Impact of a Digital Lifestyle Intervention on Diabetes Self-Management: A Pilot Study
Abstract
:1. Introduction
2. Materials and Methods
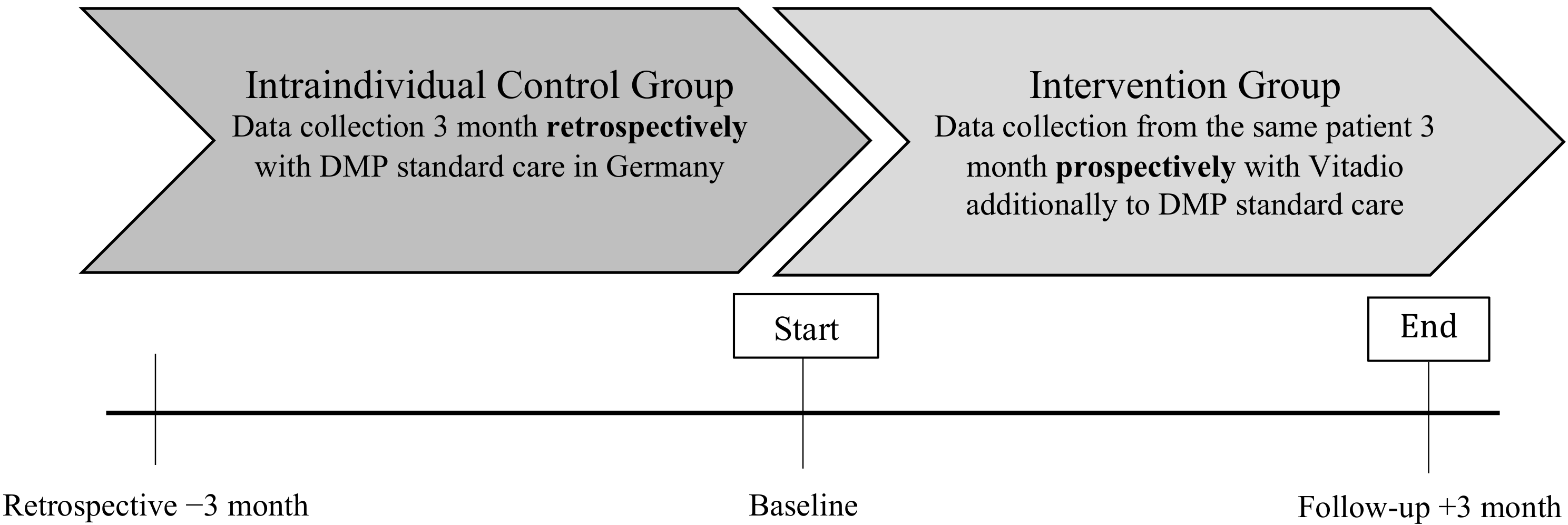
2.1. Study Design
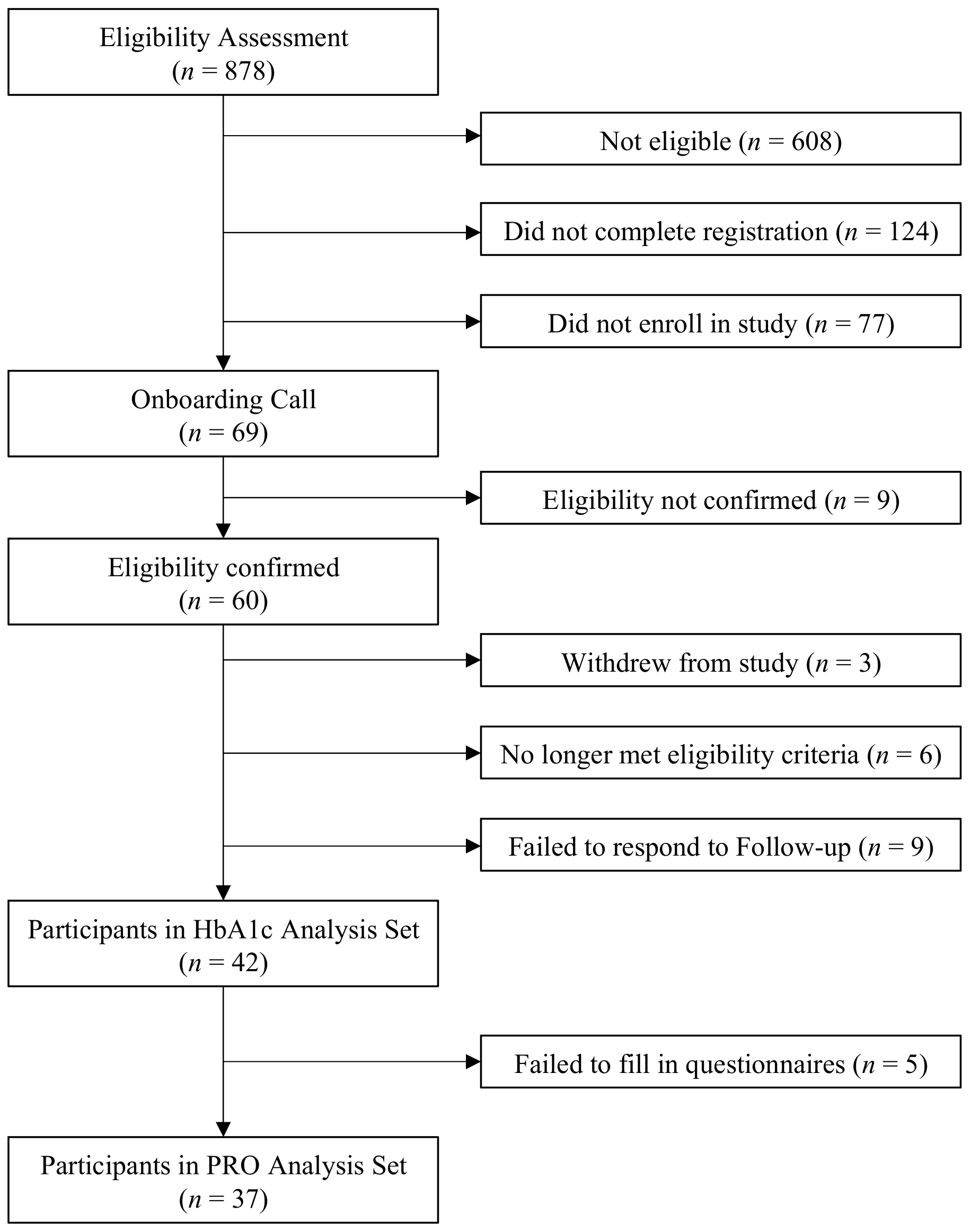
2.2. Participants
2.2.1. Eligibility
2.2.2. Recruitment
2.3. Study Procedures
2.4. Intervention
2.5. Comparator
2.6. Outcome Measurement
2.7. Sample Size
2.8. Statistical Analysis
3. Results
3.1. Participant Characteristics
3.2. Effects on Glycemic Control
3.3. Effects on Metabolic Parameters
3.4. Effects on Patient Reported Outcomes
3.5. Effects on App Reported Data
3.5.1. Food Intake
3.5.2. Physical Activity
3.5.3. Self-Management
4. Discussion
Strengths & Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. Screenshots of the App


Appendix B. Meal Photos


References
- Robert Koch-Institut. Übergewicht und Adipositas in Deutschland: Werden wir immer dicker? Bundesgesundheitsblatt–Gesundh. –Gesundh. 2012, 55, 3–4. [Google Scholar]
- Tönnies, T.; Röckl, S.; Hoyer, A.; Heidemann, C.; Baumert, J.; Du, Y.; Scheidt-Nave, C.; Brinks, R. Projected Number of People with Diagnosed Type 2 Diabetes in Germany in 2040. Diabet. Med. J. Br. Diabet. Assoc. 2019, 36, 1217–1225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmidt, C.; Reitzle, L.; Paprott, R.; Bätzing, J.; Holstiege, J. Diabetes mellitus und Komorbiditäten—Querschnittsstudie mit Kontrollgruppe anhand vertragsärztlicher Abrechnungsdaten. Robert Koch Inst. 2021, 6, 20–37. [Google Scholar] [CrossRef]
- Haftenberger, M.; Mensink, G.B.M.; Herzog, B.; Kluttig, A.; Greiser, K.H.; Merz, B.; Nöthlings, U.; Schlesinger, S.; Vogt, S.; Thorand, B.; et al. Changes in Body Weight and Obesity Status in German Adults: Results of Seven Population-Based Prospective Studies. Eur. J. Clin. Nutr. 2016, 70, 300–305. [Google Scholar] [CrossRef]
- American Diabetes Association 3. Prevention or Delay of Type 2 Diabetes: Standards of Medical Care in Diabetes—2021. Diabetes Care 2020, 44, S34–S39. [Google Scholar] [CrossRef]
- Kerrison, G.; Gillis, R.B.; Jiwani, S.I.; Alzahrani, Q.; Kok, S.; Harding, S.E.; Shaw, I.; Adams, G.G. The Effectiveness of Lifestyle Adaptation for the Prevention of Prediabetes in Adults: A Systematic Review. J. Diabetes Res. 2017, 2017, 1–20. [Google Scholar] [CrossRef]
- Sun, Y.; You, W.; Almeida, F.; Estabrooks, P.; Davy, B. The Effectiveness and Cost of Lifestyle Interventions Including Nutrition Education for Diabetes Prevention: A Systematic Review and Meta-Analysis. J. Acad. Nutr. Diet. 2017, 117, 404–421.e36. [Google Scholar] [CrossRef] [Green Version]
- Bonoto, B.C.; de Araújo, V.E.; Godói, I.P.; de Lemos, L.L.P.; Godman, B.; Bennie, M.; Diniz, L.M.; Junior, A.A.G. Efficacy of Mobile Apps to Support the Care of Patients With Diabetes Mellitus: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. JMIR MHealth UHealth 2017, 5, e6309. [Google Scholar] [CrossRef]
- Martos-Cabrera, M.B.; Velando-Soriano, A.; Pradas-Hernández, L.; Suleiman-Martos, N.; Cañadas-De la Fuente, G.A.; Albendín-García, L.; Gómez-Urquiza, J.L. Smartphones and Apps to Control Glycosylated Hemoglobin (HbA1c) Level in Diabetes: A Systematic Review and Meta-Analysis. J. Clin. Med. 2020, 9, 693. [Google Scholar] [CrossRef] [Green Version]
- American Diabetes Association 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes—2020. Diabetes Care 2019, 43, S14–S31. [Google Scholar] [CrossRef] [Green Version]
- Cosentino, F.; Grant, P.J.; Aboyans, V.; Bailey, C.J.; Ceriello, A.; Delgado, V.; Federici, M.; Filippatos, G.; Grobbee, D.E.; Hansen, T.B.; et al. 2019 ESC Guidelines on Diabetes, Pre-Diabetes, and Cardiovascular Diseases Developed in Collaboration with the EASD: The Task Force for Diabetes, Pre-Diabetes, and Cardiovascular Diseases of the European Society of Cardiology (ESC) and the European Association for the Study of Diabetes (EASD). Eur. Heart J. 2020, 41, 255–323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- American Diabetes Association 4. Foundations of Care: Education, Nutrition, Physical Activity, Smoking Cessation, Psychosocial Care, and Immunization. Diabetes Care 2015, 38, S20–S30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Landgraf, R.; Aberle, J.; Birkenfeld, A.L.; Gallwitz, B.; Kellerer, M.; Klein, H.; Müller-Wieland, D.; Nauck, M.A.; Reuter, H.-M.; Siegel, E. Therapy of Type 2 Diabetes. Exp. Clin. Endocrinol. Diabetes 2019, 127, S73–S92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schwarz, P.E.H.; Timpel, P.; Harst, L.; Greaves, C.J.; Ali, M.K.; Lambert, J.; Weber, M.B.; Almedawar, M.M.; Morawietz, H. Blood Sugar Regulation as a Key Focus for Cardiovascular Health Promotion and Prevention: An Umbrella Review. J. Am. Coll. Cardiol. 2018, 72, 1829–1844. [Google Scholar] [CrossRef]
- Grock, S.; Ku, J.; Kim, J.; Moin, T. A Review of Technology-Assisted Interventions for Diabetes Prevention. Curr. Diab. Rep. 2017, 17, 107. [Google Scholar] [CrossRef]
- Årsand, E.; Frøisland, D.H.; Skrøvseth, S.O.; Chomutare, T.; Tatara, N.; Hartvigsen, G.; Tufano, J.T. Mobile Health Applications to Assist Patients with Diabetes: Lessons Learned and Design Implications. J. Diabetes Sci. Technol. 2012, 6, 1197–1206. [Google Scholar] [CrossRef] [Green Version]
- Kaufman, N.; Khurana, I. Using Digital Health Technology to Prevent and Treat Diabetes. Diabetes Technol. Ther. 2016, 18, S-56–S-68. [Google Scholar] [CrossRef] [Green Version]
- Byambasuren, O.; Sanders, S.; Beller, E.; Glasziou, P. Prescribable MHealth Apps Identified from an Overview of Systematic Reviews. Npj Digit. Med. 2018, 1, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Timpel, P.; Oswald, S.; Schwarz, P.E.H.; Harst, L. Mapping the Evidence on the Effectiveness of Telemedicine Interventions in Diabetes, Dyslipidemia, and Hypertension: An Umbrella Review of Systematic Reviews and Meta-Analyses. J. Med. Internet Res. 2020, 22, e16791. [Google Scholar] [CrossRef]
- Kumar, S.; Moseson, H.; Uppal, J.; Juusola, J.L. A Diabetes Mobile App with In-App Coaching From a Certified Diabetes Educator Reduces A1C for Individuals with Type 2 Diabetes. Diabetes Educ. 2018, 44, 226–236. [Google Scholar] [CrossRef]
- Cafazzo, J.A.; Casselman, M.; Hamming, N.; Katzman, D.K.; Palmert, M.R. Design of an MHealth App for the Self-Management of Adolescent Type 1 Diabetes: A Pilot Study. J. Med. Internet Res. 2012, 14, e70. [Google Scholar] [CrossRef] [PubMed]
- Pagoto, S.; Tulu, B.; Waring, M.E.; Goetz, J.; Bibeau, J.; Divito, J.; Groshon, L.; Schroeder, M. Slip Buddy App for Weight Management: Randomized Feasibility Trial of a Dietary Lapse Tracking App. JMIR MHealth UHealth 2021, 9, e24249. [Google Scholar] [CrossRef] [PubMed]
- Riangkam, C.; Sriyuktasuth, A.; Pongthavornkamol, K.; Kusakunniran, W.; Sriwijitkamol, A. Effects of a Mobile Health Diabetes Self-Management Program on HbA1C, Self-Management and Patient Satisfaction in Adults with Uncontrolled Type 2 Diabetes: A Randomized Controlled Trial. J. Health Res. 2021, 2. [Google Scholar] [CrossRef]
- Turner, R.M.; Ma, Q.; Lorig, K.; Greenberg, J.; DeVries, A.R. Evaluation of a Diabetes Self-Management Program: Claims Analysis on Comorbid Illnesses, Health Care Utilization, and Cost. J. Med. Internet Res. 2018, 20, e207. [Google Scholar] [CrossRef]
- Ware, J.; Kosinski, M.; Keller, S.D. A 12-Item Short-Form Health Survey: Construction of Scales and Preliminary Tests of Reliability and Validity. Med. Care 1996, 34, 220–233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ware, J.; Kosinski, M.; Turner-Bowker, D.; Gandek, B. SF-12: How to Score SF-12 Physical and Mental Health Summary Scales, 2nd ed.; The Health Institute, New England Medical Center: Boston, MA, USA, 2002. [Google Scholar]
- Toobert, D.J.; Hampson, S.E.; Glasgow, R.E. The Summary of Diabetes Self-Care Activities Measure: Results from 7 Studies and a Revised Scale. Diabetes Care 2000, 23, 943–950. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kroenke, K.; Spitzer, R.L.; Williams, J.B. The PHQ-9: Validity of a Brief Depression Severity Measure. J. Gen. Intern. Med. 2001, 16, 606–613. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Lang, A.-G.; Buchner, A. G*Power 3: A Flexible Statistical Power Analysis Program for the Social, Behavioral, and Biomedical Sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Buchner, A.; Lang, A.-G. Statistical Power Analyses Using G*Power 3.1: Tests for Correlation and Regression Analyses. Behav. Res. Methods 2009, 41, 1149–1160. [Google Scholar] [CrossRef] [Green Version]
- Meyerowitz-Katz, G.; Ravi, S.; Arnolda, L.; Feng, X.; Maberly, G.; Astell-Burt, T. Rates of Attrition and Dropout in App-Based Interventions for Chronic Disease: Systematic Review and Meta-Analysis. J. Med. Internet Res. 2020, 22, e20283. [Google Scholar] [CrossRef]
- Jersey, D.M.L. PharmD, BCPS, BCPP, BCGP Medical Writer/Consultant Adjunct Faculty, Union County College Division of Allied Health Plainfield, New Applying Recent A1C Recommendations in Clinical Practice. Available online: https://www.uspharmacist.com/article/applying-recent-a1c-recommendations-in-clinical-practice (accessed on 14 December 2021).
- Jamieson, S. Likert Scales: How to (Ab)Use Them. Med. Educ. 2004, 38, 1217–1218. [Google Scholar] [CrossRef] [PubMed]
- Likert, R. A Technique for the Measurement of Attitudes. Arch. Psychol. 1932, 22, 55. [Google Scholar]
- Monteiro, C.A.; Cannon, G.; Levy, R.B.; Moubarac, J.-C.; Louzada, M.L.; Rauber, F.; Khandpur, N.; Cediel, G.; Neri, D.; Martinez-Steele, E.; et al. Ultra-Processed Foods: What They Are and How to Identify Them. Public Health Nutr. 2019, 22, 936–941. [Google Scholar] [CrossRef] [PubMed]
- Althoff, T.; Sosič, R.; Hicks, J.L.; King, A.C.; Delp, S.L.; Leskovec, J. Large-Scale Physical Activity Data Reveal Worldwide Activity Inequality. Nature 2017, 547, 336–339. [Google Scholar] [CrossRef] [PubMed]
- Glycemic Targets: Standards of Medical Care in Diabetes—2021|Diabetes Care. Available online: https://care.diabetesjournals.org/content/44/Supplement_1/S73 (accessed on 9 December 2021).
- Stühmann, L.M.; Paprott, R.; Heidemann, C.; Baumert, J.; Hansen, S.; Zahn, D.; Scheidt-Nave, C.; Gellert, P. Health App Use and Its Correlates Among Individuals with and Without Type 2 Diabetes: Nationwide Population-Based Survey. JMIR Diabetes 2020, 5, e14396. [Google Scholar] [CrossRef]
- Stratton, I.M.; Adler, A.I.; Neil, H.A.W.; Matthews, D.R.; Manley, S.E.; Cull, C.A.; Hadden, D.; Turner, R.C.; Holman, R.R. Association of Glycaemia with Macrovascular and Microvascular Complications of Type 2 Diabetes (UKPDS 35): Prospective Observational Study. BMJ 2000, 321, 405–412. [Google Scholar] [CrossRef] [Green Version]
- Selvin, E.; Marinopoulos, S.; Berkenblit, G.; Rami, T.; Brancati, F.L.; Powe, N.R.; Golden, S.H. Meta-Analysis: Glycosylated Hemoglobin and Cardiovascular Disease in Diabetes Mellitus. Ann. Intern. Med. 2004, 141, 421–431. [Google Scholar] [CrossRef]
- Wang, P.; Huang, R.; Lu, S.; Xia, W.; Sun, H.; Sun, J.; Cai, R.; Wang, S. HbA1c below 7% as the Goal of Glucose Control Fails to Maximize the Cardiovascular Benefits: A Meta-Analysis. Cardiovasc. Diabetol. 2015, 14, 124. [Google Scholar] [CrossRef] [Green Version]
- Balkau, B.; Picard, P.; Vol, S.; Fezeu, L.; Eschwège, E. Consequences of Change in Waist Circumference on Cardiometabolic Risk Factors Over 9 Years: Data from an Epidemiological Study on the Insulin Resistance Syndrome (DESIR). Diabetes Care 2007, 30, 1901–1903. [Google Scholar] [CrossRef] [Green Version]
- Benjamin, E.J.; Blaha, M.J.; Chiuve, S.E.; Cushman, M.; Das, S.R.; Deo, R.; de Ferranti, S.D.; Floyd, J.; Fornage, M.; Gillespie, C.; et al. Heart Disease and Stroke Statistics—2017 Update: A Report from the American Heart Association. Circulation 2017, 135, e146–e603. [Google Scholar] [CrossRef]
- Yu, E.; Malik, V.S.; Hu, F.B. Cardiovascular Disease Prevention by Diet Modification: JACC Health Promotion Series. J. Am. Coll. Cardiol. 2018, 72, 914–926. [Google Scholar] [CrossRef] [PubMed]
- Shan, Z.; Li, Y.; Baden, M.Y.; Bhupathiraju, S.N.; Wang, D.D.; Sun, Q.; Rexrode, K.M.; Rimm, E.B.; Qi, L.; Willett, W.C.; et al. Association Between Healthy Eating Patterns and Risk of Cardiovascular Disease. JAMA Intern. Med. 2020, 180, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Blonde, L.; Brunton, S.A.; Chava, P.; Zhou, R.; Meyers, J.; Davis, K.L.; Dalal, M.R.; DiGenio, A. Achievement of Target A1C <7.0% (<53 Mmol/Mol) by U.S. Type 2 Diabetes Patients Treated With Basal Insulin in Both Randomized Controlled Trials and Clinical Practice. Diabetes Spectr. 2019, 32, 93–103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mack, C.; Su, Z.; Westreich, D. Managing Missing Data in Patient Registries: Addendum to Registries for Evaluating Patient Outcomes: A User’s Guide, 3rd ed.; AHRQ Methods for Effective Health Care; Agency for Healthcare Research and Quality (US): Rockville, MD, USA, 2018. [Google Scholar]
- Jakobsen, J.C.; Gluud, C.; Wetterslev, J.; Winkel, P. When and How Should Multiple Imputation Be Used for Handling Missing Data in Randomised Clinical Trials—A Practical Guide with Flowcharts. BMC Med. Res. Methodol. 2017, 17, 162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Winkel, P. International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. In Handbook of Transnational Economic Governance Regimes; Tietje, C., Brouder, A., Eds.; Brill | Nijhoff: Leiden, The Netherlands, 2010; pp. 1041–1053. ISBN 978-90-04-18156-4. [Google Scholar]
- Shan, R.; Sarkar, S.; Martin, S.S. Digital Health Technology and Mobile Devices for the Management of Diabetes Mellitus: State of the Art. Diabetologia 2019, 62, 877–887. [Google Scholar] [CrossRef] [Green Version]
- Neugebauer, E.A.M.; Rath, A.; Antoine, S.-L.; Eikermann, M.; Seidel, D.; Koenen, C.; Jacobs, E.; Pieper, D.; Laville, M.; Pitel, S.; et al. Specific Barriers to the Conduct of Randomised Clinical Trials on Medical Devices. Trials 2017, 18, 427. [Google Scholar] [CrossRef] [Green Version]
- Timpel, P.; Cesena, F.H.Y.; da Silva Costa, C.; Soldatelli, M.D.; Gois, E.; Castrillon, E.; Díaz, L.J.J.; Repetto, G.M.; Hagos, F.; Castillo Yermenos, R.E.; et al. Efficacy of Gamification-Based Smartphone Application for Weight Loss in Overweight and Obese Adolescents: Study Protocol for a Phase II Randomized Controlled Trial. Ther. Adv. Endocrinol. Metab. 2018, 9, 167–176. [Google Scholar] [CrossRef]
- Smith, B.; Magnani, J.W. New Technologies, New Disparities: The Intersection of Electronic Health and Digital Health Literacy. Int. J. Cardiol. 2019, 292, 280–282. [Google Scholar] [CrossRef]
| Retrospective | Baseline | Follow-Up | Change | p-Value | |
|---|---|---|---|---|---|
| −3 months | +3 months | ||||
| n = 42 * | |||||
| HbA1c (%) Control group | 8.2 ± 1.3 | 7.9 ± 1.0 | - | −0.3 ± 1.1 | 0.27 |
| HbA1c (%) Intervention group | - | 7.9 ± 1.0 | 6.9 ± 0.9 | −0.9 ± 1.1 | <0.001 |
| n = 37 * | |||||
| Intervention group | |||||
| Weight (kg) | - | 105.2 ± 18.5 | 100.9 ± 17.6 | −4.3 ± 4.5 | <0.001 |
| BMI (kg/m2) | - | 35.1 ± 7.3 | 33.6 ± 7.1 | −1.4 ± 1.5 | <0.001 |
| Waist circumference (cm) | - | 121.1 ± 16.5 | 115.4 ± 17.4 | −5.7 ± 15.0 | 0.03 |
| Fasting glucose (mmol/L) | - | 7.4 ± 1.4 | 6.8 ± 1.5 | −0.6 ± 1.3 | 0.01 |
| Baseline HbA1c (%) | Follow-Up HbA1c (%) | Change in HbA1c (%) | p-Value | p-Value between Groups | |
|---|---|---|---|---|---|
| n = 42 | |||||
| <55 years (n = 12) | 8.42 ± 0.95 | 7.09 ± 1.12 | −1.32 ± 1.25 | 0.004 | 0.20 |
| >55 years (n = 30) | 7.66 ± 0.97 | 6.88 ± 0.78 | −0.78 ± 0.98 | <0.001 | |
| n = 42 | |||||
| Baseline HbA1c < 8% (n = 24) | 7.17 ± 0.44 | 6.74 ± 0.76 | −0.43 ± 0.75 | 0.01 | <0.001 |
| Baseline HbA1c > 8% (n = 18) | 8.81 ± 0.77 | 7.21 ± 0.97 | −1.61 ± 1.1 | <0.001 | |
| n = 42 | |||||
| Baseline BMI < 30 (n = 12) | 7.68 ± 1.05 | 7.07 ± 0.77 | −0.6 ± 0.98 | 0.06 | 0.16 |
| Baseline BMI > 30 (n = 30) | 7.99 ± 1 | 6.89 ± 0.94 | −1.10 ± 1.11 | <0.001 | |
| n = 26 * | |||||
| Duration < 8.5 years (n = 15) | 7.6 ± 1.14 | 6.76 ± 0.88 | −0.84 ± 1.31 | 0.008 | 0.58 |
| Duration > 8.5 years (n = 11) | 8.21 ± 0.64 | 7.1 ± 1.04 | −1.12 ± 1.19 | 0.03 | |
| n = 42 | |||||
| Male (n = 23) | 7.88 ± 0.98 | 6.86 ± 0.88 | −1.02 ± 1.1 | <0.001 | 0.57 |
| Female (n = 19) | 7.86 ± 1.08 | 7.04 ± 0.91 | −0.83 ± 1.07 | 0.003 |
| Baseline | Follow-Up | p-Value | |
|---|---|---|---|
| +3 months | |||
| n = 37 | |||
| PHQ-9 | |||
| Depression severity (n) | 0.36 | ||
| Minimal | 15 | 16 | |
| Mild | 13 | 15 | |
| Moderate | 5 | 1 | |
| Moderately severe | 3 | 3 | |
| Severe | 1 | 2 | |
| SF-12 | |||
| PCS score | 42.1 ± 9.6 | 45.4 ± 9.1 | 0.01 |
| MCS score | 42.1 ± 12.6 | 45.1 ± 13.6 | 0.06 |
| SDSCA | |||
| General Diet | 5.3 ± 1.2 | 5.5 ± 1.3 | 0.30 |
| Specific Diet | 4.6 ± 1.5 | 4.5 ± 1.7 | 0.77 |
| Exercise | 3.7 ± 2.1 | 4.2 ± 1.8 | 0.10 |
| Blood-Glucose Testing | 4.7 ± 2.9 | 4.6 ± 2.9 | 0.50 |
| Footcare | 2.4 ± 2.4 | 2.3 ± 2.4 | 0.70 |
| Overall Scale | 4.1 ± 1.2 | 4.2 ± 1.2 | 0.47 |
| Baseline | Follow-Up | Change | p-Value | |
|---|---|---|---|---|
| +3 months | ||||
| Meal Evaluation * | ||||
| n = 24 | ||||
| Portion size | 2.44 ± 0.40 | 2.18 ± 0.36 | −0.26 ± 0.46 | 0.01 |
| Protein | 2.61 ± 0.56 | 2.28 ± 0.47 | −0.32 ± 0.67 | 0.03 |
| Carbohydrate | 3.07 ± 0.51 | 2.68 ± 0.56 | −0.38 ± 0.71 | 0.01 |
| Fat | 2.96 ± 0.69 | 2.59 ± 0.50 | −0.37 ± 0.72 | 0.02 |
| Fiber | 3.33 ± 0.56 | 2.82 ± 0.54 | −0.51 ± 0.63 | <0.001 |
| Vegetable | 3.31 ± 0.92 | 2.67 ± 1.00 | −0.64 ± 0.95 | 0.003 |
| Processed food | 2.10 ± 0.50 | 1.80 ± 0.43 | −0.30 ± 0.50 | 0.007 |
| Overall grade | 3.08 ± 0.39 | 2.71 ± 0.36 | −0.36 ± 0.42 | <0.001 |
| Self-efficacy | ||||
| n = 29 | ||||
| In-app questionnaire ** | ||||
| Ability to select proper food | 4.86 ± 1.67 | 6.85 ± 1.74 | 1.99 ± 1.75 | <0.001 |
| Ability to be more active | 6.45 ± 2.16 | 7.76 ± 2.42 | 1.31 ± 2.32 | 0.005 |
| Diabetes management | 5.89 ± 2.47 | 7.76 ± 2.42 | 1.89 ± 2.50 | <0.001 |
| Weight Change | |
|---|---|
| Constant | 8.286 (3.057) * |
| Lesson reading time | −0.392 (0.183) * |
| Habit compliance | −0.068 (3.341) * |
| Self-monitoring | −0.166 (0.028) ** |
| Observations | 41 |
| R2 | 0.43 |
| Adjusted R2 | 0.39 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bretschneider, M.P.; Klásek, J.; Karbanová, M.; Timpel, P.; Herrmann, S.; Schwarz, P.E.H. Impact of a Digital Lifestyle Intervention on Diabetes Self-Management: A Pilot Study. Nutrients 2022, 14, 1810. https://doi.org/10.3390/nu14091810
Bretschneider MP, Klásek J, Karbanová M, Timpel P, Herrmann S, Schwarz PEH. Impact of a Digital Lifestyle Intervention on Diabetes Self-Management: A Pilot Study. Nutrients. 2022; 14(9):1810. https://doi.org/10.3390/nu14091810
Chicago/Turabian StyleBretschneider, Maxi Pia, Jan Klásek, Martina Karbanová, Patrick Timpel, Sandra Herrmann, and Peter E. H. Schwarz. 2022. "Impact of a Digital Lifestyle Intervention on Diabetes Self-Management: A Pilot Study" Nutrients 14, no. 9: 1810. https://doi.org/10.3390/nu14091810
APA StyleBretschneider, M. P., Klásek, J., Karbanová, M., Timpel, P., Herrmann, S., & Schwarz, P. E. H. (2022). Impact of a Digital Lifestyle Intervention on Diabetes Self-Management: A Pilot Study. Nutrients, 14(9), 1810. https://doi.org/10.3390/nu14091810







