Protein and Leucine Intake at Main Meals in Elderly People with Type 2 Diabetes
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Assessment of Clinical and Biochemical Parameters
2.3. Assessment of Dietary Habits
2.4. Statistical Analysis
3. Results
3.1. Characteristics of the Participants
3.2. Daily Energy and Nutrient Intake of the Study Population
3.3. Daily Energy, Macronutrient, and Leucine Intake at the Main Meals Consumed by the Study Population
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, Z.; Zhang, Z.; Ren, Y.; Wang, Y.; Fang, J.; Yue, H.; Ma, S.; Guan, F. Aging and age-related diseases: From mechanisms to therapeutic strategies. Biogerontology 2021, 22, 165–187. [Google Scholar] [CrossRef] [PubMed]
- Filippin, L.I.; Teixeira, V.N.; da Silva, M.P.; Miraglia, F.; da Silva, F.S. Sarcopenia: A predictor of mortality and the need for early diagnosis and intervention. Aging Clin. Exp. Res. 2015, 27, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Saeedi, P.; Petersohn, I.; Salpea, P.; Malanda, B.; Karuranga, S.; Unwin, N.; Colagiuri, S.; Guariguata, L.; Motala, A.A.; Ogurtsova, K.; et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res. Clin. Pract. 2019, 157, 107843. [Google Scholar] [CrossRef] [PubMed]
- Izzo, A.; Massimino, E.; Riccardi, G.; Della Pepa, G. A Narrative Review on Sarcopenia in Type 2 Diabetes Mellitus: Prevalence and Associated Factors. Nutrients 2021, 13, 183. [Google Scholar] [CrossRef]
- Veronese, N.; Stubbs, B.; Punzi, L.; Soysal, P.; Incalzi, R.A.; Saller, A.; Maggi, S. Effect of nutritional supplementations on physical performance and muscle strength parameters in older people: A systematic review and meta-analysis. Ageing Res. Rev. 2019, 51, 48–54. [Google Scholar] [CrossRef]
- Massimino, E.; Izzo, A.; Riccardi, G.; Della Pepa, G. The Impact of Glucose-Lowering Drugs on Sarcopenia in Type 2 Diabetes: Current Evidence and Underlying Mechanisms. Cells 2021, 10, 1958. [Google Scholar] [CrossRef]
- Traylor, D.A.; Gorissen, S.H.M.; Phillips, S.M. Perspective: Protein Requirements and Optimal Intakes in Aging: Are We Ready to Recommend More Than the Recommended Daily Allowance? Adv. Nutr. 2018, 9, 171–182. [Google Scholar] [CrossRef]
- Nygård, L.K.; Dahl, L.; Mundal, I.; Šaltytė Benth, J.; Rokstad, A.M.M. Protein Intake, Protein Mealtime Distribution and Seafood Consumption in Elderly Norwegians: Associations with Physical Function and Strength. Geriatrics 2020, 5, 100. [Google Scholar] [CrossRef]
- Houston, D.K.; Nicklas, B.J.; Ding, J.; Harris, T.B.; Tylavsky, F.A.; Newman, A.B.; Lee, J.S.; Sahyoun, N.R.; Visser, M.; Kritchevsky, S.B. Health ABC Study. Dietary protein intake is associated with lean mass change in older, community-dwelling adults: The Health, Aging, and Body Composition (Health ABC) Study. Am. J. Clin. Nutr. 2008, 87, 150–155. [Google Scholar] [CrossRef]
- Beasley, J.M.; Wertheim, B.C.; LaCroix, A.Z.; Prentice, R.L.; Neuhouser, M.L.; Tinker, L.F.; Kritchevsky, S.; Shikany, J.M.; Eaton, C.; Chen, Z.; et al. Biomarker-calibrated protein intake and physical function in the Women’s Health Initiative. J. Am. Geriatr. Soc. 2013, 61, 1863–1871. [Google Scholar] [CrossRef]
- Isanejad, M.; Mursu, J.; Sirola, J.; Kröger, H.; Rikkonen, T.; Tuppurainen, M.; Erkkilä, A.T. Dietary protein intake is associated with better physical function and muscle strength among elderly women. Br. J. Nutr. 2016, 115, 1281–1291. [Google Scholar] [CrossRef] [PubMed]
- Scott, D.; Blizzard, L.; Fell, J.; Giles, G.; Jones, G. Associations between dietary nutrient intake and muscle mass and strength in community-dwelling older adults: The Tasmanian Older Adult Cohort Study. J. Am. Geriatr. Soc. 2010, 58, 2129–2134. [Google Scholar] [CrossRef] [PubMed]
- Paulussen, K.J.M.; McKenna, C.F.; Beals, J.W.; Wilund, K.R.; Salvador, A.F.; Burd, N.A. Anabolic Resistance of Muscle Protein Turnover Comes in Various Shapes and Sizes. Front. Nutr. 2021, 8, 615849. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.R.; Churchward-Venne, T.A.; Witard, O.; Breen, L.; Burd, N.A.; Tipton, K.D.; Phillips, S.M. Protein ingestion to stimulate myofibrillar protein synthesis requires greater relative protein intakes in healthy older versus younger men. J. Gerontol. A Biol. Sci. Med. Sci. 2015, 70, 57–62. [Google Scholar] [CrossRef]
- Shad, B.J.; Wallis, G.; van Loon, L.J.; Thompson, J.L. Exercise prescription for the older population: The interactions between physical activity, sedentary time, and adequate nutrition in maintaining musculoskeletal health. Maturitas 2016, 93, 78–82. [Google Scholar] [CrossRef]
- Bauer, J.; Biolo, G.; Cederholm, T.; Cesari, M.; Cruz-Jentoft, A.J.; Morley, J.E.; Phillips, S.; Sieber, C.; Stehle, P.; Teta, D.; et al. Evidence-based recommendations for optimal dietary protein intake in older people: A position paper from the PROT-AGE Study Group. J. Am. Med. Dir. Assoc. 2013, 14, 542–559. [Google Scholar] [CrossRef]
- Deutz, N.E.; Bauer, J.M.; Barazzoni, R.; Biolo, G.; Boirie, Y.; Bosy-Westphal, A.; Cederholm, T.; Cruz-Jentoft, A.; Krznariç, Z.; Nair, K.S.; et al. Protein intake and exercise for optimal muscle function with aging: Recommendations from the ESPEN Expert Group. Clin. Nutr. 2014, 33, 929–936. [Google Scholar] [CrossRef]
- Mendonça, N.; Hengeveld, L.M.; Visser, M.; Presse, N.; Canhão, H.; Simonsick, E.M.; Kritchevsky, S.B.; Newman, A.B.; Gaudreau, P.; Jagger, C. Low protein intake, physical activity, and physical function in European and North American community-dwelling older adults: A pooled analysis of four longitudinal aging cohorts. Am. J. Clin. Nutr. 2021, 114, 29–41. [Google Scholar] [CrossRef]
- Phillips, S.M.; Martinson, W. Nutrient-rich, high-quality, protein-containing dairy foods in combination with exercise in aging persons to mitigate sarcopenia. Nutr. Rev. 2019, 77, 216–229. [Google Scholar] [CrossRef]
- Murphy, C.H.; Oikawa, S.Y.; Phillips, S.M. Dietary Protein to Maintain Muscle Mass in Aging: A Case for Per-meal Protein Recommendations. J. Frailty Aging 2016, 5, 49–58. [Google Scholar] [CrossRef]
- Devries, M.C.; McGlory, C.; Bolster, D.R.; Kamil, A.; Rahn, M.; Harkness, L.; Baker, S.K.; Phillips, S.M. Protein leucine content is a determinant of shorter- and longer-term muscle protein synthetic responses at rest and following resistance exercise in healthy older women: A randomized, controlled trial. Am. J. Clin. Nutr. 2018, 107, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Murphy, C.H.; Saddler, N.I.; Devries, M.C.; McGlory, C.; Baker, S.K.; Phillips, S.M. Leucine supplementation enhances integrative myofibrillar protein synthesis in free-living older men consuming lower- and higher-protein diets: A parallel-group crossover study. Am. J. Clin. Nutr. 2016, 104, 1594–1606. [Google Scholar] [CrossRef] [PubMed]
- Morley, J.E.; Malmstrom, T.K.; Rodriguez-Mañas, L.; Sinclair, A.J. Frailty, sarcopenia and diabetes. J. Am. Med. Dir. Assoc. 2014, 15, 853–859. [Google Scholar] [CrossRef] [PubMed]
- Fanelli, S.M.; Kelly, O.J.; Krok-Schoen, J.L.; Taylor, C.A. Low Protein Intakes and Poor Diet Quality Associate with Functional Limitations in US Adults with Diabetes: A 2005–2016 NHANES Analysis. Nutrients 2021, 13, 2582. [Google Scholar] [CrossRef] [PubMed]
- Henson, J.; Arsenyadis, F.; Redman, E.; Brady, E.M.; Coull, N.A.; Edwardson, C.L.; Hall, A.P.; James, L.J.; Khunti, K.; Rowlands, A.V.; et al. Relative protein intake and associations with markers of physical function in those with type 2 diabetes. Diabet. Med. 2022, 39, e14851. [Google Scholar] [CrossRef]
- Craig, C.L.; Marshall, A.L.; Sjöström, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef] [PubMed]
- Bailey, R.L. Overview of dietary assessment methods for measuring intakes of foods, beverages, and dietary supplements in research studies. Curr. Opin. Biotechnol. 2021, 70, 91–96. [Google Scholar] [CrossRef]
- Casey, P.H.; Goolsby, S.L.; Lensing, S.Y.; Perloff, B.P.; Bogle, M.L. The use of telephone interview methodology to obtain 24-hour dietary recalls. J. Am. Diet. Assoc. 1999, 99, 1406–1411. [Google Scholar] [CrossRef]
- Yamaoka, T.; Araki, A.; Tamura, Y.; Tanaka, S.; Fujihara, K.; Horikawa, C.; Aida, R.; Kamada, C.; Yoshimura, Y.; Moriya, T.; et al. Association between Low Protein Intake and Mortality in Patients with Type 2 Diabetes. Nutrients 2020, 12, 1629. [Google Scholar] [CrossRef]
- Lin, C.C.; Liu, C.S.; Li, C.I.; Lin, C.H.; Lin, W.Y.; Wang, M.C.; Yang, S.Y.; Li, T.C. Dietary Macronutrient Intakes and Mortality among Patients with Type 2 Diabetes. Nutrients 2020, 12, 1665. [Google Scholar] [CrossRef]
- Rousset, S.; Patureau Mirand, P.; Brandolini, M.; Martin, J.F.; Boirie, Y. Daily protein intakes and eating patterns in young and elderly French. Br. J. Nutr. 2003, 90, 1107–1115. [Google Scholar] [CrossRef] [PubMed]
- Tieland, M.; Borgonjen-Van den Berg, K.J.; Van Loon, L.J.; de Groot, L.C. Dietary Protein Intake in Dutch Elderly People: A Focus on Protein Sources. Nutrients 2015, 7, 9697–9706. [Google Scholar] [CrossRef] [PubMed]
- Smeuninx, B.; Greig, C.A.; Breen, L. Amount, Source and Pattern of Dietary Protein Intake Across the Adult Lifespan: A Cross-Sectional Study. Front. Nutr. 2020, 7, 25. [Google Scholar] [CrossRef] [PubMed]
- Berner, L.A.; Becker, G.; Wise, M.; Doi, J. Characterization of dietary protein among older adults in the United States: Amount, animal sources, and meal patterns. J. Acad. Nutr. Diet. 2013, 113, 809–815. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa-Takata, K.; Takimoto, H. Current protein and amino acid intakes among Japanese people: Analysis of the 2012 National Health and Nutrition Survey. Geriatr. Gerontol. Int. 2018, 18, 723–731. [Google Scholar] [CrossRef]
- Schlettwein-gsell, D.; Decarli, B.; de Groot, L. Meal patterns in the SENECA study of nutrition and the elderly in Europe: Assessment method and preliminary results on the role of the midday meal. Appetite 1999, 32, 15–22. [Google Scholar] [CrossRef]
- Cossu, M.; Chiavaroli, L.; Dall’Asta, M.; Francinelli, V.; Scazzina, F.; Brighenti, F. A nutritional evaluation of various typical Italian breakfast products: A comparison of macronutrient composition and glycaemic index values. Int. J. Food. Sci. Nutr. 2018, 69, 676–681. [Google Scholar] [CrossRef]
- Gaytán-González, A.; Ocampo-Alfaro, M.J.; Torres-Naranjo, F.; Arroniz-Rivera, M.; González-Mendoza, R.G.; Gil-Barreiro, M.; López-Taylor, J.R. The Consumption of Two or Three Meals per Day with Adequate Protein Content Is Associated with Lower Risk of Physical Disability in Mexican Adults Aged 60 Years and Older. Geriatrics 2020, 5, 1. [Google Scholar] [CrossRef]
- Farsijani, S.; Morais, J.A.; Payette, H.; Gaudreau, P.; Shatenstein, B.; Gray-Donald, K.; Chevalier, S. Relation between mealtime distribution of protein intake and lean mass loss in free-living older adults of the NuAge study. Am. J. Clin. Nutr. 2016, 104, 694–703. [Google Scholar] [CrossRef]
- Joint WHO/FAO/UNU Expert Consultation. Protein and amino acid requirements in human nutrition. In World Health Organization Technical Report Series; WHO: Geneva, Switzerland, 2007; Volume 935, pp. 1–265. [Google Scholar]
- Berrazaga, I.; Micard, V.; Gueugneau, M.; Walrand, S. The Role of the Anabolic Properties of Plant- versus Animal-Based Protein Sources in Supporting Muscle Mass Maintenance: A Critical Review. Nutrient 2019, 11, 1825. [Google Scholar] [CrossRef]
- Putra, C.; Konow, N.; Gage, M.; York, C.G.; Mangano, K.M. Protein Source and Muscle Health in Older Adults: A Literature Review. Nutrients 2021, 13, 743. [Google Scholar] [CrossRef]
- Lixandrão, M.E.; Longobardi, I.; Leitão, A.E.; Morais, J.V.M.; Swinton, P.A.; Aihara, A.Y.; Goes, P.C.K.; Ugrinowitsch, C.; Candow, D.G.; Gualano, B.; et al. Daily Leucine Intake Is Positively Associated with Lower Limb Skeletal Muscle Mass and Strength in the Elderly. Nutrients 2021, 13, 3536. [Google Scholar] [CrossRef] [PubMed]
- Gingrich, A.; Spiegel, A.; Kob, R.; Schoene, D.; Skurk, T.; Hauner, H.; Sieber, C.C.; Volkert, D.; Kiesswetter, E. Amount, Distribution, and Quality of Protein Intake Are Not Associated with Muscle Mass, Strength, and Power in Healthy Older Adults without Functional Limitations-An enable Study. Nutrients 2017, 9, 1358. [Google Scholar] [CrossRef] [PubMed]
- Volkert, D.; Beck, A.M.; Cederholm, T.; Cruz-Jentoft, A.; Goisser, S.; Hooper, L.; Kiesswetter, E.; Maggio, M.; Raynaud-Simon, A.; Sieber, C.C.; et al. ESPEN guideline on clinical nutrition and hydration in geriatrics. Clin. Nutr. 2019, 38, 10–47. [Google Scholar] [CrossRef]
- Mannucci, E.; Bartali, B.; Molino Lova, R.; Papucci, M.; Lauretani, F.; Luisi, M.L.; Pietrobelli, A.; Macchi, C. Eating habits in elderly diabetic subjects: Assessment in the InCHIANTI Study. Nutr. Metab. Cardiovasc. Dis. 2008, 18, 278–282. [Google Scholar] [CrossRef] [PubMed]
- Virtanen, S.M.; Feskens, E.J.; Räsänen, L.; Fidanza, F.; Tuomilehto, J.; Giampaoli, S.; Nissinen, A.; Kromhout, D. Comparison of diets of diabetic and non-diabetic elderly men in Finland, The Netherlands and Italy. Eur. J. Clin. Nutr. 2000, 54, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Bamia, C.; Orfanos, P.; Ferrari, P.; Overvad, K.; Hundborg, H.H.; Tjønneland, A.; Olsen, A.; Kesse, E.; Boutron-Ruault, M.C.; Clavel-Chapelon, F.; et al. Dietary patterns among older Europeans: The EPIC-Elderly study. Br. J. Nutr. 2005, 94, 100–113. [Google Scholar] [CrossRef]
- Rondanelli, M.; Nichetti, M.; Peroni, G.; Faliva, M.A.; Naso, M.; Gasparri, C.; Perna, S.; Oberto, L.; Di Paolo, E.; Riva, A.; et al. Where to Find Leucine in Food and How to Feed Elderly with Sarcopenia in Order to Counteract Loss of Muscle Mass: Practical Advice. Front. Nutr. 2021, 7, 622391. [Google Scholar] [CrossRef]
- Verreijen, A.M.; van den Helder, J.; Streppel, M.T.; Rotteveel, I.; Heman, D.; van Dronkelaar, C.; Memelink, R.G.; Engberink, M.F.; Visser, M.; Tieland, M.; et al. A higher protein intake at breakfast and lunch is associated with a higher total daily protein intake in older adults: A post-hoc cross-sectional analysis of four randomised controlled trials. J. Hum. Nutr. Diet. 2021, 34, 384–394. [Google Scholar] [CrossRef]
- Aoyama, S.; Kim, H.K.; Hirooka, R.; Tanaka, M.; Shimoda, T.; Chijiki, H.; Kojima, S.; Sasaki, K.; Takahashi, K.; Makino, S.; et al. Distribution of dietary protein intake in daily meals influences skeletal muscle hypertrophy via the muscle clock. Cell Rep. 2021, 36, 109336. [Google Scholar] [CrossRef]
- Park, Y.M.; Heden, T.D.; Liu, Y.; Nyhoff, L.M.; Thyfault, J.P.; Leidy, H.J.; Kanaley, J.A. A high-protein breakfast induces greater insulin and glucose-dependent insulinotropic peptide responses to a subsequent lunch meal in individuals with type 2 diabetes. J. Nutr. 2015, 145, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Rabinovitz, H.R.; Boaz, M.; Ganz, T.; Jakubowicz, D.; Matas, Z.; Madar, Z.; Wainstein, J. Big breakfast rich in protein and fat improves glycemic control in type 2 diabetics. Obesity 2014, 22, E46–E54. [Google Scholar] [CrossRef]
- Kirwan, R.P.; Mazidi, M.; Rodríguez García, C.; Lane, K.E.; Jafari, A.; Butler, T.; Perez de Heredia, F.; Davies, I.G. Protein interventions augment the effect of resistance exercise on appendicular lean mass and handgrip strength in older adults: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2022, 115, 897–913. [Google Scholar] [CrossRef] [PubMed]
- Peterson, M.D.; Sen, A.; Gordon, P.M. Influence of resistance exercise on lean body mass in aging adults: A meta-analysis. Med. Sci. Sports Exerc. 2011, 43, 249–258. [Google Scholar] [CrossRef] [PubMed]
| Variables | All Patients (n = 138) |
|---|---|
| Sex (men/women) | 91/47 |
| Age (years) | 72 ± 4 |
| BMI (kg/m2) | 29.4 ± 4.7 |
| Waist circumference (cm) | 102 ± 11 |
| Obese (%) | 42% |
| Smoking (%) | 24 |
| Inactive * (%) | 64 |
| Duration of diabetes (years) | 18 ± 9 |
| HbA1c (%) | 7.1 ± 1.0 |
| HDL-Cholesterol (mg/dL) | 49 ± 12 |
| LDL-Cholesterol (mg/dL) | 89 ± 29 |
| Triglycerides (mg/dL) | 124 ± 59 |
| Hypertension (%) | 86 |
| Dyslipidemia (%) | 91 |
| Retinopathy (%) | 21 |
| Neuropathy (%) | 23 |
| Nephropathy (%) | 24 |
| Cardiovascular diseases (%) | 36 |
| Glucose lowering therapy | |
| 69 |
| 28 |
| 16 |
| 2 |
| 12 |
| 21 |
| 41 |
| Anti-hypertensive therapy (%) | 86 |
| Lipid-lowering therapy (%) | 85 |
| Variables | All patients (n = 138) | Men (n = 91) | Women (n = 47) | p | |
|---|---|---|---|---|---|
| Energy | (Kcal/day) | 1414 ± 342 | 1462 ± 345 | 1322 ± 320 | 0.022 |
| Protein | (g/kg BW/day) | 0.9 ± 0.2 | 0.9 ± 0.2 | 1.0 ± 0.3 | 0.029 |
| (g/day) | 57 ± 16 | 57 ± 16 | 55 ± 14 | 0.493 | |
| (%TEI) | 17 ± 2.9 | 17 ± 2.9 | 17 ± 2.9 | 0.232 | |
| Vegetal protein | (g/day) | 25 ± 7.2 | 25 ± 7.3 | 23 ± 7.1 | 0.140 |
| Animal protein | (g/day) | 32 ± 14 | 32 ± 15 | 32 ± 12 | 0.962 |
| Leucine | (mg/day) | 4383 ± 1484 | 4348 ± 1292 | 4450 ± 1810 | 0.702 |
| Isoleucine | (mg/day) | 2447 ± 1113 | 2396 ± 740 | 2545 ± 1608 | 0.458 |
| Valine | (mg/day) | 2879 ± 1275 | 2826 ± 850 | 2981 ± 1842 | 0.500 |
| Fat | (g/day) | 53 ± 19 | 55 ± 20 | 50 ± 17 | 0.106 |
| (%TEI) | 35 ± 6.5 | 35 ± 6.6 | 34 ± 6.2 | 0.509 | |
| Carbohydrates | (g/day) | 167 ± 41 | 171 ± 40 | 159 ± 42 | 0.116 |
| (%TEI) | 48 ± 7.2 | 48 ± 7.2 | 48 ± 7.3 | 0.808 | |
| Simple sugars | (g/day) | 54 ± 20 | 53 ± 20 | 57 ± 21 | 0.237 |
| Fibre | (g/day) | 16 ± 6.1 | 16 ± 5.9 | 16 ± 6.5 | 0.570 |
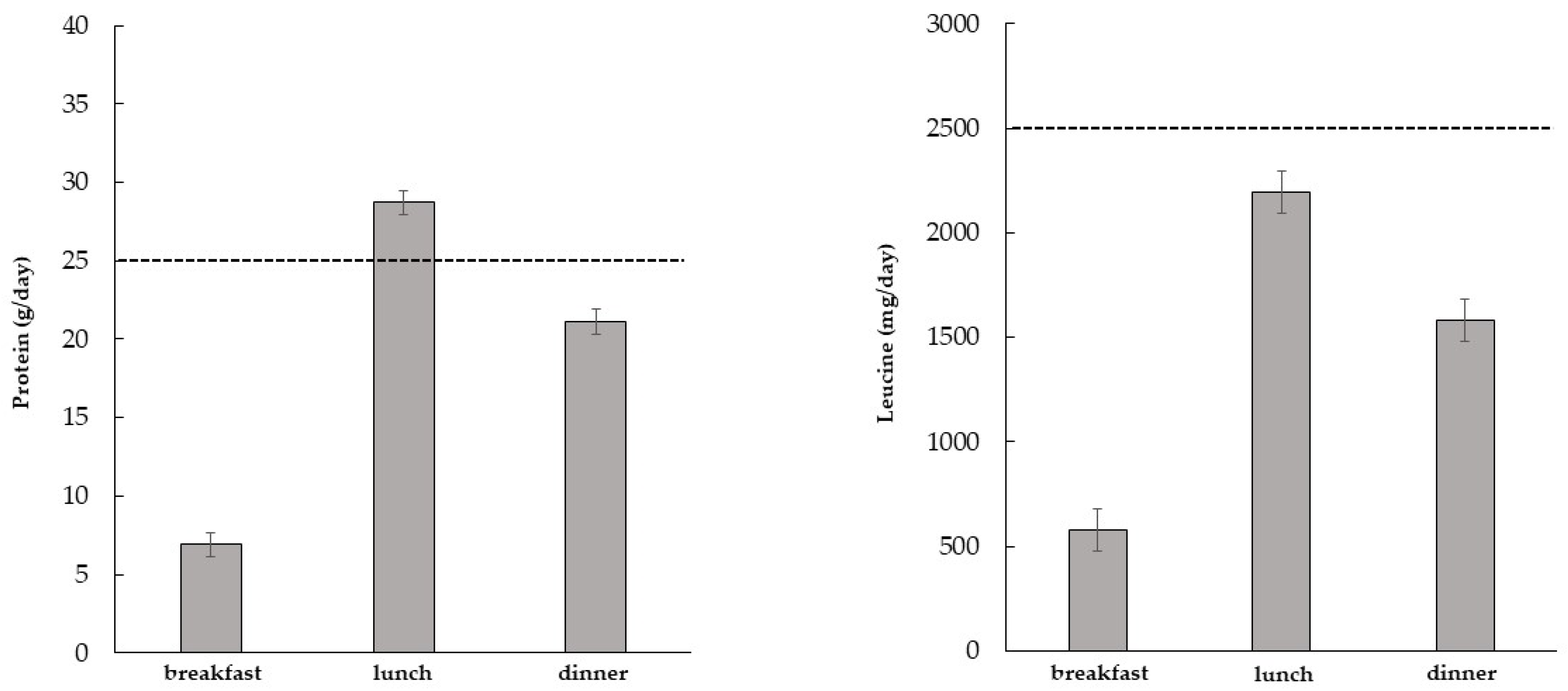
| Variables | Breakfast | Lunch | Dinner | |||
|---|---|---|---|---|---|---|
| Men (n = 91) | Women (n = 47) | Men (n = 91) | Women (n = 47) | Men (n = 91) | Women (n = 47) | |
| Energy (Kcal/day) | 175 ± 94 | 174 ± 81 | 763 ± 209 | 647 ± 210 ** | 510 ± 200 | 444 ± 161 * |
| Protein (g/day) | 6.8 ± 3.7 | 7.1 ± 3.7 | 29 ± 11 | 28 ± 9.0 | 22 ± 11 | 20 ± 9.0 |
| Vegetal protein (g/day) | 2.7 ± 1.7 | 2.5 ± 1.4 | 14 ± 4.7 | 14 ± 4.4 | 8.0 ± 4.3 | 6.5 ± 3.6 * |
| Animal protein (g/day) | 3.9 ± 2.9 | 4.5 ± 3.1 | 15 ± 10 | 14 ± 8.0 | 14 ± 9.9 | 13 ± 8.4 |
| Fat (g/day) | 4.2 ± 4.0 | 4.5 ± 3.8 | 29 ± 11 | 25 ± 12 * | 22 ± 12 | 20 ±9.4 |
| Carbohydrates (g/day) | 29 ± 15 | 28 ± 11 | 87 ± 25 | 75 ± 24 ** | 51 ± 20 | 45 ± 22 |
| Simple sugars (g/day) | 13 ± 8.0 | 13 ± 7.0 | 20 ± 10 | 18 ± 11 | 16 ± 8.4 | 17 ± 9.7 |
| Fibre (g/day) | 1.1 ± 1.0 | 1.1 ± 1.1 | 8.2 ± 3.1 | 8.3 ± 4.0 | 5.9 ± 3.5 | 5.9 ± 2.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Massimino, E.; Izzo, A.; Castaldo, C.; Amoroso, A.P.; Rivellese, A.A.; Capaldo, B.; Della Pepa, G. Protein and Leucine Intake at Main Meals in Elderly People with Type 2 Diabetes. Nutrients 2023, 15, 1345. https://doi.org/10.3390/nu15061345
Massimino E, Izzo A, Castaldo C, Amoroso AP, Rivellese AA, Capaldo B, Della Pepa G. Protein and Leucine Intake at Main Meals in Elderly People with Type 2 Diabetes. Nutrients. 2023; 15(6):1345. https://doi.org/10.3390/nu15061345
Chicago/Turabian StyleMassimino, Elena, Anna Izzo, Carmen Castaldo, Anna Paola Amoroso, Angela Albarosa Rivellese, Brunella Capaldo, and Giuseppe Della Pepa. 2023. "Protein and Leucine Intake at Main Meals in Elderly People with Type 2 Diabetes" Nutrients 15, no. 6: 1345. https://doi.org/10.3390/nu15061345
APA StyleMassimino, E., Izzo, A., Castaldo, C., Amoroso, A. P., Rivellese, A. A., Capaldo, B., & Della Pepa, G. (2023). Protein and Leucine Intake at Main Meals in Elderly People with Type 2 Diabetes. Nutrients, 15(6), 1345. https://doi.org/10.3390/nu15061345







