Effectiveness of Food-Based Intervention to Improve the Linear Growth of Children under Five: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Inclusion and Exclusion Criteria
2.3. Data Extraction
2.4. Quality of the Studies
2.5. Data Synthesis and Analysis
3. Results
3.1. Reporting Results and Study Selection
3.2. Study Characteristics
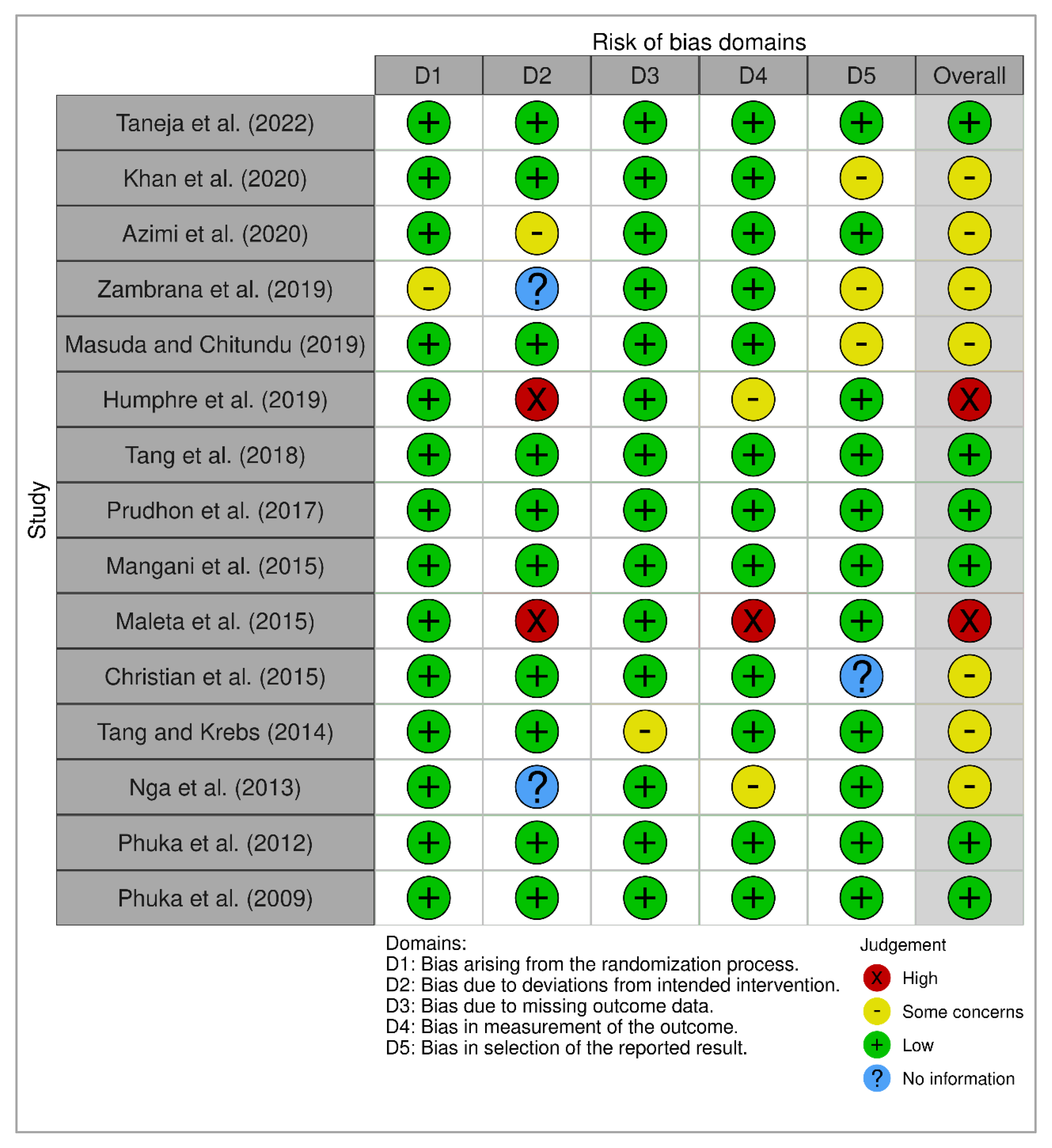
3.3. Risk of Bias
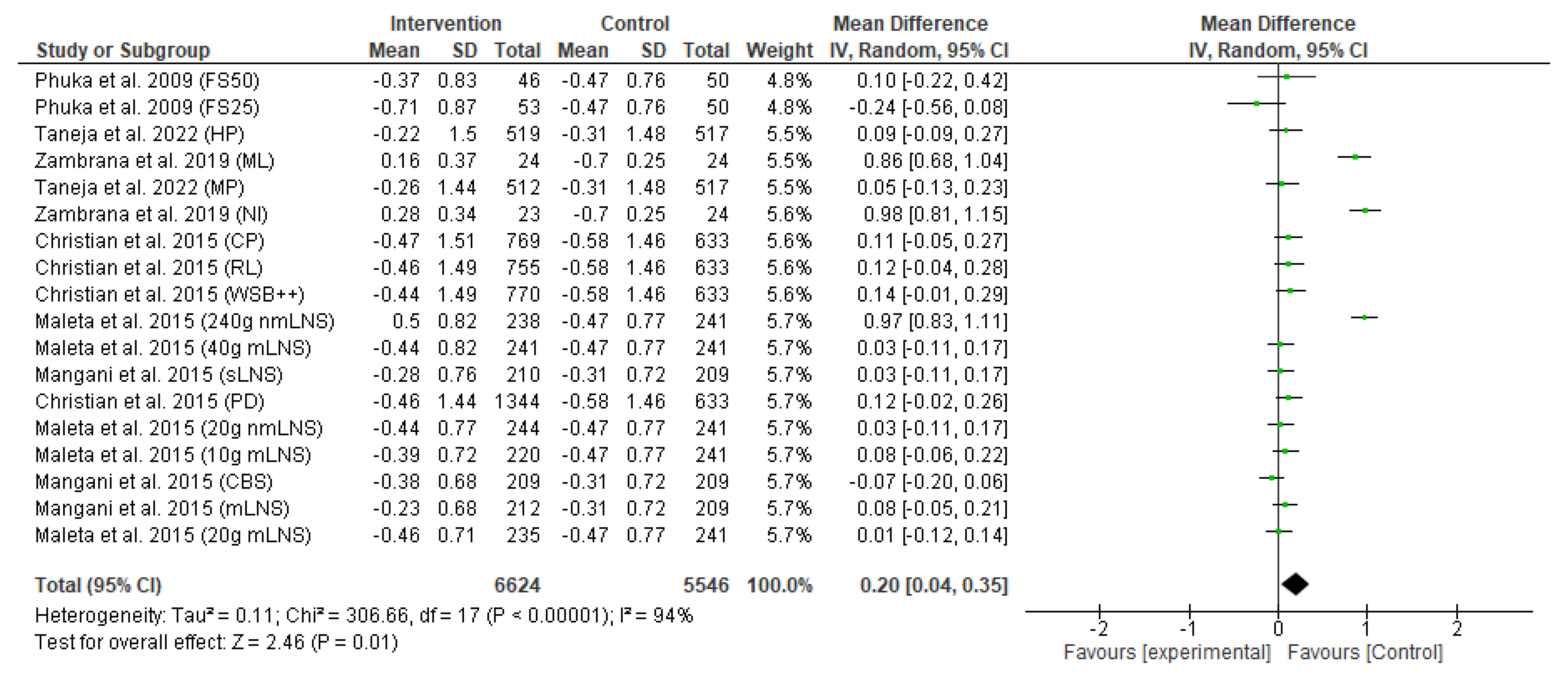
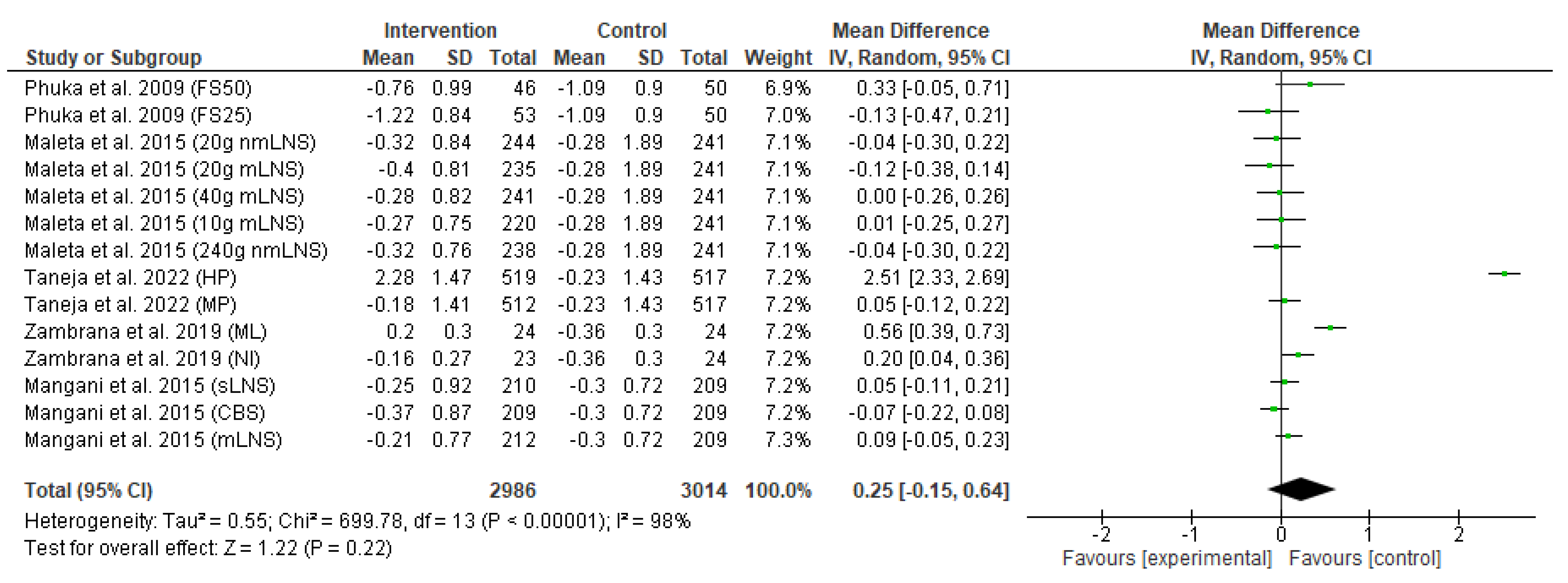
3.4. Meta-Analysis Results
3.4.1. Primary Outcome
3.4.2. Secondary Outcome
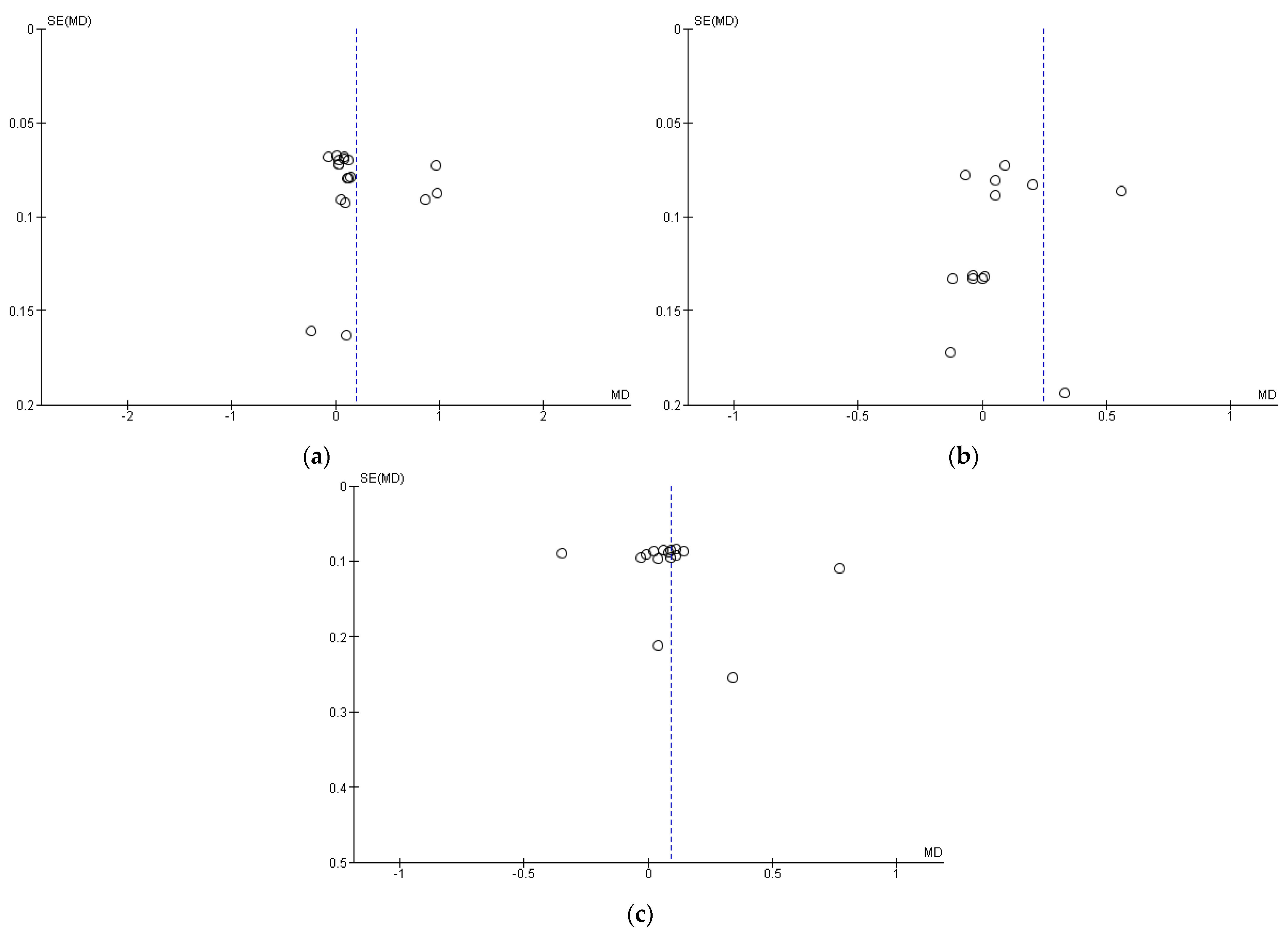
3.5. Publication Bias (Funnel Plot)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Black, R.E.; Victora, C.G.; Walker, S.P.; Bhutta, Z.A.; Christian, P.; de Onis, M.; Ezzati, M.; Grantham-Mcgregor, S.; Katz, J.; Martorell, R.; et al. Maternal and Child Undernutrition and Overweight in Low-Income and Middle-Income Countries. Lancet 2013, 382, 427–451. [Google Scholar] [CrossRef] [PubMed]
- WHO. Levels and Trends in Child Malnutrition: UNICEF/WHO/The World Bank Group Joint Child Malnutrition Estimates: Key Findings of the 2021. Available online: https://www.who.int/publications/i/item/9789240025257 (accessed on 12 January 2023).
- Stewart, C.P.; Iannotti, L.; Dewey, K.G.; Michaelsen, K.F.; Onyango, A.W. Contextualising Complementary Feeding in a Broader Framework for Stunting Prevention. Matern. Child Nutr. 2013, 9, 27–45. [Google Scholar] [CrossRef] [PubMed]
- Beal, T.; Tumilowicz, A.; Sutrisna, A.; Izwardy, D.; Neufeld, L.M. A Review of Child Stunting Determinants in Indonesia. Matern. Child Nutr. 2018, 14, e12617. [Google Scholar] [CrossRef]
- Mahfuz, M.; Alam, M.A.; Das, S.; Fahim, S.M.; Hossain, M.S.; Petri, W.A.; Ashorn, P.; Ashorn, U.; Ahmed, T. Daily Supplementation with Egg, Cow Milk, and Multiple Micronutrients Increases Linear Growth of Young Children with Short Stature. J. Nutr. 2020, 150, 394–403. [Google Scholar] [CrossRef]
- Pusparini; Isdiany, N.; Tursilowati, S. The Effects of Multiple-Nutrients Fortified Biscuits and/or Psychosocial Parenting Education Intervention Programs on Anthropometric and Cognitive Measures of Toddlers. J. Nutr. Sci. Vitaminol. 2020, 66, S443–S449. [Google Scholar] [CrossRef]
- Wang, J.; Chang, S.; Zhao, L.; Yu, W.; Zhang, J.; Man, Q.; He, L.; Duan, Y.; Wang, H.; Scherpbier, R.; et al. Effectiveness of Community-Based Complementary Food Supplement (Yingyangbao) Distribution in Children Aged 6–23 Months in Poor Areas in China. PLoS ONE 2017, 12, e0174302. [Google Scholar] [CrossRef]
- Li, Z.; Li, X.; Sudfeld, C.R.; Liu, Y.; Tang, K.; Huang, Y.; Fawzi, W. The Effect of the Yingyangbao Complementary Food Supplement on the Nutritional Status of Infants and Children: A Systematic Review and Meta-Analysis. Nutrients 2019, 11, 2404. [Google Scholar] [CrossRef] [PubMed]
- Cassinat, R.A.; Bruening, M.; Crespo, N.C.; Gutiérrez, M.; Chavez, A.; Ray, F.; Vega-López, S. Effects of a Community-Based Pilot Intervention on Home Food Availability among U.S. Households. Int. J. Env. Res. Public Health 2020, 17, 8327. [Google Scholar] [CrossRef] [PubMed]
- Thorisdottir, B.; Gunnarsdottir, I.; Palsson, G.I.; Halldorsson, T.I.; Thorsdottir, I. Animal protein intake at 12 months is associated with growth factors at the age of six. Acta Paediatr. 2014, 103, 512–517. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. BMJ 2009, 339, b2535. [Google Scholar] [CrossRef]
- Fricton, J.R.; Ouyang, W.; Schiffman, E.L.; Velly, A.M.; Look, J.O. Critical Appraisal of Methods Used in Randomized Controlled Trials of Treatments for Temporomandibular Disorders. J. Orofac. Pain 2010, 24, 139–151. [Google Scholar]
- Taneja, S.; Upadhyay, R.P.; Chowdhury, R.; Kurpad, A.V.; Bhardwaj, H.; Kumar, T.; Dwarkanath, P.; Bose, B.; Devi, S.; Kumar, G.; et al. Impact of Supplementation with Milk-Cereal Mix during 6–12 Months of Age on Growth at 12 Months: A 3-Arm Randomized Controlled Trial in Delhi, India. Am. J. Clin. Nutr. 2022, 115, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Khan, G.N.; Kureishy, S.; Ariff, S.; Rizvi, A.; Sajid, M.; Garzon, C.; Khan, A.A.; De Pee, S.; Soofi, S.B.; Bhutta, Z.A. Effect of Lipid-Based Nutrient Supplement-Medium Quantity on Reduction of Stunting in Children 6–23 Months of Age in Sindh, Pakistan: A Cluster Randomized Controlled Trial. PLoS ONE 2020, 15, e0237210. [Google Scholar] [CrossRef] [PubMed]
- Azimi, F.; Esmaillzadeh, A.; Alipoor, E.; Moslemi, M.; Yaseri, M.; Hosseinzadeh-Attar, M.J. Effect of a Newly Developed Ready-to-Use Supplementary Food on Growth Indicators in Children with Mild to Moderate Malnutrition. Public Health 2020, 185, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Zambrana, L.E.; McKeen, S.; Ibrahim, H.; Zarei, I.; Borresen, E.C.; Doumbia, L.; Boré, A.; Cissoko, A.; Douyon, S.; Koné, K.; et al. Rice Bran Supplementation Modulates Growth, Microbiota and Metabolome in Weaning Infants: A Clinical Trial in Nicaragua and Mali. Sci. Rep. 2019, 9, 13919. [Google Scholar] [CrossRef]
- Masuda, K.; Chitundu, M. Multiple Micronutrient Supplementation Using Spirulina Platensis and Infant Growth, Morbidity, and Motor Development: Evidence from a Randomized Trial in Zambia. PLoS ONE 2019, 14, e0211693. [Google Scholar] [CrossRef]
- Humphrey, J.H.; Mbuya, M.N.N.; Ntozini, R.; Moulton, L.H.; Stoltzfus, R.J.; Tavengwa, N.V.; Mutasa, K.; Majo, F.; Mutasa, B.; Mangwadu, G.; et al. Independent and Combined Effects of Improved Water, Sanitation, and Hygiene, and Improved Complementary Feeding, on Child Stunting and Anaemia in Rural Zimbabwe: A Cluster-Randomised Trial. Lancet Glob. Health 2019, 7, e132–e147. [Google Scholar] [CrossRef]
- Tang, M.; Hendricks, A.E.; Krebs, N.F. A Meat-or Dairy-Based Complementary Diet Leads to Distinct Growth Patterns in Formula-Fed Infants: A Randomized Controlled Trial. Am. J. Clin. Nutr. 2018, 107, 734–742. [Google Scholar] [CrossRef]
- Prudhon, C.; Langendorf, C.; Roederer, T.; Doyon, S.; Mamaty, A.A.; Woi-Messe, L.; Manzo, M.L.; de Pee, S.; Grais, R.F. Effect of Ready-to-Use Foods for Preventing Child Undernutrition in Niger: Analysis of a Prospective Intervention Study over 15 Months of Follow-Up. Matern. Child Nutr. 2017, 13, e12236. [Google Scholar] [CrossRef]
- Mangani, C.; Maleta, K.; Phuka, J.; Cheung, Y.B.; Thakwalakwa, C.; Dewey, K.; Manary, M.; Puumalainen, T.; Ashorn, P. Effect of Complementary Feeding with Lipid-Based Nutrient Supplements and Corn-Soy Blend on the Incidence of Stunting and Linear Growth among 6- to 18-Month-Old Infants and Children in Rural Malawi. Matern. Child Nutr. 2015, 11, 132–143. [Google Scholar] [CrossRef]
- Maleta, K.M.; Phuka, J.; Alho, L.; Cheung, Y.B.; Dewey, K.G.; Ashorn, U.; Phiri, N.; Phiri, T.E.; Vosti, S.A.; Zeilani, M.; et al. Provision of 10–40 g/d Lipid-Based Nutrient Supplements from 6 to 18 Months of Age Does Not Prevent Linear Growth Faltering in Malawi. J. Nutr. 2015, 145, 1909–1915. [Google Scholar] [CrossRef] [PubMed]
- Christian, P.; Shaikh, S.; Shamim, A.A.; Mehra, S.; Wu, L.; Mitra, M.; Ali, H.; Merrill, R.D.; Choudhury, N.; Parveen, M.; et al. Effect of Fortified Complementary Food Supplementation on Child Growth in Rural Bangladesh: A Cluster-Randomized Trial. Int. J. Epidemiol. 2015, 44, 1862–1876. [Google Scholar] [CrossRef]
- Tang, M.; Krebs, N.F. High Protein Intake from Meat as Complementary Food Increases Growth but Not Adiposity in Breastfed Infants: A Randomized Trial. Am. J. Clin. Nutr. 2014, 100, 1322–1328. [Google Scholar] [CrossRef] [PubMed]
- Nga, T.T.; Nguyen, M.; Mathisen, R.; Hoa, D.T.B.; Minh, N.H.; Berger, J.; Wieringa, F.T. Acceptability and Impact on Anthropometry of a Locally Developed Ready-to-Use Therapeutic Food in Pre-School Children in Vietnam. Nutr. J. 2013, 12, 120. [Google Scholar] [CrossRef] [PubMed]
- Phuka, J.C.; Gladstone, M.; Maleta, K.; Thakwalakwa, C.; Cheung, Y.B.; Briend, A.; Manary, M.J.; Ashorn, P. Developmental Outcomes among 18-Month-Old Malawians after a Year of Complementary Feeding with Lipid-Based Nutrient Supplements or Corn-Soy Flour. Matern. Child Nutr. 2012, 8, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Phuka, J.C.; Maleta, K.; Thakwalakwa, C.; Yin, B.C.; Briend, A.; Manary, M.J.; Ashorn, P. Postintervention Growth of Malawian Children Who Received 12-Mo Dietary Complementation with a Lipid-Based Nutrient Supplement or Maize-Soy Flour. Am. J. Clin. Nutr. 2009, 89, 382–390. [Google Scholar] [CrossRef]
- McGuinness, L.A.; Higgins, J.P.T. Risk-of-Bias VISualization (Robvis): An R Package and Shiny Web App for Visualizing Risk-of-Bias Assessments. Res. Synth. Methods 2021, 12, 55–61. [Google Scholar] [CrossRef]
- Goudet, S.M.; Bogin, B.A.; Madise, N.J.; Griffiths, P.L. Nutritional Interventions for Preventing Stunting in Children (Birth to 59 Months) Living in Urban Slums in Low-and Middle-Income Countries (LMIC). Cochrane Database Syst. Rev. 2019, 2019, CD011695. [Google Scholar] [CrossRef]
- Bhutta, Z.A.; Das, J.K.; Rizvi, A.; Gaffey, M.F.; Walker, N.; Horton, S.; Webb, P.; Lartey, A.; Black, R.E. Evidence-Based Interventions for Improvement of Maternal and Child Nutrition: What Can Be Done and at What Cost? Lancet 2013, 382, 452–477. [Google Scholar] [CrossRef]
- Elisaria, E.; Mrema, J.; Bogale, T.; Segafredo, G.; Festo, C. Effectiveness of integrated nutrition interventions on childhood stunting: A quasi-experimental evaluation design. BMC Nutr. 2021, 7, 17. [Google Scholar] [CrossRef]
- Kaimila, Y.; Divala, O.; Agapova, S.E.; Stephenson, K.B.; Thakwalakwa, C.; Trehan, I.; Manary, M.J.; Maleta, K.M. Consumption of Animal-Source Protein Is Associated with Improved Height-for-Age Z Scores in Rural Malawian Children Aged 12–36 Months. Nutrients 2019, 11, 480. [Google Scholar] [CrossRef] [PubMed]
- Lim, M.T.; Pan, B.J.; Toh, D.W.K.; Sutanto, C.N.; Kim, J.E. Animal Protein versus Plant Protein in Supporting Lean Mass and Muscle Strength: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2021, 13, 661. [Google Scholar] [CrossRef] [PubMed]
- Headey, D.; Hirvonen, K.; Hoddinott, J. Animal Sourced Foods and Child Stunting. Am. J. Agric. Econ. 2018, 100, 1302–1319. [Google Scholar] [CrossRef] [PubMed]
- Saha, J.; Chouhan, P.; Malik, N.I.; Ghosh, T.; Das, P.; Shahid, M.; Ahmed, F.; Tang, K. Effects of Dietary Diversity on Growth Outcomes of Children Aged 6 to 23 Months in India: Evidence from National Family and Health Survey. Nutrients 2023, 15, 159. [Google Scholar] [CrossRef] [PubMed]
- Potts, K.S.; Mulugeta, A.; Bazzano, A.N. Animal Source Food Consumption in Young Children from Four Regions of Ethiopia: Association with Religion, Livelihood, and Participation in the Productive Safety Net Program. Nutrients 2019, 11, 354. [Google Scholar] [CrossRef] [PubMed]
- Dewey, K.G.; Brown, K.H. Update on Technical issues concerning Complementary Feeding of Young Children in Developing Countries and Implications for Intervention Programs. Food Nutr. Bull. 2003, 24, 5–28. [Google Scholar] [CrossRef]
- Adesogan, A.T.; Havelaar, A.H.; McKune, S.L.; Eilittä, M.; Dahl, G.E. Animal Source Foods: Sustainability Problem or Malnutrition and Sustainability Solution? Perspective Matters. Glob. Food Secur. 2020, 25, 100325. [Google Scholar] [CrossRef]
- Lelijveld, N.; Beedle, A.; Farhikhtah, A.; Elrayah, E.E.; Bourdaire, J.; Aburto, N. Systematic Review of the Treatment of Moderate Acute Malnutrition Using Food Products. Matern. Child Nutr. 2020, 16, e12898. [Google Scholar] [CrossRef]
- Swareldhab, E.S.E.; Al-Jawaldeh, A.; Qureshi, A.B.; Ali, A.M.E.; Abu-Manga, M.; Al-Areeqi, M.; Dureab, F. Assessment of Micronutrient Situation among Reproductive-Age Women (15–49) and under-Five Children in Sudan. Nutrients 2021, 13, 2784. [Google Scholar] [CrossRef]
- Manaseki-Holland, S.; Manjang, B.; Hemming, K.; Martin, J.T.; Bradley, C.; Jackson, L.; Taal, M.; Gautam, O.P.; Crowe, F.; Sanneh, B.; et al. Effects on Childhood Infections of Promoting Safe and Hygienic Complementary-Food Handling Practices through a Community-Based Programme: A Cluster Randomised Controlled Trial in a Rural Area of the Gambia. PLoS Med 2021, 18, e1003260. [Google Scholar] [CrossRef]
- Chaparro, C.M.; Dewey, K.G. Use of Lipid-Based Nutrient Supplements (LNS) to Improve the Nutrient Adequacy of General Food Distribution Rations for Vulnerable Sub-Groups in Emergency Settings. Matern. Child Nutr. 2010, 6 (Suppl. 1), 1–69. [Google Scholar] [CrossRef] [PubMed]
- Arimond, M.; Zeilani, M.; Jungjohann, S.; Brown, K.H.; Ashorn, P.; Allen, L.H.; Dewey, K.G. Considerations in Developing Lipid-Based Nutrient Supplements for Prevention of Undernutrition: Experience from the International Lipid-Based Nutrient Supplements (ILiNS) Project. Matern. Child Nutr. 2015, 11, 31–61. [Google Scholar] [CrossRef] [PubMed]
- Inzaghi, E.; Pampanini, V.; Deodati, A.; Cianfarani, S. The Effects of Nutrition on Linear Growth. Nutrients 2022, 14, 1752. [Google Scholar] [CrossRef] [PubMed]



| Database | Search Strategy | Filter | Records |
|---|---|---|---|
| SCOPUS | TITLE-ABS-KEY ((“Food Intervention” OR “Nutrition Intervention”) AND (“under-five” OR “child*”) AND (“Stunting” OR “linear growth” OR “child linear growth”)) AND (LIMIT-TO (SRCTYPE, “j”)) | Year: 2000–2022 Research article, Language: English | 199 |
| Web of Science | (“Food Intervention” OR “Nutrition Intervention”) AND (“under-five” OR “child*”) AND (“Stunting” OR “linear growth” OR “child linear growth”) | Year: 2000–2022 Research article, Language: English | 74 |
| PubMed | #1: ((((“Dietary Supplements”[Mesh] OR “Food, Formulated”[Mesh]) OR “Food Intervention”[tw]) OR “Diet Therapy”[Mesh]) OR “Dietary Supplements/analysis”[Mesh]) OR “Dietary Supplements/statistics and numerical data”[Mesh]#2: (“Growth Disorders”[Mesh] OR Stunting[tw]) OR “Child Development”[Mesh] #3: ((“Child, Preschool”[Mesh] OR “under-five”[tw]) OR “under five”[tw]) OR child*[tw]) #4: “Counseling”[Mesh] OR “Health Knowledge, Attitudes, Practice”[Majr] ((#1 AND #2) AND #3) NOT #4 | 2000–2022 Randomized controlled trial, English | 449 |
| Science Direct | (“Food Intervention” OR “Nutrition Intervention”) AND (“under-five” OR “under five” OR “child”) AND (“Stunting” OR “linear growth”) NOT (“Knowledge intervention” OR “counseling intervention”) | Year: 2000–2022 Research article | 32 |
| ProQuest | (“Food Intervention” OR “Nutrition Intervention”) AND (“under-five” OR “under five” OR “child*”) AND (“Stunting” OR “linear growth” OR “child linear growth”) NOT (“counseling intervention” OR “Knowledge intervention” OR “meta-analysis” OR Review) | Year: 2000–2022 Language: English | 331 |
| Studies | Item 1 | Item 2 | Item 3 | Item 4 | Item 5 | Item 6 | Item 7 | Item 8 | Item 9 | Item 10 | Score | QR |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Taneja et al. (2022) [13] | + | + | + | - | + | + | + | + | + | + | 9/10 | Good |
| Khan et al. (2020) [14] | + | + | + | + | + | + | + | + | + | - | 9/10 | Good |
| Azimi et al. (2020) [15] | + | + | + | + | + | + | + | - | + | - | 8/10 | Good |
| Zambrana et al. (2019) [16] | + | + | + | - | + | + | + | + | + | + | 9/10 | Good |
| Masuda and Chitundu (2019) [17] | + | + | + | - | + | + | + | - | + | + | 8/10 | Good |
| Humphre et al. (2019) [18] | + | + | + | + | + | + | + | + | + | - | 9/10 | Good |
| Tang et al. (2018) [19] | + | + | + | - | + | + | + | - | + | - | 7/10 | Good |
| Prudhon et al. (2017) [20] | + | + | + | - | + | + | + | + | + | - | 8/10 | Good |
| Mangani et al. (2015) [21] | + | + | + | + | + | + | + | + | + | + | 10/10 | Good |
| Maleta et al. (2015) [22] | + | + | + | + | + | + | + | + | + | + | 10/10 | Good |
| Christian et al. (2015) [23] | + | + | + | - | + | + | - | + | + | + | 8/10 | Good |
| Tang and Krebs (2014) [24] | + | + | + | + | - | - | + | + | + | - | 7/10 | Good |
| Nga et al. (2013) [25] | + | + | + | - | - | - | + | + | + | - | 6/10 | Fair |
| Phuka et al. (2012) [26] | + | + | + | + | + | + | + | + | + | - | 9/10 | Good |
| Phuka et al. (2009) [27] | + | + | + | + | + | + | + | + | + | + | 10/10 | Good |
| Parameter | Description |
|---|---|
| Participants | Studies involving children under 5 from around the world who received food intervention for growth improvement |
| Intervention | Food-based intervention, either local food or processed food |
| Comparison | Comparison with the control group without supplementation of the target food intervention |
| Outcomes | Improvement in child linear growth or decrease in stunting condition |
| Study design | Randomized controlled trials were included in this review study |
| Author (Year) | Study Design | Participant Age Group | Sample Size; Distribution | Country | Study Settings | Study Year | Trial Registry No. | Study Limitation |
|---|---|---|---|---|---|---|---|---|
| Taneja et al. (2022) [13] | RCT | 6–12 m | 1548; M: 770 (49.7%); F: 778 (50.3%) | India | Urban Delhi | NR | CTRI/2018/04/012932 | Study was not completely blinded between 3 groups |
| Khan et al. (2020) [14] | CRT | 6–23 m | 870; M: 440 (50.6%); F: 430 (49.4%) | Pakistan | Local community in Thatta and Sujawal districts of Sindh | 2014–2016 | NCT02422953 | Large difference of age of recruited children |
| Azimi et al. (2020) [15] | RCT | 24–59 m | 100; M: 49 (49%); F: 51 (51%) | Iran | Health centers | 2017 | IRCT2017021315536N6 | Blood biomarkers could aid in a greater understanding of the RUSF’s underlying mechanisms. |
| Zambrana et al. (2019) [16] | RCT | 6–12 m | 95; M: 50 (52.6%); F: 45 (47.4%) | Nicaragua and Mali | Community health centers | 2015 | NCT02557373 | Effects of rice bran may vary (geographical location, diet, environment, and host factors) |
| Masuda and Chitundu (2019) [17] | RCT | 6–18 m | 501; NR | Zambia | Camps | 2015–2016 | NCT03523182 | Study was not blinded |
| Humphre et al. (2019) [18] | CRT | 6–18 m | 1777; NR | Zimbabwe | Rural districts | 2012–2015 | NCT01824940 | NR |
| Tang et al. (2018) [19] | RCT | 5–12 m | 64; M: 30 (47%); F: 34 (53%) | Colorado, USA | Metro area | NR | NCT02142647 | Intervention groups were compared with the WHO standards |
| Prudhon et al. (2017) [20] | RCT | 6–23 m | 2586; M: 1342 (51.9%); F: 1244 (48.1%) | Niger | Village area | 2011–2012 | NCT01828814 | Selection bias by allocation by group of nearby villages |
| Mangani et al. (2015) [21] | RCT | 6–18 m | 840; M: 419 (49.9%); F: 421 (50.1%) | Malawi | Rural area | 2008–2009 | NCT00524446 | NR |
| Maleta et al. (2015) [22] | RCT | 5.5–6.5 m | 1535; M: 58.9%; F: 41.1% | Malawi | Health center and hospital | 2009–2011 | NCT00945698 | NR |
| Christian et al. (2015) [23] | CRT | 6–18 m | 5536; M: 49.4%; F: 50.6% | Bangladesh | Rural area | 2012–2015 | NR | Study was not blinded |
| Tang and Krebs (2014) [24] | RCT | 5–6 m | 45; NR | Colorado, USA | Metropolitan area | NR | NR | Sample size was relatively small |
| Nga et al. (2013) [25] | RCT | 36–60 m | 67; NR | Vietnam | Kindergarten schools | 2010 | NR | NR |
| Phuka et al. (2012) [26] | RCT | 18 m | 163; M: 81 (49.7%); F:82 (50.3%) | Malawi | Rural area | 2005 | NCT00131209 | Lack of non-supplemented control group |
| Phuka et al. (2009) [27] | RCT | 6 months old | 182; M: 91 (50%); F: 91 (50%) | Malawi | Rural area | 2004–2008 | NCT00131209 | Lack of non-supplemented control group |
| Study | Intervention | Main Component | Duration | Frequency/Calorie | Significant Findings | Adverse Effect |
|---|---|---|---|---|---|---|
| Taneja et al. (2022) [13] | Milk–cereal mixes (2 groups: modest-protein and high-protein) Control: no food supplement | Protein, fat, and growth relevant MMN (multiple micronutrients) | 6 m | 1 packet/day; Modest-protein (2.5 g)∼125 kcal; High-protein (5.6 g)∼125 kcal | (1) Improvement in LAZ, WAZ, WAL, and MUAC in high-protein group (2) No significant improvement in modest-protein group | NR |
| Khan et al. (2020) [14] | Wawamum, a lipid-based nutrient supplement—medium quantity (LNS-MQ) Control: no food supplement | Roasted chickpeas, vegetable oil, dry skimmed milk powder, sugar, emulsifier, micronutrients, antioxidant | 6–18 m | 50 g/day (255 Kcal) | (1) The risk of stunting and wasting reduced significantly among Wawamum recipients (2) Significant reduction in anemia | NR |
| Azimi et al. (2020) [15] | Ready-to-use supplementary food (RUSF) Control: no additional food supplement, usual diet only | Soy protein isolate, whey protein, egg white, dates, vegetable oils, sugar, starch, vitamin and mineral complex | 2 m | 1–3 sachets per day (75 kcal/kg of body weight) | (1) Significant increase in weight and BMI (2) Greater daily height gain during the first month and improvement in WHZ (3) Lower prevalence of diarrhea and fever | No side effects throughout the study |
| Zambrana et al. (2019) [16] | Rice bran in food Control group: no rice bran | Rice bran without debris (rice husk, rice grain) 1 sachet > 1 g rice bran | 6 m | At 6–7, 7–8, 8–10, 10–11, 11–12 months age > 1, 2, 3, 4, 5 sachet/day, respectively | (1) Daily consumption of rice bran supported changes in LAZ from 6 to 8 and 8 to12 m of age (2) WAZ was significantly improved only for Mali infants at 8 and 12 m | No adverse events were reported in the intervention group; one case of death was reported in the control group due to respiratory infection |
| Masuda and Chitundu (2019) [17] | Spirulina in soya-maize-based porridge Control: soya-maize-based porridge without spirulina | Spirulina in mealie meal (from maize) and soya flour porridgeControl: without spirulina | 12 m | 10 g spirulina + 40 g soy per day (200.6 kcal) Control: 40 g soy per day (162 kcal) | (1) HAZ and WAZ were similar in both the intervention and control groups (2) Spirulina group had lower risk of developing a cough and were more likely to be able to walk alone at 15 m (3) positive effects on upper respiratory infection morbidity prevention and motor milestone acquisition | NR |
| Humphre et al. (2019) [18] | Small-quantity lipid-based nutrient supplement (SQ-LNSs) Control: No food supplement | NR | 12 m | 1 sachet (20 g) per day | (1) Mean LAZ and hemoglobin concentration were higher than non-intervention groups (2) Intervention did not reduce the prevalence of diarrhea | One case with congenital abnormalities complained of abdominal discomfort (possibly related) |
| Tang et al. (2018) [19] | Meat- or dairy-based complementary foods | Dairy based (yogurt, cheese stick, whey protein) Meat based (puréed ham, puréed beef, gravy) | 7 m | Total calorie intake 700 kcal/d Total protein intake 102 kcal/d (25.5 g/d) | (1) LAZ increased in the meat group and decreased in the dairy group (2) WLZ significantly increased in the dairy group | NR |
| Prudhon et al. (2017) [20] | Lipid-based nutrient supplements (LNS) Control: no control group | (a) Large-quantity LNS (LNS-LQ) Supplementary’Plumpy (b) Medium-quantity LNS (LNS-MQ) Plumpy’Doz | 15 m | (a) LNS-LQ 92 g/day (500 kcal) (b) LNS-MQ 46 g/day (247 kcal) | (1) LNS-LQ (reference) or LNS-MQ had similar effect on incidence of severe acute malnutrition, moderate acute malnutrition, severe stunting, moderate stunting, and mortality | NR |
| Mangani et al. (2015) [21] | Lipid-based nutrient supplements (LNS) Control: No food supplement | Micronutrient fortified CSB or micronutrient- fortified LNS with milk protein base (milk–LNS) or micronutrient-fortified LNS with soy protein base (soy–LNS) | 12 m | 54 g/day of soy-LNS or milk-LNS or 71 g/day of CSB (280 kcal per day) | (1) No conclusive evidence on a relationship between the LNS supplementation and reduction of stunting | NR |
| Maleta et al. (2015) [22] | Milk containing LNSs or milk-free LNSs or corn–soy blend (CSB) Control: No food supplement | Soybean oil, dry skimmed-milk powder (or maltodextrine), peanut paste, micronutrients, sugar | 12 m | 10, 20, or 40 g/day milk containing LNSs or 20 or 40 g/day milk-free LNSs or 71 g/day CSB Calories: 55–241 kcal | (1) LNS supplementation during infancy and childhood promotes length gain or prevents stunting between 6 and 18 m of age in Malawi | NR |
| Christian et al. (2015) [23] | Ready-to-use supplementary foods (RUSF) and a fortified blended food Control: No food supplement | RUSF (rice–lentil, chickpea-based), Plumpy doz, wheat-soy-blend plus plus (WSB++), or Super Cereal Plus (SC+) | 12 m | Plumpy’doz: 46 g or half dose; 28 g of rice–lentil and 23 g of chickpea product Calories: 125 or 250 kcal/day | (1) Deceleration in LAZ was lower in the Plumpy’doz, rice–lentil, and chickpea groups relative to control (2) WLZ decline was lower only in Plumpy’doz and chickpea groups (3) WSB++ had no significant impact (4) Stunting prevalence was 44% in control but lower by 5–6% in Plumpy’doz and chickpea groups | NR |
| Tang and Krebs (2014) [24] | Meat or cereal | Meat (pureed meat and gravy), iron- and zinc-fortified cereal, or iron-only-fortified cereal | 5 m | Meat: total 71 g/day (equivalent to 8 g protein) Cereals: 1 serving/day (15 g) | (1) Higher protein intake from meats was associated with greater linear growth and weight gain | No adverse effects were reported during the study |
| Nga et al. (2013) [25] | Ready-to-use-therapeutic-foods (RUTF) in form of a compressed bar | Mung and soy beans, rice, sesame, sugar, whole milk powder, whey protein, vegetable fat, vegetable oil, and a premix | 1 m | 1 sachet (500–530 kcal) of RUTF/meal (total 2 meals), providing around 1000 kcal/day | (1) The nutritional status of the children improved significantly; increases in WHZ and HAZ z-scores | Nausea, vomiting, rash, and diarrhea were measured but the prevalences were too low for any statistical analysis |
| Phuka et al. (2012) [26] | Lipid-based nutrient supplements (LNS) or corn–soy flour (control) | Micronutrient fortified LNS spread or micronutrient fortified corn–soy flour (CSF) | 12 m | CSF 70 g/day (282 Kcal) or LNS spread 25 g/day (130 Kcal) or LNS 50 g/day (264 Kcal) | (1) Daily supplementation of diet with LNS or CSF have comparable development outcomes by 18 m of age | NR |
| Phuka et al. (2009) [27] | Lipid-based nutrient supplements (LNS) or maize–soy flour (control) | LNS as micronutrient fortified spread (FS) or micronutrient fortified maize–soy flour | 12 m | Maize–soy flour 71 g/day (282 Kcal) or FS 25 g/day (130 Kcal) or FS 50 g/day (264 Kcal) | (1) FS 50 g/day is likely to have a positive and sustained impact on severe stunting (2) Half-dose intervention may not have the same effect | NR |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mamun, A.A.; Mahmudiono, T.; Yudhastuti, R.; Triatmaja, N.T.; Chen, H.-L. Effectiveness of Food-Based Intervention to Improve the Linear Growth of Children under Five: A Systematic Review and Meta-Analysis. Nutrients 2023, 15, 2430. https://doi.org/10.3390/nu15112430
Mamun AA, Mahmudiono T, Yudhastuti R, Triatmaja NT, Chen H-L. Effectiveness of Food-Based Intervention to Improve the Linear Growth of Children under Five: A Systematic Review and Meta-Analysis. Nutrients. 2023; 15(11):2430. https://doi.org/10.3390/nu15112430
Chicago/Turabian StyleMamun, Abdullah Al, Trias Mahmudiono, Ririh Yudhastuti, Nining Tyas Triatmaja, and Hsiu-Ling Chen. 2023. "Effectiveness of Food-Based Intervention to Improve the Linear Growth of Children under Five: A Systematic Review and Meta-Analysis" Nutrients 15, no. 11: 2430. https://doi.org/10.3390/nu15112430
APA StyleMamun, A. A., Mahmudiono, T., Yudhastuti, R., Triatmaja, N. T., & Chen, H.-L. (2023). Effectiveness of Food-Based Intervention to Improve the Linear Growth of Children under Five: A Systematic Review and Meta-Analysis. Nutrients, 15(11), 2430. https://doi.org/10.3390/nu15112430