Association of Gastric Antrum Echodensity and Acute Gastrointestinal Injury in Critically Ill Patients
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. POCUS Examination and Echodensity Measurement
2.3. Data Collection
2.4. Nutrition Delivery
2.5. Statistical Analysis
3. Results
3.1. Participants
3.2. Measurement of Gastric Antrum Echodensity and Consistency Analysis
3.3. Association between Gastric Antrum Echodensity and AGI Grade
3.4. Ability to Predict FI
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Blaser, A.R.; Poeze, M.; Malbrain, M.L.; Bjorck, M.; Oudemans-van Straaten, H.M.; Starkopf, J. Gastrointestinal symptoms during the first week of intensive care are associated with poor outcome: A prospective multicentre study. Intensive Care Med. 2013, 39, 899–909. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meng, M.; Klingensmith, N.J.; Coopersmith, C.M. New insights into the gut as the driver of critical illness and organ failure. Curr. Opin. Crit. Care 2017, 23, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Patel, J.J.; Rosenthal, M.D.; Miller, K.R.; Martindale, R.G. The gut in trauma. Curr. Opin. Crit. Care 2016, 22, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Blaser, A.R.; Malbrain, M.L.; Starkopf, J.; Fruhwald, S.; Jakob, S.M.; De Waele, J.; Braun, J.-P.; Poeze, M.; Spies, C. Gastrointestinal function in intensive care patients: Terminology, definitions and management. Recommendations of the ESICM Working Group on Abdominal Problems. Intensive Care Med. 2012, 38, 384–394. [Google Scholar] [CrossRef] [Green Version]
- McClave, S.A.; Gualdoni, J.; Nagengast, A.; Marsano, L.S.; Bandy, K.; Martindale, R.G. Gastrointestinal Dysfunction and Feeding Intolerance in Critical Illness: Do We Need an Objective Scoring System? Curr. Gastroenterol. Rep. 2020, 22, 1. [Google Scholar] [CrossRef]
- Asrani, V.M.; Brown, A.; Huang, W.; Bissett, I.; Windsor, J.A. Gastrointestinal Dysfunction in Critical Illness: A Review of Scoring Tools. J. Parenter. Enter. Nutr. 2020, 44, 182–196. [Google Scholar] [CrossRef]
- Blaser, A.R.; Padar, M.; Mandul, M.; Elke, G.; Engel, C.; Fischer, K.; Giabicani, M.; Gold, T.; Hess, B.; Hiesmayr, M.; et al. Development of the Gastrointestinal Dysfunction Score (GIDS) for critically ill patients—A prospective multicenter observational study (iSOFA study). Clin. Nutr. 2021, 40, 4932–4940. [Google Scholar] [CrossRef]
- Blaser, A.R.; Padar, M.; Tang, J.; Dutton, J.; Forbes, A. Citrulline and intestinal fatty acid-binding protein as biomarkers for gastrointestinal dysfunction in the critically ill. Anestezjol. Intensywna Ter. 2019, 51, 230–239. [Google Scholar] [CrossRef]
- Moonen, P.-J.; Blaser, A.R.; Starkopf, J.; Straaten, H.M.O.-V.; Van Der Mullen, J.; Vermeulen, G.; Malbrain, M.L. The black box revelation: Monitoring gastrointestinal function. Anestezjol. Intensywna Ter. 2014, 50, 72–81. [Google Scholar] [CrossRef]
- Blaser, A.R.; Preiser, J.-C.; Fruhwald, S.; Wilmer, A.; Wernerman, J.; Benstoem, C.; Casaer, M.P.; Starkopf, J.; van Zanten, A.; Rooyackers, O.; et al. Gastrointestinal dysfunction in the critically ill: A systematic scoping review and research agenda proposed by the Section of Metabolism, Endocrinology and Nutrition of the European Society of Intensive Care Medicine. Critical Care. 2020, 24, 1–17. [Google Scholar]
- Perez-Calatayud, A.A.; Carrillo-Esper, R.; Anica-Malagón, E.D.; Briones-Garduño, J.C.; Arch-Tirado, E.; Wise, R.; Malbrain, M.L. Point-of-care gastrointestinal and urinary tract sonography in daily evaluation of gastrointestinal dysfunction in critically ill patients (GUTS Protocol). Anestezjol. Intensywna Ter. 2018, 50, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Gao, T.; Cheng, M.-H.; Xi, F.-C.; Chen, Y.; Cao, C.; Su, T.; Li, W.-Q.; Yu, W.-K. Predictive value of transabdominal intestinal sonography in critically ill patients: A prospective observational study. Crit. Care 2019, 23, 378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Puthucheary, Z.; Phadke, R.; Rawal, J.; McPhail, M.J.W.; Sidhu, P.; Rowlerson, A.; Moxham, J.; Harridge, S.; Hart, N.; Montgomery, H. Qualitative Ultrasound in Acute Critical Illness Muscle Wasting. Crit. Care Med. 2015, 43, 1603–1611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiménez-Díaz, F.; Jimena, I.; Luque, E.; Mendizábal, S.; Bouffard, A.; Jiménez-Reina, L.; Peña, J. Experimental muscle injury: Correlation between ultrasound and histological findings. Muscle Nerve 2012, 45, 705–712. [Google Scholar] [CrossRef]
- Parry, S.M.; El-Ansary, D.; Cartwright, M.S.; Sarwal, A.; Berney, S.; Koopman, R.; Annoni, R.; Puthucheary, Z.; Gordon, I.R.; Morris, P.E.; et al. Ultrasonography in the intensive care setting can be used to detect changes in the quality and quantity of muscle and is related to muscle strength and function. J. Crit. Care 2015, 30, 1151.e9–1151.e14. [Google Scholar] [CrossRef]
- Coiffard, B.; Riegler, S.; Sklar, M.C.; Dres, M.; Vorona, S.; Reid, W.D.; Brochard, L.J.; Ferguson, N.D.; Goligher, E.C. Diaphragm echodensity in mechanically ventilated patients: A description of technique and outcomes. Crit. Care 2021, 25, 1–11. [Google Scholar] [CrossRef]
- Perlas, A.; Chan, V.W.S.; Lupu, C.M.; Mitsakakis, N.; Hanbidge, A. Ultrasound Assessment of Gastric Content and Volume. Anesthesiol. 2009, 111, 82–89. [Google Scholar] [CrossRef] [Green Version]
- Reintam, A.; Parm, P.; Kitus, R.; Starkopf, J.; Kern, H. Gastrointestinal failure score in critically ill patients: A prospective observational study. Crit. Care. 2008, 12, R90. [Google Scholar] [CrossRef] [Green Version]
- Heyland, D.K.; Cahill, N.E.; Dhaliwal, R.; Wang, M.; Day, A.G.; Alenzi, A.; Aris, F.; Muscedere, J.; Drover, J.W.; McClave, S.A. Enhanced protein-energy provision via the enteral route in critically ill S center feasibility trial of the PEP uP protocol. Crit. Care 2010, 14, R78. [Google Scholar] [CrossRef] [Green Version]
- Patel, J.J.; Kozeniecki, M.; Peppard, W.J.; Peppard, S.R.; Zellner-Jones, S.; Graf, J.; Szabo, A.; Heyland, D.K. Phase 3 Pilot Randomized Controlled Trial Comparing Early Trophic Enteral Nutrition With “No Enteral Nutrition” in Mechanically Ventilated Patients with Septic Shock. J. Parenter. Enter. Nutr. 2020, 44, 866–873. [Google Scholar] [CrossRef]
- Ukleja, A.; Altered, G.I. Motility in critically Ill patients: Current understanding of pathophysiology, clinical impact, and diagnostic approach. Nutr. Clin. Pract. 2010, 25, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Herbert, M.K.; Holzer, P. Standardized concept for the treatment of gastrointestinal dysmotility in critically ill patients—Current status and future options. Clin. Nutr. 2008, 27, 25–41. [Google Scholar] [CrossRef] [PubMed]
- Reignier, J.; Mercier, E.; Le Gouge, A.; Boulain, T.; Desachy, A.; Bellec, F.; Clavel, M.; Frat, J.-P.; Plantefeve, G.; Quenot, J.-P.; et al. Effect of Not Monitoring Residual Gastric Volume on Risk of Ventilator-Associated Pneumonia in Adults Receiving Mechanical Ventilation and Early Enteral Feeding: A Randomized Controlled Trial. JAMA 2013, 309, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Montejo, J.C.; Miñambres, E.; Bordeje, L.; Mesejo, A.; Acosta, J.; Heras, A.; Ferre, M.; Fernandez-Ortega, F.; Vaquerizo, C.I.; Manzanedo, R. Gastric residual volume during enteral nutrition in ICU patients: The REGANE study. Intensive Care Med. 2010, 36, 1386–1393. [Google Scholar] [CrossRef]
- Pillen, S.; Arts, I.M.P.; Zwarts, M.J. Muscle ultrasound in neuromuscular disorders. Muscle Nerve 2008, 37, 679–693. [Google Scholar] [CrossRef]
- Strasser, E.M.; Draskovits, T.; Praschak, M.; Quittan, M.; Graf, A. Association between ultrasound measurements of muscle thickness, pennation angle, echogenicity and skeletal muscle strength in the elderly. AGE 2013, 35, 2377–2388. [Google Scholar] [CrossRef] [Green Version]
- Pillen, S.; Tak, R.O.; Zwarts, M.J.; Lammens, M.M.Y.; Verrijp, K.N.; Arts, I.M.P.; van der Laak, J.A.; Hoogerbrugge, P.M.; van Engelen, B.G.M.; Verrips, A. Skeletal Muscle Ultrasound: Correlation Between Fibrous Tissue and Echo Intensity. Ultrasound Med. Biol. 2009, 35, 443–446. [Google Scholar] [CrossRef]
- Cartwright, M.S.; Kwayisi, G.; Ms, L.P.G.; Sarwal, A.; Walker, F.O.; Harris, J.M.; Berry, M.J.; Chahal, P.S.; Morris, P.E. Quantitative neuromuscular ultrasound in the intensive care unit. Muscle Nerve 2013, 47, 255–259. [Google Scholar] [CrossRef]
- Grimm, A.; Teschner, U.; Porzelius, C.; Ludewig, K.; Zielske, J.; Witte, O.W.; Brunkhorst, F.M.; Axer, H. Muscle ultrasound for early assessment of critical illness neuromyopathy in severe sepsis. Crit. Care 2013, 17, R227. [Google Scholar] [CrossRef] [Green Version]
- Formenti, P.; Umbrello, M.; Castagna, V.; Cenci, S.; Bichi, F.; Pozzi, T.; Bonifazi, M.; Coppola, S.; Chiumello, D. Respiratory and peripheral muscular ultrasound characteristics in ICU COVID-19 ARDS patients. J. Crit. Care 2022, 67, 14–20. [Google Scholar] [CrossRef]
- Umbrello, M.; Guglielmetti, L.; Formenti, P.; Antonucci, E.; Cereghini, S.; Filardo, C.; Montanari, G.; Muttini, S. Qualitative and quantitative muscle ultrasound changes in patients with COVID-19-related ARDS. Nutrition 2021, 91, 111449. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, Y.; Ikenaga, M.; Yoshimura, E.; Yamada, Y.; Kimura, M. Association between echo intensity and attenuation of skeletal muscle in young and older adults: A comparison between ultrasonography and computed tomography. Clin. Interv. Aging 2018, 13, 1871–1878. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, V.; Gudivada, D.; Gueret, R.; Bailitz, J. Ultrasound-Assessed Gastric Antral Area Correlates with Aspirated Tube Feed Volume in Enterally Fed Critically Ill Patients. Nutr. Clin. Pr. 2016, 32, 206–211. [Google Scholar] [CrossRef]
- Reintam Blaser, A.; Deane, A.M.; Preiser, J.-C.; Arabi, Y.M.; Jakob, S.M. Enteral Feeding Intolerance: Updates in Definitions and Pathophysiology. Nutr. Clin. Pract. 2021, 36, 40–49. [Google Scholar] [CrossRef] [PubMed]
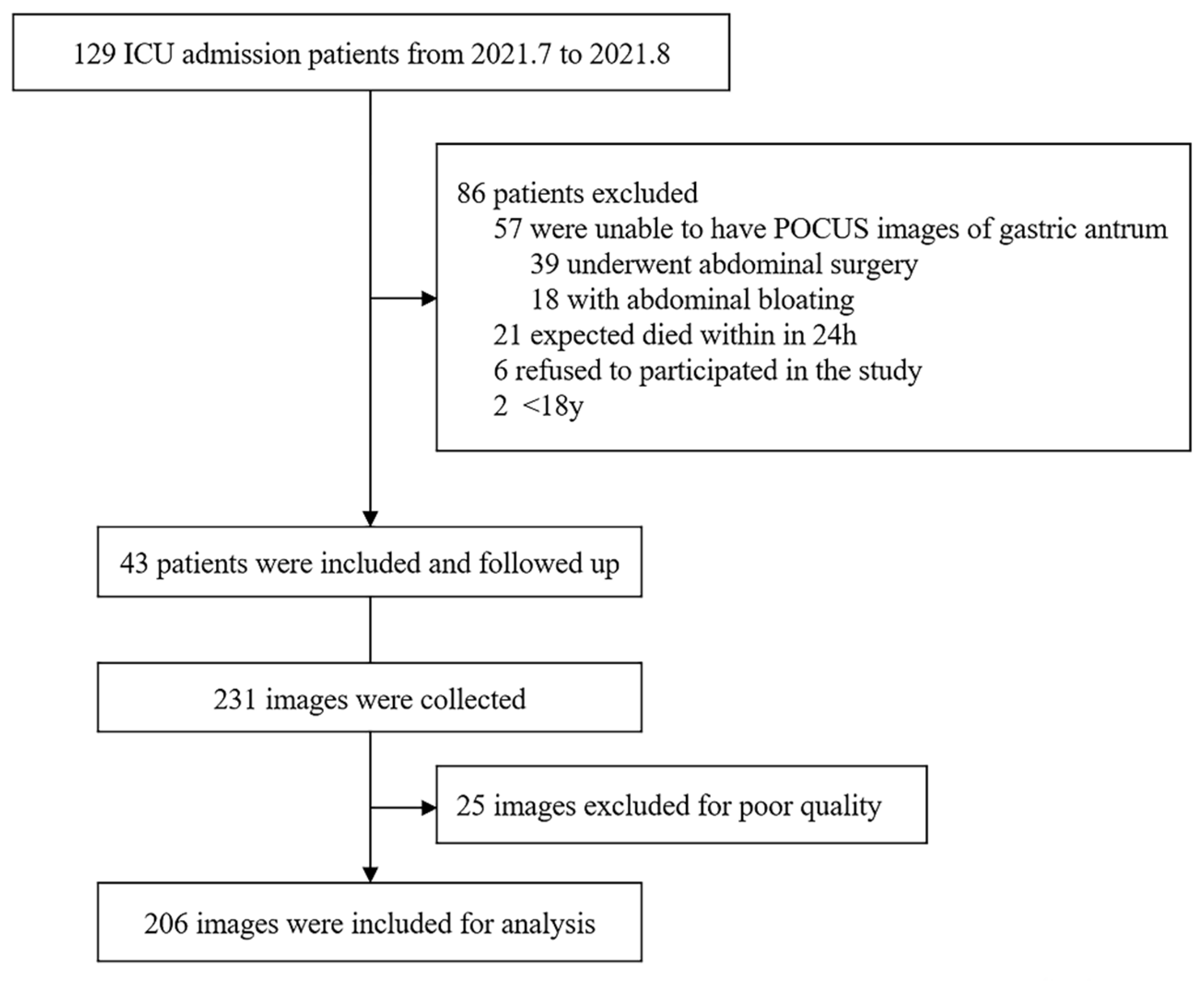
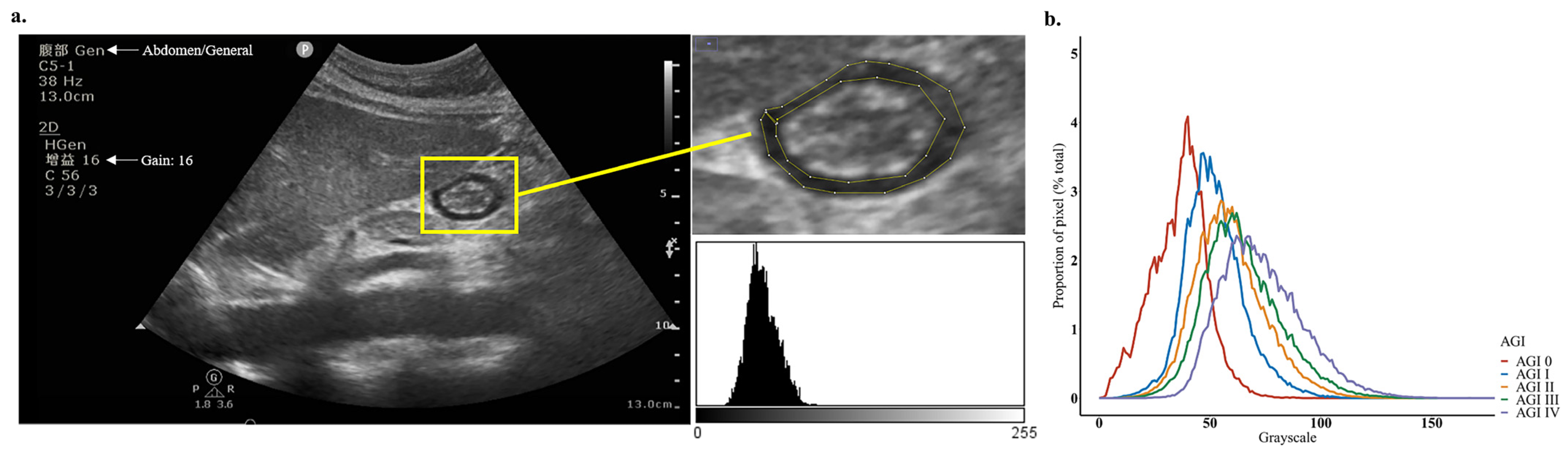
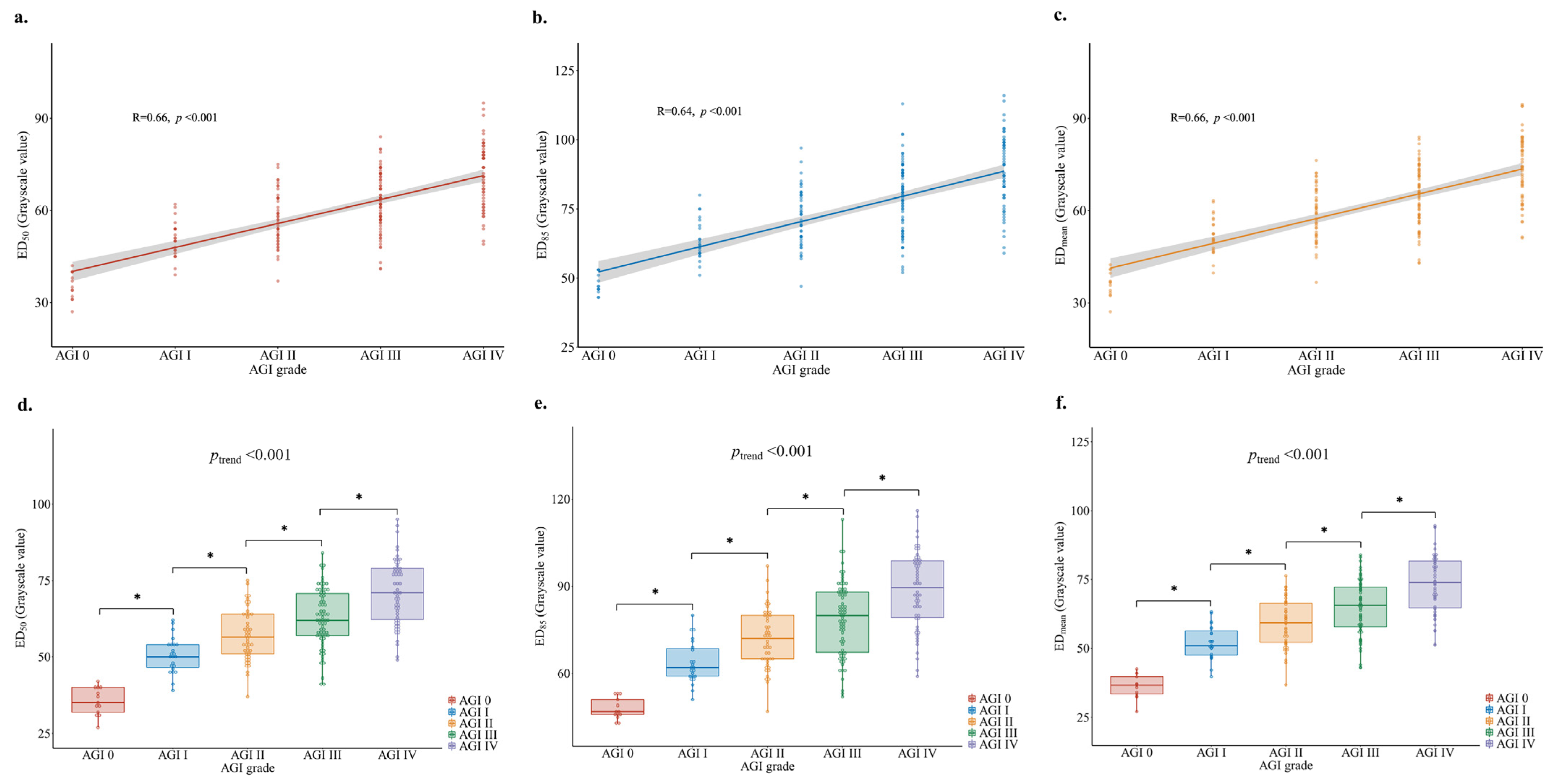
| Characteristic | Total (n = 43) |
|---|---|
| Age (years), mean ± SD | 60.1 ± 14.2 |
| Male Sex, No. (%) | 28 (65.1) |
| BMI, mean ± SD | 23.3 ± 3.4 |
| APACHE II score, mean ± SD | 18.4 ± 6.2 |
| SOFA score, mean ± SD | 9.4 ± 3.8 |
| Diagnosis, No. (%) | |
| Sepsis | 14 (32.6) |
| espiratory failure | 10 (23.3) |
| Multiple trauma | 6 (14) |
| Others 1 | 13 (30.2) |
| Comorbidities, No. (%) | |
| Cardiovascular Diseases 2 | 17 (39.5) |
| Neurological Diseases 3 | 15 (34.9) |
| Diabetes | 6 (14) |
| Laboratory tests at admission, mean ± SD | |
| White blood cell (×109/L) | 11.1 ± 7.3 |
| Hemoglobin (g/L) | 91.2 ± 22 |
| Albumin (g/L) | 31.3 ± 5.9 |
| Total bilirubin (umol/L) | 31.6 ± 45.3 |
| Serum creatinine (umol/L) | 136 ± 191 |
| Glucose (mmol/L) | 9.2 ± 4.3 |
| C-reactive protein (mg/L) | 90 ± 68.5 |
| Procalcitonin (μg/L) | 5.3 ± 9.2 |
| Lactic acid (mmol/L) | 2.2 ± 1.8 |
| ICU intervention | |
| Norepinephrine use (%) | 25 (58.1) |
| Vasopressin use (%) | 7 (16.3) |
| Prokinetics use (%) | 15 (34.9) |
| Cumulative sedation and analgesia dose within first week, mean ± SD | |
| Midazolam (mg/kg) | 5.9 ± 8.6 |
| Propofol (mg/kg) | 48.7 ± 60.2 |
| Dexmedetomidine (μg/kg) | 17.9 ± 22.7 |
| Remifentanil (mg/kg) | 0.3 ± 0.3 |
| Sufentanyl (μg/kg) | 4.1 ± 10 |
| AGI score on ICU admission | |
| AGI 0 | 2 (4.7) |
| AGI I | 9 (20.9) |
| AGI II | 3 (7) |
| AGI III | 17 (39.5) |
| AGI IV | 12 (27.9) |
| GIDS on ICU admission | |
| GIDS 0 | 10 (23.3) |
| GIDS 1 | 25 (58.1) |
| GIDS 2 | 6 (14) |
| GIDS 3 | 2 (4.7) |
| GIF score on ICU admission | |
| GIF 0 | 7 (16.3) |
| GIF 1 | 34 (79.1) |
| GIF 2 | 2 (4.7) |
| 28 d ventilation-free days (d) | 20.1 ± 5.1 |
| 28 d mortality | 10 (23.3) |
| Variable | AGI Grade | p for Trend | ||||
|---|---|---|---|---|---|---|
| AGI 0 | AGI I | AGI II | AGI III | AGI IV | ||
| ED50 | ||||||
| Median grayscale (IQR) | 35 (31.5–40) | 50 (46–54) | 56.5 (50.8–64) | 62 (57–71) | 71 (62–79) | |
| Unadjusted OR (95%CI) | 1 (Ref) | 1.8 (1.2–2.7) | 2 (1.3–3.1) | 2.1 (1.4–3.3) | 2.3 (1.5–3.6) | <0.001 |
| Model 1 OR (95%CI) | 1 (Ref) | 1.8 (1.1–2.9) | 2 (1.3–3.2) | 2.2 (1.4–3.5) | 2.4 (1.5–3.8) | <0.001 |
| Model 2 OR (95%CI) | 1 (Ref) | 1.9 (1.1–3.3) | 2.1 (1.2–3.5) | 2.3 (1.3–3.8) | 2.5 (1.5–4.2) | <0.001 |
| Model 3 OR (95%CI) | 1 (Ref) | 2.2 (1.1–4.5) | 2.6 (1.3–5.3) | 2.9 (1.4–6) | 3.2 (1.6–6.6) | <0.001 |
| ED85 | ||||||
| Median grayscale (IQR) | 47 (45.5–52) | 62 (59–69) | 72 (64.8–80) | 80 (67–88) | 89.5 (79–99) | |
| Unadjusted OR (95%CI) | 1 (Ref) | 1.6 (1.2–2.2) | 1.8 (1.3–2.5) | 1.9 (1.3–2.6) | 2 (1.4–2.7) | <0.001 |
| Model 1 OR (95%CI) | 1 (Ref) | 1.7 (1.2–2.5) | 1.9 (1.3–2.7) | 2.0 (1.4–2.9) | 2.1 (1.4–3.1) | <0.001 |
| Model 2 OR (95%CI) | 1 (Ref) | 1.8 (1.2–2.7) | 1.9 (1.3–2.9) | 2.0 (1.3–3) | 2.1 (1.4–3.2) | <0.001 |
| Model 3 OR (95%CI) | 1 (Ref) | 2.8 (1–8) | 3.2 (1.1–9.2) | 3.5 (1.2–10) | 3.7 (1.3–10.7) | <0.001 |
| EDmean | ||||||
| Median grayscale (IQR) | 36.6 (33–40.3) | 51 (47.2–57.1) | 59.3 (52.1–66.6) | 65.6 (57.8–72.4) | 73.9 (64.5–81.9) | |
| Unadjusted OR (95%CI) | 1 (Ref) | 1.7 (1.2–2.5) | 1.9 (1.3–2.8) | 2 (1.4–3) | 2.2 (1.5–3.3) | <0.001 |
| Model 1 OR (95%CI) | 1 (Ref) | 1.8 (1.2–2.7) | 2 (1.3–3) | 2.1 (1.4–3.2) | 2.3 (1.5–3.5) | <0.001 |
| Model 2 OR (95%CI) | 1 (Ref) | 1.9 (1.2–3) | 2 (1.3–3.3) | 2.2 (1.3–3.5) | 2.4 (1.5–3.8) | <0.001 |
| Model 3 OR (95%CI) | 1 (Ref) | 2 (1.1–3.5) | 2.3 (1.3–4.1) | 2.6 (1.5–4.5) | 2.8 (1.6–5) | <0.001 |
| Characteristic | Total (n = 38) | Non-FI Group (n = 21) | FI Group (n = 17) | p-Value |
|---|---|---|---|---|
| Age (years), mean ± SD | 61 ± 14.4 | 57.7 ± 13.9 | 65.1 ± 14.5 | 0.12 |
| Male Sex, No. (%) | 24 (63.2) | 13 (61.9) | 11 (64.7) | 0.86 |
| BMI, mean ± SD | 23.2 ± 3.6 | 23.4 ± 4 | 22.9 ± 3.2 | 0.66 |
| APACHE II score, mean ± SD | 18.6 ± 6 | 19.2 ± 6.2 | 17.9 ± 5.8 | 0.5 |
| SOFA score, mean ± SD | 9.4 ± 3.6 | 10.4 ± 4.1 | 8.1 ± 2.4 | 0.051 |
| Diagnosis, No. (%) | ||||
| Sepsis | 11 (28.9) | 5 (23.8) | 6 (35.3) | 0.68 |
| Respiratory failure | 10 (26.3) | 6 (28.6) | 4 (23.5) | >0.99 |
| Multiple trauma | 4 (10.5) | 3 (14.3) | 1 (5.9) | 0.76 |
| Others 1 | 13 (34.2) | 7 (33.3) | 6 (35.3) | 0.9 |
| Comorbidities—No. (%) | ||||
| Cardiovascular diseases 2 | 16 (42.1) | 5 (23.8) | 11 (64.7) | 0.01 |
| Neurological diseases 3 | 14 (36.8) | 8 (38.1) | 6 (35.3) | 0.86 |
| Diabetes | 6 (15.8) | 3 (14.3) | 3 (17.6) | >0.99 |
| Cumulative sedation and analgesia dose within first week, mean ± SD | ||||
| Midazolam (mg/kg) | 6.6 ± 8.9 | 8.6 ± 10 | 4.1 ± 6.9 | 0.12 |
| Propofol (mg/kg) | 53.6 ± 62.1 | 63.6 ± 69.1 | 41.3 ± 51.5 | 0.28 |
| Dexmedetomidine (μg/kg) | 20.1 ± 23.3 | 16.2 ± 24.5 | 24.9 ± 21.6 | 0.26 |
| Remifentanil (mg/kg) | 0.3 ± 0.3 | 0.4 ± 0.4 | 0.2 ± 0.3 | 0.13 |
| Sufentanyl (μg/kg) | 4.3 ± 10.6 | 3.6 ± 11.1 | 5.3 ± 10.1 | 0.63 |
| Time to EN initiation from ICU admission (h), mean ± SD | 56.4 ± 54.7 | 52.1 ± 50.3 | 61.7 ± 60.9 | 0.59 |
| Prokinetics use (%) | 15 (39.5) | 7 (33.3) | 8 (47.1) | 0.26 |
| Norepinephrine (μg/kg/min) at EN initiation, mean ± SD | 0.1 ± 0.2 | 0.11 ± 0.2 | 0.08 ± 0.16 | 0.65 |
| Ultrasonic parameters at EN initiation, mean ± SD | ||||
| ED50 | 61.3 ± 13.2 | 56.1 ± 12.7 | 67.8 ± 11.1 | 0.02 |
| D85 | 77.7 ± 15.7 | 71.2 ± 13.5 | 85.6 ± 14.9 | 0.01 |
| EDmean | 63.3 ± 13.6 | 57.6 ± 12.9 | 70.3 ± 11.3 | 0.01 |
| Anteroposterior diameter (cm) | 2.4 ± 0.6 | 2.4 ± 0.6 | 2.4 ± 0.6 | 0.98 |
| Craniocaudal diameter (cm) | 3 ± 0.7 | 3 ± 0.5 | 3 ± 0.9 | 0.87 |
| Antral area (cm2) | 5.6 ± 2.1 | 5.6 ± 1.7 | 5.7 ± 2.7 | 0.87 |
| 28 d ventilation-free days (d), mean ± SD | 20.1 ± 5.1 | 21.8 ± 4.4 | 19.1 ± 5.8 | 0.31 |
| 28 d mortality (%) | 8 (21.1) | 6 (28.6) | 2 (11.8) | 0.39 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, L.; Yang, H.; Lv, G.; Fu, X.; Cheng, Y.; Zhong, X.; Yang, J.; Wang, B.; Zhang, Z.; Jin, X.; et al. Association of Gastric Antrum Echodensity and Acute Gastrointestinal Injury in Critically Ill Patients. Nutrients 2022, 14, 566. https://doi.org/10.3390/nu14030566
Wang L, Yang H, Lv G, Fu X, Cheng Y, Zhong X, Yang J, Wang B, Zhang Z, Jin X, et al. Association of Gastric Antrum Echodensity and Acute Gastrointestinal Injury in Critically Ill Patients. Nutrients. 2022; 14(3):566. https://doi.org/10.3390/nu14030566
Chicago/Turabian StyleWang, Luping, Hao Yang, Guangxuan Lv, Xin Fu, Yisong Cheng, Xi Zhong, Jing Yang, Bo Wang, Zhongwei Zhang, Xiaodong Jin, and et al. 2022. "Association of Gastric Antrum Echodensity and Acute Gastrointestinal Injury in Critically Ill Patients" Nutrients 14, no. 3: 566. https://doi.org/10.3390/nu14030566
APA StyleWang, L., Yang, H., Lv, G., Fu, X., Cheng, Y., Zhong, X., Yang, J., Wang, B., Zhang, Z., Jin, X., Kang, Y., & Wu, Q. (2022). Association of Gastric Antrum Echodensity and Acute Gastrointestinal Injury in Critically Ill Patients. Nutrients, 14(3), 566. https://doi.org/10.3390/nu14030566





