Rice Bran Supplementation Ameliorates Gut Dysbiosis and Muscle Atrophy in Ovariectomized Mice Fed with a High-Fat Diet
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Experiment Design
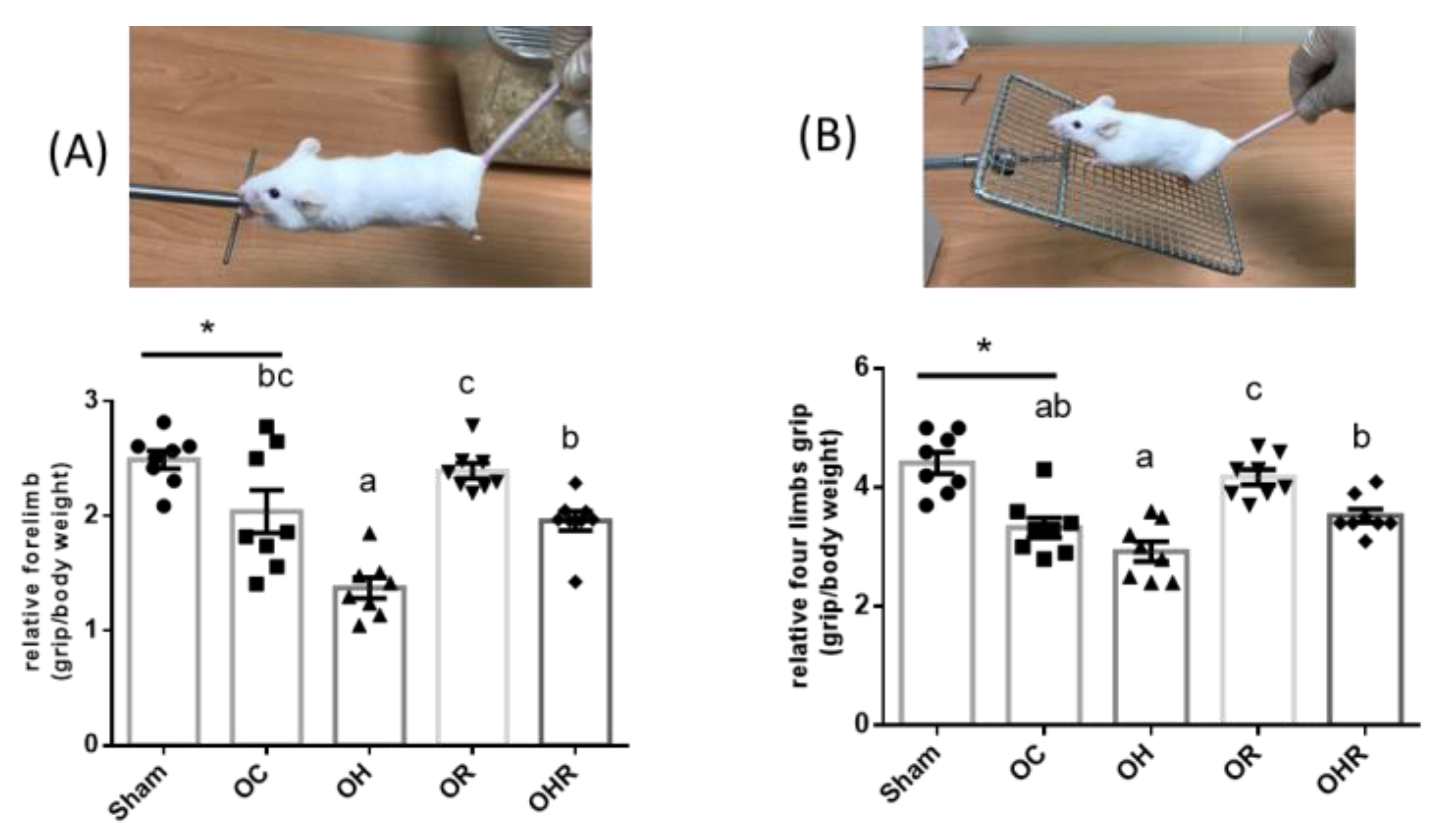
2.2. Grip Strength Measurement
2.3. Intraperitoneal Glucose Tolerance Test
2.4. Blood Biochemical Analysis
2.5. Cecum Microbiota Analysis
2.6. Gastrocnemius Muscle and Colon Gene Expression
2.7. Fecal SCFA Concentrations
2.8. Statistical Analysis
3. Results
3.1. Effect of Rice Bran on the General Characteristics of the Mice
3.1.1. Body Weight
3.1.2. Food Intake and Energy Consumption
3.2. Relative Tissue Weight of Mice
3.3. Blood Biochemical Profiles
3.3.1. Plasma Biochemical Data
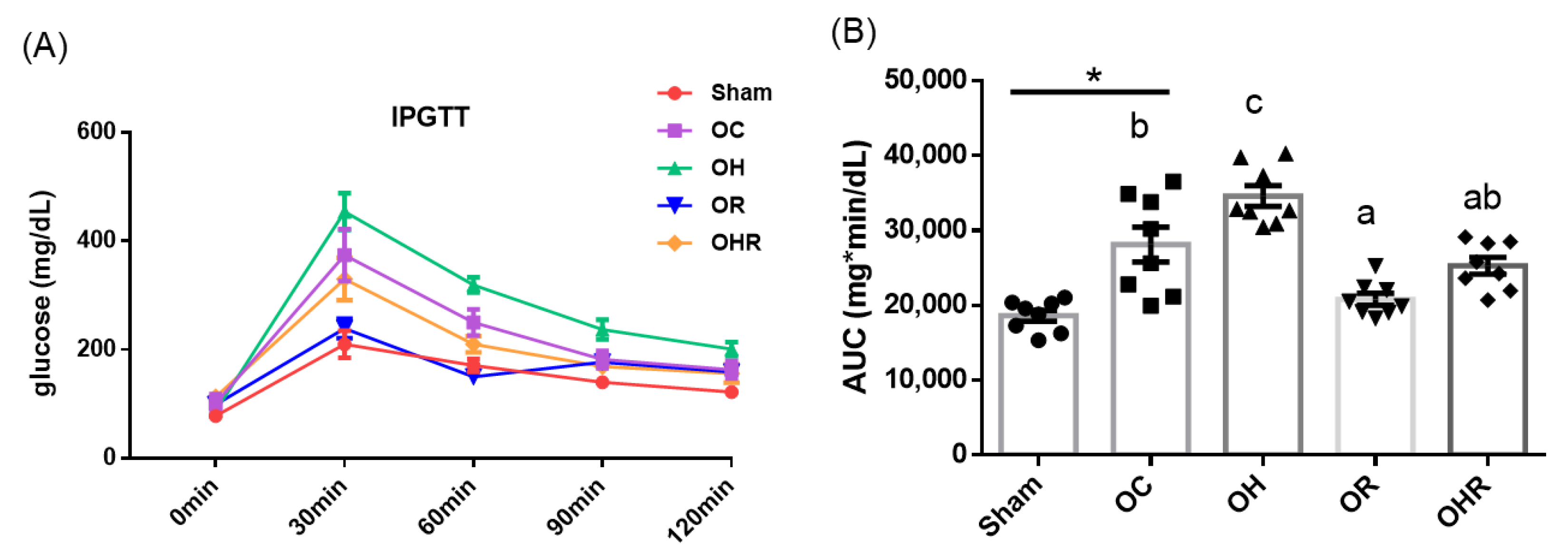
3.3.2. Results of IPGTT and Area under the Curve of Glucose
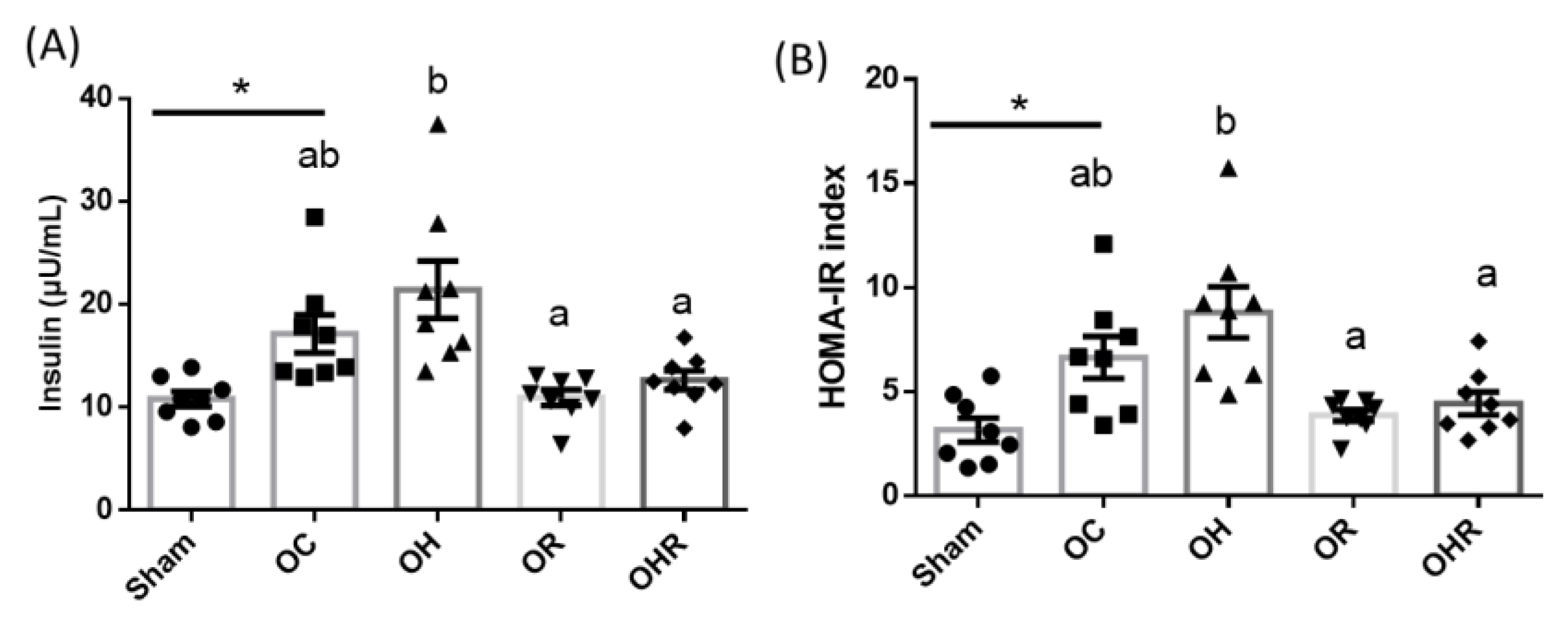
3.3.3. Insulin Levels and HOMA-IR Values
3.4. The Grip Strength Test of Mice
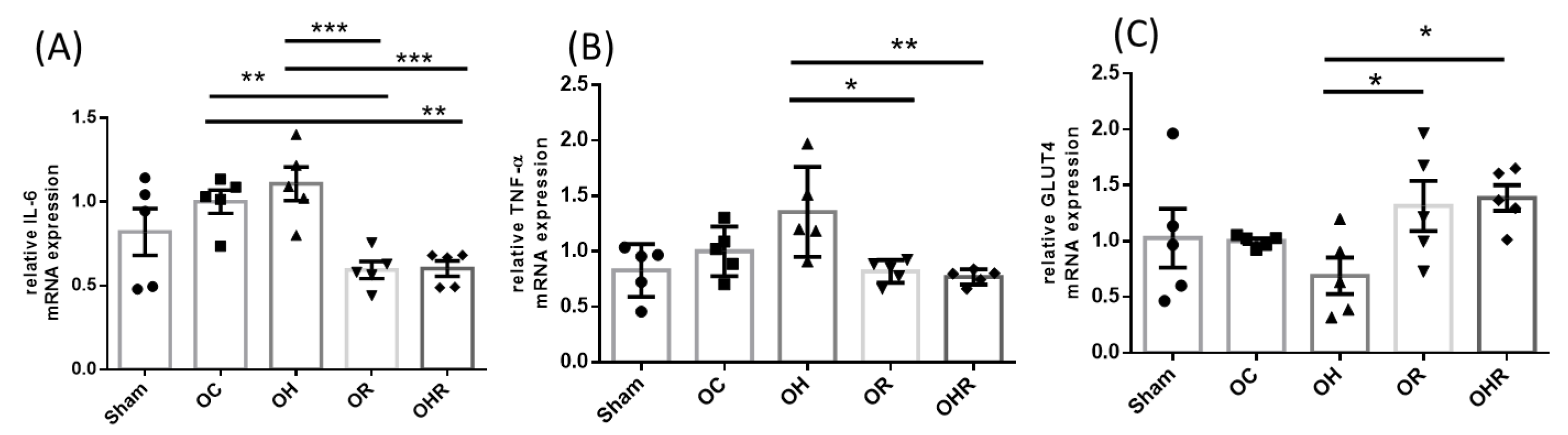
3.5. The Proinflammatory Cytokine and GLUT4 mRNA Expression in Gastrocnemius Muscle
3.6. Expression Levels of Biomarkers Associated with Synthesis and Breakdown of the Gastrocnemius Muscle of the Mice
3.7. Protein Expression Levels of Muscle Atrophy-Related Biomarkers
3.8. Gut Microbiota
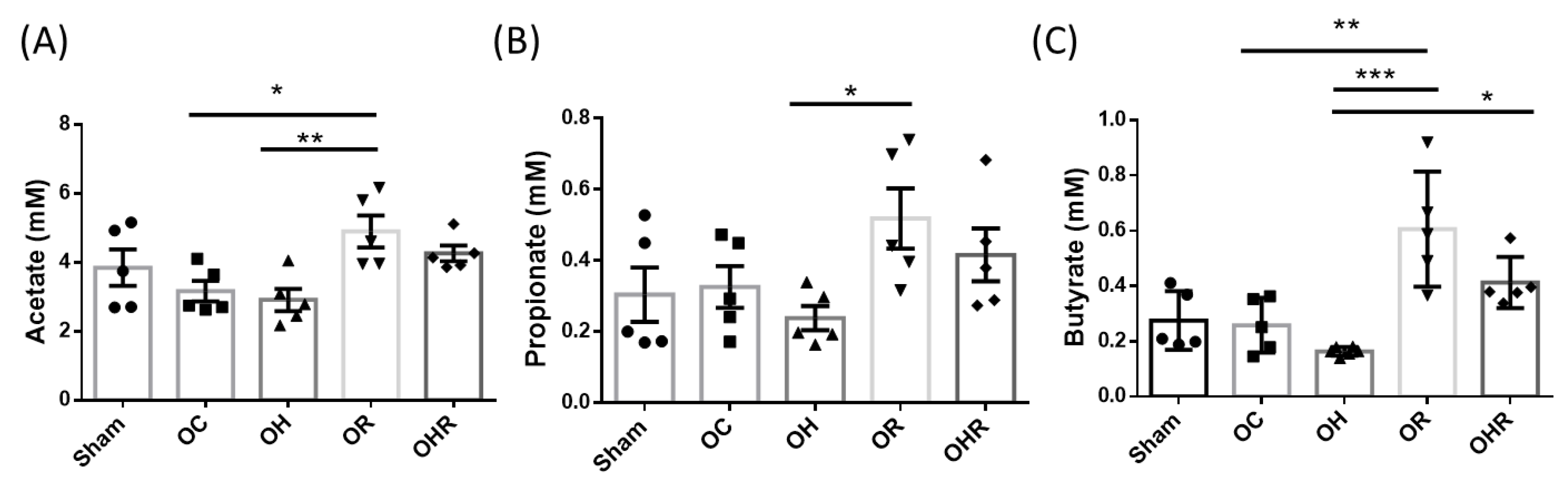
3.9. Short-Chain Fatty Acid Profiles in the Feces of the Study Groups
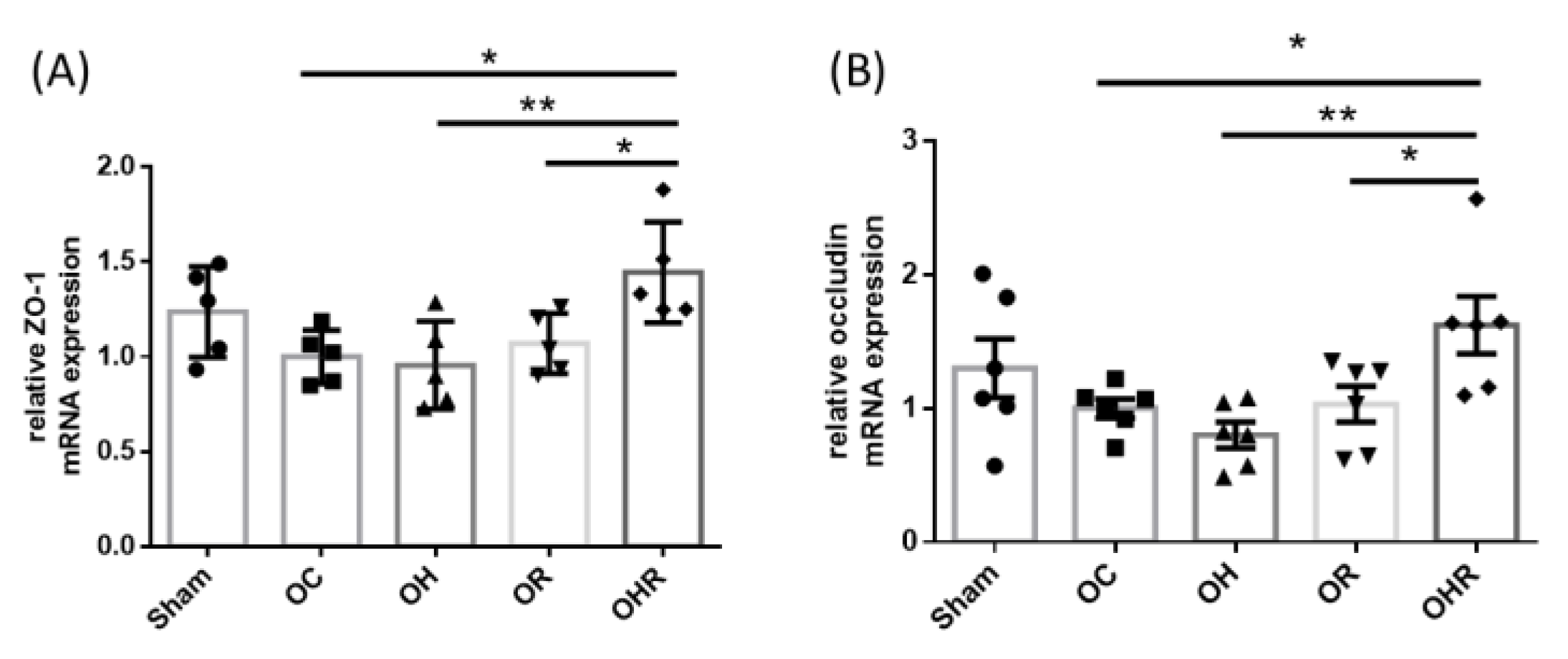
3.10. Expression Levels of Tight Junction Markers of the Colon of the Mice
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhu, D.; Chung, H.-F.; Pandeya, N.; Dobson, A.J.; Kuh, D.; Crawford, S.L.; Gold, E.B.; Avis, N.E.; Giles, G.G.; Bruinsma, F.; et al. Body mass index and age at natural menopause: An international pooled analysis of 11 prospective studies. Eur. J. Epidemiol. 2018, 33, 699–710. [Google Scholar] [CrossRef] [PubMed]
- Arthur, F.K.N.; Adu-Frimpong, M.; Osei-Yeboah, J.; Mensah, F.O.; Owusu, L. Prediction of metabolic syndrome among postmenopausal Ghanaian women using obesity and atherogenic markers. Lipids Health Dis. 2012, 11, 101. [Google Scholar] [CrossRef] [PubMed]
- Heianza, Y.; Arase, Y.; Kodama, S.; Hsieh, S.D.; Tsuji, H.; Saito, K.; Shimano, H.; Hara, S.; Sone, H. Effect of Postmenopausal Status and Age at Menopause on Type 2 Diabetes and Prediabetes in Japanese Individuals: Toranomon Hospital Health Management Center Study 17 (TOPICS 17). Diabetes Care 2013, 36, 4007–4014. [Google Scholar] [CrossRef]
- Davis, S.R.; Castelo-Branco, C.; Chedraui, P.; Lumsden, M.A.; Nappi, R.E.; Shah, D.; Villaseca, P. Understanding weight gain at menopause. Climacteric 2012, 15, 419–429. [Google Scholar] [CrossRef] [PubMed]
- Grosicki, G.J.; Fielding, R.A.; Lustgarten, M.S. Gut Microbiota Contribute to Age-Related Changes in Skeletal Muscle Size, Composition, and Function: Biological Basis for a Gut-Muscle Axis. Calcif. Tissue Int. 2018, 102, 433–442. [Google Scholar] [CrossRef]
- Yoshida, T.; Delafontaine, P. Mechanisms of IGF-1-Mediated Regulation of Skeletal Muscle Hypertrophy and Atrophy. Cells 2020, 9, 1970. [Google Scholar] [CrossRef]
- Trendelenburg, A.U.; Meyer, A.; Rohner, D.; Boyle, J.; Hatakeyama, S.; Glass, D.J. Myostatin reduces Akt/TORC1/p70S6K signaling, inhibiting myoblast differentiation and myotube size. Am. J. Physiol. Physiol. 2009, 296, C1258–C1270. [Google Scholar] [CrossRef]
- Tazawa, R.; Uchida, K.; Fujimaki, H.; Miyagi, M.; Inoue, G.; Sekiguchi, H.; Murata, K.; Takata, K.; Kawakubo, A.; Takaso, M. Elevated leptin levels induce inflammation through IL-6 in skeletal muscle of aged female rats. BMC Musculoskelet. Disord. 2019, 20, 199. [Google Scholar] [CrossRef]
- Haddad, F.; Zaldivar, F.; Cooper, D.M.; Adams, G.R. IL-6-induced skeletal muscle atrophy. J. Appl. Physiol. 2005, 98, 911–917. [Google Scholar] [CrossRef]
- Chew, W.; Lim, Y.P.; Lim, W.S.; Chambers, E.S.; Frost, G.; Wong, S.H.; Ali, Y. Gut-muscle crosstalk. A perspective on influence of microbes on muscle function. Front. Med. 2022, 9, 1065365. [Google Scholar] [CrossRef]
- Ticinesi, A.; Lauretani, F.; Milani, C.; Nouvenne, A.; Tana, C.; Del Rio, D.; Maggio, M.; Ventura, M.; Meschi, T. Aging Gut Microbiota at the Cross-Road between Nutrition, Physical Frailty, and Sarcopenia: Is There a Gut–Muscle Axis? Nutrition 2017, 9, 1303. [Google Scholar] [CrossRef]
- Ticinesi, A.; Nouvenne, A.; Cerundolo, N.; Catania, P.; Prati, B.; Tana, C.; Meschi, T. Gut Microbiota, Muscle Mass and Function in Aging: A Focus on Physical Frailty and Sarcopenia. Nutrients 2019, 11, 1633. [Google Scholar] [CrossRef]
- Chelakkot, C.; Ghim, J.; Ryu, S.H. Mechanisms regulating intestinal barrier integrity and its pathological implications. Exp. Mol. Med. 2018, 50, 1–9. [Google Scholar] [CrossRef]
- Tang, G.; Du, Y.; Guan, H.; Jia, J.; Zhu, N.; Shi, Y.; Rong, S.; Yuan, W. Butyrate ameliorates skeletal muscle atrophy in diabetic nephropathy by enhancing gut barrier function and FFA2-mediated PI3K/Akt/mTOR signals. Br. J. Pharmacol. 2022, 179, 159–178. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Zhang, P.; Shen, L.; Niu, L.; Tan, Y.; Chen, L.; Zhao, Y.; Bai, L.; Hao, X.; Li, X.; et al. Short-Chain Fatty Acids and Their Association with Signalling Pathways in Inflammation, Glucose and Lipid Metabolism. Int. J. Mol. Sci. 2020, 21, 6356. [Google Scholar] [CrossRef] [PubMed]
- Spaggiari, M.; Dall’Asta, C.; Galaverna, G.; del Castillo Bilbao, M.D. Rice Bran By-Product: From Valorization Strategies to Nutritional Perspectives. Foods 2021, 10, 85. [Google Scholar] [CrossRef]
- Cheng, H.-H.; Huang, H.-Y.; Chen, Y.-Y.; Huang, C.-L.; Chang, C.-J.; Chen, H.-L.; Lai, M.-H. Ameliorative Effects of Stabilized Rice Bran on Type 2 Diabetes Patients. Ann. Nutr. Metab. 2010, 56, 45–51. [Google Scholar] [CrossRef]
- Sivamaruthi, B.S.; Alagarsamy, K.; Thangaleela, S.; Bharathi, M.; Kesika, P.; Chaiyasut, C. Composition, Microbiota, Mechanisms, and Anti-Obesity Properties of Rice Bran. Foods 2023, 12, 1300. [Google Scholar] [CrossRef]
- Kim, S.P.; Kang, M.Y.; Nam, S.H.; Friedman, M. Dietary rice bran component γ-oryzanol inhibits tumor growth in tumor-bearing mice. Mol. Nutr. Food Res. 2012, 56, 935–944. [Google Scholar] [CrossRef]
- Suzuki, Y.; Yasui, T.; Matsukura, U.; Terao, J. Oxidative Stability of Bran Lipids from Rice Variety [Oryza sativa (L.)] Lacking Lipoxygenase-3 in Seeds. J. Agric. Food Chem. 1996, 44, 3479–3483. [Google Scholar] [CrossRef]
- Xu, Z.; Godber, J.S. Purification and Identification of Components of γ-Oryzanol in Rice Bran Oil. J. Agric. Food Chem. 1999, 47, 2724–2728. [Google Scholar] [CrossRef]
- Lotti, C.; Rubert, J.; Fava, F.; Tuohy, K.; Mattivi, F.; Vrhovsek, U. Development of a fast and cost-effective gas chromatography–mass spectrometry method for the quantification of short-chain and medium-chain fatty acids in human biofluids. Anal. Bioanal. Chem. 2017, 409, 5555–5567. [Google Scholar] [CrossRef]
- Speakman, J.R. Use of high-fat diets to study rodent obesity as a model of human obesity. Int. J. Obes. 2019, 43, 1491–1492. [Google Scholar] [CrossRef]
- Lee, I.S.; Shin, G.; Choue, R. Shifts in Diet from High Fat to High Carbohydrate Improved Levels of Adipokines and Pro-inflammatory Cytokines in Mice Fed a High-fat Diet. Endocr. J. 2010, 57, 39–50. [Google Scholar] [CrossRef]
- Wong, C.K.; Botta, A.; Pither, J.; Dai, C.; Gibson, W.T.; Ghosh, S. A high-fat diet rich in corn oil reduces spontaneous locomotor activity and induces insulin resistance in mice. J. Nutr. Biochem. 2015, 26, 319–326. [Google Scholar] [CrossRef]
- Bader, J.; Carson, M.; Enos, R.; Velazquez, K.; Sougiannis, A.; Singh, U.; Becker, W.; Nagarkatti, M.; Fan, D.; Murphy, A. High-fat diet-fed ovariectomized mice are susceptible to accelerated subcutaneous tumor growth potentially through adipose tissue inflammation, local insulin-like growth factor release, and tumor associated macrophages. Oncotarget 2020, 11, 4554–4569. [Google Scholar] [CrossRef]
- Kuryłowicz, A. Estrogens in Adipose Tissue Physiology and Obesity-Related Dysfunction. Biomedicines 2023, 11, 690. [Google Scholar] [CrossRef] [PubMed]
- Kitajima, Y.; Doi, H.; Ono, Y.; Urata, Y.; Goto, S.; Kitajima, M.; Miura, K.; Li, T.-S.; Masuzaki, H. Estrogen deficiency heterogeneously affects tissue specific stem cells in mice. Sci. Rep. 2015, 5, 12861. [Google Scholar] [CrossRef] [PubMed]
- Sitnick, M.; Foley, A.M.; Brown, M.; Spangenburg, E.E. Ovariectomy prevents the recovery of atrophied gastrocnemius skeletal muscle mass. J. Appl. Physiol. 2006, 100, 286–293. [Google Scholar] [CrossRef] [PubMed]
- Walsh, M.E.; Bhattacharya, A.; Sataranatarajan, K.; Qaisar, R.; Sloane, L.; Rahman, M.; Kinter, M.; Van Remmen, H. The histone deacetylase inhibitor butyrate improves metabolism and reduces muscle atrophy during aging. Aging Cell 2015, 14, 957–970. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Ju, X.; Chen, W.; Yuan, J.; Wang, Z.; Aluko, R.E.; He, R. Rice bran attenuated obesity via alleviating dyslipidemia, browning of white adipocytes and modulating gut microbiota in high-fat diet-induced obese mice. Food Funct. 2020, 11, 2406–2417. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Lin, Q.; Yang, T.; Liang, Y.; Nie, Y.; Luo, Y.; Shen, J.; Fu, X.; Tang, Y.; Luo, F. Oryzanol Modifies High Fat Diet-Induced Obesity, Liver Gene Expression Profile, and Inflammation Response in Mice. J. Agric. Food Chem. 2017, 65, 8374–8385. [Google Scholar] [CrossRef]
- Saha, K.R.; Rahman, M.M.; Paul, A.R.; Das, S.; Haque, S.; Jafrin, W.; Mia, A.R. Changes in lipid profile of postmenopausal women. Mymensingh Med. J. 2013, 22, 706–711. [Google Scholar]
- Kim, M.J.; Park, J.-H.; Kwon, D.Y.; Yang, H.J.; Kim, D.S.; Kang, S.; Shin, B.K.; Moon, N.R.; Song, B.-S.; Kim, J.-H.; et al. The supplementation of Korean mistletoe water extracts reduces hot flushes, dyslipidemia, hepatic steatosis, and muscle loss in ovariectomized rats. Exp. Biol. Med. 2015, 240, 477–487. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.-C.; Huang, W.-C.; Ng, X.E.; Lee, M.-C.; Hsu, Y.-J.; Huang, C.-C.; Wu, H.-H.; Yeh, C.-L.; Shirakawa, H.; Budijanto, S.; et al. Rice Bran Reduces Weight Gain and Modulates Lipid Metabolism in Rats with High-Energy-Diet-Induced Obesity. Nutrients 2019, 11, 2033. [Google Scholar] [CrossRef] [PubMed]
- Wang, O.; Liu, J.; Cheng, Q.; Guo, X.; Wang, Y.; Zhao, L.; Zhou, F.; Ji, B. Effects of Ferulic Acid and γ-Oryzanol on High-Fat and High-Fructose Diet-Induced Metabolic Syndrome in Rats. PLoS ONE 2015, 10, e0118135. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues Silva, C.; Dutra de Oliveira, J.E.; de Souza, R.A.; Silva, H.C. Effect of a rice bran fiber diet on serum glucose levels of diabetic patients in Brazil. Arch. Latinoam. Nutr. 2005, 55, 23–27. [Google Scholar]
- Ardiansyah; Shirakawa, H.; Koseki, T.; Ohinata, K.; Hashizume, K.; Komai, M. Rice Bran Fractions Improve Blood Pressure, Lipid Profile, and Glucose Metabolism in Stroke-Prone Spontaneously Hypertensive Rats. J. Agric. Food Chem. 2006, 54, 1914–1920. [Google Scholar] [CrossRef]
- Lynch, N.A.; Metter, E.J.; Lindle, R.S.; Fozard, J.L.; Tobin, J.D.; Roy, T.A.; Fleg, J.L.; Hurley, B.F. Muscle quality. I. Age-associated differences between arm and leg muscle groups. J. Appl. Physiol. 1999, 86, 188–194. [Google Scholar] [CrossRef]
- Samson, M.M.; Meeuwsen, I.B.; Crowe, A.; Dessens, J.A.; Duursma, S.A.; Verhaar, H.J. Relationships between physical performance measures, age, height and body weight in healthy adults. Age Ageing 2000, 29, 235–242. [Google Scholar] [CrossRef]
- Kitajima, Y.; Ono, Y. Estrogens maintain skeletal muscle and satellite cell functions. J. Endocrinol. 2016, 229, 267–275. [Google Scholar] [CrossRef]
- Counts, B.R.; Fix, D.K.; Hetzler, K.L.; Carson, J.A. The Effect of Estradiol Administration on Muscle Mass Loss and Cachexia Progression in Female ApcMin/+ Mice. Front. Endocrinol. 2019, 10, 720. [Google Scholar] [CrossRef]
- Bodine, S.C.; Baehr, L.M. Skeletal muscle atrophy and the E3 ubiquitin ligases MuRF1 and MAFbx/atrogin-1. Am. J. Physiol. Metab. 2014, 307, E469–E484. [Google Scholar] [CrossRef]
- Abrigo, J.; Rivera, J.C.; Aravena, J.; Cabrera, D.; Simon, F.; Ezquer, F.; Ezquer, M.; Cabello-Verrugio, C. High Fat Diet-Induced Skeletal Muscle Wasting Is Decreased by Mesenchymal Stem Cells Administration: Implications on Oxidative Stress, Ubiquitin Proteasome Pathway Activation, and Myonuclear Apoptosis. Oxidative Med. Cell. Longev. 2016, 2016, 9047821. [Google Scholar] [CrossRef]
- Kim, H.J.; Lee, W.J. Low-intensity aerobic exercise training: Inhibition of skeletal muscle atrophy in high-fat-diet-induced ovariectomized rats. J. Exerc. Nutr. Biochem. 2017, 21, 19–25. [Google Scholar] [CrossRef]
- Adhikary, S.; Kothari, P.; Choudhary, D.; Tripathi, A.K.; Trivedi, R. Glucocorticoid aggravates bone micro-architecture deterioration and skeletal muscle atrophy in mice fed on high-fat diet. Steroids 2019, 149, 108416. [Google Scholar] [CrossRef]
- Le, N.H.; Kim, C.-S.; Park, T.; Park, J.H.Y.; Sung, M.-K.; Lee, D.G.; Hong, S.-M.; Choe, S.-Y.; Goto, T.; Kawada, T.; et al. Quercetin Protects against Obesity-Induced Skeletal Muscle Inflammation and Atrophy. Mediat. Inflamm. 2014, 2014, 834294. [Google Scholar] [CrossRef] [PubMed]
- Jariwalla, R.J. Rice-bran products: Phytonutrients with potential applications in preventive and clinical medicine. Drugs Exp. Clin. Res. 2001, 27, 17–26. [Google Scholar]
- Mathew, S.; Abraham, T.E. Ferulic Acid: An Antioxidant Found Naturally in Plant Cell Walls and Feruloyl Esterases Involved in its Release and Their Applications. Crit. Rev. Biotechnol. 2004, 24, 59–83. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Malhotra, S.; Kumar, A. Nuclear factor-kappa B signaling in skeletal muscle atrophy. J. Mol. Med. 2008, 86, 1113–1126. [Google Scholar] [CrossRef]
- Sonnenburg, J.L.; Bäckhed, F. Diet–microbiota interactions as moderators of human metabolism. Nature 2016, 535, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Paul, S.; Kundu, P. NF-κB Regulation by Gut Microbiota Decides Homeostasis or Disease Outcome During Ageing. Front. Cell Dev. Biol. 2022, 10, 874940. [Google Scholar] [CrossRef]
- Everard, A.; Belzer, C.; Geurts, L.; Ouwerkerk, J.P.; Druart, C.; Bindels, L.B.; Guiot, Y.; Derrien, M.; Muccioli, G.G.; Delzenne, N.M.; et al. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc. Natl. Acad. Sci. USA 2013, 110, 9066–9071. [Google Scholar] [CrossRef] [PubMed]
- Dao, M.C.; Everard, A.; Aron-Wisnewsky, J.; Sokolovska, N.; Prifti, E.; Verger, E.O.; Kayser, B.D.; Levenez, F.; Chilloux, J.; Hoyles, L. Akkermansia muciniphila and improved metabolic health during a dietary intervention in obesity: Relationship with gut microbiome richness and ecology. Gut 2016, 65, 426–436. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wu, Y.; Zhuang, L.; Chen, X.; Min, H.; Song, S.; Liang, Q.; Li, A.-D.; Gao, Q. Puerarin prevents high-fat diet-induced obesity by enriching Akkermansia muciniphila in the gut microbiota of mice. PLoS ONE 2019, 14, e0218490. [Google Scholar] [CrossRef] [PubMed]
- Plovier, H.; Everard, A.; Druart, C.; Depommier, C.; Van Hul, M.; Geurts, L.; Chilloux, J.; Ottman, N.; Duparc, T.; Lichtenstein, L.; et al. A purified membrane protein from Akkermansia muciniphila or the pasteurized bacterium improves metabolism in obese and diabetic mice. Nat. Med. 2017, 23, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Alsanea, S.; Gao, M.; Liu, D. Phloretin Prevents High-Fat Diet-Induced Obesity and Improves Metabolic Homeostasis. AAPS J. 2017, 19, 797–805. [Google Scholar] [CrossRef]
- Rohr, M.W.; Narasimhulu, C.A.; Rudeski-Rohr, T.A.; Parthasarathy, S. Negative Effects of a High-Fat Diet on Intestinal Permeability: A Review. Adv. Nutr. 2020, 11, 77–91. [Google Scholar] [CrossRef] [PubMed]
- Peng, M.; Yi, W.; Murong, M.; Peng, N.; Tong, H.; Jiang, M.; Jin, D.; Peng, S.; Liang, W.; Quan, J.; et al. Akkermansia muciniphila improves heat stress-impaired intestinal barrier function by modulating HSP27 in Caco-2 cells. Microb. Pathog. 2023, 177, 106028. [Google Scholar] [CrossRef]
- Yao, W.; Gong, Y.; Li, L.; Hu, X.; You, L. The effects of dietary fibers from rice bran and wheat bran on gut microbiota: An overview. Food Chem. X 2022, 13, 100252. [Google Scholar] [CrossRef]
- Depommier, C.; Everard, A.; Druart, C.; Plovier, H.; Van Hul, M.; Vieira-Silva, S.; Falony, G.; Raes, J.; Maiter, D.; Delzenne, N.M.; et al. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: A proof-of-concept exploratory study. Nat. Med. 2019, 25, 1096–1103. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, J.C.; Matheus, V.A.; Oliveira, R.B.; Tada, S.F.S.; Collares-Buzato, C.B. High-Fat Diet Induces Disruption of the Tight Junction-Mediated Paracellular Barrier in the Proximal Small Intestine Before the Onset of Type 2 Diabetes and Endotoxemia. Dig. Dis. Sci. 2021, 66, 3359–3374. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.Y.-H.; Hsiao, A.W.-T.; Wong, N.; Chen, Y.-F.; Lee, C.-W.; Lee, W.Y.W. Is dexamethasone-induced muscle atrophy an alternative model for naturally aged sarcopenia model? J. Orthop. Transl. 2023, 39, 12–20. [Google Scholar] [CrossRef] [PubMed]
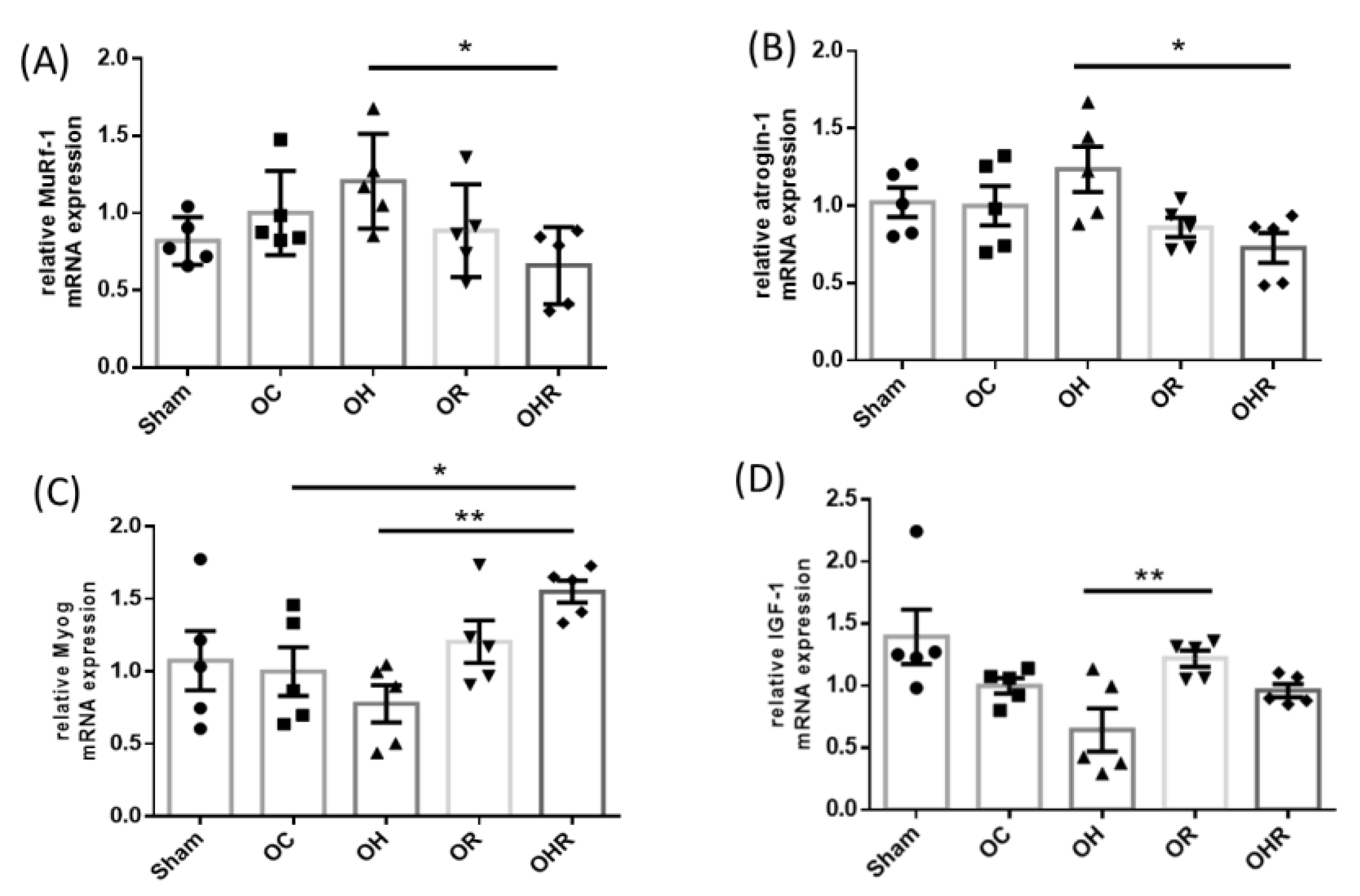
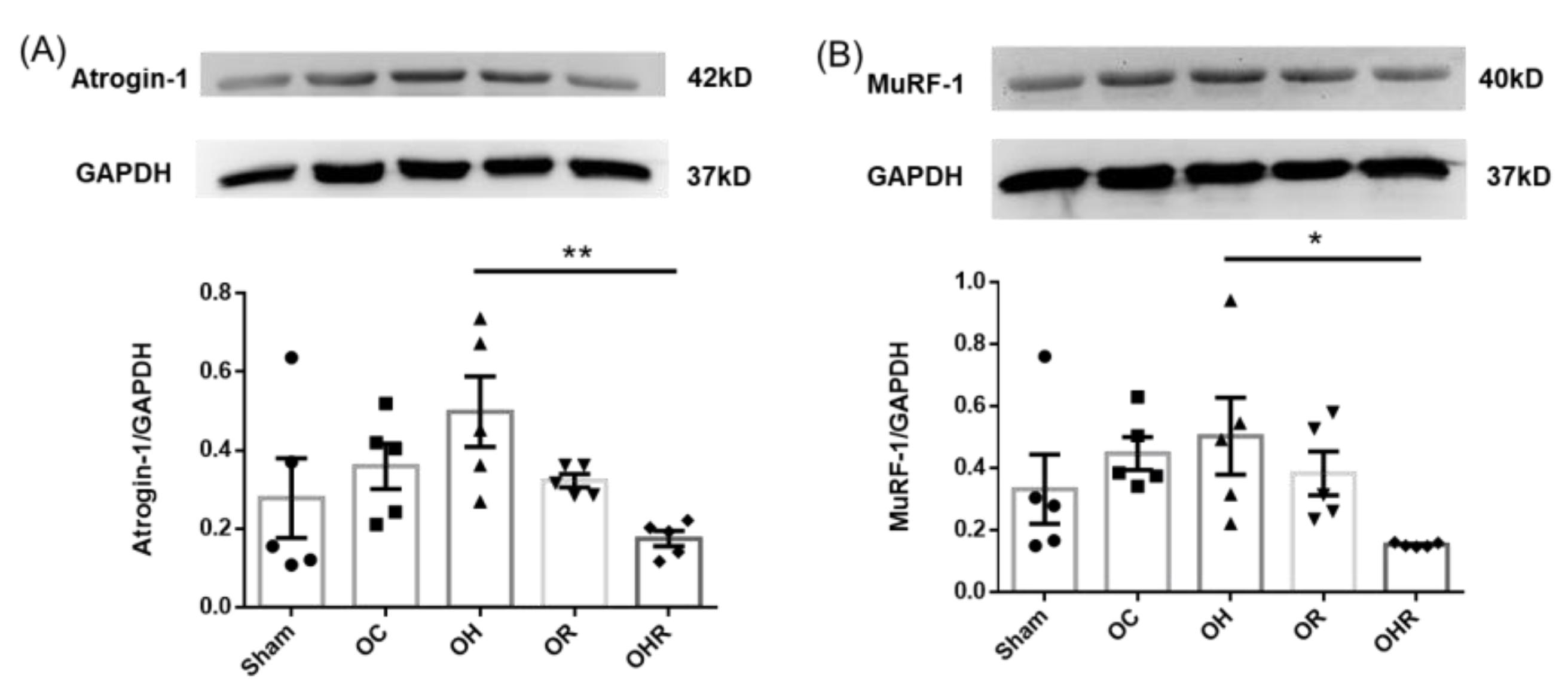
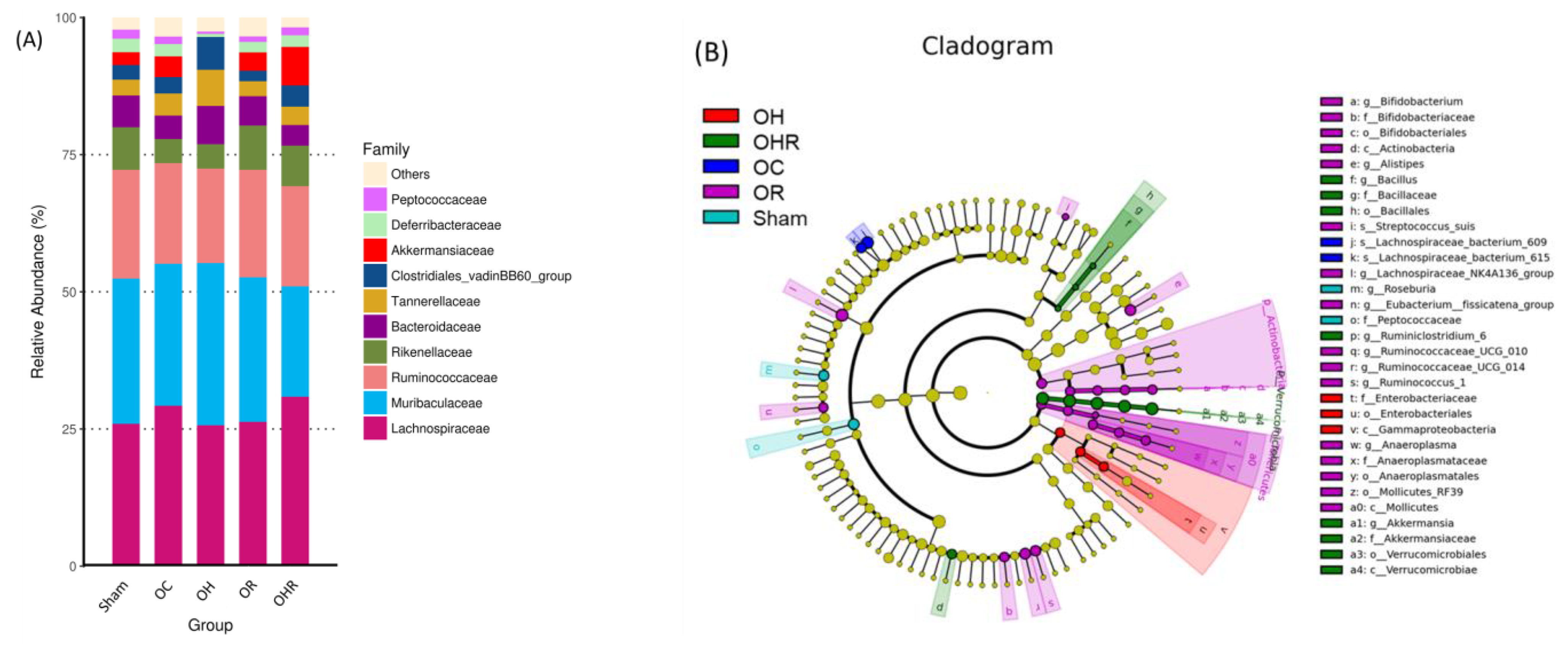
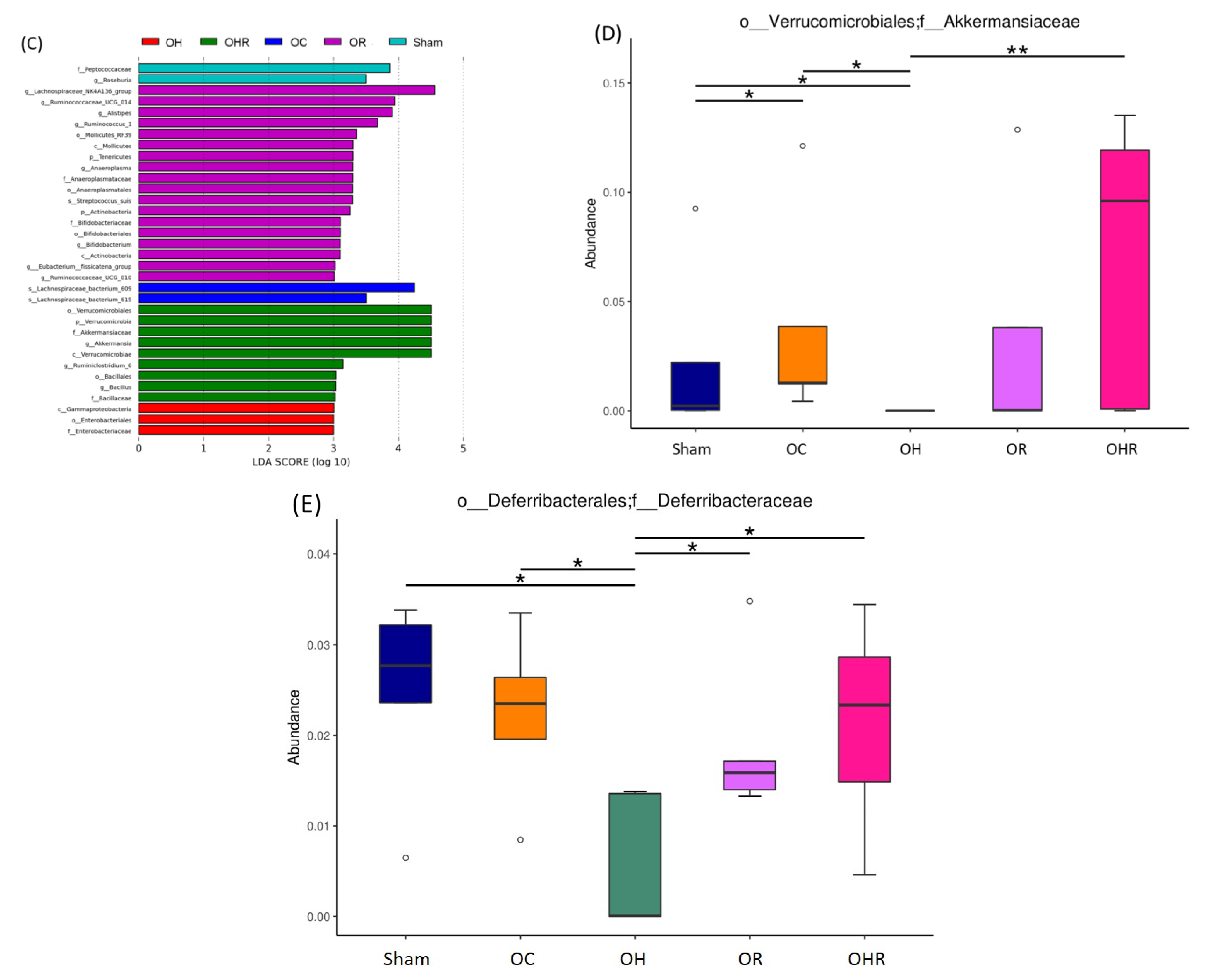
| Item | Sham | OC | OH | OR | OHR |
|---|---|---|---|---|---|
| Initial body weight (g) | 39.49 ± 0.31 | 38.81 ± 0.61 | 36.73 ± 0.54 | 38.38 ± 0.61 | 37.18 ± 0.57 |
| Final body weight (g) | 43.19 ± 0.75 | 53.94 ± 2.53 *ab | 58.58 ± 2.75 b | 45.77 ± 1.56 a | 48.41 ± 1.73 a |
| Weight gain (g) | 3.70 ± 0.99 | 15.13 ± 2.24 *bc | 21.85 ± 2.35 c | 7.39 ± 1.52 a | 11.24 ± 1.57 ab |
| Food intake (g/day) | 4.41 ± 0.20 | 4.02 ± 0.16 *c | 3.19 ± 0.21 a | 3.67 ± 0.16 bc | 3.01 ± 0.16 a |
| Energy (kcal/day) | 16.8 ± 0.35 | 15.11 ± 0.27 *b | 14.79 ± 0.38 ab | 13.96 ± 0.23 a | 13.93 ± 0.29 a |
| Tissue | Sham | OC | OH | OR | OHR |
|---|---|---|---|---|---|
| % of Body Weight | |||||
| Liver | 3.65 ± 0.06 | 3.65 ± 0.10 ab | 3.82 ± 0.20 b | 3.58 ± 0.10 ab | 3.23 ± 0.12 a |
| Perinephric adipose tissue | 3.81 ± 0.12 | 6.55 ± 0.54 * | 7.40 ± 0.65 | 5.79 ± 0.30 | 6.97 ± 0.39 |
| Uterus adipose tissue | 2.74 ± 0.16 | 3.36 ± 0.50 a | 6.16 ± 0.69 c | 2.79 ± 0.27 a | 5.17 ± 0.42 bc |
| Uterus | 0.44 ± 0.06 | 0.12 ± 0.01 * | 0.08 ± 0.01 | 0.12 ± 0.02 | 0.10 ± 0.01 |
| Colon | 1.56 ± 0.04 | 1.42 ± 0.10 bc | 1.02 ± 0.09 a | 1.60 ± 0.09 c | 1.20 ± 0.06 ab |
| Muscle | 1.36 ± 0.06 | 0.71 ± 0.06 *ab | 0.66 ± 0.04 a | 0.89 ± 0.02 c | 0.85 ± 0.03 bc |
| Sham | OC | OH | OR | OHR | |
|---|---|---|---|---|---|
| Estradiol (pg/mL) | 7.56 ± 0.45 | 4.63 ± 0.28 * | 4.74 ± 0.42 | 4.63 ± 0.34 | 4.63 ± 0.34 |
| Glucose (mg/dL) | 152.63 ± 7.16 | 158.11 ± 8.45 bc | 168.88 ± 8.82 c | 132.33 ± 4.59 a | 137.00 ± 3.79 ab |
| AST (U/L) | 115.71 ± 5.99 | 113.89 ± 7.74 bc | 119.25 ± 8.74 c | 88.00 ± 6.13 a | 76.63 ± 3.05 a |
| ALT (U/L) | 45.43 ± 10.13 | 39.44 ± 3.50 bc | 43.75 ± 5.00 c | 27.11 ± 1.35 a | 34.75 ± 1.80 abc |
| TC (mg/dL) | 108.75 ± 5.00 | 150.67 ± 8.83 * | 173.25 ± 9.7 | 164.00 ± 7.09 | 181.00 ± 11.10 |
| TG (mg/dL) | 88.13 ± 9.09 | 123.67 ± 4.32 *c | 117 ± 6.80 bc | 119.56 ± 5.14 bc | 94.00 ± 6.28 a |
| BUN (mg/dL) | 15.53 ± 0.72 | 16.81 ± 0.68 bc | 17.08 ± 0.87 c | 13.48 ± 0.42 a | 14.11 ± 0.43 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, P.-X.; Yeh, C.-L.; Yang, S.-C.; Shirakawa, H.; Chang, C.-L.; Chen, L.-H.; Chiu, Y.-S.; Chiu, W.-C. Rice Bran Supplementation Ameliorates Gut Dysbiosis and Muscle Atrophy in Ovariectomized Mice Fed with a High-Fat Diet. Nutrients 2023, 15, 3514. https://doi.org/10.3390/nu15163514
Huang P-X, Yeh C-L, Yang S-C, Shirakawa H, Chang C-L, Chen L-H, Chiu Y-S, Chiu W-C. Rice Bran Supplementation Ameliorates Gut Dysbiosis and Muscle Atrophy in Ovariectomized Mice Fed with a High-Fat Diet. Nutrients. 2023; 15(16):3514. https://doi.org/10.3390/nu15163514
Chicago/Turabian StyleHuang, Pei-Xin, Chiu-Li Yeh, Suh-Ching Yang, Hitoshi Shirakawa, Chao-Lin Chang, Li-Hsin Chen, Yen-Shuo Chiu, and Wan-Chun Chiu. 2023. "Rice Bran Supplementation Ameliorates Gut Dysbiosis and Muscle Atrophy in Ovariectomized Mice Fed with a High-Fat Diet" Nutrients 15, no. 16: 3514. https://doi.org/10.3390/nu15163514
APA StyleHuang, P.-X., Yeh, C.-L., Yang, S.-C., Shirakawa, H., Chang, C.-L., Chen, L.-H., Chiu, Y.-S., & Chiu, W.-C. (2023). Rice Bran Supplementation Ameliorates Gut Dysbiosis and Muscle Atrophy in Ovariectomized Mice Fed with a High-Fat Diet. Nutrients, 15(16), 3514. https://doi.org/10.3390/nu15163514







