The Importance of Nutritional Aspects in the Assessment of Inflammation and Intestinal Barrier in Patients with Inflammatory Bowel Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Nutritional Questionnaire
2.3. Biochemical Analysis of Selected Parameters in Serum and Fecal Samples
2.4. Determination of Organic Acid Levels in Fecal Samples
2.5. Evaluation of Intestinal Biopsies for mRNA Expression of Selected Parameters Using Real-Time Polymerase Chain Reaction
2.6. Statistical Analysis
3. Results
3.1. Characteristics of Study Participants
3.2. Nutritional Status
3.3. Fecal Organic Acid Concentrations
3.4. Serum Cytokine and Zonulin Concentrations
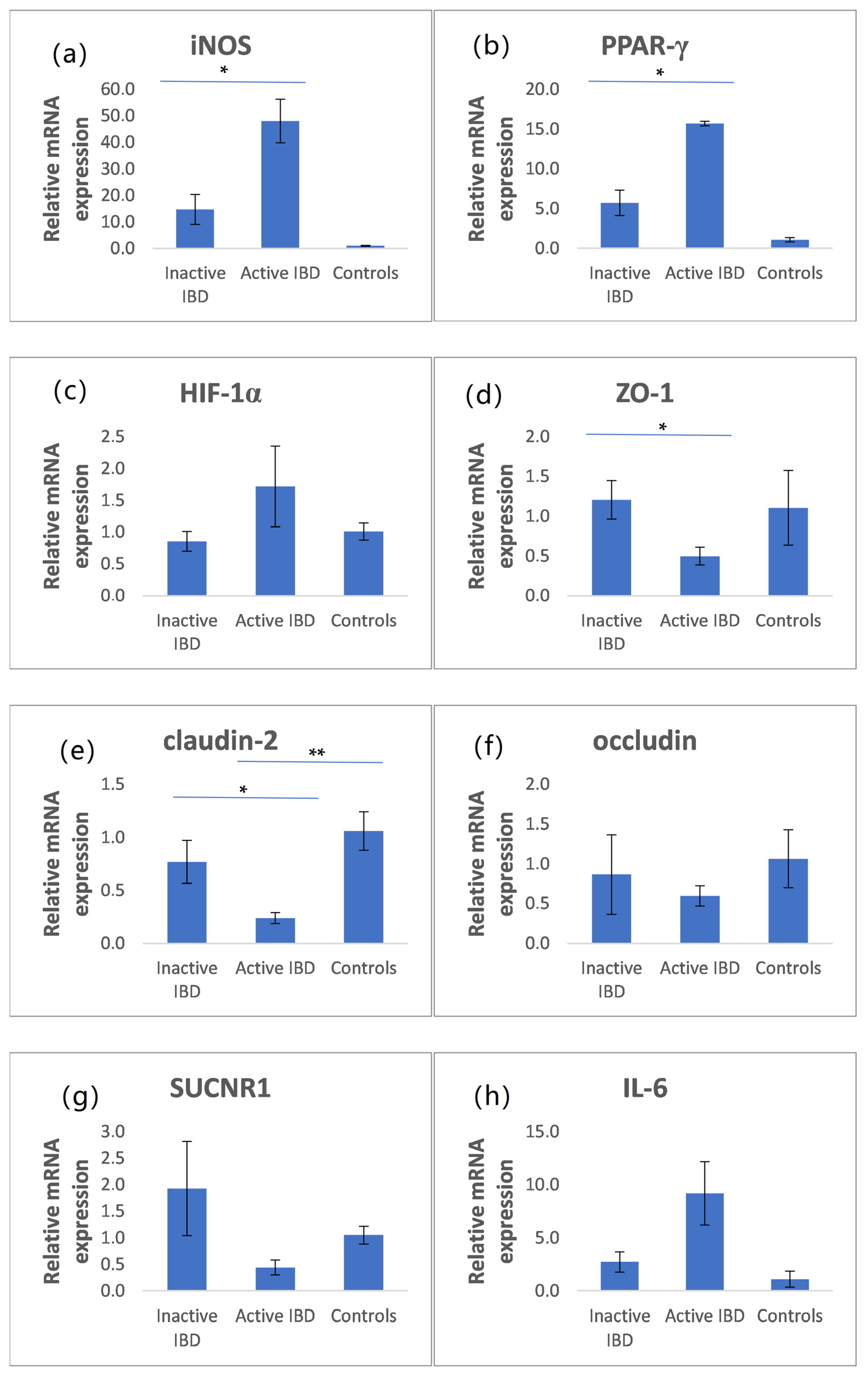
3.5. The mRNA Expression of iNOS, PPAR-γ, HIF-1α, ZO-1, Claudin-2, Occludin, SUCNR1, and IL-6 in Intestinal Biopsies
3.6. Investigation of Possible Gender Bias for the Studied Population
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, Y.Z.; Li, Y.Y. Inflammatory bowel disease: Pathogenesis. World J. Gastroenterol. 2014, 20, 91–99. [Google Scholar] [CrossRef]
- Neurath, M.F. Cytokines in inflammatory bowel disease. Nat. Rev. Immunol. 2014, 14, 329–342. [Google Scholar] [CrossRef]
- Sanchez-Muñoz, F.; Dominguez-Lopez, A.; Yamamoto-Furusho, J.K. Role of cytokines in inflammatory bowel disease. World J. Gastroenterol. 2008, 14, 4280–4288. [Google Scholar] [CrossRef]
- Antoni, L.; Nuding, S.; Wehkamp, J.; Stange, E.F. Intestinal barrier in inflammatory bowel disease. World J. Gastroenterol. 2014, 20, 1165–1179. [Google Scholar] [CrossRef]
- Lee, S.H. Intestinal Permeability Regulation by Tight Junction: Implication on Inflammatory Bowel Diseases. Intest. Res. 2015, 13, 11–18. [Google Scholar] [CrossRef]
- Landy, J.; Ronde, E.; English, N.; Clark, S.K.; Hart, A.L.; Knight, S.C.; Ciclitira, P.J.; Al-Hassi, H.O. Tight junctions in inflammatory bowel diseases and inflammatory bowel disease associated colorectal cancer. World J. Gastroenterol. 2016, 22, 3117–3126. [Google Scholar] [CrossRef]
- Liu, H.; Wang, J.; He, T.; Becker, S.; Zhang, G.; Li, D.; Ma, X. Butyrate: A double-edged sword for health? Adv. Nutr. 2018, 9, 21–29. [Google Scholar] [CrossRef]
- Yan, H.; Ajuwon, K.M. Butyrate modifies intestinal barrier function in IPEC-J2 cells through a selective upregulation of tight junction proteins and activation of the Akt signaling pathway. PLoS ONE 2017, 12, e0179586. [Google Scholar] [CrossRef]
- Byndloss, M.X.; Olsan, E.E.; Rivera-Chávez, F.; Tiffany, C.R.; Cevallos, S.A.; Lokken, K.L.; Torres, T.P.; Byndloss, A.J.; Faber, F.; Gao, Y.; et al. Microbiota-activated PPAR-γ signaling inhibits dysbiotic Enterobacteriaceae expansion. Science 2017, 357, 570–575. [Google Scholar] [CrossRef]
- Cohen, A.B.; Lee, D.; Long, M.D.; Kappelman, M.D.; Martin, C.F.; Sandler, R.S.; Lewis, J.D. Dietary patterns and self-reported associations of diet with symptoms of inflammatory bowel disease. Dig. Dis. Sci. 2013, 58, 1322–1328. [Google Scholar] [CrossRef]
- Fritsch, J.; Garces, L.; Quintero, M.A.; Pignac-Kobinger, J.; Santander, A.M.; Fernández, I.; Ban, Y.J.; Kwon, D.; Phillips, M.C.; Knight, K.; et al. Low-Fat, High-Fiber Diet Reduces Markers of Inflammation and Dysbiosis and Improves Quality of Life in Patients With Ulcerative Colitis. Clin. Gastroenterol. Hepatol. 2021, 19, 1189–1199.e30. [Google Scholar] [CrossRef]
- Desai, M.S.; Seekatz, A.M.; Koropatkin, N.M.; Kamada, N.; Hickey, C.A.; Wolter, M.; Pudlo, N.A.; Kitamoto, S.; Terrapon, N.; Muller, A.; et al. A Dietary Fiber-Deprived Gut Microbiota Degrades the Colonic Mucus Barrier and Enhances Pathogen Susceptibility. Cell 2016, 167, 1339–1353.e21. [Google Scholar] [CrossRef]
- Macias-Ceja, D.C.; Ortiz-Masiá, D.; Salvador, P.; Gisbert-Ferrándiz, L.; Hernández, C.; Hausmann, M.; Rogler, G.; Esplugues, J.V.; Hinojosa, J.; Alós, R.; et al. Succinate receptor mediates intestinal inflammation and fibrosis. Mucosal Immunol. 2019, 12, 178–187. [Google Scholar] [CrossRef]
- Scaldaferri, F.; Pizzoferrato, M.; Lopetuso, L.R.; Musca, T.; Ingravalle, F.; Sicignano, L.L.; Mentella, M.; Miggiano, G.; Mele, M.C.; Gaetani, E.; et al. Nutrition and IBD: Malnutrition and/or Sarcopenia? A Practical Guide. Gastroenterol. Res. Pract. 2017, 2017, 8646495. [Google Scholar] [CrossRef]
- Popkin, B.M.; Corvalan, C.; Grummer-Strawn, L.M. Dynamics of the double burden of malnutrition and the changing nutrition reality. Lancet 2020, 395, 65–74. [Google Scholar] [CrossRef]
- Rahmani, J.; Kord-Varkaneh, H.; Hekmatdoost, A.; Thompson, J.; Clark, C.; Salehisahlabadi, A.; Day, A.S.; Jacobson, K. Body mass index and risk of inflammatory bowel disease: A systematic review and dose-response meta-analysis of cohort studies of over a million participants. Obes. Rev. 2019, 20, 1312–1320. [Google Scholar] [CrossRef]
- Singh, S.; Dulai, P.S.; Zarrinpar, A.; Ramamoorthy, S.; Sandborn, W.J. Obesity in IBD: Epidemiology, pathogenesis, disease course and treatment outcomes. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 110–121. [Google Scholar] [CrossRef]
- Maaser, C.; Sturm, A.; Vavricka, S.R.; Kucharzik, T.; Fiorino, G.; Annese, V.; Calabrese, E.; Baumgart, D.C.; Bettenworth, D.; Borralho Nunes, P.; et al. ECCO-ESGAR Guideline for Diagnostic Assessment in IBD Part 1: Initial diagnosis, monitoring of known IBD, detection of complications. J. Crohn’s Colitis 2019, 13, 144–164. [Google Scholar] [CrossRef]
- Dobrowolska-Iwanek, J.; Zagrodzki, P.; Woźniakiewicz, M.; Woźniakiewicz, A.; Zwolińska -Wcisło, M.; Winnicka, D.; Paśko, P. Procedure optimization for extracting short-chain fatty acids from human faeces. J. Pharm. Biomed. Anal. 2016, 124, 337–340. [Google Scholar] [CrossRef]
- Kaczmarczyk, O.; Dąbek-Drobny, A.; Woźniakiewicz, M.; Paśko, P.; Dobrowolska-Iwanek, J.; Woźniakiewicz, A.; Piątek-Guziewicz, A.; Zagrodzki, P.; Mach, T.; Zwolińska-Wcisło, M. Fecal levels of lactic, succinic and short-chain fatty acids in patients with ulcerative colitis and crohn disease: A pilot study. J. Clin. Med. 2021, 10, 4701. [Google Scholar] [CrossRef]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative CT method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Gold, S.L.; Rabinowitz, L.G.; Manning, L.; Keefer, L.; Rivera-Carrero, W.; Stanley, S.; Sherman, A.; Castillo, A.; Tse, S.; Hyne, A.; et al. High Prevalence of Malnutrition and Micronutrient Deficiencies in Patients With Inflammatory Bowel Disease Early in Disease Course. Inflamm. Bowel Dis. 2022, 1–7, online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Lloyd-Price, J.; Arze, C.; Ananthakrishnan, A.N.; Schirmer, M.; Avila-Pacheco, J.; Poon, T.W.; Andrews, E.; Ajami, N.J.; Bonham, K.S.; Brislawn, C.J.; et al. Multi-omics of the gut microbial ecosystem in inflammatory bowel diseases. Nature 2019, 569, 655–662. [Google Scholar] [CrossRef]
- Kern, L.; Mittenbühler, M.J.; Vesting, A.J.; Ostermann, A.L.; Wunderlich, C.M.; Wunderlich, F.T. Obesity-induced TNFα and IL-6 signaling: The missing link between obesity and inflammation- driven liver and colorectal cancers. Cancers 2019, 11, 24. [Google Scholar] [CrossRef]
- Singer, I.I.; Kawka, D.W.; Scott, S.; Weidner, J.R.; Mumford, R.A.; Riehl, T.E.; Stenson, W.F. Expression of inducible nitric oxide synthase and nitrotyrosine in colonic epithelium in inflammatory bowel disease. Gastroenterology 1996, 111, 871–885. [Google Scholar] [CrossRef]
- Liu, T.; Li, J.; Liu, Y.; Xiao, N.; Suo, H.; Xie, K.; Yang, C.; Wu, C. Short-Chain fatty acids suppress lipopolysaccharide-Induced production of nitric oxide and proinflammatory cytokines through inhibition of NF-?B Pathway in RAW264.7 cells. Inflammation 2012, 35, 1676–1684. [Google Scholar] [CrossRef]
- Park, J.S.; Lee, E.J.; Lee, J.C.; Kim, W.K.; Kim, H.S. Anti-inflammatory effects of short chain fatty acids in IFN-γ-stimulated RAW 264.7 murine macrophage cells: Involvement of NF-κB and ERK signaling pathways. Int. Immunopharmacol. 2007, 7, 70–77. [Google Scholar] [CrossRef]
- Caioni, G.; Viscido, A.; D’angelo, M.; Panella, G.; Castelli, V.; Merola, C.; Frieri, G.; Latella, G.; Cimini, A.; Benedetti, E. Inflammatory bowel disease: New insights into the interplay between environmental factors and pparγ. Int. J. Mol. Sci. 2021, 22, 985. [Google Scholar] [CrossRef]
- Ricote, M.; Huang, J.T.; Welch, J.S.; Glass, C.K. The peroxisome proliferator-activated receptorγ (PPARγ) as a regulator of monocyte/macrophage function. J. Leukoc. Biol. 1999, 66, 733–739. [Google Scholar] [CrossRef]
- Ferré, P. The Biology of Peroxisome Proliferator-Activated Receptors. Diabetes 2004, 53, S43–S50. [Google Scholar] [CrossRef]
- Marion-Letellier, R.; Déchelotte, P.; Lacucci, M.; Ghosh, S. Dietary modulation of peroxisome proliferator-activated receptor gamma. Gut 2009, 58, 586–593. [Google Scholar] [CrossRef] [PubMed]
- Zheng, F.; Cai, Y. Concurrent exercise improves insulin resistance and nonalcoholic fatty liver disease by upregulating PPAR-γ and genes involved in the beta-oxidation of fatty acids in ApoE-KO mice fed a high-fat diet 11 Medical and Health Sciences 1103 Clinical Sciences. Lipids Health Dis. 2019, 18, 6. [Google Scholar] [CrossRef] [PubMed]
- Dubuquoy, L.; Rousseaux, C.; Thuru, X.; Peyrin-Biroulet, L.; Romano, O.; Chavatte, P.; Chamaillard, M.; Desreumaux, P. PPARγ as a new therapeutic target in inflammatory bowel diseases. Gut 2006, 55, 1341–1349. [Google Scholar] [CrossRef]
- Yamamoto-Furusho, J.K.; Peñaloza-Coronel, A.; Sánchez-Muñoz, F.; Barreto-Zuñiga, R.; Dominguez-Lopez, A. Peroxisome proliferator-activated receptor-gamma (PPAR-γ) expression is downregulated in patients with active ulcerative colitis. Inflamm. Bowel Dis. 2011, 17, 680–681. [Google Scholar] [CrossRef] [PubMed]
- Decara, J.; Rivera, P.; López-Gambero, A.J.; Serrano, A.; Pavón, F.J.; Baixeras, E.; Rodríguez de Fonseca, F.; Suárez, J. Peroxisome Proliferator-Activated Receptors: Experimental Targeting for the Treatment of Inflammatory Bowel Diseases. Front. Pharmacol. 2020, 11, 730. [Google Scholar] [CrossRef]
- Scharl, M.; Paul, G.; Barrett, K.E.; McCole, D.F. AMP-activated protein kinase mediates the interferon-γ-induced decrease in intestinal epithelial barrier function. J. Biol. Chem. 2009, 284, 27952–27963. [Google Scholar] [CrossRef]
- Zeissig, S.; Bürgel, N.; Günzel, D.; Richter, J.; Mankertz, J.; Wahnschaffe, U.; Kroesen, A.J.; Zeitz, M.; Fromm, M.; Schulzke, J.D. Changes in expression and distribution of claudin 2, 5 and 8 lead to discontinuous tight junctions and barrier dysfunction in active Crohn’s disease. Gut 2007, 56, 61–72. [Google Scholar] [CrossRef]
- Weber, C.R.; Nalle, S.C.; Tretiakova, M.; Rubin, D.T.; Turner, J.R. Claudin-1 and claudin-2 expression is elevated in inflammatory bowel disease and may contribute to early neoplastic transformation. Lab. Investig. 2008, 88, 1110–1120. [Google Scholar] [CrossRef]
- Mazzon, E.; Cuzzocrea, S. Absence of functional peroxisome proliferator-activated receptor-α enhanced ileum permeability during experimental colitis. Shock 2007, 28, 192–201. [Google Scholar] [CrossRef]
- Miao, W.; Wu, X.; Wang, K.; Wang, W.; Wang, Y.; Li, Z.; Liu, J.; Li, L.; Peng, L. Sodium butyrate promotes reassembly of tight junctions in Caco-2 monolayers involving inhibition of MLCK/MLC2 pathway and phosphorylation of PKCβ2. Int. J. Mol. Sci. 2016, 17, 1696. [Google Scholar] [CrossRef]
- Connors, J.; Dawe, N.; Van Limbergen, J. The role of succinate in the regulation of intestinal inflammation. Nutrients 2019, 11, 25. [Google Scholar] [CrossRef] [PubMed]
- Fremder, M.; Kim, S.W.; Khamaysi, A.; Shimshilashvili, L.; Eini-Rider, H.; Park, I.S.; Hadad, U.; Cheon, J.H.; Ohana, E. A transepithelial pathway delivers succinate to macrophages, thus perpetuating their pro-inflammatory metabolic state. Cell Rep. 2021, 36, 109521. [Google Scholar] [CrossRef] [PubMed]
- Osaka, T.; Moriyama, E.; Arai, S.; Date, Y.; Yagi, J.; Kikuchi, J.; Tsuneda, S. Meta-analysis of fecal microbiota and metabolites in experimental colitic mice during the inflammatory and healing phases. Nutrients 2017, 9, 1329. [Google Scholar] [CrossRef] [PubMed]
- Maslowski, K.M.; MacKay, C.R. Diet, gut microbiota and immune responses. Nat. Immunol. 2011, 12, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Harber, K.J.; de Goede, K.E.; Verberk, S.G.S.; Meinster, E.; de Vries, H.E.; van Weeghel, M.; de Winther, M.P.J.; Van den Bossche, J. Succinate is an inflammation-induced immunoregulatory metabolite in macrophages. Metabolites 2020, 10, 372. [Google Scholar] [CrossRef] [PubMed]
- Keiran, N.; Ceperuelo-Mallafré, V.; Calvo, E.; Hernández-Alvarez, M.I.; Ejarque, M.; Núñez-Roa, C.; Horrillo, D.; Maymó-Masip, E.; Rodríguez, M.M.; Fradera, R.; et al. SUCNR1 controls an anti-inflammatory program in macrophages to regulate the metabolic response to obesity. Nat. Immunol. 2019, 20, 581–592. [Google Scholar] [CrossRef]
- Kaczmarczyk, O.; Dąbek-Drobny, A.; Woźniakiewicz, M.; Paśko, P.; Dobrowolska-Iwanek, J.; Woźniakiewicz, A.; Targosz, A.; Ptak-Belowska, A.; Piątek-Guziewicz, A.; Zagrodzki, P.; et al. Association between fecal levels of Short-Chain Fatty Acids and serum Pro and Anti-Inflammatory Cytokines in patients with Inflammatory Bowel Disease. Folia Med. Cracov. 2022, 62, 43–55. [Google Scholar]
- Sideri, A.; Stavrakis, D.; Bowe, C.; Shih, D.Q.; Fleshner, P.; Arsenescu, V.; Arsenescu, R.; Turner, J.R.; Pothoulakis, C.; Karagiannides, I. Effects of obesity on severity of colitis and cytokine expression in mouse mesenteric fat. Potential role of adiponectin receptor 1. Am. J. Physiol. Gastrointest. Liver Physiol. 2015, 308, G591–G604. [Google Scholar] [CrossRef]
- Rinninella, E.; Cintoni, M.; Raoul, P.; Lopetuso, L.R.; Scaldaferri, F.; Pulcini, G.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. Food Components and Dietary Habits: Keys for a Healthy Gut Microbiota Composition. Nutrients 2019, 11, 2393. [Google Scholar] [CrossRef]
- Chang, P.V.; Hao, L.; Offermanns, S.; Medzhitov, R. The microbial metabolite butyrate regulates intestinal macrophage function via histone deacetylase inhibition. Proc. Natl. Acad. Sci. USA 2014, 111, 2247–2252. [Google Scholar] [CrossRef]
- Wang, J.J.; Zhang, Q.M.; Ni, W.W.; Zhang, X.; Li, Y.; Li, A.L.; Du, P.; Li, C.; Yu, S.S. Modulatory effect of Lactobacillus acidophilus KLDS 1.0738 on intestinal short-chain fatty acids metabolism and GPR41/43 expression in β-lactoglobulin–sensitized mice. Microbiol. Immunol. 2019, 63, 303–315. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.; Shu, D.; Zheng, M.; Wang, J.; Luo, C.; Wang, Y.; Guo, F.; Zou, X.; Lv, X.; Li, Y.; et al. Microbial metabolite butyrate facilitates M2 macrophage polarization and function. Sci. Rep. 2016, 6, 24838. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.Y.; De Zoete, M.R.; Van Putten, J.P.M.; Strijbis, K. Redirection of epithelial immune responses by short-chain fatty acids through inhibition of histone deacetylases. Front. Immunol. 2015, 6, 554. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Inactive IBD n = 11 | Active IBD n = 18 | Controls n = 6 | p Value |
|---|---|---|---|---|
| sex, female/male, n | 2/9 | 6/12 | 4/2 | - |
| age, years, mean ± SD (min–max) | 39.8 ± 16.5 (25–71) | 34.1 ± 11.7 (20–61) | 43.8 ± 22.5 (20–77) | 0.354 |
| BMI kg/m2, mean ± SD (min–max) | 21.1 ± 3.0 (16.8–26.2) | 23.2 ± 5.3 (15.7–37.1) | 24.2 ± 4.5 (20.5–32.9) | 0.363 |
| hemoglobin, g/dL, mean ± SD (min–max) | 13.8 ± 1.8 (10.4–15.7) | 12.6 ± 2.4 (7.3–16.7) | 14.1 ± 2.5 (10.2–16.9) | 0.148 |
| WBC ± SD, g/L, mean ± SD (min–max) | 8.0 ± 5.1 (3.7–19.1) | 9.7 ± 3.0 (5.5–15.7) | 7.9 ± 4.2 (4.0–10.0) | 0.431 |
| platelets, 103/μL, mean ± SD (min–max) | 293.1 ± 157.1 (90.5–542.0) | 375.8 ± 121.7 (150.0–657.0) | 224.3 ± 82.0 (160.0–343.0) | 0.040 * |
| CRP, mg/L, median (min–max) | 1.0 (1.0–57.4) | 9.4 a (1.0–154.0) | 1.0 a (1.0–6.0) | 0.013 ** |
| calprotectin, µg/g, median (min–max) | 100.0 (0–148.0) | 1541.0 b (96.0–2251.0) | <LOD b (<LOD-120.0) | 0.007 ** |
| albumin, g/L, mean ± SD (min–max) | 42.8 ± 4.7 (34.1–50.3) | 37.9 ± 5.9 (26.4–45.0) | 44.3 ± 6.7 (32.0–49.0) | 0.044 * |
| Parameter | Inactive IBD n = 11 | Active IBD n = 18 |
|---|---|---|
| Loose stools (more than 3 per day) | 3 (27.3) | 16 (88.9) |
| Abdominal pain (moderate or severe) | 3 (27.3) | 11 (61.1) |
| Anemia | 2 (18.2) | 8 (44.5) |
| Frequency | Wholegrain Products | Dried Fruit | Raw Vegetables | Legumes |
|---|---|---|---|---|
| everyday | 1 (5) | 0 | 1 (5) | 0 |
| several times/week | 3 (17) | 0 | 4 (23) | 0 |
| several times/month | 2 (10) | 7 (39) | 9 (50) | 7 (39) |
| never | 12 (78) | 11 (61) | 4 (22) | 11 (61) |
| Frequency | Wholegrain Products | Dried Fruits | Raw Vegetables | Legumes |
|---|---|---|---|---|
| everyday | 1 (9) | 0 | 1 (9) | 0 |
| several times/week | 2 (18) | 1 (9) | 0 | 0 |
| several times/month | 0 | 2 (18) | 4 (36) | 4 (36) |
| never | 8 (73) | 8 (73) | 6 (55) | 7 (64) |
| Frequency | Wholegrain Products | Dried Fruit | Raw Vegetables | Legumes |
|---|---|---|---|---|
| everyday | 1 (17) | 0 | 1 (17) | 0 |
| several times/week | 0 | 0 | 2 (34) | 2 (34) |
| several times/month | 5 (83) | 2 (34) | 3 (49) | 1 (17) |
| never | 0 | 4 (66) | 0 | 3 (49) |
| Organic Acid, µg/g | Inactive IBD n = 10 1 | Active IBD n = 18 | Controls n = 5 1 | p Value |
|---|---|---|---|---|
| succinic | 408.5 (170.4; 886.2) | 208.0 (125.3; 580.9) | 197.2 (91.9; 778.6) | 0.466 |
| acetic | 1171.5 (906.3; 1339.6) | 716.4 (552.4; 1065.1) | 940.4 (770.4; 1834.9) | 0.114 |
| lactic | 636.1 (44.2; 871.7) | 885.3 (349.0; 2408.7) | 74.5 (24.8; 1405.1) | 0.313 |
| propionic | 512.1 (214.4; 920.6) | 247.2 (85.9; 556.9) | 322.7 (117.0; 417.2) | 0.279 |
| butyric | 297.3 (35.3; 500.9) | 57 (<LOD; 215.0) | 118 (92.5; 227.0) | 0.234 |
| isobutyric | 62.6 (<LOD; 122.3) | 38.3 (<LOD; 65.4) | 36.9 (32.4; 45.1) | 0.518 |
| valeric | <LOD (<LOD; 38.0) | <LOD (<LOD; <LOD) | 21.3 (<LOD; 22.3) | 0.503 |
| isovaleric | 62.8 (<LOD; 232.4) | <LOD (<LOD; 49.9) | 77.4 (22.3–168.2) | 0.036 * |
| Parameter | Inactive IBD n = 10 1 | Active IBD n = 17 1 | Controls n = 5 1 | p Value |
|---|---|---|---|---|
| TNF-α, pg/mL | 7.1 (1.2; 28.4) | 5.8 (1.8; 10.8) | 2.0 (0.6; 3.0) | 0.187 |
| IL-17, pg/mL | 86 (70.1; 233.4) | 60.9 (49.3; 251.5) | 52 (40.8; 53.6) | 0.061 |
| IL-10, pg/mL | 28.5 (17.9; 53.5) | 20.9 (15.2; 58.1) | 14.2 (13.0; 17.0) | 0.079 |
| IL-22, pg/mL | 28.6 (24.2; 103.1) | 30.1 (15.4; 89.7) | 21.6 (19.4; 22.1) | 0.315 |
| IL-6, pg/mL | <LOD (<LOD; <LOD) | <LOD (<LOD; 4.2) | <LOD (<LOD; <LOD) | 0.198 |
| zonulin, pg/mL | 2.9 (1.6; 34.4) | 2 (1; 42.7) | 1.9 (1.4; 2.8) | 0.587 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaczmarczyk, O.; Dąbek-Drobny, A.; Piątek-Guziewicz, A.; Woźniakiewicz, M.; Paśko, P.; Dobrowolska-Iwanek, J.; Woźniakiewicz, A.; Targosz, A.; Ptak-Belowska, A.; Szczyrk, U.; et al. The Importance of Nutritional Aspects in the Assessment of Inflammation and Intestinal Barrier in Patients with Inflammatory Bowel Disease. Nutrients 2022, 14, 4622. https://doi.org/10.3390/nu14214622
Kaczmarczyk O, Dąbek-Drobny A, Piątek-Guziewicz A, Woźniakiewicz M, Paśko P, Dobrowolska-Iwanek J, Woźniakiewicz A, Targosz A, Ptak-Belowska A, Szczyrk U, et al. The Importance of Nutritional Aspects in the Assessment of Inflammation and Intestinal Barrier in Patients with Inflammatory Bowel Disease. Nutrients. 2022; 14(21):4622. https://doi.org/10.3390/nu14214622
Chicago/Turabian StyleKaczmarczyk, Olga, Agnieszka Dąbek-Drobny, Agnieszka Piątek-Guziewicz, Michał Woźniakiewicz, Paweł Paśko, Justyna Dobrowolska-Iwanek, Aneta Woźniakiewicz, Aneta Targosz, Agata Ptak-Belowska, Urszula Szczyrk, and et al. 2022. "The Importance of Nutritional Aspects in the Assessment of Inflammation and Intestinal Barrier in Patients with Inflammatory Bowel Disease" Nutrients 14, no. 21: 4622. https://doi.org/10.3390/nu14214622
APA StyleKaczmarczyk, O., Dąbek-Drobny, A., Piątek-Guziewicz, A., Woźniakiewicz, M., Paśko, P., Dobrowolska-Iwanek, J., Woźniakiewicz, A., Targosz, A., Ptak-Belowska, A., Szczyrk, U., Strzałka, M., Zagrodzki, P., & Zwolińska-Wcisło, M. (2022). The Importance of Nutritional Aspects in the Assessment of Inflammation and Intestinal Barrier in Patients with Inflammatory Bowel Disease. Nutrients, 14(21), 4622. https://doi.org/10.3390/nu14214622








