Yerba Maté (Ilex paraguariensis) Supplement Exerts Beneficial, Tissue-Specific Effects on Mitochondrial Efficiency and Redox Status in Healthy Adult Mice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Tissue Permeabilization
2.3. Mitochondrial Respirometry
2.4. ATP Quantification
2.5. Glutathione/Glutathione Disulfide Redox Potential Analysis
2.6. Statistics
3. Results
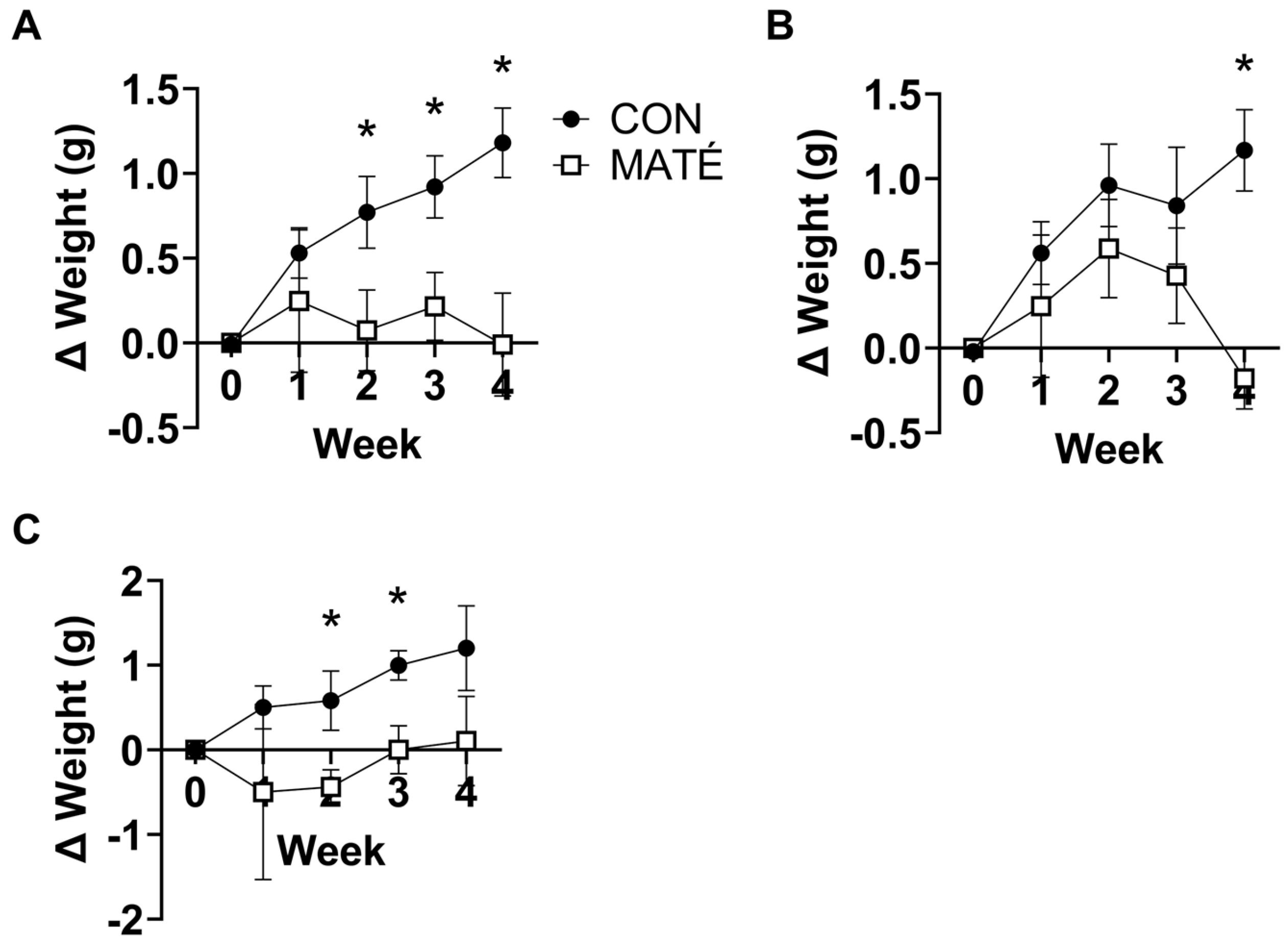
3.1. Yerba Maté Consumption Prevents Weight Gain in Male and Female Mice
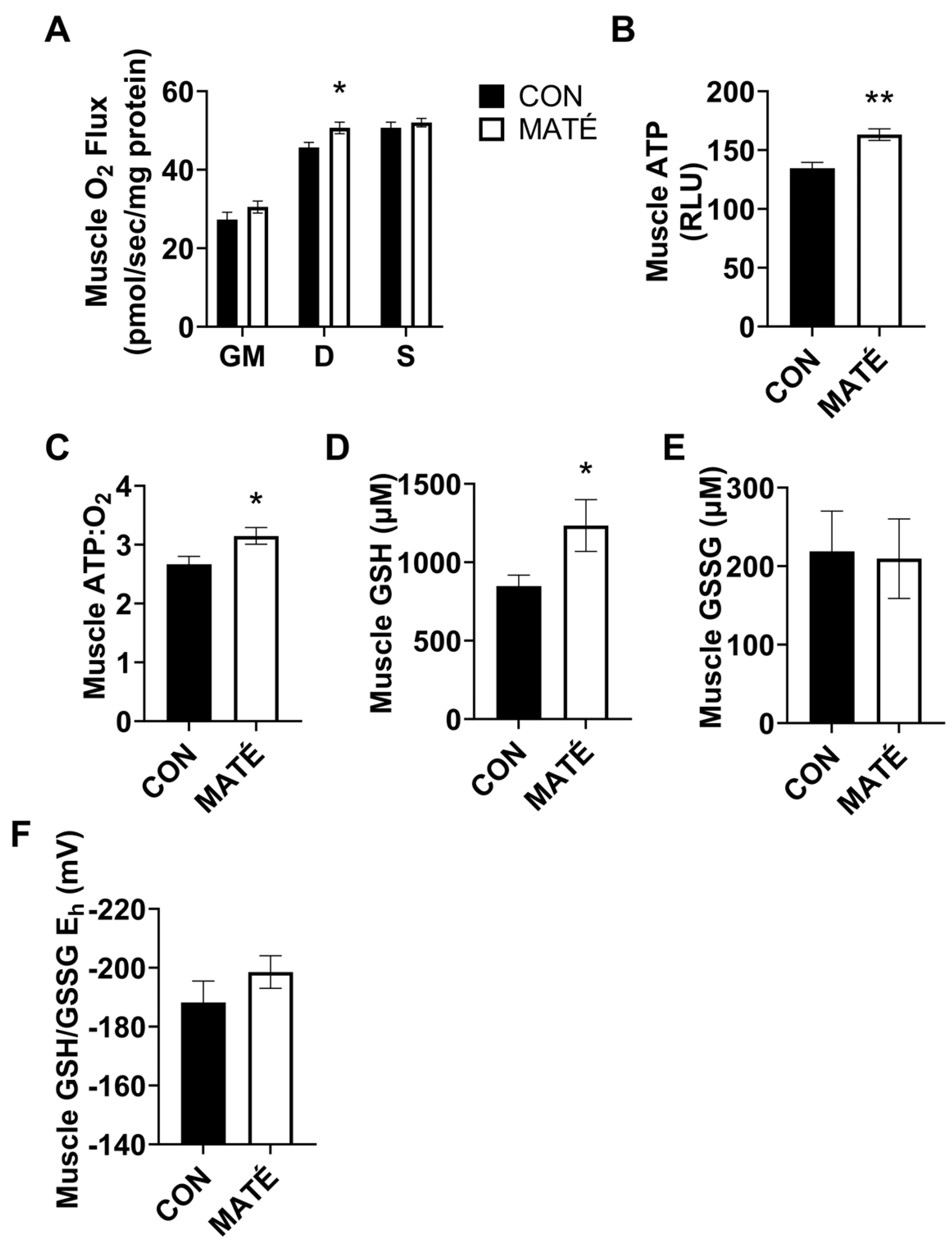
3.2. Yerba Maté Consumption Increases Mitochondrial Efficiency in Skeletal Muscle
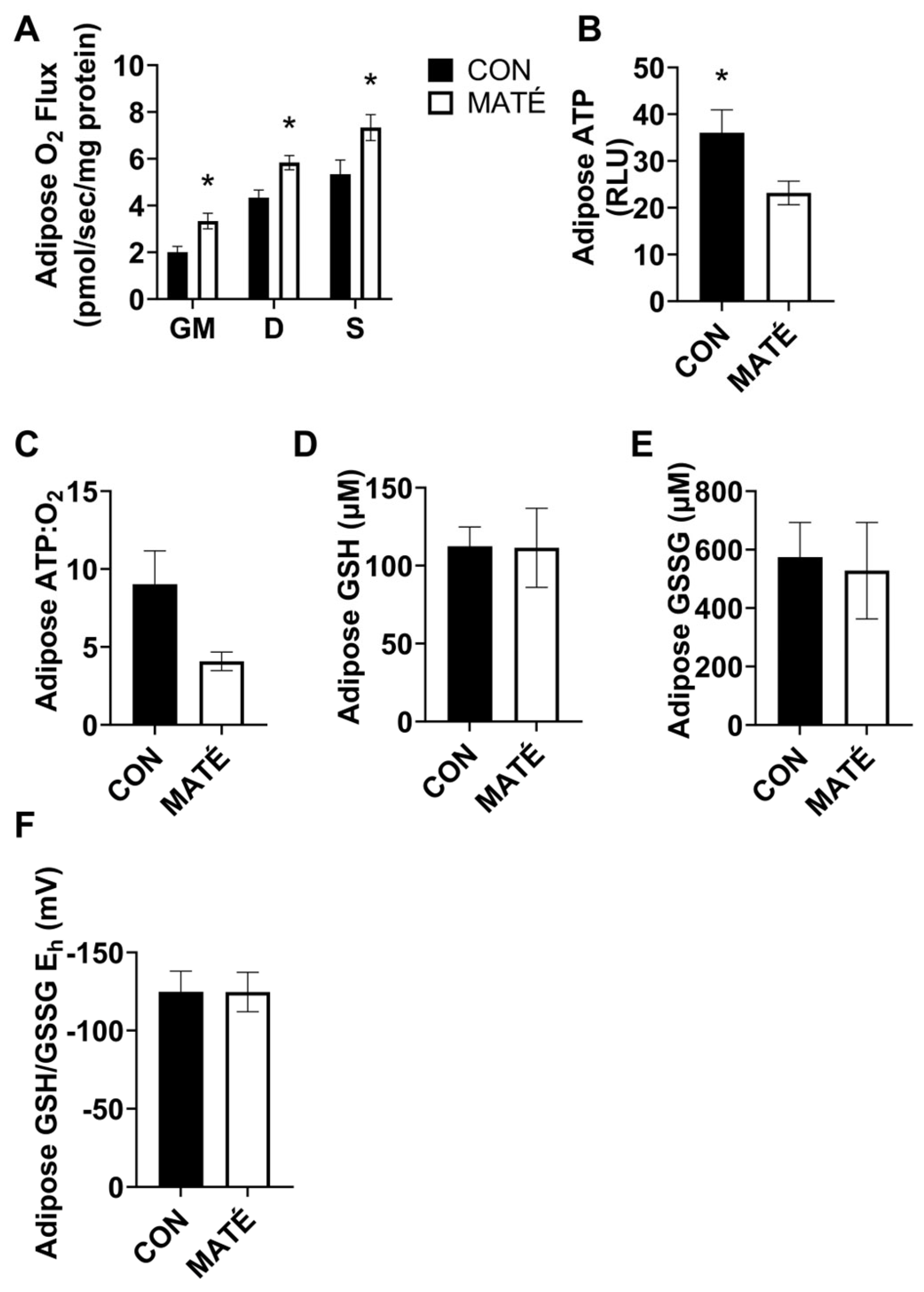
3.3. Yerba Maté Consumption Decreases Mitochondrial Efficiency in White Adipose Tissue
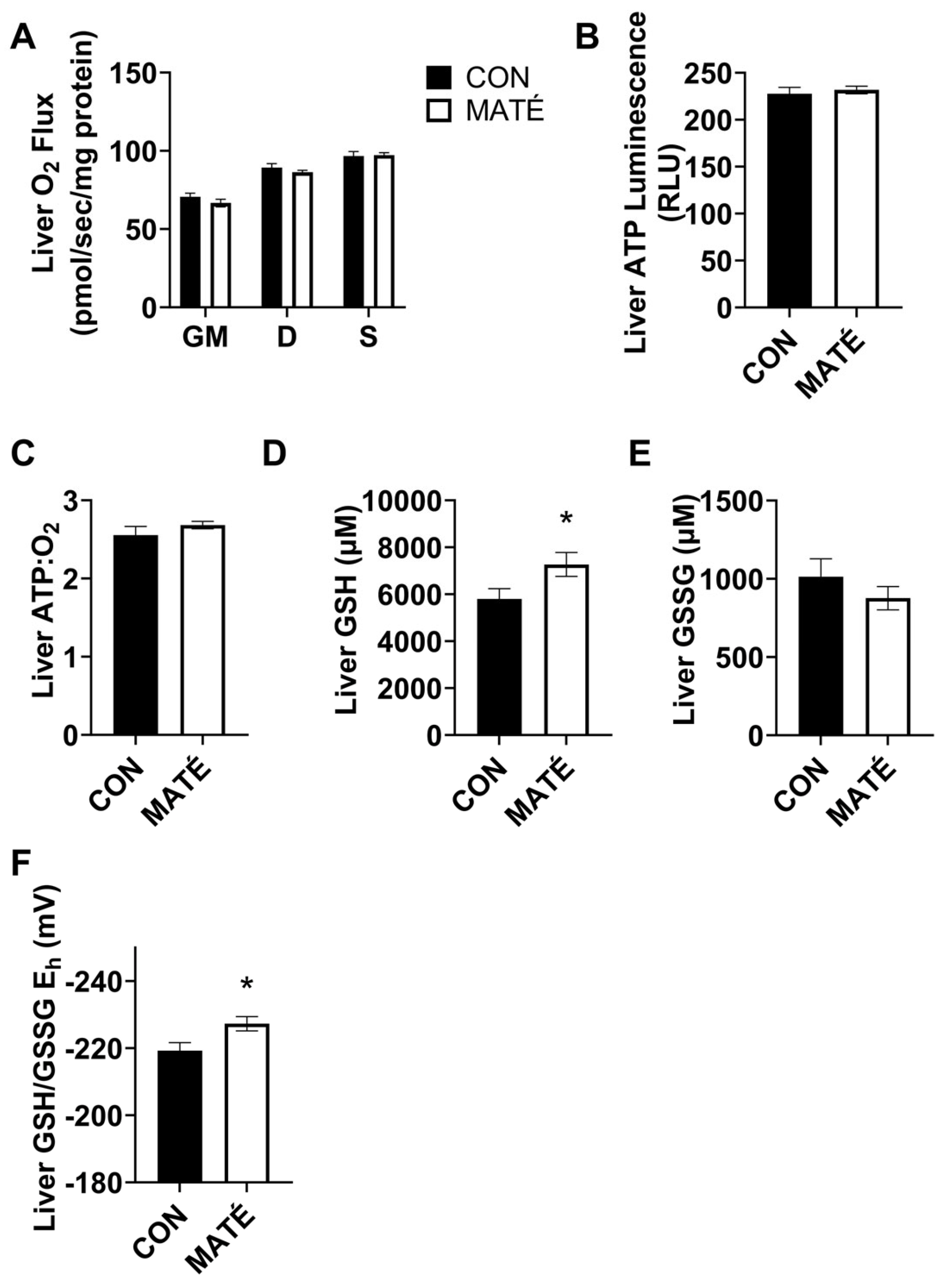
3.4. Yerba Maté Consumption Alters Hepatic Redox Potential but Not Mitochondrial Efficiency
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shai, I.; Schwarzfuchs, D.; Henkin, Y.; Shahar, D.R.; Witkow, S.; Greenberg, I.; Golan, R.; Fraser, D.; Bolotin, A.; Vardi, H.; et al. Weight loss with a low-carbohydrate, Mediterranean, or low-fat diet. N. Engl. J. Med. 2008, 359, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Hutchison, A.T.; Heilbronn, L.K. Metabolic impacts of altering meal frequency and timing—Does when we eat matter? Biochimie 2016, 124, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Plotnick, G.D.; Corretti, M.C.; Vogel, R.A.; Hesslink, R., Jr.; Wise, J.A. Effect of supplemental phytonutrients on impairment of the flow-mediated brachial artery vasoactivity after a single high-fat meal. J. Am. Coll. Cardiol. 2003, 41, 1744–1749. [Google Scholar] [CrossRef] [PubMed]
- Vasanthi, H.R.; ShriShriMal, N.; Das, D.K. Phytochemicals from plants to combat cardiovascular disease. Curr. Med. Chem. 2012, 19, 2242–2251. [Google Scholar] [CrossRef]
- Drašar, P.A.-O. Plant Secondary Metabolites Used for the Treatment of Diseases and Drug Development. Biomedicines 2022, 10, 576. [Google Scholar] [CrossRef]
- Miao, M.; Jiang, H.; Jiang, B.; Zhang, T.; Cui, S.W.; Jin, Z. Phytonutrients for controlling starch digestion: Evaluation of grape skin extract. Food Chem. 2014, 145, 205–211. [Google Scholar] [CrossRef]
- Weigel, H.J. Plant quality declines as CO2 levels rise. eLife 2014, 3, e03233. [Google Scholar] [CrossRef]
- Rowley, T.J.T.; Bitner, B.F.; Ray, J.D.; Lathen, D.R.; Smithson, A.T.; Dallon, B.W.; Plowman, C.J.; Bikman, B.T.; Hansen, J.M.; Dorenkott, M.R.; et al. Monomeric cocoa catechins enhance beta-cell function by increasing mitochondrial respiration. J. Nutr. Biochem. 2017, 49, 30–41. [Google Scholar] [CrossRef]
- Galli, R.L.; Shukitt-Hale, B.; Youdim, K.A.; Joseph, J.A. Fruit polyphenolics and brain aging: Nutritional interventions targeting age-related neuronal and behavioral deficits. Ann. N. Y. Acad. Sci. 2002, 959, 128–132. [Google Scholar] [CrossRef]
- Beecher, G.R. Phytonutrients’ role in metabolism: Effects on resistance to degenerative processes. Nutr. Rev. 1999, 57, S3–S6. [Google Scholar] [CrossRef]
- de Morais, E.C.; Stefanuto, A.; Klein, G.A.; Boaventura, B.C.; de Andrade, F.; Wazlawik, E.; Di Pietro, P.F.; Maraschin, M.; da Silva, E.L. Consumption of yerba mate (Ilex paraguariensis) improves serum lipid parameters in healthy dyslipidemic subjects and provides an additional LDL-cholesterol reduction in individuals on statin therapy. J. Agric. Food Chem. 2009, 57, 8316–8324. [Google Scholar] [CrossRef] [PubMed]
- Meinhart, A.D.; Damin, F.M.; Caldeirao, L.; da Silveira, T.F.F.; Filho, J.T.; Godoy, H.T. Chlorogenic acid isomer contents in 100 plants commercialized in Brazil. Food Res. Int. 2017, 99, 522–530. [Google Scholar] [CrossRef] [PubMed]
- Meng, S.; Cao, J.; Feng, Q.; Peng, J.; Hu, Y. Roles of chlorogenic Acid on regulating glucose and lipids metabolism: A review. Evid. Based Complement. Altern. Med. 2013, 2013, 801457. [Google Scholar] [CrossRef] [PubMed]
- Kungel, P.; Correa, V.G.; Correa, R.C.G.; Peralta, R.A.; Sokovic, M.; Calhelha, R.C.; Bracht, A.; Ferreira, I.; Peralta, R.M. Antioxidant and antimicrobial activities of a purified polysaccharide from yerba mate (Ilex paraguariensis). Int. J. Biol. Macromol. 2018, 114, 1161–1167. [Google Scholar] [CrossRef] [PubMed]
- Arçari, D.P.; Bartchewsky, W.; dos Santos, T.W.; Oliveira, K.A.; DeOliveira, C.C.; Gotardo, É.M.; Pedrazzoli, J.; Gambero, A.; Ferraz, L.F.C.; Carvalho, P.d.O. Anti-inflammatory effects of yerba maté extract (Ilex paraguariensis) ameliorate insulin resistance in mice with high fat diet-induced obesity. Mol. Cell. Endocrinol. 2011, 335, 110–115. [Google Scholar] [CrossRef]
- Maleki, S.J.; Crespo, J.F.; Cabanillas, B. Anti-inflammatory effects of flavonoids. Food Chem. 2019, 299, 125124. [Google Scholar] [CrossRef]
- Bhatti, J.S.; Bhatti, G.K.; Reddy, P.H. Mitochondrial dysfunction and oxidative stress in metabolic disorders—A step towards mitochondria based therapeutic strategies. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2017, 1863, 1066–1077. [Google Scholar] [CrossRef]
- Tippetts, T.S.; Winden, D.R.; Swensen, A.C.; Nelson, M.B.; Thatcher, M.O.; Saito, R.R.; Condie, T.B.; Simmons, K.J.; Judd, A.M.; Reynolds, P.R. Cigarette smoke increases cardiomyocyte ceramide accumulation and inhibits mitochondrial respiration. BMC Cardiovasc. Disord. 2014, 14, 165. [Google Scholar] [CrossRef]
- Hansen, M.; Tippetts, T.; Anderson, M.; Holub, Z.; Moulton, E.; Swensen, A.; Prince, J.; Bikman, B. Insulin increases ceramide synthesis in skeletal muscle. J. Diabetes Res. 2014, 2014, 765784. [Google Scholar] [CrossRef]
- Smith, M.E.; Tippetts, T.S.; Brassfield, E.S.; Tucker, B.J.; Ockey, A.; Swensen, A.C.; Anthonymuthu, T.S.; Washburn, T.D.; Kane, A.; Prince, J.T.; et al. Mitochondrial fission mediates ceramide-induced metabolic disruption in skeletal muscle. Biochem. J. 2013, 456, 427–439. [Google Scholar] [CrossRef]
- Walton, C.M.; Jacobsen, S.M.; Dallon, B.W.; Saito, E.R.; Bennett, S.L.H.; Davidson, L.E.; Thomson, D.M.; Hyldahl, R.D.; Bikman, B.T. Ketones elicit distinct alterations in adipose mitochondrial bioenergetics. Int. J. Mol. Sci. 2020, 21, 6255. [Google Scholar] [CrossRef] [PubMed]
- Piorczynski, T.B.; Lapehn, S.; Ringer, K.P.; Allen, S.A.; Johnson, G.A.; Call, K.; Lucas, S.M.; Harris, C.; Hansen, J.M. NRF2 activation inhibits valproic acid-induced neural tube defects in mice. Neurotoxicology Teratol. 2022, 89, 107039. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.P.; Carlson, J.L.; Mody, V.C., Jr.; Cai, J.; Lynn, M.J.; Sternberg, P., Jr. Redox state of glutathione in human plasma. Free Radic. Biol. Med. 2000, 28, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.P. Redox potential of GSH/GSSG couple: Assay and biological significance. In Methods in Enzymology; Elsevier: Amsterdam, The Netherlands, 2002; Volume 348, pp. 93–112. [Google Scholar]
- Saito, E.R.; Warren, C.E.; Hanegan, C.M.; Larsen, J.G.; du Randt, J.D.; Cannon, M.; Saito, J.Y.; Campbell, R.J.; Kemberling, C.M.; Miller, G.S. A novel ketone-supplemented diet improves recognition memory and hippocampal mitochondrial efficiency in healthy adult mice. Metabolites 2022, 12, 1019. [Google Scholar] [CrossRef]
- Andrade, V.M.d.M.; de Moura, A.F.; da Costa Chaves, K.; da Rocha, C.P.D.; de Andrade, C.B.V.; Trevenzoli, I.H.; Ortiga-Carvalho, T.M.; Barcellos, L.C.; Vaisman, M.; Salerno, V.P. Yerba mate consumption by ovariectomized rats alters white adipose tissue. Mol. Cell. Endocrinol. 2023, 564, 111881. [Google Scholar] [CrossRef]
- Gao, H.; Long, Y.; Jiang, X.; Liu, Z.; Wang, D.; Zhao, Y.; Li, D.; Sun, B.-l. Beneficial effects of Yerba Mate tea (Ilex paraguariensis) on hyperlipidemia in high-fat-fed hamsters. Exp. Gerontol. 2013, 48, 572–578. [Google Scholar] [CrossRef]
- Harrold, J.A.; Hughes, G.M.; O’Shiel, K.; Quinn, E.; Boyland, E.J.; Williams, N.J.; Halford, J.C.G. Acute effects of a herb extract formulation and inulin fibre on appetite, energy intake and food choice. Appetite 2013, 62, 84–90. [Google Scholar] [CrossRef]
- Andersen, T.; Fogh, J. Weight loss and delayed gastric emptying following a South American herbal preparation in overweight patients. J. Hum. Nutr. Diet. 2001, 14, 243–250. [Google Scholar] [CrossRef]
- Arçari, D.P.; Bartchewsky, W.; dos Santos, T.W.; Oliveira, K.A.; Funck, A.; Pedrazzoli, J.; de Souza, M.F.F.; Saad, M.J.; Bastos, D.H.M.; Gambero, A.; et al. Antiobesity Effects of yerba maté Extract (Ilex paraguariensis) in High-fat Diet–induced Obese Mice. Obesity 2012, 17, 2127–2133. [Google Scholar] [CrossRef]
- Silva, R.D.A.; Bueno, A.L.S.; Gallon, C.W.; Gomes, L.F.; Kaiser, S.; Pavei, C.; Ortega, G.G.; Kucharski, L.C.; Jahn, M.P. The effect of aqueous extract of gross and commercial yerba mate (Ilex paraguariensis) on intra-abdominal and epididymal fat and glucose levels in male Wistar rats. Fitoterapia 2011, 82, 818–826. [Google Scholar] [CrossRef]
- Kim, S.-Y.; Oh, M.-R.; Kim, M.-G.; Chae, H.-J.; Chae, S.-W. Anti-obesity effects of Yerba Mate (Ilex Paraguariensis): A randomized, double-blind, placebo-controlled clinical trial. BMC Complement. Altern. Med. 2015, 15, 338. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Ko, J.; Storni, C.; Song, H.J.; Cho, Y.G. Effect of green mate in overweight volunteers: A randomized placebo-controlled human study. J. Funct. Foods 2012, 4, 287–293. [Google Scholar] [CrossRef]
- Lobo, P.C.B.; da Silva, D.D.; Pimentel, G.D. Acute Supplementation of Yerba Mate Extract Did Not Change Muscle Strength in Physically Active Men Following the Strength Muscle Test: A Pilot Clinical Trial. Nutrients 2022, 14, 2619. [Google Scholar] [CrossRef] [PubMed]
- Panza, V.P.; Diefenthaeler, F.; Tamborindeguy, A.C.; de Quadros Camargo, C.; de Moura, B.M.; Brunetta, H.S.; Sakugawa, R.L.; de Oliveira, M.V.; de Oliveira Puel, E.; Nunes, E.A. Effects of mate tea consumption on muscle strength and oxidative stress markers after eccentric exercise. Br. J. Nutr. 2016, 115, 1370–1378. [Google Scholar] [CrossRef]
- Krolikowski, T.C.; Borszcz, F.K.; Panza, V.P.; Bevilacqua, L.M.; Nichele, S.; da Silva, E.L.; Amboni, R.D.M.C.; Guglielmo, L.G.A.; Phillips, S.M.; de Lucas, R.D.; et al. The Impact of Pre-Exercise Carbohydrate Meal on the Effects of Yerba Mate Drink on Metabolism, Performance, and Antioxidant Status in Trained Male Cyclists. Sports Med. Open 2022, 8, 93. [Google Scholar] [CrossRef] [PubMed]
- Alkhatib, A. Yerba Maté (Illex paraguariensis) ingestion augments fat oxidation and energy expenditure during exercise at various submaximal intensities. Nutr. Metab. 2014, 11, 42. [Google Scholar] [CrossRef]
- Gawron-Gzella, A.A.-O.; Chanaj-Kaczmarek, J.A.-O.; Cielecka-Piontek, J.A.-O. Yerba Mate-A Long but Current History. Nutrients 2021, 13, 3706. [Google Scholar] [CrossRef]
- Ruíz-Moreno, C.; Lara, B.; Brito de Souza, D.; Gutiérrez-Hellín, J.; Romero-Moraleda, B.; Cuéllar-Rayo, Á.; Del Coso, J. Acute caffeine intake increases muscle oxygen saturation during a maximal incremental exercise test. Br. J. Clin. Pharmacol. 2020, 86, 861–867. [Google Scholar] [CrossRef]
- Bracco, D.; Ferrarra, J.M.; Arnaud, M.J.; Jequier, E.; Schutz, Y. Effects of caffeine on energy metabolism, heart rate, and methylxanthine metabolism in lean and obese women. Am. J. Physiol.-Endocrinol. Metab. 1995, 269, E671–E678. [Google Scholar] [CrossRef]
- Jo, E.; Lewis, K.L.; Higuera, D.; Hernandez, J.; Osmond, A.D.; Directo, D.J.; Wong, M. Dietary Caffeine and Polyphenol Supplementation Enhances Overall Metabolic Rate and Lipid Oxidation at Rest and After a Bout of Sprint Interval Exercise. J. Strength Cond. Res. 2016, 30, 1871–1879. [Google Scholar] [CrossRef]
- Velickovic, K.; Wayne, D.; Leija, H.A.L.; Bloor, I.; Morris, D.E.; Law, J.; Budge, H.; Sacks, H.; Symonds, M.E.; Sottile, V. Caffeine exposure induces browning features in adipose tissue in vitro and in vivo. Sci. Rep. 2019, 9, 9104. [Google Scholar] [CrossRef] [PubMed]
- Bartelt, A.; Heeren, J. Adipose tissue browning and metabolic health. Nat. Rev. Endocrinol. 2014, 10, 24–36. [Google Scholar] [CrossRef] [PubMed]
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The hallmarks of aging. Cell 2013, 153, 1194–1217. [Google Scholar] [CrossRef] [PubMed]
- Rani, V.; Deep, G.; Singh, R.K.; Palle, K.; Yadav, U.C.S. Oxidative stress and metabolic disorders: Pathogenesis and therapeutic strategies. Life Sci. 2016, 148, 183–193. [Google Scholar] [CrossRef]
- Rotariu, D.; Babes, E.E.; Tit, D.M.; Moisi, M.; Bustea, C.; Stoicescu, M.; Radu, A.-F.; Vesa, C.M.; Behl, T.; Bungau, A.F. Oxidative stress–Complex pathological issues concerning the hallmark of cardiovascular and metabolic disorders. Biomed. Pharmacother. 2022, 152, 113238. [Google Scholar] [CrossRef]
- Ighodaro, O.M.; Akinloye, O.A. First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alex. J. Med. 2018, 54, 287–293. [Google Scholar] [CrossRef]
- Biswas, S.K.; Rahman, I. Environmental toxicity, redox signaling and lung inflammation: The role of glutathione. Mol. Asp. Med. 2010, 30, 60–76. [Google Scholar] [CrossRef]
- Harris, C.; Hansen, J.M. Oxidative stress, thiols, and redox profiles. Dev. Toxicol. Methods Protoc. 2012, 889, 325–346. [Google Scholar]
- Nogueira Silva Lima, M.T.; Boulanger, E.; Tessier, F.J.; Takahashi, J.A. Hibiscus, rooibos, and yerba mate for healthy aging: A review on the attenuation of in vitro and in vivo markers related to oxidative stress, glycoxidation, and neurodegeneration. Foods 2022, 11, 1676. [Google Scholar] [CrossRef]
- Lu, S.C. Dysregulation of glutathione synthesis in liver disease. Liver Res. 2020, 4, 64–73. [Google Scholar] [CrossRef]
- Ballatori, N.; Krance, S.M.; Notenboom, S.; Shi, S.; Tieu, K.; Hammond, C.L. Glutathione dysregulation and the etiology and progression of human diseases. Biol. Chem. 2009, 390, 191–214. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Walton, C.M.; Saito, E.R.; Warren, C.E.; Larsen, J.G.; Remund, N.P.; Reynolds, P.R.; Hansen, J.M.; Bikman, B.T. Yerba Maté (Ilex paraguariensis) Supplement Exerts Beneficial, Tissue-Specific Effects on Mitochondrial Efficiency and Redox Status in Healthy Adult Mice. Nutrients 2023, 15, 4454. https://doi.org/10.3390/nu15204454
Walton CM, Saito ER, Warren CE, Larsen JG, Remund NP, Reynolds PR, Hansen JM, Bikman BT. Yerba Maté (Ilex paraguariensis) Supplement Exerts Beneficial, Tissue-Specific Effects on Mitochondrial Efficiency and Redox Status in Healthy Adult Mice. Nutrients. 2023; 15(20):4454. https://doi.org/10.3390/nu15204454
Chicago/Turabian StyleWalton, Chase M., Erin R. Saito, Cali E. Warren, John G. Larsen, Nicole P. Remund, Paul R. Reynolds, Jason M. Hansen, and Benjamin T. Bikman. 2023. "Yerba Maté (Ilex paraguariensis) Supplement Exerts Beneficial, Tissue-Specific Effects on Mitochondrial Efficiency and Redox Status in Healthy Adult Mice" Nutrients 15, no. 20: 4454. https://doi.org/10.3390/nu15204454
APA StyleWalton, C. M., Saito, E. R., Warren, C. E., Larsen, J. G., Remund, N. P., Reynolds, P. R., Hansen, J. M., & Bikman, B. T. (2023). Yerba Maté (Ilex paraguariensis) Supplement Exerts Beneficial, Tissue-Specific Effects on Mitochondrial Efficiency and Redox Status in Healthy Adult Mice. Nutrients, 15(20), 4454. https://doi.org/10.3390/nu15204454








