Intestinal Dysbiosis in Patients with Histamine Intolerance
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Intestinal Microbiota and Histamine Concentration Analysis in Stool Samples
2.3. Statistical Analysis
3. Results and Discussion
3.1. Participant Characteristics
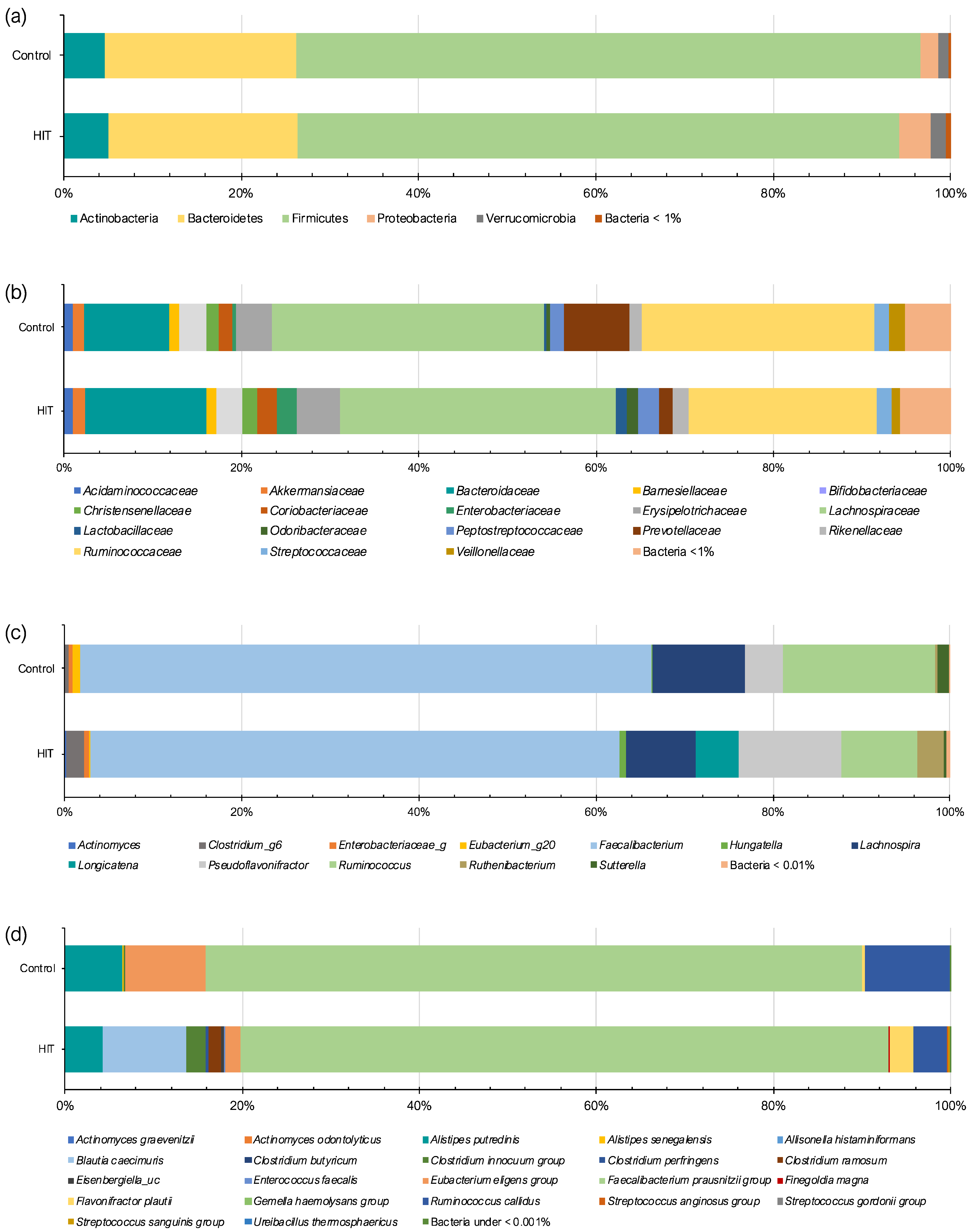
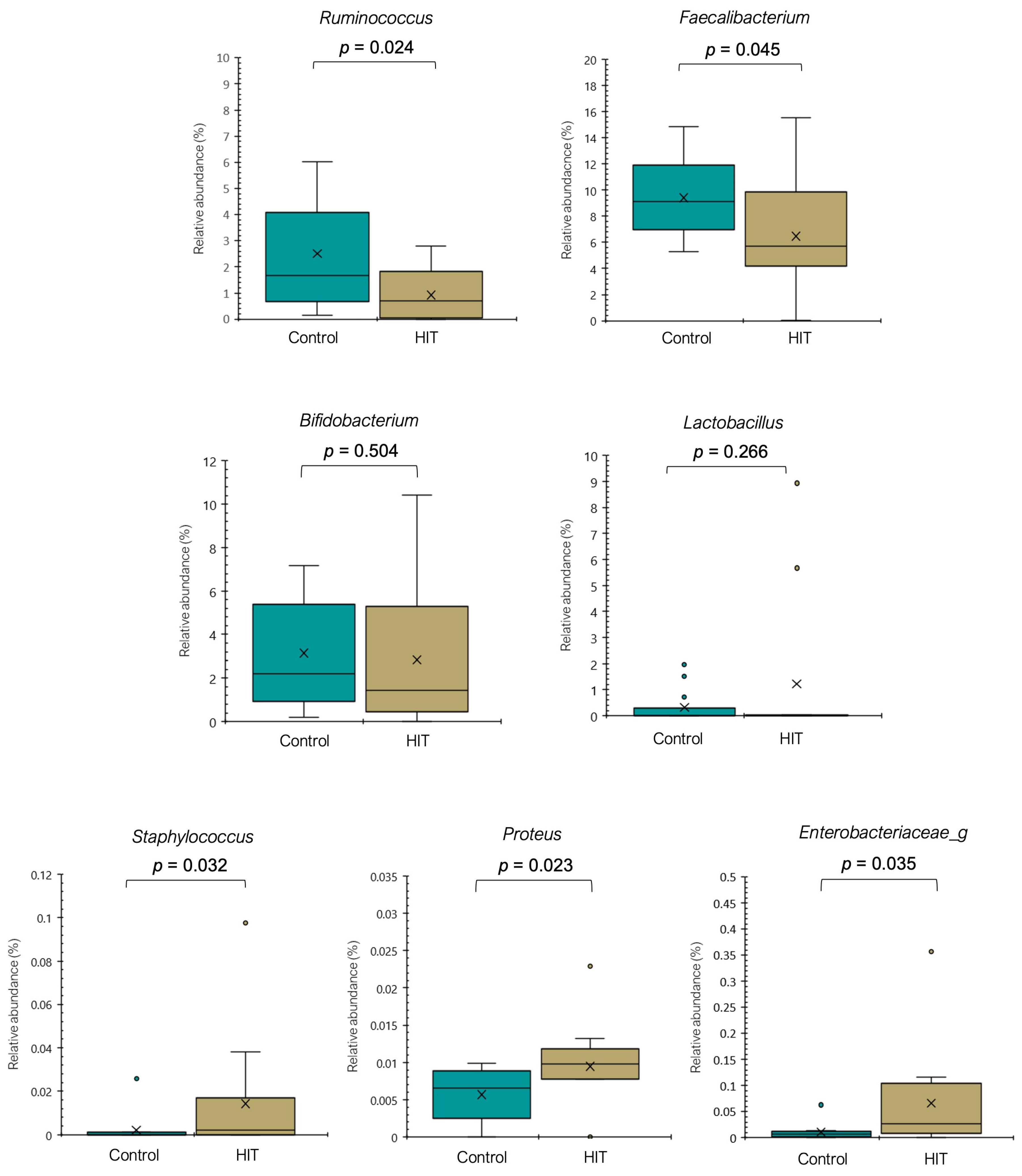
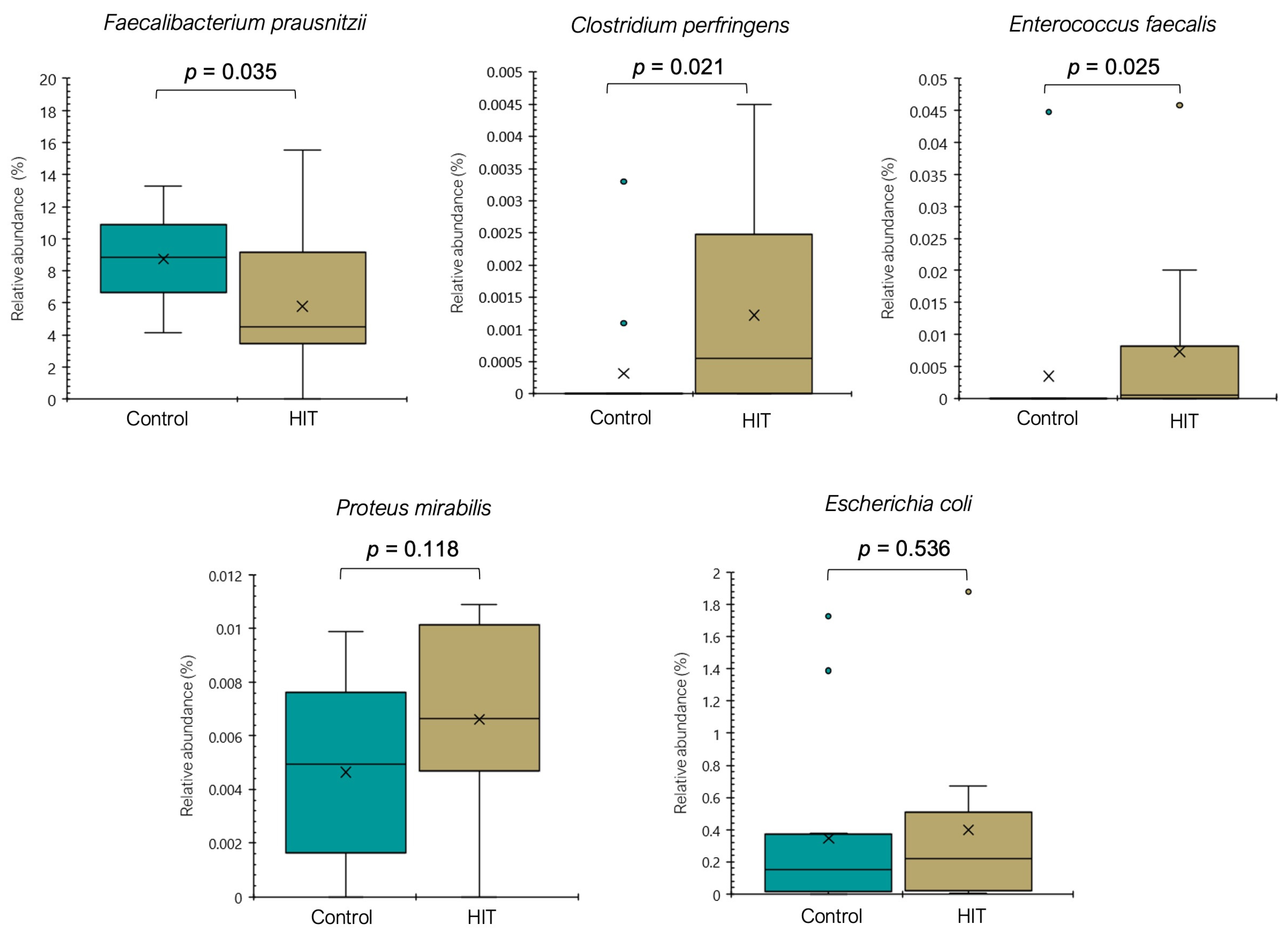
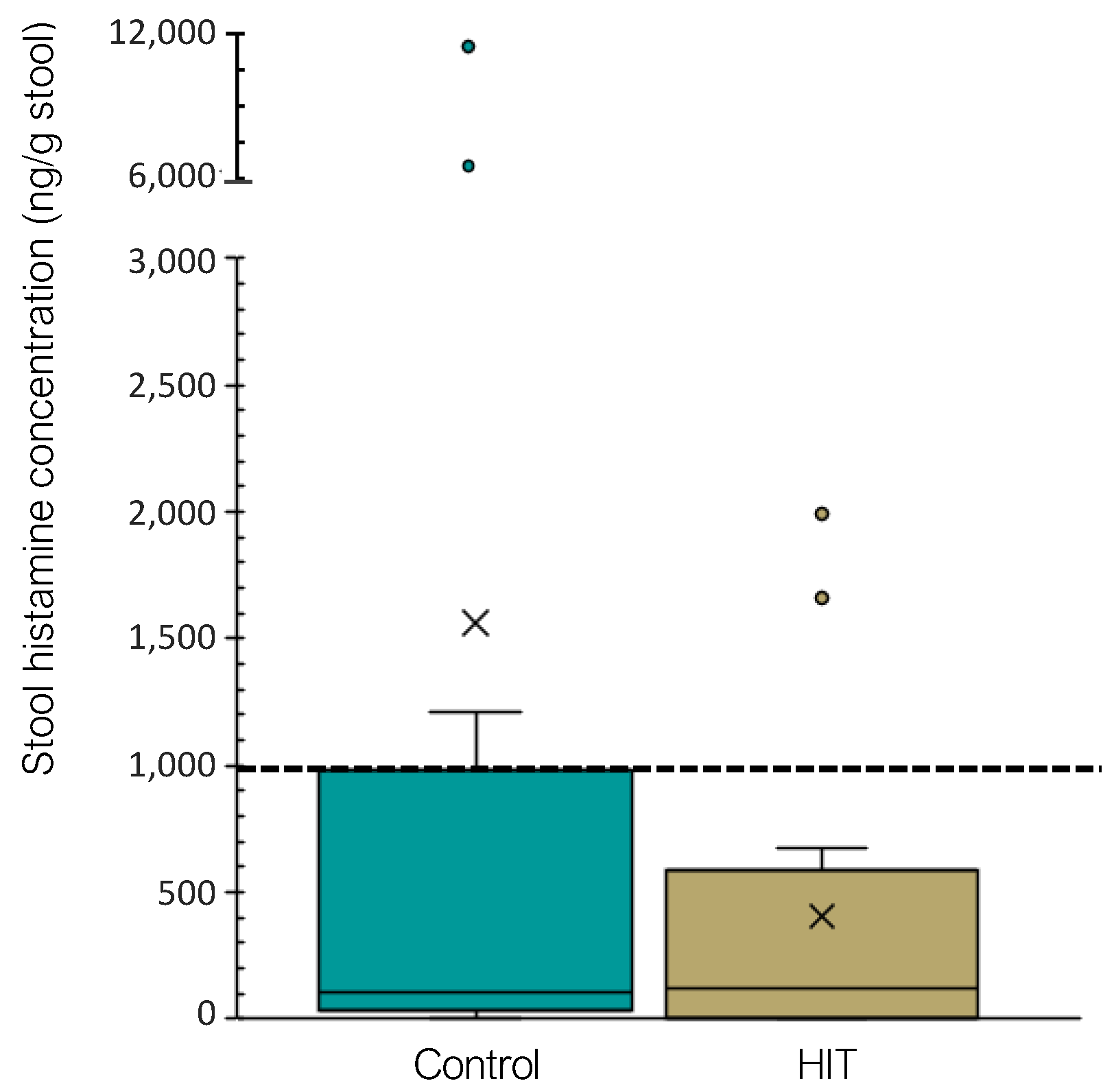
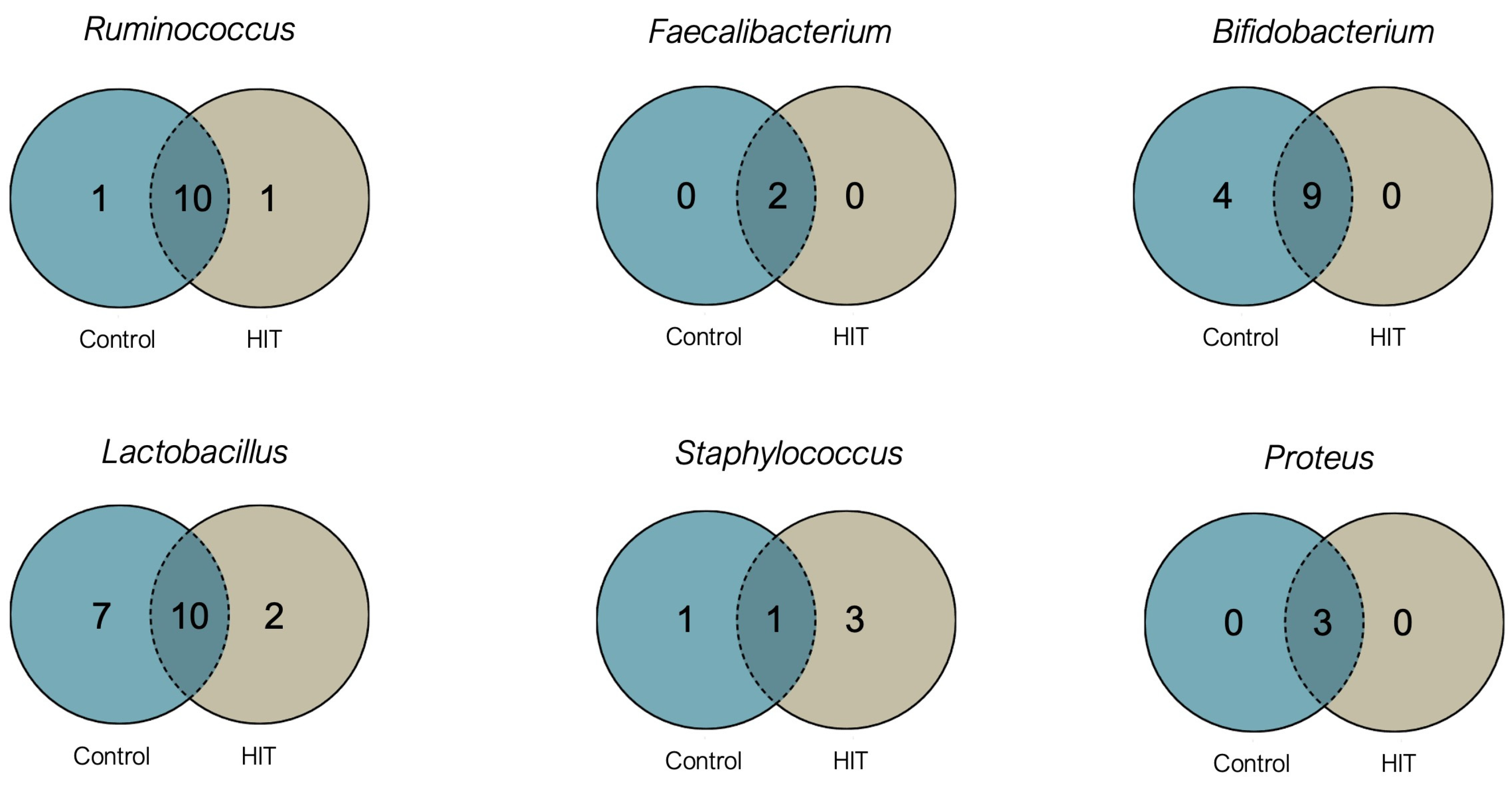
3.2. Intestinal Microbiota Composition and Stool Histamine Concentration
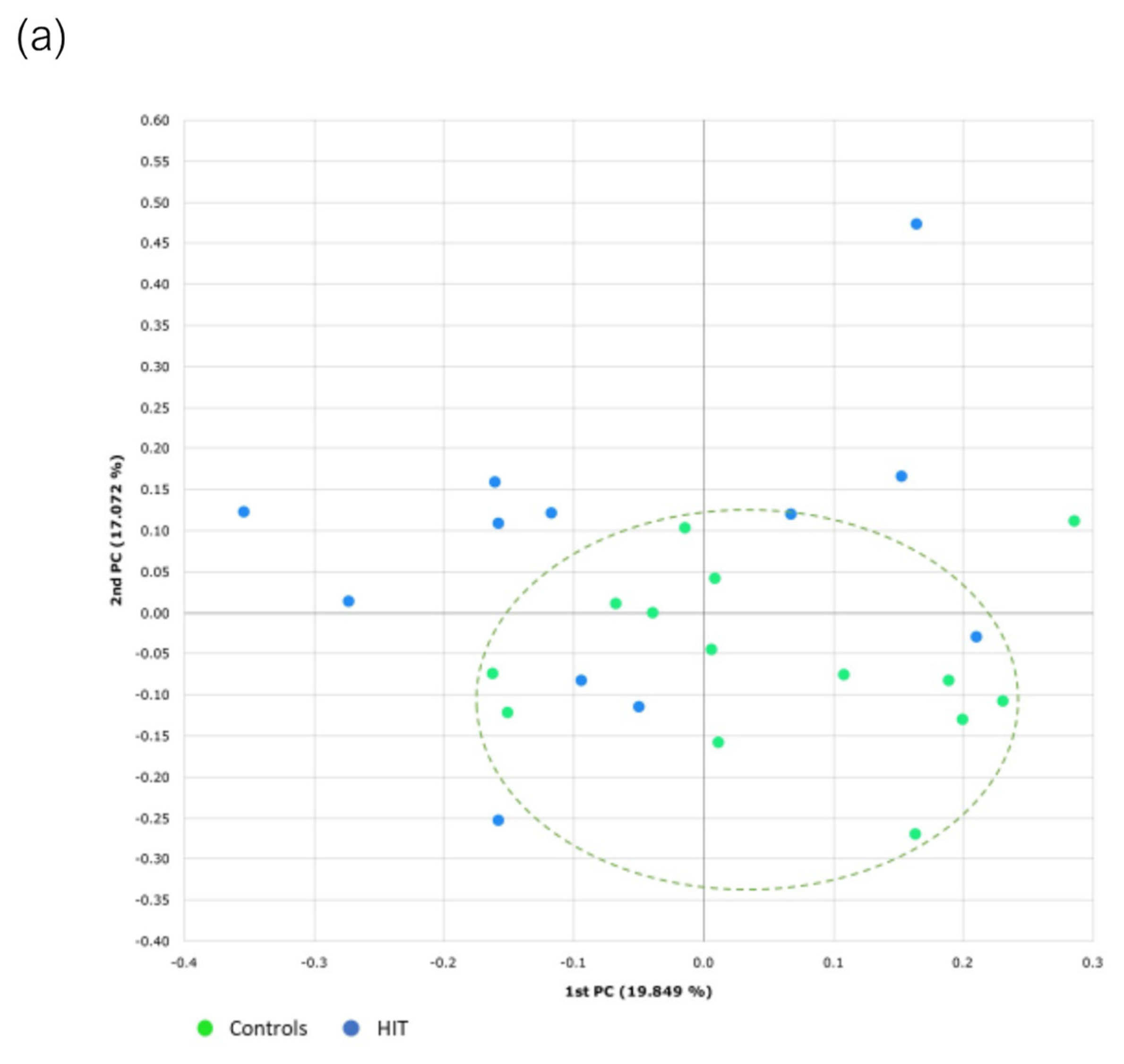
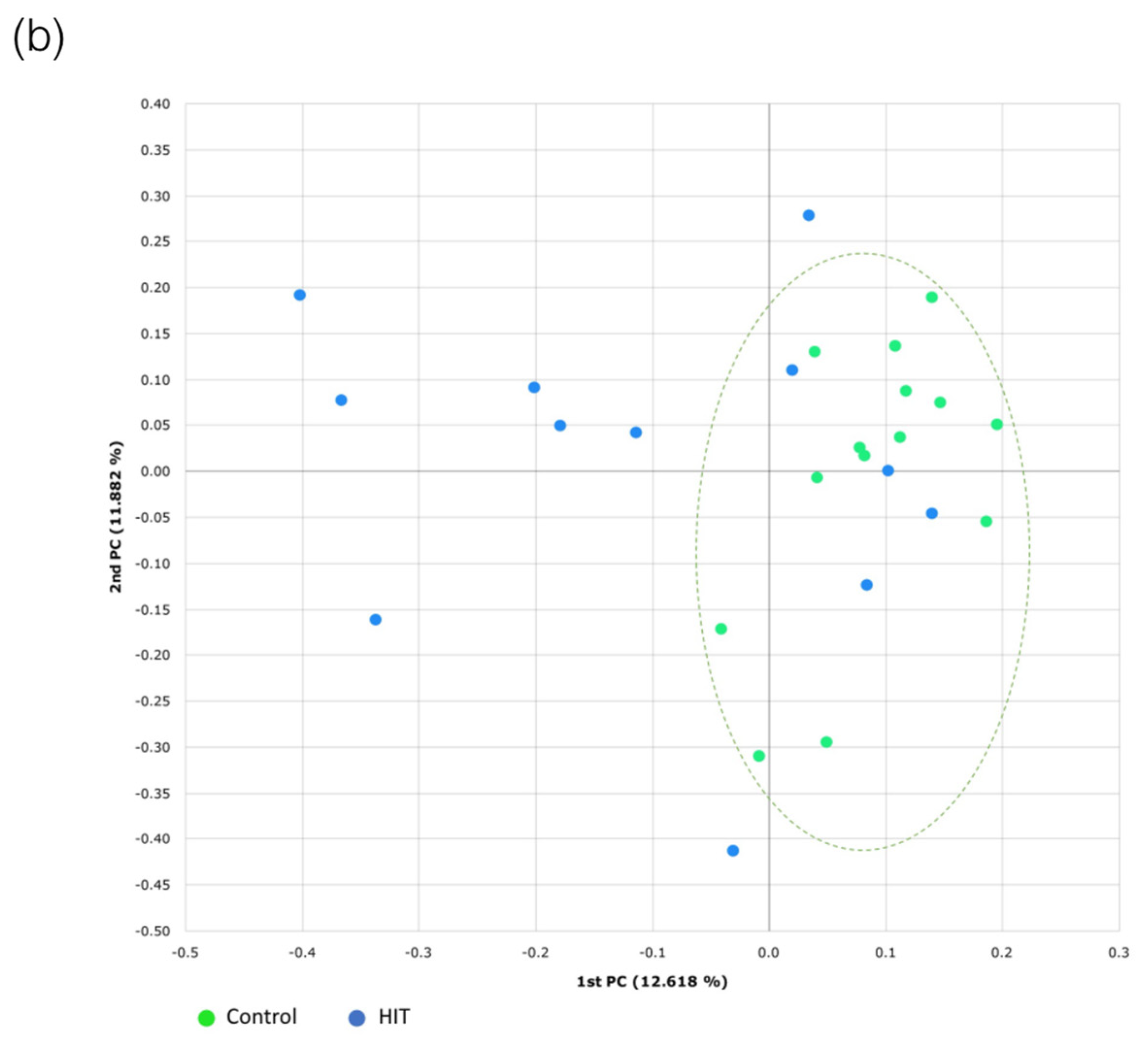
3.3. Bacterial Diversity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Power, S.E.; O’Toole, P.W.; Stanton, C.; Ross, R.P.; Fitzgerald, G.F. Intestinal Microbiota, Diet and Health. Br. J. Nutr. 2014, 111, 387–402. [Google Scholar] [CrossRef] [PubMed]
- Rinninella, E.; Cintoni, M.; Raoul, P.; Lopetuso, L.R.; Scaldaferri, F.; Pulcini, G.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. Food Components and Dietary Habits: Keys for a Healthy Gut Microbiota Composition. Nutrients 2019, 11, 2393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schnedl, W.J.; Enko, D. Histamine Intolerance Originates in the Gut. Nutrients 2021, 13, 1262. [Google Scholar] [CrossRef]
- Ley, R.E.; Bäckhed, F.; Turnbaugh, P.; Lozupone, C.A.; Knight, R.D.; Gordon, J.I. Obesity Alters Gut Microbial Ecology. Proc. Natl. Acad. Sci. USA 2005, 102, 11070–11075. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pascale, A.; Marchesi, N.; Govoni, S.; Coppola, A.; Gazzaruso, C. The Role of Gut Microbiota in Obesity, Diabetes Mellitus, and Effect of Metformin: New Insights into Old Diseases. Curr. Opin. Pharmacol. 2019, 49, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Raza, M.H.; Gul, K.; Arshad, A.; Riaz, N.; Waheed, U.; Rauf, A.; Aldakheel, F.; Alduraywish, S.; Rehman, M.U.; Abdullah, M.; et al. Microbiota in Cancer Development and Treatment. J. Cancer Res. Clin. Oncol. 2019, 145, 49–63. [Google Scholar] [CrossRef] [PubMed]
- Bhattarai, Y.; Muniz Pedrogo, D.A.; Kashyap, P.C. Irritable Bowel Syndrome: A Gut Microbiota-Related Disorder? Am. J. Physiol. Gastrointest. Liver Physiol. 2016, 312, G52–G62. [Google Scholar] [CrossRef]
- Collins, S.M.; Surette, M.; Bercik, P. The Interplay between the Intestinal Microbiota and the Brain. Nat. Rev. Microbiol. 2012, 10, 735–742. [Google Scholar] [CrossRef]
- Finegold, S.M. Desulfovibrio Species Are Potentially Important in Regressive Autism. Med. Hypotheses. 2011, 77, 270–274. [Google Scholar] [CrossRef]
- Comas-Basté, O.; Sánchez-Pérez, S.; Veciana-Nogués, M.T.; Latorre-Moratalla, M.; Vidal-Carou, M.C. Histamine Intolerance: The Current State of the Art. Biomolecules 2020, 10, 1181. [Google Scholar] [CrossRef]
- EFSA Panel on Biological Hazards (BIOHAZ). Scientific Opinion on Risk Based Control of Biogenic Amine Formation in Fermented Foods. EFSA J. 2011, 9, 2393. [Google Scholar] [CrossRef] [Green Version]
- Hrubisko, M.; Danis, R.; Huorka, M.; Wawruch, M. Histamine Intolerance—The More We Know the Less We Know. Nutrients 2021, 13, 2228. [Google Scholar] [CrossRef] [PubMed]
- Nazar, W.; Plata-Nazar, K.; Sznurkowska, K.; Szlagatys-Sidorkiewicz, A. Histamine Intolerance in Children: A Narrative Review. Nutrients 2021, 13, 1486. [Google Scholar] [CrossRef] [PubMed]
- Shulpekova, Y.O.; Nechaev, V.M.; Popova, I.R.; Deeva, T.A.; Kopylov, A.T.; Malsagova, K.A.; Kaysheva, A.L.; Ivashkin, V.T. Food Intolerance: The Role of Histamine. Nutrients 2021, 13, 3207. [Google Scholar] [CrossRef]
- Schnedl, W.J.; Lackner, S.; Enko, D.; Schenk, M.; Holasek, S.J.; Mangge, H. Evaluation of Symptoms and Symptom Combinations in Histamine Intolerance. Intest. Res. 2019, 17, 427–433. [Google Scholar] [CrossRef] [Green Version]
- García-Martín, E.; García-Menaya, J.; Sánchez, B.; Martínez, C.; Rosendo, R.; Agúndez, J.A.G. Polymorphisms of Histamine-Metabolizing Enzymes and Clinical Manifestations of Asthma and Allergic Rhinitis. Clin. Exp. Allergy 2007, 37, 1175–1182. [Google Scholar] [CrossRef]
- Maintz, L.; Yu, C.F.; Rodríguez, E.; Baurecht, H.; Bieber, T.; Illig, T.; Weidinger, S.; Novak, N. Association of Single Nucleotide Polymorphisms in the Diamine Oxidase Gene with Diamine Oxidase Serum Activities. Allergy 2011, 66, 893–902. [Google Scholar] [CrossRef]
- Leitner, R.; Zoernpfenning, E.; Missbichler, A. Evaluation of the Inhibitory Effect of Various Drugs/Active Ingredients on the Activity of Human Diamine Oxidase in Vitro. Clin. Transl. Allergy 2014, 4, 23. [Google Scholar] [CrossRef] [Green Version]
- Enko, D.; Kriegshäuser, G.; Halwachs-Baumann, G.; Mangge, H.; Schnedl, W.J. Serum Diamine Oxidase Activity Is Associated with Lactose Malabsorption Phenotypic Variation. Clin. Biochem. 2017, 50, 50–53. [Google Scholar] [CrossRef]
- Enko, D.; Meinitzer, A.; Mangge, H.; Kriegshaüser, G.; Halwachs-Baumann, G.; Reininghaus, E.Z.; Bengesser, S.A.; Schnedl, W.J. Concomitant Prevalence of Low Serum Diamine Oxidase Activity and Carbohydrate Malabsorption. Can. J. Gastroenterol. Hepatol. 2016, 2016, 4893501. [Google Scholar] [CrossRef] [Green Version]
- Griauzdaitė, K.; Maselis, K.; Žvirblienė, A.; Vaitkus, A.; Jančiauskas, D.; Banaitytė-Baleišienė, I.; Kupčinskas, L.; Rastenytė, D. Associations between Migraine, Celiac Disease, Non-Celiac Gluten Sensitivity and Activity of Diamine Oxidase. Med. Hypotheses 2020, 142, 3–6. [Google Scholar] [CrossRef] [PubMed]
- Schnedl, W.J.; Enko, D. Considering Histamine in Functional Gastrointestinal Disorders. Crit. Rev. Food Sci. Nutr. 2020, 61, 2960–2967. [Google Scholar] [CrossRef] [PubMed]
- Schink, M.; Konturek, P.C.; Tietz, E.; Dieterich, W.; Pinzer, T.C.; Wirtz, S.; Neurath, M.F.; Zopf, Y. Microbial Patterns in Patients with Histamine Intolerance. J. Physiol. Pharmacol. 2018, 69, 579–593. [Google Scholar] [CrossRef]
- Pugin, B.; Barcik, W.; Westermann, P.; Heider, A.; Wawrzyniak, M.; Hellings, P.; Akdis, C.A.; O’Mahony, L. A Wide Diversity of Bacteria from the Human Gut Produces and Degrades Biogenic Amines. Microb. Ecol. Health Dis. 2017, 28, 1353881. [Google Scholar] [CrossRef] [PubMed]
- Maintz, L.; Novak, N. Histamine and Histamine Intolerance. Am. J. Clin. Nutr. 2007, 85, 1185–1196. [Google Scholar] [CrossRef] [PubMed]
- Izquierdo-Casas, J.; Comas-Basté, O.; Latorre-Moratalla, M.L.; Lorente-Gascón, M.; Duelo, A.; Vidal-Carou, M.C.; Soler-Singla, L. Low Serum Diamine Oxidase (DAO) Activity Levels in Patients with Migraine. J. Physiol. Biochem. 2018, 74, 93–99. [Google Scholar] [CrossRef]
- Gophna, U.; Sommerfeld, K.; Gophna, S.; Doolittle, W.F.; Veldhuyzen Van Zanten, S.J.O. Differences between Tissue-Associated Intestinal Microfloras of Patients with Crohn’s Disease and Ulcerative Colitis. J. Clin. Microbiol. 2006, 44, 4136–4141. [Google Scholar] [CrossRef] [Green Version]
- Wang, T.; Cai, G.; Qiu, Y.; Fei, N.; Zhang, M.; Pang, X.; Jia, W.; Cai, S.; Zhao, L. Structural Segregation of Gut Microbiota between Colorectal Cancer Patients and Healthy Volunteers. ISME J. 2012, 6, 320–329. [Google Scholar] [CrossRef] [Green Version]
- Krogius-Kurikka, L.; Lyra, A.; Malinen, E.; Aarnikunnas, J.; Tuimala, J.; Paulin, L.; Mäkivuokko, H.; Kajander, K.; Palva, A. Microbial Community Analysis Reveals High Level Phylogenetic Alterations in the Overall Gastrointestinal Microbiota of Diarrhoea-Predominant Irritable Bowel Syndrome Sufferers. BMC Gastroenterol. 2009, 9, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Morgan, X.C.; Tickle, T.L.; Sokol, H.; Gevers, D.; Devaney, K.L.; Ward, D.V.; Reyes, J.A.; Shah, S.A.; LeLeiko, N.; Snapper, S.B.; et al. Dysfunction of the Intestinal Microbiome in Inflammatory Bowel Disease and Treatment. Genome Biol. 2012, 13, R79. [Google Scholar] [CrossRef]
- Litvak, Y.; Byndloss, M.X.; Tsolis, R.M.; Bäumler, A.J. Dysbiotic Proteobacteria Expansion: A Microbial Signature of Epithelial Dysfunction. Curr. Opin. Microbiol. 2017, 39, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Mou, Z.; Yang, Y.; Hall, A.B.; Jiang, X. The Taxonomic Distribution of Histamine-Secreting Bacteria in the Human Gut Microbiome. BMC Genom. 2021, 22, 695. [Google Scholar] [CrossRef] [PubMed]
- Scheperjans, F.; Aho, V.; Pereira, P.A.B.; Koskinen, K.; Paulin, L.; Pekkonen, E.; Haapaniemi, E.; Kaakkola, S.; Eerola-Rautio, J.; Pohja, M.; et al. Gut Microbiota Are Related to Parkinson’s Disease and Clinical Phenotype. Mov. Disord. 2015, 30, 350–358. [Google Scholar] [CrossRef] [PubMed]
- La Reau, A.J.; Suen, G. The Ruminococci: Key Symbionts of the Gut Ecosystem. J. Microbiol. 2018, 56, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Cockburn, D.W.; Koropatkin, N.M. Polysaccharide Degradation by the Intestinal Microbiota and Its Influence on Human Health and Disease. J. Mol. Biol. 2016, 428, 3230–3252. [Google Scholar] [CrossRef]
- Lopez-Siles, M.; Duncan, S.H.; Garcia-Gil, L.J.; Martinez-Medina, M. Faecalibacterium Prausnitzii: From Microbiology to Diagnostics and Prognostics. ISME J. 2017, 11, 841–852. [Google Scholar] [CrossRef] [PubMed]
- Martín, R.; Miquel, S.; Benevides, L.; Bridonneau, C.; Robert, V.; Hudault, S.; Chain, F.; Berteau, O.; Azevedo, V.; Chatel, J.M.; et al. Functional Characterization of Novel Faecalibacterium Prausnitzii Strains Isolated from Healthy Volunteers: A Step Forward in the Use of F. Prausnitzii as a next-Generation Probiotic. Front. Microbiol. 2017, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Laursen, M.F.; Laursen, R.P.; Larnkjær, A.; Mølgaard, C.; Michaelsen, K.F.; Frøkiær, H.; Bahl, M.I.; Licht, T.R. Faecalibacterium Gut Colonization Is Accelerated by Presence of Older Siblings. mSphere 2017, 2, 2–7. [Google Scholar] [CrossRef] [Green Version]
- Durak-Dados, A.; Michalski, M.; Osek, J. Histamine and Other Biogenic Amines in Food. J. Vet. Res. 2020, 64, 281–288. [Google Scholar] [CrossRef]
- Landete, J.M.; de Las Rivas, B.; Marcobal, A.; Muñoz, R. Updated Molecular Knowledge about Histamine Biosynthesis by Bacteria. Crit. Rev. Food Sci. Nutr. 2008, 48, 697–714. [Google Scholar] [CrossRef] [Green Version]
- Refai, M.A.; El-Hariri, M.; Ahmed, Z.A.M.; Jakee, J. el Histamine Producing Bacteria in Fish. Egypt. J. Aquat. Biol. Fish. 2020, 24, 1–11. [Google Scholar] [CrossRef]
- Comas-Basté, O.; Latorre-Moratalla, M.L.; Sánchez-Pérez, S.; Veciana-Nogués, M.T.; Vidal-Carou, M.C. Histamine and Other Biogenic Amines in Food. From Scombroid Poisoning to Histamine Intolerance. In Biogenic Amines; Proestos, C., Ed.; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef] [Green Version]
- Krell, T.; Gavira, J.A.; Velando, F.; Fernández, M.; Roca, A.; Monteagudo-Cascales, E.; Matilla, M.A. Histamine: A Bacterial Signal Molecule. Int. J. Mol. Sci. 2021, 22, 6312. [Google Scholar] [CrossRef] [PubMed]
- Sudo, N. Biogenic Amines: Signals Between Commensal Microbiota and Gut Physiology. Front. Endocrinol. 2019, 10, 504. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferstl, R.; Frei, R.; Schiavi, E.; Konieczna, P.; Barcik, P.; Ziegler, M.; Lauener, R.P.; Chassard, C.; Lacroix, C.; Akdis, C.A. Histamine Receptor 2 Is a Key Influence in Immune Responses to Microbial Ligands. J. Allergy Clin. Immunol. 2014, 134, 741–744. [Google Scholar] [CrossRef]
- O’Mahony, L.; Akdis, M.; Akdis, C.A. Regulation of the Immune Response and Inflammation by Histamine and Histamine Receptors. J. Allergy Clin. Immunol. 2011, 128, 1153–1162. [Google Scholar] [CrossRef]
- Mishima, Y.; Ishihara, S. Molecular Mechanisms of Microbiota-Mediated Pathology in Irritable Bowel Syndrome. Int. J. Mol. Sci. 2020, 21, 8664. [Google Scholar] [CrossRef]
- Barcik, W.; Pugin, B.; Westermann, P.; Rodriguez-Pérez, N.; Ferstl, R.; Wawrzyniak, M.; Smolinska, S.; Jutel, M.; Hessel, E.M.; Michalovich, D.; et al. Histamine-Secreting Microbes Are Increased in the Gut of Adult Asthma Patients. J. Allergy Clin. Immunol. 2016, 138, 1489–1491. [Google Scholar] [CrossRef] [Green Version]
- Frei, R.; Ferstl, R.; Konieczna, P.; Ziegler, M.; Simon, T.; Rugeles, T.M.; Mailand, S.; Watanabe, T.; Lauener, R.; Akdis, C.A.; et al. Histamine Receptor 2 Modifies Dendritic Cell Responses to Microbial Ligands. J. Allergy Clin. Immunol. 2013, 132, 194–204. [Google Scholar] [CrossRef]
- Levy, M.; Thaiss, C.A.; Zeevi, D.; Dohnalová, L.; Zilberman-Schapira, G.; Mahdi, J.A.; David, E.; Savidor, A.; Korem, T.; Herzig, Y.; et al. Microbiota-Modulated Metabolites Shape the Intestinal Microenvironment by Regulating NLRP6 Inflammasome Signaling. Cell 2015, 163, 1428–1443. [Google Scholar] [CrossRef] [Green Version]
- Sanchis-Artero, L.; Martínez-Blanch, J.F.; Manresa-Vera, S.; Cortés-Castell, E.; Valls-Gandia, M.; Iborra, M.; Paredes-Arquiola, J.M.; Boscá-Watts, M.; Huguet, J.M.; Gil-Borrás, R.; et al. Evaluation of Changes in Intestinal Microbiota in Crohn’s Disease Patients after Anti-TNF Alpha Treatment. Cells 2021, 10, 3188. [Google Scholar] [CrossRef]
- Chen, C.C.; Chen, K.J.; Kong, M.S.; Chang, H.J.; Huang, J.L. Alterations in the Gut Microbiotas of Children with Food Sensitization in Early Life. Pediatr. Allergy Immunol. 2016, 27, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Azcarate-Peril, M.A.; Ritter, A.J.; Savaiano, D.; Monteagudo-Mera, A.; Anderson, C.; Magness, S.T.; Klaenhammer, T.R. Impact of Short-Chain Galactooligosaccharides on the Gut Microbiome of Lactose-Intolerant Individuals. Proc. Natl. Acad. Sci. USA 2017, 114, E367–E375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Das, A.; O’Herlihy, E.; Shanahan, F.; O’Toole, P.W.; Jeffery, I.B. The Fecal Mycobiome in Patients with Irritable Bowel Syndrome. Sci. Rep. 2021, 11, 124. [Google Scholar] [CrossRef] [PubMed]
- Wacklin, P.; Laurikka, P.; Lindfors, K.; Collin, P.; Salmi, T.; Lähdeaho, M.L.; Saavalainen, P.; Mäki, M.; Mättö, J.; Kurppa, K.; et al. Altered Duodenal Microbiota Composition in Celiac Disease Patients Suffering from Persistent Symptoms on a Long-Term Gluten-Free Diet. Am. J. Gastroenterol. 2014, 109, 1933–1941. [Google Scholar] [CrossRef] [PubMed]
| Participants’ Characteristics | Groups | |
|---|---|---|
| Control | HIT | |
| n (%) | 14 (53.9%) | 12 (46.2%) |
| Age (average years ± SD) | 40.4 ± 12.5 | 40.4 ± 12.4 |
| Male [n (%)] | 4 (28.6%) | 0 (0%) |
| Female [n (%)] | 10 (71.4%) | 12 (100%) |
| Body Mass Index (BMI) [average ± SD] | 23.7 ± 3.2 | 22.2 ± 6.0 |
| Symptoms | Frequency (%) * |
|---|---|
| Gastrointestinal tract | |
| Bloating | 75 |
| Flatulencies | 33 |
| Abdominal pain | 25 |
| Diarrhoea | 25 |
| Heartburn | 25 |
| Constipation | 17 |
| Nausea | 17 |
| Skin | |
| Urticaria | 17 |
| Atopic skin | 17 |
| Pruritus | 8 |
| Eczema | 8 |
| Neurologic system | |
| Headache | 75 |
| Dizziness | 8 |
| Respiratory apparatus | |
| Asthma | 17 |
| Rhinitis | 8 |
| Shortness of breath | 8 |
| Other symptoms | |
| Muscular/articular pain | 25 |
| Fatigue | 17 |
| Insomnia | 8 |
| Family | Phylum | Control | HIT | p-Value |
|---|---|---|---|---|
| Acholeplasmataceae | Tenericutes | 0.001 ± 0.002 | 0.002 ± 0.001 | 0.03 |
| Actinomycetaceae | Actinobacteria | 0.012 ± 0.011 | 0.027 ± 0.024 | 0.02 |
| Prevotellaceae | Bacteroidetes | 7.422 ± 7.996 | 1.548 ± 2.408 | 0.04 |
| Staphylococcaceae | Firmicutes | 0.002 ± 0.007 | 0.014 ± 0.029 | 0.03 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez-Pérez, S.; Comas-Basté, O.; Duelo, A.; Veciana-Nogués, M.T.; Berlanga, M.; Latorre-Moratalla, M.L.; Vidal-Carou, M.C. Intestinal Dysbiosis in Patients with Histamine Intolerance. Nutrients 2022, 14, 1774. https://doi.org/10.3390/nu14091774
Sánchez-Pérez S, Comas-Basté O, Duelo A, Veciana-Nogués MT, Berlanga M, Latorre-Moratalla ML, Vidal-Carou MC. Intestinal Dysbiosis in Patients with Histamine Intolerance. Nutrients. 2022; 14(9):1774. https://doi.org/10.3390/nu14091774
Chicago/Turabian StyleSánchez-Pérez, Sònia, Oriol Comas-Basté, Adriana Duelo, M. Teresa Veciana-Nogués, Mercedes Berlanga, M. Luz Latorre-Moratalla, and M. Carmen Vidal-Carou. 2022. "Intestinal Dysbiosis in Patients with Histamine Intolerance" Nutrients 14, no. 9: 1774. https://doi.org/10.3390/nu14091774
APA StyleSánchez-Pérez, S., Comas-Basté, O., Duelo, A., Veciana-Nogués, M. T., Berlanga, M., Latorre-Moratalla, M. L., & Vidal-Carou, M. C. (2022). Intestinal Dysbiosis in Patients with Histamine Intolerance. Nutrients, 14(9), 1774. https://doi.org/10.3390/nu14091774









