Vitamin D Supplementation and Sleep: A Systematic Review and Meta-Analysis of Intervention Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Review Design
2.2. Criteria for Study Inclusion
2.3. Search Strategy
2.4. Study Selection
2.5. Data Extraction
2.6. Quality Assessment
2.7. Data Synthesis
2.8. Quality of Reporting
3. Results
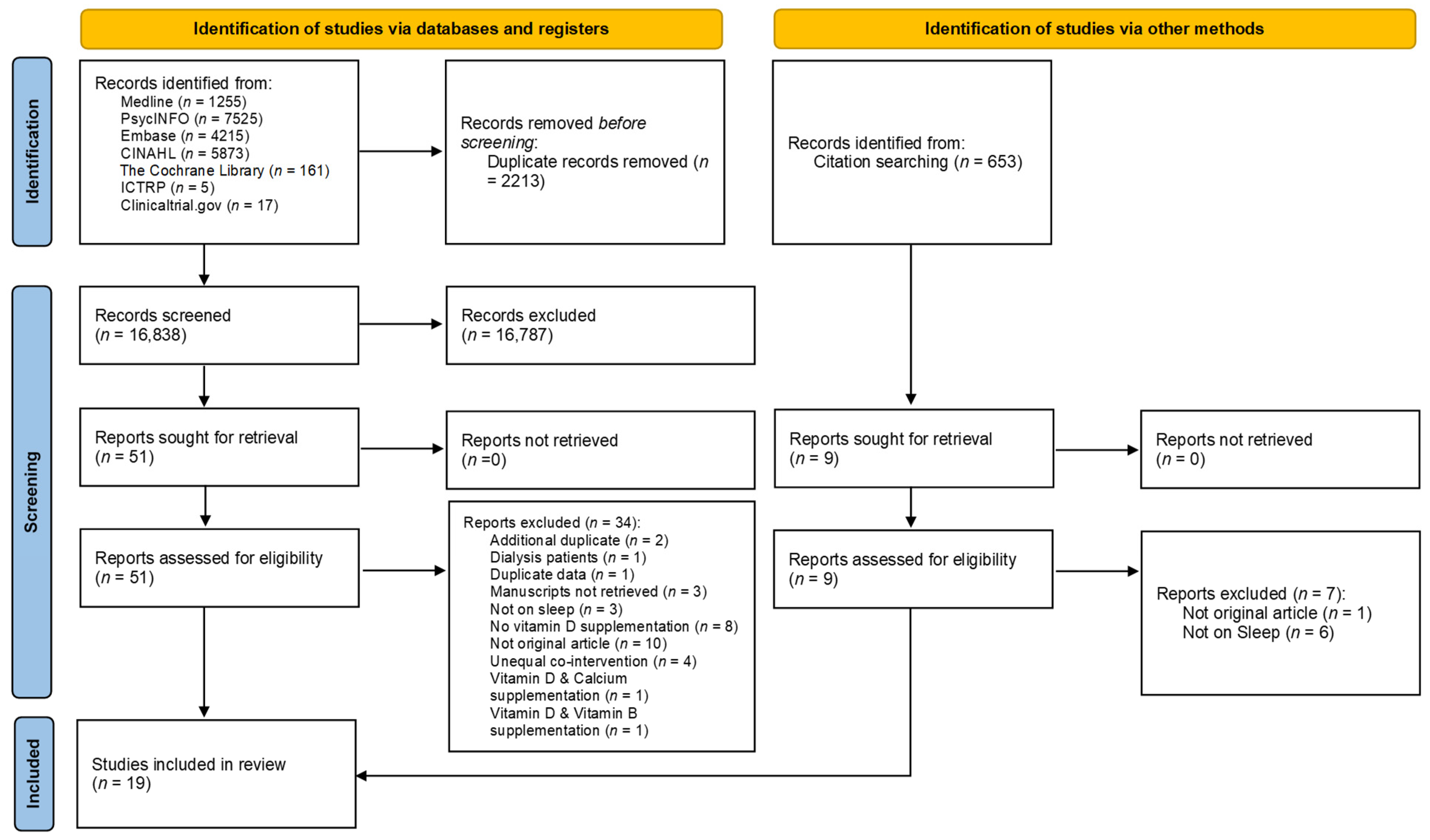
3.1. Search Results
3.2. Characteristics of Included Studies
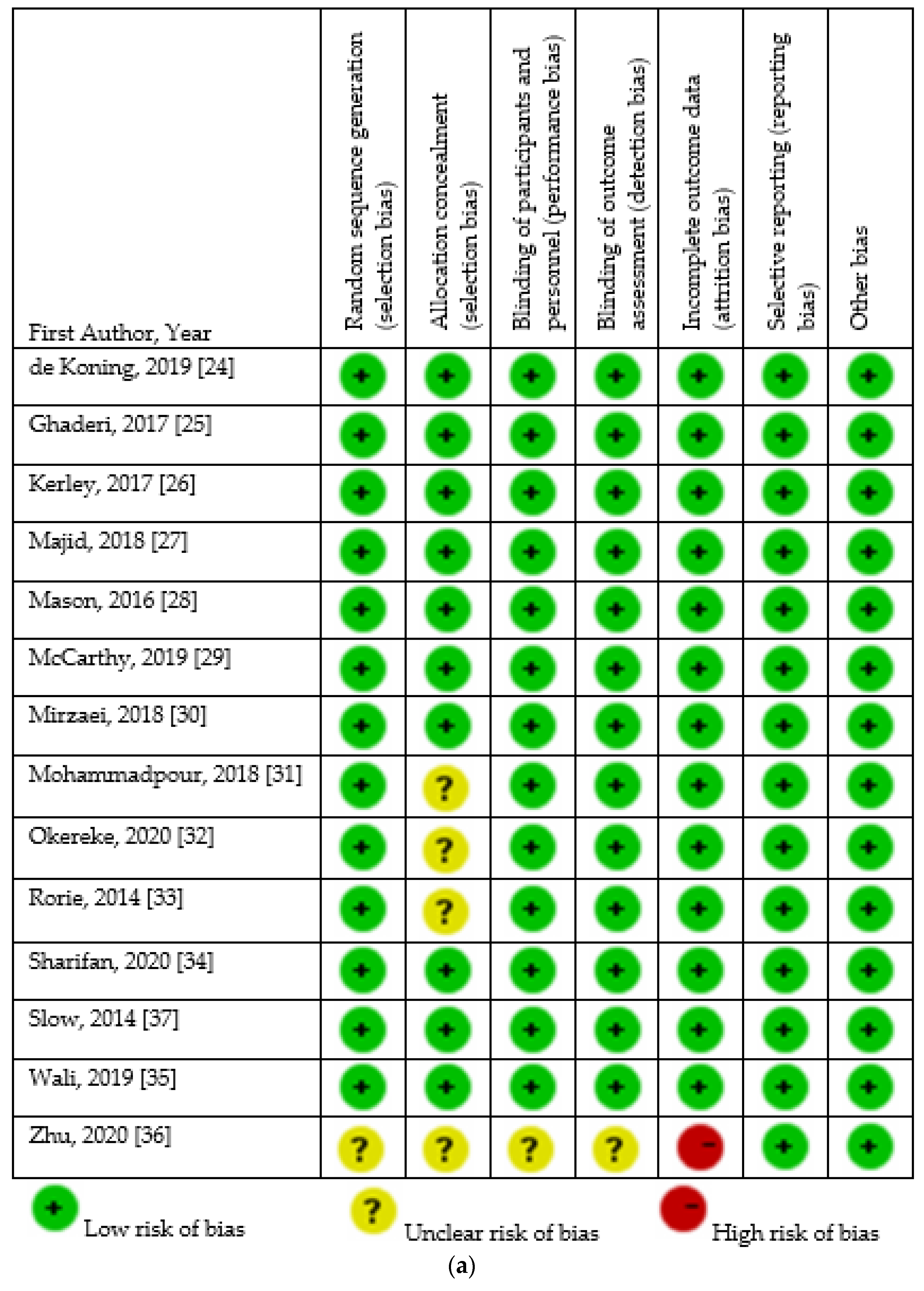
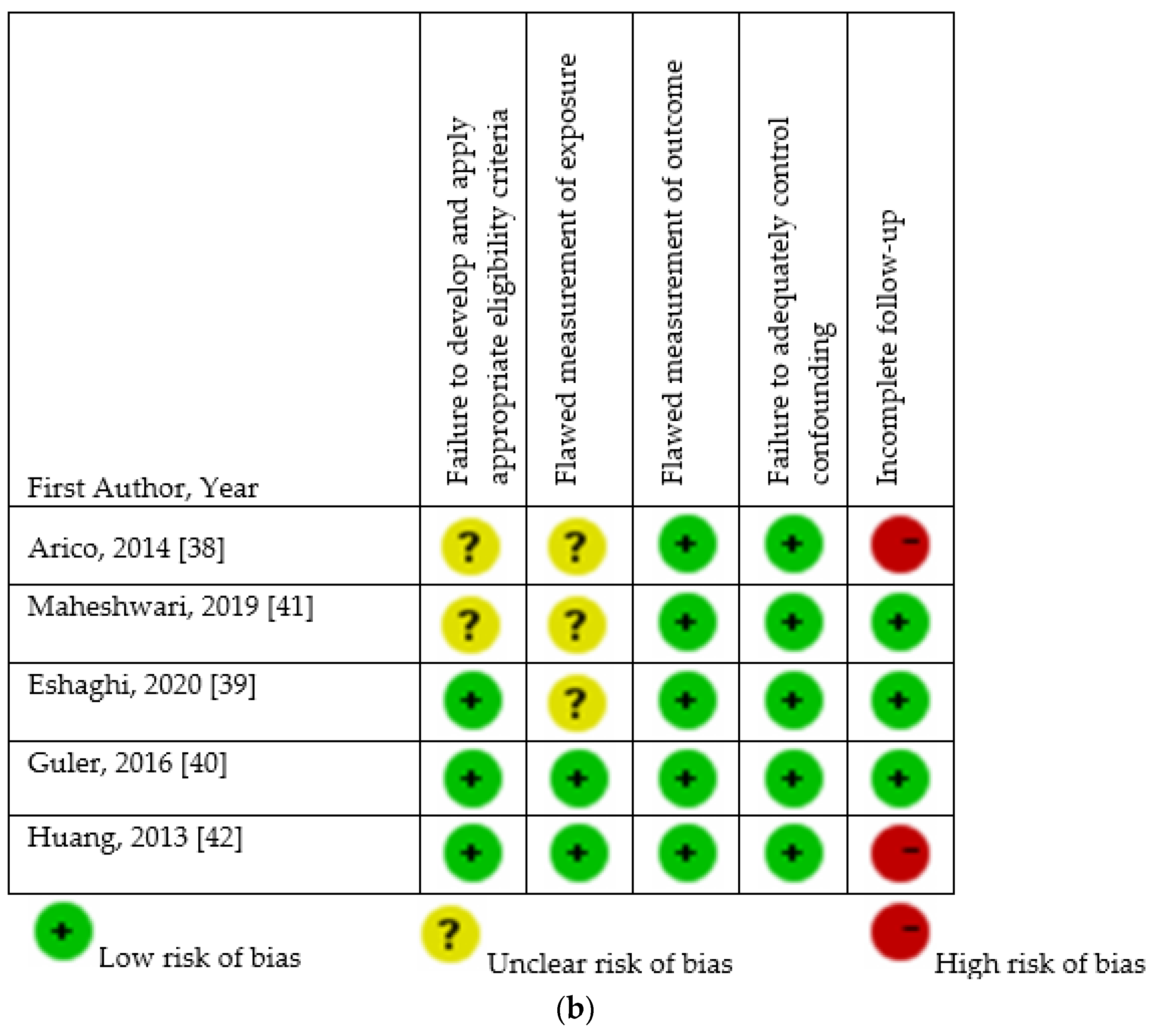
3.3. Assessment of Risk of Bias
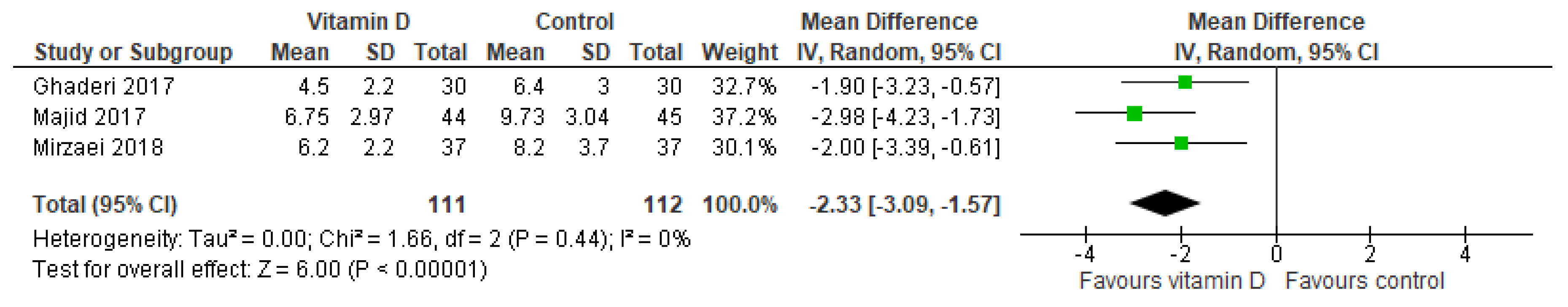
3.4. Sleep Quality
3.5. Other Outcomes
- Disturbed sleeping
- Sleepiness
- RLS
3.6. Sleep Problems as Adverse Events of VDS
4. Discussion
Strengths and Limitations
5. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Hillman, D.; Mitchell, S.; Streatfeild, J.; Burns, C.; Bruck, D.; Pezzullo, L. The economic cost of inadequate sleep. Sleep 2018, 41, zsy083. [Google Scholar] [CrossRef]
- Skaer, T.L.; Sclar, D.A. Economic implications of sleep disorders. Pharmacoeconomics 2010, 28, 1015–1023. [Google Scholar] [CrossRef] [PubMed]
- Sateia, M.J. International classification of sleep disorders-third edition: Highlights and modifications. Chest 2014, 146, 1387–1394. [Google Scholar] [CrossRef] [PubMed]
- Coren, S. The prevalence of self-reported sleep disturbances in young adults. Int. J. Neurosci. 1994, 79, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Luyster, F.S.; Strollo, P.J., Jr.; Zee, P.C.; Walsh, J.K. Sleep: A health imperative. Sleep 2012, 35, 727–734. [Google Scholar] [CrossRef] [PubMed]
- Irwin, M.R. Why sleep is important for health: A psychoneuroimmunology perspective. Annu. Rev. Psychol. 2015, 66, 143–172. [Google Scholar] [CrossRef]
- Chokroverty, S. Overview of sleep & sleep disorders. Indian J. Med. Res. 2010, 131, 126–140. [Google Scholar]
- Institute of Medicine, Committee on Sleep Medicine and Research. The National Academies Collection: Reports funded by National Institutes of Health. In Sleep Disorders and Sleep Deprivation: An Unmet Public Health Problem; Colten, H.R., Altevogt, B.M., Eds.; National Academy of Sciences: Washington, DC, USA, 2006. [Google Scholar]
- Liew, S.C.; Aung, T. Sleep deprivation and its association with diseases—A review. Sleep Med. 2021, 77, 192–204. [Google Scholar] [CrossRef]
- Andersen, M.L.; Tufik, S. Vitamin D as an underlying factor in sleep-related issues. J. Clin. Sleep Med. JCSM Off. Publ. Am. Acad. Sleep Med. 2012, 8, 699. [Google Scholar] [CrossRef]
- Anglin, R.E.; Samaan, Z.; Walter, S.D.; McDonald, S.D. Vitamin D deficiency and depression in adults: Systematic review and meta-analysis. Br. J. Psychiatry J. Ment. Sci. 2013, 202, 100–107. [Google Scholar] [CrossRef]
- Romano, F.; Muscogiuri, G.; Di Benedetto, E.; Zhukouskaya, V.V.; Barrea, L.; Savastano, S.; Colao, A.; Di Somma, C. Vitamin D and Sleep Regulation: Is there a Role for Vitamin D? Curr. Pharm. Des. 2020, 26, 2492–2496. [Google Scholar] [CrossRef] [PubMed]
- Muscogiuri, G.; Barrea, L.; Scannapieco, M.; Di Somma, C.; Scacchi, M.; Aimaretti, G.; Savastano, S.; Colao, A.; Marzullo, P. The lullaby of the sun: The role of vitamin D in sleep disturbance. Sleep Med. 2019, 54, 262–265. [Google Scholar] [CrossRef]
- Al-Shawwa, B.; Ehsan, Z.; Ingram, D.G. Vitamin D and sleep in children. J. Clin. Sleep Med. JCSM Off. Publ. Am. Acad. Sleep Med. 2020, 16, 1119–1123. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Kou, T.; Zhuang, B.; Ren, Y.; Dong, X.; Wang, Q. The Association between Vitamin D Deficiency and Sleep Disorders: A Systematic Review and Meta-Analysis. Nutrients 2018, 10, 1395. [Google Scholar] [CrossRef] [PubMed]
- Stumpf, W.E.; Bidmon, H.J.; Li, L.; Pilgrim, C.; Bartke, A.; Mayerhofer, A.; Heiss, C. Nuclear receptor sites for vitamin D-soltriol in midbrain and hindbrain of Siberian hamster (Phodopus sungorus) assessed by autoradiography. Histochemistry 1992, 98, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Stumpf, W.E.; O’Brien, L.P. 1,25 (OH)2 vitamin D3 sites of action in the brain. An autoradiographic study. Histochemistry 1987, 87, 393–406. [Google Scholar] [CrossRef]
- Patrick, R.P.; Ames, B.N. Vitamin D and the omega-3 fatty acids control serotonin synthesis and action, part 2: Relevance for ADHD, bipolar disorder, schizophrenia, and impulsive behavior. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2015, 29, 2207–2222. [Google Scholar] [CrossRef]
- Thorpy, M. International classification of sleep disorders. In Sleep Disorders Medicine; Springer: Berlin/Heidelberg, Germany, 2017; pp. 475–484. [Google Scholar]
- Higgins, J.P.T.; Savović, J.; Page, M.J.; Elbers, R.G.; Sterne, J.A.C. Chapter 8: Assessing risk of bias in a randomized trial. In Cochrane Handbook for Systematic Reviews of Interventions Version 6.0; Higgins, J.P.T., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M.J., Welch, V.A., Eds.; Cochrane: Chichester, UK, 2019. [Google Scholar]
- Guyatt, G.H.; Oxman, A.D.; Vist, G.; Kunz, R.; Brozek, J.; Alonso-Coello, P.; Montori, V.; Akl, E.A.; Djulbegovic, B.; Falck-Ytter, Y.; et al. GRADE guidelines: 4. Rating the quality of evidence--study limitations (risk of bias). J. Clin. Epidemiol. 2011, 64, 407–415. [Google Scholar] [CrossRef]
- Rethlefsen, M.L.; Kirtley, S.; Waffenschmidt, S.; Ayala, A.P.; Moher, D.; Page, M.J.; Koffel, J.B. PRISMA-S: An extension to the PRISMA Statement for Reporting Literature Searches in Systematic Reviews. Syst. Rev. 2021, 10, 39. [Google Scholar] [CrossRef]
- Page, M.J.; Moher, D.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. PRISMA 2020 explanation and elaboration: Updated guidance and exemplars for reporting systematic reviews. BMJ 2021, 372, n160. [Google Scholar] [CrossRef]
- De Koning, E.J.; Lips, P.; Penninx, B.; Elders, P.J.M.; Heijboer, A.C.; den Heijer, M.; Bet, P.M.; van Marwijk, H.W.J.; van Schoor, N.M. Vitamin D supplementation for the prevention of depression and poor physical function in older persons: The D-Vitaal study, a randomized clinical trial. Am. J. Clin. Nutr. 2019, 110, 1119–1130. [Google Scholar] [CrossRef] [PubMed]
- Ghaderi, A.; Banafshe, H.R.; Motmaen, M.; Rasouli-Azad, M.; Bahmani, F.; Asemi, Z. Clinical trial of the effects of vitamin D supplementation on psychological symptoms and metabolic profiles in maintenance methadone treatment patients. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2017, 79 Pt B, 84–89. [Google Scholar] [CrossRef]
- Kerley, C.P.; Hutchinson, K.; Bramham, J.; McGowan, A.; Faul, J.; Cormican, L. Vitamin D Improves Selected Metabolic Parameters but Not Neuropsychological or Quality of Life Indices in OSA: A Pilot Study. J. Clin. Sleep Med. JCSM Off. Publ. Am. Acad. Sleep Med. 2017, 13, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Majid, M.S.; Ahmad, H.S.; Bizhan, H.; Hosein, H.Z.M.; Mohammad, A. The effect of vitamin D supplement on the score and quality of sleep in 20–50 year-old people with sleep disorders compared with control group. Nutr. Neurosci. 2018, 21, 511–519. [Google Scholar] [CrossRef] [PubMed]
- Mason, C.; de Dieu Tapsoba, J.; Duggan, C.; Wang, C.Y.; Korde, L.; McTiernan, A. Repletion of vitamin D associated with deterioration of sleep quality among postmenopausal women. Prev. Med. 2016, 93, 166–170. [Google Scholar] [CrossRef]
- McCarthy, M.S.; Elshaw, E.B.; Szekely, B.M.; Raju, D. A Prospective Cohort Study of Vitamin D Supplementation in AD Soldiers: Preliminary Findings. Mil. Med. 2019, 184 (Suppl. S1), 498–505. [Google Scholar] [CrossRef]
- Mirzaei, A.; Zabihiyeganeh, M.; Jahed, S.A.; Khiabani, E.; Nojomi, M.; Ghaffari, S. Effects of vitamin D optimization on quality of life of patients with fibromyalgia: A randomized controlled trial. Med. J. Islamic Repub. Iran 2018, 32, 29. [Google Scholar] [CrossRef]
- Mohammadpour, N.; Jazayeri, S.; Tehrani-Doost, M.; Djalali, M.; Hosseini, M.; Effatpanah, M.; Davari-Ashtiani, R.; Karami, E. Effect of vitamin D supplementation as adjunctive therapy to methylphenidate on ADHD symptoms: A randomized, double blind, placebo-controlled trial. Nutr. Neurosci. 2018, 21, 202–209. [Google Scholar] [CrossRef]
- Okereke, O.I.; Reynolds, C.F., 3rd; Mischoulon, D.; Chang, G.; Vyas, C.M.; Cook, N.R.; Weinberg, A.; Bubes, V.; Copeland, T.; Friedenberg, G.; et al. Effect of Long-term Vitamin D3 Supplementation vs Placebo on Risk of Depression or Clinically Relevant Depressive Symptoms and on Change in Mood Scores: A Randomized Clinical Trial. JAMA 2020, 324, 471–480. [Google Scholar] [CrossRef]
- Rorie, A.; Goldner, W.S.; Lyden, E.; Poole, J.A. Beneficial role for supplemental vitamin D3 treatment in chronic urticaria: A randomized study. Ann. Allergy Asthma Immunol. Off. Publ. Am. Coll. Allergy Asthma Immunol. 2014, 112, 376–382. [Google Scholar] [CrossRef]
- Sharifan, P.; Khoshakhlagh, M.; Khorasanchi, Z.; Darroudi, S.; Rezaie, M.; Safarian, M.; Vatanparast, H.; Afshari, A.; Ferns, G.; Ghazizadeh, H.; et al. Efficacy of low-fat milk and yogurt fortified with encapsulated vitamin D3 on improvement in symptoms of insomnia and quality of life: Evidence from the SUVINA trial. Food Sci. Nutr. 2020, 8, 4484–4490. [Google Scholar] [CrossRef] [PubMed]
- Wali, S.O.; Abaalkhail, B.; Alhejaili, F.; Pandi-Perumal, S.R. Efficacy of vitamin D replacement therapy in restless legs syndrome: A randomized control trial. Sleep Breath. Schlaf Atm. 2019, 23, 595–601. [Google Scholar] [CrossRef]
- Zhu, C.; Zhang, Y.; Wang, T.; Lin, Y.; Yu, J.; Xia, Q.; Zhu, P.; Zhu, D.M. Vitamin D supplementation improves anxiety but not depression symptoms in patients with vitamin D deficiency. Brain Behav. 2020, 10, e01760. [Google Scholar] [CrossRef] [PubMed]
- Slow, S.; Florkowski, C.M.; Chambers, S.T.; Priest, P.C.; Stewart, A.W.; Jennings, L.C.; Livesey, J.H.; Camargo, C.A., Jr.; Scragg, R.; Murdoch, D.R. Effect of monthly vitamin D3 supplementation in healthy adults on adverse effects of earthquakes: Randomised controlled trial. BMJ 2014, 349, g7260. [Google Scholar] [CrossRef] [PubMed]
- Aricò, I.; Campolo, L.; Silvestri, R. Preliminary Data on Vitamin D Deficiency and Treatment in a Cohort of Sicilian RLS/WED Patients. Neuropsychiatr. Dis. Treat 2014, 10, 953–958. [Google Scholar]
- Eshaghi, S.; Morteza, T.; Khadijeh, I.; Knechtle, B.; Nikolaidis, P.T.; Chtourou, H. The effect of aerobic training and vitamin D supplements on the neurocognitive functions of elderly women with sleep disorders. Biol. Rhythm. Res. 2020, 51, 727–734. [Google Scholar] [CrossRef]
- Guler, S.; Yeşil, G.; Önal, H.; Ekici, B.; Ozdil, M. Sleep disturbances and serum vitamin D levels in children with autism spectrum disorder. Int. J. Clin. Exp. Med. 2016, 9, 14691–14697. [Google Scholar]
- Maheshwari, P.; Arun, S.; Venkatesh, N.; Sushmi, K.; Shanmugarajan, T.; Shanmugasundaram, P. Effect of Vitamin D supplement in improving pain, sleep, and quality of life on patients with chronic low back pain. Drug Invent. Today 2019, 12, 2508–2510. [Google Scholar]
- Huang, W.; Shah, S.; Long, Q.; Crankshaw, A.K.; Tangpricha, V. Improvement of pain, sleep, and quality of life in chronic pain patients with vitamin D supplementation. Clin. J. Pain 2013, 29, 341–347. [Google Scholar] [CrossRef]
- Yan, S.; Tian, Z.; Zhao, H.; Wang, C.; Pan, Y.; Yao, N.; Guo, Y.; Wang, H.; Li, B.; Cui, W. A meta-analysis: Does vitamin D play a promising role in sleep disorders? Food Sci. Nutr. 2020, 8, 5696–5709. [Google Scholar] [CrossRef]
- Eyles, D.W.; Smith, S.; Kinobe, R.; Hewison, M.; McGrath, J.J. Distribution of the vitamin D receptor and 1 alpha-hydroxylase in human brain. J. Chem. Neuroanat. 2005, 29, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Garcion, E.; Wion-Barbot, N.; Montero-Menei, C.N.; Berger, F.; Wion, D. New clues about vitamin D functions in the nervous system. Trends Endocrinol. Metab. TEM 2002, 13, 100–105. [Google Scholar] [CrossRef]
- Lucock, M.; Jones, P.; Martin, C.; Beckett, E.; Yates, Z.; Furst, J.; Veysey, M. Vitamin D: Beyond Metabolism. J. Evid. Based Complement. Altern. Med. 2015, 20, 310–322. [Google Scholar] [CrossRef] [PubMed]
- Vitaterna, M.H.; Takahashi, J.S.; Turek, F.W. Overview of circadian rhythms. Alcohol Res. Health J. Natl. Inst. Alcohol Abus. Alcohol. 2001, 25, 85–93. [Google Scholar]
- Dibner, C.; Schibler, U.; Albrecht, U. The mammalian circadian timing system: Organization and coordination of central and peripheral clocks. Annu. Rev. Physiol. 2010, 72, 517–549. [Google Scholar] [CrossRef]
- Jamilian, H.; Amirani, E.; Milajerdi, A.; Kolahdooz, F.; Mirzaei, H.; Zaroudi, M.; Ghaderi, A.; Asemi, Z. The effects of vitamin D supplementation on mental health, and biomarkers of inflammation and oxidative stress in patients with psychiatric disorders: A systematic review and meta-analysis of randomized controlled trials. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2019, 94, 109651. [Google Scholar] [CrossRef]
- Zhao, D.; Yu, Y.; Shen, Y.; Liu, Q.; Zhao, Z.; Sharma, R.; Reiter, R.J. Melatonin Synthesis and Function: Evolutionary History in Animals and Plants. Front. Endocrinol. 2019, 10, 249. [Google Scholar] [CrossRef]
- Kaneko, I.; Sabir, M.S.; Dussik, C.M.; Whitfield, G.K.; Karrys, A.; Hsieh, J.C.; Haussler, M.R.; Meyer, M.B.; Pike, J.W.; Jurutka, P.W. 1,25-Dihydroxyvitamin D regulates expression of the tryptophan hydroxylase 2 and leptin genes: Implication for behavioral influences of vitamin D. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2015, 29, 4023–4035. [Google Scholar] [CrossRef]
- Jablonski, K.L.; Chonchol, M.; Pierce, G.L.; Walker, A.E.; Seals, D.R. 25-Hydroxyvitamin D deficiency is associated with inflammation-linked vascular endothelial dysfunction in middle-aged and older adults. Hypertension 2011, 57, 63–69. [Google Scholar] [CrossRef]
- Krueger, J.M.; Majde, J.A.; Rector, D.M. Cytokines in immune function and sleep regulation. Handb. Clin. Neurol. 2011, 98, 229–240. [Google Scholar]
- Archontogeorgis, K.; Nena, E.; Papanas, N.; Steiropoulos, P. The role of vitamin D in obstructive sleep apnoea syndrome. Breathe 2018, 14, 206–215. [Google Scholar] [CrossRef]
- Chao, Y.S.; Brunel, L.; Faris, P.; Veugelers, P.J. The importance of dose, frequency and duration of vitamin D supplementation for plasma 25-hydroxyvitamin D. Nutrients 2013, 5, 4067–4078. [Google Scholar] [CrossRef]
- Chakhtoura, M.; Chamoun, N.; Rahme, M.; Fuleihan, G.E. Impact of vitamin D supplementation on falls and fractures-A critical appraisal of the quality of the evidence and an overview of the available guidelines. Bone 2020, 131, 115112. [Google Scholar] [CrossRef] [PubMed]
- Caprio, M.; Infante, M.; Calanchini, M.; Mammi, C.; Fabbri, A. Vitamin D: Not just the bone. Evidence for beneficial pleiotropic extraskeletal effects. Eat. Weight Disord. EWD 2017, 22, 27–41. [Google Scholar] [CrossRef] [PubMed]
- Egierska, D.; Pietruszka, P.; Burzyńska, P.; Chruścicka, I.; Buchta, J. Pleiotropic effects of vitamin D3. J. Educ. Health Sport 2021, 11, 143–155. [Google Scholar] [CrossRef]
| First Author, Country | Study Design | Study Population | Age %Male | Control | Co-Intervention | Intervention Duration Daily Dose Equivalent | Compliance | Vitamin D Assessment |
|---|---|---|---|---|---|---|---|---|
| Randomized, controlled trial | ||||||||
| Sleep quality | ||||||||
| Ghaderi [25], Iran | Randomized, double-blind, placebo-controlled trial | 68 patients with maintenance methadone treatment referred to a clinic (30 completed the trial: I: n = 30; C: n = 30) | Age: Range: 25–70 Mean ± SD: I: 40.1 ± 9.2 C: 42.5 ± 8.9 %Male: NR | Placebo, capsule | None | Vitamin D (unclear): capsules 12 weeks 3571.42 IU | >90% in both groups | 25(OH)D: commercial ELISA kit |
| Maheshwari [41], NR | Pre–post study | 40 patients diagnosed with chronic low back pain | Age: Range: 25–65 %Male: 60% | Self | None | Vitamin D (unclear) 8 weeks NR | NR | NR |
| Majid [27], Iran | Randomized, double-blind, placebo-controlled trial | 93 people with sleep disorders (PSQI ≥ 5), but without sleep disorders, not using sleep medications, recruited from a hospital (89 completed the trial: I: n = 44; C: n = 45) | Age: Range: 20–50 Mean ± SD: I: 37.9 ± 9.50 C: 35.5± 10.00 %Male: I: 25.00; C: 22.22 | Placebo, capsule | None | Vitamin D3: edible pearls 8 weeks 3571.42 IU | I: 97.82% C: 97.87% | 25(OH) D: Immunodiagnostic Systems kit |
| Mason [28], USA | Randomized, double-blind, placebo-controlled trial | 218 overweight (BMI ≥25 kg/m2) postmenopausal women, with serum 25(OH)D ≥10 to <32 ng/mL undergoing weight loss I: n = 109; C: n = 109 | Age: Range: 50–75 Mean ± SD: 59.6 ± 5.1 %Male: 0% | Placebo, gel capsules | Lifestyle-based weight-loss program including a reduced-calorie diet (1200–2000 kcal/day, <30% daily energy intake from fat) and 225 min/week of moderate-to-vigorous aerobic activity | Vitamin D3, gel capsules 12 months 2000 IU | I: 98% C: 96% | 25(OH)D: DiaSorin LIAISON 25-OH Vitamin D total assay |
| Mirzaei [30], Iran | Randomized, double-blind, placebo-controlled trial | 74 fibromyalgia syndrome patients (according to the American College of Rheumatology criteria) with 25(OH)D < 30 ng/mL, recruited from a rheumatology center | Age: Range: 20–70 Mean ± SD: Total: 41.6 ± 10.5 I: 42.1 ± 10.8 C: 41 ± 10.3 %Male: 0% | Placebo | Trazodone 25 mg at bedtime | Vitamin D (unclear) 8 weeks 7142.85 IU | NR | 25(OH)D: commercial radioimmunoassay kit |
| Other outcomes | ||||||||
| McCarthy [29], Pacific Northwest | Randomized, double-blind, controlled trial | 152 active-duty warfighters, divided into no-treatment group (C) (≥ 30 ng/mL) or treatment group (I) (<30 ng/mL) (131 completed the trial) | Age: Mean ± SD: I: 31.6 ± 8.2 C: 32.8 ± 10.5 %Male: 61.8% | No supplementation | None | Vitamin D3, gel capsules 12 weeks I1: 1000 IU I2: 5000 IU | I1: 84.8% I2: 87.9% C: not required to take pills | 25(OH)D: electrochemiluminescence |
| Okereke [32], USA | Randomized, double-blind, placebo-controlled trial | Participants aged ≥50 years in the VITAL-DEP (Vitamin D and Omega-3 Trial-Depression Endpoint Prevention) ancillary study to VITAL, a randomized clinical trial of cardiovascular disease and cancer prevention, without clinically relevant depressive symptoms at baseline I: n = 9181; C: n = 9172 | Age: Mean ± SD: Total: 67.5 ± 7.1 I: 67.5 ± 7.0 C: 67.4 ± 7.1 %Male: Total: 51% I: 50.6%; C: 51.1% | Placebo | None | Vitamin D3, pills Median follow-up: 5.3 years 2000 IU | Adherence rates (taking at least two-thirds of pills as assigned) I: Year 1: 94.8% Year 2: 92.2% Year 3: 91.5% Year 4: 91.4% Year 5: 90.6% C: Year 1: 94.6% Year 2: 92.1% Year 3: 91.0% Year 4: 90.5% Year 5: 89.8% | 25(OH)D: radioimmunoassay |
| Rorie [33], USA | Randomized, double-blind, controlled trial | 42 adult patients with physician-diagnosed chronic urticaria, recruited from tertiary care clinics I: n = 21; C: n = 21 | Age: I: Mean: 43.9; Range: 20–72 C: Mean: 43.1; Range: 19–79 %Male: I: 14.2%; C: 28.5% | Low-dose vitamin D3 (600 IU/d) | 10 mg of cetirizine twice daily and increase to 4 times daily as needed, 150 mg of ranitidine twice daily, and 10 mg of montelukast daily. In addition to rescue prednisone use for intolerable or uncontrolled symptoms | Vitamin D3, pills (high dose) 12 weeks 4000 IU | Excellent compliance (1 subject in the low vitamin D3 group showed <80% compliance) | 25(OH)D: Tandem mass spectroscopy |
| Zhu [36], China | Randomized, placebo-controlled trial | 158 patients with with 25(OH)D ≤ 75 nmol/L and depression with tied anxiety symptoms recruited through advertisements, assessed by the Mini-International Neuropsychiatric Interview to confirm the diagnosis of major depressive disorders (106 completed the trial: I: n = 62; C: n = 44) | Age: Mean ± SD I: 46.3 ± 9.7 C: 43.3 ± 13.7 %Male: I: 29%; C: 22.7% | Placebo | Fish oil (current intake) | Vitamin D (unclear), per os 6 months 1600 IU | NR | 25(OH)D: commercial radioimmunoassay kit |
| Kerley [26], Ireland | Randomized, double-blind, placebo-controlled trial | 26 Caucasian adults with OSA recruited from a sleep clinic (19 completed the trial: 15 on CPAP therapy and 4 CPAP naïve: I: n = 10; C: n = 9) | Age: I: Range: 42–71 Mean ± SD: 56 ± 10 C: Range: 32–68 Mean ± SD: 52 ± 13 %Male: I: 89%; C: 60% | Placebo, capsule | None | Vitamin D3: Capsule 15 weeks 4000 IU | 93% in both groups | Total 25(OH)D: Architect 25(OH)D CMIA |
| Sharifan [34], Iran | Randomized, triple-blind, placebo-controlled trial | 29 staff and students of a university, aged 30–50 years, with vitamin D deficiency (<30 ng/mL), abdominal obesity (waist circumference (≥94 cm for men and ≥80 cm for women), and symptoms of insomnia (according to validated Insomnia Severity Index) Milk: I: n = 8; C: n = 8 Yogurt: I: n = 7; C: n = 6 | Age: Mean ± SD: 43.2 ± 6.59 Milk group: I: 39.5 ± 6.23; C: 44.5 ± 5.63 Yogurt group: I: 47.42 ± 6.8; C: 41.5 ± 5.99 %Male: Milk group: I: 20%; C: 80% Yogurt group: I: 57.1%; C: 42.8% | Simple low-fat milk (200 mL/day) Simple low-fat yogurt (150 g/day) | None | I1: Vitamin D3-fortified low-fat milk containing 1500 IU Nan I2: Vitamin D3-fortified low-fat yogurt 10 weeks 1500 IU | NR | 25(OH)D: commercial ELISA kits |
| Wali [35], KSA | Randomized, double-blind, placebo-controlled trial | 35 patients with primary RLS identified based on the RLS diagnostic criteria of the IRLSSG and recruited from the Sleep Medicine and Research Center I: n = 17; C: n = 18 | Age: Mean ± SD I: 42.7 ± 4.7 C: 42.4 ± 5.5 %Male: I: 64.7%; C: 72.2% | Placebo | None | Vitamin D3, per os 12 weeks 7142.85 IU | 100% | NR |
| Sleep problems as adverse events of VDS | ||||||||
| de Koning [24], the Netherlands | Randomized, double-blind, placebo-controlled trial | 155 community-dwelling older people, aged 60–80 years, recruited from the general population or through general practitioners, with depressive symptoms, and serum 25(OH)D between 15 and 50 nmol/L during October–March or between 15 and 70 nmol/L during April–September I: n = 77; C: n = 78 | Age: Median [IQR] I: 67.8 [65.4–71.7] C: 67.3 [63.4–72.0] %Male: I: 41.6%; C: 43.6% | Placebo | Calcium tablet of 500 mg/day in case of <2 dairy consumptions/day Participants were allowed to take a (multi)VDS with a maximum of 400 IU/day in addition to the study tablets | Vitamin D3, tablet 12 months 1200 IU | 87.10% | 25(OH)D: liquid chromatography followed by tandem mass spectrometry |
| Mohammadpour [31], Iran | Randomized, double-blind, placebo-controlled trial | 62 children with ADHD (based on DSM-IV criteria), aged 5–12 years, referred from psychiatric centers, without psychiatric nor neurologic comorbidities (54 completed the trial: I: n = 25; C: n = 29) | Age: Mean ± SD: Total: 7.87 ± 1.61 I: 7.70 ± 1.77 C: 8.03 ± 1.44 %Male: I: 71%; C: 77.4% | Placebo | Methylphenidate | Vitamin D (unclear), tablet 8 weeks 2000 IU | 100% | 25(OH)D3: chemiluminescence |
| Opportunistic addition to a randomized, controlled trial | ||||||||
| Other outcomes | ||||||||
| Slow [37], New Zealand | Opportunistic addition to an established randomized, double-blind, placebo-controlled trial | 322 healthy adults already participating in the vitamin D and acute respiratory infections study (VIDARIS) staff or students recruited from a University. (308 completed the trial: I: n = 147; C: n = 146) | Age: Range: 18–67 Mean ± SD: I: 47 ± 10 C: 48 ± 10 %Male: I: 25%; C: 25% | Placebo | None | Vitamin D3, per os 18 months 6557.37 for 2 months, then 3278.68 IU | NR | NR |
| Pre–post study | ||||||||
| Sleep quality | ||||||||
| Eshaghi [39], Iran | Pre–post study | 42 elderly women referred to a sports counseling center, with a PSQI > 11, without sleep apnea, not smoking, and not taking hypnotic drugs (36 completed the trial) | Age: Range: 60–70 %Male: 0% | No supplementation (habitual daily activities) | Vitamin D (unclear) 8 weeks 1000 IU | NR | NR | |
| Other outcomes | ||||||||
| Guler [40], Turkey | Pre–post study | Cases: 60 patients with ASD according to DSM V criteria, aged between 4 and 10 years Controls: 60 age- and sex-matched apparently healthy children | Age: Mean ± SD: Cases: 7.10 ± 1.50 Controls: 6.93 ± 1.59 %Male: Cases: 73.3%; Controls: 65% | Vitamin D2 3 months Vitamin D according to deficiency level: I1: Participants with 25(OH)D: 20–29 ng/mL: 5000 IU I2: Participants with 25(OH)D < 20 ng/mL: 7142.86 IU | NR | 25(OH)D: radioimmunoassay using commercial kits | ||
| Arico [38], Italy | Pre–post study | 5 patients with RLS recruited from a sleep center | Age: NR %Male: 0% | Self | None | Vitamin D3 (unclear) 6 months NR | NR | NR |
| Pre–post study, analyzed retrospectively as a case series | ||||||||
| Sleep quality | ||||||||
| Huang [42], USA | Pre–post study, analyzed retrospectively as a case series by medical record review | 46 veterans with multiple areas of chronic pain and low serum 25(OH)D (<30 ng/mL) at baseline recruited from a major Veterans Affairs Medical Center, divided into vitamin D (1) INS: 25(OH)D: 20–29 ng/mL; and (2) DEF: 25(OH)D: <20 ng/mL (28 completed the trial: INS: n = 15; DEF: n = 13) | Age: Mean ± SD: 46.2 ± 10.8 %Male: 64.3% | Self | None | INS: Vitamin D3: per os DEF: Vitamin D2: per os 12 weeks INS: 1200 IU DEF: 7142.85 IU | NR | 25(OH)D: liquid chromatography–mass spectrometry assay |
| First Author, Country | Outcomes Evaluated and Assessment | Baseline 25OHD Level | Endline 25OHD Level | Baseline Outcomes | Endline Outcomes | Conclusion |
|---|---|---|---|---|---|---|
| Randomized, controlled trial | ||||||
| Sleep quality | ||||||
| Ghaderi [25], Iran | Sleep quality: PSQI | I: 13.9 ± 4.5 C: 13.5 ± 4.5 (NS difference between I and C) | I: 22.0 ± 7.5 (sig. increase) C: 13.1 ± 5.9 | I: 6.0 ± 2.3 C: 6.6 ± 2.2 | I: 4.5 ± 2.2 (sig. decrease) C: 6.4 ± 3.0 | PSQI sig. decreased in VDS I group compared with C group (−1.5 ± 2.2 vs. −0.2 ± 2.3) |
| Maheshwari [41], NR | Sleep quality: PSQI | NR | NR | NR | t-test: 2.965; CI: 1.8312–6.8341; p = 0.004 (sig. differences before and after VDS) | VDS improves sleep in patients with chronic low back pain |
| Majid [27], Iran | Sleep quality: PSQI Sleep duration Sleep latency Sleep efficiency: real sleep duration from the whole time passed in bed Sleep disturbances Use of sleep medications Daytime dysfunction: experiencing problems resulted by sleeplessness Subjective sleep quality | I: 25.00 ± 8.95 C: 27.60 ± 8.30 (NS difference between I and C) | I: 37.69 ± 12.25 C: 27.97 ± 7.46 (sig. increase in I, and sig. difference between I and C) | PSQI (score) (NS difference between I and C) I: 9.45 ± 2.44 C: 10.51 ± 3.14 Sleep duration (hour) (sig. higher in I compared with C) I: 5.83 ± 1.15 C: 5.22 ± 1.54 Sleep latency (minute) (NS difference between I and C) I: 49.88 ± 38.99 C: 65.00 ± 47.54 Sleep efficiency (%) (NS difference between I and C) I: 82.58 ± 9.93 C: 78.20 ± 12.90 Sleep disturbances (score) (NS difference between I and C) I: 1.23 ± 0.47 C: 1.40 ± 0.78 Use of sleep medications (time per week) (NS difference between I and C) I: 2.07 ± 1.92 C: 0.77 ± 1.02 Day time dysfunction (score) (NS difference between I and C) I: 1.57 ± 0.99 C: 1.17 ± 0.93 Subjective sleep quality (score) (NS difference between I and C) I: 1.68 ± 0.77 C: 1.57 ± 0.62 | PSQI (score) (sig. lower in I compared with C) I: 6.75 ± 2.97 (sig. decrease) C: 9.73 ± 3.04 Sleep duration (hour) (sig. higher in I compared with C) I: 6.50 ± 1.49 C: 5.21 ± 1.44 Sleep latency (minute) (sig. lower in I compared with C) I: 33.18 ± 27.91 C: 58.57 ± 36.81 Sleep efficiency (%) (NS difference between I and C) I: 86.97 ± 11.39 (sig. decrease) C: 80.89 ± 11.46 Sleep disturbances (score) (NS difference between I and C) I: 1.14 ± 0.46 (NS) C: 1.41 ± 0.65 (NS) Use of sleep medications (time per week) (NS difference between I and C) I: 1.07 ± 0.94 (sig. decrease) C: 1.20 ± 0.99 Day time dysfunction (score) (NS difference between I and C) I: 0.70 ± 0.96 (sig. decrease) C: 0.75 ± 0.98 Subjective sleep quality (score) (sig. lower in I compared with C) I: 1.18 ± 0.62 (sig. decrease) C: 1.46 ± 0.58 | Reduced PSQI (improved sleep score), reduced sleep latency, increased sleep duration, and subjective sleep quality with VDS. NS difference in sleep efficiency, sleep disturbances, and use of sleep medications |
| Mason [28], USA | Sleep quality: PSQI | NR | NR | NR | NR | NS change in overall sleep quality between VDS I and C groups A greater magnitude of change in serum 25(OH)D was associated with an increased need to take medications to sleep and overall worse sleep quality Deterioration in total PSQI among women who became vitamin D replete (≥32 ng/mL) compared with those who remained <32 ng/mL (despite VDS) VDS of 2000 IU/d may result in overall worse sleep quality for postmenopausal women with low circulating vitamin D undergoing weight loss |
| Mirzaei [30], Iran | Sleep quality: PSQI | I: 11.4 ± 6.7 C: 13.4 ± 7.3 | I: 33.5 ± 12.2 (sig. higher in I compared with C) C: 13.3 ± 7.2 | I: 10 ± 3.3 C: 10.75 ± 4.4 | I: 6.2 ± 2.2 C: 8.2 ± 3.7 (sig. lower in I compared with C) | Considerable improvements were observed in the PSQI score of the both study groups; yet there was a sig. greater decrease in mean PSQI score in the I compared with C group |
| Other outcomes | ||||||
| McCarthy [29], Pacific Northwest | Sleep-related impairment: Questions from the National Institutes of Health Patient-Reported Outcomes Measurement Information System | I1: 22.2 ± 5.0 I2: 22.9 ± 4.7 C: 37.8 ± 5.6 | I1: 30.80 ± 10.0 I2: 40.15 ± 7.5 (sig. higher in I2 compared with I1 and C) C: 34.46 ± 9.9 | I1: 53.0 ± 7.0 I2: 48.3 ± 9.5 C: 51.5 ± 7.0 | I1: 49.5 ± 9.5 I2: 45.2 ± 8.4 C: 49.3 ± 8.2 | Statistically significant improvements seen across groups and over time |
| Okereke [32], USA | Sleep difficulty (sleep problems) as specific depressive feature (item-level symptom) from the 8 item Patient Health Questionnaire depression scale: Trouble falling or staying asleep, or sleeping too much | 25(OH)D < 20 ng/mL I: 11.0% C: 12.3% Mean ± SD I: 31.2 ± 9.8 C: 31.1 ± 10.0 | NR | NR | NR | NS differences in likelihood of sleep problems in the I compared with C group Adjusted differences in change in likelihood of PHQ-8 item-level symptoms, comparing vitamin D3 to Placebo: Sleep problems: Likelihood ratio: 95% CI: 1.00 (0.89–1.12) Analyses were from repeated measures logistic regression models, with follow-up time modeled as an indicator; models were controlled for age, sex, and n-3 fatty acid randomization group. Results show likelihood ratios and 95% confidence intervals (95% CIs), which reflect differences in the change in likelihood of burden from each PHQ-8 item-level symptom, comparing vitamin D3 to placebo treatment group. Differences reflect the average effect over all follow-up times since baseline |
| Rorie [33], USA | Nights of hives and sleep interference: from the Urticaria Symptom Severity scores | Mean(SE) C: 37.1(3.4) I: 28.8(2.2) | Mean(SE) C: 35.8(2.3) I: 56.0(3.9) (sig. higher in I compared with C) | NR | NR | Beneficial trends for sleep quality and towards decreased interference with sleep were observed with high vitamin D3 |
| Zhu [36], China | Sleep disorder: NR | I: 15.66 ± 4.20 C: 16.86 ± 5.04 (NS difference between I and C group) | NR | NR | NR | Between-group linear mixed-model analysis showed sig. decrease in Sleep disorder (β: −0.588; 95% CI: −1.061,−0.115), that was rendered NS after controlling for confounding variables (β: −0.355; 95% CI: −0.963,0.227) |
| Kerley [26], Ireland | Sleepiness: ESS | I: 13.38 ± 4.64 C: 16.58 ± 8.81 (NS difference between I and C) | I: 40.38 ± 15.98 (sig. increase) C: 17.22 ± 8.57 | I: 11.00 ± 5.00 C: 10.00 ± 6.00 (NS difference between I and C) | I: 6.00 ± 2.00 C: 7.00 ± 5.00 (NS difference between I and C) | No difference in ESS between the VDS I group and C group |
| Sharifan [34], Iran | Changes in sleepiness symptoms: Insomnia Severity Index | Milk I: 15.03 ± 3.91 C: 14.9 ± 7.34 (NS difference between I and C group) Yogurt I: 15.82 ± 4.09 C: 16.72 ± 2.96 (NS difference between I and C group) | Milk: I: 18.57 (sig. increase compared with baseline) C: 14.66 Yogurt: I: 19.93 (sig. increase compared with baseline) C: 16.26 (SD not reported) | Milk: I: 18.5 ± 3.33 C: 17.25 ± 2.34 Yogurt: I: 13.28 ± 5.12 C: 13 ± 3.54 | Milk I: 13.62 ± 3.29 (sig. increase compared with baseline) C: 16.5 ± 4.02 (NS difference compared with baseline) Yogurt I: 17.57 ± 13.28 (NS difference compared with baseline) C: 16.66 ± 1.36 (NS difference compared with baseline) | Fortified low-fat milk containing 1500 IU vitamin D3 can improve insomnia symptoms |
| Wali [35], KSA | RLS severity: IRLSSG rating scale | I: 17.06 ± 12.6 C: 22.95 ± 16.98 (NS difference between I and C group) | I: 6.09 ± 15.38 (sig. higher in I compared with C group) C: 21.23 ± 13.74 | Total I: 14.60 ± 4.5 C: 16.11 ± 6.2 In DEF patients I: 14.82 ± 5.2 C: 16.81 ± 6.3 | Total: I: 14.5 ± 08.2 (NS difference compared with baseline) C: 10.3 ± 11.1 (sig. decrease compared with baseline) In DEF patients I: 13.8 ± 8.9 (NS difference compared with baseline) C: 7.8 ± 13.9 (sig. decrease compared with baseline) | NS difference in RLS severity score with VDS suggesting that VDS does not improve RLS symptoms |
| Sleep problems as adverse events of VDS | ||||||
| de Koning [24], the Netherlands | Sleep problems as adverse event: registered by telephone or face-to-face contact | median [IQR] I: 18.42 [13.02–22.83] C: 17.68 [14.42–22.13] (significance NR) | mean ± SD I: 34.05 ± 6.41 C: 17.22 ± 7.21 (sig. difference between I and C) | NA (sleep problems as adverse event of VDS) | I: n = 1 C: n = 5 (NS difference between I and C) | NS difference in sleep problems as adverse effect of VDS in community-dwelling people with depressive symptoms |
| Mohammadpour [31], Iran | Sleep problems as adverse events: questionnaire | mean ± SD I: 15.792 ± 5.259 C: 12.979 ± 5.804 (NS difference between I and C) | mean ± SD: I: 34.63 ± 9.54 (sig. increase between baseline and endline) C: 11.22 ± 5.11 (NS difference between baseline and endline) Sig. difference between I and C | NA (sleep problems as adverse event of VDS) | Rate I: 4 C: 1 (NS difference between I and C) | NS difference in sleep problems as adverse effect of VDS as adjunctive therapy in children with ADHD |
| Opportunistic addition to a randomized, controlled trial | ||||||
| Other outcomes | ||||||
| Slow [37], New Zealand | Specific questionnaire about disruptions in sleeping patterns as a consequence of the earthquake on the 22nd February 2011 | I: 29.24 ± 8.81 C: 28.44 ± 8.81 | NR | NR | I: 79% C: 70% (NS difference between I and C) | VDS did not reduce the adverse impact of earthquakes in healthy adults |
| Pre–post study | ||||||
| Sleep quality | ||||||
| Eshaghi [39], Iran | Sleep quality: PSQI | NR | NR | Mean ± SD I: 12.55 ± 1.01 C: 12.55 ± 1.01 | Mean ± SD I: 10.11 ± 1.26 (significant difference compared with baseline) C: 12.44 ± 0.88 (contradictory evidence regarding C group between written (NS difference) and tabulated (sig. difference) results) | Sleep quality sig. improved by 19.1% with VDS |
| Other outcomes | ||||||
| Guler [40], Turkey | Sleep habits and disorders: short version of the CSHQ with a higher score reflecting more disturbed sleep behavior | Cases: 25(OH)D < 10 ng/mL: 23.3% 25(OH)D: 10–30 ng/mL: 45% 25(OH)D > 30 ng/mL: 31.7% mean ± SD: 25.58 ± 10.31 Controls: 25(OH)D < 10 ng/mL: 23.3% 25(OH)D: 10–30 ng/mL: 38.3% 25(OH)D > 30 ng/mL: 38.3% mean ± SD: 25.35 ± 9.92 (NS difference between Cases and Controls) In DEF participants: 25(OH)D: Cases: 19.68 ± 6.22 Controls: 19.21 ± 7.35 | Cases: 25(OH)D < 10 ng/mL: 0% 25(OH)D: 10–30 ng/mL: 11.7% 25(OH)D > 30 ng/mL): 88.3% mean ± SD: 37.27 ± 6.51 Controls: 25(OH)D < 10 ng/mL: 0% 25(OH)D: 10–30 ng/mL: 6.7% 25(OH)D > 30 ng/mL: 93.3% mean ± SD: 37.15 ± 6.78 (NS difference between Cases and Controls) In DEF participants: 25(OH)D: Cases: 37.26 ± 7.34; sig Controls: 39.13 ± 7.74; sig | CSHQ total score Cases: <41: 21.7%; ≥41: 78.3% Controls: <41: 66.7%; ≥41: 33.3% mean ± SD total sleep time (hours) Cases: 8.10 ± 0.97 Controls: 9.24 ± 0.89 In DEF participants Total score Cases: 52.05 ± 8.24 Controls: 42.00 ± 4.78 Bedtime resistance Cases: 11.24 ± 2.49 Controls: 7.38 ± 1.01 Sleep-onset delay Cases: 2.32 ± 0.79 Controls: 1.86 ± 0.89 Sleep duration Cases: 5.41 ± 0.92 Controls: 4.41 ± 0.98 Sleep anxiety Cases: 7.73 ± 2.59 Controls: 4.41 ± 0.80 Night wakings Cases: 5.59 ± 1.61 Controls: 4.11 ± 1.39 Parasomnias Cases: 9.80 ± 2.52 Controls: 8.51 ± 1.76 Sleep-disordered breathing Cases: 3.63 ± 0.73 Controls: 3.59 ± 0.90 Daytime sleepiness Cases: 10.44 ± 1.84 Controls: 9.89 ± 1.85 Total sleep time Cases: 8.16 ± 0.89 Controls: 9.11 ± 0.89 | CSHQ total score Cases: <41: 28.3%; ≥41: 71.7% Controls: <41: 86.7%; ≥41: 13.3% mean ± SD total sleep time (hours) Cases: 8.58 ± 0.96 Controls: 9.38 ± 0.88 (sleep time sig. different between Cases and Controls) In DEF participants Total score Cases: 46.43 ± 8.04; sig Controls: 37.56 ± 2.80; sig Bedtime resistance Cases: 10.17 ± 2.66; sig Controls: 7.21 ± 0.75; NS Sleep-onset delay Cases: 1.82 ± 0.80; sig Controls: 1.08 ± 0.36; sig Sleep duration Cases: 4.70 ± 0.95; sig Controls: 4.18 ± 0.90; sig Sleep anxiety Cases: 6.78 ± 2.35; sig Controls: 4.05 ± 0.4; sig Night wakings Cases: 4.24 ± 1.59; sig Controls: 3.32 ± 0.62; sig Parasomnias Cases: 8.75 ± 1.84; sig Controls: 7.16 ± 0.44; sig Sleep-disordered breathing Cases: 3.41 ± 059; sig Controls: 3.40 ± 0.76; sig Daytime sleepiness Cases: 10.17 ± 1.93; NS Controls: 9.21 ± 1.08; sig Total sleep time Cases: 8.63 ± 0.85; sig Controls: 9.29 ± 0.89; sig | VDS may be beneficial in ASD patients and healthy individuals with sleep disturbances |
| Arico [38], Italy | RLS severity: IRLS-RS | 10.3 | 30.4 (no information on statistical significance) | 19.8 | 8.6 (no information on statistical significance) | VDS has a therapeutic effect in decreasing RLS severity |
| Pre–post study, analyzed retrospectively as a case series | ||||||
| Sleep quality | ||||||
| Huang [42], USA | Sleep quality: PSQI Sleep latency: #2 “how long has it usually taken you to fall asleep each night” of the PSQI Sleep duration: #4 “how many hours of actual sleep did you get at night” of the PSQI Sleep efficiency: #4 + #1 “what time have you usually gone to bed at night” + #3 “what time have you usually gotten up in the morning” of the PSQI | Total: 18.57 ± 5.42 INS: 22.73 ± 1.83 DEF: 13.77 ± 3.94 | Total: 26.00 ± 8.38 (sig. increase) INS: 29.60 ± 11.67 DEF: 24.00 ± 5.79 | Global PSQI score Total: 13.46 ± 4.92 INS: 12.27 ± 5.55 DEF: 14.85 ± 3.83 Sleep latency (min) Total: 67.22 ± 56.13 INS: 41.61 ± 48.21 DEF: 94.81 ± 52.15 Sleep duration (h) Total: 4.59 ± 1.84 INS: 5.33 ± 1.88 DEF: 3.73 ± 1.41 Sleep efficiency (%) Total: 59.79 ± 25.31 INS: 66.97 ± 23.7 DEF: 52.61 ± 25.70 | Global PSQI score Total: 12.22 ± 4.61 (sig. decrease) INS: 11.29 ± 4.66 DEF: 13.23 ± 4.51 (sig. decrease) Sleep latency (min) Total: 57.86 ± 44.03 (sig. decrease) INS: 39.83 ± 39.05 DEF: 78.65 ± 41.33 (sig. decrease) Sleep duration (h) Total: 5.30 ± 1.57 (sig. decrease) INS: 5.90 ± 1.55 (sig. decrease) DEF: 4.62 ± 1.33 (sig. decrease) Sleep efficiency (%) Total: 66.62 ± 18.61 (sig. increase) INS: 70.34 ± 17.76 DEF: 62.33 ± 19.34 (sig. increase) | Sig. improvement in overall sleep quality, sleep latency, sleep duration, and sleep efficiency in veterans with multiple areas of chronic pain with VDS (after controlling for potential confounders improvement in sleep efficiency became borderline significant) The magnitudes of sleep improvement in latency, duration, and efficiency were all larger in the DEF subgroup; however, the difference in improvements between the subgroups was NS except for sleep latency |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abboud, M. Vitamin D Supplementation and Sleep: A Systematic Review and Meta-Analysis of Intervention Studies. Nutrients 2022, 14, 1076. https://doi.org/10.3390/nu14051076
Abboud M. Vitamin D Supplementation and Sleep: A Systematic Review and Meta-Analysis of Intervention Studies. Nutrients. 2022; 14(5):1076. https://doi.org/10.3390/nu14051076
Chicago/Turabian StyleAbboud, Myriam. 2022. "Vitamin D Supplementation and Sleep: A Systematic Review and Meta-Analysis of Intervention Studies" Nutrients 14, no. 5: 1076. https://doi.org/10.3390/nu14051076
APA StyleAbboud, M. (2022). Vitamin D Supplementation and Sleep: A Systematic Review and Meta-Analysis of Intervention Studies. Nutrients, 14(5), 1076. https://doi.org/10.3390/nu14051076






