Curcumin-Added Whey Protein Positively Modulates Skeletal Muscle Inflammation and Oxidative Damage after Exhaustive Exercise
Abstract
1. Introduction
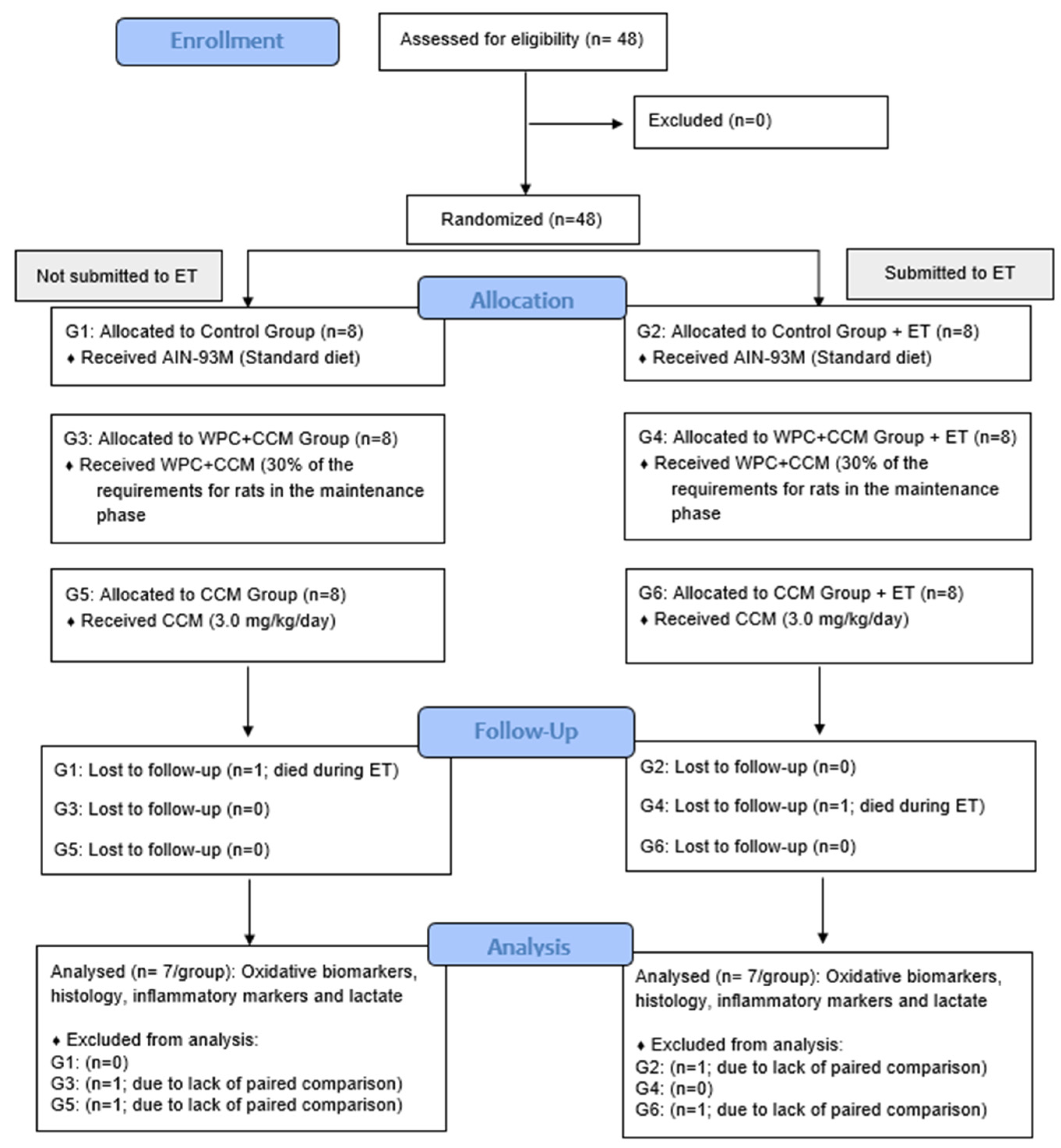
2. Materials and Methods
2.1. Feedstock
2.1.1. Curcumin
2.1.2. Whey Protein Concentrate (WPC)
2.1.3. Curcumin-Added Whey Protein Concentrate
2.2. Animals and Experimental Diets
2.3. Analysis of Oxidation Products and Activity of Antioxidant Enzymes
2.4. Cytokine Profile in Muscle Homogenate
2.5. Real-Time Polymerase Chain Reaction (RT-qPCR)
2.6. Histopathological Analyzes
2.7. Blood Lactate Analysis during the Exhaustion Test
2.8. Statistical Analysis
3. Results
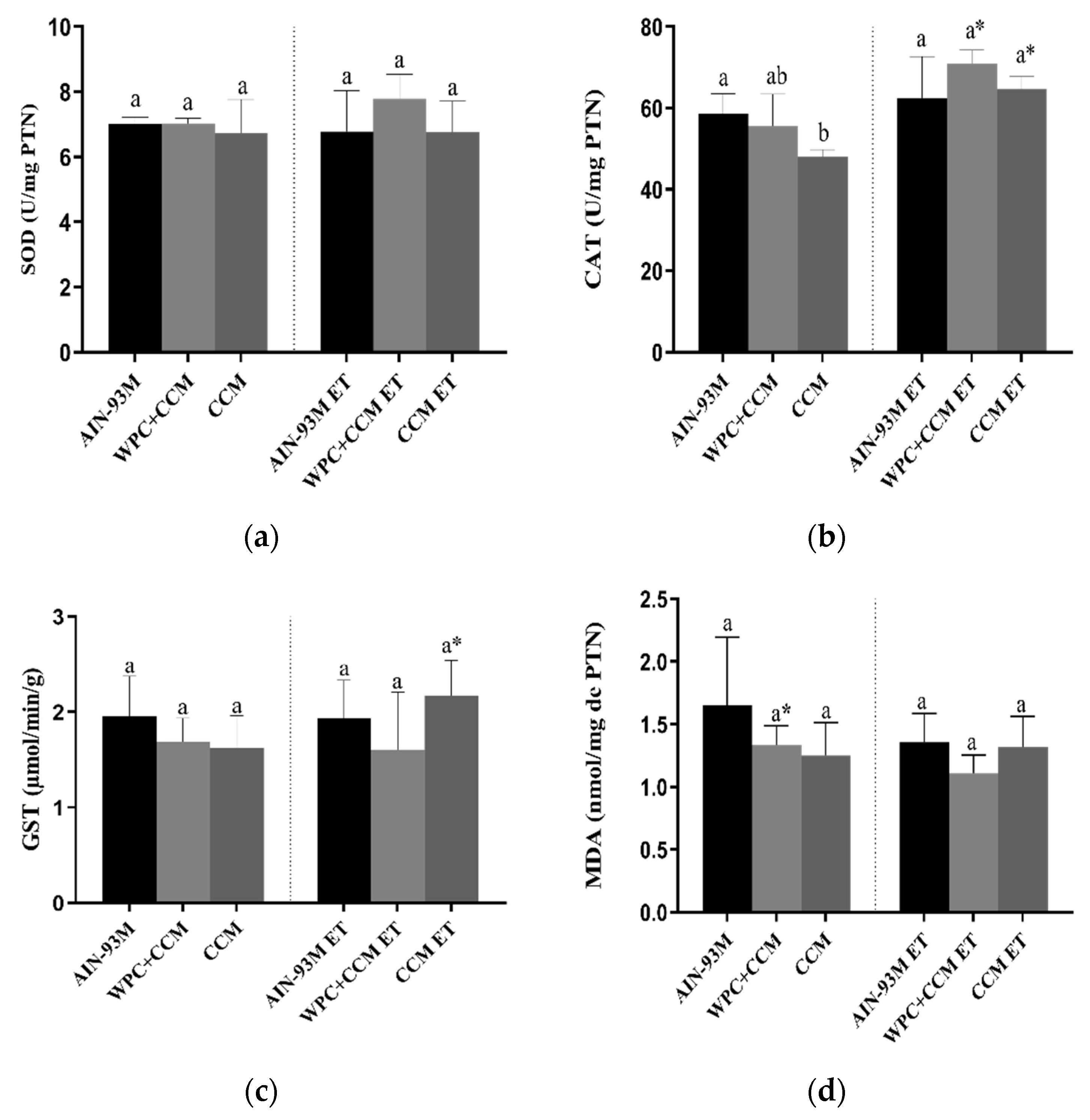
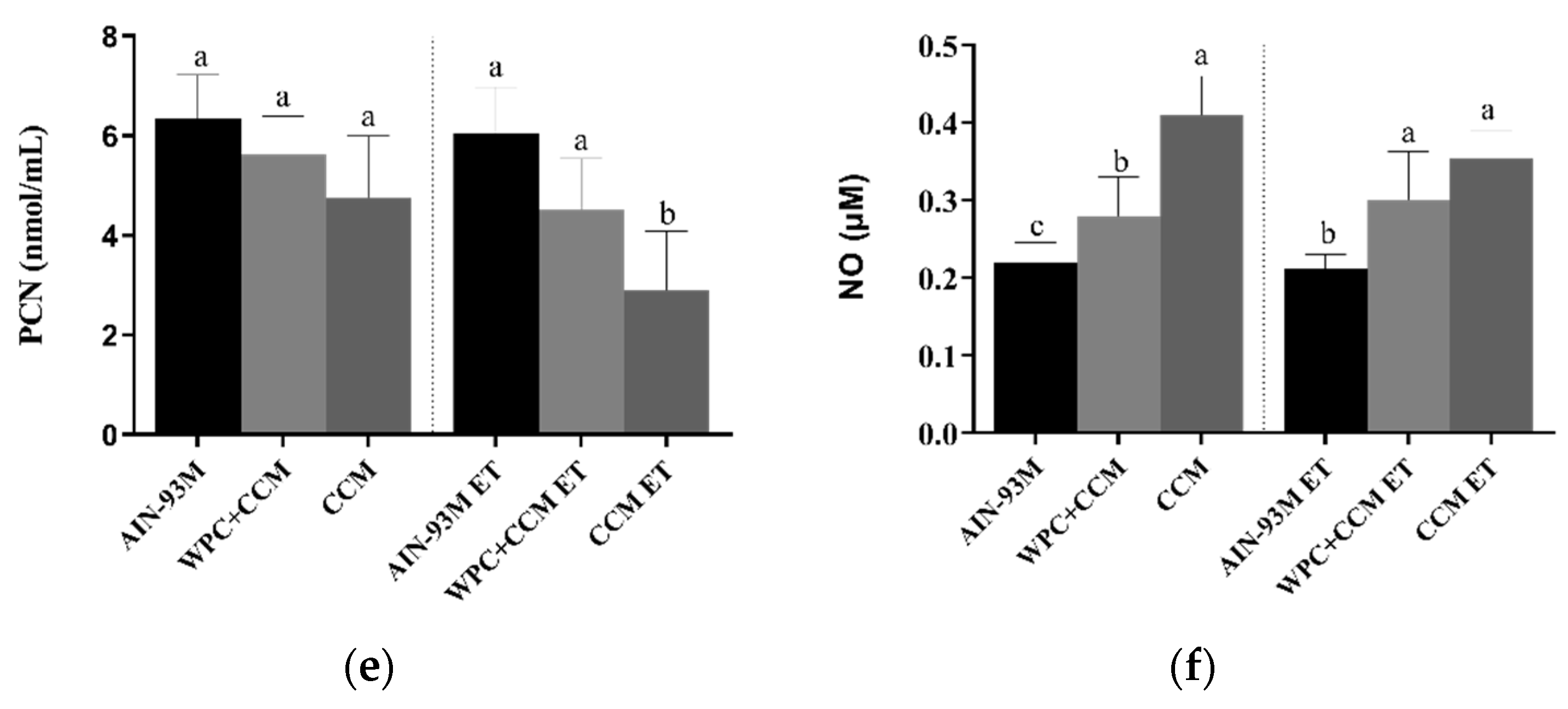
3.1. Antioxidant Enzymes and Markers Oxidatives
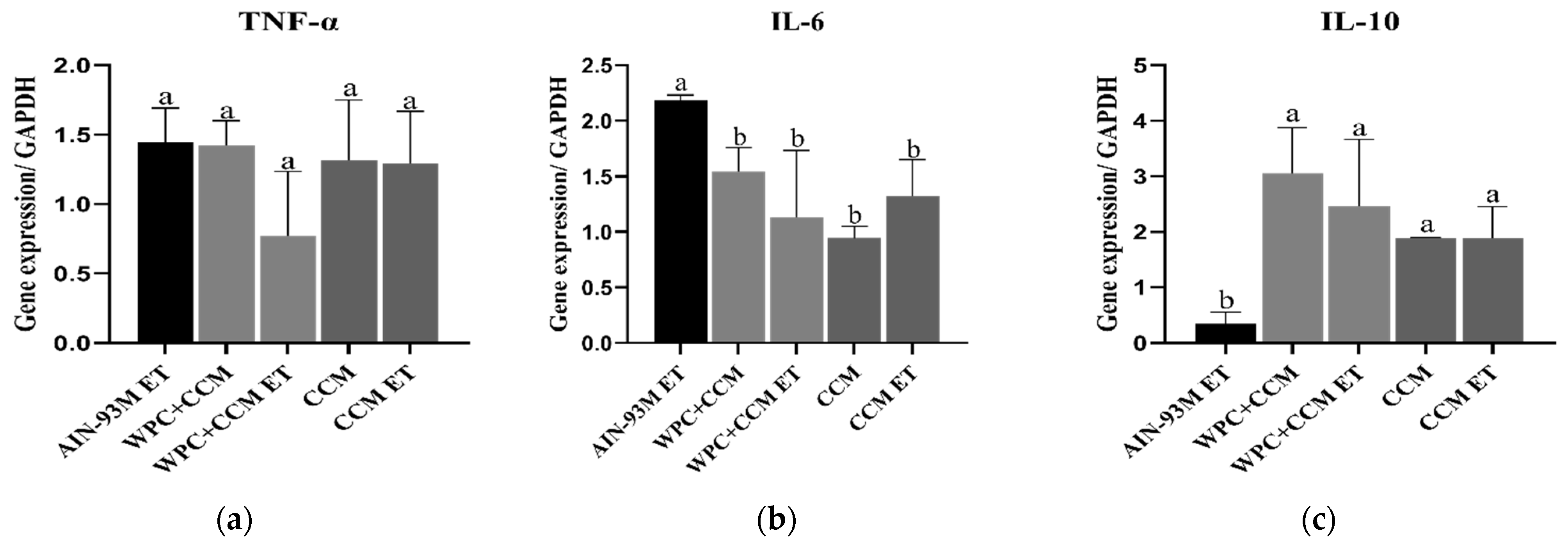
3.2. Gene Expression of TNF-α, IL-6, and IL-10
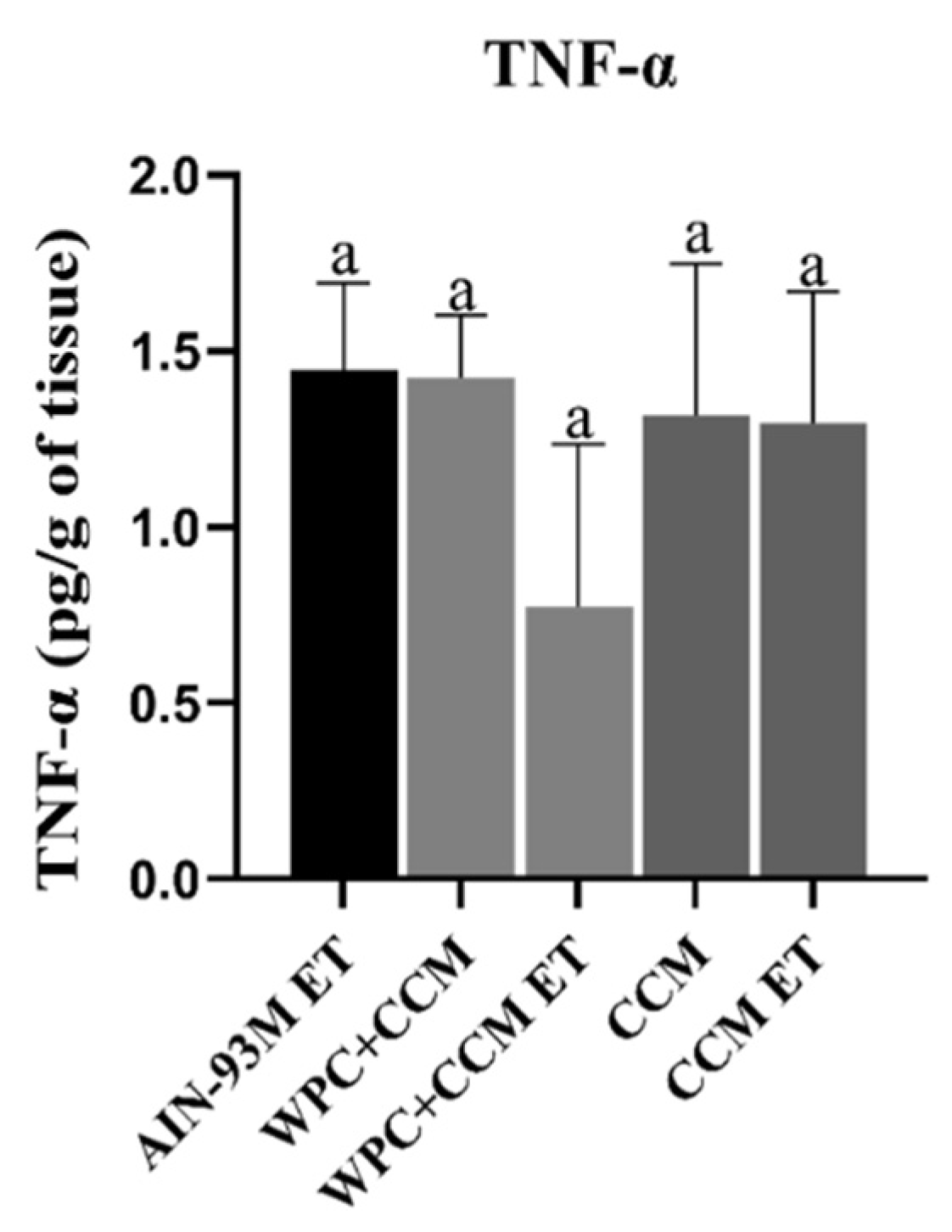
3.3. Cytokine Production Profile in Muscle Tissue
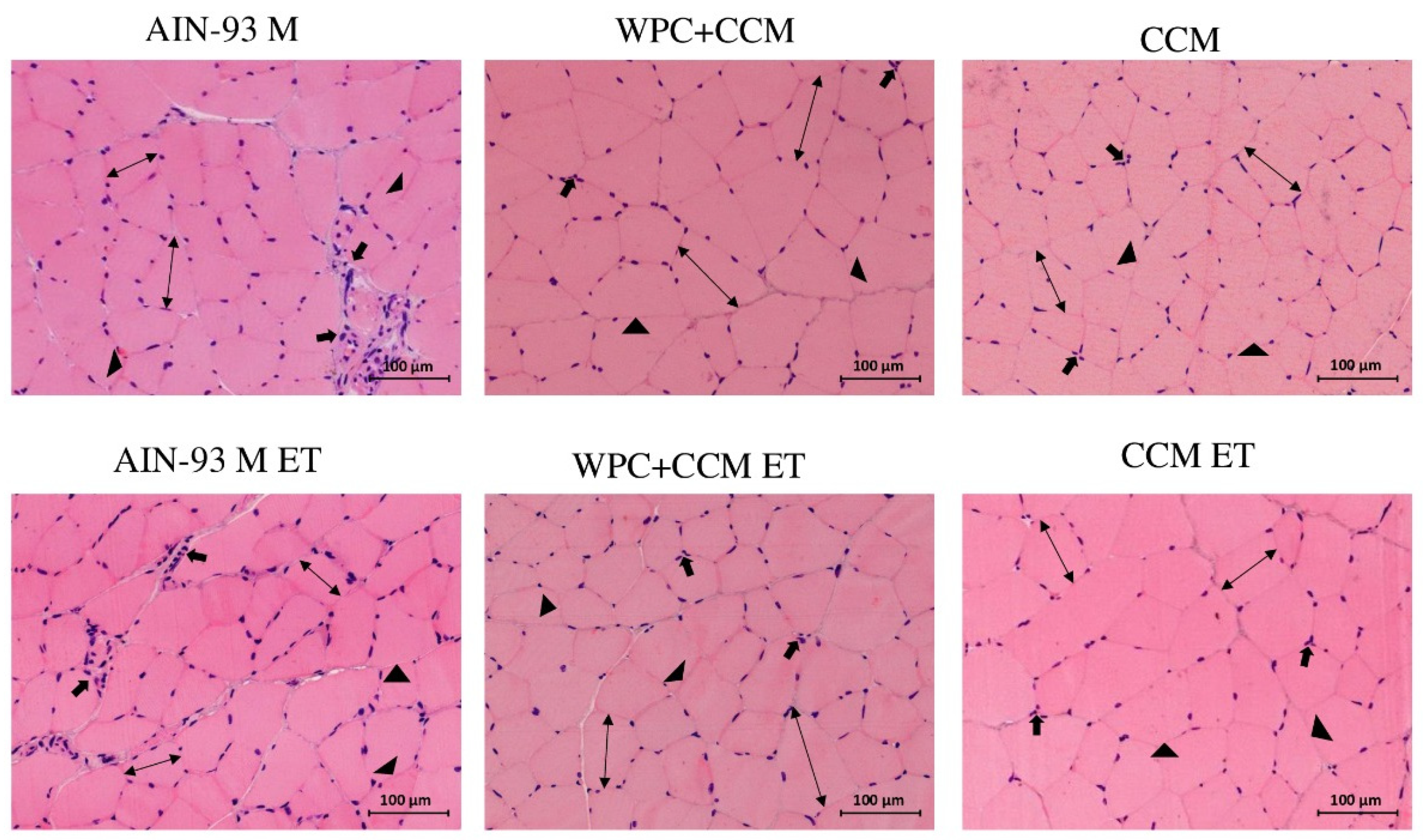
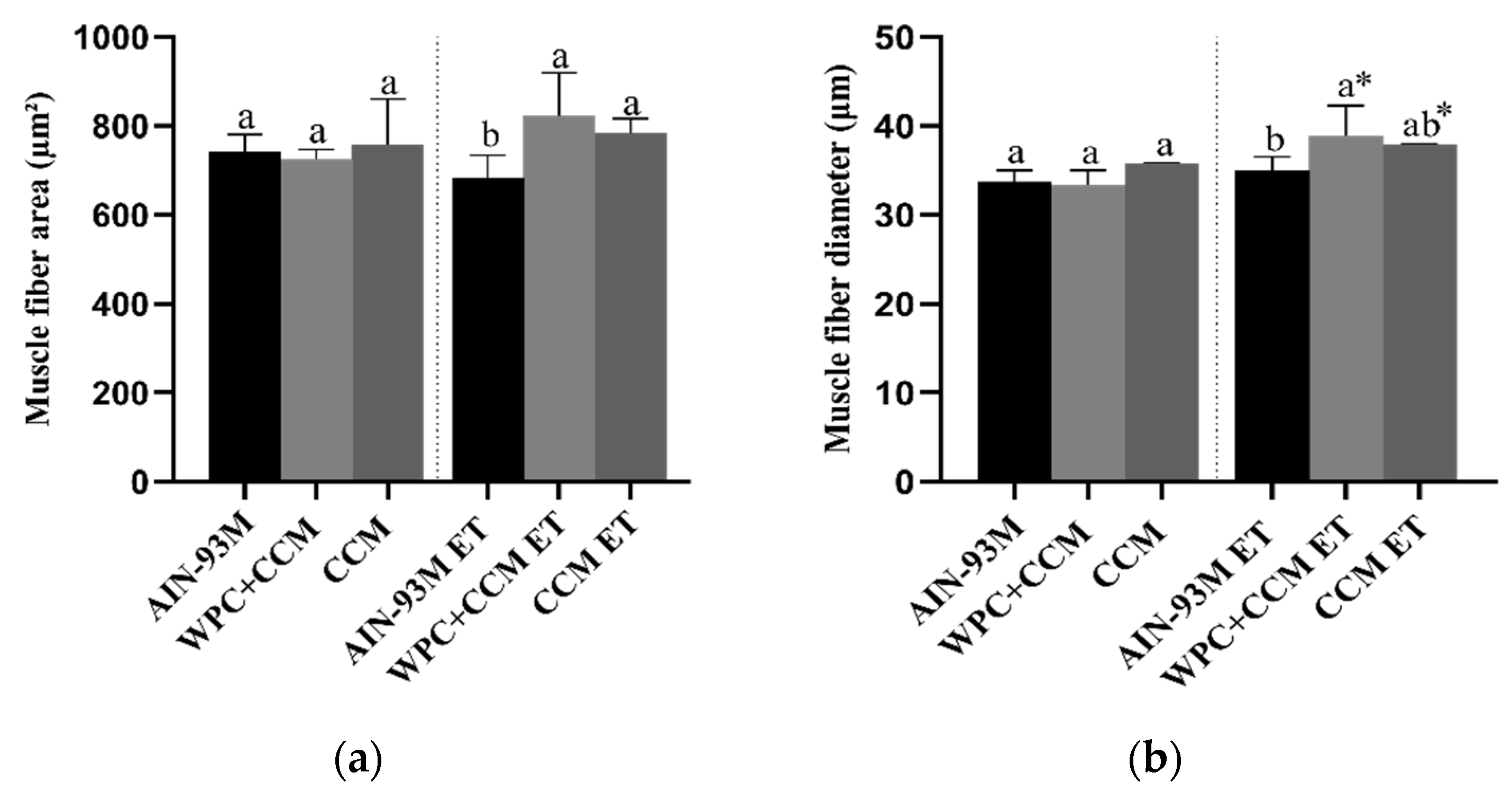
3.4. Histopathology
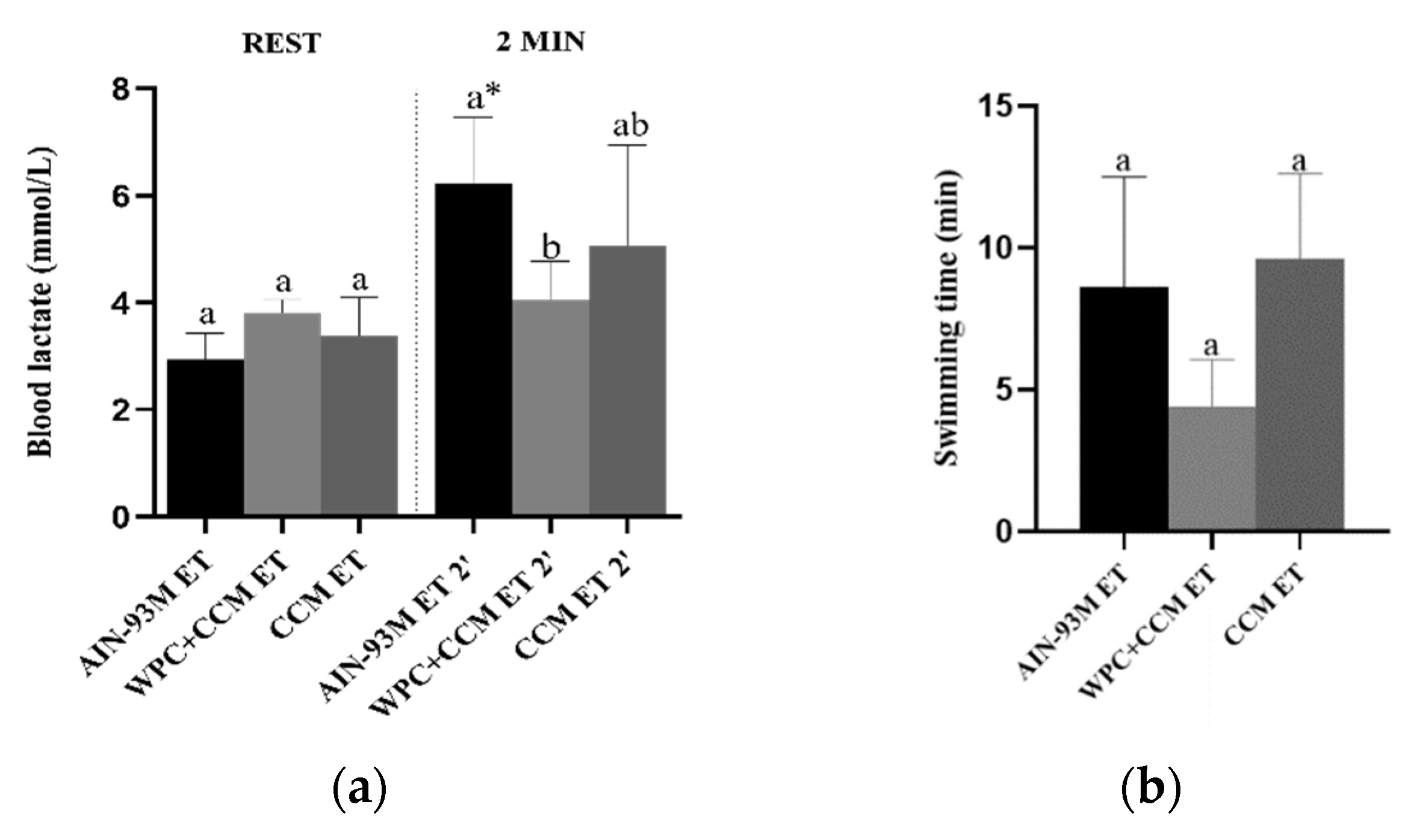
3.5. Blood Lactate and Swimming Time
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Clarkson, P.M.; Nosaka, K.; Braun, B. Muscle Function after Exercise-Induced Muscle Damage and Rapid Adaptation. Med. Sci. Sports Exerc. 1992, 24, 512–520. [Google Scholar] [CrossRef] [PubMed]
- González-Bartholin, R.; Mackay, K.; Valladares, D.; Zbinden-Foncea, H.; Nosaka, K.; Peñailillo, L. Changes in Oxidative Stress, Inflammation and Muscle Damage Markers Following Eccentric versus Concentric Cycling in Older Adults. Eur. J. Appl. Physiol. 2019, 119, 2301–2312. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, Y.; Chino, K.; Sagayama, H.; Lee, H.J.; Ozawa, H.; Maeda, S.; Takahashi, H. Effective Timing of Curcumin Ingestion to Attenuate Eccentric Exercise–Induced Muscle Soreness in Men. J. Nutr. Sci. Vitaminol. 2019, 65, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Howatson, G.; Van Someren, K.A. The Prevention and Treatment of Exercise-Induced Muscle Damage. Sport. Med. 2008, 38, 483–503. [Google Scholar] [CrossRef]
- Huang, W.C.; Chiu, W.C.; Chuang, H.L.; Tang, D.W.; Lee, Z.M.; Li, W.; Chen, F.A.; Huang, C.C. Effect of Curcumin Supplementation on Physiological Fatigue and Physical Performance in Mice. Nutrients 2015, 7, 905–921. [Google Scholar] [CrossRef] [PubMed]
- Faria, F.R.; Gomes, A.C.; Antunes, A.; Rezende, K.R.; Pimentel, G.D.; Oliveira, C.L.P.; Antunes, B.M.; Lira, F.S.; Aoki, M.S.; Mota, J.F. Effects of Turmeric Extract Supplementation on Inflammation and Muscle Damage after a Half-Marathon Race: A Randomized, Double-Blind, Placebo-Controlled Trial. Eur. J. Appl. Physiol. 2020, 120, 1531–1540. [Google Scholar] [CrossRef]
- Fuster-Muñoz, E.; Roche, E.; Funes, L.; Martínez-Peinado, P.; Sempere, J.M.; Vicente-Salar, N. Effects of Pomegranate Juice in Circulating Parameters, Cytokines, and Oxidative Stress Markers in Endurance-Based Athletes: A Randomized Controlled Trial. Nutrition 2016, 32, 539–545. [Google Scholar] [CrossRef]
- Howatson, G.; McHugh, M.P.; Hill, J.A.; Brouner, J.; Jewell, A.P.; Van Someren, K.A.; Shave, R.E.; Howatson, S.A. Influence of Tart Cherry Juice on Indices of Recovery Following Marathon Running. Scand. J. Med. Sci. Sport. 2010, 20, 843–852. [Google Scholar] [CrossRef]
- Hewlings, S.J.; Kalman, D.S. Curcumin: A Review of Its Effects on Human Health. Foods 2017, 6, 92. [Google Scholar] [CrossRef]
- Liu, F.; Gao, S.; Yang, Y.; Zhao, X.; Fan, Y.; Ma, W.; Yang, D.; Yang, A.; Yu, Y. Antitumor Activity of Curcumin by Modulation of Apoptosis and Autophagy in Human Lung Cancer A549 Cells through Inhibiting PI3K/Akt/MTOR Pathway. Oncol. Rep. 2018, 39, 1523–1531. [Google Scholar] [CrossRef]
- Li, M.; Liu, F. Effect of Whey Protein Supplementation during Resistance Training Sessions on Body Mass and Muscular Strength: A Meta-Analysis. Food Funct. 2019, 10, 2766–2773. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, Y.; Maeda, S.; Akazawa, N.; Zempo-Miyaki, A.; Choi, Y.; Ra, S.G.; Imaizumi, A.; Otsuka, Y.; Nosaka, K. Attenuation of Indirect Markers of Eccentric Exercise-Induced Muscle Damage by Curcumin. Eur. J. Appl. Physiol. 2015, 115, 1949–1957. [Google Scholar] [CrossRef] [PubMed]
- Nicol, L.M.; Rowlands, D.S.; Fazakerly, R.; Kellett, J. Curcumin Supplementation Likely Attenuates Delayed Onset Muscle Soreness (DOMS). Eur. J. Appl. Physiol. 2015, 115, 1769–1777. [Google Scholar] [CrossRef] [PubMed]
- Sahin, K.; Pala, R.; Tuzcu, M.; Ozdemir, O.; Orhan, C.; Sahin, N.; Juturu, V. Curcumin Prevents Muscle Damage by Regulating NF-ΚB and Nrf2 Pathways and Improves Performance: An in Vivo Model. J. Inflamm. Res. 2016, 9, 147–154. [Google Scholar] [CrossRef]
- Fridén, J.; Lieber, R.L. Structural and Mechanical Basis of Exercise-Induced Muscle Injury. Med. Sci. Sports Exerc. 1992, 24, 521–529. [Google Scholar] [CrossRef]
- Davies, R.W.; Carson, B.P.; Jakeman, P.M. The Effect of Whey Protein Supplementation on the Temporal Recovery of Muscle Function Following Resistance Training: A Systematic Review and Meta-Analysis. Nutrients 2018, 10, 221. [Google Scholar] [CrossRef]
- Atherton, P.J.; Smith, K. Muscle Protein Synthesis in Response to Nutrition and Exercise. J. Physiol. 2012, 590, 1049–1057. [Google Scholar] [CrossRef]
- Teixeira, K.R.; Silva, M.E.; de Lima, W.G.; Pedrosa, M.L.; Haraguchi, F.K. Whey Protein Increases Muscle Weight Gain through Inhibition of Oxidative Effects Induced by Resistance Exercise in Rats. Nutr. Res. 2016, 36, 1081–1089. [Google Scholar] [CrossRef]
- Athira, S.; Mann, B.; Sharma, R.; Kumar, R. Ameliorative Potential of Whey Protein Hydrolysate against Paracetamol-Induced Oxidative Stress. J. Dairy Sci. 2013, 96, 1431–1437. [Google Scholar] [CrossRef]
- Kerasioti, E.; Stagos, D.; Jamurtas, A.; Kiskini, A.; Koutedakis, Y.; Goutzourelas, N.; Pournaras, S.; Tsatsakis, A.M.; Kouretas, D. Anti-Inflammatory Effects of a Special Carbohydrate-Whey Protein Cake after Exhaustive Cycling in Humans. Food Chem. Toxicol. 2013, 61, 42–46. [Google Scholar] [CrossRef]
- Ma, Y.; Liu, J.; Shi, H.; Yu, L. Isolation and Characterization of Anti-Inflammatory Peptides Derived from Whey Protein. J. Dairy Sci. 2016, 99, 6902–6912. [Google Scholar] [CrossRef] [PubMed]
- Combaret, L.; Dardevet, D.; Rieu, I.; Pouch, M.N.; Béchet, D.; Taillandier, D.; Grizard, J.; Attaix, D. A Leucine-Supplemented Diet Restores the Defective Postprandial Inhibition of Proteasome-Dependent Proteolysis in Aged Rat Skeletal Muscle. J. Physiol. 2005, 569, 489–499. [Google Scholar] [CrossRef]
- Tanabe, Y.; Chino, K.; Ohnishi, T.; Ozawa, H.; Sagayama, H.; Maeda, S.; Takahashi, H. Effects of Oral Curcumin Ingested before or after Eccentric Exercise on Markers of Muscle Damage and Inflammation. Scand. J. Med. Sci. Sport. 2019, 29, 524–534. [Google Scholar] [CrossRef] [PubMed]
- Wang, I.L.; Hsiao, C.Y.; Li, Y.H.; Meng, F.B.; Huang, C.C.; Chen, Y.M. Nanobubbles Water Curcumin Extract Reduces Injury Risks on Drop Jumps in Women: A Pilot Study. Evidence-Based Complement. Altern. Med. 2019, 2019, 8647587. [Google Scholar] [CrossRef] [PubMed]
- Perrone, Í.T.; Simeão, M.; Rodrigues Júnior, P.H.; Stephani, R.; Carvalho, A.F. Influence of Pilot Spray Dryer Settings on Moisture and Water Activity of Whole Milk Powder. Rev. Do Inst. Laticínios Cândido Tostes 2013, 68, 5–9. [Google Scholar] [CrossRef]
- Stankovic, I. Curcumin—Chemical and Technical Assessment. FAO 2004, 61st JECFA, 1–8. [Google Scholar]
- Schoenfeld, B.J.; Aragon, A.A. How Much Protein Can the Body Use in a Single Meal for Muscle-Building? Implications for Daily Protein Distribution. J. Int. Soc. Sports Nutr. 2018, 15, 10. [Google Scholar] [CrossRef]
- Moher, D.; Schulz, K.F.; Altman, D.G.; Lepage, L. The CONSORT Statement: Revised Recommendations for Improving the Quality of Reports of Parallel-Group Randomised Trials. Lancet 2001, 357, 1191–1194. [Google Scholar] [CrossRef]
- Reeves, P.G.; Nielsen, F.H.; Fahey, G.C. AIN-93 Purified Diets for Laboratory Rodents: Final Report of the American Institute of Nutrition Ad Hoc Writing Committee on the Reformulation of the AIN-76A Rodent Diet. J. Nutr. 1993, 123, 1939–1951. [Google Scholar] [CrossRef]
- Vendetti, P.; Di Meo, S. Antioxidants, Tissue Damage, and Endurance in Trained and Untrained Young Male Rats. Arch. Biochem. Biophys. 1996, 331, 63–68. [Google Scholar] [CrossRef]
- Dawson, C.; Horvath, S.M. Swimming in Small Laboratory Animals. Med. Sci. Sport. Exerc. 1970, 2, 51–78. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in Vitro. Methods Enzymol. 1984, 105, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Dieterich, S.; Bieligk, U.; Beulich, K.; Hasenfuss, G.; Prestle, J. Gene Expression of Antioxidative Enzymes in the Human Heart: Increased Expression of Catalase in the End-Stage Failing Heart. Circulation 2000, 101, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Habig, W.H.; Jakoby, W.B. Glutathione S-Transferases (Rat and Human). Methods Enzymol. 1981, 77, 218–231. [Google Scholar] [CrossRef] [PubMed]
- Buege, J.A.; Aust, S.D. Microsomal Lipid Peroxidation. Methods Enzymol. 1978, 52, 302–310. [Google Scholar] [CrossRef]
- Tsikas, D. Analysis of Nitrite and Nitrate in Biological Fluids by Assays Based on the Griess Reaction: Appraisal of the Griess Reaction in the l-Arginine/Nitric Oxide Area of Research. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2007, 851, 51–70. [Google Scholar] [CrossRef]
- Levine, R.L.; Garland, D.; Oliver, C.N.; Amici, A.; Climent, I.; Lenz, A.G.; Ahn, B.W.; Shaltiel, S.; Stadtman, E.R. Determination of Carbonyl Content in Oxidatively Modified Proteins. Methods Enzymol. 1990, 186, 464–478. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L. Protein Measurement with the Folin Phenol Reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Sabarense, C.M.; Rocha, K.S.O.; Rosa, D.D.; Martins, J.H.; da Pereira, M.M.L.S.; Silva, F.F.; Steward, B.L. A New Computational Method for Hepatic Fat Microvesicles Counting in Histological Study in Rats. Biochem. Biophys. Res. Commun. 2012, 418, 284–289. [Google Scholar] [CrossRef]
- Gobatto, C.A.; De Mello, M.A.R.; Sibuya, C.Y.; De Azevedo, J.R.M.; Dos Santos, L.A.; Kokubun, E. Maximal Lactate Steady State in Rats Submitted to Swimming Exercise. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2001, 130, 21–27. [Google Scholar] [CrossRef]
- Dias, K.A.; Da Conceição, A.R.; Oliveira, L.A.; Pereira, S.M.S.; Paes, S.D.S.; Monte, L.F.; Sarandy, M.M.; Novaes, R.D.; Gonçalves, R.V.; Della Lucia, C.M. Effects of Curcumin Supplementation on Inflammatory Markers, Muscle Damage, and Sports Performance during Acute Physical Exercise in Sedentary Individuals. Oxid. Med. Cell. Longev. 2021, 2021, 9264639. [Google Scholar] [CrossRef] [PubMed]
- Campbell, M.S.; Carlini, N.A.; Fleenor, B.S. Influence of Curcumin on Performance and Post-Exercise Recovery. Crit. Rev. Food Sci. Nutr. 2021, 61, 1152–1162. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, R.V.; Costa, A.M.A.; Grzeskowiak, L. Oxidative Stress and Tissue Repair: Mechanism, Biomarkers, and Therapeutics|Hindawi. Oxid. Med. Cell. Longev. 2021, 2021, 6204096. [Google Scholar] [CrossRef] [PubMed]
- Radak, Z.; Taylor, A.W. Exercise and Hormesis. In The Science of Hormesis in Health and Longevity; Elsevier: Amsterdam, The Netherlands, 2018; pp. 63–73. ISBN 9780128142547. [Google Scholar]
- Tsikas, D. Assessment of Lipid Peroxidation by Measuring Malondialdehyde (MDA) and Relatives in Biological Samples: Analytical and Biological Challenges. Anal. Biochem. 2017, 524, 13–30. [Google Scholar] [CrossRef]
- Hendrix, J.; Nijs, J.; Ickmans, K.; Godderis, L.; Ghosh, M.; Polli, A. The Interplay between Oxidative Stress, Exercise, and Pain in Health and Disease: Potential Role of Autonomic Regulation and Epigenetic Mechanisms. Antioxidants 2020, 9, 1166. [Google Scholar] [CrossRef]
- Da Conceição, A.R.; Dias, K.A.; Pereira, S.M.S.; Saraiva, L.C.; Oliveira, L.A.; De Souza, E.C.G.; Gonçalves, R.V.; Da Matta, S.L.P.; Natali, A.J.; Martino, H.S.D.; et al. Protective Effects of Whey Protein Concentrate Admixtured of Curcumin on Metabolic Control, Inflammation and Oxidative Stress in Wistar Rats Submitted to Exhaustive Exercise. Br. J. Nutr. 2021, 127, 526–539. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine, 5th ed.; Oxford University Press: New York, NY, USA, 2015. [Google Scholar]
- Robbins, R.A.; Grisham, M.B. Nitric Oxide. Int. J. Biochem. Cell Biol. 1997, 29, 857–860. [Google Scholar] [CrossRef]
- Garcia, V.; Sessa, W.C. Endothelial NOS: Perspective and Recent Developments. Br. J. Pharmacol. 2019, 176, 189–196. [Google Scholar] [CrossRef]
- Prestes, J.; Donatto, F.F.; Dias, R.; Frollini, A.B.; Cavaglieri, C.R. Papel Da Interleucina-6 Como Um Sinalizador Em Diferentes Tecidos Durante O Exercício Físico. Fit. Perform. J. 2006, 5, 348–353. [Google Scholar] [CrossRef]
- Drobnic, F.; Riera, J.; Appendino, G.; Togni, S.; Franceschi, F.; Valle, X.; Pons, A.; Tur, J. Reduction of Delayed Onset Muscle Soreness by a Novel Curcumin Delivery System (Meriva®): A Randomised, Placebo-Controlled Trial. J. Int. Soc. Sports Nutr. 2014, 11, 31. [Google Scholar] [CrossRef]
- Davis, J.M.; Murphy, E.A.; Carmichael, M.D.; Zielinski, M.R.; Groschwitz, C.M.; Brown, A.S.; Gangemi, J.D.; Ghaffar, A.; Mayer, E.P. Curcumin Effects on Inflammation and Performance Recovery Following Eccentric Exercise-Induced Muscle Damage. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007, 292, 2168–2173. [Google Scholar] [CrossRef] [PubMed]
- Mosser, D.M.; Zhang, X. Interleukin-10: New Perspectives on an Old Cytokine. Immunol. Rev. 2008, 226, 205–218. [Google Scholar] [CrossRef] [PubMed]
- Mollazadeh, H.; Cicero, A.F.G.; Blesso, C.N.; Pirro, M.; Majeed, M.; Sahebkar, A. Immune Modulation by Curcumin: The Role of Interleukin-10. Crit. Rev. Food Sci. Nutr. 2019, 59, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Kocaadam, B.; Şanlier, N. Curcumin, an Active Component of Turmeric (Curcuma longa), and Its Effects on Health. Crit. Rev. Food Sci. Nutr. 2017, 57, 2889–2895. [Google Scholar] [CrossRef]
- Hargis, A.M.; Myers, S. The Integument. In Pathologic Basis of Veterinary Disease; Mosby: Maryland Heights, MI, USA, 2017; pp. 1009–1146.e1. [Google Scholar] [CrossRef]
- Nogueira, B.C.F.; Campos, A.K.; Alves, R.S.; de Cássia Vieira Faria, R.; Sarandy, M.M.; Fonseca e Silva, F.; Gonçalves, R.V. Oxidative and Local Histopathological Response on Skin Wound of Horses Due to Amblyomma Sculptum Tick Parasitism. Res. Vet. Sci. 2021, 136, 550–560. [Google Scholar] [CrossRef]
- Kostricky, I.M.; Basso, R.D.B.; Dos Santos, A.B.; Sulzbacher, M.M.; Goettems-Fiorin, P.B.; Frizzo, M.N.; Ludwig, M.S.; Heck, T.G. Effects of Water Temperature on Blood Lactate Concentration in Swimming Exercise Rat Model. J. Exerc. Physiol. Online 2021, 24, 13–21. [Google Scholar]
- Kantanista, A.; Kusy, K.; Zarębska, E.; Włodarczyk, M.; Ciekot-Sołtysiak, M.; Zieliński, J. Blood Ammonia and Lactate Responses to Incremental Exercise in Highly-Trained Male Sprinters and Triathletes. Biomed. Hum. Kinet. 2016, 8, 32–38. [Google Scholar] [CrossRef]
| Ingredients | g/kg of Diet | ||
|---|---|---|---|
| AIN-93M | WPC + CCM | CCM | |
| Cornstarch | 455.7 | 455.7 | 455.7 |
| Albumin 1 | 150.0 | 105.0 | 150.0 |
| Maltodextrin | 155.0 | 155.0 | 155.0 |
| Sucrose | 100.0 | 99.0 | 99.0 |
| Soybean oil | 40.0 | 40.0 | 40.0 |
| Cellulose | 50.0 | 50.0 | 50.0 |
| Mineral blend | 35.0 | 35.0 | 35.0 |
| Vitamin blend | 10.0 | 10.0 | 10.0 |
| L-cystine | 1.8 | 1.8 | 1.8 |
| Choline bitartrate | 2.5 | 2.5 | 2.5 |
| WPC 2 | - | 44.0 | - |
| Theracurmin® | - | 1.2 | 1.2 |
| Genes | Oligonucleotides (5′–3′) | |
|---|---|---|
| Forward | Reverse | |
| GAPDH | CCCCCAATGTATCCGTTGTG | TAGCCCAGGATGCCCTTTAGT |
| IL-6 | TCCTACCCCAACTTCCAATGCTC | TTGGATGGTCTTGGTCCTTAGCC |
| IL-10 | TTGAACCACCCGGCATCTAC | CCAAGGAGTTGTTCCCGTTA |
| TNF-α | TGGGCTACGGGCTTGTCACTC | GGGGGCCACCACGCTCTT |
| Biomarkers | CV (%) AIN-93M | CV (%) AIN-93M ET | CV (%) WPC + CCM | CV (%) WPC + CCM ET | CV (%) CCM | CV (%) CCM ET |
|---|---|---|---|---|---|---|
| SOD | 2.8 | 19 | 2.4 | 9.4 | 16.6 | 15.2 |
| CAT | 7.9 | 15.7 | 15 | 4.8 | 3.5 | 5 |
| GST | 21.2 | 21.7 | 15.6 | 41.5 | 21.6 | 15.9 |
| MDA | 33.7 | 18.3 | 11.2 | 12.9 | 22.3 | 19.5 |
| PCN | 14.2 | 15.8 | 13.7 | 23.4 | 25.5 | 42.8 |
| NO | 11.2 | 8.5 | 17.2 | 21.8 | 12.1 | 9.8 |
| IL-6 | - | 2.4 | 13.9 | 69 | 11.1 | 26.5 |
| TNF | - | 16.2 | 12.3 | 58.8 | 37.9 | 28.9 |
| IL-10 | - | 75.7 | 23.9 | 54.5 | 0.3 | 30.7 |
| TNF (CBA) | 48.6 | 9.2 | 44.5 | 18.7 | 8.8 | 25.2 |
| Blood lactate | - | 20.37 | - | 16.7 | - | 41.9 |
| Swimming time | - | 37.9 | - | 41.8 | - | 30 |
| Histopathology: Inflammation | 14.4 | 22.2 | 14.6 | 8.1 | 14.3 | 28.3 |
| Point on muscle fiber | 2.4 | 2.3 | 1.8 | 1.9 | 2 | 1.9 |
| Point on connective tissue | 13.6 | 16.4 | 7.8 | 8.2 | 18.3 | 9.6 |
| Muscle fiber area | 5.1 | 2.7 | 14.4 | 7.1 | 12.5 | 4.3 |
| Muscle fiber diameter | 3.5 | 4.8 | 0.2 | 4.4 | 9.2 | 0.1 |
| Groups | Inflammation (%) | Count a Point on Muscle Fiber | Count a Point on Connective Tissue |
|---|---|---|---|
| AIN-93M | 3.69 ± 0.53 a | 243.38 ± 5.92 b | 43.01 ± 5.88 a |
| WPC + CCM | 2.03 ± 0.45 b | 254.00 ± 5.72 a | 33.70 ± 5.52 b |
| CCM | 1.92 ± 0.28 b | 258.60 ± 4.59 a | 24.60 ± 1.93 c |
| AIN-93M ET | 3.59 ± 0.29 aª | 236.80 ± 4.43 b | 39.31 ± 3.23 a |
| WPC + CCM ET | 2.02 ± 0.29 b | 259.15 ± 5.08 a | 26.65 ± 4.88 b |
| CCM ET | 1.73 ± 0.49 b | 258.06 ± 4.88 a | 26.46 ± 2.55 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dias, K.A.; da Conceição, A.R.; Pereira, S.M.S.; Oliveira, L.A.; Rodrigues, J.S.; Dias, R.S.; de Paula, S.O.; Natali, A.J.; da Matta, S.L.P.; Gonçalves, R.V.; et al. Curcumin-Added Whey Protein Positively Modulates Skeletal Muscle Inflammation and Oxidative Damage after Exhaustive Exercise. Nutrients 2022, 14, 4905. https://doi.org/10.3390/nu14224905
Dias KA, da Conceição AR, Pereira SMS, Oliveira LA, Rodrigues JS, Dias RS, de Paula SO, Natali AJ, da Matta SLP, Gonçalves RV, et al. Curcumin-Added Whey Protein Positively Modulates Skeletal Muscle Inflammation and Oxidative Damage after Exhaustive Exercise. Nutrients. 2022; 14(22):4905. https://doi.org/10.3390/nu14224905
Chicago/Turabian StyleDias, Kelly Aparecida, Aline Rosignoli da Conceição, Stephanie Michelin Santana Pereira, Lívya Alves Oliveira, Jojo Silva Rodrigues, Roberto Sousa Dias, Sérgio Oliveira de Paula, Antônio José Natali, Sérgio Luis Pinto da Matta, Reggiani Vilela Gonçalves, and et al. 2022. "Curcumin-Added Whey Protein Positively Modulates Skeletal Muscle Inflammation and Oxidative Damage after Exhaustive Exercise" Nutrients 14, no. 22: 4905. https://doi.org/10.3390/nu14224905
APA StyleDias, K. A., da Conceição, A. R., Pereira, S. M. S., Oliveira, L. A., Rodrigues, J. S., Dias, R. S., de Paula, S. O., Natali, A. J., da Matta, S. L. P., Gonçalves, R. V., Tako, E., Martino, H. S. D., & Lucia, C. M. D. (2022). Curcumin-Added Whey Protein Positively Modulates Skeletal Muscle Inflammation and Oxidative Damage after Exhaustive Exercise. Nutrients, 14(22), 4905. https://doi.org/10.3390/nu14224905








