Beta-Hydroxybutyrate (BHB), Glucose, Insulin, Octanoate (C8), and Decanoate (C10) Responses to a Medium-Chain Triglyceride (MCT) Oil with and without Glucose: A Single-Center Study in Healthy Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Test Ingredients (MCT and Glucose)
2.3. Experimental Procedure
2.4. Plasma Metabolite Analyses
2.5. Questionnaire on Satiety and Tolerability
2.6. Cognitive Test
2.7. Statistical Analysis
3. Results
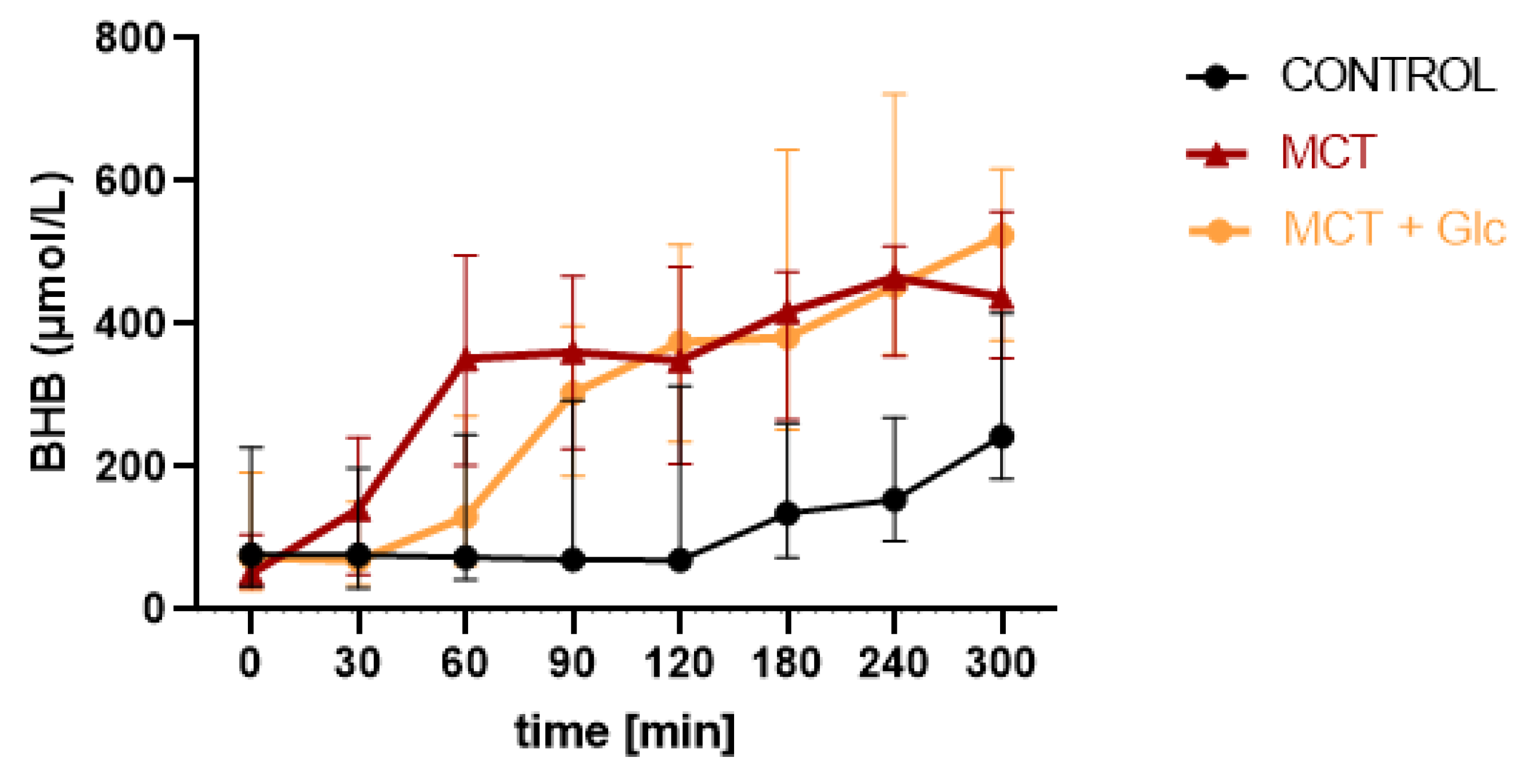
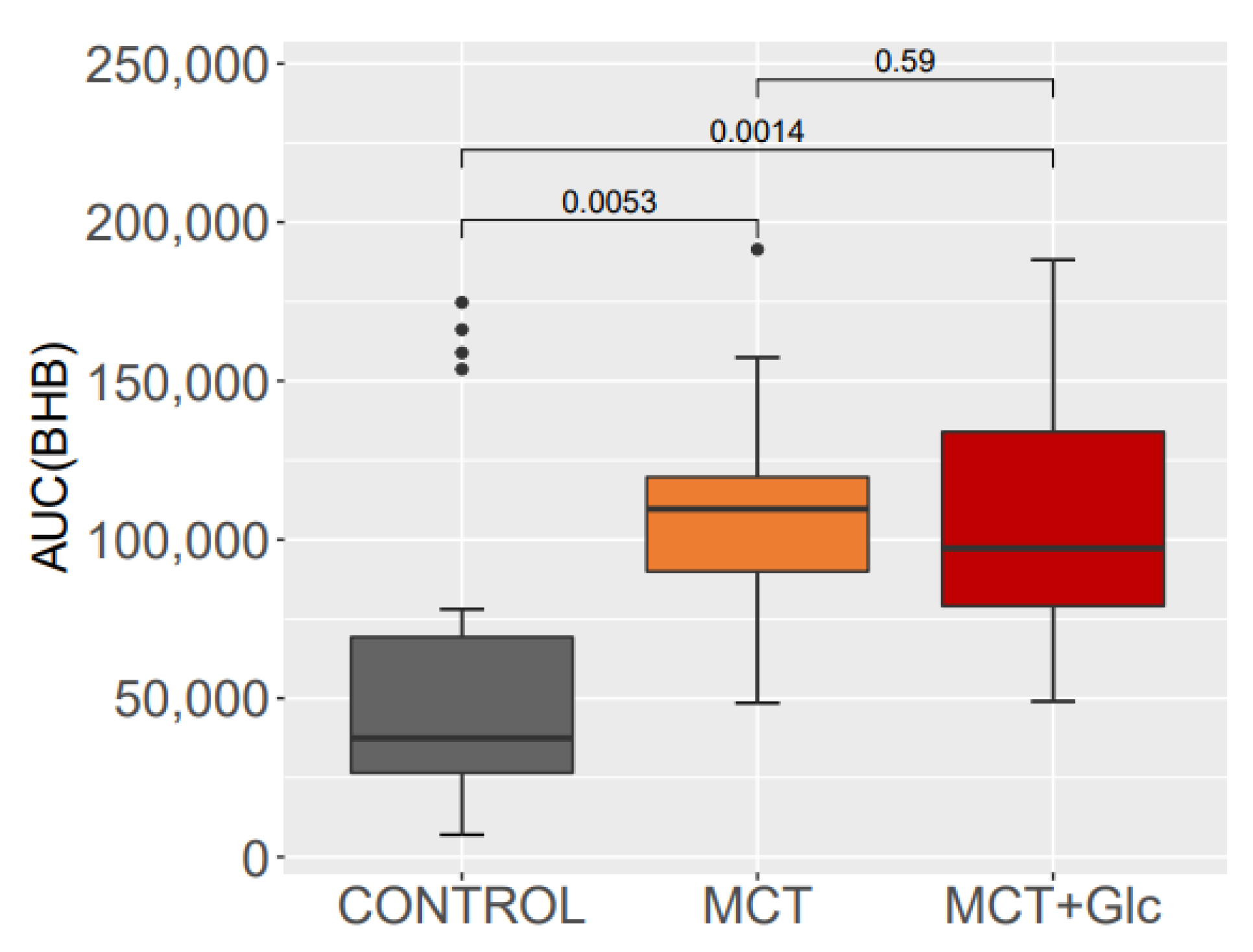
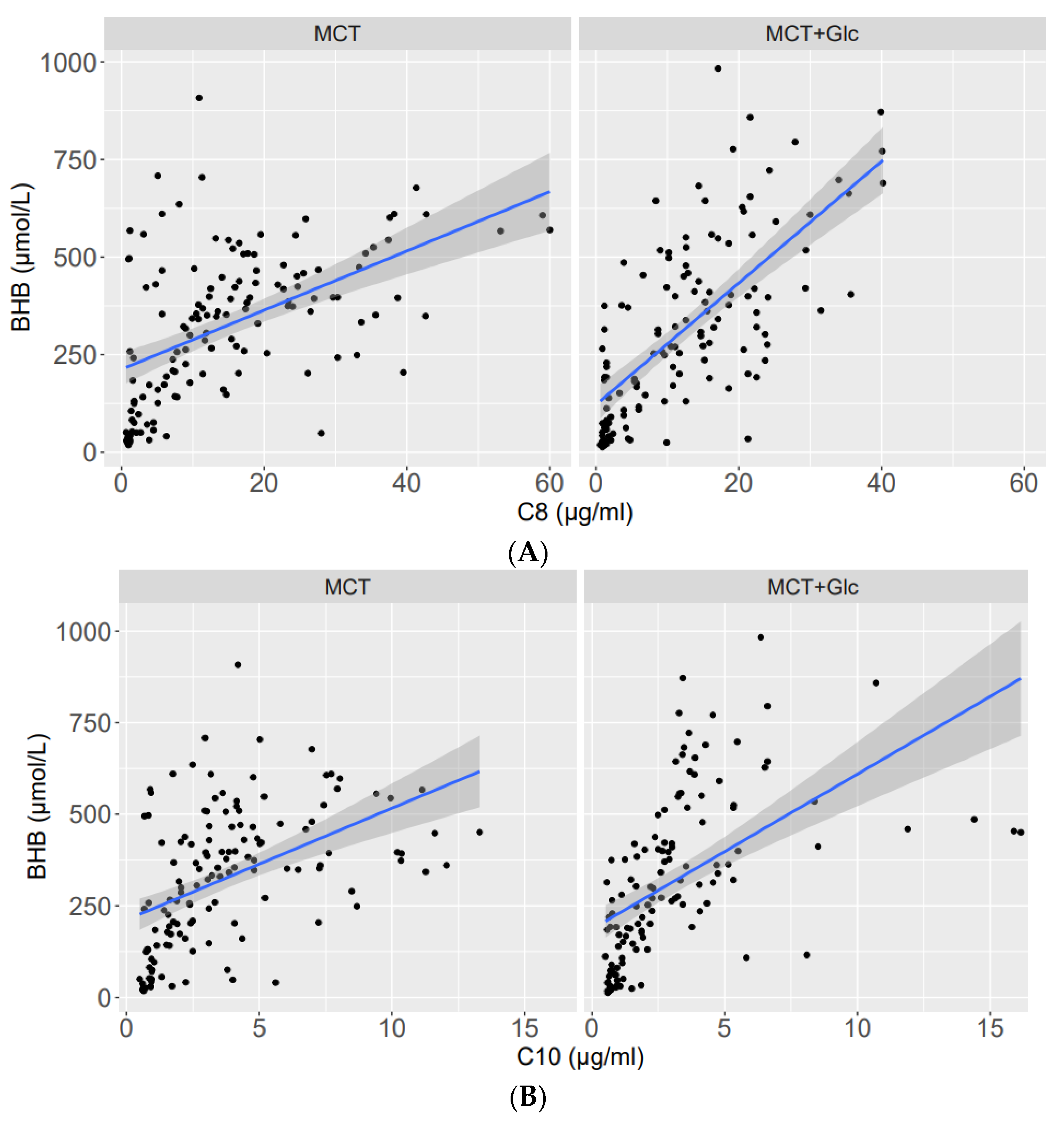
3.1. Beta-Hydroxybutyrate
3.1.1. Responses to MCT Oil Alone
3.1.2. MCT Oil Alone versus MCT Plus Glucose
3.1.3. Responses to MCT Oil Plus Glucose
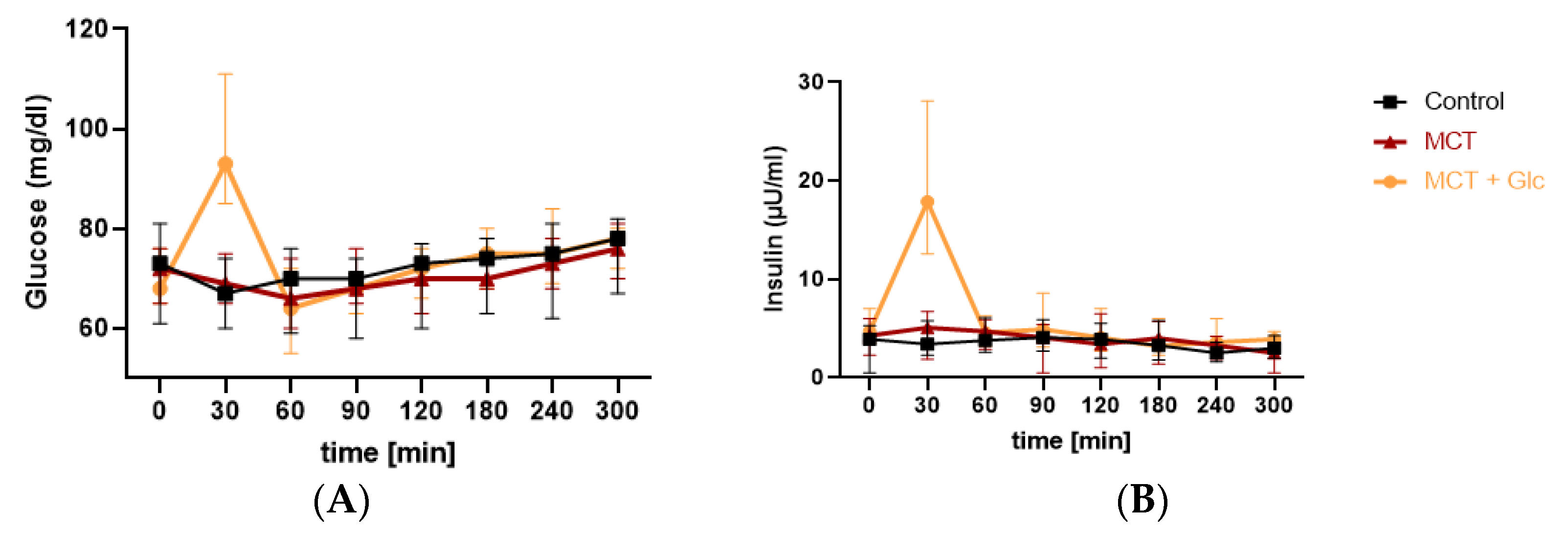
3.2. Glucose Responses
Responses to MCT Oil Alone
3.3. Insulin Responses
3.3.1. Responses to MCT Oil Alone
3.3.2. Responses to MCT Oil Plus Glucose
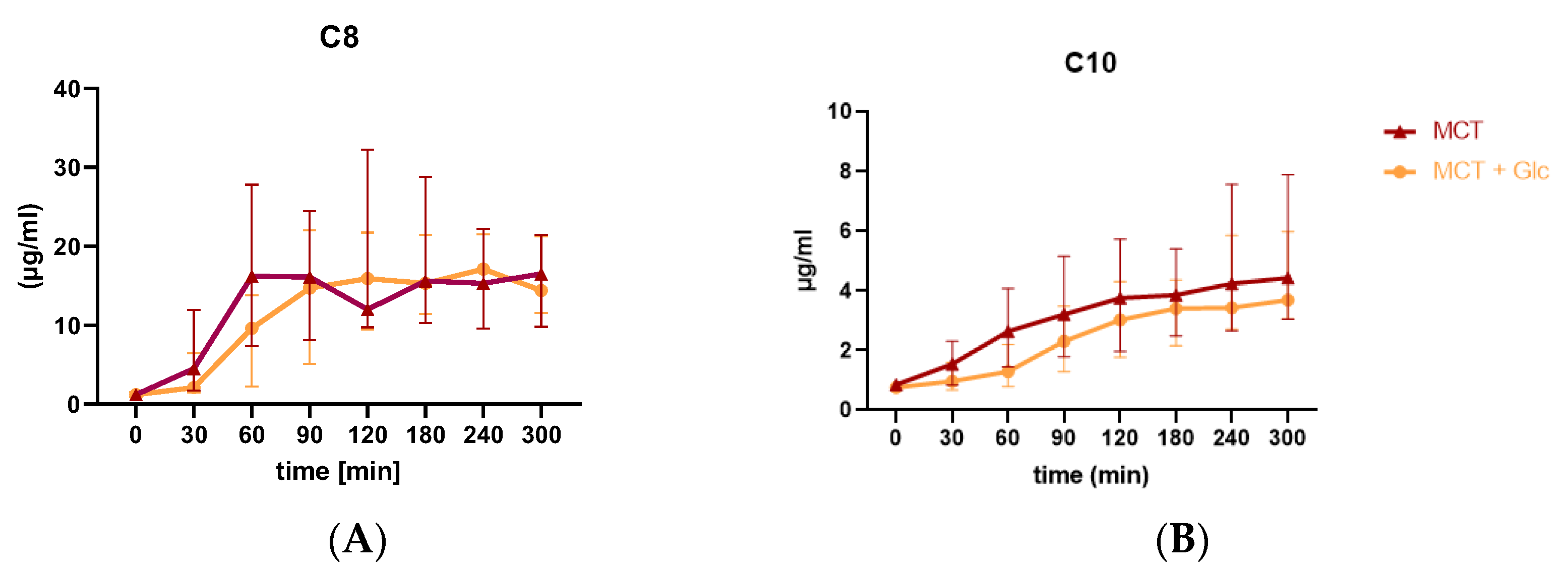
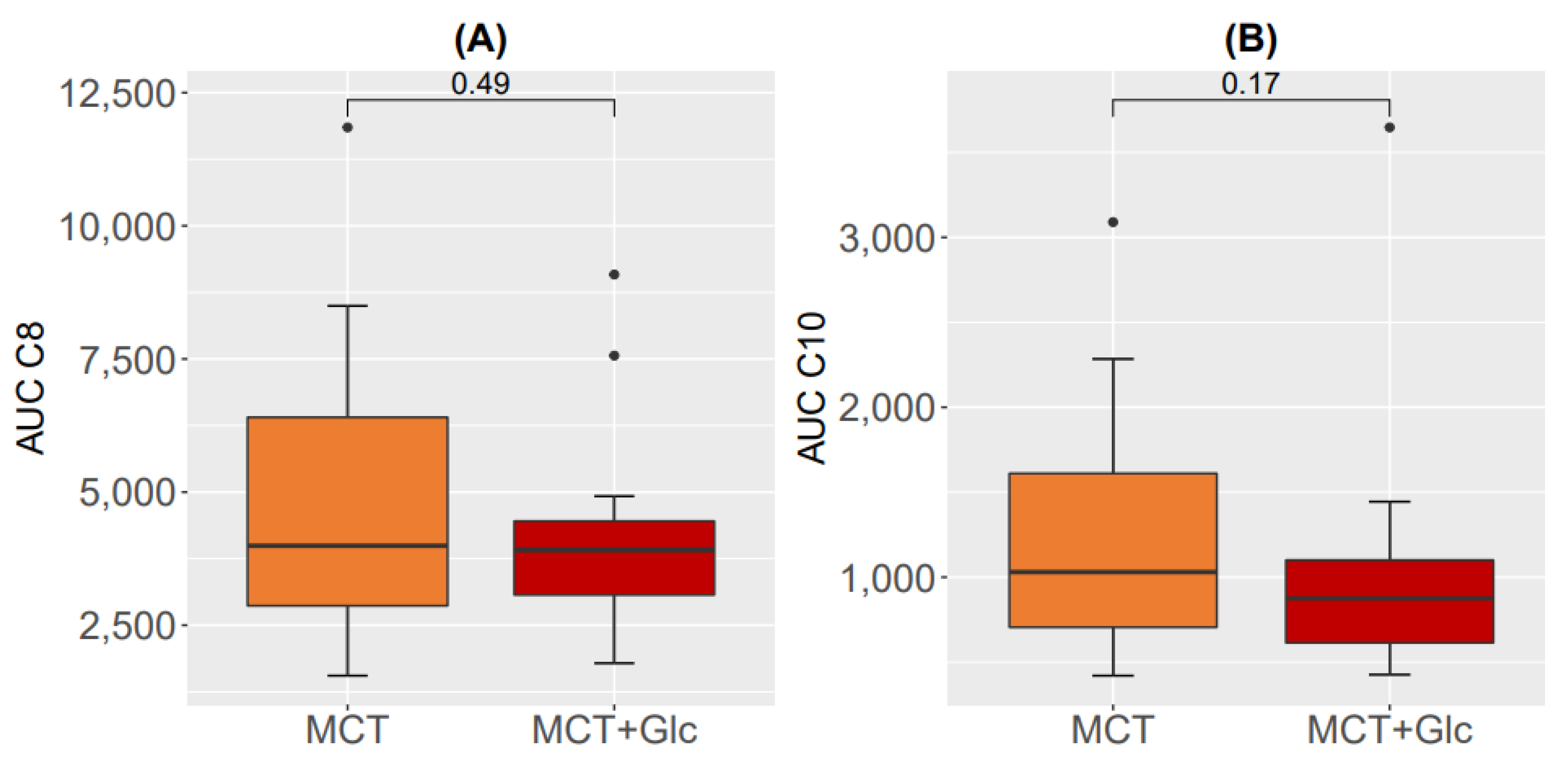
3.4. Plasma C8 and C10 Levels and Area under the Curve for C8 and C10 Response
3.4.1. Responses to MCT Oil Alone
3.4.2. Responses to MCT Oil Plus Glucose
3.5. Satiety and Side Effects
3.5.1. Satiety
3.5.2. Side Effects
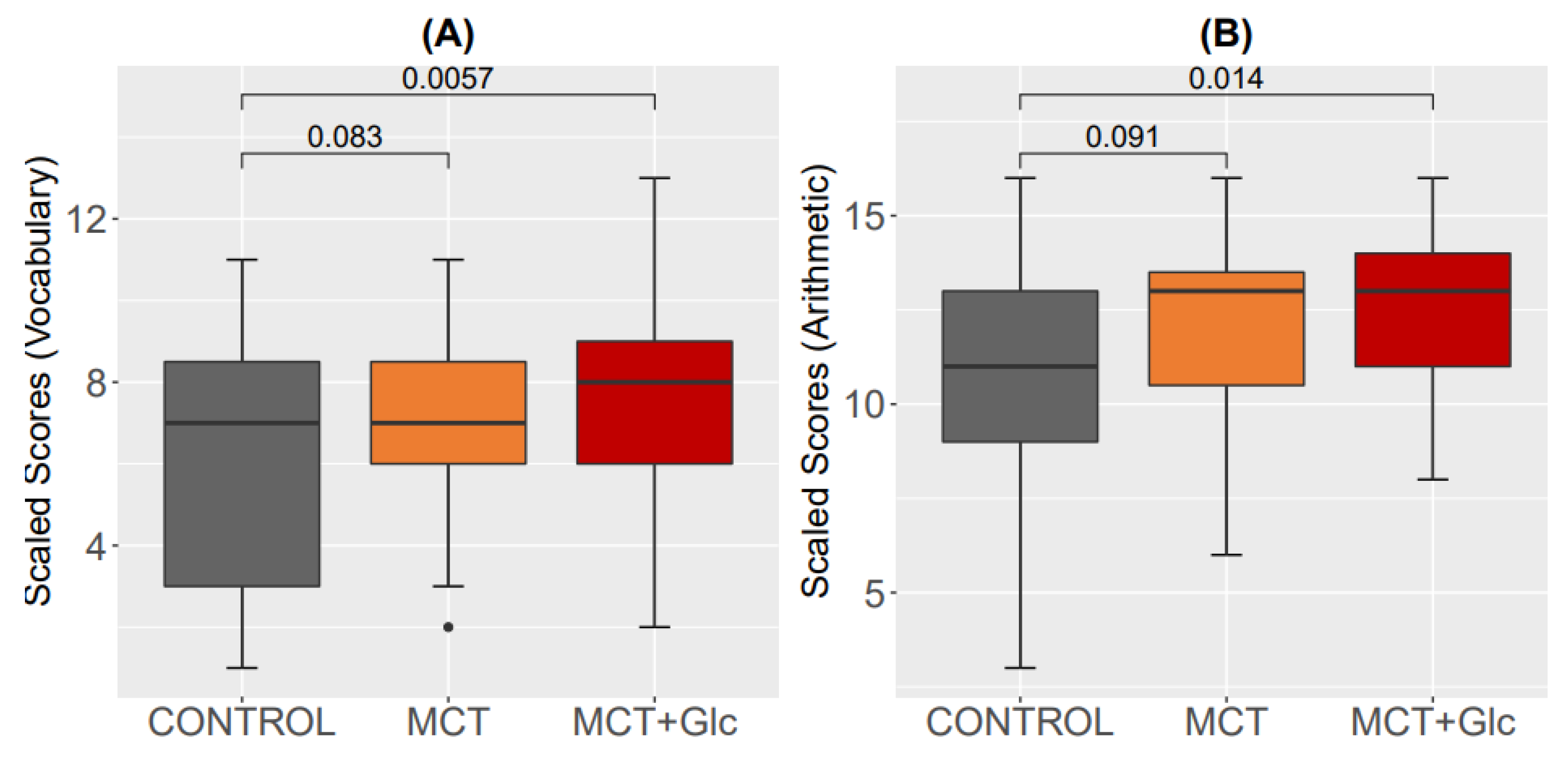
3.6. Cognitive Assessment
3.6.1. Subtests “Vocabulary”
3.6.2. Subtest “Arithmetic”
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bach, A.C.; Babayan, V.K. Medium-chain triglycerides: An update. Am. J. Clin. Nutr. 1982, 36, 950–962. [Google Scholar] [CrossRef] [PubMed]
- Babayan, V.K. Medium chain triglycerides and structured lipids. Lipids 1987, 22, 417–420. [Google Scholar] [CrossRef]
- Bodkowski, R.; Czyż, K.; Kupczyński, R.; Patkowska-Sokoła, B.; Nowakowski, P.; Wiliczkiewicz, A. Lipid complex effect on fatty acid profile and chemical composition of cow milk and cheese. J. Dairy Sci. 2016, 99, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Edem, D.O. Palm oil: Biochemical, physiological, nutritional, hematological, and toxicological aspects: A review. Plant Foods Hum. Nutr. 2002, 57, 319–341. [Google Scholar] [CrossRef]
- Traul, K.A.; Driedger, A.; Ingle, D.L.; Nakhasi, D. Review of the toxicologic properties of medium-chain triglycerides. Food Chem. Toxicol. 2000, 38, 79–98. [Google Scholar] [CrossRef]
- Marten, B.; Pfeuffer, M.; Schrezenmeir, J. Medium-chain triglycerides. Int. Dairy J. 2006, 16, 1374–1382. [Google Scholar] [CrossRef]
- Dai, X.; Yuan, T.; Zhang, X.; Zhou, Q.; Bi, H.; Yu, R.; Wei, W.; Wang, X. Short-chain fatty acid (SCFA) and medium-chain fatty acid (MCFA) concentrations in human milk consumed by infants born at different gestational ages and the variations in concentration during lactation stages. Food Funct. 2020, 11, 1869–1880. [Google Scholar] [CrossRef] [PubMed]
- Schönfeld, P.; Wojtczak, L. Short- and medium-chain fatty acids in energy metabolism: The cellular perspective. J. Lipid Res. 2016, 57, 943–954. [Google Scholar] [CrossRef]
- Miyagawa, Y.; Mori, T.; Goto, K.; Kawahara, I.; Fujiwara-Tani, R.; Kishi, S.; Sasaki, T.; Fujii, K.; Ohmori, H.; Kuniyasu, H. Intake of medium-chain fatty acids induces myocardial oxidative stress and atrophy. Lipids Health Dis. 2018, 17, 258. [Google Scholar] [CrossRef]
- Nguyen, P.; Leray, V.; Diez, M.; Serisier, S.; Le Bloc’h, J.; Siliart, B.; Dumon, H. Liver lipid metabolism. J. Anim. Physiol. Anim. Nutr. 2008, 92, 272–283. [Google Scholar] [CrossRef]
- Shcherbakova, K.; Schwarz, A.; Apryatin, S.; Karpenko, M.; Trofimov, A. Supplementation of Regular Diet With Medium-Chain Triglycerides for Procognitive Effects: A Narrative Review. Front. Nutr. 2022, 9, 934497. [Google Scholar] [CrossRef]
- Juby, A.G.; Brocks, D.R.; Jay, D.A.; Davis, C.M.J.; Mager, D.R. Assessing the Impact of Factors that Influence the Ketogenic Response to Varying Doses of Medium Chain Triglyceride (MCT) Oil. J. Prev. Alzheimers Dis. 2021, 8, 19–28. [Google Scholar] [CrossRef]
- Courchesne-Loyer, A.; Fortier, M.; Tremblay-Mercier, J.; Chouinard-Watkins, R.; Roy, M.; Nugent, S.; Castellano, C.A.; Cunnane, S.C. Stimulation of mild, sustained ketonemia by medium-chain triacylglycerols in healthy humans: Estimated potential contribution to brain energy metabolism. Nutrition 2013, 29, 635–640. [Google Scholar] [CrossRef]
- Fortier, M.; Castellano, C.A.; St-Pierre, V.; Myette-Côté, É.; Langlois, F.; Roy, M.; Morin, M.C.; Bocti, C.; Fulop, T.; Godin, J.P.; et al. A ketogenic drink improves cognition in mild cognitive impairment: Results of a 6-month RCT. Alzheimers Dement. 2021, 17, 543–552. [Google Scholar] [CrossRef]
- Roy, M.; Fortier, M.; Rheault, F.; Edde, M.; Croteau, E.; Castellano, C.A.; Langlois, F.; St-Pierre, V.; Cuenoud, B.; Bocti, C.; et al. A ketogenic supplement improves white matter energy supply and processing speed in mild cognitive impairment. Alzheimers Dement. 2021, 7, e12217. [Google Scholar] [CrossRef] [PubMed]
- Ota, M.; Matsuo, J.; Ishida, I.; Takano, H.; Yokoi, Y.; Hori, H.; Yoshida, S.; Ashida, K.; Nakamura, K.; Takahashi, T.; et al. Effects of a medium-chain triglyceride-based ketogenic formula on cognitive function in patients with mild-to-moderate Alzheimer’s disease. Neurosci. Lett. 2019, 690, 232–236. [Google Scholar] [CrossRef]
- Cunnane, S.C.; Courchesne-Loyer, A.; Vandenberghe, C.; St-Pierre, V.; Fortier, M.; Hennebelle, M.; Croteau, E.; Bocti, C.; Fulop, T.; Castellano, C.A. Can Ketones Help Rescue Brain Fuel Supply in Later Life? Implications for Cognitive Health during Aging and the Treatment of Alzheimer’s Disease. Front. Mol. Neurosci. 2016, 9, 53. [Google Scholar] [CrossRef] [PubMed]
- Cunnane, S.C.; Trushina, E.; Morland, C.; Prigione, A.; Casadesus, G.; Andrews, Z.B.; Beal, M.F.; Bergersen, L.H.; Brinton, R.D.; de la Monte, S.; et al. Brain energy rescue: An emerging therapeutic concept for neurodegenerative disorders of ageing. Nat. Rev. Drug Discov. 2020, 19, 609–633. [Google Scholar] [CrossRef]
- Freund, G.; Weinsier, R.L. Standardized ketosis in man following medium chain triglyceride ingestion. Metabolism 1966, 15, 980–991. [Google Scholar] [CrossRef] [PubMed]
- Kolb, S.; Sailer, D. Effect of fat emulsions containing medium-chain triglycerides and glucose on ketone body production and excretion. JPEN J. Parenter. Enteral. Nutr. 1984, 8, 285–289. [Google Scholar] [CrossRef]
- Dubois, B.; Feldman, H.H.; Jacova, C.; Dekosky, S.T.; Barberger-Gateau, P.; Cummings, J.; Delacourte, A.; Galasko, D.; Gauthier, S.; Jicha, G.; et al. Research criteria for the diagnosis of Alzheimer’s disease: Revising the NINCDS-ADRDA criteria. Lancet Neurol. 2007, 6, 734–746. [Google Scholar] [CrossRef]
- Courchesne-Loyer, A.; Lowry, C.M.; St-Pierre, V.; Vandenberghe, C.; Fortier, M.; Castellano, C.A.; Wagner, J.R.; Cunnane, S.C. Emulsification Increases the Acute Ketogenic Effect and Bioavailability of Medium-Chain Triglycerides in Humans: Protein, Carbohydrate, and Fat Metabolism. Curr. Dev. Nutr. 2017, 1, e000851. [Google Scholar] [CrossRef] [PubMed]
- Vandenberghe, C.; St-Pierre, V.; Pierotti, T.; Fortier, M.; Castellano, C.A.; Cunnane, S.C. Tricaprylin Alone Increases Plasma Ketone Response More Than Coconut Oil or Other Medium-Chain Triglycerides: An Acute Crossover Study in Healthy Adults. Curr. Dev. Nutr. 2017, 1, e000257. [Google Scholar] [CrossRef] [PubMed]
- Kossoff, E.H.; Zupec-Kania, B.A.; Auvin, S.; Ballaban-Gil, K.R.; Christina Bergqvist, A.G.; Blackford, R.; Buchhalter, J.R.; Caraballo, R.H.; Cross, J.H.; Dahlin, M.G.; et al. Optimal clinical management of children receiving dietary therapies for epilepsy: Updated recommendations of the International Ketogenic Diet Study Group. Epilepsia Open 2018, 3, 175–192. [Google Scholar] [CrossRef]
- Bartolucci, G.; Pallecchi, M.; Menicatti, M.; Moracci, L.; Pucciarelli, S.; Agostini, M.; Crotti, S. A method for assessing plasma free fatty acids from C2 to C18 and its application for the early detection of colorectal cancer. J. Pharm. Biomed. Anal. 2022, 215, 114762. [Google Scholar] [CrossRef] [PubMed]
- Ward, L.C.; Bergman, M.A.; Hebert, K.R. WAIS-IV subtest covariance structure: Conceptual and statistical considerations. Psychol. Assess. 2012, 24, 328–340. [Google Scholar] [CrossRef] [PubMed]
- Wechsler, D. Wechsler Adult Intelligence Scale–Fourth Edition (WAIS–IV); NCS Pearson: San Antonio, TX, USA, 2008; Volume 22, p. 1. [Google Scholar]
- Wlodarek, D. The possibility of use of the ketogenic diet and medium chain triglycerides supplementation in the support therapy of Alzheimer disease. Curr. Opin. Clin. Nutr. Metab. Care 2021, 24, 385–391. [Google Scholar] [CrossRef]
- Huttenlocher, P.; Wilbourn, A.; Signore, J. Medium-chain triglycerides as a therapy for intractable childhood epilepsy. Neurology 1971, 21, 1097–1103. [Google Scholar] [CrossRef]
- Lambrechts, D.A.; de Kinderen, R.J.; Vles, H.S.; de Louw, A.J.; Aldenkamp, A.P.; Majoie, M.J. The MCT-ketogenic diet as a treatment option in refractory childhood epilepsy: A prospective study with 2-year follow-up. Epilepsy Behav. 2015, 51, 261–266. [Google Scholar] [CrossRef]
- Neal, E.G.; Chaffe, H.; Schwartz, R.H.; Lawson, M.S.; Edwards, N.; Fitzsimmons, G.; Whitney, A.; Cross, J.H. A randomized trial of classical and medium-chain triglyceride ketogenic diets in the treatment of childhood epilepsy. Epilepsia 2009, 50, 1109–1117. [Google Scholar] [CrossRef]
- Liu, Y.-M.; Wang, H.-S. Medium-chain triglyceride ketogenic diet, an effective treatment for drug-resistant epilepsy and a comparison with other ketogenic diets. Biomed J. 2013, 36, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-M.C.; Williams, S.; Basualdo-Hammond, C.; Stephens, D.; Curtis, R. A prospective study: Growth and nutritional status of children treated with the ketogenic diet. J. Am. Diet. Assoc. 2003, 103, 707–712. [Google Scholar] [CrossRef] [PubMed]
- Harvey, C.J.; Schofield, G.M.; Williden, M.; McQuillan, J.A. The effect of medium chain triglycerides on time to nutritional ketosis and symptoms of keto-induction in healthy adults: A randomised controlled clinical trial. J. Nutr. Metab. 2018, 2018. [Google Scholar] [CrossRef]
- Yurista, S.R.; Chong, C.-R.; Badimon, J.J.; Kelly, D.P.; de Boer, R.A.; Westenbrink, B.D. Therapeutic potential of ketone bodies for patients with cardiovascular disease: JACC state-of-the-art review. J. Am. Coll. Cardiol. 2021, 77, 1660–1669. [Google Scholar] [CrossRef] [PubMed]
- Witters, L.; Kemp, B. Insulin activation of acetyl-CoA carboxylase accompanied by inhibition of the 5′-AMP-activated protein kinase. J. Biol. Chem. 1992, 267, 2864–2867. [Google Scholar] [CrossRef]
- Bernini, A.; Masoodi, M.; Solari, D.; Miroz, J.-P.; Carteron, L.; Christinat, N.; Morelli, P.; Beaumont, M.; Abed-Maillard, S.; Hartweg, M. Modulation of cerebral ketone metabolism following traumatic brain injury in humans. J. Cereb. Blood Flow Metab. 2020, 40, 177–186. [Google Scholar] [CrossRef]
- Norgren, J.; Sindi, S.; Sandebring-Matton, A.; Kåreholt, I.; Daniilidou, M.; Akenine, U.; Nordin, K.; Rosenborg, S.; Ngandu, T.; Kivipelto, M. Ketosis after intake of coconut oil and caprylic acid—With and without glucose: A cross-over study in healthy older adults. Front. Nutr. 2020, 7, 40. [Google Scholar] [CrossRef]
- St-Pierre, V.; Vandenberghe, C.; Lowry, C.-M.; Fortier, M.; Castellano, C.-A.; Wagner, R.; Cunnane, S.C. Plasma ketone and medium chain fatty acid response in humans consuming different medium chain triglycerides during a metabolic study day. Front. Nutr. 2019, 6, 46. [Google Scholar] [CrossRef]
- Freemantle, E.; Vandal, M.; Tremblay-Mercier, J.; Plourde, M.; Poirier, J.; Cunnane, S.C. Metabolic response to a ketogenic breakfast in the healthy elderly. JNHA-J. Nutr. Health Aging 2009, 13, 293–298. [Google Scholar] [CrossRef]
- Vandenberghe, C.; St-Pierre, V.; Fortier, M.; Castellano, C.-A.; Cuenoud, B.; Cunnane, S.C. Medium chain triglycerides modulate the ketogenic effect of a metabolic switch. Front. Nutr. 2020, 7, 3. [Google Scholar] [CrossRef]
- Lin, T.-Y.; Liu, H.-W.; Hung, T.-M. The Ketogenic effect of medium-chain triacylglycerides. Front. Nutr. 2021, 8, 952. [Google Scholar] [CrossRef] [PubMed]
- Henderson, S.T.; Vogel, J.L.; Barr, L.J.; Garvin, F.; Jones, J.J.; Costantini, L.C. Study of the ketogenic agent AC-1202 in mild to moderate Alzheimer’s disease: A randomized, double-blind, placebo-controlled, multicenter trial. Nutr. Metab. 2009, 6, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Zeevi, D.; Korem, T.; Zmora, N.; Israeli, D.; Rothschild, D.; Weinberger, A.; Ben-Yacov, O.; Lador, D.; Avnit-Sagi, T.; Lotan-Pompan, M. Personalized nutrition by prediction of glycemic responses. Cell 2015, 163, 1079–1094. [Google Scholar] [CrossRef]
- Wanten, G.J.; Naber, A.H. Cellular and physiological effects of medium-chain triglycerides. Mini Rev. Med. Chem. 2004, 4, 847–857. [Google Scholar] [CrossRef] [PubMed]
- Knottnerus, S.J.; van Harskamp, D.; Schierbeek, H.; Bleeker, J.C.; Crefcoeur, L.L.; Ferdinandusse, S.; van Goudoever, J.B.; Houtkooper, R.H.; IJlst, L.; Langeveld, M. Exploring the metabolic fate of medium-chain triglycerides in healthy individuals using a stable isotope tracer. Clin. Nutr. 2021, 40, 1396–1404. [Google Scholar] [CrossRef]
- Sills, M.; Forsythe, W.; Haidukewych, D. Role of octanoic and decanoic acids in the control of seizures. Arch. Dis. Child. 1986, 61, 1173–1177. [Google Scholar] [CrossRef]
- Ulrich, H.; Pastores, S.M.; Katz, D.P.; Kvetan, V. Parenteral use of medium-chain triglycerides: A reappraisal. Nutrition 1996, 12, 231–238. [Google Scholar] [CrossRef]
- Wunderling, K.; Leopold, C.; Jamitzky, I.; Yaghmour, M.; Zink, F.; Kratky, D.; Thiele, C. Hepatic synthesis of triacylglycerols containing medium-chain fatty acids is dominated by diacylglycerol acyltransferase 1 and efficiently inhibited by etomoxir. Mol. Metab. 2021, 45, 101150. [Google Scholar] [CrossRef]
- Sonnay, S.; Chakrabarti, A.; Thevenet, J.; Wiederkehr, A.; Christinat, N.; Masoodi, M. Differential Metabolism of Medium-Chain Fatty Acids in Differentiated Human-Induced Pluripotent Stem Cell-Derived Astrocytes. Front. Physiol. 2019, 10, 657. [Google Scholar] [CrossRef]
- Turner, N.; Hariharan, K.; TidAng, J.; Frangioudakis, G.; Beale, S.M.; Wright, L.E.; Zeng, X.Y.; Leslie, S.J.; Li, J.Y.; Kraegen, E.W.; et al. Enhancement of muscle mitochondrial oxidative capacity and alterations in insulin action are lipid species dependent: Potent tissue-specific effects of medium-chain fatty acids. Diabetes 2009, 58, 2547–2554. [Google Scholar] [CrossRef]
- Ota, M.; Matsuo, J.; Ishida, I.; Hattori, K.; Teraishi, T.; Tonouchi, H.; Ashida, K.; Takahashi, T.; Kunugi, H. Effect of a ketogenic meal on cognitive function in elderly adults: Potential for cognitive enhancement. Psychopharmacology 2016, 233, 3797–3802. [Google Scholar] [CrossRef] [PubMed]
- Giannos, P.; Prokopidis, K.; Lidoriki, I.; Triantafyllidis, K.K.; Kechagias, K.S.; Celoch, K.; Candow, D.G.; Ostojic, S.M.; Forbes, S.C. Medium-chain triglycerides may improve memory in non-demented older adults: A systematic review of randomized controlled trials. BMC Geriatr. 2022, 22, 817. [Google Scholar] [CrossRef] [PubMed]
- Ashton, J.S.; Roberts, J.W.; Wakefield, C.J.; Page, R.M.; MacLaren, D.P.; Marwood, S.; Malone, J.J. The effects of medium chain triglyceride (MCT) supplementation using a C8: C10 ratio of 30: 70 on cognitive performance in healthy young adults. Physiol. Behav. 2021, 229, 113252. [Google Scholar] [CrossRef]
- Augustin, K.; Khabbush, A.; Williams, S.; Eaton, S.; Orford, M.; Cross, J.H.; Heales, S.J.; Walker, M.C.; Williams, R.S. Mechanisms of action for the medium-chain triglyceride ketogenic diet in neurological and metabolic disorders. Lancet Neurol. 2018, 17, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Andersen, J.V.; Westi, E.W.; Neal, E.S.; Aldana, B.I.; Borges, K. β-Hydroxybutyrate and Medium-Chain Fatty Acids are Metabolized by Different Cell Types in Mouse Cerebral Cortex Slices. Neurochem. Res. 2023, 48, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Myette-Côté, É.; Neudorf, H.; Rafiei, H.; Clarke, K.; Little, J.P. Prior ingestion of exogenous ketone monoester attenuates the glycaemic response to an oral glucose tolerance test in healthy young individuals. J. Physiol. 2018, 596, 1385–1395. [Google Scholar] [CrossRef]
- Scharfen, J.; Jansen, K.; Holling, H. Retest effects in working memory capacity tests: A meta-analysis. Psychon. Bull. Rev. 2018, 25, 2175–2199. [Google Scholar] [CrossRef]
- Basso, M.R.; Carona, F.D.; Lowery, N.; Axelrod, B.N. Practice effects on the WAIS-III across 3-and 6-month intervals. Clin. Neuropsychol. 2002, 16, 57–63. [Google Scholar] [CrossRef]
- Atkinson, T.M.; Ryan, J.P.; Kryza, M.; Charette, L.M. Using versions of the Trail Making Test as alternate forms. Clin. Neuropsychol. 2011, 25, 1193–1206. [Google Scholar] [CrossRef]


| Characteristics | Values | |
|---|---|---|
| n | (female/male) | 19 (12/7) |
| age | years | 25 (20–27) * |
| weight | kg | 64 (61–71) * |
| height | cm | 173.2 (169–177) * |
| BMI | kg/m2 | 22 (20–23) * |
| BHB | µmol/L | 77 (34–227) * |
| fasting glucose | mg/dL | 85 (79–89) * |
| fasting insulin | µU/mL | 3.9 (0.5–5.3) * |
| Side Effects (SE) | MCT Oil | MCT Oil Plus Glucose |
|---|---|---|
| Total SE (n) | ||
| t1 | 20 | 10 |
| t7 | 0 | 0 |
| Nausea | ||
| t1 | 5 | 0 |
| t7 | 0 | 0 |
| Diarrhea | ||
| t1 | 2 | 2 |
| t7 | 0 | 0 |
| Stomach discomfort | ||
| t1 | 4 | 4 |
| t7 | 0 | 0 |
| Abdominal pain | ||
| t1 | 4 | 4 |
| t7 | 0 | 0 |
| Dizziness | ||
| t1 | 2 | 0 |
| t7 | 0 | 0 |
| Sweating | ||
| t1 | 1 | 0 |
| t7 | 0 | 0 |
| Brain fog | ||
| t1 | 1 | 0 |
| t7 | 0 | 0 |
| Vomiting | ||
| t1 | 1 | 0 |
| t7 | 0 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Heidt, C.; Fobker, M.; Newport, M.; Feldmann, R.; Fischer, T.; Marquardt, T. Beta-Hydroxybutyrate (BHB), Glucose, Insulin, Octanoate (C8), and Decanoate (C10) Responses to a Medium-Chain Triglyceride (MCT) Oil with and without Glucose: A Single-Center Study in Healthy Adults. Nutrients 2023, 15, 1148. https://doi.org/10.3390/nu15051148
Heidt C, Fobker M, Newport M, Feldmann R, Fischer T, Marquardt T. Beta-Hydroxybutyrate (BHB), Glucose, Insulin, Octanoate (C8), and Decanoate (C10) Responses to a Medium-Chain Triglyceride (MCT) Oil with and without Glucose: A Single-Center Study in Healthy Adults. Nutrients. 2023; 15(5):1148. https://doi.org/10.3390/nu15051148
Chicago/Turabian StyleHeidt, Christina, Manfred Fobker, Mary Newport, Reinhold Feldmann, Tobias Fischer, and Thorsten Marquardt. 2023. "Beta-Hydroxybutyrate (BHB), Glucose, Insulin, Octanoate (C8), and Decanoate (C10) Responses to a Medium-Chain Triglyceride (MCT) Oil with and without Glucose: A Single-Center Study in Healthy Adults" Nutrients 15, no. 5: 1148. https://doi.org/10.3390/nu15051148
APA StyleHeidt, C., Fobker, M., Newport, M., Feldmann, R., Fischer, T., & Marquardt, T. (2023). Beta-Hydroxybutyrate (BHB), Glucose, Insulin, Octanoate (C8), and Decanoate (C10) Responses to a Medium-Chain Triglyceride (MCT) Oil with and without Glucose: A Single-Center Study in Healthy Adults. Nutrients, 15(5), 1148. https://doi.org/10.3390/nu15051148







