Effectiveness of a Per-Meal Protein Prescription and Nutrition Education with versus without Diet Coaching on Dietary Protein Intake and Muscle Health in Middle-Aged Women
Abstract
:1. Introduction
2. Materials and Methods
2.1. Assessments
2.2. Procedures
2.3. Statistical Analyses
3. Results
3.1. Anthropometric Findings
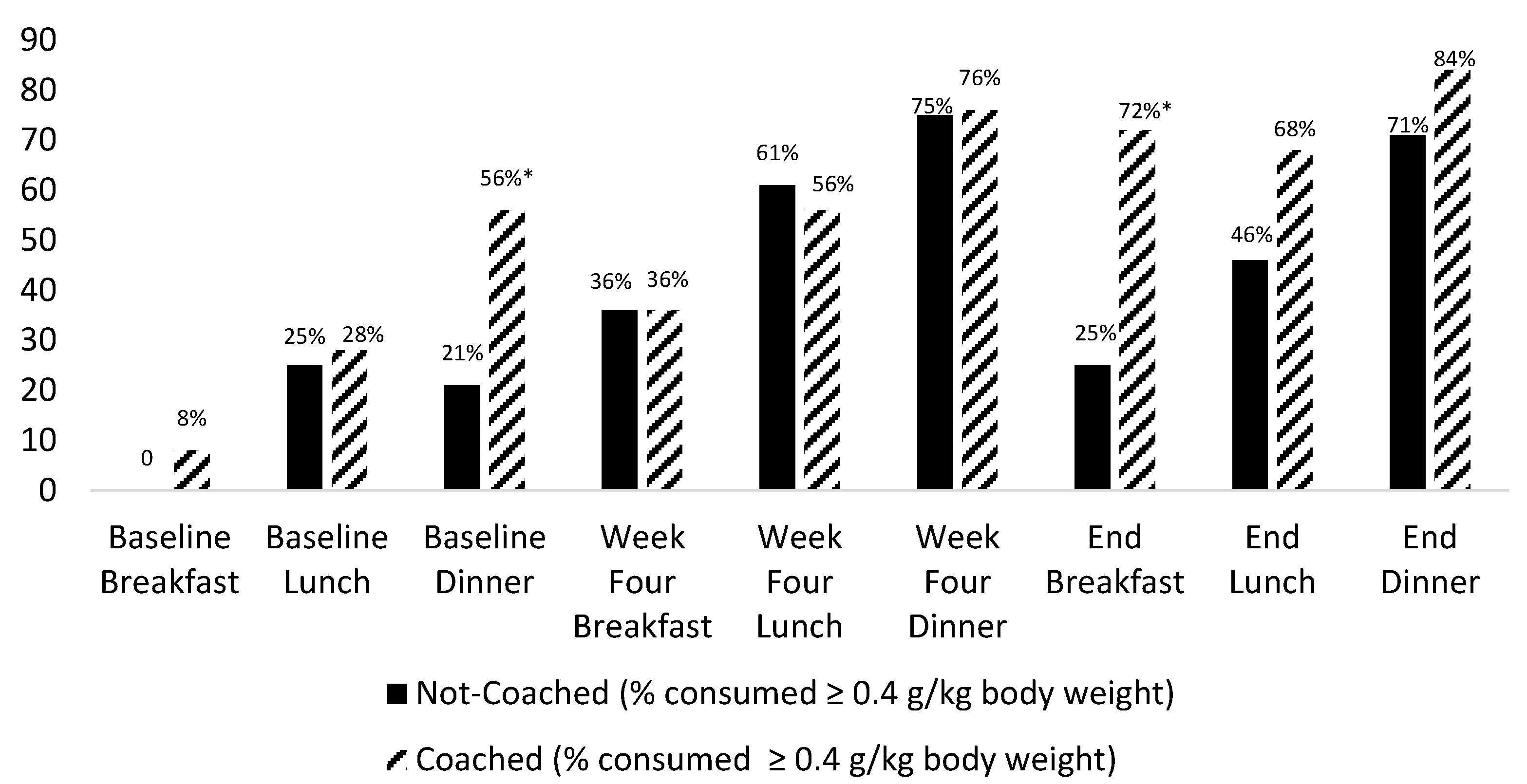
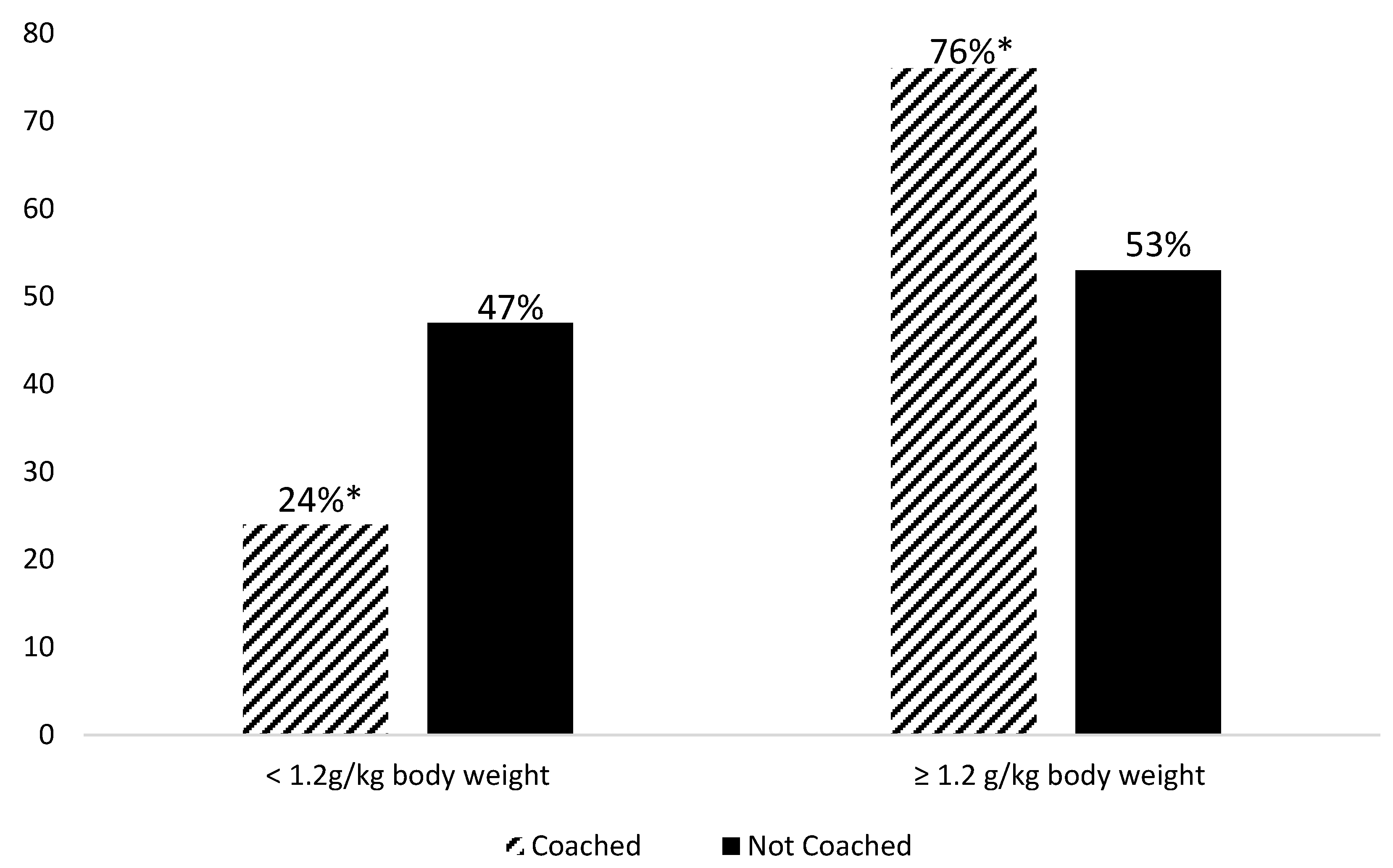
3.2. Dietary Intake Findings
3.3. Muscle Mass, Strength and Function Findings
3.4. Risk for Sarcopenia
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Berner, L.A.; Becker, G.; Wise, M.; Dot, J. Characterization of dietary protein among older adults in the United States: Amount, animal sources, and meal patterns. J. Acad. Nutr. Diet. 2013, 113, 809–815. [Google Scholar] [CrossRef] [PubMed]
- Gropper, S.S.; Tappen, R.M.; Vieira, E.R. Differences in nutritional and physical health indicators among older African Americans, European Americans, and Hispanic Americans. J. Nutr. Gerontol. Geriatr. 2019, 38, 205–217. [Google Scholar] [CrossRef] [PubMed]
- Gropper, S.S.; Exantus, M.; Jackson, K.L.; Spiers, S.M.; Viera, E.R.; D’Avolio, D.; Opalinski, A.; Tappen, R. Increasing protein intake to help older adults increase muscle strength and function: A pilot, single-arm investigation using coaching and a per-meal protein prescription. J. Aging Res. Lifestyle 2020, 9, 9–13. [Google Scholar] [CrossRef]
- Krok-Schoen, J.L.; Price, A.A.; Luo, M.; Kelly, O.J.; Taylor, C.A. Low dietary protein intakes and associated dietary patterns and functional limitations in an aging population: A NHANES analysis. J. Nutr. Health Aging 2019, 24, 338–347. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- USDA. Agricultural Research Service. Energy Intake: Percentages of Energy from Protein, Carbohydrate, Fat and Alcohol by Gender and Age, What We Eat in America, NHANES 2009–2010. 2012. Available online: http://www.ars.usda.gov/ba/bhnrc/fsrg (accessed on 1 November 2021).
- Food and Nutrition Board. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Protein and Amino Acids; National Academy Press: Washington, DC, USA, 2002. [Google Scholar]
- Beasley, J.M.; Rillamas-Sun, E.; Tinker, L.F.; Wylie-Rosett, J.; Mossavar-Rahmani, Y.; Datta, M.; Caan, B.J.; LaCroix, A.Z. Dietary intakes of women’s health initiative long life study participants falls short of the dietary reference intakes. J. Acad. Nutr. Diet. 2020, 120, 1530–1537. [Google Scholar] [CrossRef]
- Hengeveld, L.M.; Boer, J.M.; Gaudreau, P.; Heymans, M.W.; Jagger, C.; Mendonça, N.; Ocké, M.C.; Presse, N.; Sette, S.; Simonsick, E.M.; et al. Prevalence of protein intake below recommended in community-dwelling older adults: A meta-analysis across cohorts from the PROMISS consortium. J. Cachexia Sarcopenia Muscle 2020, 11, 1212–1222. [Google Scholar] [CrossRef]
- Bhasin, S.; Travison, T.G.; Manini, T.M.; Patel, S.; Pencina, K.M.; Fielding, R.A.; Magaziner, J.M.; Newman, A.B.; Kiel, D.P.; Cooper, C.; et al. Sarcopenia definition: The position statements of the sarcopenia definition and outcomes consortium. J. Am. Geriatr. Soc. 2020, 68, 1410–1418. [Google Scholar] [CrossRef] [PubMed]
- Borsheim, E.; Bui, Q.T.; Tissier, S.; Kobayashi, H.; Ferrando, A.A.; Wolfe, R.R. Effect of amino acid supplementation on muscle mass, strength and physical function in elderly. Clin. Nutr. 2008, 27, 189–195. [Google Scholar] [CrossRef] [Green Version]
- Bradlee, M.L.; Mustafa, J.; Singer, M.R.; Moore, L.L. High-protein foods and physical activity protect against age-related muscle loss and functional decline. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2018, 73, 88–94. [Google Scholar] [CrossRef]
- Chanet, A.; Verlaan, S.; Salles, J.; Giraudet, C.; Patrac, V.; Pidou, V.; Pouyet, C.; Hafnaoui, N.; Blot, A.; Cano, N.; et al. Supplementing breakfast with a vitamin D and leucine-rich whey protein medical nutrition drink enhances postprandial muscle protein synthesis and muscle mass in healthy older men. J. Nutr. 2017, 147, 2262–2271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coelho-Junior, H.; Marzetti, E.; Picca, A.; Cesari, M.; Uchida, M.C.; Calvani, R. Protein intake and frailty: A matter of quantity, quality and timing. Nutrients 2020, 12, 2915. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef] [Green Version]
- Dillon, E.L.; Sheffield-Moore, M.; Paddon-Jones, D.; Gilkison, C.; Sanford, A.P.; Casperson, S.L.; Jiang, J.; Chinkes, D.L.; Urban, R.J. Amino acid supplementation increases lean body mass, basal muscle protein synthesis, and insulin-like growth factor-1 expression in older women. J. Clin. Endocrinol. Metab. 2009, 94, 1630–1637. [Google Scholar] [CrossRef] [Green Version]
- Food and Nutrition Board. Recommended Dietary Allowances; National Research Council: Washington, DC, USA, 1943. [Google Scholar]
- Janssen, I.; Heymsfield, S.B.; Wang, Z.M.; Ross, R. Skeletal muscle mass and distribution in 468 men and women aged 18–88 yr. J. Appl. Physiol. 2000, 89, 81–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kirk, B.; Zanker, J.; Bani Hassan, E.; Bird, S.; Brennan-Olsen, S.; Duque, G. Sarcopenia definitions and outcomes consortium (SDOC) criteria are strongly associated with malnutrition, depression, falls, and fractures in high-risk older persons. J. Am. Med. Dir. Assoc. 2021, 22, 741–745. [Google Scholar] [CrossRef] [PubMed]
- Mamerow, M.M.; Mettler, J.A.; English, K.L.; Casperson, S.L.; Arentson-Lantz, E.; Sheffield-Moore, M.; Layman, D.K.; Paddon-Jones, D. Dietary protein distribution positively influences 24-h muscle protein synthesis in healthy adults. J. Nutr. 2014, 144, 876–880. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McGrath, R.; Stastny, S.; Casperson, S.; Jahns, L.; Roemmich, J.; Hackney, K.J. Daily protein intake and distribution of daily protein consumed decreases odds for functional disability in older Americans. J. Aging Health 2020, 32, 1075–1083. [Google Scholar] [CrossRef]
- Mitchell, C.J.; Milan, A.M.; Mitchell, S.M.; Zeng, N.; Ramzan, F.; Sharma, P.; Knowles, S.O.; Roy, N.C.; Sjödin, A.; Wagner, K.H.; et al. The effects of dietary protein intake on appendicular lean mass and muscle function in elderly men: A 10-week randomized controlled trial. Am. J. Clin. Nutr. 2017, 106, 1375–1388. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murphy, C.H.; Oikawa, S.Y.; Phillips, S.M. Dietary protein to maintain muscle mass in aging: A case for per-meal protein recommendations. J. Frailty Aging 2016, 5, 49–58. [Google Scholar] [CrossRef]
- Norton, C.; Toomey, C.; McCormack, W.G.; Francis, P.; Saunders, J.; Kerin, E.; Jakeman, P. Protein supplementation at breakfast and lunch for 24 weeks beyond habitual intakes increases whole-body lean tissue mass in healthy older adults. J. Nutr. 2016, 146, 65–69. [Google Scholar] [CrossRef] [Green Version]
- Wall, B.T.; Gorissen, S.H.; Pennings, B.; Koopman, R.; Groen, B.B.; Verdijk, L.B.; van Loon, L.J. Aging is accompanied by a blunted muscle protein synthetic response to protein ingestion. PLoS ONE 2015, 10, e014903. [Google Scholar]
- Bauer, J.; Biolo, G.; Cederholm, T.; Cesari, M.; Cruz-Jentoft, A.J.; Morley, J.E.; Phillips, S.; Sieber, C.; Stehle, P.; Teta, D.; et al. Evidence-based recommendations for optimal dietary protein intake in older people: A position paper from the PROT-AGE Study Group. J. Am. Med. Dir. Assoc. 2013, 14, 542–559. [Google Scholar] [CrossRef]
- Coelho-Júnior, H.J.; Milano-Teixeira, L.; Rodrigues, B.; Bacurau, R.; Marzetti, E.; Uchida, M. Relative protein intake and physical function in older adults: A systematic review and meta-analysis of observational studies. Nutrients 2018, 10, 1330. [Google Scholar] [CrossRef] [Green Version]
- Deutz, N.E.; Bauer, J.M.; Barazzoni, R.; Biolo, G.; Boirie, Y.; Bosy-Westphal, A.; Cederholm, T.; Cruz-Jentoft, A.; Krznariç, Z.; Nair, K.S.; et al. Protein intake and exercise for optimal muscle function with aging: Recommendations from the ESPEN Expert Group. Clin. Nutr. 2014, 33, 929–936. [Google Scholar] [CrossRef] [Green Version]
- Houston, D.K.; Nicklas, B.J.; Ding, J.; Harris, T.B.; Tylavsky, F.A.; Newman, A.B.; Lee, J.S.; Sahyoun, N.R.; Visser, M.; Kritchevsky, S.B. Dietary protein intake is associated with lean mass change in older, community-dwelling adults: The health, aging, and body composition (Health ABC) study. Am. J. Clin. Nutr. 2008, 87, 150–155. [Google Scholar] [CrossRef] [Green Version]
- Kim, I.Y.; Schutzler, S.; Schrader, A.; Spencer, H.; Kortebein, P.; Deutz, N.E.; Wolfe, R.R.; Ferrando, A.A. Quantity of dietary protein intake, but not pattern of intake, affects net protein balance primarily through differences in protein synthesis in older adults. Am. J. Physiol. Endocrin. Metab. 2015, 308, E21–E28. [Google Scholar] [CrossRef]
- Loenneke, J.P.; Loprinzi, P.D.; Murphy, C.H.; Phillips, S.M. Per meal dose and frequency of protein consumption is associated with lean mass and muscle performance. Clin. Nutr. 2016, 35, 1506–1511. [Google Scholar] [CrossRef]
- Cardon-Thomas, D.K.; Rieviere, T.; Tieges, Z.; Greig, C.A. Dietary protein in older adults: Adequate daily intake but potential for improved distribution. Nutrients 2017, 9, 184. [Google Scholar] [CrossRef] [PubMed]
- Farsijani, S.; Morais, J.A.; Payette, H.; Gaudreau, P.; Shatenstein, B.; Gray-Donald, K.; Chevalier, S. Relation between mealtime distribution of protein intake and lean mass loss in free-living older adults of the NuAge study. Am. J. Clin. Nutr. 2016, 104, 694–703. [Google Scholar] [CrossRef] [PubMed]
- Farsijani, S.; Payette, H.; Morais, J.A.; Shatenstein, B.; Gaudreau Chevalier, S. Even mealtime distribution of protein intake is associated with greater muscle strength, but not with 3-y physical function decline, in free-living older adults (NuAge study). Am. J. Clin. Nutr. 2017, 106, 113–124. [Google Scholar] [CrossRef] [PubMed]
- Smeuninx, B.; Greig, C.A.; Breen, L. Amount, source and pattern of dietary protein intake across the adult lifespan: A cross-sectional study. Front. Nutr. 2020, 7, 25. [Google Scholar] [CrossRef] [Green Version]
- Park, Y.; Choi, J.E.; Hwang, H.S. Protein supplementation improves muscle mass and physical performance in undernourished prefrail and frail elderly subjects: A randomized, double-blind, placebo-controlled trial. Am. J. Clin. Nutr. 2018, 108, 1026–1033. [Google Scholar] [CrossRef]
- Van den Heuvel, E.; Murphy, J.L.; Appleton, K.M. The provision of recipes and single-use herb/spice packets to increase egg and protein intake in community-dwelling older adults: A randomized controlled trial. Pub. Health Nutr. 2021, 24, 2109–2122. [Google Scholar] [CrossRef]
- Borkent, J.W.; Beelen, J.; Linschooten, J.O.; Roodenburg, A.J.C.; de van der Schueren, M.A.E. The consumer study: A randomized trial towards the effectiveness of protein-rich ready-made meals and protein-rich dairy products in increasing protein intake of community-dwelling older adults after switching from self-prepared meals towards ready-made meals. J. Nutr. Sci. 2019, 8. [Google Scholar] [CrossRef] [Green Version]
- Aleman-Mateo, H.; Carreon, V.R.; Macias, L.; Astiazaran-Garcia, H.; Gallegos-Aguilar, A.C.; Enriquez, J.R. Nutrient-rich dairy proteins improve appendicular skeletal muscle mass and physical performance and attenuate the loss of muscle strength in older men and women subjects: A single-blind randomized clinical trial. Clin. Interv. Aging 2014, 9, 1517–1525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hill, B.; Richardson, B.; Skouteris, H. Do we know how to design effective health coaching interventions: A systematic review of the state of the literature. Am. J. Health Promotion 2015, 29, e158–e168. [Google Scholar] [CrossRef] [PubMed]
- Schneider, K.L.; Coons, M.J.; McFadden, H.; Pellegrini, C.A.; DeMott, A.; Siddique, J.; Hedeker, D.; Aylward, L.; Spring, B. Mechanisms of change in diet and activity in the Make Better Choices 1 trial. Health Psychol. 2016, 35, 723. [Google Scholar] [CrossRef]
- Olsen, J.M.; Nesbitt, B.J. Health coaching to improve healthy lifestyle behaviors: An integrative review. Am. J. Health Promotion 2010, 25, e1–e12. [Google Scholar] [CrossRef]
- Vincent, A.E.; Sanchez Birkhead, A.C. Evaluation of the effectiveness of nurse coaching in improving health outcomes in chronic conditions. Holist. Nurs. Pract. 2013, 27, 148–161. [Google Scholar] [CrossRef]
- ESHA Research. ESHA Food and Nutrition Databases. Available online: https://esha.com/nutrition-database/ (accessed on 15 June 2021).
- Stumbo, P. Considerations for selecting a dietary assessment system. J. Food Compost. Anal. 2008, 21, S13–S19. [Google Scholar] [CrossRef] [Green Version]
- Janssen, I.; Heymsfield, S.B.; Ross, R. Low relative skeletal muscle mass (sarcopenia) in older persons is associated with functional impairment and physical disability. J. Am. Geriatr Soc. 2002, 50, 889–896. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bohannon, R.W. Reference values for the Timed Up and Go Test: A descriptive meta-analysis. J. Geriatr. Phys. Ther. 2006, 29, 64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guralnik, J.M.; Simonsick, E.M.; Ferrucci, L.; Glynn, R.J.; Berkman, L.F.; Blazer, D.G.; Scherr, P.A.; Wallace, R.B. A short physical performance battery assessing lower extremity function: Association with self-reported disability and prediction of morality and nursing home admission. J. Gerontol. 1994, 49, M85–M94. [Google Scholar] [CrossRef]
- Roberts, H.C.; Denison, H.J.; Martin, H.J.; Patel, H.P.; Syddall, H.; Cooper, C.; Sayer, A.A. A review of the measurement of grip strength in clinical and epidemiological studies: Towards a standardised approach. Age Ageing 2011, 40, 423–429. [Google Scholar] [CrossRef] [Green Version]
- Cesari, M.; Kritchevsky, S.B.; Newman, A.B.; Simonsick, E.M.; Harris, T.B.; Penninx, B.W.; Brach, J.S.; Tylavsky, F.A.; Satterfield, S.; Bauer, D.C.; et al. Added value of physical performance measures in predicting adverse health-related events: Results from the health, aging and body composition study. J. Am. Geriatr. Soc. 2009, 57, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Dodds, R.M.; Syddall, H.E.; Cooper, R.; Benzeval, M.; Deary, I.J.; Dennison, E.M.; Der, G.; Gale, C.R.; Inskip, H.M.; Jagger, C.; et al. Grip strength across the life course: Normative data from twelve British studies. PLoS ONE 2014, 9, e113637. [Google Scholar] [CrossRef] [Green Version]
- Dossey, B.M.; Schaub, B.G.; Luck, S.; International Nurse Coach Association. Nurse Coaching: Integrative Approaches for Health and Wellbeing; International Nurse Coach Association: North Miami, FL, USA, 2015. [Google Scholar]
- Hess, D.R.; Dossey, B.M.; Southard, M.E.; Luck, S.; Schaub, B.G.; Bark, L. The Art and Science of Nurse Coaching: Provider’s Guide to Coaching Scope and Competencies; American Nurses Association: Silver Spring, MD, USA, 2013. [Google Scholar]
- Mishra, S.; Goldman, J.D.; Sahyoun, N.R.; Moshfegh, A.J. Association between dietary protein intake and grip strength among adults aged 51 years and over: What we eat in America, National Health and Nutrition Examination Survey 2011–2014. PLoS ONE 2018, 13, e0191368. [Google Scholar] [CrossRef] [Green Version]
- Norman, K.; Stobaus, N.; Gonzalez, M.C.; Schulzke, J.; Pirlich, M. Hand grip strength: Outcome predictor and marker of nutritional status. Clin. Nutr. 2011, 30, 135–142. [Google Scholar] [CrossRef]
- Rossato, L.T.; Nahas, P.C.; De Branco, F.; Martins, F.M.; Souza, A.P.; Carneiro, M.A.; Orsatti, F.L.; De Oliveira, E.P. Higher protein intake does not improve lean mass gain when compared with RDA recommendation in postmenopausal women following resistance exercise protocol: A randomized clinical trial. Nutrients 2017, 9, 1007. [Google Scholar] [CrossRef] [Green Version]
- Morton, R.W.; Murphy, K.T.; McKellar, S.R. A systematic review, meta-analysis and meta-regression of the effect of protein supplementation on resistance training-induced gains in muscle mass and strength in healthy adults. Brit. J. Sports Med. 2018, 52, 376–384. [Google Scholar] [CrossRef]
| Baseline Characteristics | Not-Coached (n = 28) | Coached (n = 25) | ||||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | t(51) | p | |
| Age (years) | 56.4 | 4.8 | 55.1 | 5.7 | 0.95 | 0.35 |
| Height (cm) | 164.1 | 7.3 | 162.1 | 6.4 | 1.35 | 0.33 |
| Weight (kg) | 67.6 | 11.1 | 63.4 | 11.9 | 1.33 | 0.19 |
| Body Mass Index (kg/m2) | 25.1 | 3.7 | 24.2 | 3.9 | 0.84 | 0.41 |
| Protein Intake | Not-Coached | Coached | |||
|---|---|---|---|---|---|
| (n = 28) | (n = 25) | ||||
| Mean | SD | Mean | SD | p | |
| Breakfast | |||||
| Protein (g) | 9.6 | 6.3 | 11.5 | 6.5 | 0.29 |
| Protein (g/kg body weight) | 0.1 | 0.1 | 0.2 | 0.1 | 0.17 |
| Lunch | |||||
| Protein (g) | 18.1 | 7.1 | 19.9 | 7.8 | 0.38 |
| Protein (g/kg body weight) | 0.3 | 0.1 | 0.3 | 0.1 | 0.10 |
| Dinner | |||||
| Protein (g) | 21.6 | 6.9 | 24.1 | 6.5 | 0.18 |
| Protein (g/kg body weight) | 0.3 | 0.1 | 0.4 | 0.1 | 0.06 |
| 3-meals | |||||
| Protein 3-meals (g) | 49.0 | 13.9 | 53.8 | 14.8 | 0.07 |
| Protein 3-meals (g/kg body weight) | 0.7 | 0.2 | 0.9 | 0.2 | 0.01 |
| Outcomes | Not-Coached | Coached | Within Group Change by Time | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Baseline | 12 Weeks | Baseline | 12 Weeks | F | Df | p | n2 | |||||
| Mean SD | Mean SD | Mean SD | Mean SD | |||||||||
| SMM (kg) | 19.8 | 2.0 | 19.5 | 2.1 | 19.1 | 2.1 | 19.0 | 2.3 | 0.223 | 1.50 | 0.64 | 0.004 |
| SMI (%) | 30.3 | 4.6 | 29.7 | 4.7 | 30.6 | 3.6 | 29.9 | 3.5 | 0.018 | 1.50 | 0.89 | 0.000 |
| GS (kg) | 25.0 | 5.4 | 25.8 | 5.1 | 21.5 | 4.5 | 21.9 | 5.2 | 1.62 | 1.48 | 0.21 | 0.033 |
| 5-Chair Rise (s) | 9.2 | 2.8 | 7.3 | 2.3 | 7.9 | 2.0 | 6.8 | 1.8 | 42.41 | 1.49 | <0.001 | 0.464 |
| 4 m Gait Speed (s) | 3.3 | 0.6 | 3.0 | 0.4 | 3.1 | 0.5 | 2.9 | 0.4 | 26.68 | 1.50 | <0.001 | 0.348 |
| Between Group Change by Time Intervention | ||||||||||||
| SMM (kg) | 1.13 | 1.50 | 0.29 | 0.022 | ||||||||
| SMI (%) | 0.063 | 1.50 | 0.80 | 0.001 | ||||||||
| GS (kg) | 0.21 | 1.48 | 0.32 | 0.004 | ||||||||
| 5-Chair Rise (s) | 3.50 | 1.50 | 0.07 | 0.067 | ||||||||
| 4 m Gait Speed (s) | 0.10 | 1.50 | 0.38 | 0.002 | ||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jackson, K.L.; Gropper, S.S.; Hunt, D.; D’Avolio, D.; Newman, D. Effectiveness of a Per-Meal Protein Prescription and Nutrition Education with versus without Diet Coaching on Dietary Protein Intake and Muscle Health in Middle-Aged Women. Nutrients 2022, 14, 375. https://doi.org/10.3390/nu14020375
Jackson KL, Gropper SS, Hunt D, D’Avolio D, Newman D. Effectiveness of a Per-Meal Protein Prescription and Nutrition Education with versus without Diet Coaching on Dietary Protein Intake and Muscle Health in Middle-Aged Women. Nutrients. 2022; 14(2):375. https://doi.org/10.3390/nu14020375
Chicago/Turabian StyleJackson, Kelley L., Sareen S. Gropper, Dennis Hunt, Deborah D’Avolio, and David Newman. 2022. "Effectiveness of a Per-Meal Protein Prescription and Nutrition Education with versus without Diet Coaching on Dietary Protein Intake and Muscle Health in Middle-Aged Women" Nutrients 14, no. 2: 375. https://doi.org/10.3390/nu14020375
APA StyleJackson, K. L., Gropper, S. S., Hunt, D., D’Avolio, D., & Newman, D. (2022). Effectiveness of a Per-Meal Protein Prescription and Nutrition Education with versus without Diet Coaching on Dietary Protein Intake and Muscle Health in Middle-Aged Women. Nutrients, 14(2), 375. https://doi.org/10.3390/nu14020375






