Hydration Status in Older Adults: Current Knowledge and Future Challenges
Abstract
1. Introduction
2. Body Water Regulation
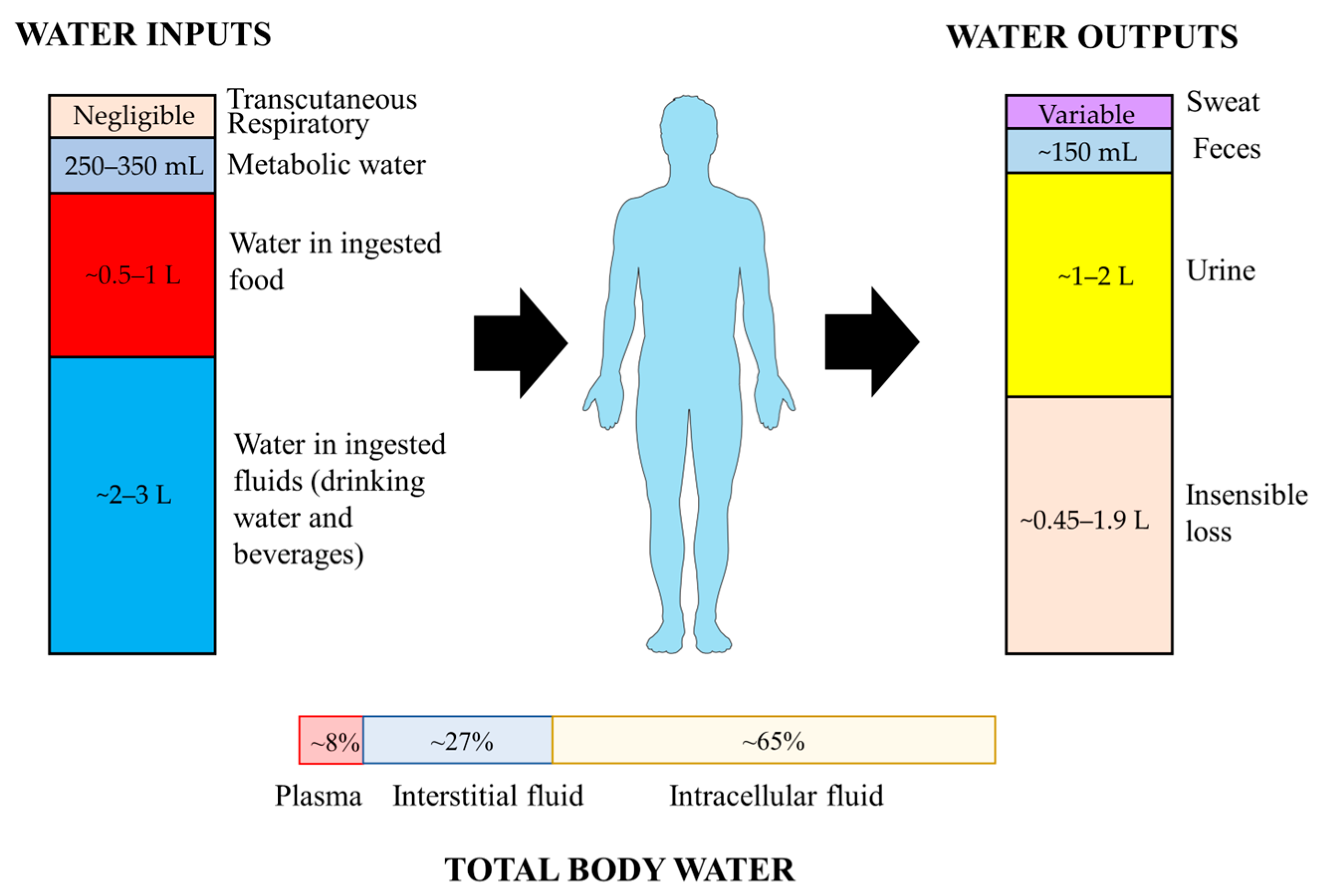
2.1. Water Inputs and Outputs
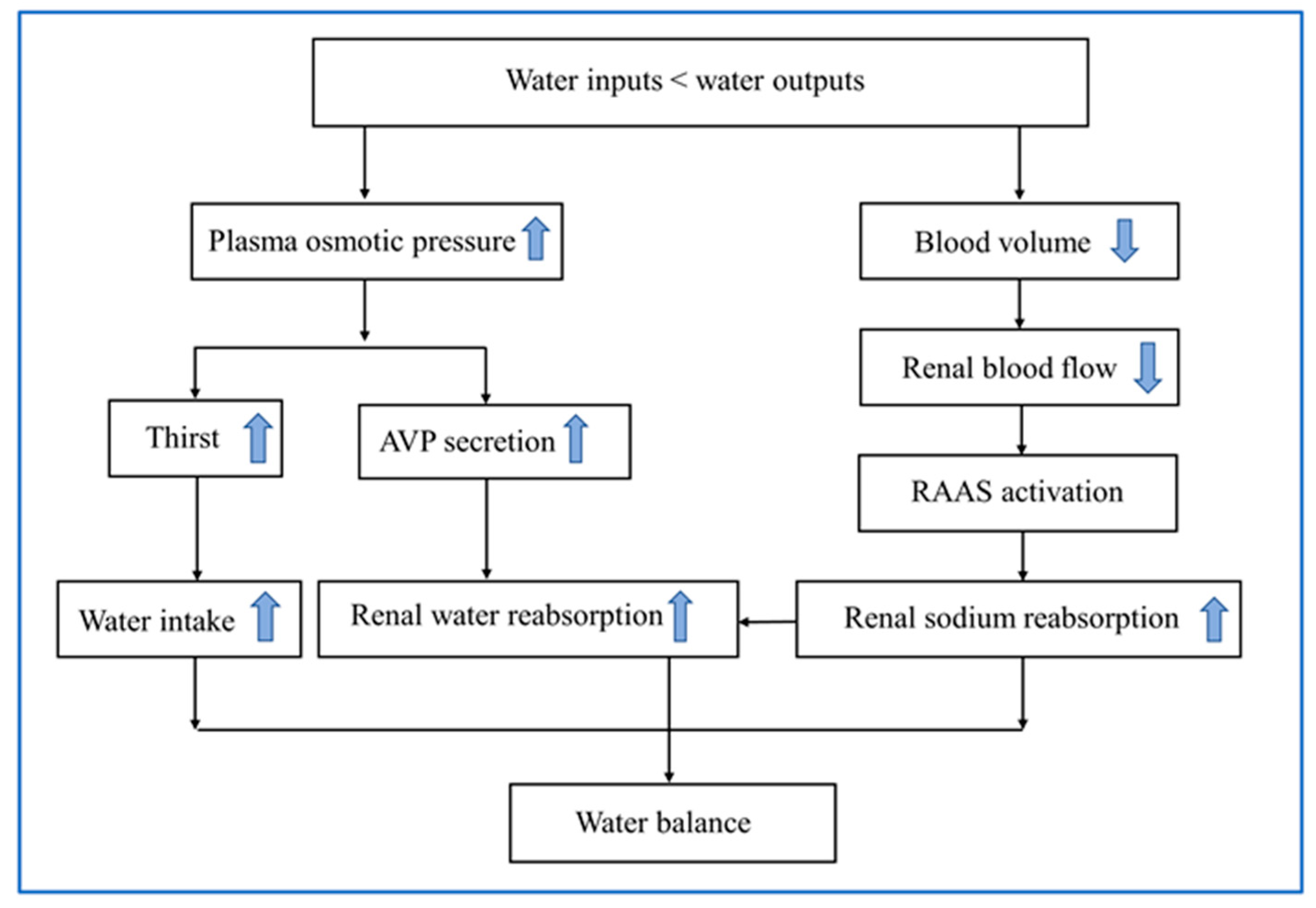
2.2. Physiological Mechanisms of Water Balance
2.3. Age-Related Changes in Water Balance Regulation
3. Definition and Causes of Dehydration
4. Measurement of Hydration Status
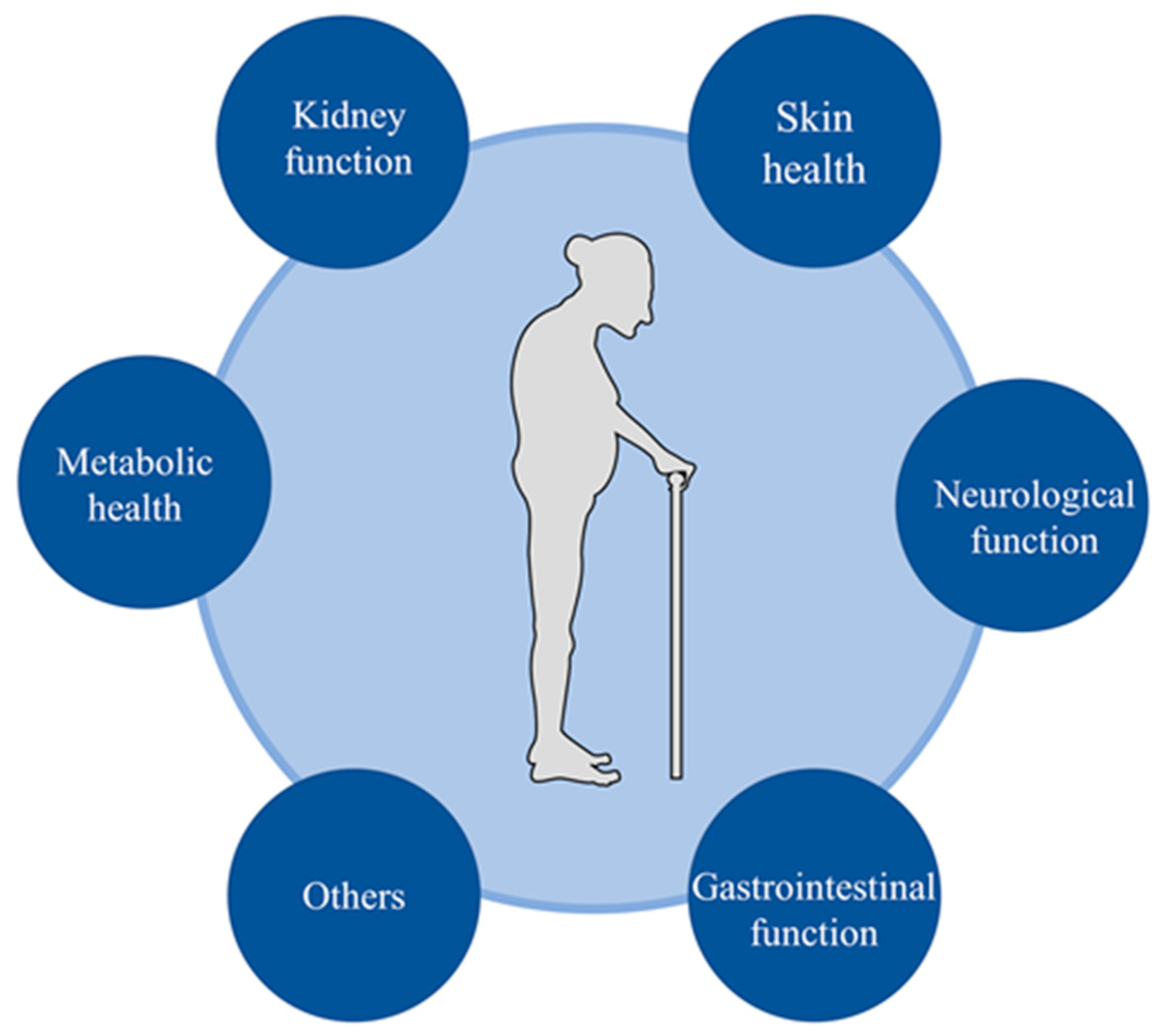
5. Water Intake, Hydration and Health Outcomes
5.1. Skin Health
5.2. Neurological Function
5.3. Gastrointestinal Function
5.4. Kidney Function
5.5. Metabolic Health
6. Management and Treatment of Low-Intake Dehydration
7. Challenges and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Petraccia, L.; Liberati, G.; Masciullo, S.G.; Grassi, M.; Fraioli, A. Water, mineral waters and health. Clin. Nutr. 2006, 25, 377–385. [Google Scholar] [CrossRef]
- Jéquier, E.; Constant, F. Water as an essential nutrient: The physiological basis of hydration. Eur. J. Clin. Nutr. 2010, 64, 115–123. [Google Scholar] [CrossRef]
- El-Sharkawy, A.M.; Sahota, O.; Maughan, R.J.; Lobo, D.N. The pathophysiology of fluid and electrolyte balance in the older adult surgical patient. Clin. Nutr. 2014, 33, 6–13. [Google Scholar] [CrossRef]
- Lacey, J.; Corbett, J.; Forni, L.; Hooper, L.; Hughes, F.; Minto, G.; Moss, C.; Price, S.; Whyte, G.; Woodcock, T.; et al. A multidisciplinary consensus on dehydration: Definitions, diagnostic methods and clinical implications. Ann. Med. 2019, 51, 232–251. [Google Scholar] [CrossRef]
- Volkert, D.; Beck, A.M.; Cederholm, T.; Cruz-Jentoft, A.; Goisser, S.; Hooper, L.; Kiesswetter, E.; Maggio, M.; Raynaud-Simon, A.; Sieber, C.C.; et al. ESPEN guideline on clinical nutrition and hydration in geriatrics. Clin. Nutr. 2019, 38, 10–47. [Google Scholar] [CrossRef]
- Hooper, L. Why, Oh Why, Are So Many Older Adults Not Drinking Enough Fluid? J. Acad. Nutr. Diet. 2016, 116, 774–778. [Google Scholar] [CrossRef]
- Hooper, L.; Bunn, D.; Jimoh, F.O.; Fairweather-Tait, S.J. Water-loss dehydration and aging. Mech. Ageing Dev. 2014, 136–137, 50–58. [Google Scholar] [CrossRef]
- Stookey, J.D.; Pieper, C.F.; Cohen, H.J. Is the prevalence of dehydration among community-dwelling older adults really low? Informing current debate over the fluid recommendation for adults aged 70+ years. Public Health Nutr. 2005, 8, 1275–1285. [Google Scholar] [CrossRef]
- Marra, M.V.; Simmons, S.F.; Shotwell, M.S.; Hudson, A.; Hollingsworth, E.K.; Long, E.; Kuertz, B.; Silver, H.J. Elevated Serum Osmolality and Total Water Deficit Indicate Impaired Hydration Status in Residents of Long-Term Care Facilities Regardless of Low or High Body Mass Index. J. Acad. Nutr. Diet. 2016, 116, 828–836. [Google Scholar] [CrossRef]
- Hooper, L.; Bunn, D.K.; Downing, A.; Jimoh, F.O.; Groves, J.; Free, C.; Cowap, V.; Potter, J.F.; Hunter, P.R.; Shepstone, L. Which Frail Older People Are Dehydrated? The UK DRIE Study. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2016, 71, 1341–1347. [Google Scholar] [CrossRef]
- Mentes, J.C. A typology of oral hydration problems exhibited by frail nursing home residents. J. Gerontol. Nurs. 2006, 32, 13–19, 20-21. [Google Scholar] [CrossRef]
- El-Sharkawy, A.M.; Watson, P.; Neal, K.R.; Ljungqvist, O.; Maughan, R.J.; Sahota, O.; Lobo, D.N. Hydration and outcome in older patients admitted to hospital (The HOOP prospective cohort study). Age Ageing 2015, 44, 943–947. [Google Scholar] [CrossRef] [PubMed]
- Kayser-Jones, J.; Schell, E.S.; Porter, C.; Barbaccia, J.C.; Shaw, H. Factors contributing to dehydration in nursing homes: Inadequate staffing and lack of professional supervision. J. Am. Geriatr. Soc. 1999, 47, 1187–1194. [Google Scholar] [CrossRef] [PubMed]
- Liska, D.; Mah, E.; Brisbois, T.; Barrios, P.L.; Baker, L.B.; Spriet, L.L. Narrative Review of Hydration and Selected Health Outcomes in the General Population. Nutrients 2019, 11, 70. [Google Scholar] [CrossRef] [PubMed]
- Wald, R.; Jaber, B.L.; Price, L.L.; Upadhyay, A.; Madias, N.E. Impact of hospital-associated hyponatremia on selected outcomes. Arch. Intern. Med. 2010, 170, 294–302. [Google Scholar] [CrossRef]
- Warren, J.L.; Bacon, W.E.; Harris, T.; McBean, A.M.; Foley, D.J.; Phillips, C. The burden and outcomes associated with dehydration among US elderly, 1991. Am. J. Public Health 1994, 84, 1265–1269. [Google Scholar] [CrossRef]
- Dimaria-Ghalili, R.A.; Nicolo, M. Nutrition and hydration in older adults in critical care. Crit. Care Nurs. Clin. N. Am. 2014, 26, 31–45. [Google Scholar] [CrossRef]
- Sanson, G.; Marzinotto, I.; De Matteis, D.; Boscutti, G.; Barazzoni, R.; Zanetti, M. Impaired hydration status in acutely admitted older patients: Prevalence and impact on mortality. Age Ageing 2021, 50, 1151–1158. [Google Scholar] [CrossRef]
- Grandjean, A.C.; Reimers, K.J.; Buyckx, M.E. Hydration: Issues for the 21st century. Nutr. Rev. 2003, 61, 261–271. [Google Scholar] [CrossRef]
- Sawka, M.N.; Cheuvront, S.N.; Kenefick, R.W. Hypohydration and Human Performance: Impact of Environment and Physiological Mechanisms. Sport. Med. 2015, 45 (Suppl. S1), S51–S60. [Google Scholar] [CrossRef]
- Yamada, Y.; Zhang, X.; Henderson, M.; Sagayama, H.; Pontzer, H.; Watanabe, D.; Yoshida, T.; Kimura, M.; Ainslie, P.N.; Andersen, L.F.; et al. Variation in human water turnover associated with environmental and lifestyle factors. Science 2022, 378, 909–915. [Google Scholar] [CrossRef] [PubMed]
- Luckey, A.E.; Parsa, C.J. Fluid and electrolytes in the aged. Arch. Surg. 2003, 138, 1055–1060. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, I.; Serra-Prat, M.; Yébenes, J.C. The Role of Water Homeostasis in Muscle Function and Frailty: A Review. Nutrients 2019, 11, 1857. [Google Scholar] [CrossRef] [PubMed]
- Sawka, M.N.; Cheuvront, S.N.; Carter, R.R. Human water needs. Nutr. Rev. 2005, 63, S30–S39. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Dietetic Products Nutrition and Allergic (NDA) Scientific Opinion on Dietary reference values for water/European Food Safety Authority. EFSA J. 2010, 8, 48.
- Institute of Medicine. Dietary Reference Intakes for Water, Potassium, Sodium, Chloride and Sulfate. Available online: http://www.nal.usda.gov/fnic/DRI//DRI_Water/73-185.pdf (accessed on 12 August 2014).
- Chinese Nutrition Society. Dietary Guide for Elderly Adults. WS/T 556-2017. Available online: https://www.cnsoc.org/policystand/page3.html (accessed on 1 April 2023).
- Volkert, D.; Beck, A.M.; Cederholm, T.; Cruz-Jentoft, A.; Hooper, L.; Kiesswetter, E.; Maggio, M.; Raynaud-Simon, A.; Sieber, C.; Sobotka, L.; et al. ESPEN practical guideline: Clinical nutrition and hydration in geriatrics. Clin. Nutr. 2022, 41, 958–989. [Google Scholar] [CrossRef]
- Perrier, E.; Demazières, A.; Girard, N.; Pross, N.; Osbild, D.; Metzger, D.; Guelinckx, I.; Klein, A. Circadian variation and responsiveness of hydration biomarkers to changes in daily water intake. Eur. J. Appl. Physiol. 2013, 113, 2143–2151. [Google Scholar] [CrossRef]
- Kiela, P.R.; Ghishan, F.K. Physiology of Intestinal Absorption and Secretion. Best Pract. Res. Clin. Gastroenterol. 2016, 30, 145–159. [Google Scholar] [CrossRef]
- Ritz, P.; Berrut, G. The importance of good hydration for day-to-day health. Nutr. Rev. 2005, 63, S6–S13. [Google Scholar] [CrossRef]
- Leiper, J.B.; Seonaid, P.C.; Primrose, W.R.; Phillimore, J.; Maughan, R.J. A comparison of water turnover in older people in community and institutional settings. J. Nutr. Health Aging 2005, 9, 189–193. [Google Scholar]
- Bankir, L.; Bichet, D.G.; Morgenthaler, N.G. Vasopressin: Physiology, assessment and osmosensation. J. Intern. Med. 2017, 282, 284–297. [Google Scholar] [CrossRef]
- Gizowski, C.; Bourque, C.W. The neural basis of homeostatic and anticipatory thirst. Nat. Rev. Nephrol. 2018, 14, 11–25. [Google Scholar] [CrossRef]
- Koshy, R.M.; Jamil, R.T. Physiology, Osmoreceptors; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Leib, D.E.; Zimmerman, C.A.; Knight, Z.A. Thirst. Curr. Biol. 2016, 26, R1260–R1265. [Google Scholar] [CrossRef]
- Phillips, P.A.; Rolls, B.J.; Ledingham, J.G.; Forsling, M.L.; Morton, J.J.; Crowe, M.J.; Wollner, L. Reduced thirst after water deprivation in healthy elderly men. N. Engl. J. Med. 1984, 311, 753–759. [Google Scholar] [CrossRef] [PubMed]
- Stachenfeld, N.S.; Mack, G.W.; Takamata, A.; DiPietro, L.; Nadel, E.R. Thirst and fluid regulatory responses to hypertonicity in older adults. Am. J. Physiol. 1996, 271, R757–R765. [Google Scholar] [CrossRef] [PubMed]
- Davies, I.; O’Neill, P.A.; McLean, K.A.; Catania, J.; Bennett, D. Age-associated alterations in thirst and arginine vasopressin in response to a water or sodium load. Age Ageing 1995, 24, 151–159. [Google Scholar] [CrossRef]
- Danziger, J.; Zeidel, M.L. Osmotic homeostasis. Clin. J. Am. Soc. Nephrol. 2015, 10, 852–862. [Google Scholar] [CrossRef]
- Watso, J.C.; Farquhar, W.B. Hydration Status and Cardiovascular Function. Nutrients 2019, 11, 1866. [Google Scholar] [CrossRef] [PubMed]
- Epstein, M. Aging and the kidney. J. Am. Soc. Nephrol. 1996, 7, 1106–1122. [Google Scholar] [CrossRef] [PubMed]
- Denic, A.; Glassock, R.J.; Rule, A.D. Structural and Functional Changes with the Aging Kidney. Adv. Chronic Kidney Dis. 2016, 23, 19–28. [Google Scholar] [CrossRef]
- Lindeman, R.D.; Tobin, J.; Shock, N.W. Longitudinal studies on the rate of decline in renal function with age. J. Am. Geriatr. Soc. 1985, 33, 278–285. [Google Scholar] [CrossRef]
- Rowe, J.W.; Shock, N.W.; DeFronzo, R.A. The influence of age on the renal response to water deprivation in man. Nephron 1976, 17, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Helderman, J.H.; Vestal, R.E.; Rowe, J.W.; Tobin, J.D.; Andres, R.; Robertson, G.L. The response of arginine vasopressin to intravenous ethanol and hypertonic saline in man: The impact of aging. J. Gerontol. 1978, 33, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Chiodera, P.; Capretti, L.; Marchesi, M.; Caiazza, A.; Bianconi, L.; Cavazzini, U.; Marchesi, C.; Volpi, R.; Coiro, V. Abnormal arginine vasopressin response to cigarette smoking and metoclopramide (but not to insulin-induced hypoglycemia) in elderly subjects. J. Gerontol. 1991, 46, M6–M10. [Google Scholar] [CrossRef]
- Faull, C.M.; Holmes, C.; Baylis, P.H. Water balance in elderly people: Is there a deficiency of vasopressin? Age Ageing 1993, 22, 114–120. [Google Scholar] [CrossRef]
- Johnson, A.G.; Crawford, G.A.; Kelly, D.; Nguyen, T.V.; Gyory, A.Z. Arginine vasopressin and osmolality in the elderly. J. Am. Geriatr. Soc. 1994, 42, 399–404. [Google Scholar] [CrossRef]
- Cheuvront, S.N.; Kenefick, R.W. Dehydration: Physiology, assessment, and performance effects. Compr. Physiol. 2014, 4, 257–285. [Google Scholar] [PubMed]
- Shah, M.K.; Workeneh, B.; Taffet, G.E. Hypernatremia in the geriatric population. Clin. Interv. Aging 2014, 9, 1987–1992. [Google Scholar] [PubMed]
- Lambert, K.; Carey, S. Dehydration in geriatrics: Consequences and practical guidelines. Curr. Opin. Clin. Nutr. Metab. Care 2023, 26, 36–41. [Google Scholar] [CrossRef]
- Barrado-Martín, Y.; Hatter, L.; Moore, K.J.; Sampson, E.L.; Rait, G.; Manthorpe, J.; Smith, C.H.; Nair, P.; Davies, N. Nutrition and hydration for people living with dementia near the end of life: A qualitative systematic review. J. Adv. Nurs. 2021, 77, 664–680. [Google Scholar] [CrossRef]
- Leibovitz, A.; Baumoehl, Y.; Lubart, E.; Yaina, A.; Platinovitz, N.; Segal, R. Dehydration among long-term care elderly patients with oropharyngeal dysphagia. Gerontology 2007, 53, 179–183. [Google Scholar] [CrossRef]
- Hooper, L.; Abdelhamid, A.; Attreed, N.J.; Campbell, W.W.; Channell, A.M.; Chassagne, P.; Culp, K.R.; Fletcher, S.J.; Fortes, M.B.; Fuller, N.; et al. Clinical symptoms, signs and tests for identification of impending and current water-loss dehydration in older people. Cochrane Database Syst. Rev. 2015, 2015, D9647. [Google Scholar] [CrossRef]
- Hooper, L.; Abdelhamid, A.; Ali, A.; Bunn, D.K.; Jennings, A.; John, W.G.; Kerry, S.; Lindner, G.; Pfortmueller, C.A.; Sjöstrand, F.; et al. Diagnostic accuracy of calculated serum osmolarity to predict dehydration in older people: Adding value to pathology laboratory reports. BMJ Open 2015, 5, e8846. [Google Scholar] [CrossRef] [PubMed]
- AlDisi, R.; Bader, Q.; Bermak, A. Hydration Assessment Using the Bio-Impedance Analysis Method. Sensors 2022, 22, 6350. [Google Scholar] [CrossRef] [PubMed]
- Lukaski, H.C.; Talluri, A. Phase angle as an index of physiological status: Validating bioelectrical assessments of hydration and cell mass in health and disease. Rev. Endocr. Metab. Disord. 2023, 24, 371–379. [Google Scholar] [CrossRef]
- Kyle, U.G.; Bosaeus, I.; De Lorenzo, A.D.; Deurenberg, P.; Elia, M.; Manuel, G.J.; Lilienthal, H.B.; Kent-Smith, L.; Melchior, J.C.; Pirlich, M.; et al. Bioelectrical impedance analysis-part II: Utilization in clinical practice. Clin. Nutr. 2004, 23, 1430–1453. [Google Scholar] [CrossRef] [PubMed]
- Garrett, D.C.; Rae, N.; Fletcher, J.R.; Zarnke, S.; Thorson, S.; Hogan, D.B.; Fear, E.C. Engineering Approaches to Assessing Hydration Status. IEEE Rev. Biomed. Eng. 2018, 11, 233–248. [Google Scholar] [CrossRef]
- Armstrong, L.E. Hydration assessment techniques. Nutr. Rev. 2005, 63, S40–S54. [Google Scholar] [CrossRef]
- Allen, M.D.; Springer, D.A.; Burg, M.B.; Boehm, M.; Dmitrieva, N.I. Suboptimal hydration remodels metabolism, promotes degenerative diseases, and shortens life. JCI Insight 2019, 4, e130949. [Google Scholar] [CrossRef]
- Walker, K.A.; Gottesman, R.F.; Wu, A.; Knopman, D.S.; Gross, A.L.; Mosley, T.J.; Selvin, E.; Windham, B.G. Systemic inflammation during midlife and cognitive change over 20 years: The ARIC Study. Neurology 2019, 92, e1256–e1267. [Google Scholar]
- Nadruz, W.J.; Kitzman, D.; Windham, B.G.; Kucharska-Newton, A.; Butler, K.; Palta, P.; Griswold, M.E.; Wagenknecht, L.E.; Heiss, G.; Solomon, S.D.; et al. Cardiovascular Dysfunction and Frailty among Older Adults in the Community: The ARIC Study. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2017, 72, 958–964. [Google Scholar] [CrossRef] [PubMed]
- Chambers, E.S.; Vukmanovic-Stejic, M. Skin barrier immunity and ageing. Immunology 2020, 160, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Verdier-Sévrain, S.; Bonté, F. Skin hydration: A review on its molecular mechanisms. J. Cosmet. Dermatol. 2007, 6, 75–82. [Google Scholar] [CrossRef]
- Camilion, J.V.; Khanna, S.; Anasseri, S.; Laney, C.; Mayrovitz, H.N. Physiological, Pathological, and Circadian Factors Impacting Skin Hydration. Cureus 2022, 14, e27666. [Google Scholar] [CrossRef] [PubMed]
- Samadi, A.; Yazdanparast, T.; Shamsipour, M.; Hassanzadeh, H.; Hashemi, O.M.; Firooz, R.; Firooz, A. Stratum corneum hydration in healthy adult humans according to the skin area, age and sex: A systematic review and meta-analysis. J. Eur. Acad. Dermatol. Venereol. 2022, 36, 1713–1721. [Google Scholar] [CrossRef]
- Yang, B.; Lv, C.; Ye, L.; Wang, Z.; Kim, Y.; Luo, W.; Elias, P.M.; Man, M.Q. Stratum corneum hydration inversely correlates with certain serum cytokine levels in the elderly, possibly contributing to inflammaging. Immun. Ageing 2023, 20, 7. [Google Scholar]
- Ye, L.; Wang, Z.; Kim, Y.; Elias, P.M.; Li, T.; Wen, S.; Song, J.; Lv, C.; Yang, B.; Man, M.Q. A topical emollient mitigates the progression of cognitive impairment in the elderly: A randomized, open-label pilot trial. J. Eur. Acad. Dermatol. Venereol. 2022, 36, 1382–1388. [Google Scholar] [CrossRef]
- Palma, L.; Marques, L.T.; Bujan, J.; Rodrigues, L.M. Dietary water affects human skin hydration and biomechanics. Clin. Cosmet. Investig. Dermatol. 2015, 8, 413–421. [Google Scholar]
- Palma, M.L.; Tavares, L.; Fluhr, J.W.; Bujan, M.J.; Rodrigues, L.M. Positive impact of dietary water on in vivo epidermal water physiology. Ski. Res. Technol. 2015, 21, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Mac-Mary, S.; Creidi, P.; Marsaut, D.; Courderot-Masuyer, C.; Cochet, V.; Gharbi, T.; Guidicelli-Arranz, D.; Tondu, F.; Humbert, P. Assessment of effects of an additional dietary natural mineral water uptake on skin hydration in healthy subjects by dynamic barrier function measurements and clinic scoring. Ski. Res. Technol. 2006, 12, 199–205. [Google Scholar] [CrossRef]
- Masento, N.A.; Golightly, M.; Field, D.T.; Butler, L.T.; van Reekum, C.M. Effects of hydration status on cognitive performance and mood. Br. J. Nutr. 2014, 111, 1841–1852. [Google Scholar] [CrossRef]
- Spigt, M.; Weerkamp, N.; Troost, J.; van Schayck, C.P.; Knottnerus, J.A. A randomized trial on the effects of regular water intake in patients with recurrent headaches. Fam. Pract. 2012, 29, 370–375. [Google Scholar] [CrossRef] [PubMed]
- Majdi, M.; Hosseini, F.; Naghshi, S.; Djafarian, K.; Shab-Bidar, S. Total and drinking water intake and risk of all-cause and cardiovascular mortality: A systematic review and dose-response meta-analysis of prospective cohort studies. Int. J. Clin. Pract. 2021, 75, e14878. [Google Scholar] [CrossRef] [PubMed]
- Anjo, I.; Amaral, T.F.; Afonso, C.; Borges, N.; Santos, A.; Moreira, P.; Padrão, P. Are hypohydrated older adults at increased risk of exhaustion? J. Hum. Nutr. Diet. 2020, 33, 23–30. [Google Scholar] [CrossRef]
- Białecka-Dębek, A.; Pietruszka, B. The association between hydration status and cognitive function among free-living elderly volunteers. Aging Clin. Exp. Res. 2019, 31, 695–703. [Google Scholar] [CrossRef]
- Lieberman, H.R. Hydration and cognition: A critical review and recommendations for future research. J. Am. Coll. Nutr. 2007, 26, 555S–561S. [Google Scholar] [CrossRef]
- Suhr, J.A.; Hall, J.; Patterson, S.M.; Niinistö, R.T. The relation of hydration status to cognitive performance in healthy older adults. Int. J. Psychophysiol. 2004, 53, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Suhr, J.A.; Patterson, S.M.; Austin, A.W.; Heffner, K.L. The relation of hydration status to declarative memory and working memory in older adults. J. Nutr. Health Aging 2010, 14, 840–843. [Google Scholar] [CrossRef]
- Bethancourt, H.J.; Kenney, W.L.; Almeida, D.M.; Rosinger, A.Y. Cognitive performance in relation to hydration status and water intake among older adults, NHANES 2011–2014. Eur. J. Nutr. 2020, 59, 3133–3148. [Google Scholar] [CrossRef]
- Nishi, S.K.; Babio, N.; Paz-Graniel, I.; Serra-Majem, L.; Vioque, J.; Fitó, M.; Corella, D.; Pintó, X.; Bueno-Cavanillas, A.; Tur, J.A.; et al. Water intake, hydration status and 2-year changes in cognitive performance: A prospective cohort study. BMC Med. 2023, 21, 82. [Google Scholar] [CrossRef]
- Gallegos-Orozco, J.F.; Foxx-Orenstein, A.E.; Sterler, S.M.; Stoa, J.M. Chronic constipation in the elderly. Am. J. Gastroenterol. 2012, 107, 18–25. [Google Scholar] [CrossRef]
- Bellini, M.; Tonarelli, S.; Barracca, F.; Rettura, F.; Pancetti, A.; Ceccarelli, L.; Ricchiuti, A.; Costa, F.; de Bortoli, N.; Marchi, S.; et al. Chronic Constipation: Is a Nutritional Approach Reasonable? Nutrients 2021, 13, 3386. [Google Scholar] [CrossRef] [PubMed]
- Lindeman, R.D.; Romero, L.J.; Liang, H.C.; Baumgartner, R.N.; Koehler, K.M.; Garry, P.J. Do elderly persons need to be encouraged to drink more fluids? J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2000, 55, M361–M365. [Google Scholar] [CrossRef] [PubMed]
- Robson, K.M.; Kiely, D.K.; Lembo, T. Development of constipation in nursing home residents. Dis. Colon Rectum 2000, 43, 940–943. [Google Scholar] [CrossRef]
- Anti, M.; Pignataro, G.; Armuzzi, A.; Valenti, A.; Iascone, E.; Marmo, R.; Lamazza, A.; Pretaroli, A.R.; Pace, V.; Leo, P.; et al. Water supplementation enhances the effect of high-fiber diet on stool frequency and laxative consumption in adult patients with functional constipation. Hepatogastroenterology 1998, 45, 727–732. [Google Scholar] [PubMed]
- Yurtdaş, D.G.; Acar, T.N.; Akbulut, G.; Günel, Z.; Kamanlı, B. Functional Constipation in Elderly and Related Determinant Risk Factors: Malnutrition and Dietary Intake. J. Am. Nutr. Assoc. 2022, 1–7. [Google Scholar] [CrossRef]
- Yurtdaş, G.; Acar-Tek, N.; Akbulut, G.; Cemali, Ö.; Arslan, N.; Beyaz, C.A.; Zengin, F.H. Risk Factors for Constipation in Adults: A Cross-Sectional Study. J. Am. Coll. Nutr. 2020, 39, 713–719. [Google Scholar] [CrossRef]
- Adak, A.; Khan, M.R. An insight into gut microbiota and its functionalities. Cell. Mol. Life Sci. 2019, 76, 473–493. [Google Scholar] [CrossRef]
- Ziętak, M.; Kovatcheva-Datchary, P.; Markiewicz, L.H.; Ståhlman, M.; Kozak, L.P.; Bäckhed, F. Altered Microbiota Contributes to Reduced Diet-Induced Obesity upon Cold Exposure. Cell Metab. 2016, 23, 1216–1223. [Google Scholar] [CrossRef]
- Mach, N.; Fuster-Botella, D. Endurance exercise and gut microbiota: A review. J. Sport Health Sci. 2017, 6, 179–197. [Google Scholar] [CrossRef]
- Willis, N.B.; Muñoz, C.X.; Mysonhimer, A.R.; Edwards, C.G.; Wolf, P.G.; Hillman, C.H.; Burd, N.A.; Holscher, H.D.; Khan, N.A. Hydration Biomarkers Are Related to the Differential Abundance of Fecal Microbiota and Plasma Lipopolysaccharide-Binding Protein in Adults. Ann. Nutr. Metab. 2021, 77 (Suppl. S4), 37–45. [Google Scholar] [CrossRef]
- Knepper, M.A.; Kwon, T.H.; Nielsen, S. Molecular physiology of water balance. N. Engl. J. Med. 2015, 372, 1349–1358. [Google Scholar] [CrossRef] [PubMed]
- Clark, W.F.; Sontrop, J.M.; Huang, S.H.; Moist, L.; Bouby, N.; Bankir, L. Hydration and Chronic Kidney Disease Progression: A Critical Review of the Evidence. Am. J. Nephrol. 2016, 43, 281–292. [Google Scholar] [CrossRef] [PubMed]
- Clark, W.F.; Sontrop, J.M.; Huang, S.H.; Gallo, K.; Moist, L.; House, A.A.; Cuerden, M.S.; Weir, M.A.; Bagga, A.; Brimble, S.; et al. Effect of Coaching to Increase Water Intake on Kidney Function Decline in Adults with Chronic Kidney Disease: The CKD WIT Randomized Clinical Trial. JAMA 2018, 319, 1870–1879. [Google Scholar] [CrossRef]
- Wagner, S.; Merkling, T.; Metzger, M.; Bankir, L.; Laville, M.; Frimat, L.; Combe, C.; Jacquelinet, C.; Fouque, D.; Massy, Z.A.; et al. Water intake and progression of chronic kidney disease: The CKD-REIN cohort study. Nephrol. Dial. Transplant. 2022, 37, 730–739. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Watanabe, H.; Tanaka, A.; Yasui, M.; Nishihira, J.; Murayama, N. Effect of Increased Daily Water Intake and Hydration on Health in Japanese Adults. Nutrients 2020, 12, 1191. [Google Scholar] [CrossRef]
- Weber, M.B.; Gujral, U.P.; Jagannathan, R.; Shah, M. Lifestyle Interventions for Diabetes Prevention in South Asians: Current Evidence and Opportunities. Curr. Diabetes Rep. 2021, 21, 23. [Google Scholar] [CrossRef]
- Yamaoka, K.; Tango, T. Effects of lifestyle modification on metabolic syndrome: A systematic review and meta-analysis. BMC Med. 2012, 10, 138. [Google Scholar] [CrossRef]
- Johnson, E.C.; Bardis, C.N.; Jansen, L.T.; Adams, J.D.; Kirkland, T.W.; Kavouras, S.A. Reduced water intake deteriorates glucose regulation in patients with type 2 diabetes. Nutr. Res. 2017, 43, 25–32. [Google Scholar] [CrossRef]
- Roussel, R.; Fezeu, L.; Bouby, N.; Balkau, B.; Lantieri, O.; Alhenc-Gelas, F.; Marre, M.; Bankir, L. Low water intake and risk for new-onset hyperglycemia. Diabetes Care 2011, 34, 2551–2554. [Google Scholar] [CrossRef]
- Vanhaecke, T.; Perrier, E.T.; Melander, O. A Journey through the Early Evidence Linking Hydration to Metabolic Health. Ann. Nutr. Metab. 2020, 76 (Suppl. S1), 4–9. [Google Scholar] [CrossRef]
- Enhörning, S.; Brunkwall, L.; Tasevska, I.; Ericson, U.; Persson, T.J.; Persson, M.; Lemetais, G.; Vanhaecke, T.; Dolci, A.; Perrier, E.T.; et al. Water Supplementation Reduces Copeptin and Plasma Glucose in Adults with High Copeptin: The H2O Metabolism Pilot Study. J. Clin. Endocrinol. Metab. 2019, 104, 1917–1925. [Google Scholar] [CrossRef] [PubMed]
- Shirreffs, S.M.; Sawka, M.N. Fluid and electrolyte needs for training, competition, and recovery. J. Sports Sci. 2011, 29 (Suppl. S1), S39–S46. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, L.E. Rehydration during Endurance Exercise: Challenges, Research, Options, Methods. Nutrients 2021, 13, 887. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.R.; Cote, T.R.; Lawhorne, L.; Levenson, S.A.; Rubenstein, L.Z.; Smith, D.A.; Stefanacci, R.G.; Tangalos, E.G.; Morley, J.E. Understanding clinical dehydration and its treatment. J. Am. Med. Dir. Assoc. 2008, 9, 292–301. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, L.; Imamura, F.; Lentjes, M.A.; Khaw, K.T.; Wareham, N.J.; Forouhi, N.G. Prospective associations and population impact of sweet beverage intake and type 2 diabetes, and effects of substitutions with alternative beverages. Diabetologia 2015, 58, 1474–1483. [Google Scholar] [CrossRef]
- Bruno, C.; Collier, A.; Holyday, M.; Lambert, K. Interventions to Improve Hydration in Older Adults: A Systematic Review and Meta-Analysis. Nutrients 2021, 13, 3640. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, L.E.; Muñoz, C.X.; Armstrong, E.M. Distinguishing Low and High Water Consumers-A Paradigm of Disease Risk. Nutrients 2020, 12, 858. [Google Scholar] [CrossRef]
- Valtin, H. “Drink at least eight glasses of water a day.” Really? Is there scientific evidence for “8 × 8”? Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2002, 283, R993–R1004. [Google Scholar] [CrossRef]
| Institution | Year | Daily Fluid Intake | Age |
|---|---|---|---|
| EFSA [25] | 2010 | ≥1.6 L for women ≥2.0 L for men | Adults (≥18 years) |
| IOM [26] | 2014 | 2.2 L for women 3.0 L for men | Adults (≥50 years) |
| CNS [27] | 2019 | 1.5 L for women 1.7 L for men | Older adults (≥65 years) |
| ESPEN [28] | 2022 | ≥1.6 L for women ≥2.0 L for men | Older adults (≥65 years) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, S.; Xiao, X.; Zhang, X. Hydration Status in Older Adults: Current Knowledge and Future Challenges. Nutrients 2023, 15, 2609. https://doi.org/10.3390/nu15112609
Li S, Xiao X, Zhang X. Hydration Status in Older Adults: Current Knowledge and Future Challenges. Nutrients. 2023; 15(11):2609. https://doi.org/10.3390/nu15112609
Chicago/Turabian StyleLi, Shizhen, Xun Xiao, and Xiangyu Zhang. 2023. "Hydration Status in Older Adults: Current Knowledge and Future Challenges" Nutrients 15, no. 11: 2609. https://doi.org/10.3390/nu15112609
APA StyleLi, S., Xiao, X., & Zhang, X. (2023). Hydration Status in Older Adults: Current Knowledge and Future Challenges. Nutrients, 15(11), 2609. https://doi.org/10.3390/nu15112609






