Effects of Exercise on Nutritional Status in People with Cystic Fibrosis: A Systematic Review
Abstract
:1. Introduction
2. Materials and Methods
2.1. Systematic Review Design and Registration and Design
2.2. Data Sources and Searches
2.3. Eligibility Criteria
2.4. Study Selection
2.5. Data Extraction
2.6. Quality Assessment
2.7. Data Synthesis and Analysis
3. Results
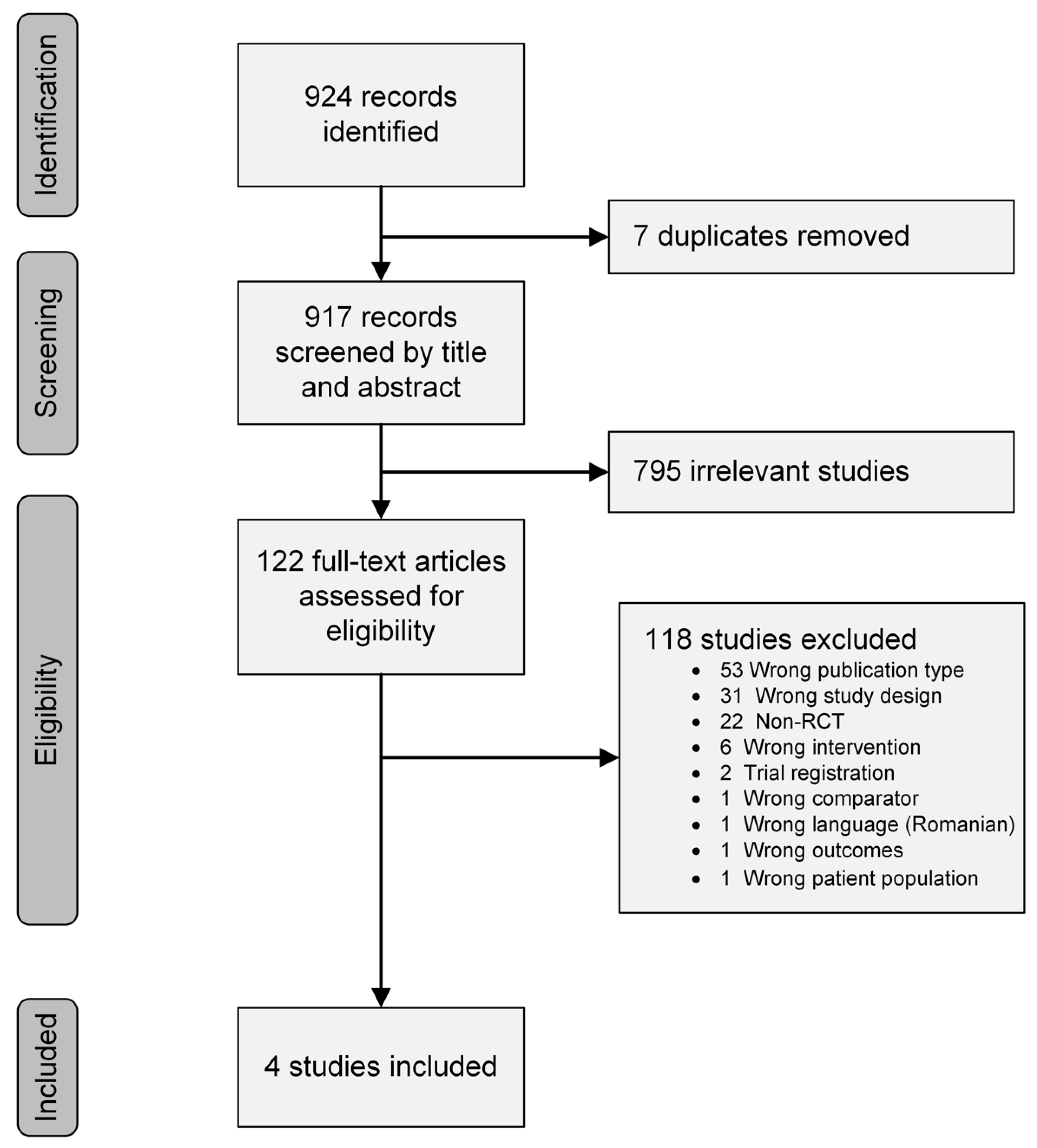
3.1. Study Search Results and Selection
3.2. Description of the Studies
Individual Study Descriptions
3.3. Study Quality
3.4. Effects of Intervention
3.4.1. Nutritional Status Outcomes
3.4.2. Physiologic Outcomes
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Cystic Fibrosis Foundation. What Is Cystic Fibrosis? Available online: https://www.cff.org/intro-cf/about-cystic-fibrosis#what-is-cystic-fibrosis? (accessed on 4 January 2022).
- O’Sullivan, B.P.; Freedman, S.D. Cystic fibrosis. Lancet 2009, 373, 1891–1904. [Google Scholar] [CrossRef]
- Wouthuyzen-Bakker, M.; Bodewes, F.A.; Verkade, H.J. Persistent fat malabsorption in cystic fibrosis; lessons from patients and mice. J. Cyst. Fibros. 2011, 10, 150–158. [Google Scholar] [CrossRef] [Green Version]
- Ionescu, A.A.; Nixon, L.S.; Luzio, S.; Lewis-Jenkins, V.; Evans, W.D.; Stone, M.D.; Owens, D.R.; Routledge, P.A.; Shale, D.J. Pulmonary function, body composition, and protein catabolism in adults with cystic fibrosis. Am. J. Respir. Crit. Care Med. 2002, 165, 495–500. [Google Scholar] [CrossRef]
- Culhane, S.; George, C.; Pearo, B.; Spoede, E. Malnutrition in cystic fibrosis: A review. Nutr. Clin. Pract. 2013, 28, 676–683. [Google Scholar] [CrossRef]
- Hulzebos, E.H.; Bomhof-Roordink, H.; van de Weert-van Leeuwen, P.B.; Twisk, J.W.; Arets, H.G.; van der Ent, C.K.; Takken, T. Prediction of mortality in adolescents with cystic fibrosis. Med. Sci. Sports Exerc. 2014, 46, 2047–2052. [Google Scholar] [CrossRef] [PubMed]
- Steinkamp, G.; Wiedemann, B. Relationship between nutritional status and lung function in cystic fibrosis: Cross sectional and longitudinal analyses from the German CF quality assurance (CFQA) project. Thorax 2002, 57, 596–601. [Google Scholar] [CrossRef] [Green Version]
- Stallings, V.A.; Stark, L.J.; Robinson, K.A.; Feranchak, A.P.; Quinton, H. Evidence-based practice recommendations for nutrition-related management of children and adults with cystic fibrosis and pancreatic insufficiency: Results of a systematic review. J. Am. Diet. Assoc. 2008, 108, 832–839. [Google Scholar] [CrossRef]
- Nixon, P.A.; Orenstein, D.M.; Kelsey, S.F.; Doershuk, C.F. The Prognostic Value of Exercise Testing in Patients with Cystic Fibrosis. N. Engl. J. Med. 1992, 327, 1785–1788. [Google Scholar] [CrossRef]
- Hebestreit, H.; Hulzebos, E.H.J.; Schneiderman, J.E.; Karila, C.; Boas, S.R.; Kriemler, S.; Dwyer, T.; Sahlberg, M.; Urquhart, D.S.; Lands, L.C.; et al. Cardiopulmonary Exercise Testing Provides Additional Prognostic Information in Cystic Fibrosis. Am. J. Respir. Crit. Care Med. 2019, 199, 987–995. [Google Scholar] [CrossRef] [Green Version]
- Schneiderman, J.E.; Wilkes, D.L.; Atenafu, E.G.; Nguyen, T.; Wells, G.D.; Alarie, N.; Tullis, E.; Lands, L.C.; Coates, A.L.; Corey, M.; et al. Longitudinal relationship between physical activity and lung health in patients with cystic fibrosis. Eur. Respir. J. 2014, 43, 817–823. [Google Scholar] [CrossRef] [Green Version]
- Gabel, M.E.; Fox, C.K.; Grimes, R.A.; Lowman, J.D.; McDonald, C.M.; Stallings, V.A.; Michel, S.H. Overweight and cystic fibrosis: An unexpected challenge. Pediatr. Pulmonol. 2022, 57, S40–S49. [Google Scholar] [CrossRef]
- Bailey, J.; Rozga, M.; McDonald, C.M.; Bowser, E.K.; Farnham, K.; Mangus, M.; Padula, L.; Porco, K.; Alvarez, J.A. Effect of CFTR Modulators on Anthropometric Parameters in Individuals with Cystic Fibrosis: An Evidence Analysis Center Systematic Review. J. Acad. Nutr. Diet. 2021, 121, 1364–1378.e1362. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Syst. Rev. 2021, 10, 89. [Google Scholar] [CrossRef] [PubMed]
- Lowman, J.; Nicolson, W.; Alotaibi, N.; Krick, S.; Bailey, J. Exercise for Weight Management in People with Cystic Fibrosis: A Systematic Review. PROSPERO 2021 CRD42021273303. Available online: https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42021273303 (accessed on 10 January 2022).
- Covidence Systematic Review Software, Veritas Health Innovation, Melbourne, Australia. Available online: www.covidence.org (accessed on 10 January 2022).
- National Center for Health Statistics in Collaboration with the National Center for Chronic Disease Prevention and Health Promotion. Clinical Growth Charts. Available online: http://www.cdc.gov/growthcharts (accessed on 4 January 2022).
- Hommerding, P.X.; Baptista, R.R.; Makarewicz, G.T.; Schindel, C.S.; Donadio, M.V.; Pinto, L.A.; Marostica, P.J. Effects of an educational intervention of physical activity for children and adolescents with cystic fibrosis: A randomized controlled trial. Respir. Care 2015, 60, 81–87. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Selvadurai, H.C.; Blimkie, C.J.; Meyers, N.; Mellis, C.M.; Cooper, P.J.; Van Asperen, P.P. Randomized controlled study of in-hospital exercise training programs in children with cystic fibrosis. Pediatr. Pulmonol. 2002, 33, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Santana-Sosa, E.; Groeneveld, I.F.; Gonzalez-Saiz, L.; López-Mojares, L.M.; Villa-Asensi, J.R.; Barrio Gonzalez, M.I.; Fleck, S.J.; Pérez, M.; Lucia, A. Intrahospital weight and aerobic training in children with cystic fibrosis: A randomized controlled trial. Med. Sci. Sports Exerc. 2012, 44, 2–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santana-Sosa, E.; Gonzalez-Saiz, L.; Groeneveld, I.F.; Villa-Asensi, J.R.; Barrio Gómez de Aguero, M.I.; Fleck, S.J.; López-Mojares, L.M.; Pérez, M.; Lucia, A. Benefits of combining inspiratory muscle with ‘whole muscle’ training in children with cystic fibrosis: A randomised controlled trial. Br. J. Sports Med. 2014, 48, 1513–1517. [Google Scholar] [CrossRef] [PubMed]
- Hebestreit, H.; Arets, H.G.; Aurora, P.; Boas, S.; Cerny, F.; Hulzebos, E.H.; Karila, C.; Lands, L.C.; Lowman, J.D.; Swisher, A.; et al. Statement on Exercise Testing in Cystic Fibrosis. Respiration 2015, 90, 332–351. [Google Scholar] [CrossRef]
- Shei, R.J.; Dekerlegand, R.L.; Mackintosh, K.A.; Lowman, J.D.; McNarry, M.A. Inspiration for the Future: The Role of Inspiratory Muscle Training in Cystic Fibrosis. Sports Med. Open 2019, 5, 36. [Google Scholar] [CrossRef] [Green Version]
- Cashin, A.G.; McAuley, J.H. Clinimetrics: Physiotherapy Evidence Database (PEDro) Scale. J. Physiother. 2020, 66, 59. [Google Scholar] [CrossRef]
- Must, A.; Anderson, S.E. Body mass index in children and adolescents: Considerations for population-based applications. Int. J. Obes. 2006, 30, 590–594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radtke, T.; Nevitt, S.J.; Hebestreit, H.; Kriemler, S. Physical exercise training for cystic fibrosis. Cochrane Database Syst. Rev. 2017, 11, Cd002768. [Google Scholar] [CrossRef] [PubMed]
- Elce, A.; Nigro, E.; Gelzo, M.; Iacotucci, P.; Carnovale, V.; Liguori, R.; Izzo, V.; Corso, G.; Castaldo, G.; Daniele, A.; et al. Supervised physical exercise improves clinical, anthropometric and biochemical parameters in adult cystic fibrosis patients: A 2-year evaluation. Clin. Respir. J. 2018, 12, 2228–2234. [Google Scholar] [CrossRef] [PubMed]
- Van Biervliet, S.; Declercq, D.; Dereeper, S.; Vermeulen, D.; Würth, B.; De Guschtenaere, A. The effect of an intensive residential rehabilitation program on body composition in patients with cystic fibrosis. Eur. J. Pediatr. 2021, 180, 1981–1985. [Google Scholar] [CrossRef]
- Hebestreit, H.; Kriemler, S.; Schindler, C.; Stein, L.; Karila, C.; Urquhart, D.S.; Orenstein, D.M.; Lands, L.C.; Schaeff, J.; Eber, E.; et al. Effects of a Partially Supervised Conditioning Program in Cystic Fibrosis: An International Multicenter, Randomized Controlled Trial (ACTIVATE-CF). Am. J. Respir. Crit. Care Med. 2022, 205, 330–339. [Google Scholar] [CrossRef]
- Hebestreit, H.; Lands, L.C.; Alarie, N.; Schaeff, J.; Karila, C.; Orenstein, D.M.; Urquhart, D.S.; Hulzebos, E.H.J.; Stein, L.; Schindler, C.; et al. Effects of a partially supervised conditioning programme in cystic fibrosis: An international multi-centre randomised controlled trial (ACTIVATE-CF): Study protocol. BMC Pulm. Med. 2018, 18, 31. [Google Scholar] [CrossRef] [Green Version]
- Swisher, A.K.; Hebestreit, H.; Mejia-Downs, A.; Lowman, J.D.; Gruber, W.; Nippins, M.; Alison, J.; Schneiderman, J. Exercise and Habitual Physical Activity for PeopleWith Cystic Fibrosis: Expert Consensus, Evidence-Based Guide for Advising Patients. Cardiopulm. Phys. Ther. J. 2015, 26, 85–98. [Google Scholar] [CrossRef]
- Bradley, J.; O’Neill, B.; Kent, L.; Hulzebos, E.H.; Arets, B.; Hebestreit, H. Physical activity assessment in cystic fibrosis: A position statement. J. Cyst. Fibros. 2015, 14, e25–e32. [Google Scholar] [CrossRef] [Green Version]
- McDonald, C.M.; Alvarez, J.A.; Bailey, J.; Bowser, E.K.; Farnham, K.; Mangus, M.; Padula, L.; Porco, K.; Rozga, M. Academy of Nutrition and Dietetics: 2020 Cystic Fibrosis Evidence Analysis Center Evidence-Based Nutrition Practice Guideline. J. Acad. Nutr. Diet. 2021, 121, 1591–1636.e1593. [Google Scholar] [CrossRef]
| PICOS Parameter | Inclusion Criteria | Exclusion Criteria |
|---|---|---|
| Population | Children and adults with cystic fibrosis (underweight, normal weight, or overweight) | Infants, toddlers and preschoolers (<5 years old) |
| Intervention | Exercise or physical activity | Passive exercise (e.g., stretching, range of motion) |
| Comparison | Non-exposed control group | |
| Outcome | Body mass index, body mass, body composition (e.g., fat-free mass) | |
| Study design | Randomized controlled trials | Language other than English, German, Spanish, or French |
| Ref. | First Author | Year | State/Country | Number of Participants (Female) | Age | FEV1% | Body Mass (kg) | Weight for Age% † | BMI (kg/m2) | BMI for Age% § |
|---|---|---|---|---|---|---|---|---|---|---|
| [19] | Selvadurai, H.C. | 2002 | New South Wales, Australia | 66 (38) | 13 (2) | 57 (17) | 38.0 (7.8) | 16th | NA | NA |
| [20] | Santana-Sosa, E. | 2012 | Madrid, Spain | 22 (9) | 10.5 (2) | 83 (11) * | 37.0 (3.0) | 65th | 17.8 | 61st |
| [21] | Santana-Sosa, E. | 2014 | Madrid, Spain | 20 (8) | 10.5 (1) | 73 (9) * | 34.0 (3.8) | 47th | 16.1 | 34th |
| [18] | Hommerding, P.X. | 2015 | Rio Grande do Sul, Brazil | 34 (14) | 13 (3) | 98 (20) | 45.6 (15.3) | 50th | NA | NA |
| Ref. | Setting (Duration) | Exercise Group(s) | Control Group | |
|---|---|---|---|---|
| [19] | Acute/ Inpatient (~2–3 wks) | Aerobic exercise training | Resistance exercise training | No exercise |
| Mode: treadmill or stationary cycling | Mode: Isotonic weight machines | |||
| Intensity: 70% of HRpeak | Intensity: 70% 1 RM | |||
| Duration: 30 min | Duration: 5 sets of 10 repetitions | |||
| Frequency: 5 d/wk | Frequency: 5 d/wk | |||
| Other: Supplemental oxygen was titrated to keep SpO2 > 90% (if needed). Training was stopped if dyspnea ≥ 7 on Borg CR10 scale. Each session was individually supervised | Other: Upper- and lower-extremity exercises (specific exercises and number not defined). Each session was individually supervised | |||
| [20] | Hospital-based, outpatient gym (8 weeks) | Aerobic exercise training | Chest physiotherapy twice daily and provided verbal instruction on the benefits of physical activity | |
| Mode: Cycle ergometer | ||||
| Intensity: HR at ventilatory threshold (determined during exercise test) | ||||
| Duration: 20–40 min | ||||
| Frequency: 3 d/wk | ||||
| Other: HR monitor was worn during aerobic exercise. 10 min warmup on cycle. Each session was individually supervised | ||||
| Resistance exercise training | ||||
| Mode: Isotonic weight machines (bench press, shoulder press, leg extension, leg press, leg curl, abdominal crunch, low back extension, arm curl, elbow extension, seated row, and lateral pulldown) | ||||
| Intensity: Progressive, from 40 to 60% of 5 RM | ||||
| Duration: 3 circuits of 1 set of 12–15 repetitions of each exercise | ||||
| Frequency: 3 d/wk (following aerobic exercise session) | ||||
| [21] | Hospital-based, outpatient gym (8 weeks) | Aerobic exercise | Chest physiotherapy twice daily, IMT at 10% of PImax, and provided instruction on the benefits of physical activity | |
| Mode: Cycle ergometer and “active playing” (i.e., running and soccer) | ||||
| Intensity: HR at ventilatory threshold (determined during exercise test) | ||||
| Duration: 20–40 min | ||||
| Frequency: 3 d/wk | ||||
| Other: HR monitor was worn during aerobic exercise. 10 min warmup on cycle. Each session was individually supervised | ||||
| Resistance exercise | ||||
| Mode: isotonic weight machine (leg press, pull down, leg extension, bench press, leg curl, seated row and abdominal crunch) | ||||
| Intensity: Progressive, beginning at 50% of 5 RM | ||||
| Duration: 3 circuits of 1 set of 12–15 repetitions of each exercise | ||||
| Frequency: 3 d/wk (following aerobic exercise) | ||||
| Inspiratory muscle training | ||||
| Mode: PowerBreathe threshold loading device | ||||
| Intensity: 40–50% of PImax. | ||||
| Duration: 30 breaths | ||||
| Frequency: twice daily, 6–7 d/wk | ||||
| Other: One IMT session was performed during the 3 d/wk supervised sessions; the remaining IMT sessions were performed independently at home | ||||
| [18] | Home-based with tele-health follow-up every 2 weeks (3 months) | Aerobic exercise | Verbal instructions regarding aerobic exercise which are part of routine outpatient care | |
| Mode: Self-selected per recommendations (e.g., walking, jogging, swimming, dancing skipping rope) | ||||
| Intensity: No recommendations given | ||||
| Duration: ≥20 min | ||||
| Frequency: at least 2 d/wk | ||||
| Other: Written manual of aerobic and stretching exercises provided | ||||
| Ref. | Nutritional Status Outcomes | Physiologic Outcomes | Conclusions | |
|---|---|---|---|---|
| BMI | Other | |||
| [19] | Not reported/unable to calculate based on data reported | ∆ body mass (kg): | ∆ VO2 peak (mL/kg/min): | AET improved body composition (2%) and peak VO2 (22%) RET improved body composition (7%) and was the only group to improve LE strength (18%) The CTL group had an improvement in body mass (2.7%), but an insignificant loss of strength and aerobic capacity |
| AET ↑ 0.80 (0.64) * | AET ↑ 7.3 (6.3) * | |||
| RET ↑ 2.76 (0.70) * | RET ↑ 0.7 (5.9) | |||
| CTL ↑ 1.03 (0.58) * | CTL ↓ 1.2 (6.2) | |||
| ∆ fat-free mass (kg): | ∆ strength (Nm): | |||
| AET ↑ 0.61 (0.37) * | AET ↑ 1.8 (6.2) | |||
| RET ↑ 2.40 (0.46) * | RET ↑ 18.3 (7.0) * | |||
| CTL ↑ 0.60 (0.32) * | CTL ↓ 6.3 (6.1) | |||
| [20] | ∆ BMI (kg/m2): ET ↓ 0.1 CTL ↓ 0.1 | ∆ body mass | ∆ VO2 peak | No significant changes in body composition variables. Peak VO2 improved ~10% and strength ~25% in the ET group compared to a ~6% decrease in peak VO2 and −2 to +5% change in strength of the CTL group |
| (kg): | (mL/kg/min): | |||
| ET ↑ 0.6 | ET ↑ 3.9 (2–6) * | |||
| CTL ↑ 1.1 | CTL ↓ 2.2 (−5–0) | |||
| ∆ fat-free mass (%): | ∆ strength (kg): | |||
| ET ↑ 1.3 | ET ↑ 10.5 (7–14) * | |||
| CTL ↓ 0.2 | CTL not reported | |||
| [21] | Not reported/unable to calculate based on data reported | ∆ body mass | ∆ VO2 peak (mL/kg/min): | No significant changes in body mass, but fat-free mass increased in the ET group. Peak VO2, LE strength, and inspiratory muscle strength increased 22%, 43%, and 58%, respectively, in the ET group. There were no significant changes in the CTL group |
| ET ↑ 6.9 * | ||||
| (kg): | CTL ↓ 0.6 | |||
| ET ↑ 1.4 | ∆ strength (kg): | |||
| CTL ↑ 0.9 | ET ↑ 27 * | |||
| ∆ fat-free mass (%): | CTL ↓ 1.3 | |||
| ET ↑ 1.0 | ∆ PImax (mm Hg): | |||
| CTL ↓ 0.1 | ET ↑ 39 * | |||
| CTL ↑2.3 | ||||
| [18] | ∆ BMI z-score: ET ↑ 0.2 (0.5) CTL ↑ 0.1 (0.2) | ∆ Triceps skin fold | ∆ VO2 peak | In spite of self-reported increase in regular physical activity, there were no significant changes in any outcome measures in either group |
| ET ↑ 0.3 (1.3) | ||||
| CTL ↓ 0.1 (1.0) | (mL/kg/min): | |||
| ∆ Arm muscle circ. (cm) | ET ↑ 1.1 (4.6) | |||
| ET ↑ 0.1 (0.4) | CTL ↑ 2.3 (11.9) | |||
| CTL ↓ 0.1 (0.2) | ||||
| PEDro Criteria | Selvadurai 2002 [19] | Santana-Sosa 2012 [20] | Santana-Sosa 2014 [21] | Hommerding 2015 [18] |
|---|---|---|---|---|
| 1—Eligibility criteria |  |  |  |  |
| 2—Random allocation |  |  |  |  |
| 3—Concealed allocation |  |  |  |  |
| 4—Baseline comparability |  |  |  |  |
| 5—Blind subjects |  |  |  |  |
| 6—Blind therapists |  |  |  |  |
| 7—Blind assessors |  |  |  |  |
| 8—Adequate follow-up |  |  |  |  |
| 9—Intention-to-treat analysis |  |  |  |  |
| 10—Between-group comparisons |  |  |  |  |
| 11—Points estimates and variability |  |  |  |  |
| Total score | 6 | 7 | 7 | 5 |
 indicates criteria was fulfilled;
indicates criteria was fulfilled;  indicates criteria was not met.
indicates criteria was not met.Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nicolson, W.B.; Bailey, J.; Alotaibi, N.Z.; Krick, S.; Lowman, J.D. Effects of Exercise on Nutritional Status in People with Cystic Fibrosis: A Systematic Review. Nutrients 2022, 14, 933. https://doi.org/10.3390/nu14050933
Nicolson WB, Bailey J, Alotaibi NZ, Krick S, Lowman JD. Effects of Exercise on Nutritional Status in People with Cystic Fibrosis: A Systematic Review. Nutrients. 2022; 14(5):933. https://doi.org/10.3390/nu14050933
Chicago/Turabian StyleNicolson, William B., Julianna Bailey, Najlaa Z. Alotaibi, Stefanie Krick, and John D. Lowman. 2022. "Effects of Exercise on Nutritional Status in People with Cystic Fibrosis: A Systematic Review" Nutrients 14, no. 5: 933. https://doi.org/10.3390/nu14050933
APA StyleNicolson, W. B., Bailey, J., Alotaibi, N. Z., Krick, S., & Lowman, J. D. (2022). Effects of Exercise on Nutritional Status in People with Cystic Fibrosis: A Systematic Review. Nutrients, 14(5), 933. https://doi.org/10.3390/nu14050933







