25-Hydroxy-Vitamin D and Risk of Recurrent Stroke: A Dose Response Meta-Analysis
Abstract
:1. Introduction
1.1. Stroke: Definition and Epidemiology
1.2. Stroke Recurrence
1.3. Vitamin D System
2. Materials and Methods
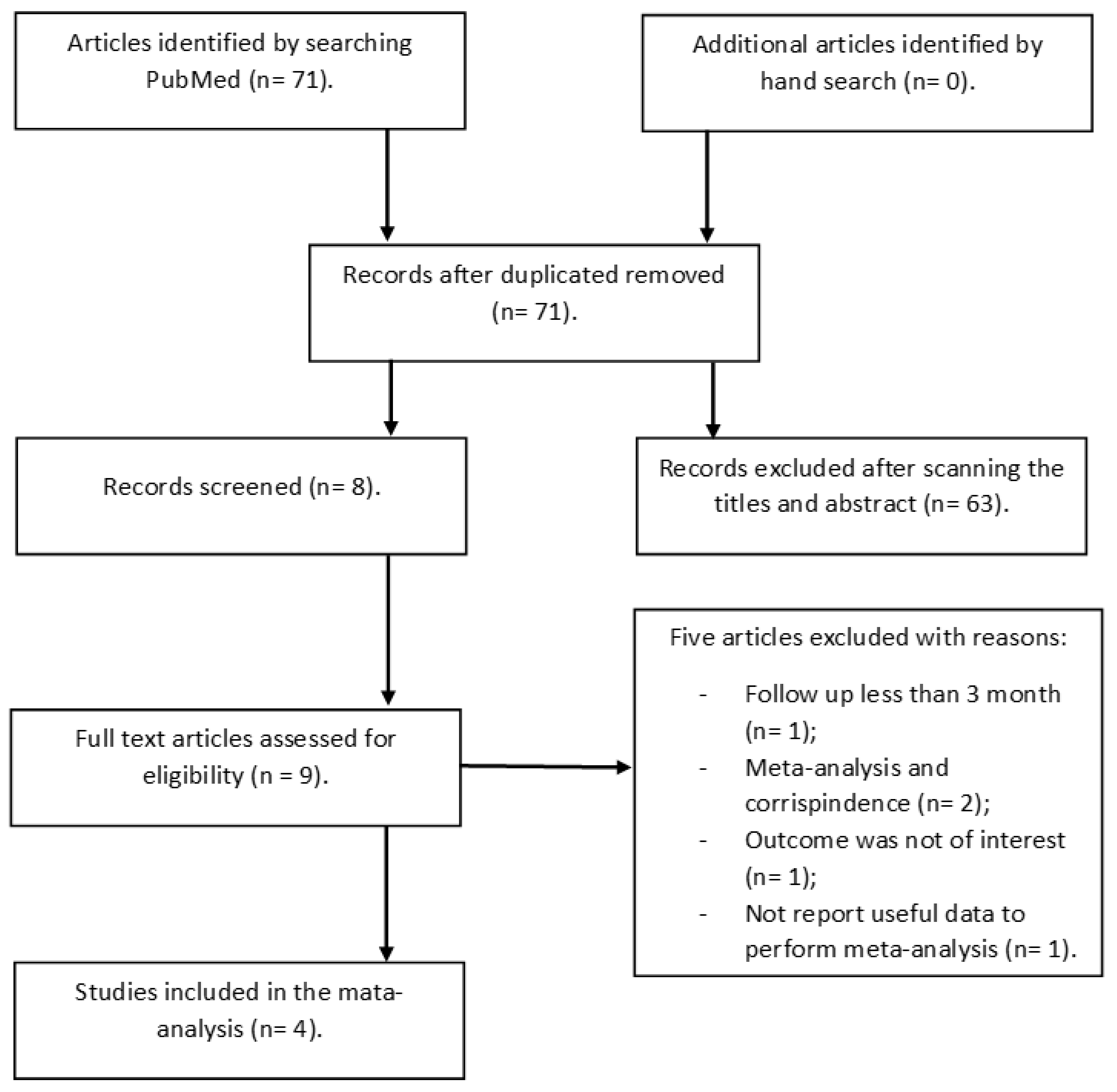
2.1. Data Source and Literature Search
2.2. Study Selection
2.3. Data Extraction and Quality Assessment
2.4. Statistical Analysis
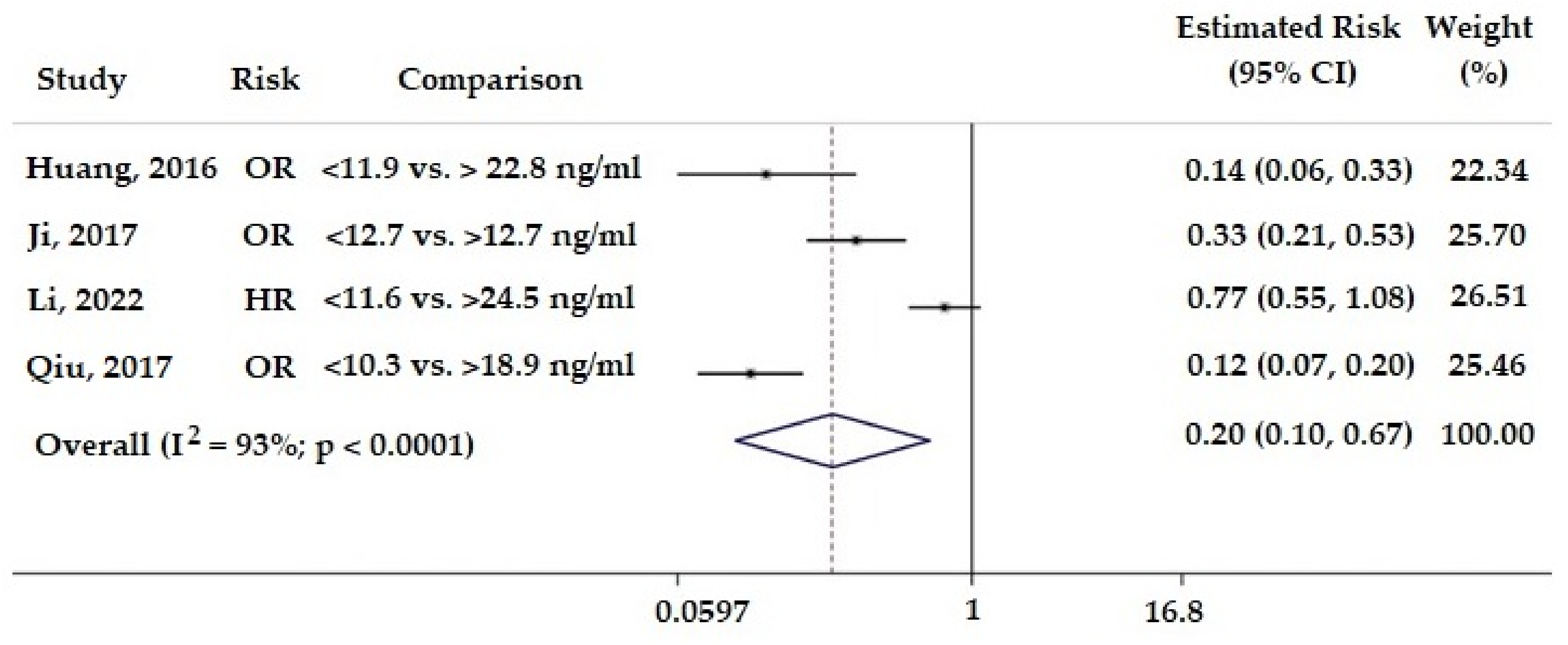
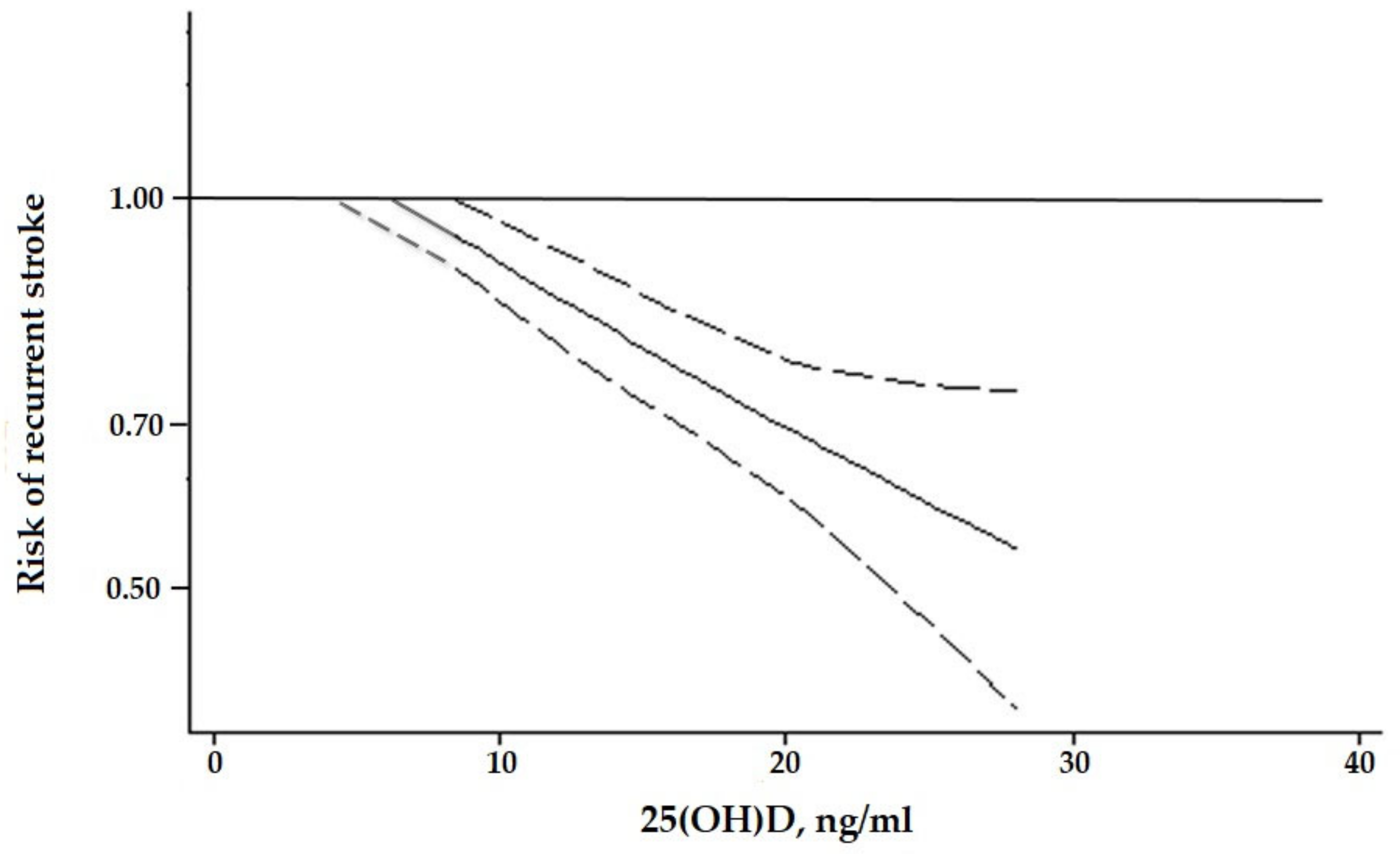
3. Results
4. Discussion
Study Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Campbell, B.C.V.; Khatri, P. Stroke. Lancet 2020, 396, 129–142. [Google Scholar] [CrossRef] [PubMed]
- GBD 2019 Stroke Collaborators. Global, regional, and national burden of stroke and its risk factors, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet Neurol. 2021, 20, 795–820. [Google Scholar] [CrossRef]
- Virani, S.S.; Alonso, A.; Aparicio, H.J.; Benjamin, E.J.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Cheng, S.; Delling, F.N.; et al. American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart Disease and Stroke Statistics-2021 Update: A Report from the American Heart Association. Circulation 2021, 143, e254–e743. [Google Scholar] [CrossRef]
- Kleindorfer, D.O.; Towfighi, A.; Chaturvedi, S.; Cockroft, K.M.; Gutierrez, J.; Lombardi-Hill, D.; Kamel, H.; Kernan, W.N.; Kittner, S.J.; Leira, E.C.; et al. 2021 Guideline for the Prevention of Stroke in Patients with Stroke and Transient Ischemic Attack: A Guideline From the American Heart Association/American Stroke Association. Stroke 2021, 52, e364–e467. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Li, L.; Adachi, J.D.; Wang, R.; Ye, Z.; Liu, X.; Thabane, L.; Lip, G. Relationship between Serum 25-Hydroxyvitamin D Level and Risk of Recurrent Stroke. Nutrients 2022, 14, 1908. [Google Scholar] [CrossRef] [PubMed]
- Flach, C.; Muruet, W.; Wolfe, C.; Bhalla, A.; Douiri, A. Risk and Secondary Prevention of Stroke Recurrence: A Population-Base Cohort Study. Stroke 2020, 51, 2435–2444. [Google Scholar] [CrossRef] [PubMed]
- Mohan, K.M.; Wolfe, C.D.; Rudd, A.G.; Heuschmann, P.U.; Kolominsky-Rabas, P.L.; Grieve, A.P. Risk and cumulative risk of stroke recurrence: A systematic review and meta-analysis. Stroke 2011, 42, 1489–1494. [Google Scholar] [CrossRef] [Green Version]
- Lin, B.; Zhang, Z.; Mei, Y.; Wang, C.; Xu, H.; Liu, L.; Wang, W. Cumulative risk of stroke recurrence over the last 10 years: A systematic review and meta-analysis. Neurol. Sci. 2021, 42, 61–71. [Google Scholar] [CrossRef]
- Khanevski, A.N.; Bjerkreim, A.T.; Novotny, V.; Naess, H.; Thomassen, L.; Logallo, N.; Kvistad, C.E.; NOR-STROKE Study Group. Recurrent ischemic stroke: Incidence, predictors, and impact on mortality. Acta Neurol. Scand. 2019, 140, 3–8. [Google Scholar] [CrossRef] [Green Version]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Hassan Murad, M.; Weaver, C.M. Evaluation, Treatment, and Prevention of Vitamin D Deficiency: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef]
- Haussler, M.R.; Haussler, C.A.; Bartik, L.; Whitfield, G.K.; Hsieh, J.C.; Slater, S.; Jurutka, P.W. Vitamin D receptor: Molecular signaling and actions of nutritional ligands in disease prevention. Nutr. Rev. 2008, 66, S98–S112. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Gooch, H.; Petty, A.; McGrath, J.J.; Eyles, D. Vitamin D and the brain: Genomic and non-genomic actions. Mol. Cell Endocrinol. 2017, 453, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Deo, R.; Katz, R.; Shlipak, M.G.; Sotoodehnia, N.; Psaty, B.M.; Sarnak, M.J.; Fried, L.F.; Chonchol, M.; de Boer, I.H.; Enquobahrie, D.; et al. Vitamin D, parathyroid hormone, and sudden cardiac death: Results from the Cardiovascular Health Study. Hypertension 2011, 58, 1021–1028. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valcour, A.; Blocki, F.; Hawkins, D.M.; Rao, S.D. Effects of age and serum 25-OH-vitamin D on serum parathyroid hormone levels. J. Clin. Endocrinol. Metab. 2012, 97, 3989–3995. [Google Scholar] [CrossRef]
- Zhang, Z.; Yang, Y.; Ng, C.Y.; Wang, D.; Wang, J.; Li, G.; Liu, T. Meta-analysis of Vitamin D Deficiency and Risk of Atrial Fibrillation. Clin. Cardiol. 2016, 39, 537–543. [Google Scholar] [CrossRef] [Green Version]
- Brøndum-Jacobsen, P.; Nordestgaard, B.G.; Schnohr, P.; Benn, M. 25-hydroxyvitamin D and symptomatic ischemic stroke: An original study and meta-analysis. Ann. Neurol. 2013, 73, 38–47. [Google Scholar] [CrossRef]
- Zhou, R.; Wang, M.; Huang, H.; Li, W.; Hu, Y.; Wu, T. Lower Vitamin D Status Is Associated with an Increased Risk of Ischemic Stroke: A Systematic Review and Meta-Analysis. Nutrients 2018, 10, 277. [Google Scholar] [CrossRef] [Green Version]
- Su, C.; Jin, B.; Xia, H.; Zhao, K. Association between Vitamin D and Risk of Stroke: A PRISMA-Compliant Systematic Review and Meta-Analysis. Eur. Neurol. 2021, 84, 399–408. [Google Scholar] [CrossRef]
- Liu, H.; Wang, J.; Xu, Z. Prognostic utility of serum 25-hydroxyvitamin D in patients with stroke: A meta-analysis. J. Neurol. 2020, 267, 3177–3186. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. J. Clin. Epidemiol. 2009, 62, e1–e34. [Google Scholar] [CrossRef]
- Greenland, S.; Longnecker, M.P. Methods for trend estimation from summarized dose-response data, with applications to meta-analysis. Am. J. Epidemiol. 1992, 135, 1301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DerSimonian, R.; Laird, N. Meta-analysis in clinical trials revisited. Contemp. Clin. Trials 2015, 45, 139–145. [Google Scholar] [CrossRef] [Green Version]
- Qiu, H.; Wang, M.; Mi, D.; Zhao, J.; Tu, W.; Liu, Q. Vitamin D Status and the Risk of Recurrent Stroke and Mortality in Ischemic Stroke Patients: Data from a 24-Month Follow-Up Study in China. J. Nutr. Health Aging 2017, 21, 766–771. [Google Scholar] [CrossRef]
- Huang, H.; Zheng, T.; Wang, S.; Wei, L.; Wang, Q.; Sun, Z. Serum 25-hydroxyvitamin D predicts early recurrent stroke in ischemic stroke patients. Nutr. Metab. Cardiovasc. Dis. 2016, 26, 908–914. [Google Scholar] [CrossRef]
- Ji, W.; Zhou, H.; Wang, S.; Cheng, L.; Fang, Y. Low Serum Levels of 25-Hydroxyvitamin D Are Associated with Stroke Recurrence and Poor Functional Outcomes in Patients with Ischemic Stroke. J. Nutr. Health Aging 2017, 21, 892–896. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.J.; Dusso, A.; Slatopolsky, E. Vitamin D. Am. J. Physiol. 1999, 277, F157–F175. [Google Scholar] [CrossRef]
- Tonney, F.O. Vitamin D in Child Health. Am. J. Public Health Nations Health 1936, 26, 665–671. [Google Scholar] [CrossRef] [Green Version]
- Dusso, A.S.; Brown, A.J.; Slatopolsky, E. Vitamin D. Am. J. Physiol. Renal Physiol. 2005, 289, F8–F28. [Google Scholar] [CrossRef]
- Thomas, M.K.; Lloyd-Jones, D.M.; Thadhani, R.I.; Shaw, A.C.; Deraska, D.J.; Kitch, B.T.; Vamvakas, E.C.; Dick, I.M.; Prince, R.L.; Finkelstein, J.S. Hypovitaminosis D in medical inpatients. N. Engl. J. Med. 1998, 338, 777–783. [Google Scholar] [CrossRef] [PubMed]
- Pilz, S.; Tomaschitz, A.; Ritz, E.; Pieber, T.R. Vitamin D status and arterial hypertension: A systematic review. Nat. Rev. Cardiol. 2009, 6, 621–630. [Google Scholar] [CrossRef] [PubMed]
- Mirhosseini, N.; Vatanparast, H.; Kimball, S.M. The Association between Serum 25(OH)D Status and Blood Pressure in Participants of a Community-Based Program Taking Vitamin D Supplements. Nutrients 2017, 9, 1244. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pittas, A.G.; Dawson-Hughes, B. Vitamin D and diabetes. J. Steroid. Biochem. Mol. Biol. 2010, 121, 425–429. [Google Scholar] [CrossRef] [PubMed]
- Dawson-Hughes, B.; Staten, M.A.; Knowler, W.C.; Nelson, J.; Vickery, E.M.; LeBlanc, E.S.; Neff, L.M.; Park, J.; Pittas, A.G.; D2d Research Group. Intratrial Exposure to Vitamin D and New-Onset Diabetes Among Adults with Prediabetes: A Secondary Analysis from the Vitamin D and Type 2 Diabetes (D2d) Study. Diabetes Care 2020, 43, 2916–2922. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, N.; Brandsch, C.; Kühne, H.; Thiele, A.; Hirche, F.; Stangl, G.I. Vitamin D receptor deficiency and low vitamin D diet stimulate aortic calcification and osteogenic key factor expression in mice. PLoS ONE 2012, 7, e35316. [Google Scholar] [CrossRef]
- Surdu, A.M.; Pînzariu, O.; Ciobanu, D.M.; Negru, A.G.; Căinap, S.S.; Lazea, C.; Iacob, D.; Săraci, G.; Tirinescu, D.; Borda, I.M.; et al. Vitamin D and Its Role in the Lipid Metabolism and the Development of Atherosclerosis. Biomedicines 2021, 9, 172. [Google Scholar] [CrossRef] [PubMed]
- Hewison, M. Vitamin D and the immune system: New perspectives on an old theme. Endocrinol. Metab. Clin. N. Am. 2010, 39, 365–379. [Google Scholar] [CrossRef] [Green Version]
- Muscogiuri, G.; Annweiler, C.; Duval, G.; Karras, S.; Tirabassi, G.; Salvio, G.; Balercia, G.; Kimball, S.; Kotsa, K.; Mascitelli, L.; et al. Vitamin D and cardiovascular disease: From atherosclerosis to myocardial infarction and stroke. Int. J. Cardiol. 2017, 230, 577–584. [Google Scholar] [CrossRef]
- Uberti, F.; Lattuada, D.; Morsanuto, V.; Nava, U.; Bolis, G.; Vacca, G.; Squarzanti, D.F.; Cisari, C.; Molinari, C. Vitamin D protects human endothelial cells from oxidative stress through the autophagic and survival pathways. J. Clin. Endocrinol. Metab. 2014, 99, 1367–1374. [Google Scholar] [CrossRef] [Green Version]
- Holick, M.F. Vitamin D deficiency. N Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Ashouri, R.; Fangman, M.; Brielmaier, J.; Fields, Z.A.; Campo, N.; Doré, S. Nutritional Supplementation of Naturally Occurring Vitamin D to Improve Hemorrhagic Stroke Outcomes. Front. Neurol. 2021, 12, 670245. [Google Scholar] [CrossRef] [PubMed]
- Charoenngam, N.; Holick, M.F. Immunologic Effects of Vitamin D on Human Health and Disease. Nutrients 2020, 12, 2097. [Google Scholar] [CrossRef]
- Charoenngam, N.; Shirvani, A.; Holick, M.F. Vitamin D for skeletal and non-skeletal health: What we should know. J. Clin. Orthop. Trauma 2019, 10, 1082–1093. [Google Scholar] [CrossRef] [PubMed]
- Verdoia, M.; Pergolini, P.; Rolla, R.; Sartori, C.; Nardin, M.; Schaffer, A.; Barbieri, L.; Daffara, V.; Marino, P.; Bellomo, G.; et al. Vitamin D levels and high-residual platelet reactivity in patients receiving dual antiplatelet therapy with clopidogrel or ticagrelor. Platelets 2016, 27, 576–582. [Google Scholar] [CrossRef] [PubMed]
- Giovannucci, E.; Liu, Y.; Hollis, B.W.; Rimm, E.B. 25-hydroxyvitamin D and risk of myocardial infarction in men: A prospective study. Arch. Intern. Med. 2008, 168, 1174–1180. [Google Scholar] [CrossRef] [Green Version]
- Nibbelink, K.A.; Tishkoff, D.X.; Hershey, S.D.; Rahman, A.; Simpson, R.U. 1,25(OH)2-vitamin D3 actions on cell proliferation, size, gene expression, and receptor localization, in the HL-1 cardiac myocyte. J. Steroid. Biochem. Mol. Biol. 2007, 103, 533–537. [Google Scholar] [CrossRef] [Green Version]
- Zhou, A.; Selvanayagam, J.B.; Hyppönen, E. Non-linear Mendelian randomization analyses support a role for vitamin D deficiency in cardiovascular disease risk. Eur. Heart J. 2022, 43, 1731–1739. [Google Scholar] [CrossRef]
- Monraats, P.S.; Fang, Y.; Pons, D.; Pires, N.M.; Pols, H.A.; Zwinderman, A.H.; de Maat, M.P.; Doevendans, P.A.; DeWinter, R.J.; Tio, R.A.; et al. Vitamin D receptor: A new risk marker for clinical restenosis after percutaneous coronary intervention. Expert Opin. Ther. Targets 2010, 14, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Hagström, E.; Hellman, P.; Larsson, T.E.; Ingelsson, E.; Berglund, L.; Sundström, J.; Melhus, H.; Held, C.; Lind, L.; Michaëlsson, K.; et al. Plasma parathyroid hormone and the risk of cardiovascular mortality in the community. Circulation 2009, 119, 2765–2771. [Google Scholar] [CrossRef] [PubMed]
- Cortese, F.; Costantino, M.F.; Luzi, G.; Di Marino, S.; Giordano, P.; Monitillo, F. Vitamin D and cardiovascular disease risk. A literature overview. Mol. Biol. Rep. 2022, 49, 8925–8942. [Google Scholar] [CrossRef] [PubMed]
- Rendina, D.; De Filippo, G.; Muscariello, R.; De Palma, D.; Fiengo, A.; De Pascale, F.; Strazzullo, P. Vitamin D and cardiometabolic disorders. High Blood Press. Cardiovasc. Prev. 2014, 21, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. High prevalence of vitamin D inadequacy and implications for health. Mayo Clin. Proc. 2006, 81, 353–373. [Google Scholar] [CrossRef] [Green Version]
- Sutherland, J.P.; Zhou, A.; Hyppönen, E. Vitamin D Deficiency Increases Mortality Risk in the UK Biobank: A Nonlinear Mendelian Randomization Study. Ann. Intern. Med. 2022, 175, 1552–1559. [Google Scholar] [CrossRef] [PubMed]
- Manson, J.E.; Cook, N.R.; Lee, I.M.; Christen, W.; Bassuk, S.S.; Mora, S.; Gibson, H.; Gordon, D.; Copeland, T.; D’Agostino, D.; et al. Vitamin D Supplements and Prevention of Cancer and Cardiovascular Disease. N. Engl. J. Med. 2019, 380, 33–44. [Google Scholar] [CrossRef]
- Pilz, S.; Zittermann, A.; Trummer, C.; Theiler-Schwetz, V.; Lerchbaum, E.; Keppel, M.H.; Grübler, M.R.; März, W.; Pandis, M. Vitamin D testing and treatment: A narrative review of current evidence. Endocr. Connect 2019, 8, R27–R43. [Google Scholar] [CrossRef] [Green Version]
- Pilz, S.; Trummer, C.; Theiler-Schwetz, V.; Grübler, M.R.; Verheyen, N.D.; Odler, B.; Karras, S.N.; Zittermann, A.; März, W. Critical Appraisal of Large Vitamin D Randomized Controlled Trials. Nutrients 2022, 14, 303. [Google Scholar] [CrossRef]
- Gaksch, M.; Jorde, R.; Grimnes, G.; Joakimsen, R.; Schirmer, H.; Wilsgaard, T.; Mathiesen, E.B.; Njølstad, I.; Løchen, M.L.; März, W.; et al. Vitamin D and mortality: Individual participant data meta-analysis of standardized 25-hydroxyvitamin D in 26916 individuals from a European consortium. PLoS ONE 2017, 12, e0170791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ross, A.C.; Taylor, C.L.; Yaktine, A.L.; Del Valle, H.B. Institute of Medicine (US) Committee to Review Dietary Reference Intakes for Vitamin D and Calcium. In Dietary Reference Intakes for Calcium and Vitamin D; National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Pludowski, P.; Takacs, I.; Boyanov, M.; Belaya, Z.; Diaconu, C.C.; Mokhort, T.; Zherdova, N.; Rasa, I.; Payer, J.; Pilz, S. Clinical Practice in the Prevention, Diagnosis and Treatment of Vitamin D Deficiency: A Central and Eastern European Expert Consensus Statement. Nutrients 2022, 14, 1483. [Google Scholar] [CrossRef] [PubMed]
- Bouillon, R.; Carmeliet, G. Vitamin D insufficiency: Definition, diagnosis and management. Best Pract. Res. Clin. Endocrinol. Metab. 2018, 32, 669–684. [Google Scholar] [CrossRef]
- Chan, Y.H.; Schooling, C.M.; Zhao, J.; Au Yeung, S.L.; Hai, J.J.; Thomas, G.N.; Cheng, K.K.; Jiang, C.Q.; Wong, Y.K.; Au, K.W.; et al. Mendelian Randomization Focused Analysis of Vitamin D on the Secondary Prevention of Ischemic Stroke. Stroke 2021, 52, 3926–3937. [Google Scholar] [CrossRef]
- Hyppönen, E.; Vimaleswaran, K.S.; Zhou, A. Genetic Determinants of 25-Hydroxyvitamin D Concentrations and Their Relevance to Public Health. Nutrients 2022, 14, 4408. [Google Scholar] [CrossRef]

| Author | Year | Ref | SD | P | M | Age | FU | HTN | T2D | AF | HCL | SM | R-S | Death | NOS |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Qiu H | 2017 | [23] | p | 220 | 61.6 | 65 | 24 | 80.1 | 43.1 | 25.0 | 45.8 | 25.9 | 40 | 30.1 | 8 |
| Ji W | 2017 | [25] | p | 277 | 58.9 | 65 | 6 | 59.6 | 28.2 | 22.4 | 29.2 | 26.4 | 31 | - | 7 |
| Huang H | 2016 | [24] | p | 396 | 58.2 | 68 | 3 | 54.2 | 32.1 | 26.1 | 35.8 | 18.6 | 37 | 11.9 | 7 |
| Li G | 2022 | [5] | p | 6824 | 59.2 | 60.6 | 86 | 61.9 | 14.7 | 7.8 | 43.9 | 16.2 | 388 | 9.6 | 7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vergatti, A.; Abate, V.; Zarrella, A.F.; Manganelli, F.; Tozza, S.; Iodice, R.; De Filippo, G.; D’Elia, L.; Strazzullo, P.; Rendina, D. 25-Hydroxy-Vitamin D and Risk of Recurrent Stroke: A Dose Response Meta-Analysis. Nutrients 2023, 15, 512. https://doi.org/10.3390/nu15030512
Vergatti A, Abate V, Zarrella AF, Manganelli F, Tozza S, Iodice R, De Filippo G, D’Elia L, Strazzullo P, Rendina D. 25-Hydroxy-Vitamin D and Risk of Recurrent Stroke: A Dose Response Meta-Analysis. Nutrients. 2023; 15(3):512. https://doi.org/10.3390/nu15030512
Chicago/Turabian StyleVergatti, Anita, Veronica Abate, Aquilino Flavio Zarrella, Fiore Manganelli, Stefano Tozza, Rosa Iodice, Gianpaolo De Filippo, Lanfranco D’Elia, Pasquale Strazzullo, and Domenico Rendina. 2023. "25-Hydroxy-Vitamin D and Risk of Recurrent Stroke: A Dose Response Meta-Analysis" Nutrients 15, no. 3: 512. https://doi.org/10.3390/nu15030512






