Lipid Profile in Children Born Small for Gestational Age
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hong, Y.H.; Chung, S. Small for gestational age and obesity related comorbidities. Ann. Pediatr. Endocrinol. Metab. 2018, 23, 4–8. [Google Scholar] [CrossRef]
- Barker, D.J. The fetal and infant origins of adult disease. Br. Med. J. 1990, 301, 1111. [Google Scholar] [CrossRef]
- Thomas, P.; Peabody, J.; Turnier, V.; Clark, R.H. A new look at intrauterine growth and the impact of race, altitude, and gender. Pediatrics 2000, 106, E21. [Google Scholar] [CrossRef]
- World Health Organization. International Statistical Classification of Diseases and Related Health Problems, 10th revision, 2nd ed.; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Figueras, F.; Eixarch, E.; Gratacos, E.; Gardosi, J. Predictiveness of antenatal umbilical artery Doppler for adverse pregnancy outcome in small-for-gestational-age babies according to customised birthweight centiles: Population-based study. BJOG 2008, 115, 590–594. [Google Scholar] [CrossRef]
- Alisi, A.; Panera, N.; Agostoni, C.; Nobili, V. Intrauterine growth retardation and nonalcoholic Fatty liver disease in children. Int. J. Endocrinol. 2011, 2011, 269853. [Google Scholar] [CrossRef] [PubMed]
- Hofman, P.L.; Cutfield, W.S.; Robinson, E.M.; Bergman, R.N.; Menon, R.K.; Sperling, M.A.; Gluckman, P.D. Insulin resistance in short children with intrauterine growth retardation. J. Clin. Endocrinol. Metab. 1997, 82, 402–406. [Google Scholar] [CrossRef] [PubMed]
- Desta, M.; Tadese, M.; Kassie, B.; Gedefaw, M. Determinants and adverse perinatal outcomes of low birth weight newborns delivered in Hawassa University Comprehensive Specialized Hospital, Ethiopia: A cohort study. BMC Res. Notes 2019, 12, 118. [Google Scholar] [CrossRef]
- Lubchenco, L.O.; Hansman, C.; Boyd, E. Intrauterine growth in length and head circumference as estimated from live births at gestational ages from 26 to 42 weeks. Pediatrics 1966, 37, 403–408. [Google Scholar] [CrossRef]
- Suhag, A.; Berghella, V. Intrauterine growth restriction (IUGR): Etiology and diagnosis. Curr. Obstet. Gynecol. Rep. 2013, 2, 102–111. [Google Scholar] [CrossRef]
- Sharma, D.; Shastri, S.; Sharma, P. Intrauterine growth restriction: Antenatal and postnatal aspects. Clin. Med. Insights Pediatr. 2016, 10, 67–83. [Google Scholar] [CrossRef]
- Bakketeig, L.S. Current growth standards, definitions, diagnosis and classification of fetal growth retardation. Eur. J. Clin. Nutr. 1998, 52 (Suppl. S1), S1–S4. [Google Scholar] [PubMed]
- Hwang, J.K.; Kang, H.N.; Ahn, J.H.; Lee, H.J.; Park, H.K.; Kim, C.R. Effects of Ponderal Index on Neonatal Mortality and Morbidities in Extremely Premature Infants. J. Korean Med. Sci. 2022, 37, e198. [Google Scholar] [CrossRef] [PubMed]
- Mohseni, R.; Mohammed, S.H.; Safabakhsh, M.; Mohseni, F.; Monfared, Z.S.; Seyyedi, J.; Mejareh, Z.N.; Alizadeh, S. Birth Weight and Risk of Cardiovascular Disease Incidence in Adulthood: A Dose-Response Meta-analysis. Curr. Atheroscler. Rep. 2020, 22, 12. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Tang, Z.; Yu, X.; Gao, Y.; Guan, F.; Li, C.; Huang, S.; Zheng, J.; Zeng, P. Birth Weight and Stroke in Adult Life: Genetic Correlation and Causal Inference with Genome-Wide Association Data Sets. Front. Neurosci. 2020, 14, 479. [Google Scholar] [CrossRef] [PubMed]
- Zerbeto, A.B.; Cortelo, F.M.; CFilho, É.B. Association between gestational age and birth weight on the language development of Brazilian children: A systematic review. J. Pediatr. 2015, 91, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Cutland, C.L.; Lackritz, E.M.; Mallett-Moore, T.; Bardají, A.; Chandrasekaran, R.; Lahariya, C.; Nisar, M.I.; Tapia, M.D.; Pathirana, J.; Kochhar, S.; et al. Low birth weight: Case definition & guidelines for data collection, analysis, and presentation of maternal immunization safety data. Vaccine 2017, 35, 6492–6500. [Google Scholar] [CrossRef]
- Nordman, H.; Jääskeläinen, J.; Voutilainen, R. Birth Size as a Determinant of Cardiometabolic Risk Factors in Children. Horm. Res. Paediatr. 2020, 93, 144–153. [Google Scholar] [CrossRef]
- Lurbe, E.; Aguilar, F.; Álvarez, J.; Redon, P.; Torró, M.I.; Redon, J. Determinants of Cardiometabolic Risk Factors in the First Decade of Life: A Longitudinal Study Starting at Birth. Hypertension 2018, 71, 437–443. [Google Scholar] [CrossRef]
- Mullett, M.D.; Cottrell, L.; Lilly, C.; Gadikota, K.; Dong, L.; Hobbs, G.; Neal, W.A. Association between birth characteristics and coronary disease risk factors among fifth graders. J. Pediatr. 2014, 164, 78–82. [Google Scholar] [CrossRef]
- Tenhola, S.; Martikainen, A.; Rahiala, E.; Herrgârd, E.; Halonen, P.; Voutilainen, R. Serum lipid concentrations and growth characteristics in 12-year-old children born small for gestational age. Pediatr. Res. 2000, 48, 623–628. [Google Scholar] [CrossRef]
- Barker, D.J.; Martyn, C.N.; Osmond, C.; Hales, C.N.; Fall, C.H. Growth in utero and serum cholesterol concentrations in adult life. BMJ 1993, 307, 1524–1527. [Google Scholar] [CrossRef] [PubMed]
- Donker, G.A.; Labarthe, D.R.; Harrist, R.B.; Selwyn, B.J.; Srinivasan, S.R.; Wattigney, W.; Berenson, G.S. Low birth weight and serum lipid concentrations at age 7–11 years in a biracial sample. Am. J. Epidemiol. 1997, 145, 398–407. [Google Scholar] [CrossRef] [PubMed]
- Antal, M.; Agfalvi, R.; Nagy, K.; Szepvölgyi, J.; Banto, E.; Regöly-Merei, A.; Biro, L.; Biro, G. Lipid status in adolescents born with low birth weight. Z. Ernahrungswiss. 1998, 37 (Suppl. S1), 131–133. [Google Scholar] [PubMed]
- Ibanez, L.; Potau, N.; de Zegher, F. Precocious pubarche, dyslipidemia, and low IGF binding protein-1 in girls: Relation to reduced prenatal growth. Pediatr. Res. 1999, 46, 320–322. [Google Scholar] [CrossRef]
- Okosun, I.S.; Dever, G.E.; Choi, S.T. Low birth weight is associated with elevated serum lipoprotein(a) in white and black American children ages 5-11 y. Public. Health 2002, 116, 33–38. [Google Scholar] [CrossRef]
- Krochik, A.G.; Chaler, E.A.; Maceiras, M.; Aspres, N.; Mazza, C.S. Presence of early risk markers of metabolic syndrome in prepubertal children with a history of intrauterine growth restriction. Arch. Argent. Pediatr. 2010, 108, 10–16. [Google Scholar]
- Umer, A.; Hamilton, C.; Cottrell, L.; Giacobbi, P.; Innes, K.; Kelley, G.A.; Neal, W.; John, C.; Lilly, C. Association between birth weight and childhood cardiovascular disease risk factors in West Virginia. J. Dev. Orig. Health Dis. 2020, 11, 86–95. [Google Scholar] [CrossRef]
- Starnberg, J.; Norman, M.; Westrup, B.; Domellöf, M.; Berglund, S.K. Cardiometabolic risk factors in children born with marginally low birth weight: A longitudinal cohort study up to 7 years-of-age. PLoS ONE 2019, 14, e0215866. [Google Scholar] [CrossRef]
- Boguszewski, M.C.; Mericq, V.; Bergada, I.; Damiani, D.; Belgorosky, A.; Gunczler, P.; Ortiz, T.; Llano, M.; Domené, H.M.; Calzada-León, R.; et al. Latin American consensus: Children born small for gestational age. BMC Pediatr. 2011, 11, 66. [Google Scholar] [CrossRef]
- Finken, M.J.; van der Steen, M.; Smeets, C.C.; Walenkamp, M.J.; de Bruin, C.; Hokken-Koelega, A.C.; Wit, J.M. Children Born Small for Gestational Age: Differential Diagnosis, Molecular Genetic Evaluation, and Implications. Endocr. Rev. 2018, 39, 851–894. [Google Scholar] [CrossRef]
- Goedegebuure, W.J.; Van der Steen, M.; Smeets, C.C.J.; Kerkhof, G.F.; Hokken-Koelega, A.C.S. SGA-born adults with postnatal catch-up have a persistently unfavourable metabolic health profile and increased adiposity at age 32 years. Eur. J. Endocrinol. 2022, 187, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Yang, X.; Zhao, Y.; Luo, X.P.; Liang, Y.; Wu, W.; Ying, Y.Q.; Ye, F.; Hou, L. A comparison of the growth status, level of blood glucose, and lipid metabolism in small for gestational age and appropriate for gestational age girls with central precocious puberty: A retrospective study. Transl. Pediatr. 2021, 10, 783–789. [Google Scholar] [CrossRef] [PubMed]
- Dunger, D.; Darendeliler, F.; Kandemir, N.; Harris, M.; Rabbani, A.; Kappelgaard, A.M. What is the evidence for beneficial effects of growth hormone treatment beyond height in short children born small for gestational age? A review of published literature. J. Pediatr. Endocrinol. Metab. 2020, 33, 53–70. [Google Scholar] [CrossRef]
- Porta, M. (Ed.) A Dictionary of Epidemiology, 6th ed.; Oxford University Press: New York, NY, USA, 2014; pp. 33–225. [Google Scholar]
- Liu, K.; Daviglus, M.L.; Loria, C.M.; Colangelo, L.A.; Spring, B.; Moller, A.C.; Lloyd-Jones, D.M. Healthy lifestyle through young adulthood and the presence of low cardiovascular disease risk profile in middle age: The Coronary Artery Risk Development in (Young) Adults (CARDIA) study. Circulation 2012, 125, 996–1004. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.N.; Abreu, G.R.; Resende, R.S.; Goncalves, W.L.; Gouvea, S.A. Cardiovascular risk factor investigation: A pediatric issue. Int. J. Gen. Med. 2013, 6, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Zouridis, A.; Manousopoulou, A.; Potiris, A.; Sarli, P.M.; Aravantinos, L.; Pervanidou, P.; Deligeoroglou, E.; Garbis, S.D.; Eleftheriades, M. Impact of Maternal Food Restriction on Heart Proteome in Appropriately Grown and Growth-Restricted Wistar-Rat Offspring. Nutrients 2021, 13, 466. [Google Scholar] [CrossRef]
- Howe, L.D.; Tilling, K.; Benfield, L.; Logue, J.; Sattar, N.; Ness, A.R.; Smith, G.D.; Lawlor, D.A. Changes in ponderal index and body mass index across childhood and their associations with fat mass and cardiovascular risk factors at age 15. PLoS ONE 2010, 5, e15186. [Google Scholar] [CrossRef] [PubMed]
- Oostvogels, A.J.; Stronks, K.; Roseboom, T.J.; van der Post, J.A.; van Eijsden, M.; Vrijkotte, T.G. Maternal prepregnancy BMI, offspring’s early postnatal growth, and metabolic profile at age 5-6 years: The ABCD Study. J. Clin. Endocrinol. Metab. 2014, 99, 3845–3854. [Google Scholar] [CrossRef]
- Koklu, E.; Kurtoglu, S.; Akcakus, M.; Koklu, S.; Buyukkayhan, D.; Gumus, H.; Yikilmaz, A. Increased aortic intima-media thickness is related to lipid profile in newborns with intrauterine growth restriction. Horm. Res. 2006, 65, 269–275. [Google Scholar] [CrossRef]
- Guo, H.J.; Li, C.C.; Bian, X.Y.; Hao, Q. Correlation study on the relationship between dyslipidemia and carotid intima-media thickness in patients with diabetes mellitus. Pak. J. Med. Sci. 2023, 39, 875–879. [Google Scholar] [CrossRef]
- Molina, M.; Casanueva, V.; Cid, X.; Ferrada, M.C.; Pérez, R.; Dios, G.; Reyes, M.; Venegas, H.; Cid, L. Lipid profile in newborns with intrauterine growth retardation. Rev. Med. Chil. 2000, 128, 741–748. [Google Scholar] [PubMed]
- Zheng, R.D.; Wang, W.J.; Ying, Y.Q.; Luo, X.P. Effects of intrauterine growth retardation with catch-up growth on sugar-lipid metabolism and adipocyte function in young rats. Chin. J. Contemp. Pediatr. 2015, 17, 1124–1130. [Google Scholar]
- Huang, Y.; Li, Y.; Chen, Q.; Chen, H.; Ma, H.; Su, Z.; Du, M. Low serum adiponectin levels are associated with reduced insulin sensitivity and lipid disturbances in short children born small for gestational age. Clin. Endocrinol. 2015, 83, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Evagelidou, E.N.; Giapros, V.I.; Challa, A.S.; Kiortsis, D.N.; Tsatsoulis, A.A.; Andronikou, S.K. Serum adiponectin levels, insulin resistance, and lipid profile in children born small for gestational age are affected by the severity of growth retardation at birth. Eur. J. Endocrinol. 2007, 156, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Chiavaroli, V.; Marcovecchio, M.L.; de Giorgis, T.; Diesse, L.; Chiarelli, F.; Mohn, A. Progression of cardio-metabolic risk factors in subjects born small to karge for gestational age. PLoS ONE 2014, 9, e104278. [Google Scholar] [CrossRef]
- Deng, H.Z.; Deng, H.; Su, Z.; Li, Y.H.; Ma, H.M.; Chen, H.S.; Du, M.L. Insulin resistance and adiponectin levels are associated with height catch-up growth in pre-pubertal Chinese individuals born small for gestational age. Nutr. Metab. 2012, 9, 107. [Google Scholar] [CrossRef]
| SGA Group (N = 93) Mean ± SD | Control Group (N = 47) Mean ± SD | p Value | |
|---|---|---|---|
| Sex (Male/Female) | 43/50 | 23/24 | 0.76 (NS) |
| Age on examination | 7.53 ± 1.35 | 7.4 ± 1.77 | 0.67 (NS) |
| Birth weight (g) | 2545.22 ± 209.07 | 3370.29 ± 503.11 | p < 0.001 |
| Birth height (cm) | 51.43 ± 2.61 | 54.51 ± 3.20 | p < 0.001 |
| Gestational age (hbd) | 38.98 ± 0.86 | 39.09 ± 0.74 | 0.52 (NS) |
| Actual height (cm) | 123.78 ± 10.06 | 128.11 ± 10.18 | p < 0.05 |
| Actual weight (kg) | 24.79 ± 8.54 | 26.53 ± 7.78 | 0.30 (NS) |
| BMI | 15.80 ± 2.83 | 15.97 ± 2.43 | 0.76 (NS) |
| SGA Symmetrical (N = 43) Mean ± SD | SGA Asymmetrical (N = 50) Mean ± SD | p Value | |
|---|---|---|---|
| Sex (Male/Female) | 22/21 | 21/29 | 0.38 (NS) |
| Age on examination | 7.72 ± 1.15 | 7.79 ± 1.46 | 0.16 (NS) |
| Birth weight (g) | 2506.74 ± 256.66 | 2578.30 ± 152.28 | 0.51 (NS) |
| Birth height (cm) | 49.79 ± 2.65 | 52.84 ± 1.53 | p < 0.001 |
| Gestational age (hbd) | 38.94 ± 0.82 | 39.02 ± 0.91 | 0.62 (NS) |
| Current height (cm) | 121.51 ± 9.34 | 125.73 ± 10.34 | p < 0.05 |
| Current weight (kg) | 23.22 ± 7.59 | 26.14 ± 9.13 | p < 0.05 |
| BMI | 15.35 ± 2.63 | 16.19 ± 2.97 | 0.12 (NS) |
| Ponderal index (g/cm3) | 2.02 ± 0.23 | 1.75 ± 0.16 | p < 0.00001 |
| Parameter | SGA Group (N = 93) Mean ± SD | Control Group (N = 47) Mean ± SD | p Value |
|---|---|---|---|
| Glucose (mg/dL) | 94.51 ± 12.47 | 84.64 ± 6.39 | <0.001 |
| Total cholesterol (mg/dL) | 190.61 ± 24.66 | 143.23 ± 23.90 | <0.001 |
| HDL cholesterol (mg/dL) | 71.46 ± 13.31 | 57.94 ± 18.50 | <0.001 |
| LDL cholesterol (mg/dL) | 105.35 ± 20.96 | 82.67 ± 16.92 | <0.001 |
| Triglycerides | 112.32 ± 38.10 | 55.89 ± 18.31 | <0.001 |
| Parameter | SGA Symmetrical (N = 43) Mean ± SD | SGA Asymmetrical (N = 50) Mean ± SD | p Value |
|---|---|---|---|
| Glucose (mg/dL) | 98.95 ± 14.79 | 90.56 ± 10.21 | <0.001 |
| Total cholesterol (mg/dL) | 190.90 ± 27.26 | 188.64 ± 23.70 | 0.73 (NS) |
| HDL cholesterol (mg/dL) | 70.95 ± 13.00 | 71.86 ± 13.76 | 0.85 (NS) |
| LDL cholesterol (mg/dL) | 104.65 ± 23.71 | 103.03 ± 19.49 | 0.87 (NS) |
| Triglycerides | 109.09 ± 38.42 | 111.4 ± 39.31 | 0.91 (NS) |
| Parameter | Glucose | Total Cholesterol | HDL Cholesterol | LDL Cholesterol | Triglycerides |
|---|---|---|---|---|---|
| Birth weight | 0.33 | 0.03 | 0.16 | −0.01 | 0.02 |
| Birth height | 0.26 | 0.31 | 0.19 | −0.11 | −0.02 |
| Actual weight | 0.18 | 0.05 | −0.24 | −0.17 | −0.18 |
| BMI | 0.10 | 0.04 | −0.38 * | 0.05 | −0.17 |
| Parameter | Glucose | Total Cholesterol | HDL Cholesterol | LDL Cholesterol | Triglycerides |
|---|---|---|---|---|---|
| Birth weight | −0.17 | −0.01 | 0.08 | −0.13 | −0.03 |
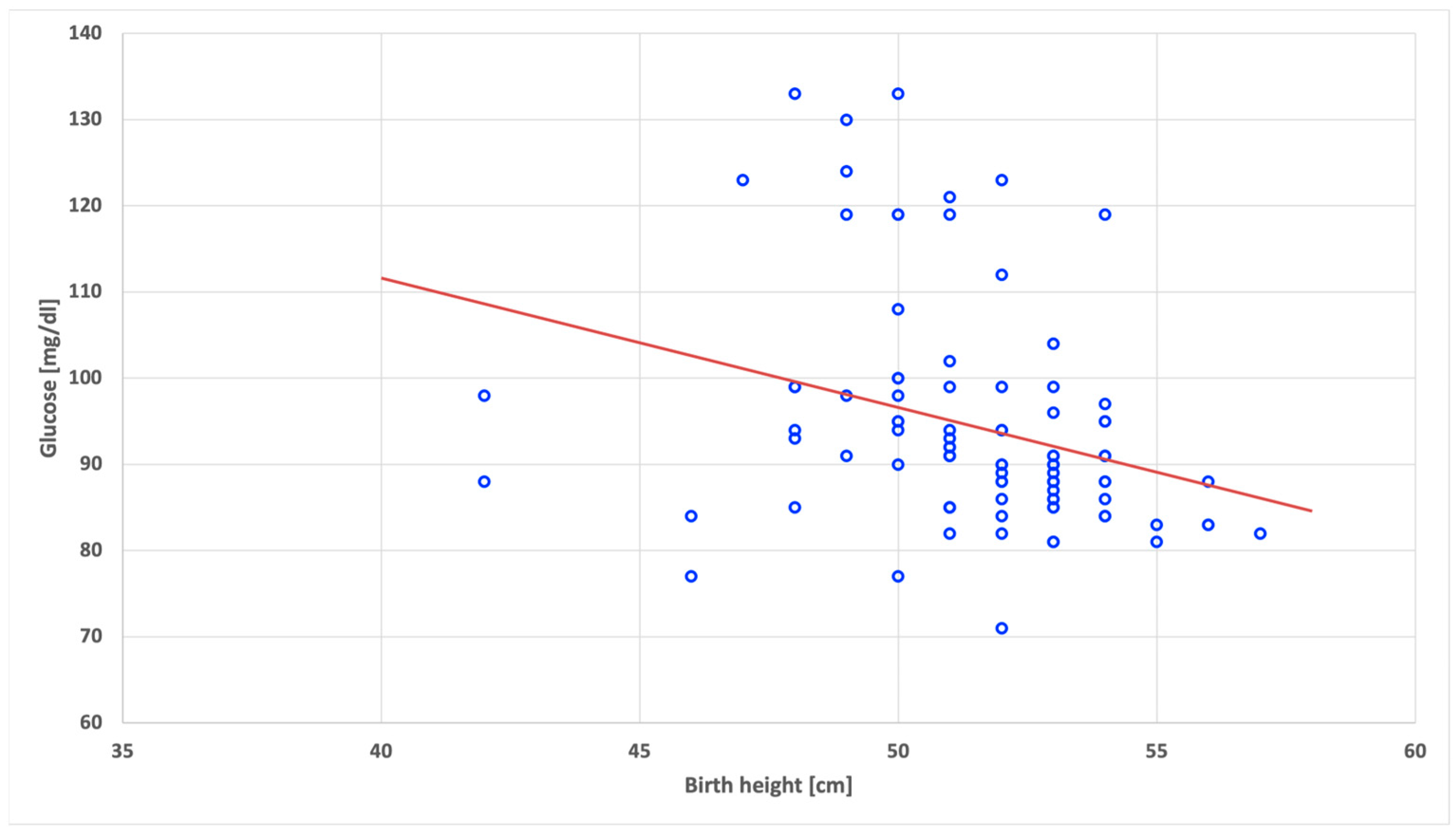
| Birth height | −0.40 * | 0.09 | 0.09 | 0.13 | −0.04 |
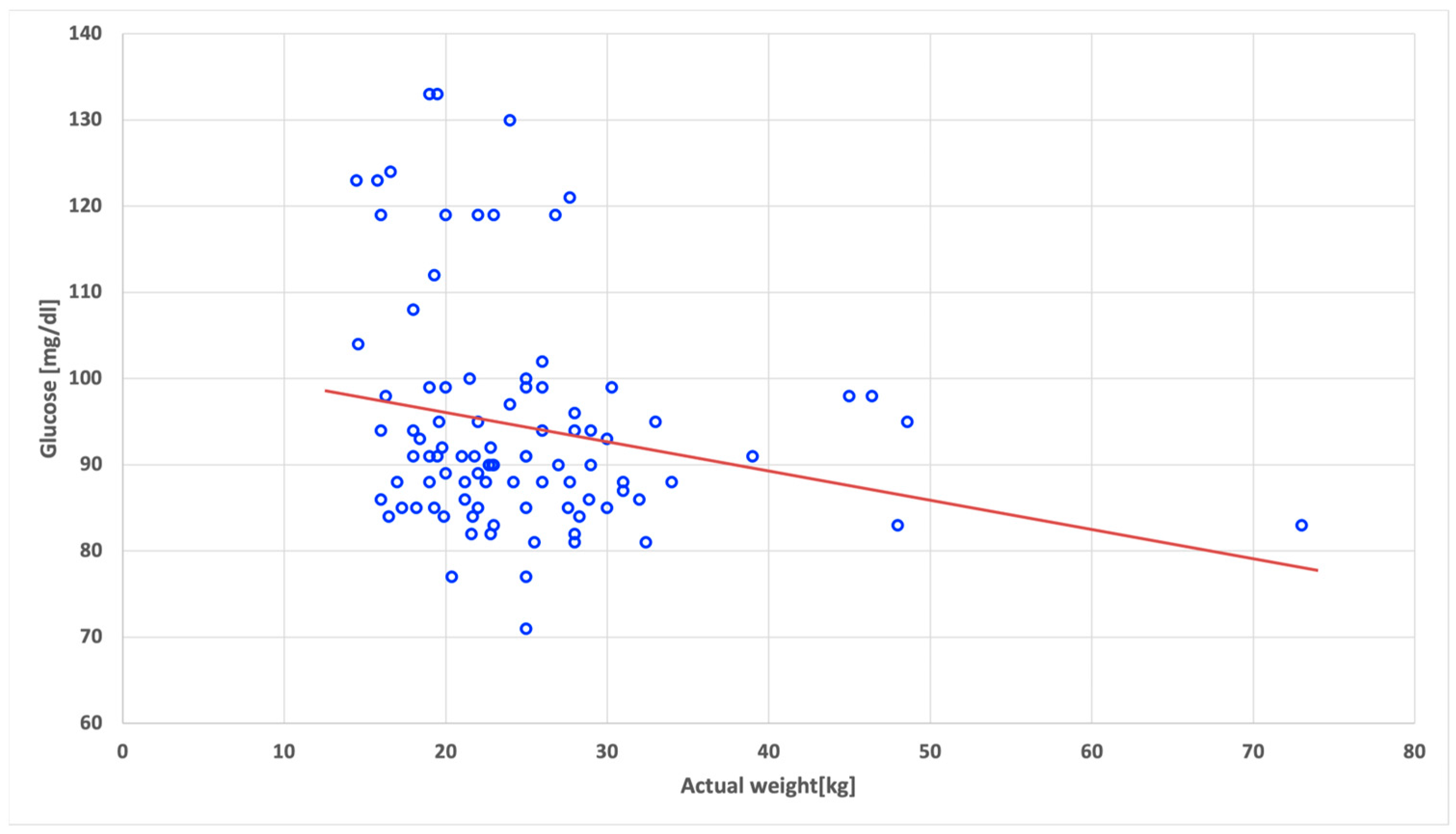
| Actual weight | −0.22 * | −0.01 | −0.002 | −0.03 | 0.08 |
| BMI | −0.16 | −0.09 | −0.06 | −0.08 | 0.10 |
| Parameter | Glucose | Total Cholesterol | HDL Cholesterol | LDL Cholesterol | Triglycerides |
|---|---|---|---|---|---|
| Birth weight | −0.14 | −0.14 | 0.09 | −0.07 | −0.11 |
| Birth height | −0.18 | 0.27 | 0.45 * | 0.23 | −0.002 |
| Actual weight | −0.26 | −0.05 | −0.05 | −0.14 | 0.01 |
| BMI | −0.15 | −0.18 | −0.19 | −0.22 | 0.08 |
| Parameter | Glucose | Total Cholesterol | HDL Cholesterol | LDL Cholesterol | Triglycerides |
|---|---|---|---|---|---|
| Birth weight | −0.06 | 0.03 | 0.01 | 0.07 | 0.001 |
| Birth height | −0.27 * | 0.10 | −0.06 | 0.16 | −0.04 |
| Actual weight | −0.08 | 0.11 | −0.01 | 0.09 | 0.11 |
| BMI | 0.03 | −0.05 | −0.04 | −0.04 | −0.006 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zamojska, J.; Niewiadomska-Jarosik, K.; Kierzkowska, B.; Gruca, M.; Wosiak, A.; Smolewska, E. Lipid Profile in Children Born Small for Gestational Age. Nutrients 2023, 15, 4781. https://doi.org/10.3390/nu15224781
Zamojska J, Niewiadomska-Jarosik K, Kierzkowska B, Gruca M, Wosiak A, Smolewska E. Lipid Profile in Children Born Small for Gestational Age. Nutrients. 2023; 15(22):4781. https://doi.org/10.3390/nu15224781
Chicago/Turabian StyleZamojska, Justyna, Katarzyna Niewiadomska-Jarosik, Beata Kierzkowska, Marta Gruca, Agnieszka Wosiak, and Elżbieta Smolewska. 2023. "Lipid Profile in Children Born Small for Gestational Age" Nutrients 15, no. 22: 4781. https://doi.org/10.3390/nu15224781
APA StyleZamojska, J., Niewiadomska-Jarosik, K., Kierzkowska, B., Gruca, M., Wosiak, A., & Smolewska, E. (2023). Lipid Profile in Children Born Small for Gestational Age. Nutrients, 15(22), 4781. https://doi.org/10.3390/nu15224781







