Impact of SARS-CoV-2 Lockdown on the Preoperative Care Program of Patients Scheduled for Bariatric Surgery
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Characteristics of the Study Patients at Baseline
2.2. Endpoints
2.3. Development of the KMED
2.4. Nutritional Counseling, FDR, and Nutrient Intake Assessment
2.5. Anthropometric Evaluation, Blood Tests, and Micronutrient Status of the Study Population
2.6. Left Hepatic Lobe Volume Measurement
2.7. Statistical Analysis
3. Results
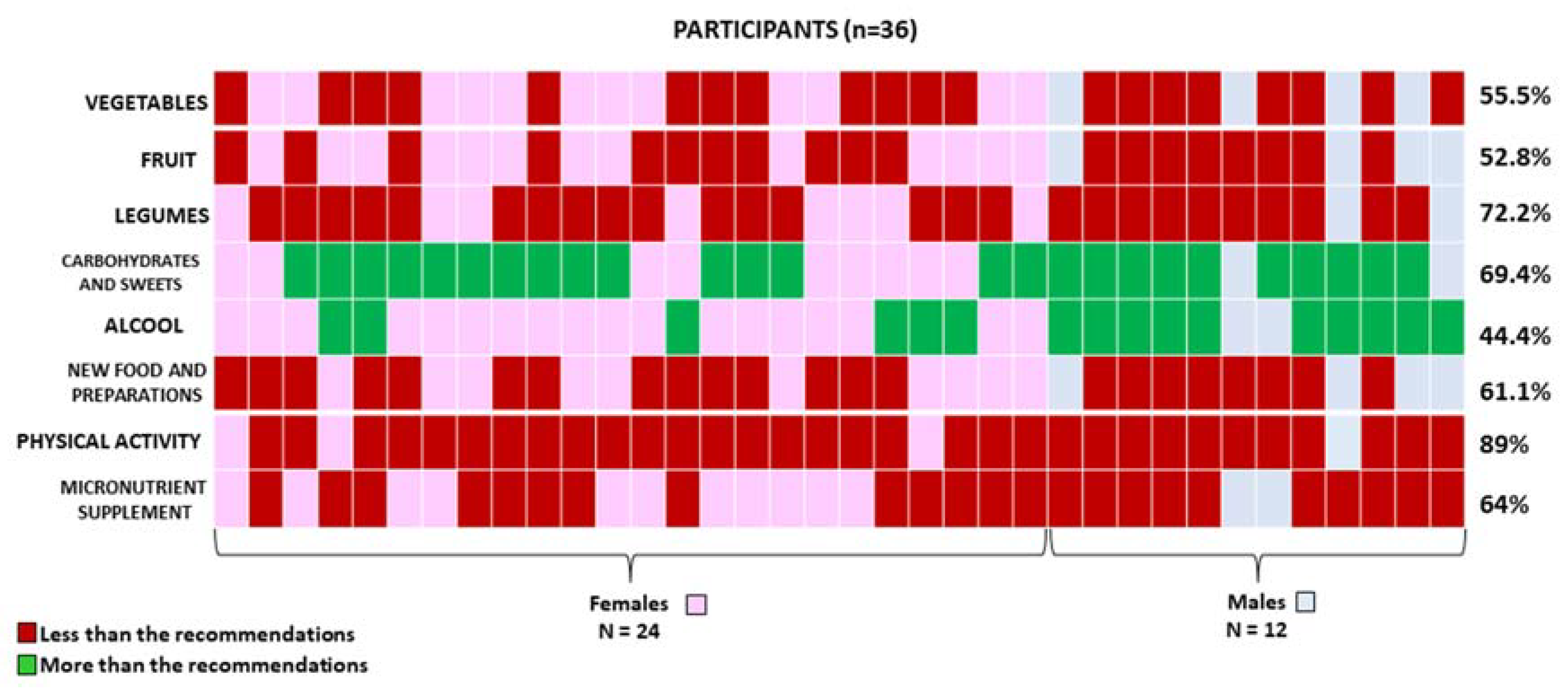
3.1. Dietary Habits Assessment and Comparison with the Prescribed KMED
3.2. Changes in BW and Left Hepatic Lobe Volume
3.3. Use of Micronutrient Supplements, Micronutrient Status, Frequency of Physical Activity, and Evolution of Comorbidities
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Osuchowski, M.F.; Winkler, M.S.; Skirecki, T.; Cajander, S.; Shankar-Hari, M.; Lachmann, G.; Monneret, G.; Venet, F.; Bauer, M.; Brunkhorst, F.M.; et al. The COVID-19 puzzle: Deciphering pathophysiology and phenotypes of a new disease entity. Lancet Respir. Med. 2021, 9, 622–642. [Google Scholar] [CrossRef]
- Cereda, A.A.; Toselli, M.; Palmisano, A.; Vignale, D.; Khokhar, A.; Campo, G.; Bertini, M.; Loffi, M.; Andreini, D.; Pontone, G.; et al. Coronary calcium score as a predictor of outcomes in the hypertensive COVID-19 population: Results from the Italian (S) Core-COVID-19 Registry. Hypertens. Res. 2022, 45, 333–343. [Google Scholar] [CrossRef]
- Thorell, A. The 2020 ESPEN Arvid Wretlind lecture: Metabolic response in bariatric surgery—Mechanisms and clinical implications. Clin. Nutr. 2021, 40, 2602–2608. [Google Scholar] [CrossRef] [PubMed]
- Schiavo, L.; Scalera, G.; Sergio, R.; De Sena, G.; Pilone, V.; Barbarisi, A. Clinical impact of Mediterranean enriched-protein diet on liver size, visceral fat, fat mass, and fat-free mass in patients undergoing sleeve gastrectomy. Surg. Obes. Relat. Dis. 2015, 11, 1164–1170. [Google Scholar] [CrossRef] [PubMed]
- Iannelli, A.; Martini, F.; Schneck, A.S.; Ghavami, B.; Baudin, G.; Anty, R.; Gugenheim, J. Pre-operative 4-week supplementation with omega-3 polyunsaturated fatty acid reduces liver volume and facilitates bariatric surgery in morbidly obese patients. Obes. Surg. 2013, 23, 1761–1765. [Google Scholar] [CrossRef]
- Schiavo, L.; Sans, A.; Scalera, G.; Barbarisi, A.; Iannelli, A. Why Preoperative Weight Loss in Preparation for Bariatric Surgery Is Important. Obes. Surg. 2016, 26, 2790–2792. [Google Scholar] [CrossRef]
- Parrott, J.; Frank, L.; Rabena, R.; Craggs-Dino, L.; Isom, K.A.; Greiman, L. American Society for Metabolic and Bariatric Surgery Integrated Health Nutritional Guidelines for the Surgical Weight Loss Patient 2016 Update: Micronutrients. Surg. Obes. Relat. Dis. 2017, 13, 727–741. [Google Scholar] [CrossRef]
- Schiavo, L.; Scalera, G.; Pilone, V.; De Sena, G.; Capuozzo, V.; Barbarisi, A. Micronutrient Deficiencies in Patients Candidate for Bariatric Surgery: A Prospective, Preoperative Trial of Screening, Diagnosis, and Treatment. Int. J. Vitam. Nutr. Res. 2015, 85, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Barrea, L.; Verde, L.; Vetrani, C.; Marino, F.; Aprano, S.; Savastano, S.; Colao, A.; Muscogiuri, G. VLCKD: A real time safety study in obesity. J. Transl. Med. 2022, 20, 23. [Google Scholar] [CrossRef]
- Schiavo, L.; Pilone, V.; Rossetti, G.; Barbarisi, A.; Cesaretti, M.; Iannelli, A. A 4-Week Preoperative Ketogenic Micronutrient-Enriched Diet Is Effective in Reducing Body Weight, Left Hepatic Lobe Volume, and Micronutrient Deficiencies in Patients Undergoing Bariatric Surgery: A Prospective Pilot Study. Obes. Surg. 2018, 28, 2215–2224. [Google Scholar] [CrossRef]
- Pellegrini, M.; Ponzo, V.; Rosato, R.; Scumaci, E.; Goitre, I.; Benso, A.; Belcastro, S.; Crespi, C.; De Michieli, F.; Ghigo, E.; et al. Changes in Weight and Nutritional Habits in Adults with Obesity during the “Lockdown” Period Caused by the COVID-19 Virus Emergency. Nutrients 2020, 12, 2016. [Google Scholar] [CrossRef] [PubMed]
- Schiavo, L.; Pierro, R.; Asteria, C.; Calabrese, P.; Di Biasio, A.; Coluzzi, I.; Severino, L.; Giovanelli, A.; Pilone, V.; Silecchia, G. Low-Calorie Ketogenic Diet with Continuous Positive Airway Pressure to Alleviate Severe Obstructive Sleep Apnea Syndrome in Patients with Obesity Scheduled for Bariatric/Metabolic Surgery: A Pilot, Prospective, Randomized Multicenter Comparative Study. Obes. Surg. 2022, 32, 634–642. [Google Scholar] [CrossRef] [PubMed]
- Ortega, R.M.; Pérez-Rodrigo, C.; López-Sobaler, A.M. Dietary assessment methods: Dietary records. Nutr. Hosp. 2015, 31, 38–45. [Google Scholar]
- Schiavo, L.; Di Rosa, M.; Tramontano, S.; Rossetti, G.; Iannelli, A.; Pilone, V. Long-Term Results of the Mediterranean Diet after Sleeve Gastrectomy. Obes. Surg. 2020, 30, 3792–3802. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.Y.; Bansal, N. Measured GFR as “gold standard”—all that glitters is not gold? Clin. J. Am. Soc. Nephrol. 2011, 6, 1813–1814. [Google Scholar] [CrossRef] [Green Version]
- Smith, J.C., Jr.; Butrimovitz, G.P.; Purdy, W.C. Direct measurement of zinc in plasma by atomic adsorption spectroscopy. Clin. Chem. 1979, 25, 1487–1491. [Google Scholar] [CrossRef]
- Fris, R.J. Preoperative low energy diet diminishes liver size. Obes. Surg. 2004, 14, 1165–1170. [Google Scholar] [CrossRef] [PubMed]
- Carter, J.; Chang, J.; Birriel, T.J.; Moustarah, F.; Sogg, S.; Goodpaster, K.; Benson-Davies, S.; Chapmon, K.; Eisenberg, D. ASMBS position statement on preoperative patient optimization before metabolic and bariatric surgery. Surg. Obes. Relat. Dis. 2021, 17, 1956–1976. [Google Scholar] [CrossRef] [PubMed]
- Schiavo, L.; Scalera, G.; Barbarisi, A. Micronutrient deficiencies and sleeve gastrectomy for weight reduction. In Metabolism and Pathophysiology of Bariatric Surgery, Nutrition, Procedures, Outcomes, and Adverse Effects; Preedy, V.R., Rajendram, R., Martin, C.R., Eds.; Academic Press: Cambridge, MA, USA, 2016; pp. 469–477. [Google Scholar]
- Bettini, S.; Belligoli, A.; Fabris, R.; Busetto, L. Diet approach before and after bariatric surgery. Rev. Endocr. Metab. Disord. 2020, 21, 297–306. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, B.; Raggi, P. The ketogenic diet: Pros and cons. Atherosclerosis 2020, 292, 119–126. [Google Scholar] [CrossRef] [Green Version]
- Tarantino, G.; Citro, V.; Capone, D. Nonalcoholic Fatty Liver Disease: A Challenge from Mechanisms to Therapy. J. Clin. Med. 2019, 9, 15. [Google Scholar] [CrossRef] [Green Version]
- Schiavo, L.; Busetto, L.; Cesaretti, M.; Zelber-Sagi, S.; Deutsch, L.; Iannelli, A. Nutritional issues in patients with obesity and cirrhosis. World J. Gastroenterol. 2018, 24, 3330–3346. [Google Scholar] [CrossRef] [PubMed]
- Schübel, R.; Nonnenmacher, T.; Sookthai, D.; Maldonado, S.G.; Sowah, S.A.; Von Stackelberg, O.; Schlett, C.L.; Grafetstätter, M.; Nabers, D.; Johnson, T.; et al. Similar Weight Loss Induces Greater Improvements in Insulin Sensitivity and Liver Function among Individuals with NAFLD Compared to Individuals without NAFLD. Nutrients 2019, 11, 544. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pan, C.S.; Stanley, T.L. Effect of Weight Loss Medications on Hepatic Steatosis and Steatohepatitis: A Systematic Review. Front. Endocrinol. 2020, 11, 70. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hydes, T.J.; Ravi, S.; Loomba, R.; Gray, M. Evidence-based clinical advice for nutrition and dietary weight loss strategies for the management of NAFLD and NASH. Clin. Mol. Hepatol. 2020, 26, 383–400. [Google Scholar] [CrossRef] [PubMed]
- Regan, J.P.; Inabnet, W.B.; Gagner, M.; Pomp, A. Early experience with 380 two-stage laparoscopic Roux-en-Y gastric bypass as an alternative in 381 the super-super obese patient. Obes. Surg. 2003, 13, 861–864. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.T.; Longoria, M.; Gelfand, D.V.; Sabio, A.; Wilson, S.E. Staged laparoscopic Roux-en-Y: A novel two-stage bariatric operation as an alternative in the super-obese with massively enlarged liver. Obes. Surg. 2005, 15, 1077–1081. [Google Scholar] [CrossRef] [Green Version]
- Iannelli, A.; Kassir, R.; Gugenheim, J. The long and narrow gastric pouch for laparoscopic Roux-en-Y gastric bypass. Obes. Surg. 2014, 24, 1744–1745. [Google Scholar] [CrossRef] [PubMed]
- Iannelli, A.; Schneck, A.S.; Hébuterne, X.; Gugenheim, J. Gastric pouch resizing for Roux-en-Y gastric bypass failure. Surg. Obes. Relat. Dis. 2013, 9, 260–267. [Google Scholar] [CrossRef]
- Ben-Porat, T.; Elazary, R.; Yuval, J.B.; Wieder, A.; Khalaileh, A.; Weiss, R. Nutritional deficiencies following sleeve gastrectomy—Can they be predicted pre-operatively? Surg. Obes. Relat. Dis. 2015, 11, 1029–1036. [Google Scholar] [CrossRef] [PubMed]
- Thibault, R.; Huber, O.; Azagury, D.E.; Pichard, C. Twelve key nutritional issues in bariatric surgery. Clin. Nutr. 2016, 35, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Schiavo, L.; Pilone, V.; Rossetti, G.; Iannelli, A. The Role of the Nutritionist in a Multidisciplinary Bariatric Surgery Team. Obes. Surg. 2019, 29, 1028–1030. [Google Scholar] [CrossRef] [PubMed]
- Schiavo, L.; Pilone, V.; Rossetti, G.; Romano, M.; Pieretti, G.; Schneck, A.S.; Iannelli, A. Correcting micronutrient deficiencies before sleeve gastrectomy may be useful in preventing early postoperative micronutrient deficiencies. Int. J. Vitam. Nutr. Res. 2019, 89, 22–28. [Google Scholar] [CrossRef] [PubMed]
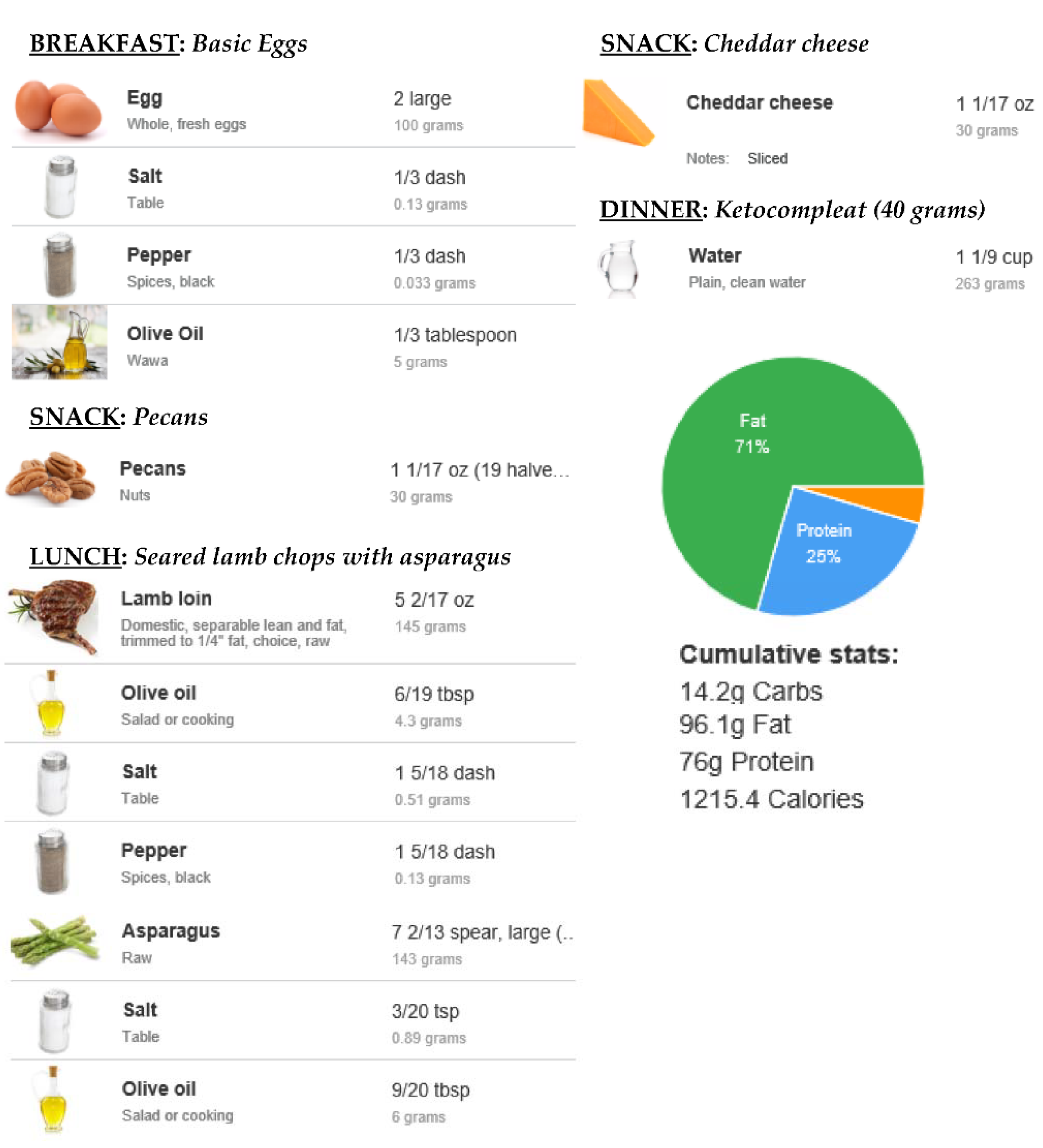
| Element | Daily Value (40 g) | RDA * |
|---|---|---|
| Soy Protein | 33.31 g | - |
| Vitamin A | 0.483 mg | 60.38% |
| Vitamin B1 | 0.496 mg | 45.09% |
| Vitamin B2 | 0.833 mg | 59.50% |
| Vitamin B3 | 8.33 mg | 52.06% |
| Vitamin B5 | 1.28 mg | 21.33% |
| Vitamin B6 | 0.92 mg | 65.71% |
| Biotin | 0.028 mg | 56% |
| Vitamin B12 | 0.007 mg | 280% |
| Folic Acid | 0.222 mg | 111% |
| Vitamin C | 83.28 mg | 104.10% |
| Vitamin D3 | 0.011 mg | 222% |
| Vitamin E | 12.65 mg | 105.42% |
| Vitamin K1 | 11.1 mg | 14.80% |
| Iron | 5.5 mg | 39.29% |
| Copper | 0.08 mg | 8% |
| Magnesium | 39.9 mg | 10.64% |
| Selenium | 0.015 mg | 27.27% |
| Manganese | 0.19 mg | 9.50% |
| Chromium | 0.003 mg | 7.50% |
| Calcium | 116.76 mg | 14.60% |
| Zinc | 8.74 mg | 87.40% |
| Inulin | 1.38 g | - |
| Phaseolamin | 1.11 g | - |
| Fructo-oligo-saccharides | 1.11 g | - |
| Lactobacillus plantarum | 2.24 mld | - |
| Lactobacillus acidophilus | 2.2 mld | - |
| Lactobacillus rhamnosus | 2.2 mld | - |
| Bifidobacterium longum | 2.24 mld | - |
| Saccharomyces boulardi | 4.48 mld | - |
| Clinical Characteristics | Baseline Mean ± SD | 4-Week Follow Up Mean ± SD | p |
|---|---|---|---|
| Patients (M/F) | 12/24 | 12/24 | - |
| Body weight, kg (M/F) | 143.6 ± 23.6 | 151 ± 16.1 | 0.115 |
| BMI (kg/m2) | 50.1 ± 5.9 | 52.8 ± 6.5 | 0.0692 |
| Left hepatic lobe volume (cm3) | 627 ± 85 | 631 ± 91 | 0.0848 |
| Glucose (mg/dL) | 115 ± 26.3 | 119 ± 23.9 | 0.502 |
| Insulin (mU/L) | 10.5 ± 5.9 | 10.9 ± 5.7 | 0.771 |
| Iron (g/dL) | 64.9 ± 12.7 | 66.5 ± 11.7 | 0.580 |
| Hb (mcg/dL) | 12.5 ± 5.9 | 12.9 ± 6.3 | 0.782 |
| Creatinine (mg/dL) | 0.86 ± 1.3 | 0.81 ± 1.2 | 0.866 |
| Ketonemia (mmol/L) | 0.05 ± 0.04 | 0.04 ± 0.02 | 0.184 |
| Total Cholesterol (mg/dL) | 224 ± 19.4 | 235 ± 21.5 | 0.0257 |
| HDL (mg/dL) | 51 ± 11.5 | 48 ± 11.9 | 0.280 |
| LDL (mg/dL) | 162 ± 27.9 | 167 ± 22.9 | 0.409 |
| Total Cholesterol/HDL ratio | 4.4 ± 2.9 | 4.9 ± 2.4 | 0.428 |
| Triglycerides (mg/dL) | 204 ± 35.4 | 221 ± 34.1 | 0.0416 |
| GOT (U/L) | 53 ± 14.1 | 56 ± 16.3 | 0.406 |
| GPT (U/L) | 39 ± 19.3 | 41 ± 18.6 | 0.656 |
| GGT (U/L) | 31 ± 19.6 | 33 ± 17.2 | 0.647 |
| Uric Acid (mg/dL) | 6.7 ± 1.2 | 6.9 ± 1.8 | 0.581 |
| Urea/mg/dL) | 31.4 ± 11.2 | 30.6 ± 13.2 | 0.782 |
| GFR (mL/min) | 95.4 ± 23.2 | 96.2 ± 26.3 | 0.891 |
| † Vitamin B12 deficiency (M/F) | 7/8 | 6/8 | - |
| † Folate (M/F) | 6/8 | 5/8 | - |
| † Vitamin D deficiency (M/F) | 10/12 | 10/12 | - |
| † Iron deficiency (M/F) | 4/8 | 2/8 | - |
| † Zinc deficiency (M/F) | 2/7 | 2/7 | - |
| † Hypertension (M/F) | 5/3 | 5/3 | - |
| † Diabetes type 2 (M/F) | 4/2 | 4/2 | - |
| † Dyslipidemia (M/F) | 4/4 | 4/4 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schiavo, L.; Calabrese, P.; Aliberti, S.M.; Tramontano, S.; Iannelli, A.; Pilone, V. Impact of SARS-CoV-2 Lockdown on the Preoperative Care Program of Patients Scheduled for Bariatric Surgery. Nutrients 2022, 14, 1488. https://doi.org/10.3390/nu14071488
Schiavo L, Calabrese P, Aliberti SM, Tramontano S, Iannelli A, Pilone V. Impact of SARS-CoV-2 Lockdown on the Preoperative Care Program of Patients Scheduled for Bariatric Surgery. Nutrients. 2022; 14(7):1488. https://doi.org/10.3390/nu14071488
Chicago/Turabian StyleSchiavo, Luigi, Pietro Calabrese, Silvana Mirella Aliberti, Salvatore Tramontano, Antonio Iannelli, and Vincenzo Pilone. 2022. "Impact of SARS-CoV-2 Lockdown on the Preoperative Care Program of Patients Scheduled for Bariatric Surgery" Nutrients 14, no. 7: 1488. https://doi.org/10.3390/nu14071488
APA StyleSchiavo, L., Calabrese, P., Aliberti, S. M., Tramontano, S., Iannelli, A., & Pilone, V. (2022). Impact of SARS-CoV-2 Lockdown on the Preoperative Care Program of Patients Scheduled for Bariatric Surgery. Nutrients, 14(7), 1488. https://doi.org/10.3390/nu14071488






