Plant-Based Dietary Patterns for Human and Planetary Health
Abstract
:1. Introduction
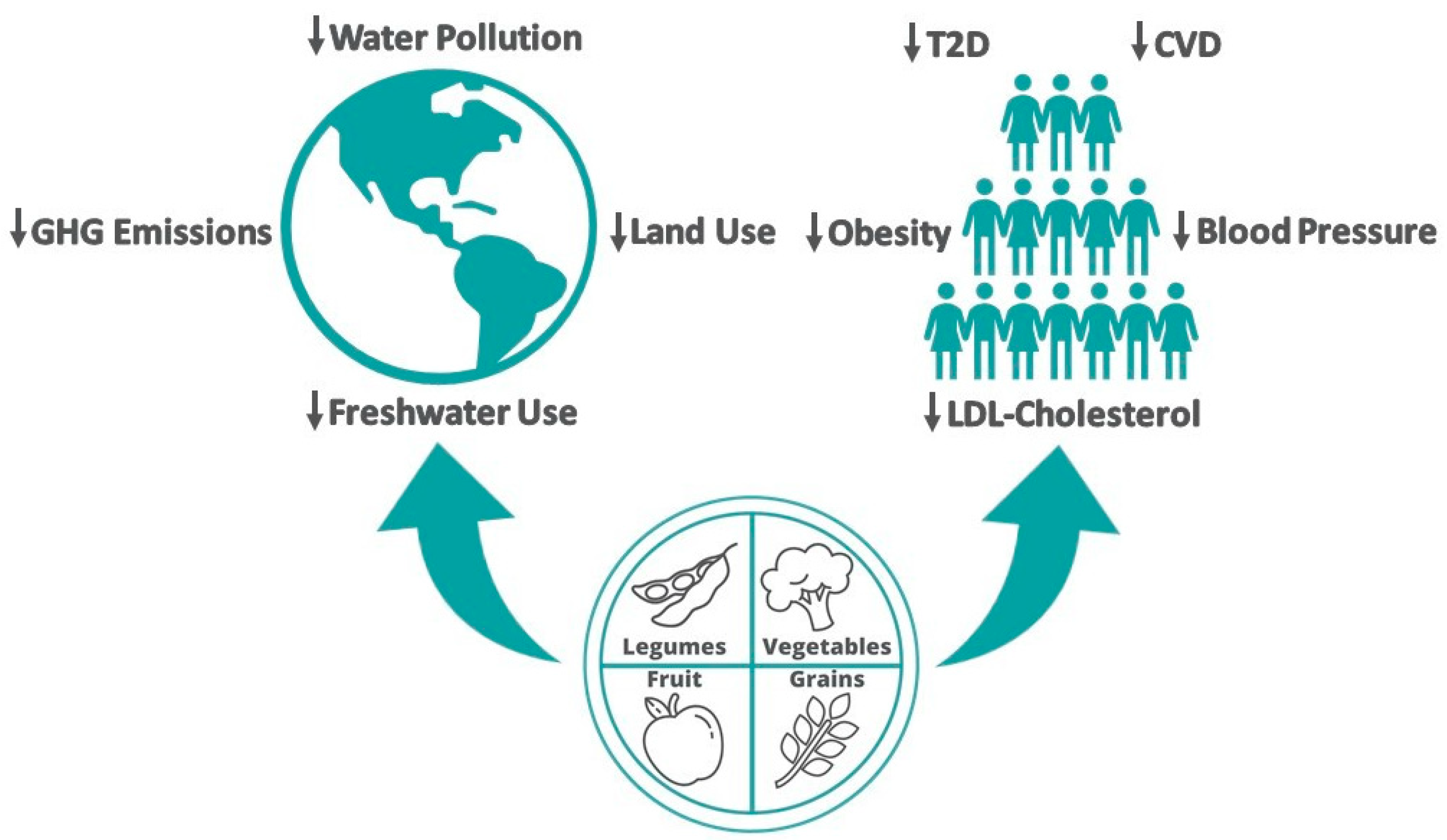
2. Planetary Health
2.1. Greenhouse Gas (GHG) Emission
2.2. Agricultural Land Use
2.3. Water Use
2.4. Healthy Reference Dietary Pattern
3. Human Health
3.1. Obesity
3.2. Type 2 Diabetes
3.3. CVD Risk
3.4. Hypertension and Hypercholesterolemia
3.5. CVD Prevention
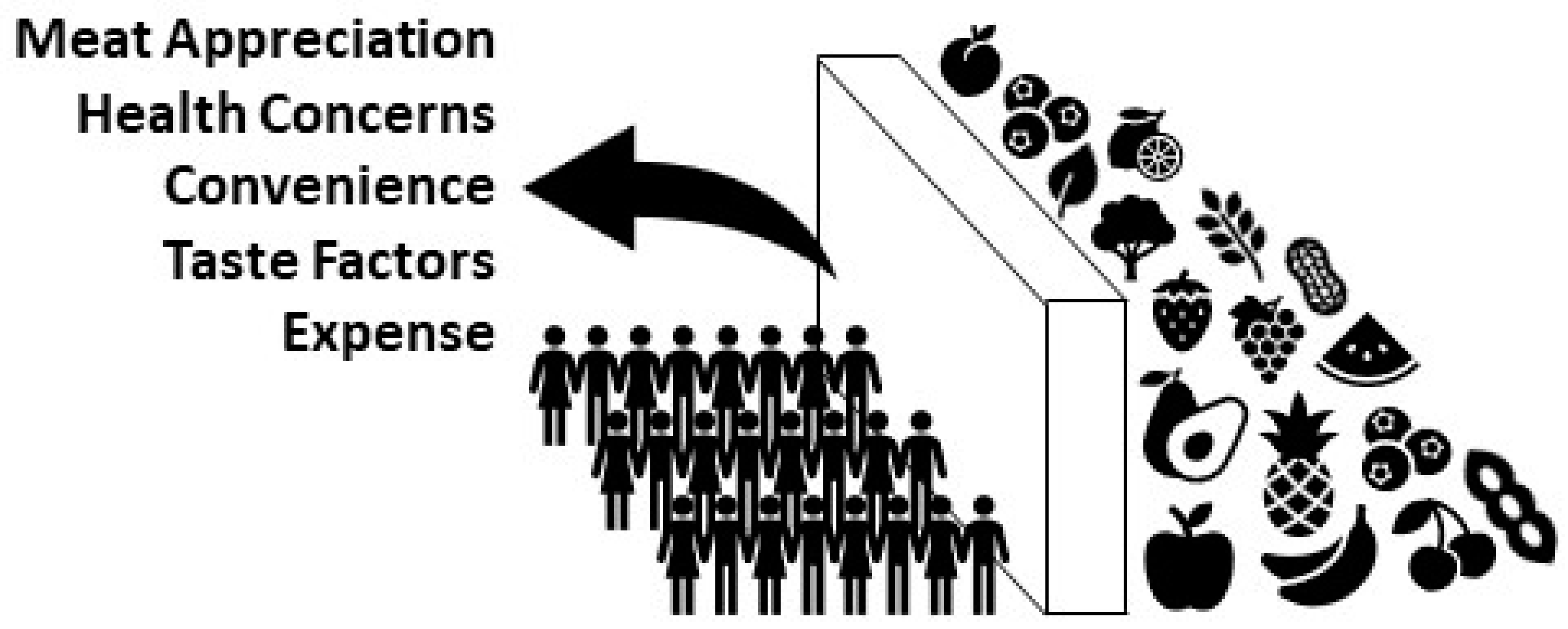
4. Barriers and Potential Solutions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stocker, T.F.; Qin, D.; Plattner, G.-K.; Tignor, M.; Allen, S.K.; Boschung, J.; Nauels, A.; Xia, Y.; Bex, B.; Midgley, B.M. IPCC, 2013: Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013. [Google Scholar]
- Horowitz, C.A. Paris agreement. Int. Leg. Mater. 2016, 55, 740–755. [Google Scholar] [CrossRef]
- Schwab, K.; Malleret, T. The Great Reset; World Economic Forum: Geneva, Switzerland, 2020. [Google Scholar]
- Johnson, B. The Ten Point Plan for a Green Industrial Revolution; H.M. Government: London, UK, 2020.
- Willett, W.; Rockström, J.; Loken, B.; Springmann, M.; Lang, T.; Vermeulen, S.; Garnett, T.; Tilman, D.; DeClerck, F.; Wood, A. Food in the Anthropocene: The EAT–Lancet Commission on healthy diets from sustainable food systems. Lancet 2019, 393, 447–492. [Google Scholar] [CrossRef]
- Vermeulen, S.J.; Campbell, B.M.; Ingram, J.S.I. Climate change and food systems. Annu. Rev. Environ. Resour. 2012, 37, 195–222. [Google Scholar] [CrossRef] [Green Version]
- Steffen, W.; Richardson, K.; Rockström, J.; Cornell, S.E.; Fetzer, I.; Bennett, E.M.; Biggs, R.; Carpenter, S.R.; De Vries, W.; De Wit, C.A. Planetary boundaries: Guiding human development on a changing planet. Science 2015, 347, 1259855. [Google Scholar] [CrossRef] [Green Version]
- Foley, J.A.; DeFries, R.; Asner, G.P.; Barford, C.; Bonan, G.; Carpenter, S.R.; Chapin, F.S.; Coe, M.T.; Daily, G.C.; Gibbs, H.K. Global consequences of land use. Science 2005, 309, 570–574. [Google Scholar] [CrossRef] [Green Version]
- Poore, J.; Nemecek, T. Reducing food’s environmental impacts through producers and consumers. Science 2018, 360, 987–992. [Google Scholar] [CrossRef] [Green Version]
- Hemler, E.C.; Hu, F.B. Plant-based diets for personal, population, and planetary health. Adv. Nutr. 2019, 10, S275–S283. [Google Scholar] [CrossRef]
- Fresán, U.; Sabaté, J. Vegetarian diets: Planetary health and its alignment with human health. Adv. Nutr. 2019, 10, S380–S388. [Google Scholar] [CrossRef] [Green Version]
- Wilson, N.; Cleghorn, C.L.; Cobiac, L.J.; Mizdrak, A.; Nghiem, N. Achieving healthy and sustainable diets: A review of the results of recent mathematical optimization studies. Adv. Nutr. 2019, 10, S389–S403. [Google Scholar] [CrossRef] [Green Version]
- Shukla, P.R.; Skeg, J.; Buendia, E.C.; Masson-Delmotte, V.; Pörtner, H.O.; Roberts, D.C.; Zhai, P.; Slade, R.; Connors, S.; van Diemen, S. Climate Change and Land: An IPCC Special Report on Climate Change, Desertification, Land Degradation, Sustainable Land Management, Food Security, and Greenhouse Gas Fluxes in Terrestrial Ecosystems; IPCC: Geneva, Switzerland, 2019.
- Romanello, M.; McGushin, A.; Di Napoli, C.; Drummond, P.; Hughes, N.; Jamart, L.; Kennard, H.; Lampard, P.; Rodriguez, B.S.; Arnell, N. The 2021 report of the Lancet Countdown on health and climate change: Code red for a healthy future. Lancet 2021, 398, 1619–1662. [Google Scholar] [CrossRef]
- Lynch, H.; Johnston, C.; Wharton, C. Plant-based diets: Considerations for environmental impact, protein quality, and exercise performance. Nutrients 2018, 10, 1841. [Google Scholar] [CrossRef] [Green Version]
- Carlsson-Kanyama, A.; Ekström, M.P.; Shanahan, H. Food and life cycle energy inputs: Consequences of diet and ways to increase efficiency. Ecol. Econ. 2003, 44, 293–307. [Google Scholar] [CrossRef]
- Eshel, G.; Stainier, P.; Shepon, A.; Swaminathan, A. Environmentally optimal, nutritionally sound, protein and energy conserving plant based alternatives to US meat. Sci. Rep. 2019, 9, 10345. [Google Scholar] [CrossRef] [Green Version]
- Tilman, D.; Clark, M.; Williams, D.R.; Kimmel, K.; Polasky, S.; Packer, C. Future threats to biodiversity and pathways to their prevention. Nature 2017, 546, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Cardinale, B.J.; Duffy, J.E.; Gonzalez, A.; Hooper, D.U.; Perrings, C.; Venail, P.; Narwani, A.; Mace, G.M.; Tilman, D.; Wardle, D.A. Biodiversity loss and its impact on humanity. Nature 2012, 486, 59–67. [Google Scholar] [CrossRef]
- Cassidy, E.S.; West, P.C.; Gerber, J.S.; Foley, J.A. Redefining agricultural yields: From tonnes to people nourished per hectare. Environ. Res. Lett. 2013, 8, 034015. [Google Scholar] [CrossRef]
- Pimentel, D.; Berger, B.; Filiberto, D.; Newton, M.; Wolfe, B.; Karabinakis, E.; Clark, S.; Poon, E.; Abbett, E.; Nandagopal, S. Water resources: Agricultural and environmental issues. BioScience 2004, 54, 909–918. [Google Scholar] [CrossRef] [Green Version]
- Jalava, M.; Kummu, M.; Porkka, M.; Siebert, S.; Varis, O. Diet change—A solution to reduce water use? Environ. Res. Lett. 2014, 9, 074016. [Google Scholar] [CrossRef]
- Laine, J.E.; Huybrechts, I.; Gunter, M.J.; Ferrari, P.; Weiderpass, E.; Tsilidis, K.; Aune, D.; Schulze, M.B.; Bergmann, M.; Temme, E.H.M. Co-benefits from sustainable dietary shifts for population and environmental health: An assessment from a large European cohort study. Lancet Planet. Health 2021, 5, e786–e796. [Google Scholar] [CrossRef]
- Smyth, S.; Heron, A. Diabetes and obesity: The twin epidemics. Nat. Med. 2006, 12, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Rahmouni, K.; Correia, M.L.G.; Haynes, W.G.; Mark, A.L. Obesity-associated hypertension: New insights into mechanisms. Hypertension 2005, 45, 9–14. [Google Scholar] [CrossRef]
- Van Gaal, L.F.; Mertens, I.L.; De Block, C.E. Mechanisms linking obesity with cardiovascular disease. Nature 2006, 444, 875–880. [Google Scholar] [CrossRef]
- Wolin, K.Y.; Carson, K.; Colditz, G.A. Obesity and cancer. Oncologist 2010, 15, 556–565. [Google Scholar] [CrossRef]
- Lim, S.S.; Vos, T.; Flaxman, A.D.; Danaei, G.; Shibuya, K.; Adair-Rohani, H.; Almazroa, M.A.; Amann, M.; Anderson, H.R.; Andrews, K.G.; et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2224–2260. [Google Scholar] [CrossRef] [Green Version]
- Tonstad, S.; Stewart, K.; Oda, K.; Batech, M.; Herring, R.P.; Fraser, G.E. Vegetarian diets and incidence of diabetes in the Adventist Health Study-2. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 292–299. [Google Scholar] [CrossRef] [Green Version]
- Rosell, M.; Appleby, P.; Spencer, E.; Key, T. Weight gain over 5 years in 21,966 meat-eating, fish-eating, vegetarian, and vegan men and women in EPIC-Oxford. Int. J. Obes. 2006, 30, 1389–1396. [Google Scholar] [CrossRef] [Green Version]
- Vergnaud, A.-C.; Norat, T.; Romaguera, D.; Mouw, T.; May, A.M.; Travier, N.; Luan, J.a.; Wareham, N.; Slimani, N.; Rinaldi, S. Meat consumption and prospective weight change in participants of the EPIC-PANACEA study. Am. J. Clin. Nutr. 2010, 92, 398–407. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.B.; Shivappa, N.; Hébert, J.R.; Page, A.J.; Gill, T.K.; Melaku, Y.A. Association between dietary inflammatory index, dietary patterns, plant-based dietary index and the risk of obesity. Nutrients 2021, 13, 1536. [Google Scholar] [CrossRef]
- Barnard, N.D.; Levin, S.M.; Yokoyama, Y. A systematic review and meta-analysis of changes in body weight in clinical trials of vegetarian diets. J. Acad. Nutr. Diet. 2015, 115, 954–969. [Google Scholar] [CrossRef]
- Austin, G.; Ferguson, J.J.A.; Garg, M.L. Effects of Plant-Based Diets on Weight Status in Type 2 Diabetes: A Systematic Review and Meta-Analysis of Randomised Controlled Trials. Nutrients 2021, 13, 4099. [Google Scholar] [CrossRef]
- Turner-McGrievy, G.M.; Davidson, C.R.; Wingard, E.E.; Wilcox, S.; Frongillo, E.A. Comparative effectiveness of plant-based diets for weight loss: A randomized controlled trial of five different diets. Nutrition 2015, 31, 350–358. [Google Scholar] [CrossRef] [PubMed]
- Turner-McGrievy, G.M.; Davidson, C.R.; Wingard, E.E.; Billings, D.L. Low glycemic index vegan or low-calorie weight loss diets for women with polycystic ovary syndrome: A randomized controlled feasibility study. Nutr. Res. 2014, 34, 552–558. [Google Scholar] [CrossRef] [PubMed]
- Talreja, A.; Talreja, S.; Talreja, R.; Talreja, D. CRT-601 The VA beach diet study: An investigation of the effects of plant-based, mediterranean, paleolithic, and dash diets on cardiovascular disease risk. JACC Cardiovasc. Interv. 2015, 8, S41. [Google Scholar] [CrossRef] [Green Version]
- Wright, N.; Wilson, L.; Smith, M.; Duncan, B.; McHugh, P. The BROAD study: A randomised controlled trial using a whole food plant-based diet in the community for obesity, ischaemic heart disease or diabetes. Nutr. Diabetes 2017, 7, e256. [Google Scholar] [CrossRef]
- Kahleova, H.; Fleeman, R.; Hlozkova, A.; Holubkov, R.; Barnard, N.D. A plant-based diet in overweight individuals in a 16-week randomized clinical trial: Metabolic benefits of plant protein. Nutr. Diabetes 2018, 8, 58. [Google Scholar] [CrossRef] [Green Version]
- Barnard, N.D.; Levin, S.M.; Gloede, L.; Flores, R. Turning the waiting room into a classroom: Weekly classes using a vegan or a portion-controlled eating plan improve diabetes control in a randomized translational study. J. Acad. Nutr. Diet. 2018, 118, 1072–1079. [Google Scholar] [CrossRef] [Green Version]
- Zhou, B.; Lu, Y.; Hajifathalian, K.; Bentham, J.; Di Cesare, M.; Danaei, G.; Bixby, H.; Cowan, M.J.; Ali, M.K.; Taddei, C. Worldwide trends in diabetes since 1980: A pooled analysis of 751 population-based studies with 4·4 million participants. Lancet 2016, 387, 1513–1530. [Google Scholar] [CrossRef] [Green Version]
- Sun, H.; Saeedi, P.; Karuranga, S.; Pinkepank, M.; Ogurtsova, K.; Duncan, B.B.; Stein, C.; Basit, A.; Chan, J.C.N.; Mbanya, J.C. IDF diabetes atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res. Clin. Pract. 2022, 183, 109119. [Google Scholar] [CrossRef]
- Tonstad, S.; Butler, T.; Yan, R.; Fraser, G.E. Type of Vegetarian Diet, Body Weight, and Prevalence of Type 2 Diabetes. Diabetes Care 2009, 32, 791–796. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vang, A.; Singh, P.N.; Lee, J.W.; Haddad, E.H.; Brinegar, C.H. Meats, processed meats, obesity, weight gain and occurrence of diabetes among adults: Findings from Adventist Health Studies. Ann. Nutr. Metab. 2008, 52, 96–104. [Google Scholar] [CrossRef]
- Chen, Z.; Drouin-Chartier, J.-P.; Li, Y.; Baden, M.Y.; Manson, J.E.; Willett, W.C.; Voortman, T.; Hu, F.B.; Bhupathiraju, S.N. Changes in plant-based diet indices and subsequent risk of type 2 diabetes in women and men: Three US prospective cohorts. Diabetes Care 2021, 44, 663–671. [Google Scholar] [CrossRef] [PubMed]
- Qian, F.; Liu, G.; Hu, F.B.; Bhupathiraju, S.N.; Sun, Q. Association between plant-based dietary patterns and risk of type 2 diabetes: A systematic review and meta-analysis. JAMA Intern. Med. 2019, 179, 1335–1344. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, Y.; Barnard, N.D.; Levin, S.M.; Watanabe, M. Vegetarian diets and glycemic control in diabetes: A systematic review and meta-analysis. Cardiovasc. Diagn. Ther. 2014, 4, 373. [Google Scholar] [PubMed]
- Johansen, K. Efficacy of metformin in the treatment of NIDDM. Meta-analysis. Diabetes Care 1999, 22, 33–37. [Google Scholar] [CrossRef] [PubMed]
- Roth, G.A.; Mensah, G.A.; Johnson, C.O.; Addolorato, G.; Ammirati, E.; Baddour, L.M.; Barengo, N.C.; Beaton, A.Z.; Benjamin, E.J.; Benziger, C.P. Global burden of cardiovascular diseases and risk factors, 1990–2019: Update from the GBD 2019 study. J. Am. Coll. Cardiol. 2020, 76, 2982–3021. [Google Scholar] [CrossRef]
- Gan, Z.H.; Cheong, H.C.; Tu, Y.-K.; Kuo, P.-H. Association between Plant-Based Dietary Patterns and Risk of Cardiovascular Disease: A Systematic Review and Meta-Analysis of Prospective Cohort Studies. Nutrients 2021, 13, 3952. [Google Scholar] [CrossRef]
- Quek, J.; Lim, G.; Lim, W.H.; Ng, C.H.; So, W.Z.; Toh, J.; Pan, X.H.; Chin, Y.H.; Muthiah, M.D.; Chan, S.P. The Association of Plant-Based Diet With Cardiovascular Disease and Mortality: A Meta-Analysis and Systematic Review of Prospect Cohort Studies. Front. Cardiovasc. Med. 2021, 8, 756810. [Google Scholar] [CrossRef]
- De Lorgeril, M.; Salen, P.; Martin, J.-L.; Monjaud, I.; Delaye, J.; Mamelle, N. Mediterranean diet, traditional risk factors, and the rate of cardiovascular complications after myocardial infarction: Final report of the Lyon Diet Heart Study. Circulation 1999, 99, 779–785. [Google Scholar] [CrossRef]
- Ornish, D.; Scherwitz, L.W.; Billings, J.H.; Gould, K.L.; Merritt, T.A.; Sparler, S.; Armstrong, W.T.; Ports, T.A.; Kirkeeide, R.L.; Hogeboom, C. Intensive lifestyle changes for reversal of coronary heart disease. JAMA 1998, 280, 2001–2007. [Google Scholar] [CrossRef]
- Pettersen, B.J.; Anousheh, R.; Fan, J.; Jaceldo-Siegl, K.; Fraser, G.E. Vegetarian diets and blood pressure among white subjects: Results from the Adventist Health Study-2 (AHS-2). Public Health Nutr. 2012, 15, 1909–1916. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gibbs, J.; Gaskin, E.; Ji, C.; Miller, M.A.; Cappuccio, F.P. The effect of plant-based dietary patterns on blood pressure: A systematic review and meta-analysis of controlled intervention trials. J. Hypertens. 2021, 39, 23–37. [Google Scholar] [CrossRef]
- Yokoyama, Y.; Levin, S.M.; Barnard, N.D. Association between plant-based diets and plasma lipids: A systematic review and meta-analysis. Nutr. Rev. 2017, 75, 683–698. [Google Scholar] [CrossRef]
- Wang, D.D.; Li, Y.; Nguyen, X.-M.T.; Song, R.J.; Ho, Y.-L.; Hu, F.B.; Willett, W.C.; Wilson, P.W.F.; Cho, K.; Gaziano, J.M. Degree of Adherence to Based Diet and Total and Cause-Specific Mortality: Prospective Cohort Study in the Million Veteran Program. Public Health Nutr. 2022, 25, 1–38. [Google Scholar] [CrossRef]
- English, L.K.; Ard, J.D.; Bailey, R.L.; Bates, M.; Bazzano, L.A.; Boushey, C.J.; Brown, C.; Butera, G.; Callahan, E.H.; De Jesus, J. Evaluation of dietary patterns and all-cause mortality: A systematic review. JAMA Netw. Open 2021, 4, e2122277. [Google Scholar] [CrossRef]
- Fehér, A.; Gazdecki, M.; Véha, M.; Szakály, M.; Szakály, Z. A Comprehensive Review of the Benefits of and the Barriers to the Switch to a Plant-Based Diet. Sustainability 2020, 12, 4136. [Google Scholar] [CrossRef]
- Heller, M.C.; Keoleian, G.A. Beyond Meat’s Beyond Burger Life Cycle Assessment: A Detailed Comparison between a Plant-Based and an Animal-Based Protein Source; University of Michigan: Michigan, MI, USA, 2018. [Google Scholar]
- Khan, S.; Dettling, J.; Hester, J.; Moses, R.; Foods, I. Comparative Environmental LCA of the Impossible Burger with Conventional Ground Beef Burger; Quantis: Lausanne, Switzerland, 2019. [Google Scholar]
- Bohrer, B.M. An investigation of the formulation and nutritional composition of modern meat analogue products. Food Sci. Hum. Wellness 2019, 8, 320–329. [Google Scholar] [CrossRef]
- Crimarco, A.; Springfield, S.; Petlura, C.; Streaty, T.; Cunanan, K.; Lee, J.; Fielding-Singh, P.; Carter, M.M.; Topf, M.A.; Wastyk, H.C. A randomized crossover trial on the effect of plant-based compared with animal-based meat on trimethylamine-N-oxide and cardiovascular disease risk factors in generally healthy adults: Study With Appetizing Plantfood—Meat Eating Alternative Trial (SWAP-MEAT). Am. J. Clin. Nutr. 2020, 112, 1188–1199. [Google Scholar]
- Adams, K.M.; Kohlmeier, M.; Zeisel, S.H. Nutrition education in US medical schools: Latest update of a national survey. Acad. Med. J. Assoc. Am. Med. Coll. 2010, 85, 1537. [Google Scholar] [CrossRef]
- Chung, M.; Van Buul, V.J.; Wilms, E.; Nellessen, N.; Brouns, F. Nutrition education in European medical schools: Results of an international survey. Eur. J. Clin. Nutr. 2014, 68, 844–846. [Google Scholar] [CrossRef]
- Dumic, A.; Miskulin, M.; Pavlovic, N.; Orkic, Z.; Bilic-Kirin, V.; Miskulin, I. The nutrition knowledge of Croatian general practitioners. J. Clin. Med. 2018, 7, 178. [Google Scholar] [CrossRef] [Green Version]
- Grammatikopoulou, M.G.; Katsouda, A.; Lekka, K.; Tsantekidis, K.; Bouras, E.; Kasapidou, E.; Poulia, K.-A.; Chourdakis, M. Is continuing medical education sufficient? Assessing the clinical nutrition knowledge of medical doctors. Nutrition 2019, 57, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Hyska, J.; Mersini, E.; Mone, I.; Bushi, E.; Sadiku, E.; Hoti, K.; Bregu, A. Assessment of knowledge, attitudes and practices about public health nutrition among students of the University of Medicine in Tirana, Albania. S. East. Eur. J. Public Health (SEEJPH) 2015, I. [Google Scholar] [CrossRef]
- Devries, S.; Agatston, A.; Aggarwal, M.; Aspry, K.E.; Esselstyn, C.B.; Kris-Etherton, P.; Miller, M.; O’Keefe, J.H.; Ros, E.; Rzeszut, A.K. A deficiency of nutrition education and practice in cardiology. Am. J. Med. 2017, 130, 1298–1305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Devries, S.; Dalen, J.E.; Eisenberg, D.M.; Maizes, V.; Ornish, D.; Prasad, A.; Sierpina, V.; Weil, A.T.; Willett, W. A deficiency of nutrition education in medical training. Am. J. Med. 2014, 127, 804–806. [Google Scholar] [CrossRef]
- Crowley, J.; Ball, L.; Hiddink, G.J. Nutrition in medical education: A systematic review. Lancet Planet. Health 2019, 3, e379–e389. [Google Scholar] [CrossRef]
- Sanne, I.; Bjørke-Monsen, A.-L. Lack of nutritional knowledge among Norwegian medical students concerning vegetarian diets. J. Public Health 2020, 30, 495–501. [Google Scholar] [CrossRef]
- Bettinelli, M.E.; Bezze, E.; Morasca, L.; Plevani, L.; Sorrentino, G.; Morniroli, D.; Giannì, M.L.; Mosca, F. Knowledge of health professionals regarding vegetarian diets from pregnancy to adolescence: An observational study. Nutrients 2019, 11, 1149. [Google Scholar] [CrossRef] [Green Version]
- Hamiel, U.; Landau, N.; Fuhrer, A.E.; Shalem, T.; Goldman, M. The knowledge and attitudes of pediatricians in Israel towards vegetarianism. J. Pediatric Gastroenterol. Nutr. 2020, 71, 119–124. [Google Scholar] [CrossRef]
- Nestle, M.; Baron, R.B. Nutrition in medical education: From counting hours to measuring competence. JAMA Intern. Med. 2014, 174, 843–844. [Google Scholar] [CrossRef]
- Vetter, M.L.; Herring, S.J.; Sood, M.; Shah, N.R.; Kalet, A.L. What do resident physicians know about nutrition? An evaluation of attitudes, self-perceived proficiency and knowledge. J. Am. Coll. Nutr. 2008, 27, 287–298. [Google Scholar] [CrossRef] [Green Version]
- American Dietetic Association. Nutrition and You: Trends 2008; ADA: Chicago, IL, USA, 2008. [Google Scholar]
- Storz, M. Barriers to the Plant-Based Movement: A Physician’s Perspective. Int. J. Dis. Reversal Prev. 2020, 2, 27–30. [Google Scholar] [CrossRef]
- Jakše, B. Placing a Well-Designed Vegan Diet for Slovenes. Nutrients 2021, 13, 4545. [Google Scholar] [CrossRef] [PubMed]
- Lea, E.; Worsley, A. Influences on meat consumption in Australia. Appetite 2001, 36, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Pancrazi, R.; van Rens, T.; Vukotić, M. How distorted food prices discourage a healthy diet. Sci. Adv. 2022, 8, eabi8807. [Google Scholar] [CrossRef] [PubMed]
| Dietary Pattern | Description |
|---|---|
| Healthful plant-based | High consumption of fruits, vegetables, legumes, whole grains, nuts, and unsaturated vegetable oils, and lower or no consumption of animal products (meat, fish, poultry, dairy, and eggs) and processed foods |
| Unhealthful plant-based | High consumption of fruit juices, sugar-sweetened beverages, refined grains, potatoes, and sweets and desserts, and lower consumption of animal products (meat, fish, poultry, dairy, and eggs) and healthy plant foods (fruits, vegetables, legumes, whole grains, nuts, and unsaturated vegetable oils). |
| Vegan | Excludes all animal products (meat, fish, poultry, dairy, and eggs) and is based solely on plant-based foods |
| Vegetarian | Excludes meat, fish, and poultry but does include eggs and dairy, in addition to plant-based foods |
| Pescatarian | Excludes meat and poultry but includes fish, dairy, and eggs, in addition to plant-based foods |
| Semi-vegetarian | Includes all animal products, including meat, fish, poultry, dairy, and eggs, in addition to plant-based foods. However, red meat intake is limited |
| EAT-Lancet reference | Consists of fruits and vegetables, whole grains, legumes, nuts, and unsaturated oils; low to moderate consumption of seafood and poultry; zero to low consumption of red meat, processed meat, added sugar, refined grains, and starchy vegetables |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gibbs, J.; Cappuccio, F.P. Plant-Based Dietary Patterns for Human and Planetary Health. Nutrients 2022, 14, 1614. https://doi.org/10.3390/nu14081614
Gibbs J, Cappuccio FP. Plant-Based Dietary Patterns for Human and Planetary Health. Nutrients. 2022; 14(8):1614. https://doi.org/10.3390/nu14081614
Chicago/Turabian StyleGibbs, Joshua, and Francesco P. Cappuccio. 2022. "Plant-Based Dietary Patterns for Human and Planetary Health" Nutrients 14, no. 8: 1614. https://doi.org/10.3390/nu14081614
APA StyleGibbs, J., & Cappuccio, F. P. (2022). Plant-Based Dietary Patterns for Human and Planetary Health. Nutrients, 14(8), 1614. https://doi.org/10.3390/nu14081614







