Nutritional Strategies in the Rehabilitation of Musculoskeletal Injuries in Athletes: A Systematic Integrative Review
Abstract
1. Introduction
2. Methods
2.1. Eligibility Criteria
2.2. Information Sources
2.3. Search Strategy
2.4. Selection Process
2.5. Data Collection Process and Items
2.6. Study Risk of Bias Assessment
3. Results
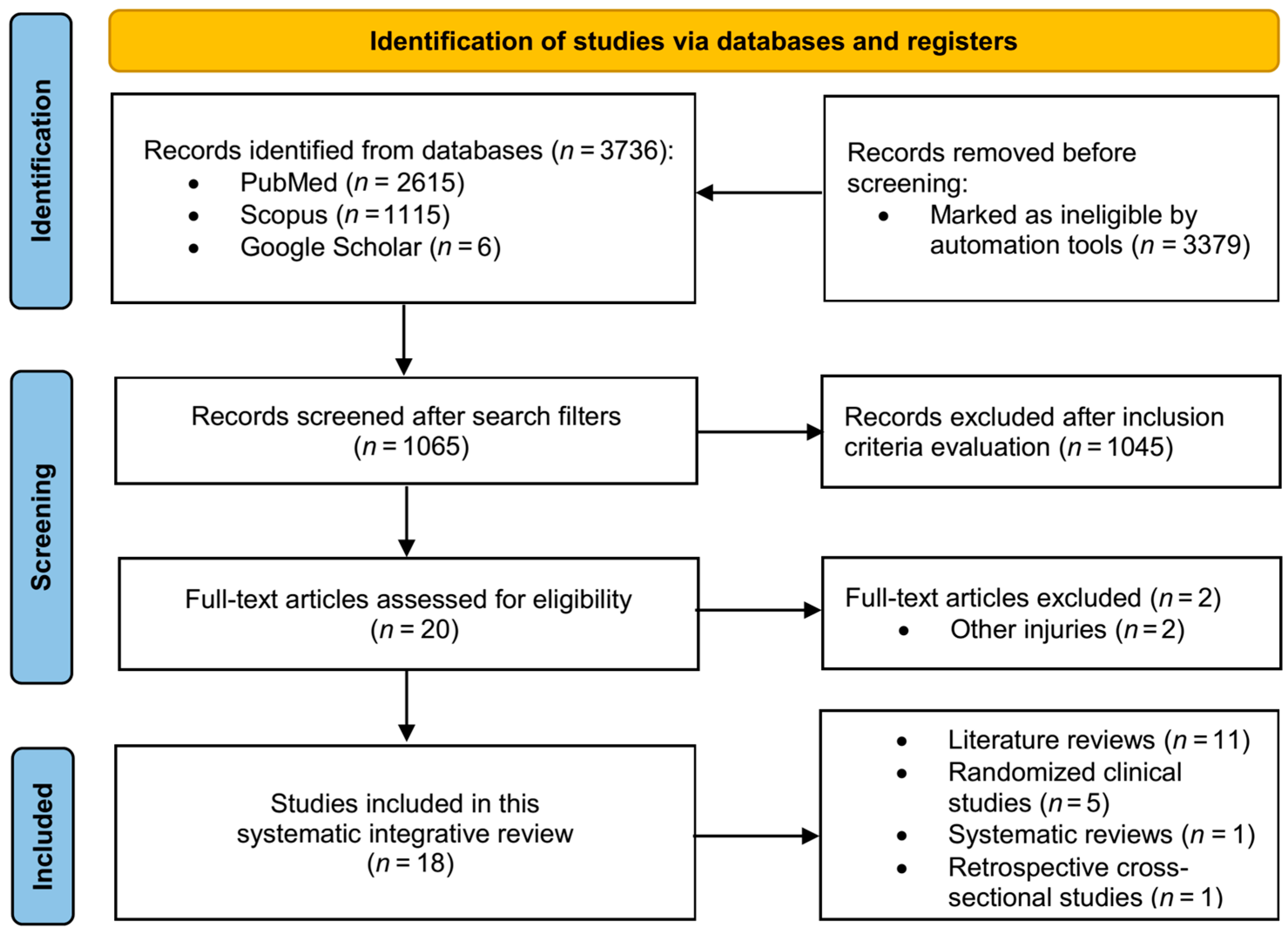
3.1. Study Selection
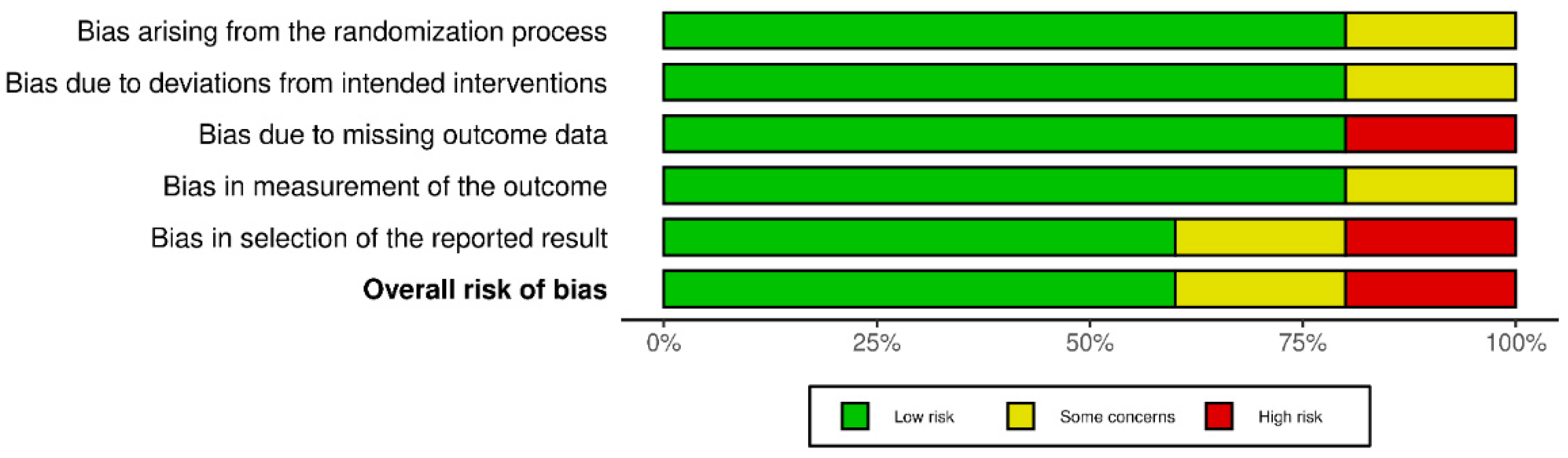
3.2. Risk of Bias within Studies
3.3. Results of Individual Studies
4. Discussion
4.1. Energy Availability
4.2. Loss of Muscle Mass and Protein Intake
4.3. Tissue Repair and Inflammation
4.3.1. Creatine Monohydrate
4.3.2. Omega-3 Fatty Acids
4.3.3. Collagen Peptides and Specific Gelatin Products
4.3.4. HMB
4.3.5. Vitamin D
4.4. Future Directions
5. Limitations and Strengths
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wall, B.T.; van Loon, L.J. Nutritional strategies to attenuate muscle disuse atrophy. Nutr. Rev. 2013, 71, 195–208. [Google Scholar] [CrossRef]
- Seki, K.; Taniguchi, Y.; Narusawa, M. Effects of joint immobilization on firing rate modulation of human motor units. J. Physiol. 2001, 530, 507–519. [Google Scholar] [CrossRef] [PubMed]
- Shackelford, L.; LeBlanc, A.; Driscoll, T.; Evans, H.; Rianon, N.; Smith, S.; Spector, E.; Feeback, D.; Lai, D. Resistance exercise as a countermeasure to disuse-induced bone loss. J. Appl. Physiol. 2004, 97, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Capelli, C.; Antonutto, G.; Kenfack, M.A.; Cautero, M.; Lador, F.; Moia, C.; Tam, E.; Ferretti, G. Factors determining the time course of VO2(max) decay during bedrest: Implications for VO2(max) limitation. Eur. J. Appl. Physiol. 2006, 98, 152–160. [Google Scholar] [CrossRef] [PubMed]
- McGuire, D.K.; Levine, B.D.; Williamson, J.W.; Snell, P.G.; Blomqvist, C.G.; Saltin, B.; Mitchell, J.H. A 30-year follow-up of the Dallas Bedrest and Training Study: I. Effect of age on the cardiovascular response to exercise. Circulation 2001, 104, 1350–1357. [Google Scholar] [CrossRef] [PubMed]
- Dirks, M.L.; Wall, B.T.; van de Valk, B.; Holloway, T.M.; Holloway, G.P.; Chabowski, A.; Goossens, G.H.; van Loon, L.J. One Week of Bed Rest Leads to Substantial Muscle Atrophy and Induces Whole-Body Insulin Resistance in the Absence of Skeletal Muscle Lipid Accumulation. Diabetes 2016, 65, 2862–2875. [Google Scholar] [CrossRef]
- Abadi, A.; Glover, E.I.; Isfort, R.J.; Raha, S.; Safdar, A.; Yasuda, N.; Kaczor, J.J.; Melov, S.; Hubbard, A.; Qu, X.; et al. Limb immobilization induces a coordinate down-regulation of mitochondrial and other metabolic pathways in men and women. PLoS ONE 2009, 4, e6518. [Google Scholar] [CrossRef]
- Ferrando, A.A.; Lane, H.W.; Stuart, C.A.; Davis-Street, J.; Wolfe, R.R. Prolonged bed rest decreases skeletal muscle and whole body protein synthesis. Am. J. Physiol. 1996, 270, E627–E633. [Google Scholar] [CrossRef]
- Ryan, J.L.; Pracht, E.E.; Orban, B.L. Inpatient and emergency department costs from sports injuries among youth aged 5-18 years. BMJ Open Sport Exerc. Med. 2019, 5. [Google Scholar] [CrossRef]
- Papadopoulou, S.K.; Mantzorou, M.; Kondyli-Sarika, F.; Alexandropoulou, I.; Papathanasiou, J.; Voulgaridou, G.; Nikolaidis, P.T. The Key Role of Nutritional Elements on Sport Rehabilitation and the Effects of Nutrients Intake. Sports 2022, 10, 84. [Google Scholar] [CrossRef]
- Tipton, K.D.; Phillips, S.M. Dietary protein for muscle hypertrophy. In Limits of Human Endurance; Nestle Nutr Inst Workshop Series; Karger Publishers: Basel, Switzerland, 2013; Volume 76, pp. 73–84. [Google Scholar]
- Owoeye, O.B.A.; VanderWey, M.J.; Pike, I. Reducing Injuries in Soccer (Football): An Umbrella Review of Best Evidence Across the Epidemiological Framework for Prevention. Sports Med Open 2020, 6, 46. [Google Scholar] [CrossRef] [PubMed]
- Close, G.L.; Sale, C.; Baar, K.; Bermon, S. Nutrition for the Prevention and Treatment of Injuries in Track and Field Athletes. Int. J. Sport Nutr. Exerc. Metab. 2019, 29, 189–197. [Google Scholar] [CrossRef]
- Wall, B.T.; Dirks, M.L.; van Loon, L.J. Skeletal muscle atrophy during short-term disuse: Implications for age-related sarcopenia. Ageing Res. Rev. 2013, 12, 898–906. [Google Scholar] [CrossRef] [PubMed]
- Jackson, T.J.; Starkey, C.; McElhiney, D.; Domb, B.G. Epidemiology of Hip Injuries in the National Basketball Association: A 24-Year Overview. Orthop. J. Sports Med. 2013, 1, 2325967113499130. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.M. Physiologic and molecular bases of muscle hypertrophy and atrophy: Impact of resistance exercise on human skeletal muscle (protein and exercise dose effects). Appl. Physiol. Nutr. Metab. Physiol. Appl. Nutr. Et Metab. 2009, 34, 403–410. [Google Scholar] [CrossRef]
- Tipton, K.D. Nutritional Support for Exercise-Induced Injuries. Sports Med. 2015, 45 (Suppl. S1), S93–S104. [Google Scholar] [CrossRef]
- Tipton, K.D. Dietary strategies to attenuate muscle loss during recovery from injury. Nestle Nutr. Inst. Workshop Ser. 2013, 75, 51–61. [Google Scholar] [CrossRef]
- Phillips, S.M. A brief review of critical processes in exercise-induced muscular hypertrophy. Sports Med. 2014, 44 (Suppl. S1), S71–S77. [Google Scholar] [CrossRef]
- Tooth, C.; Gofflot, A.; Schwartz, C.; Croisier, J.L.; Beaudart, C.; Bruyère, O.; Forthomme, B. Risk Factors of Overuse Shoulder Injuries in Overhead Athletes: A Systematic Review. Sports Health 2020, 12, 478–487. [Google Scholar] [CrossRef]
- Toresdahl, B.G.; McElheny, K.; Metzl, J.; Ammerman, B.; Chang, B.; Kinderknecht, J. A Randomized Study of a Strength Training Program to Prevent Injuries in Runners of the New York City Marathon. Sports Health 2020, 12, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Minghelli, B.; Cadete, J. Epidemiology of musculoskeletal injuries in tennis players: Risk factors. J. Sports Med. Phys. Fit. 2019, 59, 2045–2052. [Google Scholar]
- Frankenfield, D. Energy expenditure and protein requirements after traumatic injury. Nutr. Clin. Pract. Off. Publ. Am. Soc. Parenter. Enter. Nutr. 2006, 21, 430–437. [Google Scholar] [CrossRef]
- Wolfe, R.R. The underappreciated role of muscle in health and disease. Am. J. Clin. Nutr. 2006, 84, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Pérez, V.; Ruiz, J.; Vazquez-Guerrero, J.; Rodas, G.; Del Coso, J. Training and competition injury epidemiology in professional basketball players: A prospective observational study. Physician Sportsmed. 2021, 17, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gravina, L.; Ruiz, F.; Diaz, E.; Lekue, J.A.; Badiola, A.; Irazusta, J.; Gil, S.M. Influence of nutrient intake on antioxidant capacity, muscle damage and white blood cell count in female soccer players. J. Int. Soc. Sports Nutr. 2012, 9, 32. [Google Scholar] [CrossRef]
- Peeling, P.; Binnie, M.J.; Goods, P.S.; Sim, M.; Burke, L.M. Evidence-based supplements for the enhancement of athletic performance. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 178–187. [Google Scholar] [CrossRef]
- Grondin, J.; Crenn, V.; Gernigon, M.; Quinette, Y.; Louguet, B.; Menu, P.; Fouasson-Chailloux, A.; Dauty, M. Relevant Strength Parameters to Allow Return to Running after Primary Anterior Cruciate Ligament Reconstruction with Hamstring Tendon Autograft. Int. J. Environ. Res. Public Health 2022, 19, 8245. [Google Scholar] [CrossRef]
- Vélez-Gutiérrez, J.M.; Petro, J.L.; Aburto-Corona, J.A.; Vargas-Molina, S.; Kreider, R.B.; Bonilla, D.A. Cortical Changes as a Result of Sports Injuries: A Short Commentary. Cuerpo Cult. Y Mov. 2022, 12, 7884. [Google Scholar] [CrossRef]
- Pasiakos, S.M.; Vislocky, L.M.; Carbone, J.W.; Altieri, N.; Konopelski, K.; Freake, H.C.; Anderson, J.M.; Ferrando, A.A.; Wolfe, R.R.; Rodriguez, N.R. Acute energy deprivation affects skeletal muscle protein synthesis and associated intracellular signaling proteins in physically active adults. J. Nutr. 2010, 140, 745–751. [Google Scholar] [CrossRef]
- Biolo, G.; Agostini, F.; Simunic, B.; Sturma, M.; Torelli, L.; Preiser, J.C.; Deby-Dupont, G.; Magni, P.; Strollo, F.; di Prampero, P.; et al. Positive energy balance is associated with accelerated muscle atrophy and increased erythrocyte glutathione turnover during 5 wk of bed rest. Am. J. Clin. Nutr. 2008, 88, 950–958. [Google Scholar] [CrossRef] [PubMed]
- Bonilla, D.A.; Moreno, Y.; Petro, J.L.; Forero, D.A.; Vargas-Molina, S.; Odriozola-Martinez, A.; Orozco, C.A.; Stout, J.R.; Rawson, E.S.; Kreider, R.B. A Bioinformatics-Assisted Review on Iron Metabolism and Immune System to Identify Potential Biomarkers of Exercise Stress-Induced Immunosuppression. Biomedicines 2022, 10, 724. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S. Stress, Adaptation, and Disease: Allostasis and Allostatic Load. Ann. N. Y. Acad. Sci. 1998, 840, 33–44. [Google Scholar] [CrossRef]
- Krause, J.; DiPiro, N.; Saunders, L.; Newman, S.; Banik, N.; Park, S. Allostatic Load and Spinal Cord Injury: Review of Existing Research and Preliminary Data. Top. Spinal Cord Inj. Rehabil. 2014, 20, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Whittemore, R.; Knafl, K. The integrative review: Updated methodology. J. Adv. Nurs. 2005, 52, 546–553. [Google Scholar] [CrossRef]
- Bonilla, D.A.; Cardozo, L.A.; Velez-Gutierrez, J.M.; Arevalo-Rodriguez, A.; Vargas-Molina, S.; Stout, J.R.; Kreider, R.B.; Petro, J.L. Exercise Selection and Common Injuries in Fitness Centers: A Systematic Integrative Review and Practical Recommendations. Int. J. Environ. Res. Public Health 2022, 19, 2710. [Google Scholar] [CrossRef]
- Hopia, H.; Latvala, E.; Liimatainen, L. Reviewing the methodology of an integrative review. Scand. J. Caring Sci. 2016, 30, 662–669. [Google Scholar] [CrossRef] [PubMed]
- Ardern, C.L.; Büttner, F.; Andrade, R.; Weir, A.; Ashe, M.C.; Holden, S.; Impellizzeri, F.M.; Delahunt, E.; Dijkstra, H.P.; Mathieson, S.J. Implementing the 27 PRISMA 2020 Statement items for systematic reviews in the sport and exercise medicine, musculoskeletal rehabilitation and sports science fields: The PERSiST (implementing Prisma in Exercise, Rehabilitation, Sport medicine and SporTs science) guidance. Br. J. Sports Med. 2022, 56, 175–195. [Google Scholar]
- Higgins, J.P.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons: Hoboken, NJ, USA, 2019. [Google Scholar]
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef]
- Büttner, F.; Winters, M.; Delahunt, E.; Elbers, R.; Lura, C.B.; Khan, K.M.; Weir, A.; Ardern, C.L. Identifying the ‘incredible’! Part 2: Spot the difference-a rigorous risk of bias assessment can alter the main findings of a systematic review. Br. J. Sports Med. 2020, 54, 801–808. [Google Scholar] [CrossRef]
- Shea, B.; Grimshaw, J.; Wells, G.; Boers, M.; Andersson, N.; Hamel, C.; Porter, A.; Tugwell, P.; Moher, D.; Bouter, L.J.H. AMSTAR: Assessing Methodological Quality of Systematic Reviews; McMaster University: Hamilton, ON, Canada, 2011. [Google Scholar]
- Shea, B.J.; Reeves, B.C.; Wells, G.; Thuku, M.; Hamel, C.; Moran, J.; Moher, D.; Tugwell, P.; Welch, V.; Kristjansson, E. AMSTAR 2: A critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ 2017, 358, j4008. [Google Scholar] [CrossRef]
- Lundy, B.; Suni, V.; Drew, M.; Trease, L.; Burke, L.M. Nutrition factors associated with rib stress injury history in elite rowers. J. Sci. Med. Sport 2022, 25, 979–985. [Google Scholar] [CrossRef] [PubMed]
- Burton, I.; McCormack, A. Nutritional Supplements in the Clinical Management of Tendinopathy: A Scoping Review. SportRχiv 2022. pre-print. [Google Scholar] [CrossRef]
- Turnagol, H.H.; Kosar, S.N.; Guzel, Y.; Aktitiz, S.; Atakan, M.M. Nutritional Considerations for Injury Prevention and Recovery in Combat Sports. Nutrients 2021, 14, 53. [Google Scholar] [CrossRef]
- Khatri, M.; Naughton, R.J.; Clifford, T.; Harper, L.D.; Corr, L. The effects of collagen peptide supplementation on body composition, collagen synthesis, and recovery from joint injury and exercise: A systematic review. Amino Acids 2021, 53, 1493–1506. [Google Scholar] [CrossRef] [PubMed]
- Smith-Ryan, A.E.; Hirsch, K.R.; Saylor, H.E.; Gould, L.M.; Blue, M.N.M. Nutritional Considerations and Strategies to Facilitate Injury Recovery and Rehabilitation. J. Athl. Train. 2020, 55, 918–930. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulou, S.K. Rehabilitation Nutrition for Injury Recovery of Athletes: The Role of Macronutrient Intake. Nutrients 2020, 12, 2449. [Google Scholar] [CrossRef] [PubMed]
- Quintero, K.J.; Resende, A.d.S.; Leite, G.S.F.; Lancha Junior, A.H. An overview of nutritional strategies for recovery process in sports-related muscle injuries. Nutrire 2018, 43, 27. [Google Scholar] [CrossRef]
- Kahn, D.S.; Shuler, F.D.; Qazi, Z.N.; Belmaggio, T.D.; Mehta, S.P.J.C.R.i.P.; Medicine, R. The emerging role of vitamin D in sports physical therapy: A review. Crit. Rev. Phys. Rehabil. Med. 2015, 27, 1–10. [Google Scholar] [CrossRef]
- Wall, B.T.; Morton, J.P.; Van Loon, L.J. Strategies to maintain skeletal muscle mass in the injured athlete: Nutritional considerations and exercise mimetics. Eur. J. Sport Sci. 2015, 15, 53–62. [Google Scholar] [CrossRef]
- Pyne, D.B.; Verhagen, E.A.; Mountjoy, M. Nutrition, illness, and injury in aquatic sports. Int. J. Sport Nutr. Exerc. Metab. 2014, 24, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Laboute, E.; France, J.; Trouve, P.; Puig, P.-L.; Boireau, M.; Blanchard, A.J.A.o.P.; Medicine, R. Rehabilitation and leucine supplementation as possible contributors to an athlete’s muscle strength in the reathletization phase following anterior cruciate ligament surgery. Ann. Phys. Rehabil. Med. 2013, 56, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Eraslan, A.; Ulkar, B. Glucosamine supplementation after anterior cruciate ligament reconstruction in athletes: A randomized placebo-controlled trial. Res. Sports Med. 2015, 23, 14–26. [Google Scholar] [CrossRef]
- Flueck, J.L.; Schlaepfer, M.W.; Perret, C.J.N. Effect of 12-week vitamin D supplementation on 25 [OH] D status and performance in athletes with a spinal cord injury. Nutrients 2016, 8, 586. [Google Scholar] [CrossRef]
- Juhasz, I.; Kopkane, J.P.; Hajdu, P.; Szalay, G.; Kopper, B.; Tihanyi, J. Creatine Supplementation Supports the Rehabilitation of Adolescent Fin Swimmers in Tendon Overuse Injury Cases. J. Sports Sci. Med. 2018, 17, 279–288. [Google Scholar]
- Collins, J.; Maughan, R.J.; Gleeson, M.; Bilsborough, J.; Jeukendrup, A.; Morton, J.P.; Phillips, S.M.; Armstrong, L.; Burke, L.M.; Close, G.L.; et al. UEFA expert group statement on nutrition in elite football. Current evidence to inform practical recommendations and guide future research. Br. J. Sports Med. 2021, 55, 416. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Gómez, Á.; Jurado-Castro, J.M.; Mata, F.; Sánchez-Oliver, A.J.; Domínguez, R. Effects of β-Hydroxy β-Methylbutyric Supplementation in Combination with Conservative Non-Invasive Treatments in Athletes with Patellar Tendinopathy: A Pilot Study. Int. J. Environ. Res. Public Heal. 2022, 19, 471. [Google Scholar] [CrossRef] [PubMed]
- Hotfiel, T.; Mayer, I.; Huettel, M.; Hoppe, M.W.; Engelhardt, M.; Lutter, C.; Pöttgen, K.; Heiss, R.; Kastner, T.; Grim, C. Accelerating Recovery from Exercise-Induced Muscle Injuries in Triathletes: Considerations for Olympic Distance Races. Sports 2019, 7, 143. [Google Scholar] [CrossRef]
- Aasa, U.; Svartholm, I.; Andersson, F.; Berglund, L. Injuries among weightlifters and powerlifters: A systematic review. Br. J. Sports Med. 2017, 51, 211. [Google Scholar] [CrossRef]
- Baron, J.E.; Parker, E.A.; Duchman, K.R.; Westermann, R.W. Perioperative and Postoperative Factors Influence Quadriceps Atrophy and Strength After ACL Reconstruction: A Systematic Review. Orthop. J. Sports Med. 2020, 8, 2325967120930296. [Google Scholar] [CrossRef]
- Lynch, M.; Marinov, G.K. The bioenergetic costs of a gene. Proc. Natl. Acad. Sci. USA 2015, 112, 15690–15695. [Google Scholar] [CrossRef] [PubMed]
- DeWane, G.; Salvi, A.M.; DeMali, K.A. Fueling the cytoskeleton–links between cell metabolism and actin remodeling. J. Cell Sci. 2021, 134, jcs248385. [Google Scholar] [CrossRef] [PubMed]
- Bobba-Alves, N.; Juster, R.-P.; Picard, M. The energetic cost of allostasis and allostatic load. Psychoneuroendocrinology 2022, 146, 105951. [Google Scholar] [CrossRef] [PubMed]
- Areta, J.L.; Taylor, H.L.; Koehler, K. Low energy availability: History, definition and evidence of its endocrine, metabolic and physiological effects in prospective studies in females and males. Eur. J. Appl. Physiol. 2021, 121, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Wasserfurth, P.; Palmowski, J.; Hahn, A.; Krüger, K. Reasons for and Consequences of Low Energy Availability in Female and Male Athletes: Social Environment, Adaptations, and Prevention. Sports Med.–Open 2020, 6, 44. [Google Scholar] [CrossRef] [PubMed]
- Dave, S.C.; Fisher, M. Relative energy deficiency in sport (RED–S). Curr. Probl. Pediatr. Adolesc. Health Care 2022, 52. [Google Scholar] [CrossRef]
- Mountjoy, M.; Sundgot-Borgen, J.; Burke, L.; Ackerman, K.E.; Blauwet, C.; Constantini, N.; Lebrun, C.; Lundy, B.; Melin, A.; Meyer, N.J.; et al. International Olympic Committee (IOC) consensus statement on relative energy deficiency in sport (RED-S): 2018 update. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 316–331. [Google Scholar] [CrossRef]
- Loucks, A.B.; Verdun, M.; Heath, E.M. Low energy availability, not stress of exercise, alters LH pulsatility in exercising women. J. Appl. Physiol. 1998, 84, 37–46. [Google Scholar] [CrossRef]
- Holtzman, B.; Ackerman, K. Recommendations and Nutritional Considerations for Female Athletes: Health and Performance. Sports Med 2021, 51 (Suppl. 1), 43–57. [Google Scholar] [CrossRef]
- Heikura, I.A.; Uusitalo, A.L.T.; Stellingwerff, T.; Bergland, D.; Mero, A.A.; Burke, L.M. Low Energy Availability Is Difficult to Assess but Outcomes Have Large Impact on Bone Injury Rates in Elite Distance Athletes. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 403–411. [Google Scholar] [CrossRef]
- Hutson, M.J.; O’Donnell, E.; Brooke-Wavell, K.; Sale, C.; Blagrove, R.C. Effects of Low Energy Availability on Bone Health in Endurance Athletes and High-Impact Exercise as A Potential Countermeasure: A Narrative Review. Sports Med. 2021, 51, 391–403. [Google Scholar] [CrossRef]
- Torres-McGehee, T.M.; Pritchett, K.L.; Zippel, D.; Minton, D.M.; Cellamare, A.; Sibilia, M.J. Sports nutrition knowledge among collegiate athletes, coaches, athletic trainers, and strength and conditioning specialists. J. Athl. Train. 2012, 47, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Mountjoy, M.; Sundgot-Borgen, J.; Burke, L.; Carter, S.; Constantini, N.; Lebrun, C.; Meyer, N.; Sherman, R.; Steffen, K.; Budgett, R.J. The IOC consensus statement: Beyond the female athlete triad—Relative energy deficiency in sport (RED-S). Br. J. Sports Med. 2014, 48, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Shamim, B.; Hawley, J.A.; Camera, D.M. Protein Availability and Satellite Cell Dynamics in Skeletal Muscle. Sports Med 2018, 48, 1329–1343. [Google Scholar] [CrossRef] [PubMed]
- Jäger, R.; Kerksick, C.M.; Campbell, B.I.; Cribb, P.J.; Wells, S.D.; Skwiat, T.M.; Purpura, M.; Ziegenfuss, T.N.; Ferrando, A.A.; Arent, S.M.J. International society of sports nutrition position stand: Protein and exercise. J. Int. Soc. Sports Nutr. 2017, 14, 20. [Google Scholar] [CrossRef] [PubMed]
- Hector, A.J.; Phillips, S.M. Protein Recommendations for Weight Loss in Elite Athletes: A Focus on Body Composition and Performance. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 170–177. [Google Scholar] [CrossRef]
- Mettler, S.; Mitchell, N.; Tipton, K.D. Increased protein intake reduces lean body mass loss during weight loss in athletes. Med. Sci. Sports Exerc. 2010, 42, 326–337. [Google Scholar] [CrossRef]
- Baum, J.I.; Kim, I.Y.; Wolfe, R.R. Protein Consumption and the Elderly: What Is the Optimal Level of Intake? Nutrients 2016, 8, 359. [Google Scholar] [CrossRef]
- Paddon-Jones, D.; Rasmussen, B.B. Dietary protein recommendations and the prevention of sarcopenia. Curr. Opin. Clin. Nutr. Metab. Care 2009, 12, 86–90. [Google Scholar] [CrossRef]
- Mamerow, M.M.; Mettler, J.A.; English, K.L.; Casperson, S.L.; Arentson-Lantz, E.; Sheffield-Moore, M.; Layman, D.K.; Paddon-Jones, D. Dietary protein distribution positively influences 24-h muscle protein synthesis in healthy adults. J. Nutr. 2014, 144, 876–880. [Google Scholar] [CrossRef] [PubMed]
- Tipton, K.D. Nutritional Support for Injuries Requiring Reduced Activity. Sports Sci. Exch. 2017, 28, 1–6. [Google Scholar]
- Bonilla, D.A.; Perez-Idarraga, A.; Odriozola-Martinez, A.; Kreider, R.B. The 4R’s Framework of Nutritional Strategies for Post-Exercise Recovery: A Review with Emphasis on New Generation of Carbohydrates. Int. J. Environ. Res. Public Health 2020, 18, 103. [Google Scholar] [CrossRef]
- Kreider, R.B.; Jager, R.; Purpura, M. Bioavailability, Efficacy, Safety, and Regulatory Status of Creatine and Related Compounds: A Critical Review. Nutrients 2022, 14, 1035. [Google Scholar] [CrossRef] [PubMed]
- Bonilla, D.A.; Kreider, R.B.; Stout, J.R.; Forero, D.A.; Kerksick, C.M.; Roberts, M.D.; Rawson, E.S.J.N. Metabolic basis of creatine in health and disease: A Bioinformatics-assisted review. Nutrients 2021, 13, 1238. [Google Scholar] [CrossRef]
- Wax, B.; Kerksick, C.M.; Jagim, A.R.; Mayo, J.J.; Lyons, B.C.; Kreider, R.B.J.N. Creatine for exercise and sports performance, with recovery considerations for healthy populations. Nutrients 2021, 13, 1915. [Google Scholar] [CrossRef]
- Bonilla, D.A.; Moreno, Y.; Rawson, E.S.; Forero, D.A.; Stout, J.R.; Kerksick, C.M.; Roberts, M.D.; Kreider, R.B.J.N. A convergent functional genomics analysis to identify biological regulators mediating effects of creatine supplementation. Nutrients 2021, 13, 2521. [Google Scholar] [CrossRef]
- Wallimann, T.; Tokarska-Schlattner, M.; Schlattner, U. The creatine kinase system and pleiotropic effects of creatine. Amino Acids 2011, 40, 1271–1296. [Google Scholar] [CrossRef]
- Marshall, R.P.; Droste, J.-N.; Giessing, J.; Kreider, R.B.J.N. Role of creatine supplementation in conditions involving mitochondrial dysfunction: A narrative review. Nutrients 2022, 14, 529. [Google Scholar] [CrossRef]
- Harmon, K.K.; Stout, J.R.; Fukuda, D.H.; Pabian, P.S.; Rawson, E.S.; Stock, M.S.J.N. The application of creatine supplementation in medical rehabilitation. Nutrients 2021, 13, 1825. [Google Scholar] [CrossRef]
- MacDougall, J.D.; Ward, G.R.; Sale, D.G.; Sutton, J.R. Biochemical adaptation of human skeletal muscle to heavy resistance training and immobilization. J. Appl. Physiol. Respir. Environ. Exerc. Physiol. 1977, 43, 700–703. [Google Scholar] [CrossRef] [PubMed]
- Rawson, E.S.; Miles, M.P.; Larson-Meyer, D.E. Dietary Supplements for Health, Adaptation, and Recovery in Athletes. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 188–199. [Google Scholar] [CrossRef] [PubMed]
- Padilha, C.S.; Cella, P.S.; Salles, L.R.; Deminice, R. Oral creatine supplementation attenuates muscle loss caused by limb immobilization: A systematic review. Fisioter. Mov. 2017, 30, 831–838. [Google Scholar] [CrossRef]
- Op ‘t Eijnde, B.; Urso, B.; Richter, E.A.; Greenhaff, P.L.; Hespel, P. Effect of oral creatine supplementation on human muscle GLUT4 protein content after immobilization. Diabetes 2001, 50, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Johnston, A.P.; Burke, D.G.; MacNeil, L.G.; Candow, D.G. Effect of creatine supplementation during cast-induced immobilization on the preservation of muscle mass, strength, and endurance. J. Strength Cond. Res. 2009, 23, 116–120. [Google Scholar] [CrossRef]
- Backx, E.M.P.; Hangelbroek, R.; Snijders, T.; Verscheijden, M.L.; Verdijk, L.B.; de Groot, L.; van Loon, L.J.C. Creatine Loading Does Not Preserve Muscle Mass or Strength During Leg Immobilization in Healthy, Young Males: A Randomized Controlled Trial. Sports Med. 2017, 47, 1661–1671. [Google Scholar] [CrossRef]
- Hespel, P.; Op’t Eijnde, B.; Van Leemputte, M.; Urso, B.; Greenhaff, P.L.; Labarque, V.; Dymarkowski, S.; Van Hecke, P.; Richter, E.A. Oral creatine supplementation facilitates the rehabilitation of disuse atrophy and alters the expression of muscle myogenic factors in humans. J. Physiol. 2001, 536, 625–633. [Google Scholar] [CrossRef]
- Hirsch, K.R.; Wolfe, R.R.; Ferrando, A.A. Pre- and Post-Surgical Nutrition for Preservation of Muscle Mass, Strength, and Functionality Following Orthopedic Surgery. Nutrients 2021, 13, 1675. [Google Scholar] [CrossRef]
- Mistry, D.; Gee, T.I.; Lee, P. Systematic Review for Protein and Creatine Supplements in Peri-operative Period in Elective Musculoskeletal Surgery Knee and Hip Replacement. J. Arthritis 2022, 11, 1–6. [Google Scholar] [CrossRef]
- Smith, C.; Kruger, M.J.; Smith, R.M.; Myburgh, K.H. The inflammatory response to skeletal muscle injury: Illuminating complexities. Sports Med. 2008, 38, 947–969. [Google Scholar] [CrossRef]
- Tidball, J.G. Regulation of muscle growth and regeneration by the immune system. Nat. Rev. Immunol. 2017, 17, 165–178. [Google Scholar] [CrossRef] [PubMed]
- Peake, J.M.; Neubauer, O.; Della Gatta, P.A.; Nosaka, K. Muscle damage and inflammation during recovery from exercise. J. Appl. Physiol. 2017, 122, 559–570. [Google Scholar] [CrossRef] [PubMed]
- Patsalos, A.; Pap, A.; Varga, T.; Trencsenyi, G.; Contreras, G.A.; Garai, I.; Papp, Z.; Dezso, B.; Pintye, E.; Nagy, L. In situ macrophage phenotypic transition is affected by altered cellular composition prior to acute sterile muscle injury. J. Physiol. 2017, 595, 5815–5842. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Omega-3 fatty acids and inflammatory processes: From molecules to man. Biochem. Soc. Trans. 2017, 45, 1105–1115. [Google Scholar] [CrossRef]
- Tachtsis, B.; Camera, D.; Lacham-Kaplan, O. Potential roles of n-3 PUFAs during skeletal muscle growth and regeneration. Nutrients 2018, 10, 309. [Google Scholar] [CrossRef]
- Maughan, R.J.; Burke, L.M.; Dvorak, J.; Larson-Meyer, D.E.; Peeling, P.; Phillips, S.M.; Rawson, E.S.; Walsh, N.P.; Garthe, I.; Geyer, H.; et al. IOC consensus statement: Dietary supplements and the high-performance athlete. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 104–125. [Google Scholar] [CrossRef]
- Cuñado-González, Á.; Martín-Pintado-Zugasti, A.; Rodríguez-Fernández, Á.L.J.J.o.s.r. Prevalence and factors associated with injuries in elite Spanish volleyball. J. Sport Rehabil. 2019, 28, 796–802. [Google Scholar] [CrossRef]
- Whitehouse, T.; Orr, R.; Fitzgerald, E.; Harries, S.; McLellan, C.P. The Epidemiology of Injuries in Australian Professional Rugby Union 2014 Super Rugby Competition. Orthop. J. Sports Med. 2016, 4, 2325967116634075. [Google Scholar] [CrossRef]
- Hess, M.C.; Swedler, D.I.; Collins, C.S.; Ponce, B.A.; Brabston, E.W. Descriptive Epidemiology of Injuries in Professional Ultimate Frisbee Athletes. J. Athl. Train. 2020, 55, 195–204. [Google Scholar] [CrossRef]
- Shaw, G.; Lee-Barthel, A.; Ross, M.L.; Wang, B.; Baar, K. Vitamin C-enriched gelatin supplementation before intermittent activity augments collagen synthesis. Am. J. Clin. Nutr. 2017, 105, 136–143. [Google Scholar] [CrossRef]
- McAlindon, T.E.; Nuite, M.; Krishnan, N.; Ruthazer, R.; Price, L.L.; Burstein, D.; Griffith, J.; Flechsenhar, K. Change in knee osteoarthritis cartilage detected by delayed gadolinium enhanced magnetic resonance imaging following treatment with collagen hydrolysate: A pilot randomized controlled trial. Osteoarthr. Cartil. 2011, 19, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Clark, K.L.; Sebastianelli, W.; Flechsenhar, K.R.; Aukermann, D.F.; Meza, F.; Millard, R.L.; Deitch, J.R.; Sherbondy, P.S.; Albert, A. 24-Week study on the use of collagen hydrolysate as a dietary supplement in athletes with activity-related joint pain. Curr. Med. Res. Opin. 2008, 24, 1485–1496. [Google Scholar] [CrossRef] [PubMed]
- Szczesniak, K.A.; Ostaszewski, P.; Fuller, J.C., Jr.; Ciecierska, A.; Sadkowski, T. Dietary supplementation of beta-hydroxy-beta-methylbutyrate in animals—A review. J. Anim. Physiol. Anim. Nutr. 2015, 99, 405–417. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, D.J.; Hossain, T.; Hill, D.S.; Phillips, B.E.; Crossland, H.; Williams, J.; Loughna, P.; Churchward-Venne, T.A.; Breen, L.; Phillips, S.M.; et al. Effects of leucine and its metabolite beta-hydroxy-beta-methylbutyrate on human skeletal muscle protein metabolism. J. Physiol. 2013, 591, 2911–2923. [Google Scholar] [CrossRef] [PubMed]
- Rowlands, D.S.; Thomson, J.S. Effects of beta-hydroxy-beta-methylbutyrate supplementation during resistance training on strength, body composition, and muscle damage in trained and untrained young men: A meta-analysis. J. Strength Cond. Res. 2009, 23, 836–846. [Google Scholar] [CrossRef]
- Sanchez-Martinez, J.; Santos-Lozano, A.; Garcia-Hermoso, A.; Sadarangani, K.P.; Cristi-Montero, C. Effects of beta-hydroxy-beta-methylbutyrate supplementation on strength and body composition in trained and competitive athletes: A meta-analysis of randomized controlled trials. J. Sci. Med. Sport 2018, 21, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Deutz, N.E.; Pereira, S.L.; Hays, N.P.; Oliver, J.S.; Edens, N.K.; Evans, C.M.; Wolfe, R.R. Effect of beta-hydroxy-beta-methylbutyrate (HMB) on lean body mass during 10 days of bed rest in older adults. Clin. Nutr. 2013, 32, 704–712. [Google Scholar] [CrossRef]
- Christakos, S.; Dhawan, P.; Liu, Y.; Peng, X.; Porta, A. New insights into the mechanisms of vitamin D action. J. Cell. Biochem. 2003, 88, 695–705. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, L.; Xu, H.J.; Li, Y.; Hu, C.M.; Yang, J.Y.; Sun, M.Y. The Anti-Inflammatory Effects of Vitamin D in Tumorigenesis. Int. J. Mol. Sci. 2018, 19, 2736. [Google Scholar] [CrossRef]
- Pike, J.W.; Christakos, S. Biology and Mechanisms of Action of the Vitamin D Hormone. Endocrinol. Metab. Clin. North Am. 2017, 46, 815–843. [Google Scholar] [CrossRef]
- Wyon, M.A.; Wolman, R.; Nevill, A.M.; Cloak, R.; Metsios, G.S.; Gould, D.; Ingham, A.; Koutedakis, Y. Acute Effects of Vitamin D3 Supplementation on Muscle Strength in Judoka Athletes: A Randomized Placebo-Controlled, Double-Blind Trial. Clin. J. Sport Med. 2016, 26, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Ruohola, J.P.; Laaksi, I.; Ylikomi, T.; Haataja, R.; Mattila, V.M.; Sahi, T.; Tuohimaa, P.; Pihlajamaki, H. Association between serum 25(OH)D concentrations and bone stress fractures in Finnish young men. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2006, 21, 1483–1488. [Google Scholar] [CrossRef]
- Rebolledo, B.J.; Bernard, J.A.; Werner, B.C.; Finlay, A.K.; Nwachukwu, B.U.; Dare, D.M.; Warren, R.F.; Rodeo, S.A.; Surgery, R. The association of vitamin D status in lower extremity muscle strains and core muscle injuries at the National Football League Combine. J. Arthrosc. Relat. Surg. 2018, 34, 1280–1285. [Google Scholar] [CrossRef] [PubMed]
- Lappe, J.; Cullen, D.; Haynatzki, G.; Recker, R.; Ahlf, R.; Thompson, K. Calcium and vitamin d supplementation decreases incidence of stress fractures in female navy recruits. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2008, 23, 741–749. [Google Scholar] [CrossRef] [PubMed]
- Maroon, J.C.; Mathyssek, C.M.; Bost, J.W.; Amos, A.; Winkelman, R.; Yates, A.P.; Duca, M.A.; Norwig, J.A. Vitamin D profile in National Football League players. Am. J. Sports Med. 2015, 43, 1241–1245. [Google Scholar] [CrossRef]
- Pilch, W.; Kita, B.; Piotrowska, A.; Tota, Ł.; Maciejczyk, M.; Czerwińska-Ledwig, O.; Sadowska-Krepa, E.; Kita, S.; Pałka, T. The effect of vitamin D supplementation on the muscle damage after eccentric exercise in young men: A randomized, control trial. J. Int. Soc. Sports Nutr. 2020, 17, 1–10. [Google Scholar] [CrossRef]
- Barker, T.; Schneider, E.D.; Dixon, B.M.; Henriksen, V.T.; Weaver, L.K. Supplemental vitamin D enhances the recovery in peak isometric force shortly after intense exercise. Nutr. Metab. 2013, 10, 69. [Google Scholar] [CrossRef]
- Sikora-Klak, J.; Narvy, S.J.; Yang, J.; Makhni, E.; Kharrazi, F.D.; Mehran, N. The Effect of Abnormal Vitamin D Levels in Athletes. Perm. J. 2018, 22, 17–216. [Google Scholar] [CrossRef]
- Minshull, C.; Biant, L.C.; Ralston, S.H.; Gleeson, N. A Systematic Review of the Role of Vitamin D on Neuromuscular Remodelling Following Exercise and Injury. Calcif. Tissue Int. 2016, 98, 426–437. [Google Scholar] [CrossRef]
- Barker, T.; Henriksen, V.T.; Martins, T.B.; Hill, H.R.; Kjeldsberg, C.R.; Schneider, E.D.; Dixon, B.M.; Weaver, L.K. Higher serum 25-hydroxyvitamin D concentrations associate with a faster recovery of skeletal muscle strength after muscular injury. Nutrients 2013, 5, 1253–1275. [Google Scholar] [CrossRef]
- Powers, S.K. Can antioxidants protect against disuse muscle atrophy? Sports Med. 2014, 44 (Suppl. S2), S155–S165. [Google Scholar] [CrossRef] [PubMed]
- Powers, S.K.; Nelson, W.B.; Hudson, M.B. Exercise-induced oxidative stress in humans: Cause and consequences. Free Radic. Biol. Med. 2011, 51, 942–950. [Google Scholar] [CrossRef] [PubMed]
- Shadel, G.S.; Horvath, T.L. Mitochondrial ROS signaling in organismal homeostasis. Cell 2015, 163, 560–569. [Google Scholar] [CrossRef]
- Scheele, C.; Nielsen, S.; Pedersen, B.K. ROS and myokines promote muscle adaptation to exercise. Trends Endocrinol. Metab.: TEM 2009, 20, 95–99. [Google Scholar] [CrossRef]
- Niess, A.M.; Simon, P. Response and adaptation of skeletal muscle to exercise--the role of reactive oxygen species. Front. Biosci.: J. Virtual Libr. 2007, 12, 4826–4838. [Google Scholar] [CrossRef] [PubMed]
- Nemes, R.; Koltai, E.; Taylor, A.W.; Suzuki, K.; Gyori, F.; Radak, Z. Reactive Oxygen and Nitrogen Species Regulate Key Metabolic, Anabolic, and Catabolic Pathways in Skeletal Muscle. Antioxidants 2018, 7, 85. [Google Scholar] [CrossRef]
- Powers, S.K.; Smuder, A.J.; Judge, A.R. Oxidative stress and disuse muscle atrophy: Cause or consequence? Curr. Opin. Clin. Nutr. Metab. Care 2012, 15, 240–245. [Google Scholar] [CrossRef]
- Barker, T.; Leonard, S.W.; Hansen, J.; Trawick, R.H.; Ingram, R.; Burdett, G.; Lebold, K.M.; Walker, J.A.; Traber, M.G. Vitamin E and C supplementation does not ameliorate muscle dysfunction after anterior cruciate ligament surgery. Free Radic. Biol. Med. 2009, 47, 1611–1618. [Google Scholar] [CrossRef] [PubMed]
- Close, G.L.; Jackson, M.J. Antioxidants and exercise: A tale of the complexities of relating signalling processes to physiological function? J. Physiol. 2014, 592, 1721–1722. [Google Scholar] [CrossRef]
- Hewlings, S.J.; Kalman, D.S. Curcumin: A Review of Its’ Effects on Human Health. Foods 2017, 6, 92. [Google Scholar] [CrossRef]
- Oh, S.N.; Myung, S.K.; Jho, H.J. Analgesic Efficacy of Melatonin: A Meta-Analysis of Randomized, Double-Blind, Placebo-Controlled Trials. J. Clin. Med. 2020, 9, 1553. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Palaksha, K.J.; Park, K.; Park, S.; Kim, H.D.; Reiter, R.J.; Chang, K.T. Melatonin plus exercise-based neurorehabilitative therapy for spinal cord injury. J. Pineal Res. 2010, 49, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Mendonca, L.M.; Schuermans, J.; Denolf, S.; Napier, C.; Bittencourt, N.F.N.; Romanuk, A.; Tak, I.; Thorborg, K.; Bizzini, M.; Ramponi, C.; et al. Sports injury prevention programmes from the sports physical therapist’s perspective: An international expert Delphi approach. Phys. Sport 2022, 55, 146–154. [Google Scholar] [CrossRef] [PubMed]
| AMSTAR QUESTIONS | Papadopoulou et al. 2022 [10] | Burton et al. 2022 [45] | Turnagol et al. 2022 [46] | Khatri et al. 2021 [47] | Smith-Ryan et al. 2020 [48] | Papadopoulou et al. 2020 [49] | Close et al. 2019 [13] | Quintero et al. 2018 [50] | Kahn et al. 2015 [51] | Tipton 2015 [17] | Wall et al. 2015 [52] | Pyne et al. 2014 [53] |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Did the research questions and inclusion criteria for the review include the components of PICO? | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| Did the report of the review contain an explicit statement that the review methods were established prior to the conduct of the review and did the report justify any significant deviations from the protocol? | No | Yes | No | Medium | No | No | No | Medium | No | No | No | No |
| Did the review authors explain their selection of the study designs for inclusion in the review? | No | Yes | No | Yes | No | No | No | Yes | No | No | No | No |
| Did the review authors use a comprehensive literature search strategy? | No | Medium | No | Medium | No | No | No | Medium | No | No | No | No |
| Did the review authors perform study selection in duplicate? | No | Yes | No | Yes | No | No | No | No | No | No | No | No |
| Did the review authors perform data extraction in duplicate? | No | Yes | No | Yes | No | No | No | No | No | No | No | No |
| Did the review authors provide a list of excluded studies and justify the exclusions? | No | Yes | No | Yes | No | No | No | Yes | No | No | No | No |
| Did the review authors describe the included studies in adequate detail? | No | No | No | Medium | No | No | No | No | No | No | No | No |
| Did the review authors use a satisfactory technique for assessing the risk of bias (RoB) in individual studies that were included in the review? | No | 0 | No | Yes | No | No | No | 0 | No | No | No | No |
| Did the review authors report on the sources of funding for the studies included in the review? | No | No | No | Yes | No | No | No | No | No | No | No | No |
| If meta-analysis was performed did the review authors use appropriate methods for statistical combination of results? | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| If meta-analysis was performed, did the review authors assess the potential impact of RoB in individual studies on the results of the meta-analysis or other evidence synthesis? | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Did the review authors account for RoB in individual studies when interpreting/discussing the results of the review? | No | No | No | Yes | No | No | No | No | No | No | No | No |
| Did the review authors provide a satisfactory explanation for, and discussion of, any heterogeneity observed in the results of the review? | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| If they performed quantitative synthesis did the review authors carry out an adequate investigation of publication bias (small study bias) and discuss its likely impact on the results of the review? | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Did the review authors report any potential sources of conflict of interest, including any funding they received for conducting the review? | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| Type of Study | Participants (M; F) | Aim | Methodology | Dosage and Timing | Main Findings | Reference |
|---|---|---|---|---|---|---|
| RCT/Quantitative Analysis | n = 45 (32 M; 13 F) high-performance athletes. Rugby (n = 17), Soccer (n = 10), Handball (n = 5), Judo (n = 4), Basketball (n = 3), Tennis (n = 2), Climbing (n = 1), Motocross (n = 1), Kitesurf (n = 1). | To determine the effect of a muscle 2–3-week rehabilitation program following ACL reconstruction and the influence of L-leucine supplementation on muscle strength in athletes undergoing sports reathletization. | Muscle strengthening exercises, proprioception, and running. Athletes were randomly assigned to receive L-leucine (n = 22) or placebo (n = 23). Thigh perimeter, isokinetic strength, single-leg long jump, and body fat (based on skinfolds) were measured. | 330 mg of L-leucine per capsule four times per day (1.2 g leucine daily). | A muscle rehabilitation program with or without leucine favored the improvement of muscle quality. However, leucine supplementation favored the recovery of the injured muscle and a reduction of 1.28% in body fat. | Laboute et al. (2013) [54] |
| Narrative Review/Qualitative Analysis | 74 references (from year of publication to 2015). | To summarize the physiological basis of muscle atrophy/disuse and discuss nutritional intervention strategies to limit muscle tissue loss during recovery from injury (including non-immobilization-induced disuse). | Expert view and non-structured analysis of the scientific literature. |
| Specific nutritional compounds, such as Ω3, high protein diet (including leucine), CrM, and HMB may assist in maintaining muscle protein synthesis rates during a period of injury. | Wall et al. (2015) [52] |
| RCT/Quantitative Analysis | n = 30 (30 M; 0 F) Athletes who underwent arthroscopic ACL reconstruction (73% were soccer players). | To examine the effectiveness of glucosamine sulfate administration on the rehabilitation outcomes of ACL reconstructed male athletes. | Athletes were assigned to receive glucosamine (n = 15) or placebo (n = 15) during 8 weeks. Knee pain (VAS), functional status, and isokinetic strength were measured. | 1000 mg of glucosamine sulfate per day for 8 weeks. | Glucosamine sulfate supplementation did not positively affect the rehabilitation outcomes. | Eraslan and Ulkar (2015) [55] |
| Narrative Review/Qualitative Analysis | 74 references (from year of publication to 2015). | To translate the knowledge regarding the role of vitamin D in athletic injuries to sports physical therapy practice. | Expert view and non-structured analysis of the scientific literature. | 4000 IU of Vitamin D per day or 50,000 IU per week for 8 weeks (to correct deficiency during rehabilitation). | Athletes with musculoskeletal injuries have significantly lower vitamin D levels relative to athletes without injuries. Treatment of vitamin D deficiency would lead to a decrease in the recurrence of musculoskeletal injuries. | Kahn et al. (2015) [51] |
| Narrative Review/Qualitative Analysis | 136 references (from year of publication to 2015). | To examine and update the evidence for nutritional strategies to support the enhancement of recovery and return to training and competition (focus on the first stage of injury, i.e., wound healing and reduced activity or immobilization). | Expert view and non-structured analysis of the scientific literature. |
| The best recommendation would be to adopt a ‘first, do no harm’ approach. The basis of nutritional strategy for an injured athlete should be a well-balanced diet based on whole foods from nature that are minimally processed. | Tipton (2015) [17] |
| RCT/Quantitative Analysis | n = 21 (21 M; 0 F) Swiss elite wheelchair indoor athletes with a spinal cord injury. Wheelchair rugby (n = 15), basketball (n = 4), or table tennis (n = 2). | To investigate the effect of vitamin D supplementation on muscle strength and performance in indoor wheelchair athletes. | Athletes received vitamin D for 12 weeks after detecting insufficiency at baseline. Muscle strength, power, and the extremity function and symptoms (DASH questionnaire) were measured. | 6000 IU of vitamin D3 (cholecalciferol) daily over 12 weeks. | Twelve weeks of vitamin D3 supplementation appears to be sufficient to reach an optimal vitamin D status in indoor wheelchair athletes. The real effect of vitamin D supplementation on upper body exercise performance in athletes with a spinal cord injury still remains unclear. | Flueck et al. (2016) [56] |
| Narrative Review/Qualitative Analysis | 89 references (from year of publication to 2019). | To identify nutritional strategies to assist with the most common injuries and consideration of the change in energy requirements during the injury period. | Expert view and non-structured analysis of the scientific literature. |
| The nutritional strategies discussed in this review can be implemented to decrease risk, marked loss of muscle mass due to disuse, and recovery time in the injured athlete. Supportive supervision should be provided to avoid low energy availability. | Close et al. (2019) [13] |
| Narrative Review/Qualitative Analysis | 80 references (from year of publication to 2018). | An overview of the nutritional strategies and recommendations after a muscular sports injury, emphasizing on muscle recovery. | Semi-structured literature search in PubMed, Science Direct, Scielo, Embase, and Google Scholar databases using specific search terms (MeSH and DeCS). |
| A high protein diet is recommended to maintain muscle mass. An adequate supply of antioxidant compounds and the use of probiotics might accelerate the muscle recovery process. | Quintero et al. (2018) [50] |
| RCT/Quantitative Analysis | n = 18 (10 M; 8 F) Injured adolescent male and female competitive fin swimmers. | To investigate the effect of creatine (Cr) supplementation on regeneration periods in tendon overuse injury rehabilitation of adolescent fin swimmers. | Athletes were assigned to receive CrM (n = 9) or placebo (n = 9) during 6 weeks as part of the conservative treatment of the tendinopathy. Segmental lean mass, ankle plantar flexion peak torque, pain intensity, and muscle damage were measured. | 20 g of CrM for 5 days (loading phase) followed by 5 g daily for 37 days (maintenance phase). | The results of this study indicate that CrM supplementation combined with therapeutic strategy effectively supports the rehabilitation of tendon overuse injury of adolescent fin swimmers. | Juhasz et al. (2018) [57] |
| Narrative Review/Qualitative Analysis | 316 references (from year of publication to 2020). | To provide a narrative synthesis of the scientific background related to selected topics (Expert Group Topic 7: Nutrition for Injury) within an elite sports setting. | Expert group statement with non-structured analysis of the scientific literature. |
| Given the metabolic demand of tissue/wound recovery processes, staying as close to energy balance as possible and thus avoiding drastic reductions in energy intake, is perhaps the most crucial nutritional aspect during rehabilitation. | Collins et al. (2020) [58] |
| Narrative Review/Qualitative Analysis | 77 references (from year of publication to 2020). | To define the proper nutrition for athletes in order to hasten their return to the sport after surgery or injury. | Expert view and non-structured analysis of the scientific literature. |
| Adequate intake of macronutrients can support anabolism in athletes. Dietary protocols should consider doses, timing, rehabilitation time, type, and quality of nutrients, as well as the type of injury, and the injured body part. | Papadopoulou et al. (2020) [49] |
| Narrative Review/Qualitative Analysis | 106 references (from year of publication to 2020). | To provide an evidence-based, practical guide for athletes with injuries treated surgically or conservatively, along with healing and rehabilitation considerations. | Expert view and non-structured analysis of the scientific literature. |
| The athlete’s energy requirements should be identified to avoid energy deficit. Higher protein intakes, with special attention to evenly distributed consumption throughout the day, will minimize loss of muscle mass and strength during times of immobilization. | Smith-Ryan et al. (2020) [48] |
| Systematic Review/Qualitative Analysis | 48 references (from year of publication to 2020). | To evaluate the effect of COL and exercise on joint function and athletic recovery. | Structured literature search in PubMed, Web of Science, and CINAHL. Fifteen references met the inclusion criteria. | 5–15 g of COL at least 1 h prior to exercise for over 3 months. | Strong evidence of COL use in improving joint pain and functionality (15 g/day may be a more effective dose). | Khatri et al. (2021) [47] |
| RCT/Quantitative Analysis | n = 8 (4 M; 4 F) Federated athletes (including basketball, volleyball, handball, and athletics) with patellar tendinopathy. | To analyze the effect of 4 weeks of physical rehabilitation with HMB supplementation in athletes diagnosed with patellar tendinopathy. | Athletes were assigned to receive HMB (n = 4) or placebo (n = 4) during 4 weeks. Body composition, perceived pain, and muscular function were measured. | 3 g of HMB per day 60 min before exercise. | HMB supplementation might enhance muscle power in athletes with patellar tendinopathy. It seems to optimize adaptions during the non-invasive treatment of the injury. | Sánchez-Gómez et al. (2022) [59] |
| Narrative Review/Qualitative Analysis | 77 references (from year of publication to 2022). | To define the proper nutritional elements tailored by athletes’ needs in order to facilitate their fast return to sports after surgery or injury. | Expert view and non-structured analysis of the scientific literature. |
| Diets that include high quality nutrients, rich in macro, micro, and bioactive compounds are recommended. Biomedical indices and vitamin and mineral levels should be evaluated and monitored to avoid deficiencies. | Papadopoulou et al. (2022) [10] |
| Narrative Review/Qualitative Analysis | 182 references (from year of publication to 2022). | To present various nutritional strategies for reducing the risk of injury and improving the treatment and rehabilitation process in combat sports. | Expert view and non-structured analysis of the scientific literature. |
| It is important to provide athletes with an adequate amount of macro- and micro-nutrients and nutritional supplements to meet the demands of the catabolic state and contribute to the injury-healing process. | Turnagöl et al. (2022) [46] |
| Scoping Review/Qualitative Analysis | 155 references (from year of publication to 2022). | To evaluate current research on the use of nutritional supplements for treating tendon injuries. | Structured literature search in Medline, Cinahl, Amed, EMBase, SPORTDiscus, and Cochrane. Sixteen references met the inclusion criteria. | COL, hydrolyzed COL, amino acids, vitamin C, glucosamine, HMB, Ω3, antioxidants, and CrM have been studied. | Certain nutritional supplements might have pain relieving, anti-inflammatory, and structural tendon effects that augment the positive functional outcomes gained from progressive exercise rehabilitation. | Burton et al. (2022) [45] |
| Cross-sectional Study/Quantitative Analysis | n = 133 (77 M; 56) International level Australian rowers from seniors (n = 115) and under 23-year-old levels (n = 18). | To identify nutrition-related factors associated with a history of rib stress injuries in elite rowers (including the injury time course). | Online questionnaire for historical records and a qualified dietitian collected information regarding habitual Ca intake. Body composition and BMD were measured with DXA. A sub-group of participants (n = 68) were assessed for vitamins D and K. | NA | Nutritional strategies to support injury prevention should focus on energy availability and its contribution to health and function, including menstrual status. | Lundy B et al. 2022 [44] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giraldo-Vallejo, J.E.; Cardona-Guzmán, M.Á.; Rodríguez-Alcivar, E.J.; Kočí, J.; Petro, J.L.; Kreider, R.B.; Cannataro, R.; Bonilla, D.A. Nutritional Strategies in the Rehabilitation of Musculoskeletal Injuries in Athletes: A Systematic Integrative Review. Nutrients 2023, 15, 819. https://doi.org/10.3390/nu15040819
Giraldo-Vallejo JE, Cardona-Guzmán MÁ, Rodríguez-Alcivar EJ, Kočí J, Petro JL, Kreider RB, Cannataro R, Bonilla DA. Nutritional Strategies in the Rehabilitation of Musculoskeletal Injuries in Athletes: A Systematic Integrative Review. Nutrients. 2023; 15(4):819. https://doi.org/10.3390/nu15040819
Chicago/Turabian StyleGiraldo-Vallejo, John E., Miguel Á. Cardona-Guzmán, Ericka J. Rodríguez-Alcivar, Jana Kočí, Jorge L. Petro, Richard B. Kreider, Roberto Cannataro, and Diego A. Bonilla. 2023. "Nutritional Strategies in the Rehabilitation of Musculoskeletal Injuries in Athletes: A Systematic Integrative Review" Nutrients 15, no. 4: 819. https://doi.org/10.3390/nu15040819
APA StyleGiraldo-Vallejo, J. E., Cardona-Guzmán, M. Á., Rodríguez-Alcivar, E. J., Kočí, J., Petro, J. L., Kreider, R. B., Cannataro, R., & Bonilla, D. A. (2023). Nutritional Strategies in the Rehabilitation of Musculoskeletal Injuries in Athletes: A Systematic Integrative Review. Nutrients, 15(4), 819. https://doi.org/10.3390/nu15040819











