Athletes Can Benefit from Increased Intake of EPA and DHA—Evaluating the Evidence
Abstract
1. Introduction
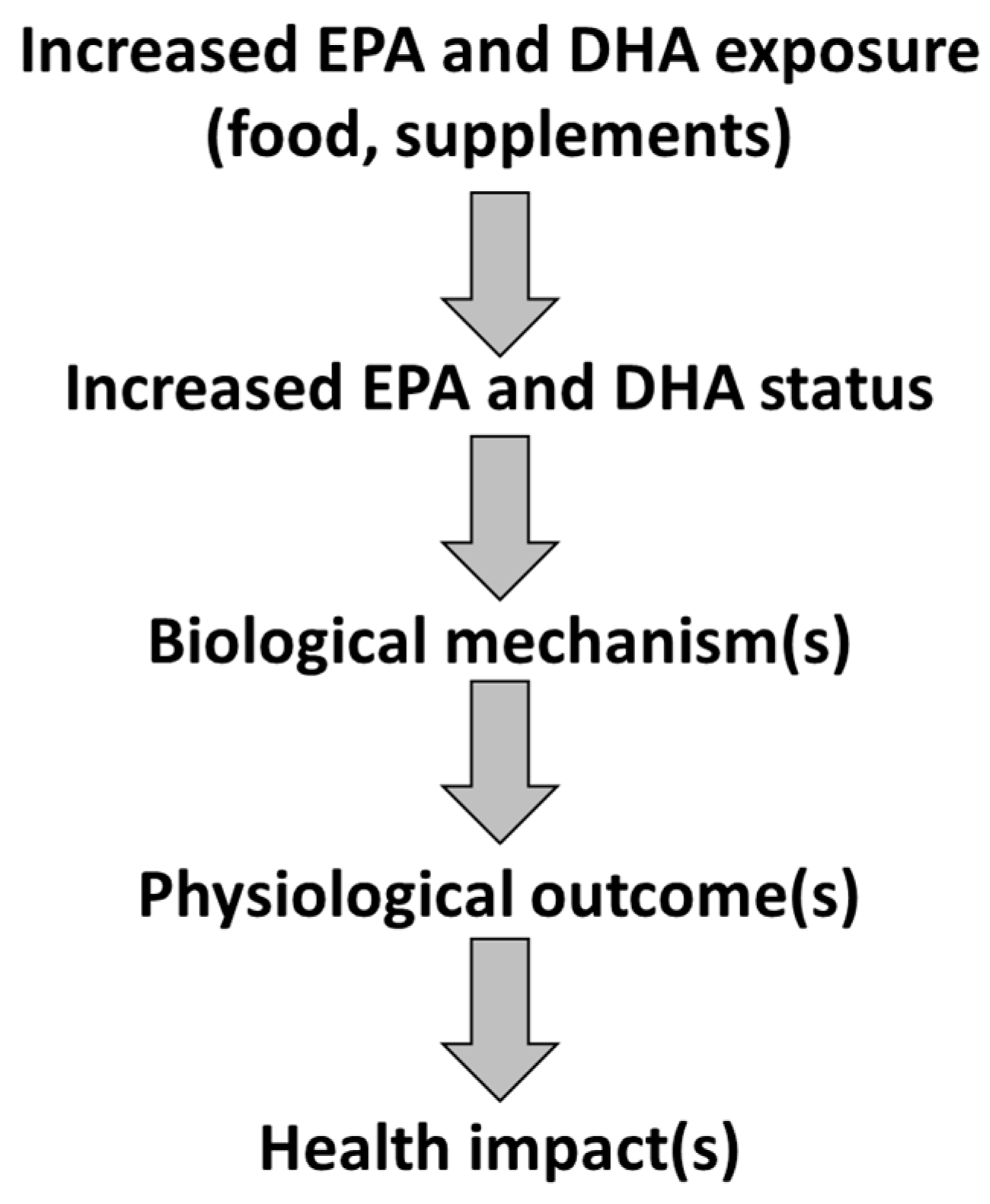
2. Effects of EPA and DHA and Underlying Mechanisms
3. Nutritional Considerations about EPA and DHA
4. Status of EPA and DHA in Athletes
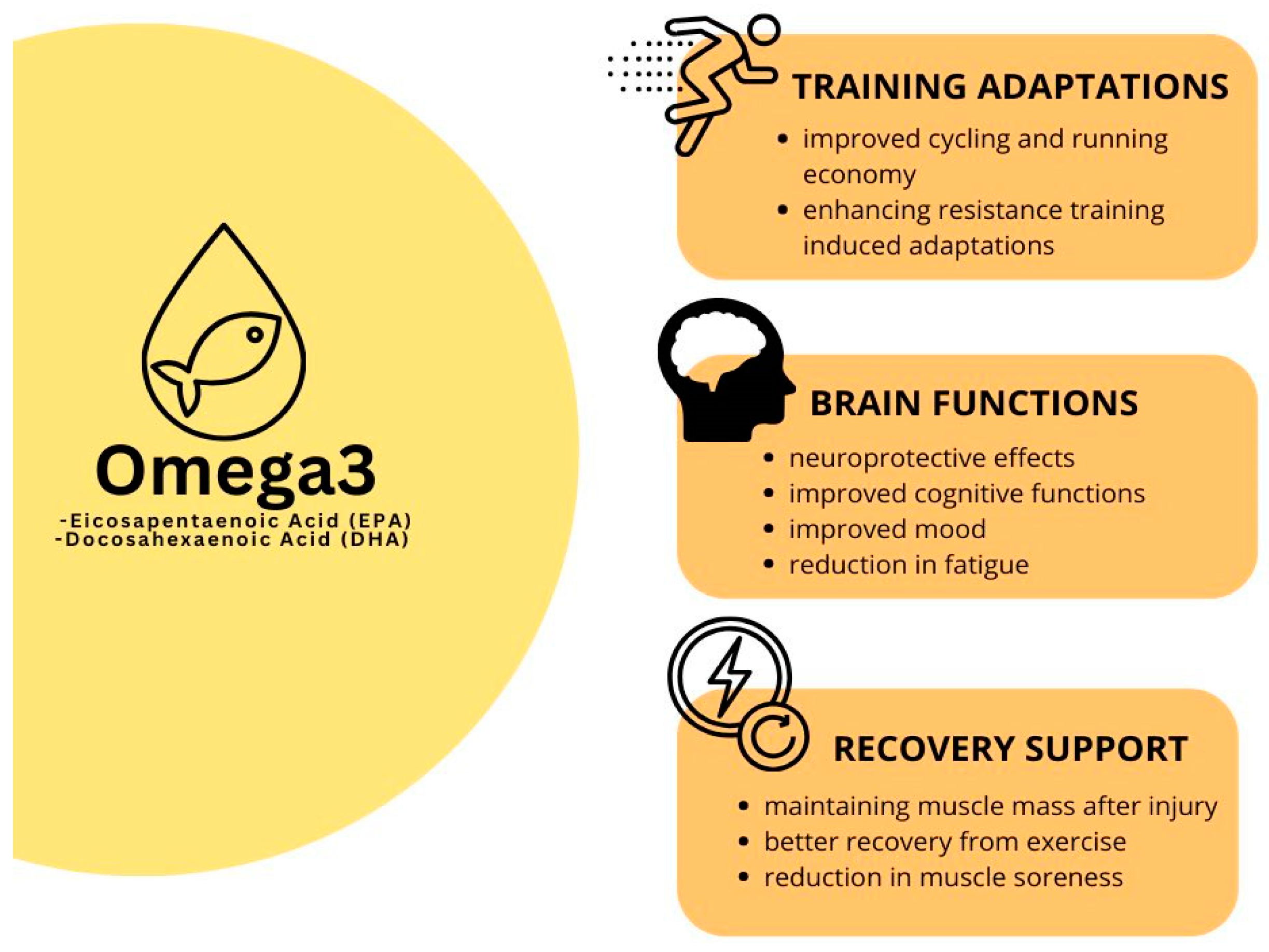
5. EPA and DHA for Athletes’ Health
6. EPA and DHA and Recovery from Exercise
7. EPA and DHA and Training Adaptation
8. Potential Adverse Impacts of EPA and DHA Supplementation
- “long-term supplemental intakes of EPA and DHA combined up to about 5 g/day do not increase the risk of spontaneous bleeding episodes or bleeding complications even in subjects at high risk of bleeding”;
- “supplemental intakes of EPA and DHA consumed either alone or in combination at doses up to about 5 g/day for up to 16 weeks do not induce changes in lipid peroxidation which might raise concern in relation to cardiovascular disease risk as long as the oxidative stability of these [fatty acids] is guaranteed”;
- “supplemental intakes of EPA and DHA combined at doses up to 5 g/day, and supplemental intakes of EPA alone up to 1.8 g/day, do not raise safety concerns for the adult population”;
- “supplemental intakes of DHA alone up to about 1 g/day do not raise safety concerns for the general population”.
9. Other Points to Consider
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Australian Institute of Sport; Australian Sports Commission. The AIS Sports Supplement Framework; Australian Institute of Sport: Bruce, Australia, 2021.
- Calder, P.C. Very long-chain n-3 fatty acids and human health: Fact, fiction and the future. Proc. Nutr. Soc. 2018, 77, 52–72. [Google Scholar] [CrossRef] [PubMed]
- Baker, E.J.; Miles, E.A.; Burdge, G.C.; Yaqoob, P.; Calder, P.C. Metabolism and functional effects of plant-derived omega-3 fatty acids in humans. Prog. Lipid Res. 2016, 64, 30–56. [Google Scholar] [CrossRef] [PubMed]
- Emken, E.A.; Adlof, R.O.; Duval, S.M.; Nelson, G.J. Effect of dietary docosahexaenoic acid on desaturation and uptake in vivo of isotope-labeled oleic, linoleic, and linolenic acids by male subjects. Lipids 1999, 34, 785–791. [Google Scholar] [CrossRef] [PubMed]
- Salem, N.; Pawlosky, R.; Wegher, B.; Hibbeln, J. In vivo conversion of linoleic acid to arachidonic acid in human adults. Prostaglandins Leukot. Essent. Fat. Acids 1999, 60, 407–410. [Google Scholar] [CrossRef] [PubMed]
- Burdge, G.C.; Jones, A.E.; Wootton, S.A. Eicosapentaenoic and docosapentaenoic acids are the principal products of α-linolenic acid metabolism in young men. Br. J. Nutr. 2002, 88, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Burdge, G.C.; Wootton, S.A. Conversion of α-linolenic acid to eicosapentaenoic, docosapentaenoic and docosahexaenoic acids in young women. Br. J. Nutr. 2002, 88, 411–420. [Google Scholar] [CrossRef] [PubMed]
- Hussein, N.; Ah-Sing, E.; Wilkinson, P.; Leach, C.; Griffin, B.A.; Millward, D.J. Long-chain conversion of [13C]linoleic acid and α-linolenic acid in response to marked changes in their dietary intake in men. J. Lipid Res. 2005, 46, 269–280. [Google Scholar] [CrossRef]
- Pawlosky, R.J.; Hibbeln, J.R.; Novotny, J.A.; Salem, N., Jr. Physiological compartmental analysis of alpha-linolenic acid metabolism in adult humans. J. Lipid Res. 2001, 42, 1257–1265. [Google Scholar] [CrossRef]
- Harris, W.S.; von Schacky, C. The Omega-3 Index: A new risk factor for death from coronary heart disease? Prev. Med. 2004, 39, 212–220. [Google Scholar] [CrossRef]
- Von Schacky, C.; Kemper, M.; Haslbauer, R.; Halle, M. Low omega-3 index in 106 German elite winter endurance athletes: A pilot study. Int. J. Sport Nutr. Exerc. Metab. 2014, 24, 559564. [Google Scholar] [CrossRef]
- Craddock, J.C.; Probst, Y.C.; Neale, E.P.; Peoples, G.E. A cross-sectional comparison of the whole blood fatty acid profile and omega-3 index of male vegan and omnivorous endurance athletes. J. Am. Nutr. Assoc. 2022, 41, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Anzalone, A.; Carbuhn, A.; Jones, L.; Gallop, A.; Smith, A.; Johnson, P.; Swearingen, L.; Moore, C.; Rimer, E.; McBeth, J.; et al. The omega-3 index in National Collegiate Athletic Association division I collegiate football athletes. J. Athl. Train. 2019, 54, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Lust, C.A.C.; Burns, J.L.; Jones, M.T.; Smith, S.B.; Choi, S.H.; Krk, M.; Gable, D.A.; Oliver, J.M.; Ma, D.W.L. The dose-response effect of docosahexaenoic acid on the omega-3 index in American football athletes. Med. Sci. Sports Exerc. 2023, 55, 865–872. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.K.; Freese, E.C.; Wolfe, A.S.; Basham, S.A.; Stein, K.M.W. Evaluation of omega-3 status in professional basketball players. J. Strength Cond. Res. 2021, 35, 1794–1799. [Google Scholar] [CrossRef] [PubMed]
- Kunces, L.J.; Keenan, J.; Schmidt, C.M.; Schmidt, M.A. Molecular deficits relevant to concussion are prevalent in top-ranked football players entering the national football league draft. J. Strength Cond. Res. 2021, 35, 339–3144. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, A.; Anzalone, A.J.; Pethick, W.; Murray, H.; Dahlquist, D.T.; Askow, A.T.; Heileson, J.L.; Hillyer, L.M.; Ma, D.W.L.; Oliver, J.M. An evaluation of omega-3 status and intake in Canadian elite rugby 7s players. Nutrients 2021, 13, 3777. [Google Scholar] [CrossRef] [PubMed]
- Arsić, A.; Vučić, V.; Tepšić, J.; Mazić, S.; Djelić, M.; Glibetić, M. Altered plasma and erythrocyte phospholipid fatty acid profile in elite female water polo and football players. Appl. Physiol. Nutr. Metab. 2012, 37, 40–47. [Google Scholar] [CrossRef]
- Fontani, G.; Corradeschi, F.; Felici, A.; Alfatti, F.; Migliorini, S.; Lodi, L. Cognitive and physiological effects of Omega-3 polyunsaturated fatty acid supplementation in healthy subjects. Eur. J. Clin. Investig. 2005, 35, 691–699. [Google Scholar] [CrossRef]
- Guzmán, J.F.; Esteve, H.; Pablos, C.; Pablos, A.; Blasco, C.; Villegas, J.A. DHA-rich fish oil improves complex reaction time in female elite soccer players. J. Sports Sci. Med. 2011, 10, 301–305. [Google Scholar]
- Oliver, J.M.; Jones, M.T.; Kirk, K.M.; Gable, D.A.; Repshas, J.T.; Johnson, T.A.; Andréasson, U.; Norgren, N.; Blennow, K.; Zetterberg, H. Effect of docosahexaenoic acid on a biomarker of head trauma in American Football. Med. Sci. Sports Exerc. 2016, 48, 974–982. [Google Scholar] [CrossRef]
- Heileson, J.L.; Machek, S.B.; Harris, D.R.; Tomek, S.; de Souza, L.C.; Kieffer, A.J.; Barringer, N.D.; Gallucci, A.; Forsse, J.S.; Funderburk, L.L.K. The effect of fish oil supplementation on resistance training-induced adaptations. J. Int. Soc. Sports Nutr. 2023, 20, 2174704. [Google Scholar] [CrossRef] [PubMed]
- You, J.S.; Park, M.N.; Song, W.; Lee, Y.S. Dietary fish oil alleviates soleus atrophy during immobilization in association with Akt signaling to p70s6k and E3 ubiquitin ligases in rats. Appl. Physiol. Nutr. Metab. 2010, 35, 310–318. [Google Scholar] [CrossRef] [PubMed]
- You, J.S.; Park, M.N.; Lee, Y.S. Dietary fish oil inhibits the early stage of recovery of atrophied soleus muscle in rats via Akt-p70s6k signaling and PGF2α. J. Nutr. Biochem. 2010, 21, 929–934. [Google Scholar] [CrossRef] [PubMed]
- McGlory, C.; Gorissen, S.H.M.; Kamal, M.; Bahniwal, R.; Hector, A.J.; Baker, S.K.; Chabowski, A.; Phillips, S.M. Omega-3 fatty acid supplementation attenuates skeletal muscle disuse atrophy during two weeks of unilateral leg immobilization in healthy young women. FASEB J. 2019, 33, 4586–4597. [Google Scholar] [CrossRef] [PubMed]
- Tomczyk, M.; Jost, Z.; Chroboczek, M.; Urbański, R.; Calder, P.C.; Fisk, H.L.; Sprengel, M.; Antosiewicz, J. Effects of 12 wk of omega-3 fatty acid supplementation in long-distance runners. Med. Sci. Sports Exerc. 2023, 55, 216–224. [Google Scholar] [CrossRef]
- Tsuchiya, Y.; Yanagimoto, K.; Nakazato, K.; Hayamizu, K.; Ochi, E. Eicosapentaenoic and docosahexaenoic acids-rich fish oil supplementation attenuates strength loss and limited joint range of motion after eccentric contractions: A randomized, double-blind, placebo-controlled, parallel-group trial. Eur. J. Appl. Physiol. 2016, 116, 216–224. [Google Scholar] [CrossRef]
- Calder, P.C. Mechanisms of action of (n-3) fatty acids. J. Nutr. 2012, 142, 592S–599S. [Google Scholar] [CrossRef]
- Deckelbaum, R.J.; Worgall, T.S.; Seo, T. n-3 fatty acids and gene expression. Am. J. Clin. Nutr. 2006, 83, 1520S–1525S. [Google Scholar] [CrossRef]
- Calder, P.C. N-3 PUFA and inflammation: From membrane to nucleus and from bench to bedside. Proc. Nutr. Soc. 2020, 79, 404–416. [Google Scholar] [CrossRef]
- Christiansen, E.; Watterson, K.R.; Stocker, C.J.; Sokol, E.; Jenkins, L.; Simon, K.; Grundmann, M.; Petersen, R.K.; Wargent, E.T.; Hudson, B.D.; et al. Activity of dietary fatty acids on FFA1 and FFA4 and characterisation of pinolenic acid as a dual FFA1/FFA4 agonist with potential effect against metabolic diseases. Br. J. Nutr. 2015, 113, 1677–1688. [Google Scholar] [CrossRef]
- Moniri, N.H. Free-fatty acid receptor-4 (GPR120): Cellular and molecular function and its role in metabolic disorders. Biochem. Pharmacol. 2016, 110–111, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Im, D.S. Functions of omega-3 fatty acids and FFA4 (GPR120) in macrophages. Eur. J. Pharmacol. 2016, 785, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Sohn, K.H.; Rhee, S.H.; Hwang, D. Saturated Fatty acids, but not unsaturated fatty acids, induce the expression of cyclooxygenase-2 mediated through Toll-like Receptor 4. J. Biol. Chem. 2001, 276, 16683–16689. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Ye, J.; Gao, Z.; Youn, H.S.; Lee, W.H.; Zhao, L.; Sizemore, N.; Hwang, D.H. Reciprocal modulation of toll-like receptor-4 signaling pathways involving MyD88 and phosphatidylinositol 3-kinase/AKT by saturated and polyunsaturated fatty acids. J. Biol. Chem. 2003, 278, 37041–37051. [Google Scholar] [CrossRef] [PubMed]
- Weatherill, A.R.; Lee, J.Y.; Zhao, L.; Lemay, D.G.; Youn, H.S.; Hwang, D.H. Saturated and polyunsaturated fatty acids reciprocally modulate dendritic cell functions mediated through TLR4. J. Immunol. 2005, 174, 5390–5397. [Google Scholar] [CrossRef]
- Wong, S.W.; Kwon, M.J.; Choi, A.M.K.; Kim, H.P.; Nakahira, K.; Hwang, D.H. Fatty acids modulate toll-like receptor 4 activation through regulation of receptor dimerization and recruitment into lipid rafts in a reactive oxygen species-dependent manner. J. Biol. Chem. 2009, 284, 27384–27392. [Google Scholar] [CrossRef] [PubMed]
- Gani, O.A.; Sylte, I. Molecular recognition of docosahexaenoic acid by peroxisome proliferator-activated receptors and retinoid-X receptor α. J. Mol. Graph. Model. 2008, 27, 217–224. [Google Scholar] [CrossRef]
- Allred, C.D.; Talbert, D.R.; Southard, R.C.; Wang, X.; Kilgore, M.W. PPARγ1 as a molecular target of eicosapentaenoic acid in human colon cancer (HT-29) cells. J. Nutr. 2008, 138, 250–256. [Google Scholar] [CrossRef]
- Genolet, R.; Wahli, W.; Michalik, L. PPARs as drug targets to modulate inflammatory responses? Curr. Drug Targets Inflamm. Allergy 2004, 3, 361–375. [Google Scholar] [CrossRef]
- Dyall, S.C.; Balas, L.; Bazan, N.G.; Brenna, J.T.; Chiang, N.; Souza, F.d.C.; Dalli, J.; Durand, T.; Galano, J.-M.; Lein, P.J.; et al. Polyunsaturated fatty acids and fatty acid-derived lipid mediators: Recent advances in the understanding of their biosynthesis, structures, and functions. Prog. Lipid Res. 2022, 86, 101165. [Google Scholar] [CrossRef]
- Panigrahy, D.; Gilligan, M.M.; Serhan, C.N.; Kashfi, K. Resolution of inflammation: An organizing principle in biology and medicine. Pharmacol. Ther. 2021, 227, 107879. [Google Scholar] [CrossRef] [PubMed]
- Chiang, N.; Serhan, C.N. Specialized pro-resolving mediator network: An update on production and actions. Essays Biochem. 2020, 64, 443–462. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C.; Albers, R.; Antoine, J.-M.; Blum, S.; Bourdet-Sicard, R.; Ferns, G.A.; Folkerts, G.; Friedmann, P.S.; Frost, G.S.; Guarner, F.; et al. Inflammatory disease processes and interactions with nutrition. Br. J. Nutr. 2009, 101 (Suppl. S1), S1–S45. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C.; Ahluwalia, N.; Albers, R.; Bosco, N.; Bourdet-Sicard, R.; Haller, D.; Holgate, S.T.; Jönsson, L.S.; Latulippe, M.E.; Marcos, A.; et al. A consideration of biomarkers to be used for evaluation of inflammation in human nutritional studies. Br. J. Nutr. 2013, 109 (Suppl. S1), S1–S34. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C.; Bosco, N.; Bourdet-Sicard, R.; Capuron, L.; Delzenne, N.; Doré, J.; Franceschi, C.; Lehtinen, M.J.; Recker, T.; Salvioli, S.; et al. Health relevance of the modification of low grade inflammation in ageing (inflammageing) and the role of nutrition. Ageing Res. Rev. 2017, 40, 95–119. [Google Scholar] [CrossRef]
- Minihane, A.M.; Vinoy, S.; Russell, W.R.; Baka, A.; Roche, H.M.; Tuohy, K.M.; Teeling, J.L.; Blaak, E.E.; Fenech, M.; Vauzour, D.; et al. Low-grade inflammation, diet composition and health: Current research evidence and its translation. Br. J. Nutr. 2015, 114, 999–1012. [Google Scholar] [CrossRef] [PubMed]
- Meijerink, J.; Balvers, M.; Witkamp, R. N-acyl amines of docosahexaenoic acid and other n-3 polyunsatured fatty acids—From fishy endocannabinoids to potential leads. Br. J. Pharmacol. 2013, 169, 772–783. [Google Scholar] [CrossRef]
- Wainwright, C.L.; Michel, L. Endocannabinoid system as a potential mechanism for n-3 long-chain polyunsaturated fatty acid mediated cardiovascular protection. Proc. Nutr. Soc. 2013, 72, 460–469. [Google Scholar] [CrossRef]
- Dyall, S.C. Interplay between n-3 and n-6 long-chain polyunsaturated fatty acids and the endocannabinoid system in brain protection and repair. Lipids 2017, 52, 885–900. [Google Scholar] [CrossRef]
- Kim, H.Y.; Spector, A.A. N-docosahexaenoylethanolamine: A neurotrophic and neuroprotective metabolite of docosahexaenoic acid. Mol. Asp. Med. 2018, 64, 34–44. [Google Scholar] [CrossRef]
- de Bus, I.; Witkamp, R.; Zuilhof, H.; Albada, B.; Balvers, M. The role of n-3 PUFA-derived fatty acid derivatives and their oxygenated metabolites in the modulation of inflammation. Prostaglandins Other Lipid Mediat. 2019, 144, 106351. [Google Scholar] [CrossRef] [PubMed]
- Mallick, R.; Basak, S.; Duttaroy, A.K. Docosahexaenoic acid,22:6n-3: Its roles in the structure and function of the brain. Int. J. Dev. Neurosci. 2019, 79, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Docosahexaenoic acid. Ann. Nutr. Metab. 2016, 69 (Suppl. S1), S7–S21. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.I.; Atherton, P.; Reeds, D.N.; Mohammed, B.S.; Rankin, D.; Rennie, M.J.; Mittendorfer, B. Omega-3 polyunsaturated fatty acids augment the muscle protein anabolic response to hyperinsulinaemia-hyperaminoacidaemia in healthy young and middle-aged men and women. Clin. Sci. 2011, 121, 267–278. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Omega-3: The good oil. Nutr. Bull. 2017, 42, 132–140. [Google Scholar] [CrossRef]
- Aidos, I.; van der Padt, A.; Luten, J.B.; Boom, R.M. Seasonal changes in crude and lipid composition of herring fillets, byproducts, and respective produced oils. J. Agric. Food Chem. 2002, 50, 4589–4599. [Google Scholar] [CrossRef] [PubMed]
- Hooks, M.P.; Madigan, S.M.; Woodside, J.V.; Nugent, A.P. Dietary intake, biological status, and barriers towards omega-3 intake in elite level (tier 4), female athletes: Pilot study. Nutrients 2023, 15, 2821. [Google Scholar] [CrossRef]
- Bogard, J.R.; Farmery, A.K.; Little, D.C.; Fulton, E.A.; Cook, M. Will fish be part of future healthy and sustainable diets? Lancet Planet. Health 2019, 3, e159–e160. [Google Scholar] [CrossRef]
- Chang, M.; Zhang, T.; Guo, X.; Liu, Y.; Liu, R.; Jin, Q.; Wang, X. Optimization of cultivation conditions for efficient production of carotenoid-rich DHA oil by Schizochytrium sp. S31. Process Biochem. 2020, 94, 190–197. [Google Scholar] [CrossRef]
- Zeb, L.; Wang, X.D.; Zheng, W.L.; Teng, X.N.; Shafiq, M.; Mu, Y.; Chi, Z.Y.; Xiu, Z.L. Microwave-assisted three-liquid-phase salting-out extraction of docosahexaenoic acid (DHA)-rich oil from cultivation broths of Schizochytrium limacinium SR21. Food Bioprod. Process. 2019, 118, 237–247. [Google Scholar] [CrossRef]
- European Food Safety Authority. Scientific opinion on dietary reference values for fats, including saturated fatty acids, polyunsaturated fatty acids, monounsaturated fatty acids, trans fatty acids and cholesterol. EFSA J. 2010, 8, 1461. [Google Scholar]
- Food and Agriculture Organisation of the United Nations. Fats and Fatty Acids in Human Nutrition: Report of an Expert Consultation; FAO: Rome, Italy, 2010. [Google Scholar]
- US Department of Agriculture. Dietary Guidelines for Americans, 2015–2020. Available online: https://health.gov/our-work/food-nutrition/previous-dietary-guidelines/2015 (accessed on 1 August 2023).
- Vannice, G.; Rasmussen, H. Position of the academy of nutrition and dietetics: Dietary fatty acids for healthy adults. J. Acad. Nutr. Diet. 2014, 114, 136–153. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P.; Leaf, A.; Salem, N., Jr. Workshop statement on the essentiality of and recommended dietary intakes for Omega-6 and Omega-3 fatty acids. Prostagland Leukot Essent Fatty Acids 2000, 63, 119–121. [Google Scholar] [CrossRef] [PubMed]
- Jackson, K.H.; Polreis, J.M.; Tintle, N.L.; Kris-Etherton, P.M.; Harris, W.S. Association of reported fish intake and supplementation status with the omega-3 index. Prostaglandins Leukot. Essent. Fat. Acids 2019, 142, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Block, R.C.; Harris, W.S.; Pottala, J.V. Determinants of blood cell omega-3 fatty acid content. Open Biomark. J. 2008, 1, 1–6. [Google Scholar] [CrossRef]
- Dempsey, M.; Rockwell, M.S.; Wentz, L.M. The influence of dietary and supplemental omega-3 fatty acids on the omega-3 index: A scoping review. Front. Nutr. 2023, 10, 1072653. [Google Scholar] [CrossRef]
- Maughan, R.J.; Burke, L.M.; Dvorak, J.; Larson-Meyer, D.E.; Peeling, P.; Phillips, S.M.; Rawson, E.S.; Walsh, N.P.; Garthe, I.; Geyer, H.; et al. IOC consensus statement: Dietary supplements and the high-performance athlete. Br. J. Sports Med. 2018, 52, 439–455. [Google Scholar] [CrossRef]
- Black, K.E.; Witard, O.C.; Baker, D.; Healey, P.; Lewis, V.; Tavares, F.; Christensen, S.; Pease, T.; Smith, B. Adding omega-3 fatty acids to a protein-based supplement during pre-season training results in reduced muscle soreness and the better maintenance of explosive power in professional Rugby Union players. Eur. J. Sport Sci. 2018, 18, 1357–1367. [Google Scholar] [CrossRef]
- Theadom, A.; Mahon, S.; Hume, P.; Starkey, N.; Barker-Collo, S.; Jones, K.; Majdan, M.; Feigin, V.L. Incidence of sports-related traumatic brain injury of all severities: A systematic review. Neuroepidemiology 2020, 54, 1516–1525. [Google Scholar] [CrossRef]
- Lemme, N.J.; Johnston, B.; DeFroda, S.F.; Owens, B.D.; Kriz, P.K. Incidence of combat sport-related mild traumatic brain injuries presenting to the emergency department from 2012 to 2016. Clin. J. Sport Med. Off. J. Can. Acad. Sport Med. 2020, 30, 585–590. [Google Scholar] [CrossRef]
- Langlois, J.A.; Rutland-Brown, W.; Wald, M.M. The epidemiology and impact of traumatic brain injury: A brief overview. J. Head Trauma Rehabil. 2006, 21, 375–378. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Ding, Y.; Kong, W.; Li, T.; Chen, H. Docosahexaenoic acid (DHA) provides neuroprotection in traumatic brain injury models via activating Nrf2-ARE signaling. Inflammation 2018, 41, 1182–1193. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Cui, G.; Li, T.; Chen, H.; Zhu, J.; Ding, Y.; Zhao, L. Docosahexaenoic acid protects traumatic brain injury by regulating NOX2 generation via Nrf2 signaling pathway. Neurochem. Res. 2020, 45, 1839–1850. [Google Scholar] [CrossRef] [PubMed]
- Lust, C.A.; Mountjoy, M.; Robinson, L.E.; Oliver, J.M.; Ma, D.W. Sports-related concussions and subconcussive impacts in athletes: Incidence, diagnosis, and the emerging role of EPA and DHA. Appl. Physiol. Nutr. Metab. 2020, 45, s886–s892. [Google Scholar] [CrossRef]
- Patch, C.S.; Hill-Yardin, E.L.; Lewis, M.; Ryan, L.; Daly, E.; Pearce, A.J. The more, the better: High-dose omega-3 fatty acids improve behavioural and molecular outcomes in preclinical models in mild brain injury. Curr. Neurol. Neurosci. Rep. 2021, 21, 45. [Google Scholar] [CrossRef] [PubMed]
- Heileson, J.L.; Anzalone, A.J.; Carbuhn, A.F.; Askow, A.T.; Stone, J.D.; Turner, S.M.; Hillyer, L.M.; Ma, D.W.L.; Luedke, J.A.; Jagim, A.R.; et al. The effect of omega-3 fatty acids on a biomarker of head trauma in NCAA football athletes: A multi-site, non-randomized study. J. Int. Soc. Sports Nutr. 2021, 18, 65. [Google Scholar] [CrossRef]
- Mullins, V.A.; Graham, S.; Cummings, D.; Wood, A.; Ovando, V.; Skulas-Ray, A.C.; Polian, D.; Wang, Y.; Hernandez, G.D.; Lopez, C.M.; et al. Effects of fish oil on biomarkers of axonal injury and inflammation in American football players: A placebo-controlled randomized controlled trial. Nutrients 2022, 14, 2139. [Google Scholar] [CrossRef]
- Raikes, A.C.; Hernandez, G.D.; Mullins, V.A.; Wang, Y.; Lopez, C.; Killgore WD, S.; Chilton, F.H.; Brinton, R.D. Effects of docosahexaenoic acid and eicosapentaoic acid supplementation on white matter integrity after repetitive sub-concussive head impacts during American football: Exploratory neuroimaging findings from a pilot RCT. Front. Neurol. 2022, 13, 891531. [Google Scholar] [CrossRef]
- Miller, S.M.; Zynda, A.J.; Sabatino, M.J.; Jo, C.; Ellis, H.B.; Dimeff, R.J. A pilot randomized controlled trial of docosahexaenoic acid for the treatment of sport-related concussion in adolescents. Clin. Pediatr. 2022, 61, 785–794. [Google Scholar] [CrossRef]
- Bostock, E.L.; Morse, C.I.; Winwood, K.; McEwan, I.M.; Onambélé-Pearson, G.L. Omega-3 fatty acids and vitamin D in immobilisation: Part A—Modulation of appendicular mass content, composition and structure. J. Nutr. Health Aging 2017, 21, 51–58. [Google Scholar] [CrossRef]
- Owens, D.J.; Twist, C.; Cobley, J.N.; Howatson, G.; Close, G.L. Exercise-induced muscle damage: What is it, what causes it and what are the nutritional solutions? Eur. J. Sport Sci. 2019, 19, 71–85. [Google Scholar] [CrossRef]
- Heileson, J.L.; Funderburk, L.L.K. The effect of fish oil supplementation on the promotion and preservation of lean body mass, strength, and recovery from physiological stress in young, healthy adults: A systematic review. Nutr. Rev. 2020, 78, 1001–1014. [Google Scholar] [CrossRef] [PubMed]
- Harty, P.S.; Cottet, M.L.; Malloy, J.K.; Kerksick, C.M. Nutritional and supplementation strategies to prevent and attenuate exercise-induced muscle damage: A brief review. Sports Med. Open 2019, 5, 1. [Google Scholar] [CrossRef] [PubMed]
- Tartibian, B.; Maleki, B.H.; Abbasi, A. The effects of ingestion of omega-3 fatty acids on perceived pain and external symptoms of delayed onset muscle soreness in untrained men. Clin. J. Sport Med. 2009, 19, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Campo, D.J.; Ávila-Gandía, V.; López-Román, F.J.; Miñarro, J.; Contreras, C.; Soto-Méndez, F.; Pedrol JC, D.; Luque-Rubia, A.J. Supplementation of re-esterified docosahexaenoic and eicosapentaenoic acids reduce inflammatory and muscle damage markers after exercise in endurance athletes: A randomized, controlled crossover trial. Nutrients 2020, 12, 719. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, Y.; Yanagimoto, K.; Ueda, H.; Ochi, E. Supplementation of eicosapentaenoic acid-rich fish oil attenuates muscle stiffness after eccentric contractions of human elbow flexors. J. Int. Soc. Sports Nutr. 2019, 16, 19. [Google Scholar] [CrossRef]
- Corder, K.E.; Newsham, K.R.; McDaniel, J.L.; Ezekiel, U.R.; Weiss, E.P. Effects of short-term docosahexaenoic acid supplementation on markers of inflammation after eccentric strength exercise in women. J. Sports Sci. Med. 2016, 15, 176–183. [Google Scholar]
- Jouris, K.B.; McDaniel, J.L.; Weiss, E.P. The effect of omega-3 fatty acid supplementation on the inflammatory response to eccentric strength exercise. J. Sports Sci. Med. 2011, 10, 432–438. [Google Scholar] [CrossRef][Green Version]
- Lembke, P.; Capodice, J.; Hebert, K.; Swenson, T. Influence of omega-3 (N3) index on performance and wellbeing in young adults after heavy eccentric exercise. J. Sports Sci. Med. 2014, 13, 151–156. [Google Scholar]
- van Dusseldorp, T.A.; Escobar, K.A.; Johnson, K.E.; Stratton, M.T.; Moriarty, T.; Kerksick, C.M.; Mangine, G.T.; Holmes, A.J.; Lee, M.; Endito, M.R.; et al. Impact of varying dosages of fish oil on recovery and soreness following eccentric exercise. Nutrients 2020, 12, 2246. [Google Scholar] [CrossRef]
- Philpott, J.D.; Donnelly, C.; Walshe, I.H.; MacKinley, E.E.; Dick, J.; Galloway SD, R.; Tipton, K.D.; Witard, O.C. Adding fish oil to whey protein, leucine, and carbohydrate over a six-week supplementation period attenuates muscle soreness following eccentric exercise in competitive soccer players. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Kyriakidou, Y.; Wood, C.; Ferrier, C.; Dolci, A.; Elliott, B. The effect of omega-3 polyunsaturated fatty acid supplementation on exercise-induced muscle damage. J. Int. Soc. Sports Nutr. 2021, 18, 9. [Google Scholar] [CrossRef] [PubMed]
- Anthony, R.; MaCartney, M.J.; Heileson, J.L.; McLennan, P.L.; Peoples, G.E. A review and evaluation of study design considerations for omega-3 fatty acid supplementation trials in physically trained participants. Nutr. Res. Rev. 2023; in press. [Google Scholar] [CrossRef] [PubMed]
- Philpott, J.D.; Bootsma, N.J.; Rodriguez-Sanchez, N.; Hamilton, D.L.; MacKinlay, E.; Dick, J.; Mettler, S.; Galloway, S.D.R.; Tipton, K.D.; Witard, O.C. Influence of fish oil-derived n-3 fatty acid supplementation on changes in body composition and muscle strength during short-term weight loss in resistance-trained men. Front. Nutr. 2019, 6, 102. [Google Scholar] [CrossRef] [PubMed]
- Hingley, L.; Macartney, M.J.; Brown, M.A.; McLennan, P.L.; Peoples, G.E. DHA-rich fish oil increases the omega-3 index and lowers the oxygen cost of physiologically stressful cycling in trained individuals. Int. J. Sport Nutr. Exerc. Metab. 2017, 27, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Raastad, T.; Hostmark, A.T.; Stramme, S.B. Omega-3 fatty acid supplementation does not improve maximal aerobic power, anaerobic threshold and running performance in well-trained soccer players. Scand. J. Med. Sci. Sports 1997, 7, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Żebrowska, A.; Mizia-Stec, K.; Mizia, M.; Gąsior, Z.; Poprzęcki, S. Omega-3 fatty acids supplementation improves endothelial function and maximal oxygen uptake in endurance-trained athletes. Eur. J. Sport Sci. 2015, 15, 305–314. [Google Scholar] [CrossRef]
- Thielecke, F.; Blannin, A. Omega-3 fatty acids for sport performance—Are they equally beneficial for athletes and amateurs? A narrative review. Nutrients 2020, 12, 3712. [Google Scholar] [CrossRef]
- Browning, L.M.; Walker, C.G.; Mander, A.P.; West, A.L.; Madden, J.; Gambell, J.M.; Young, S.; Wang, L.; Jebb, S.A.; Calder, P.C. Incorporation of eicosapentaenoic and docosahexaenoic acids into lipid pools when given as supplements providing doses equivalent to typical intakes of oily fish. Am. J. Clin. Nutr. 2012, 96, 748–758. [Google Scholar] [CrossRef]
- Villani, A.M.; Crotty, M.; Cleland, L.G.; James, M.J.; Fraser, R.J.; Cobiac, L.; Miller, M.D. Fish oil administration in older adults: Is there potential for adverse events? A systematic review of the literature. BMC Geriatr. 2013, 13, 41. [Google Scholar] [CrossRef]
- Chang, C.H.; Tseng, P.T.; Chen, N.Y.; Lin, P.C.; Lin, P.Y.; Chang JP, C.; Kuo, F.Y.; Lin, J.; Wu, M.C.; Su, K.P. Safety and tolerability of prescription omega-3 fatty acids: A systematic review and meta-analysis of randomized controlled trials. Prostaglandins Leukot. Essent. Fat. Acids 2018, 129, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Dyerberg, J.; Bang, H.O. Haemostatic function and platelet polyunsaturated fatty acids in Eskimos. Lancet 1979, 8140, 433–435. [Google Scholar] [CrossRef] [PubMed]
- Bang, H.O.; Dyerberg, J.; Hjørne, N. The composition of food consumed by Greenland Eskimos. Acta Medica Scand. 1976, 200, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Wachira, J.K.; Larson, M.K.; Harris, W.S. n-3 fatty acids affect haemostasis but do not increase the risk of bleeding: Clinical observations and mechanistic insights. Br. J. Nutr. 2014, 111, 1652–1662. [Google Scholar] [CrossRef]
- Jeansen, S.; Witkamp, R.F.; Garthoff, J.A.; van Helvoort, A.; Calder, P.C. Fish oil LC-PUFAs do not affect blood coagulation parameters and bleeding manifestations: Analysis of 8 clinical studies with selected patient groups on omega-3-enriched medical nutrition. Clin. Nutr. 2018, 37, 948–957. [Google Scholar] [CrossRef]
- Akintoye, E.; Sethi, P.; Harris, W.S.; Thompson, P.A.; Marchioli, R.; Tavazzi, L.; Latini, R.; Pretorius, M.; Brown, N.J.; Libby, P.; et al. Fish oil and perioperative bleeding. Circ. Cardiovasc. Qual. Outcomes 2018, 11, e004584. [Google Scholar] [CrossRef]
- Jairoun, A.A.; Shahwan, M.; Zyoud, S.H. Fish oil supplements, oxidative status, and compliance behaviour: Regulatory challenges and opportunities. PLoS ONE 2020, 15, e0244688. [Google Scholar] [CrossRef]
- EFSA. Scientific opinion on the tolerable upper intake level of eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA) and docosapentaenoic acid (DPA). EFSA J. 2012, 10, 2815. [Google Scholar] [CrossRef]
- Lien, E.L. Toxicology and safety of DHA. Prostaglandins Leukot. Essent. Fat. Acids 2009, 81, 125–132. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. Qualified Health Claims: Letters of Enforcement Discretion; U.S. Food and Drug Administration: Washington, DC, USA, 2019.
- Siscovick, D.S.; Barringer, T.A.; Fretts, A.M.; Wu, J.H.Y.; Lichtenstein, A.H.; Costello, R.B.; Kris-Etherton, P.M.; Jacobson, T.A.; Engler, M.B.; Alger, H.M.; et al. Omega-3 polyunsaturated fatty acid (fish oil) supplementation and the prevention of clinical cardiovascular disease: A Science Advisory from the American Heart Association. Circulation 2017, 135, e864–e884. [Google Scholar] [CrossRef]
- Mozaffarian, D.; Rimm, E.B. Fish intake, contaminants, and human health evaluating the risks and the benefits. JAMA 2006, 296, 1885–1899. [Google Scholar] [CrossRef] [PubMed]
- von Schacky, C.; Fischer, S.; Weber, P.C. Long-term effects of dietary marine ω-3 fatty acids upon plasma and cellular lipids, platelet function, and eicosanoid formation in humans. J. Clin. Investig. 1985, 76, 1626–1631. [Google Scholar] [CrossRef] [PubMed]
- Blonk, M.C.; Bilo, H.J.G.; Nauta, J.J.P.; Popp-Snijders, C.; Mulder, C.; Donker, A.J.M. Dose-response effects of fish-oil supplementation in healthy volunteers. Am. J. Clin. Nutr. 1990, 52, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S.; Windsor, S.L.; Dujovne, C.A. Effects of four doses of n-3 fatty acids given to hyperlipidemic patients for six months. J. Am. Coll. Nutr. 1991, 10, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Rees, D.; Miles, E.A.; Banerjee, T.; Wells, S.J.; Roynette, C.E.; Wahle, K.W.J.; Calder, P.C. Dose-related effects of eicosapentaenoic acid on innate immune function in healthy humans: A comparison of young and older men. Am. J. Clin. Nutr. 2006, 83, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Faber, J.; Berkhout, M.; Vos, A.P.; Sijben, J.W.C.; Calder, P.C.; Garssen, J.; van Helvoort, A. Supplementation with a fish oil-enriched, high-protein medical food leads to rapid incorporation of EPA into white blood cells and modulates immune responses within one week in healthy men and women. J. Nutr. 2011, 141, 964–970. [Google Scholar] [CrossRef] [PubMed]
- Yaqoob, P.; Pala, H.S.; Cortina-Borja, M.; Newsholme, E.A.; Calder, P.C. Encapsulated fish oil enriched in α-tocopherol alters plasma phospholipid and mononuclear cell fatty acid compositions but not mononuclear cell functions. Eur. J. Clin. Investig. 2000, 30, 260–274. [Google Scholar] [CrossRef]
- Katan, M.B.; Deslypere, J.P.; van Birgelen, A.P.J.M.; Penders, M.; Zegwaard, M. Kinetics of the incorporation of dietary fatty acids into serum cholesteryl esters, erythrocyte membranes, and adipose tissue: An 18-month controlled study. J. Lipid Res. 1997, 38, 2012–2022. [Google Scholar] [CrossRef]
- Marsen, T.A.; Pollok, M.; Oette, K.; Baldamus, C.A. Pharmacokinetics of omega-3-fatty acids during ingestion of fish oil preparations. Prostaglandins Leukot. Essent. Fat. Acids 1992, 46, 191–196. [Google Scholar] [CrossRef]
- McGlory, C.; Galloway, S.D.R.; Hamilton, D.L.; McClintock, C.; Breen, L.; Dick, J.R.; Bell, J.G.; Tipton, K.D. Temporal changes in human skeletal muscle and blood lipid composition with fish oil supplementation. Prostaglandins Leukot. Essent. Fat. Acids 2014, 90, 199–206. [Google Scholar] [CrossRef]
- West, A.L.; Burdge, G.C.; Calder, P.C. Lipid structure does not modify incorporation of EPA and DHA into blood lipids in healthy adults: A randomised-controlled trial. Br. J. Nutr. 2016, 116, 788–797. [Google Scholar] [CrossRef] [PubMed]
- El-Boustani, S.; Colette, C.; Monnier, L.; Descomps, B.; de Paulet, A.C.; Mendy, F. Enteral absorption in man of eicosapentaenoic acid in different chemical forms. Lipids 1987, 22, 711–714. [Google Scholar] [CrossRef] [PubMed]
- Schuchardt, J.P.; Neubronner, J.; Kressel, G.; Merkel, M.; von Schacky, C.; Hahn, A. Moderate doses of EPA and DHA from re-esterified triacylglycerols but not from ethyl-esters lower fasting serum triacylglycerols in statin-treated dyslipidemic subjects: Results from a six month randomized controlled trial. Prostaglandins Leukot. Essent. Fat. Acids 2011, 85, 381–386. [Google Scholar] [CrossRef] [PubMed]
- Davidson, M.H.; Johnson, J.; Rooney, M.W.; Kyle, M.L.; Kling, D.F. A novel omega-3 free fatty acid formulation has dramatically improved bioavailability during a low-fat diet compared with omega-3-acid ethyl esters: The ECLIPSE (Epanova® compared to Lovaza® in a pharmacokinetic single-dose evaluation) study. J. Clin. Lipidol. 2012, 6, 573–584. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Nyheim, H.; Haram, E.M.; Moritz, J.M.; Hustvedt, S.O. A novel self-micro-emulsifying delivery system (SMEDS) formulation significantly improves the fasting absorption of EPA and DHA from a single dose of an omega-3 ethyl ester concentrate. Lipids Health Dis. 2017, 16, 204. [Google Scholar] [CrossRef] [PubMed]
- West, A.L.; Kindberg, G.M.; Hustvedt, S.O.; Calder, P.C. A novel self-micro-emulsifying delivery system enhances enrichment of eicosapentaenoic acid and docosahexaenoic acid after single and repeated dosing in healthy adults in a randomized trial. J. Nutr. 2018, 148, 1704–1715. [Google Scholar] [CrossRef]
- Fisk, H.L.; Kindberg, G.M.; Hustvedt, S.O.; Calder, P.C. A novel n-3 glyceride mixture enhances enrichment of EPA and DHA after single dosing in healthy older adults: Results from a double-blind crossover trial. Br. J. Nutr. 2021, 126, 244–252. [Google Scholar] [CrossRef]
- Kagan, M.L.; West, A.L.; Zante, C.; Calder, P.C. Acute appearance of fatty acids in human plasma—A comparative study between polar-lipid rich oil from the microalgae Nannochloropsis oculata and krill oil in healthy young males. Lipids Health Dis. 2013, 12, 102. [Google Scholar] [CrossRef]
- Schuchardt, J.P.; Hahn, A. Bioavailability of long-chain omega-3 fatty acids. Prostaglandins Leukot. Essent. Fat. Acids 2013, 89, 18. [Google Scholar] [CrossRef]
- Innes, J.K.; Calder, P.C. The differential effects of eicosapentaenoic acid and docosahexaenoic acid on cardiometabolic risk factors: A systematic review. Int. J. Mol. Sci. 2018, 19, 532. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomczyk, M.; Heileson, J.L.; Babiarz, M.; Calder, P.C. Athletes Can Benefit from Increased Intake of EPA and DHA—Evaluating the Evidence. Nutrients 2023, 15, 4925. https://doi.org/10.3390/nu15234925
Tomczyk M, Heileson JL, Babiarz M, Calder PC. Athletes Can Benefit from Increased Intake of EPA and DHA—Evaluating the Evidence. Nutrients. 2023; 15(23):4925. https://doi.org/10.3390/nu15234925
Chicago/Turabian StyleTomczyk, Maja, Jeffery L. Heileson, Mirosław Babiarz, and Philip C. Calder. 2023. "Athletes Can Benefit from Increased Intake of EPA and DHA—Evaluating the Evidence" Nutrients 15, no. 23: 4925. https://doi.org/10.3390/nu15234925
APA StyleTomczyk, M., Heileson, J. L., Babiarz, M., & Calder, P. C. (2023). Athletes Can Benefit from Increased Intake of EPA and DHA—Evaluating the Evidence. Nutrients, 15(23), 4925. https://doi.org/10.3390/nu15234925






