Serum 25-Hydroxyvitamin D and Cancer Risk: A Systematic Review of Mendelian Randomization Studies
Abstract
1. Introduction
2. Materials and Methods
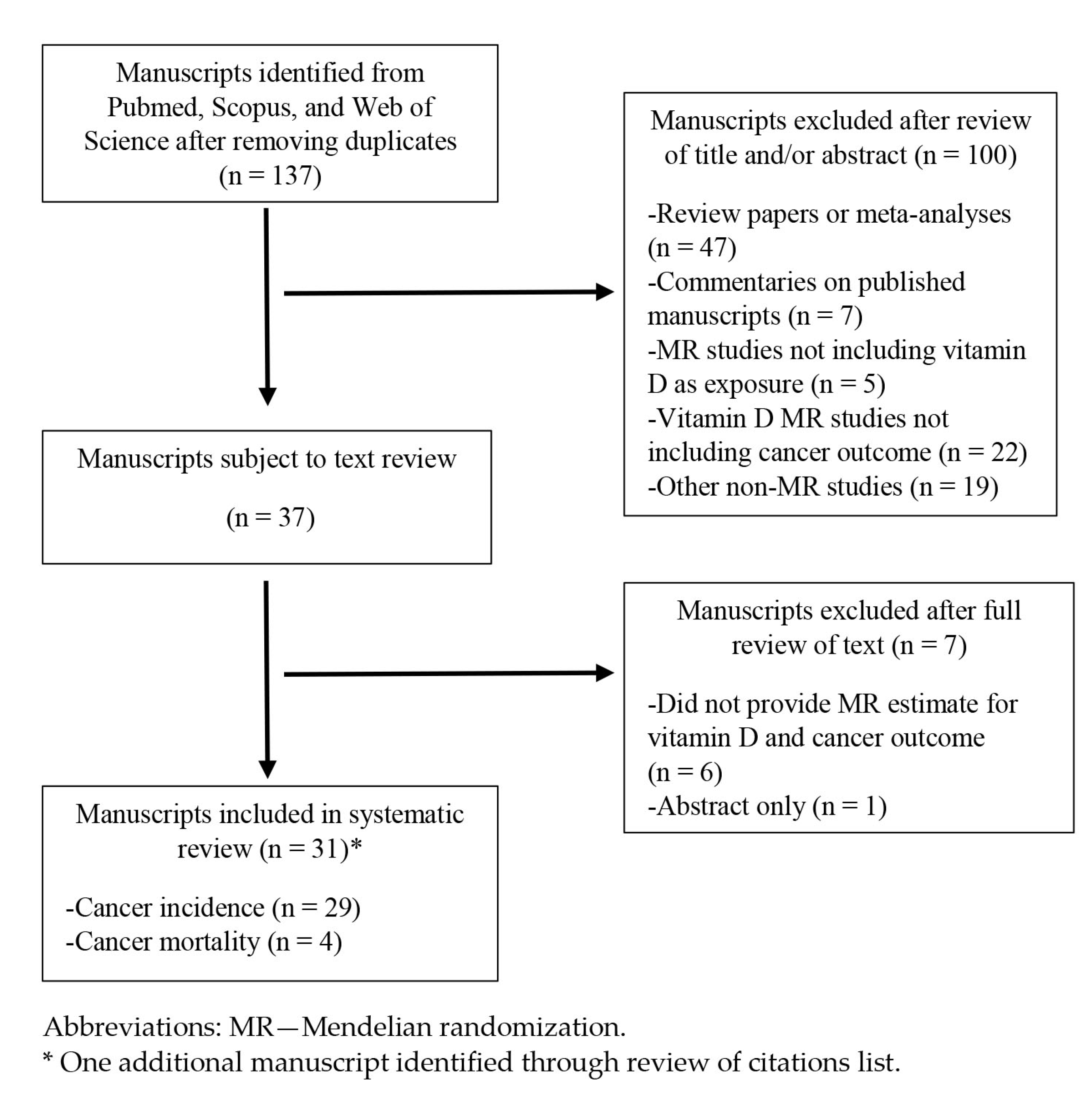
2.1. Literature Review
2.2. Data Extraction
3. Results
3.1. Overview of MR Studies
3.2. Summary of MR Estimates for Cancer Incidence
3.3. Summary of MR Estimates for Cancer-Specific Mortality
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dou, R.; Ng, K.; Giovannucci, E.L.; Manson, J.E.; Qian, Z.R.; Ogino, S. Vitamin D and colorectal cancer: Molecular, epidemiological and clinical evidence. Br. J. Nutr. 2016, 115, 1643–1660. [Google Scholar] [CrossRef] [PubMed]
- Bandera Merchan, B.; Morcillo, S.; Martin-Nuñez, G.; Tinahones, F.J.; Macías-González, M. The role of vitamin D and VDR in carcinogenesis: Through epidemiology and basic sciences. J. Steroid Biochem. Mol. Biol. 2017, 167, 203–218. [Google Scholar] [CrossRef] [PubMed]
- Feldman, D.; Krishnan, A.V.; Swami, S.; Giovannucci, E.; Feldman, B.J. The role of vitamin D in reducing cancer risk and progression. Nat. Rev. Cancer 2014, 14, 342–357. [Google Scholar] [CrossRef] [PubMed]
- Moradi, S.; Shahdadian, F.; Mohammadi, H.; Rouhani, M.H. A comparison of the effect of supplementation and sunlight exposure on serum vitamin D and parathyroid hormone: A systematic review and meta-analysis. Crit. Rev. Food Sci. Nutr. 2020, 60, 1881–1889. [Google Scholar] [CrossRef] [PubMed]
- Garland, C.F.; Gorham, E.D. Dose-response of serum 25-hydroxyvitamin D in association with risk of colorectal cancer: A meta-analysis. J. Steroid Biochem. Mol. Biol. 2017, 168, 1–8. [Google Scholar] [CrossRef]
- Lee, J.E.; Li, H.; Chan, A.T.; Hollis, B.W.; Lee, I.-M.; Stampfer, M.J.; Wu, K.; Giovannucci, E.; Ma, J. Circulating levels of vitamin D and colon and rectal cancer: The Physicians’ Health Study and a meta-analysis of prospective studies. Cancer Prev. Res. 2011, 4, 735–743. [Google Scholar] [CrossRef]
- McCullough, M.L.; Zoltick, E.S.; Weinstein, S.J.; Fedirko, V.; Wang, M.; Cook, N.R.; Eliassen, A.H.; Zeleniuch-Jacquotte, A.; Agnoli, C.; Albanes, D.; et al. Circulating Vitamin D and Colorectal Cancer Risk: An International Pooling Project of 17 Cohorts. J. Natl. Cancer Inst. 2019, 111, 158–169. [Google Scholar] [CrossRef]
- Song, D.; Deng, Y.; Liu, K.; Zhou, L.; Li, N.; Zheng, Y.; Hao, Q.; Yang, S.; Wu, Y.; Zhai, Z.; et al. Vitamin D intake, blood vitamin D levels, and the risk of breast cancer: A dose-response meta-analysis of observational studies. Aging 2019, 11, 12708–12732. [Google Scholar] [CrossRef]
- Hossain, S.; Beydoun, M.A.; Beydoun, H.A.; Chen, X.; Zonderman, A.B.; Wood, R.J. Vitamin D and breast cancer: A systematic review and meta-analysis of observational studies. Clin. Nutr. ESPEN 2019, 30, 170–184. [Google Scholar] [CrossRef]
- Estébanez, N.; Gómez-Acebo, I.; Palazuelos, C.; Llorca, J.; Dierssen-Sotos, T. Vitamin D exposure and Risk of Breast Cancer: A meta-analysis. Sci. Rep. 2018, 8, 9039. [Google Scholar] [CrossRef]
- Wei, H.; Jing, H.; Wei, Q.; Wei, G.; Heng, Z. Associations of the risk of lung cancer with serum 25-hydroxyvitamin D level and dietary vitamin D intake: A dose-response PRISMA meta-analysis. Medicine 2018, 97, e12282. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, S.; Che, X.; Li, X. Vitamin D and lung cancer risk: A comprehensive review and meta-analysis. Cell. Physiol. Biochem. 2015, 36, 299–305. [Google Scholar] [CrossRef]
- Feng, Q.; Zhang, H.; Dong, Z.; Zhou, Y.; Ma, J. Circulating 25-hydroxyvitamin D and lung cancer risk and survival: A dose-response meta-analysis of prospective cohort studies. Medicine 2017, 96, e8613. [Google Scholar] [CrossRef]
- Jensen, S.S.; Madsen, M.W.; Lukas, J.; Binderup, L.; Bartek, J. Inhibitory effects of 1alpha,25-dihydroxyvitamin D(3) on the G(1)-S phase-controlling machinery. Mol. Endocrinol. 2001, 15, 1370–1380. [Google Scholar] [CrossRef]
- Yang, K.; Lamprecht, S.A.; Shinozaki, H.; Fan, K.; Yang, W.; Newmark, H.L.; Kopelovich, L.; Edelmann, W.; Jin, B.; Gravaghi, C.; et al. Dietary calcium and cholecalciferol modulate cyclin D1 expression, apoptosis, and tumorigenesis in intestine of adenomatous polyposis coli1638N/+ mice. J. Nutr. 2008, 138, 1658–1663. [Google Scholar] [CrossRef]
- Xin, Y.; He, L.; Luan, Z.; Lv, H.; Yang, H.; Zhou, Y.; Zhao, X.; Zhou, W.; Yu, S.; Tan, B.; et al. E-cadherin Mediates the Preventive Effect of Vitamin D3 in Colitis-associated Carcinogenesis. Inflamm. Bowel Dis. 2017, 23, 1535–1543. [Google Scholar] [CrossRef]
- Blutt, S.E.; Allegretto, E.A.; Pike, J.W.; Weigel, N.L. 1,25-dihydroxyvitamin D3 and 9-cis-retinoic acid act synergistically to inhibit the growth of LNCaP prostate cells and cause accumulation of cells in G1. Endocrinology 1997, 138, 1491–1497. [Google Scholar] [CrossRef]
- Chung, I.; Han, G.; Seshadri, M.; Gillard, B.M.; Yu, W.; Foster, B.A.; Trump, D.L.; Johnson, C.S. Role of vitamin D receptor in the antiproliferative effects of calcitriol in tumor-derived endothelial cells and tumor angiogenesis in vivo. Cancer Res. 2009, 69, 967–975. [Google Scholar] [CrossRef]
- Ben-Shoshan, M.; Amir, S.; Dang, D.T.; Dang, L.H.; Weisman, Y.; Mabjeesh, N.J. 1alpha,25-dihydroxyvitamin D3 (Calcitriol) inhibits hypoxia-inducible factor-1/vascular endothelial growth factor pathway in human cancer cells. Mol. Cancer Ther. 2007, 6, 1433–1439. [Google Scholar] [CrossRef]
- Bao, B.-Y.; Yao, J.; Lee, Y.-F. 1alpha, 25-dihydroxyvitamin D3 suppresses interleukin-8-mediated prostate cancer cell angiogenesis. Carcinogenesis 2006, 27, 1883–1893. [Google Scholar] [CrossRef]
- Cohen-Lahav, M.; Shany, S.; Tobvin, D.; Chaimovitz, C.; Douvdevani, A. Vitamin D decreases NFkappaB activity by increasing IkappaBalpha levels. Nephrol. Dial. Transplant. 2006, 21, 889–897. [Google Scholar] [CrossRef] [PubMed]
- van Harten-Gerritsen, A.S.; Balvers, M.G.J.; Witkamp, R.F.; Kampman, E.; van Duijnhoven, F.J.B. Vitamin D, Inflammation, and Colorectal Cancer Progression: A Review of Mechanistic Studies and Future Directions for Epidemiological Studies. Cancer Epidemiol. Biomark. Prev. 2015, 24, 1820–1828. [Google Scholar] [CrossRef] [PubMed]
- Moreno, J.; Krishnan, A.V.; Swami, S.; Nonn, L.; Peehl, D.M.; Feldman, D. Regulation of prostaglandin metabolism by calcitriol attenuates growth stimulation in prostate cancer cells. Cancer Res. 2005, 65, 7917–7925. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Sun, H.; Hu, M.; Zhang, Y.; Chen, S.; Tighe, S.; Zhu, Y. The Role of Cyclooxygenase-2 in Colorectal Carcinogenesis. Clin. Colorectal Cancer 2017, 16, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Zhang, H.; Wu, H.; Li, H.; Liu, L.; Guo, J.; Li, C.; Shih, D.Q.; Zhang, X. Protective role of 1,25(OH)2 vitamin D3 in the mucosal injury and epithelial barrier disruption in DSS-induced acute colitis in mice. BMC Gastroenterol. 2012, 12, 57. [Google Scholar] [CrossRef]
- Ross, A.C.; Manson, J.E.; Abrams, S.A.; Aloia, J.F.; Brannon, P.M.; Clinton, S.K.; Durazo-Arvizu, R.A.; Gallagher, J.C.; Gallo, R.L.; Jones, G.; et al. The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: What clinicians need to know. J. Clin. Endocrinol. Metab. 2011, 96, 53–58. [Google Scholar] [CrossRef]
- Giovannucci, E. Modifiable risk factors for colon cancer. Gastroenterol. Clin. N. Am. 2002, 31, 925–943. [Google Scholar] [CrossRef]
- Wactawski-Wende, J.; Kotchen, J.M.; Anderson, G.L.; Assaf, A.R.; Brunner, R.L.; O’Sullivan, M.J.; Margolis, K.L.; Ockene, J.K.; Phillips, L.; Pottern, L.; et al. Calcium plus vitamin D supplementation and the risk of colorectal cancer. N. Engl. J. Med. 2006, 354, 684–696. [Google Scholar] [CrossRef]
- Manson, J.E.; Cook, N.R.; Lee, I.-M.; Christen, W.; Bassuk, S.S.; Mora, S.; Gibson, H.; Gordon, D.; Copeland, T.; D’Agostino, D.; et al. Vitamin D Supplements and Prevention of Cancer and Cardiovascular Disease. N. Engl. J. Med. 2019, 380, 33–44. [Google Scholar] [CrossRef]
- Scragg, R.; Khaw, K.-T.; Toop, L.; Sluyter, J.; Lawes, C.M.M.; Waayer, D.; Giovannucci, E.; Camargo, C.A. Monthly High-Dose Vitamin D Supplementation and Cancer Risk: A Post Hoc Analysis of the Vitamin D Assessment Randomized Clinical Trial. JAMA Oncol. 2018, 4, e182178. [Google Scholar] [CrossRef]
- Chlebowski, R.T.; Johnson, K.C.; Kooperberg, C.; Pettinger, M.; Wactawski-Wende, J.; Rohan, T.; Rossouw, J.; Lane, D.; O’Sullivan, M.J.; Yasmeen, S.; et al. Calcium plus vitamin D supplementation and the risk of breast cancer. J. Natl. Cancer Inst. 2008, 100, 1581–1591. [Google Scholar] [CrossRef]
- Brunner, R.L.; Wactawski-Wende, J.; Caan, B.J.; Cochrane, B.B.; Chlebowski, R.T.; Gass, M.L.S.; Jacobs, E.T.; LaCroix, A.Z.; Lane, D.; Larson, J.; et al. The effect of calcium plus vitamin D on risk for invasive cancer: Results of the Women’s Health Initiative (WHI) calcium plus vitamin D randomized clinical trial. Nutr. Cancer 2011, 63, 827–841. [Google Scholar] [CrossRef]
- Chandler, P.D.; Chen, W.Y.; Ajala, O.N.; Hazra, A.; Cook, N.; Bubes, V.; Lee, I.-M.; Giovannucci, E.L.; Willett, W.; Buring, J.E.; et al. Effect of Vitamin D3 Supplements on Development of Advanced Cancer: A Secondary Analysis of the VITAL Randomized Clinical Trial. JAMA Netw. Open 2020, 3, e2025850. [Google Scholar] [CrossRef]
- Herrick, K.A.; Storandt, R.J.; Afful, J.; Pfeiffer, C.M.; Schleicher, R.L.; Gahche, J.J.; Potischman, N. Vitamin D status in the United States, 2011-2014. Am. J. Clin. Nutr. 2019, 110, 150–157. [Google Scholar] [CrossRef]
- Keum, N.; Lee, D.H.; Greenwood, D.C.; Manson, J.E.; Giovannucci, E. Vitamin D supplementation and total cancer incidence and mortality: A meta-analysis of randomized controlled trials. Ann. Oncol. 2019, 30, 733–743. [Google Scholar] [CrossRef]
- Zhou, L.; Chen, B.; Sheng, L.; Turner, A. The effect of vitamin D supplementation on the risk of breast cancer: A trial sequential meta-analysis. Breast Cancer Res. Treat. 2020, 182, 1–8. [Google Scholar] [CrossRef]
- Bolland, M.J.; Grey, A.; Gamble, G.D.; Reid, I.R. Calcium and vitamin D supplements and health outcomes: A reanalysis of the Women’s Health Initiative (WHI) limited-access data set. Am. J. Clin. Nutr. 2011, 94, 1144–1149. [Google Scholar] [CrossRef]
- Smith, G.D.; Ebrahim, S. “Mendelian randomization”: Can genetic epidemiology contribute to understanding environmental determinants of disease? Int. J. Epidemiol. 2003, 32, 1–22. [Google Scholar] [CrossRef]
- Burgess, S.; Scott, R.A.; Timpson, N.J.; Davey Smith, G.; Thompson, S.G. EPIC-InterAct Consortium Using published data in Mendelian randomization: A blueprint for efficient identification of causal risk factors. Eur. J. Epidemiol. 2015, 30, 543–552. [Google Scholar] [CrossRef]
- Cornish, A.J.; Tomlinson, I.P.M.; Houlston, R.S. Mendelian randomisation: A powerful and inexpensive method for identifying and excluding non-genetic risk factors for colorectal cancer. Mol. Aspects Med. 2019, 69, 41–47. [Google Scholar] [CrossRef]
- Hemani, G.; Zheng, J.; Elsworth, B.; Wade, K.H.; Haberland, V.; Baird, D.; Laurin, C.; Burgess, S.; Bowden, J.; Langdon, R.; et al. The MR-Base platform supports systematic causal inference across the human phenome. eLife 2018, 7, e34408. [Google Scholar] [CrossRef] [PubMed]
- Glymour, M.M.; Tchetgen, E.J.; Robins, J.M. Credible Mendelian randomization studies: Approaches for evaluating the instrumental variable assumptions. Am. J. Epidemiol. 2012, 175, 332–339. [Google Scholar] [CrossRef] [PubMed]
- Burgess, S.; Thompson, S.G.; CRP CHD Genetics Collaboration. Avoiding bias from weak instruments in Mendelian randomization studies. Int. J. Epidemiol. 2011, 40, 755–764. [Google Scholar] [CrossRef] [PubMed]
- Bowden, J.; Davey Smith, G.; Burgess, S. Mendelian randomization with invalid instruments: Effect estimation and bias detection through Egger regression. Int. J. Epidemiol. 2015, 44, 512–525. [Google Scholar] [CrossRef] [PubMed]
- Bowden, J.; Davey Smith, G.; Haycock, P.C.; Burgess, S. Consistent Estimation in Mendelian Randomization with Some Invalid Instruments Using a Weighted Median Estimator. Genet. Epidemiol. 2016, 40, 304–314. [Google Scholar] [CrossRef]
- Jiang, X.; O’Reilly, P.F.; Aschard, H.; Hsu, Y.-H.; Richards, J.B.; Dupuis, J.; Ingelsson, E.; Karasik, D.; Pilz, S.; Berry, D.; et al. Genome-wide association study in 79,366 European-ancestry individuals informs the genetic architecture of 25-hydroxyvitamin D levels. Nat. Commun. 2018, 9, 260. [Google Scholar] [CrossRef]
- Revez, J.A.; Lin, T.; Qiao, Z.; Xue, A.; Holtz, Y.; Zhu, Z.; Zeng, J.; Wang, H.; Sidorenko, J.; Kemper, K.E.; et al. Genome-wide association study identifies 143 loci associated with 25 hydroxyvitamin D concentration. Nat. Commun. 2020, 11, 1647. [Google Scholar] [CrossRef]
- Manousaki, D.; Mitchell, R.; Dudding, T.; Haworth, S.; Harroud, A.; Forgetta, V.; Shah, R.L.; Luan, J.; Langenberg, C.; Timpson, N.J.; et al. Genome-wide Association Study for Vitamin D Levels Reveals 69 Independent Loci. Am. J. Hum. Genet. 2020, 106, 327–337. [Google Scholar] [CrossRef]
- Ong, J.-S.; MacGregor, S. Implementing MR-PRESSO and GCTA-GSMR for pleiotropy assessment in Mendelian randomization studies from a practitioner’s perspective. Genet. Epidemiol. 2019, 43, 609–616. [Google Scholar] [CrossRef]
- Ye, Y.; Yang, H.; Wang, Y.; Zhao, H. A comprehensive genetic and epidemiological association analysis of vitamin D with common diseases/traits in the UK Biobank. Genet. Epidemiol. 2021, 45, 24–35. [Google Scholar] [CrossRef]
- Dimitrakopoulou, V.I.; Tsilidis, K.K.; Haycock, P.C.; Dimou, N.L.; Al-Dabhani, K.; Martin, R.M.; Lewis, S.J.; Gunter, M.J.; Mondul, A.; Shui, I.M.; et al. Circulating vitamin D concentration and risk of seven cancers: Mendelian randomisation study. BMJ 2017, 359, j4761. [Google Scholar] [CrossRef]
- Chandler, P.D.; Tobias, D.K.; Wang, L.; Smith-Warner, S.A.; Chasman, D.I.; Rose, L.; Giovannucci, E.L.; Buring, J.E.; Ridker, P.M.; Cook, N.R.; et al. Association between Vitamin D Genetic Risk Score and Cancer Risk in a Large Cohort of U.S. Women. Nutrients 2018, 10, 55. [Google Scholar] [CrossRef]
- Ong, J.-S.; Gharahkhani, P.; An, J.; Law, M.H.; Whiteman, D.C.; Neale, R.E.; MacGregor, S. Vitamin D and overall cancer risk and cancer mortality: A Mendelian randomization study. Hum. Mol. Genet. 2018, 27, 4315–4322. [Google Scholar] [CrossRef]
- Wang, S.; Huo, D.; Kupfer, S.; Alleyne, D.; Ogundiran, T.O.; Ojengbede, O.; Zheng, W.; Nathanson, K.L.; Nemesure, B.; Ambs, S.; et al. Genetic variation in the vitamin D related pathway and breast cancer risk in women of African ancestry in the root consortium. Int. J. Cancer 2018, 142, 36–43. [Google Scholar] [CrossRef]
- Jiang, X.; Dimou, N.L.; Al-Dabhani, K.; Lewis, S.J.; Martin, R.M.; Haycock, P.C.; Gunter, M.J.; Key, T.J.; Eeles, R.A.; Muir, K.; et al. Circulating vitamin D concentrations and risk of breast and prostate cancer: A Mendelian randomization study. Int. J. Epidemiol. 2019, 48, 1416–1424. [Google Scholar] [CrossRef]
- Cheng, W.-W.; Wang, Z.-K.; Shangguan, H.-F.; Zhu, Q.; Zhang, H.-Y. Are vitamins relevant to cancer risks? A Mendelian randomization investigation. Nutrition 2020, 78, 110870. [Google Scholar] [CrossRef]
- Jiang, X.; Ge, T.; Chen, C.Y. The causal role of circulating vitamin D concentrations in human complex traits and diseases: A large-scale Mendelian randomization study. Sci. Rep. 2021, 11, 184. [Google Scholar] [CrossRef]
- Ong, J.-S.; Dixon-Suen, S.C.; Han, X.; An, J.; Esophageal Cancer Consortium; 23 and Me Research Team; Liyanage, U.; Dusingize, J.-C.; Schumacher, J.; Gockel, I.; et al. A comprehensive re-assessment of the association between vitamin D and cancer susceptibility using Mendelian randomization. Nat. Commun. 2021, 12, 246. [Google Scholar] [CrossRef]
- Theodoratou, E.; Palmer, T.; Zgaga, L.; Farrington, S.M.; McKeigue, P.; Din, F.V.N.; Tenesa, A.; Davey-Smith, G.; Dunlop, M.G.; Campbell, H. Instrumental variable estimation of the causal effect of plasma 25-hydroxy-vitamin D on colorectal cancer risk: A mendelian randomization analysis. PLoS ONE 2012, 7, e37662. [Google Scholar] [CrossRef]
- He, Y.; Timofeeva, M.; Farrington, S.M.; Vaughan-Shaw, P.; Svinti, V.; Walker, M.; Zgaga, L.; Meng, X.; Li, X.; Spiliopoulou, A.; et al. Exploring causality in the association between circulating 25-hydroxyvitamin D and colorectal cancer risk: A large Mendelian randomisation study. BMC Med. 2018, 16, 142. [Google Scholar] [CrossRef]
- Cornish, A.J.; Law, P.J.; Timofeeva, M.; Palin, K.; Farrington, S.M.; Palles, C.; Jenkins, M.A.; Casey, G.; Brenner, H.; Chang-Claude, J.; et al. Modifiable pathways for colorectal cancer: A mendelian randomisation analysis. Lancet Gastroenterol. Hepatol. 2020, 5, 55–62. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Zhang, X.; Timofeeva, M.; Farrington, S.M.; Li, X.; Xu, W.; Campbell, H.; Houlston, R.S.; Tomlinson, I.P.; Theodoratou, E.; et al. Bidirectional Mendelian randomisation analysis of the relationship between circulating vitamin D concentration and colorectal cancer risk. Int. J. Cancer 2022, 150, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Gharahkhani, P.; Chow, W.-H.; Gammon, M.D.; Liu, G.; Caldas, C.; Wu, A.H.; Ye, W.; Onstad, L.; Anderson, L.A.; et al. No Association Between Vitamin D Status and Risk of Barrett’s Esophagus or Esophageal Adenocarcinoma: A Mendelian Randomization Study. Clin. Gastroenterol. Hepatol. 2019, 17, 2227–2235.e1. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.; Cornish, A.J.; Sud, A.; Law, P.J.; Kinnersley, B.; Ostrom, Q.T.; Labreche, K.; Eckel-Passow, J.E.; Armstrong, G.N.; Claus, E.B.; et al. Mendelian randomisation study of the relationship between vitamin D and risk of glioma. Sci. Rep. 2018, 8, 2339. [Google Scholar] [CrossRef] [PubMed]
- Saunders, C.N.; Cornish, A.J.; Kinnersley, B.; Law, P.J.; Claus, E.B.; Il’yasova, D.; Schildkraut, J.; Barnholtz-Sloan, J.S.; Olson, S.H.; Bernstein, J.L.; et al. Lack of association between modifiable exposures and glioma risk: A Mendelian randomization analysis. Neuro-Oncol. 2020, 22, 207–215. [Google Scholar] [CrossRef]
- Liu, H.; Jiang, X.; Qiao, Q.; Chen, L.; Matsuda, K.; Jiang, G.; Yu, T.; Wang, Y.; Lin, H.; Liang, X.; et al. Association of circulating 25-Hydroxyvitamin D and its related genetic variations with hepatocellular carcinoma incidence and survival. Ann. Transl. Med. 2020, 8, 1080. [Google Scholar] [CrossRef]
- Sun, Y.-Q.; Brumpton, B.M.; Bonilla, C.; Lewis, S.J.; Burgess, S.; Skorpen, F.; Chen, Y.; Nilsen, T.I.L.; Romundstad, P.R.; Mai, X.-M. Serum 25-hydroxyvitamin D levels and risk of lung cancer and histologic types: A Mendelian randomisation analysis of the HUNT study. Eur. Respir. J. 2018, 51, 1800329. [Google Scholar] [CrossRef]
- Went, M.; Cornish, A.J.; Law, P.J.; Kinnersley, B.; van Duin, M.; Weinhold, N.; Foersti, A.; Hansson, M.; Sonneveld, P.; Goldschmidt, H.; et al. Search for multiple myeloma risk factors using Mendelian randomization. Blood Adv. 2020, 4, 2172–2179. [Google Scholar] [CrossRef]
- Dudding, T.; Johansson, M.; Thomas, S.J.; Brennan, P.; Martin, R.M.; Timpson, N.J. Assessing the causal association between 25-hydroxyvitamin D and the risk of oral and oropharyngeal cancer using Mendelian randomization. Int. J. Cancer 2018, 143, 1029–1036. [Google Scholar] [CrossRef]
- Ong, J.-S.; Cuellar-Partida, G.; Lu, Y.; Australian Ovarian Cancer Study; Fasching, P.A.; Hein, A.; Burghaus, S.; Beckmann, M.W.; Lambrechts, D.; Van Nieuwenhuysen, E.; et al. Association of vitamin D levels and risk of ovarian cancer: A Mendelian randomization study. Int. J. Epidemiol. 2016, 45, 1619–1630. [Google Scholar] [CrossRef]
- Yarmolinsky, J.; Relton, C.L.; Lophatananon, A.; Muir, K.; Menon, U.; Gentry-Maharaj, A.; Walther, A.; Zheng, J.; Fasching, P.; Zheng, W.; et al. Appraising the role of previously reported risk factors in epithelial ovarian cancer risk: A Mendelian randomization analysis. PLoS Med. 2019, 16, e1002893. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Gentiluomo, M.; Lorenzo-Bermejo, J.; Morelli, L.; Obazee, O.; Campa, D.; Canzian, F. Mendelian randomisation study of the effects of known and putative risk factors on pancreatic cancer. J. Med. Genet. 2020, 57, 820–828. [Google Scholar] [CrossRef] [PubMed]
- Kazmi, N.; Haycock, P.; Tsilidis, K.; Lynch, B.M.; Truong, T.; PRACTICAL Consortium; CRUK; BPC3; Martin, R.M.; Lewis, S.J.; et al. Appraising causal relationships of dietary, nutritional and physical-activity exposures with overall and aggressive prostate cancer: Two-sample Mendelian-randomization study based on 79148 prostate-cancer cases and 61106 controls. Int. J. Epidemiol. 2020, 49, 587–596. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Qin, J.; Berndt, S.I.; Albanes, D.; Deng, L.; Gail, M.H.; Yu, K. On Mendelian randomization analysis of case-control study. Biometrics 2020, 76, 380–391. [Google Scholar] [CrossRef] [PubMed]
- Gu, D.; Tang, M.; Wang, Y.; Cui, H.; Zhang, M.; Bai, Y.; Zeng, Z.; Tan, Y.; Wang, X.; Zhang, B. The Causal Relationships Between Extrinsic Exposures and Risk of Prostate Cancer: A Phenome-Wide Mendelian Randomization Study. Front. Oncol. 2022, 12, 829248. [Google Scholar] [CrossRef]
- Winsløw, U.C.; Nordestgaard, B.G.; Afzal, S. High plasma 25-hydroxyvitamin D and high risk of nonmelanoma skin cancer: A Mendelian randomization study of 97 849 individuals. Br. J. Dermatol. 2018, 178, 1388–1395. [Google Scholar] [CrossRef]
- Liyanage, U.E.; Law, M.H.; Melanoma Meta-analysis Consortium; Barrett, J.H.; Iles, M.M.; MacGregor, S. Is there a causal relationship between vitamin D and melanoma risk? A Mendelian randomization study. Br. J. Dermatol. 2020, 182, 97–103. [Google Scholar] [CrossRef]
- Yuan, S.; Baron, J.A.; Michaëlsson, K.; Larsson, S.C. Serum calcium and 25-hydroxyvitamin D in relation to longevity, cardiovascular disease and cancer: A Mendelian randomization study. NPJ Genom. Med. 2021, 6, 86. [Google Scholar] [CrossRef]
- Afzal, S.; Brøndum-Jacobsen, P.; Bojesen, S.E.; Nordestgaard, B.G. Genetically low vitamin D concentrations and increased mortality: Mendelian randomisation analysis in three large cohorts. BMJ 2014, 349, g6330. [Google Scholar] [CrossRef]
- Emerging Risk Factors Collaboration/EPIC-CVD/Vitamin D Studies Collaboration. Estimating dose-response relationships for vitamin D with coronary heart disease, stroke, and all-cause mortality: Observational and Mendelian randomisation analyses. Lancet Diabetes Endocrinol. 2021, 9, 837–846. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA. Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef]
- Charles, B.A.; Shriner, D.; Rotimi, C.N. Accounting for linkage disequilibrium in association analysis of diverse populations. Genet. Epidemiol. 2014, 38, 265–273. [Google Scholar] [CrossRef]
- Hutchinson, A.; Asimit, J.; Wallace, C. Fine-mapping genetic associations. Hum. Mol. Genet. 2020, 29, R81–R88. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.; Yu, K.; Stolzenberg-Solomon, R.; Simon, K.C.; McCullough, M.L.; Gallicchio, L.; Jacobs, E.J.; Ascherio, A.; Helzlsouer, K.; Jacobs, K.B.; et al. Genome-wide association study of circulating vitamin D levels. Hum. Mol. Genet. 2010, 19, 2739–2745. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.J.; Zhang, F.; Richards, J.B.; Kestenbaum, B.; van Meurs, J.B.; Berry, D.; Kiel, D.P.; Streeten, E.A.; Ohlsson, C.; Koller, D.L.; et al. Common genetic determinants of vitamin D insufficiency: A genome-wide association study. Lancet 2010, 376, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Vimaleswaran, K.S.; Cavadino, A.; Berry, D.J.; Jorde, R.; Dieffenbach, A.K.; Lu, C.; Alves, A.C.; Heerspink, H.J.L.; Tikkanen, E.; Eriksson, J.; et al. Association of vitamin D status with arterial blood pressure and hypertension risk: A mendelian randomisation study. Lancet Diabetes Endocrinol. 2014, 2, 719–729. [Google Scholar] [CrossRef] [PubMed]
- Mokry, L.E.; Ross, S.; Ahmad, O.S.; Forgetta, V.; Smith, G.D.; Goltzman, D.; Leong, A.; Greenwood, C.M.T.; Thanassoulis, G.; Richards, J.B. Vitamin D and Risk of Multiple Sclerosis: A Mendelian Randomization Study. PLoS Med. 2015, 12, e1001866. [Google Scholar] [CrossRef]
- Hiraki, L.T.; Qu, C.; Hutter, C.M.; Baron, J.A.; Berndt, S.I.; Bézieau, S.; Brenner, H.; Caan, B.J.; Casey, G.; Chang-Claude, J.; et al. Genetic predictors of circulating 25-hydroxyvitamin d and risk of colorectal cancer. Cancer Epidemiol. Biomark. Prev. 2013, 22, 2037–2046. [Google Scholar] [CrossRef]
- Padi, S.K.R.; Zhang, Q.; Rustum, Y.M.; Morrison, C.; Guo, B. MicroRNA-627 mediates the epigenetic mechanisms of vitamin D to suppress proliferation of human colorectal cancer cells and growth of xenograft tumors in mice. Gastroenterology 2013, 145, 437–446. [Google Scholar] [CrossRef]
- Williams, J.D.; Aggarwal, A.; Swami, S.; Krishnan, A.V.; Ji, L.; Albertelli, M.A.; Feldman, B.J. Tumor Autonomous Effects of Vitamin D Deficiency Promote Breast Cancer Metastasis. Endocrinology 2016, 157, 1341–1347. [Google Scholar] [CrossRef]
- Vaughan-Shaw, P.G.; Buijs, L.F.; Blackmur, J.P.; Theodoratou, E.; Zgaga, L.; Din, F.V.N.; Farrington, S.M.; Dunlop, M.G. The effect of vitamin D supplementation on survival in patients with colorectal cancer: Systematic review and meta-analysis of randomised controlled trials. Br. J. Cancer 2020, 123, 1705–1712. [Google Scholar] [CrossRef]
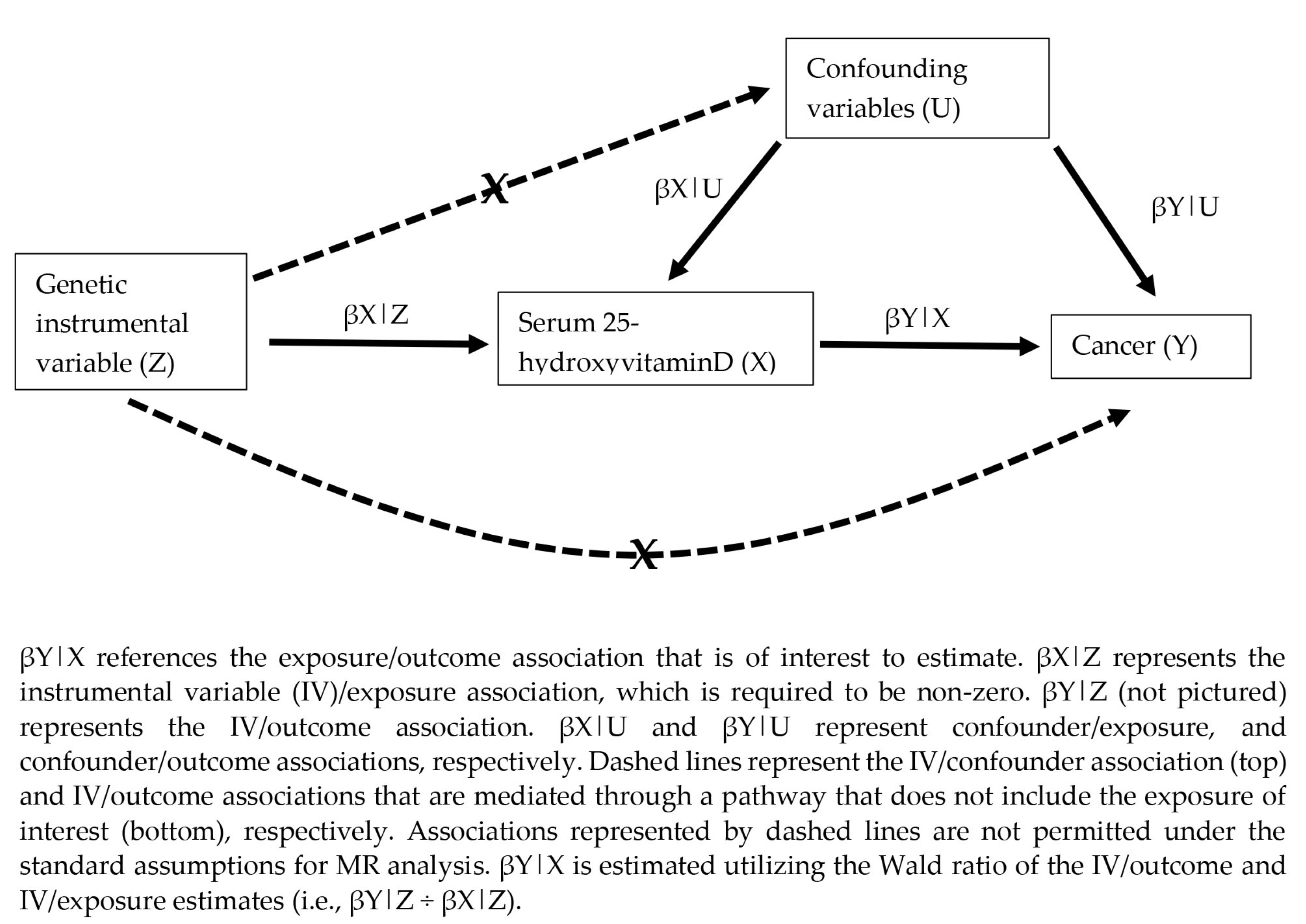
| Author, Year | Primary Ancestry | Cases | Controls | IV SNPs | Instrument Strength (PVE) a | Instrument Strength (F-Statistic) | OR (95% CI) | Per Unit Change | p-Trend |
|---|---|---|---|---|---|---|---|---|---|
| Bladder cancer | |||||||||
| Ye, 2021 [50] | European | NR | NR | 61 | 7.5% | NR | 1.00 (0.99–1.02) | 1 SD increase | 0.58 |
| Breast cancer | |||||||||
| Dimitrakopoulou, 2017 [51] | European | 15,748 | 18,084 | 4 | 1.0–2.5% | NR | 1.05 (0.89–1.24) | 25 nmol/L increase | 0.59 |
| Chandler, 2018 [52] | European | 1560 | N/A b | 5 | 2.6% | 48 | 1.14 (0.92–1.41) | 20 nmol/L increase | 0.22 |
| Ong, 2018 [53] | European | 11,703 | NR | 5 | 3.6% | NR | 0.94 (0.85–1.03) | 20 nmol/L increase | 0.19 |
| Wang, 2018 [54] | African | 1657 | 2029 | 4 | 1.0–2.5% | NR | 1.04 (0.97–1.11) | 1 SD increase | 0.23 |
| Jiang, 2019 [55] | European | 122,977 | 105,974 | 6 | 2.8% | NR | 1.02 (0.78–1.08) | 25 nmol/L increase | 0.47 |
| Cheng, 2020 [56] | European | 1145 | 1142 | 3 | NR | NR | 1.09 (0.55–2.15) | 1 SD increase | 0.80 |
| Jiang, 2021 [57] | European | 122,977 | 105,974 | 88 | 4.9% | NR | 1.02 (0.97–1.07) | 1 SD increase | 0.51 |
| Ong, 2021 [58] | European | 122,977 | 105,974 | 74 | 3.9% | NR | 1.03 (0.93–1.13) | 25 nmol/L increase | 0.60 |
| Ye, 2021 [50] | European | 122,977 | 105,974 | 91 | 7.5% | NR | 1.00 (0.98–1.02) | 1 SD increase | 0.95 |
| Colorectal cancer | |||||||||
| Theodoratou, 2012 [59] | European | 2001 | 2237 | 4 | 1.0–2.5% | 16.52 | 1.16 (0.60–2.23) | NR | >0.05 |
| Dimitrakopoulou, 2017 [51] | European | 11,488 | 11,679 | 4 | 1.0–2.5% | NR | 0.92 (0.67–1.10) | 25 nmol/L increase | 0.36 |
| European | 5100 | 4831 | 4 | 1.0–2.5% | NR | 1.04 (0.78–1.38) | 25 nmol/L increase | 0.81 | |
| Chandler, 2018 [52] | European | 329 | N/A b | 5 | 2.6% | 48 | 1.54 (0.96–2.47) | 20 nmol/L increase | 0.07 |
| He, 2018 [60] | European | 9940 | 22,848 | 6 | 2.8% | 46.0 | 1.03 (0.51–2.07) | 1 unit increase c | 0.93 |
| European | 17,716 | 40,095 | 6 | 2.8% | 46.0 | 0.91 (0.69–1.19) | 1 unit increase c | 0.48 | |
| Ong, 2018 [53] | European | 4442 | NR | 5 | 3.6% | NR | 0.94 (0.79–1.13) | 20 nmol/L increase | 0.52 |
| Cheng, 2020 [56] | Japanese | 6692 | 27,178 | 7 | NR | NR | 1.01 (0.99–1.03) | 1 SD increase | 0.42 |
| Cornish, 2020 [61] | European | 26,397 | 41,481 | 5 | 2.6% | 431.37 | 0.99 (0.90–1.09) | 1 SD increase | 0.89 |
| Ye, 2021 [50] | European | NR | NR | 61 | 7.5% | NR | 1.00 (0.99–1.02) | 1 SD increase | 0.71 |
| He, 2022 [62] | European | 26,397 | 41,181 | 110 | 7.5% | 25,241 | 0.97 (0.88–1.07) | 1 unit increase c | 0.57 |
| Endometrial cancer | |||||||||
| Ong, 2018 [53] | European | 1938 | NR | 5 | 3.6% | NR | 0.90 (0.72–1.13) | 20 nmol/L increase | 0.38 |
| Ong, 2021 [58] | European | 12,906 | 108,979 | 75 | 3.9% | NR | 0.93 (0.80–1.07) | 20 nmol/L increase | 0.32 |
| Esophageal cancer | |||||||||
| Dong, 2019 [63] | European | 4112 | 17,159 | 6 | 2.8% | NR | 0.68 (0.39–1.19) | 20 nmol/L increase | 0.18 |
| Ong, 2021 [58] | European | 4112 | 17,159 | 76 | 3.9% | NR | 0.97 (0.78–1.20) | 20 nmol/L increase | 0.76 |
| Glioma | |||||||||
| Takahashi, 2018 [64] | European | 12,488 | 18,169 | 4 | 1.0–2.5% | 12.57 | 1.21 (0.90–1.62) | NR | 0.20 |
| Saunders, 2020 [65] | European | 12,488 | 18,169 | 5 | 2.7% | 431.37 | 0.99 (0.86–1.15) | 1 SD increase | 0.93 |
| Kidney cancer | |||||||||
| Ong, 2018 [53] | European | 1012 | NR | 5 | 3.6% | NR | 1.21 (0.84–1.76) | 20 nmol/L increase | 0.31 |
| Ye, 2021 [50] | European | NR | NR | 62 | 7.5% | NR | 1.00 (0.99–1.01) | 1 SD increase | 0.96 |
| Leukemia | |||||||||
| Ye, 2021 [50] | European | NR | NR | 57 | 7.5% | NR | 1.01 (1.00–1.03) | 1 SD increase | 0.10 |
| Liver cancer (hepatocellular carcinoma) | |||||||||
| Liu, 2020 [66] | Chinese | 721 | 2890 | 6 | 2.8% | NR | 1.03 (0.31–3.47) | NR | >0.05 |
| Lung cancer | |||||||||
| Dimitrakopoulou, 2017 [51] | European | 12,537 | 17,285 | 4 | 1.0–2.5% | NR | 1.03 (0.87 to 1.23) | 25 nmol/L increase | 0.72 |
| Chandler, 2018 [52] | European | 330 | N/A b | 5 | 2.6% | 48 | 0.96 (0.55–1.68) | 20 nmol/L increase | 0.89 |
| Ong, 2018 [53] | European | 1863 | NR | 5 | 3.6% | NR | 1.04 (0.83–1.30) | 20 nmol/L increase | 0.73 |
| Sun, 2018 [67] One-sample MR | European | 676 | N/A b | 3 | 3.4% | 197 | 0.96 (0.54–1.69) | 25 nmol/L increase | 0.88 |
| Sun, 2018 [67] Two-sample MR | European | 676 | N/A b | 3 | 1.0–2.5% | 197 | 0.99 (0.88–1.12) | 10% increase | 0.85 |
| Jiang, 2021 [57] | European | 11,348 | 15,861 | 81 | 4.9% | NR | 1.13 (0.98–1.32) | 1 SD increase | 0.10 |
| Ong, 2021 [58] | European | 11,348 | 15,861 | 65 | 3.9% | NR | 0.94 (0.78–1.13) | 25 nmol/L increase | 0.50 |
| Ye, 2021 [50] | European | NR | NR | 82 | 7.5% | NR | 1.00 (0.97–1.03) | 1 SD increase | 0.84 |
| Lymphoid cancer | |||||||||
| Ong, 2018 [53] | European | 3576 | NR | 5 | 3.6% | NR | 1.10 (0.92–1.31) | 20 nmol/L increase | 0.29 |
| Multiple myeloma | |||||||||
| Went, 2020 [68] | European | 7717 | 29,304 | 5 | 2.7% | 431.37 | 1.08 (0.93–1.26) c | 1 SD increase | >0.05 |
| Neuroblastoma | |||||||||
| Dimitrakopoulou, 2017 [51] | European | 1627 | 3254 | 4 | 1.0–2.5% | NR | 0.76 (0.47–1.21) | 25 nmol/L increase | 0.24 |
| Ong, 2021 [58] | European | 1627 | 3254 | 26 | 3.9% | NR | 0.74 (0.42–1.29) | 25 nmol/L increase | 0.29 |
| Ye, 2021 [50] | European | 1627 | 3254 | 10 | 7.5% | NR | 0.92 (0.63–1.34) | 1 SD increase | 0.67 |
| Non-Hodgkin’s lymphoma | |||||||||
| Ye, 2021 [50] | European | NR | NR | 60 | 7.5% | NR | 1.00 (0.98–1.03) | 1 SD increase | 0.87 |
| Cancer of the oral cavity and pharynx | |||||||||
| Dudding, 2018 [69] | European | 5133 | 5984 | 5 | 2.0–3.5% | NR | 1.01 (0.74–1.40) | 1 SD increase | 0.93 |
| European | 585 | 336,523 | 5 | 2.0–3.5% | NR | 0.86 (0.58–1.27) | 1 SD increase | 0.44 | |
| Ovarian cancer | |||||||||
| Ong, 2016 [70] | European | 10,065 | 21,654 | 3 | 1.3% | NR | 0.79 (0.66–0.94) | 20 nmol/L increase | <0.05 |
| Dimitrakopoulou, 2017 [51] | European | 4369 | 9123 | 4 | 1.0–2.5% | NR | 1.12 (0.86–1.47) | 25 nmol/L increase | 0.40 |
| Ong, 2018 [53] | European | 1031 | NR | 5 | 3.6% | NR | 1.10 (0.80–1.51) | 20 nmol/L increase | 0.57 |
| Yarmolinsky, 2019 [71] | European | 25,509 | 40,941 | 5 | 2.6% | 423 | 1.02 (0.72–1.44) | 1 unit increase c | 0.93 |
| Ong, 2021 [58] | European | 25,509 | 40,941 | 76 | 3.9% | NR | 0.78 (0.63–0.96) | 1 unit increase c | 0.03 |
| Ye, 2021 [50] | Unclear | 18,174 | 26,134 | 104 | 7.5% | NR | 0.96 (0.93–0.99) | 1 SD increase | 0.02 |
| Pancreatic cancer | |||||||||
| Dimitrakopoulou, 2017 [51] | European | 1896 | 1939 | 4 | 1.0–2.5% | NR | 1.36 (0.81–2.27) | 25 nmol/L increase | 0.25 |
| Ong, 2018 [53] | European | 500 | NR | 5 | 3.6% | NR | 1.09 (0.63–1.88) | 20 nmol/L increase | 0.76 |
| Lu, 2020 [72] | European | 8769 | 7055 | 6 | 2.8% | NR | 1.13 (0.71–1.80) | 1 unit increase c | 0.60 |
| Ong, 2021 [58] | European | 1896 | 1939 | 27 | 3.9% | NR | 0.93 (0.46–1.92) | 25 nmol/L increase | 0.99 |
| Ye, 2021 [50] | European | 3851 | 3934 | 12 | 7.5% | NR | 0.92 (0.76–1.11) | 1 SD increase | 0.37 |
| Prostate cancer | |||||||||
| Dimitrakopoulou, 2017 [51] | Unclear | 22,898 | 23,054 | 4 | 1.0–2.5% | NR | 0.89 (0.77–1.02) | 25 nmol/L increase | 0.08 |
| European | 14,159 | 12,712 | 4 | 1.9% | NR | 1.08 (0.88–1.33) | 25 nmol/L increase | 0.47 | |
| Ong, 2018 [53] | European | 7532 | NR | 5 | 3.6% | NR | 0.91 (0.80–1.05) | 20 nmol/L increase | 0.19 |
| Jiang, 2019 [55] | European | 79,148 | 61,106 | 6 | 2.8% | NR | 1.00 (0.93–1.07) | 25 nmol/L increase | 0.99 |
| Cheng, 2020 [56] | European | NR | NR | 8 | NR | NR | 1.00 (0.99–1.00) | 1 SD increase | 0.37 |
| Kazmi, 2020 [73] | European | 15,167 | 58,308 | 4 | 2.4% | 253.15 | 1.00 (0.97–1.03) | 1 SD increase | 0.90 |
| Zhang, 2020 [74] | European | 4600 | 2941 | 3 | 1.0–2.5% | NR | 1.16 (0.86–1.57) | NR | 0.34 |
| Jiang, 2021 [57] | European | 79,194 | 61,112 | 51 | 4.9% | NR | 0.98 (0.91–1.05) | 1 SD increase | 0.57 |
| Ong, 2021 [58] | European | 79,148 | 61,106 | 75 | 3.9% | NR | 1.07 (0.89–1.29) | 25 nmol/L increase | 0.46 |
| Ye, 2021 [50] | European | 79,194 | 61,112 | 78 | 7.5% | NR | 0.99 (0.98–1.01) | 1 SD increase | 0.42 |
| Gu, 2022 [75] | European | 51,704 | 227,795 | 138 | 8.2% | 286.33 | 0.999 (0.995–1.003) | NR | 0.72 |
| Skin cancer (non-Melanoma) | |||||||||
| Winsløw, 2018 [76] | European | 8643 | N/A b | 4 | 1.0% | 314 | 1.11 (0.91–1.35) | 20 nmol/L increase | >0.05 |
| Skin cancer (squamous cell carcinoma) | |||||||||
| Cheng, 2020 [56] | European | NR | NR | 8 | NR | NR | 1.00 (0.99–1.00) | 1 SD increase | 0.47 |
| Ong, 2021 [58] | European | 7400 | 285,355 | 77 | 3.9% | NR | 1.02 (0.88–1.19) | 20 nmol/L increase | 0.77 |
| Skin cancer (basal cell carcinoma) | |||||||||
| Ong, 2021 [58] | European | 14,940 | 279,049 | 77 | 3.9% | NR | 1.18 (1.05–1.33) d | 20 nmol/L increase | 0.01 |
| Skin cancer (not specified) | |||||||||
| Ye, 2021 [50] | European | NR | NR | 52 | 7.5% | NR | 1.02 (0.99–1.04) | 1 SD increase | 0.15 |
| Skin cancer (melanoma) | |||||||||
| Ong, 2018 [53] | European | 2758 | NR | 5 | 3.6% | NR | 0.88 (0.71–1.10) | 20 nmol/L increase | 0.26 |
| Cheng, 2020 [56] | European | NR | NR | 8 | NR | NR | 1.00 (0.99–1.00) | 1 SD increase | 0.56 |
| Liyanage, 2020 [77] | European | 12,874 | 23,203 | 5 | 3.6% | NR | 0.94 (0.84–1.05) | 20 nmol/L increase | >0.05 |
| Ong, 2021 [58] | European | 15,990 | 26,409 | 69 | 3.9% | NR | 1.09 (0.92–1.28) | 20 nmol/L increase | 0.31 |
| Thyroid cancer | |||||||||
| Ye, 2021 [50] | European | NR | NR | 55 | 7.5% | NR | 0.99 (0.96–1.02) | 1 SD increase | 0.56 |
| Total cancer | |||||||||
| Chandler, 2018 [52] | European | 3985 | N/A b | 5 | 2.6% | 48 | 1.10 (0.96–1.25) | 20 nmol/L increase | 0.17 |
| Ong, 2018 [53] | European | 46,155 | 264,638 | 5 | 3.6% | NR | 0.97 (0.90–1.04) | 20 nmol/L increase | 0.40 |
| Ye, 2021 [50] | European | NR | NR | 54 | 7.5% | NR | 1.01 (1.00–1.02) | 1 SD increase | 0.19 |
| Yuan, 2021 [78] | European | 38,036 | 180,756 | 7 | 3.7% e | NR f | 1.01 (0.97–1.05) | 1 SD increase | 0.68 |
| European | 38,036 | 180,756 | 115 | 7.5% | NR f | 0.98 (0.93–1.04) | 1 SD increase | 0.50 | |
| Uterine cancer | |||||||||
| Ye, 2021 [50] | European | NR | NR | 59 | 7.5% | NR | 1.01 (0.99–1.03) | 1 SD increase | 0.30 |
| Author, Year | Primary Ancestry | Study Type | Sample Size | Cancer Deaths | IV SNPs | Instrument Strength (PVE) a | Instrument Strength (F-Statistic) | OR (95% CI) | Per Unit Change | p-Trend |
|---|---|---|---|---|---|---|---|---|---|---|
| Afzal, 2014 [79] | European | Prospective cohort | 95,766 | 2839 | 4 | 1.0% | NR | 0.70 (0.50–0.98) | 20 nmol/L increase | <0.05 |
| Chandler, 2018 [52] | European | Prospective cohort | 23,394 | 770 | 5 | 2.6% | 48 | 0.98 (0.73–1.32) | 20 nmol/L increase | 0.90 |
| Ong, 2018 [53] | European | Case-control | 277,340 b | 6998 | 5 | 3.6% | NR | 0.97 (0.84–1.11) | 20 nmol/L increase | >0.05 |
| Sofianopoulou, 2021 [80] | European | Prospective cohort | 386,406 | 12,804 | 3–21 c | 1.8–5.8% c | NR | 0.98 (0.93–1.02) | 10 nmol/L increase | 0.29 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lawler, T.; Warren Andersen, S. Serum 25-Hydroxyvitamin D and Cancer Risk: A Systematic Review of Mendelian Randomization Studies. Nutrients 2023, 15, 422. https://doi.org/10.3390/nu15020422
Lawler T, Warren Andersen S. Serum 25-Hydroxyvitamin D and Cancer Risk: A Systematic Review of Mendelian Randomization Studies. Nutrients. 2023; 15(2):422. https://doi.org/10.3390/nu15020422
Chicago/Turabian StyleLawler, Thomas, and Shaneda Warren Andersen. 2023. "Serum 25-Hydroxyvitamin D and Cancer Risk: A Systematic Review of Mendelian Randomization Studies" Nutrients 15, no. 2: 422. https://doi.org/10.3390/nu15020422
APA StyleLawler, T., & Warren Andersen, S. (2023). Serum 25-Hydroxyvitamin D and Cancer Risk: A Systematic Review of Mendelian Randomization Studies. Nutrients, 15(2), 422. https://doi.org/10.3390/nu15020422






