0.1 | |
| DILI | 0.98 | ||
| FDAMDD | 0.31 | ||
| Respiratory toxicity | 0.072 | ||
| Baicalein | Absorption | Caco-2 permeability | −4.981 |
| HIA | 0.018 | ||
| Distribution | PPB | 98.99% | |
| BBB penetration | 0.013 | ||
| Metabolism | CYP1A2 inhibitor | 0.971 | |
| CYP2C19 inhibitor | 0.144 | ||
| CYP2C9 inhibitor | 0.644 | ||
| CYP2D6 inhibitor | 0.638 | ||
| CYP3A4 inhibitor | 0.183 | ||
| Excretion | CL | 4.082 | |
| T ½ | 0.881 | ||
| Toxicity | H-HT | 0.084 | |
| DILI | 0.958 | ||
| FDAMDD | 0.084 | ||
| Respiratory toxicity | 0.332 | ||
| Baicalin | Absorption | Caco-2 permeability | −6.34 |
| HIA | 0.793 | ||
| Distribution | PPB | 83.35% | |
| BBB penetration | 0.05 | ||
| Metabolism | CYP1A2 inhibitor | 0.037 | |
| CYP2C19 inhibitor | 0.017 | ||
| CYP2C9 inhibitor | 0.005 | ||
| CYP2D6 inhibitor | 0.01 | ||
| CYP3A4 inhibitor | 0.004 | ||
| Excretion | CL | 1.0 | |
| T ½ | 0.855 | ||
| Toxicity | H-HT | 0.178 | |
| DILI | 0.977 | ||
| FDAMDD | 0.005 | ||
| Respiratory toxicity | 0.046 | ||
| Silibinin | Absorption | Caco-2 permeability | −6.255 |
| HIA | 0.366 | ||
| Distribution | PPB | 96.65% | |
| BBB penetration | 0.024 | ||
| Metabolism | CYP1A2 inhibitor | 0.038 | |
| CYP2C19 inhibitor | 0.12 | ||
| CYP2C9 inhibitor | 0.664 | ||
| CYP2D6 inhibitor | 0.31 | ||
| CYP3A4 inhibitor | 0.785 | ||
| Excretion | CL | 5.144 | |
| T ½ | 0.274 | ||
| Toxicity | H-HT | 0.079 | |
| DILI | 0.921 | ||
| FDAMDD | 0.035 | ||
| Respiratory toxicity | 0.027 | ||
| Apigenin | Absorption | Caco-2 permeability | −4.847 |
| HIA | 0.015 | ||
| Distribution | PPB | 97.25% | |
| BBB penetration | 0.012 | ||
| Metabolism | CYP1A2 inhibitor | 0.988 | |
| CYP2C19 inhibitor | 0.588 | ||
| CYP2C9 inhibitor | 0.602 | ||
| CYP2D6 inhibitor | 0.792 | ||
| CYP3A4 inhibitor | 0.833 | ||
| Excretion | CL | 7.022 | |
| T ½ | 0.856 | ||
| Toxicity | H-HT | 0.072 | |
| DILI | 0.854 | ||
| FDAMDD | 0.433 | ||
| Respiratory toxicity | 0.266 | ||
| Luteolin | Absorption | Caco-2 permeability | −5.208 |
| HIA | 0.047 | ||
| Distribution | PPB | 95.43% | |
| BBB penetration | 0.009 | ||
| Metabolism | CYP1A2 inhibitor | 0.981 | |
| CYP2C19 inhibitor | 0.124 | ||
| CYP2C9 inhibitor | 0.576 | ||
| CYP2D6 inhibitor | 0.559 | ||
| CYP3A4 inhibitor | 0.549 | ||
| Excretion | CL | 8.146 | |
| T ½ | 0.898 | ||
| Toxicity | H-HT | 0.084 | |
| DILI | 0.905 | ||
| FDAMDD | 0.741 | ||
| Respiratory toxicity | 0.22 | ||
| Naringenin | Absorption | Caco-2 permeability | −4.803 |
| HIA | 0.018 | ||
| Distribution | PPB | 93.76% | |
| BBB penetration | 0.042 | ||
| Metabolism | CYP1A2 inhibitor | 0.917 | |
| CYP2C19 inhibitor | 0.793 | ||
| CYP2C9 inhibitor | 0.823 | ||
| CYP2D6 inhibitor | 0.745 | ||
| CYP3A4 inhibitor | 0.855 | ||
| Excretion | CL | 17.388 | |
| T ½ | 0.774 | ||
| Toxicity | H-HT | 0.098 | |
| DILI | 0.853 | ||
| FDAMDD | 0.177 | ||
| Respiratory toxicity | 0.34 | ||
| Fisetin | Absorption | Caco-2 permeability | −4.987 |
| HIA | 0.009 | ||
| Distribution | PPB | 97.04% | |
| BBB penetration | 0.009 | ||
| Metabolism | CYP1A2 inhibitor | 0.95 | |
| CYP2C19 inhibitor | 0.097 | ||
| CYP2C9 inhibitor | 0.535 | ||
| CYP2D6 inhibitor | 0.532 | ||
| CYP3A4 inhibitor | 0.62 | ||
| Excretion | CL | 8.273 | |
| T ½ | 0.92 | ||
| Toxicity | H-HT | 0.127 | |
| DILI | 0.978 | ||
| FDAMDD | 0.259 | ||
| Respiratory toxicity | 0.074 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pandey, P.; Khan, F.; Seifeldin, S.A.; Alshaghdali, K.; Siddiqui, S.; Abdelwadoud, M.E.; Vyas, M.; Saeed, M.; Mazumder, A.; Saeed, A. Targeting Wnt/β-Catenin Pathway by Flavonoids: Implication for Cancer Therapeutics. Nutrients 2023, 15, 2088. https://doi.org/10.3390/nu15092088
Pandey P, Khan F, Seifeldin SA, Alshaghdali K, Siddiqui S, Abdelwadoud ME, Vyas M, Saeed M, Mazumder A, Saeed A. Targeting Wnt/β-Catenin Pathway by Flavonoids: Implication for Cancer Therapeutics. Nutrients. 2023; 15(9):2088. https://doi.org/10.3390/nu15092088
Chicago/Turabian StylePandey, Pratibha, Fahad Khan, Sara A. Seifeldin, Khalid Alshaghdali, Samra Siddiqui, Mohamed Elfatih Abdelwadoud, Manish Vyas, Mohd Saeed, Avijit Mazumder, and Amir Saeed. 2023. "Targeting Wnt/β-Catenin Pathway by Flavonoids: Implication for Cancer Therapeutics" Nutrients 15, no. 9: 2088. https://doi.org/10.3390/nu15092088
APA StylePandey, P., Khan, F., Seifeldin, S. A., Alshaghdali, K., Siddiqui, S., Abdelwadoud, M. E., Vyas, M., Saeed, M., Mazumder, A., & Saeed, A. (2023). Targeting Wnt/β-Catenin Pathway by Flavonoids: Implication for Cancer Therapeutics. Nutrients, 15(9), 2088. https://doi.org/10.3390/nu15092088








