Prenatal Folate and Choline Levels and Brain and Cognitive Development in Children: A Critical Narrative Review
Abstract
:1. Introduction
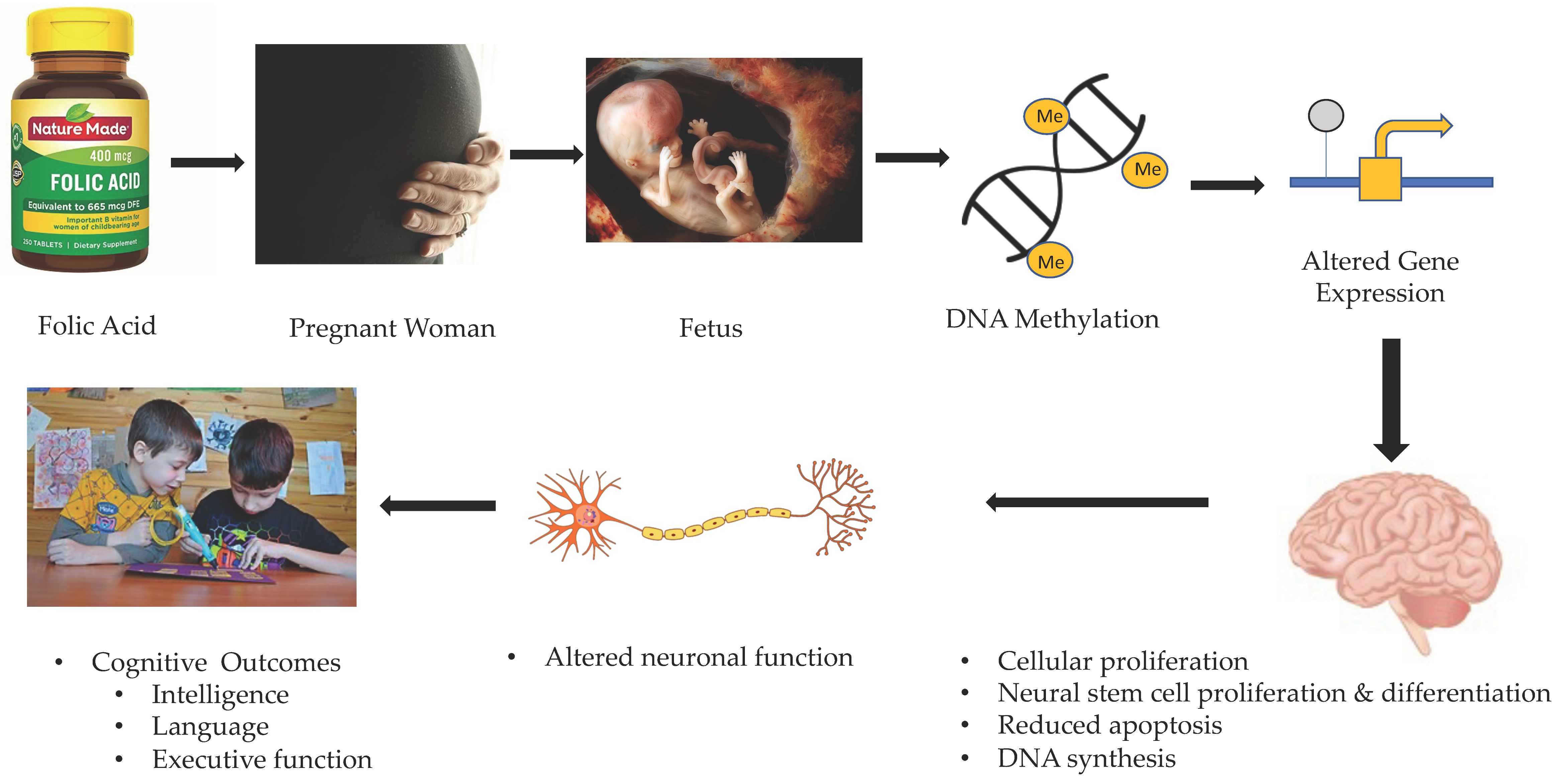
2. Gestational Folate and Fetal Brain Development
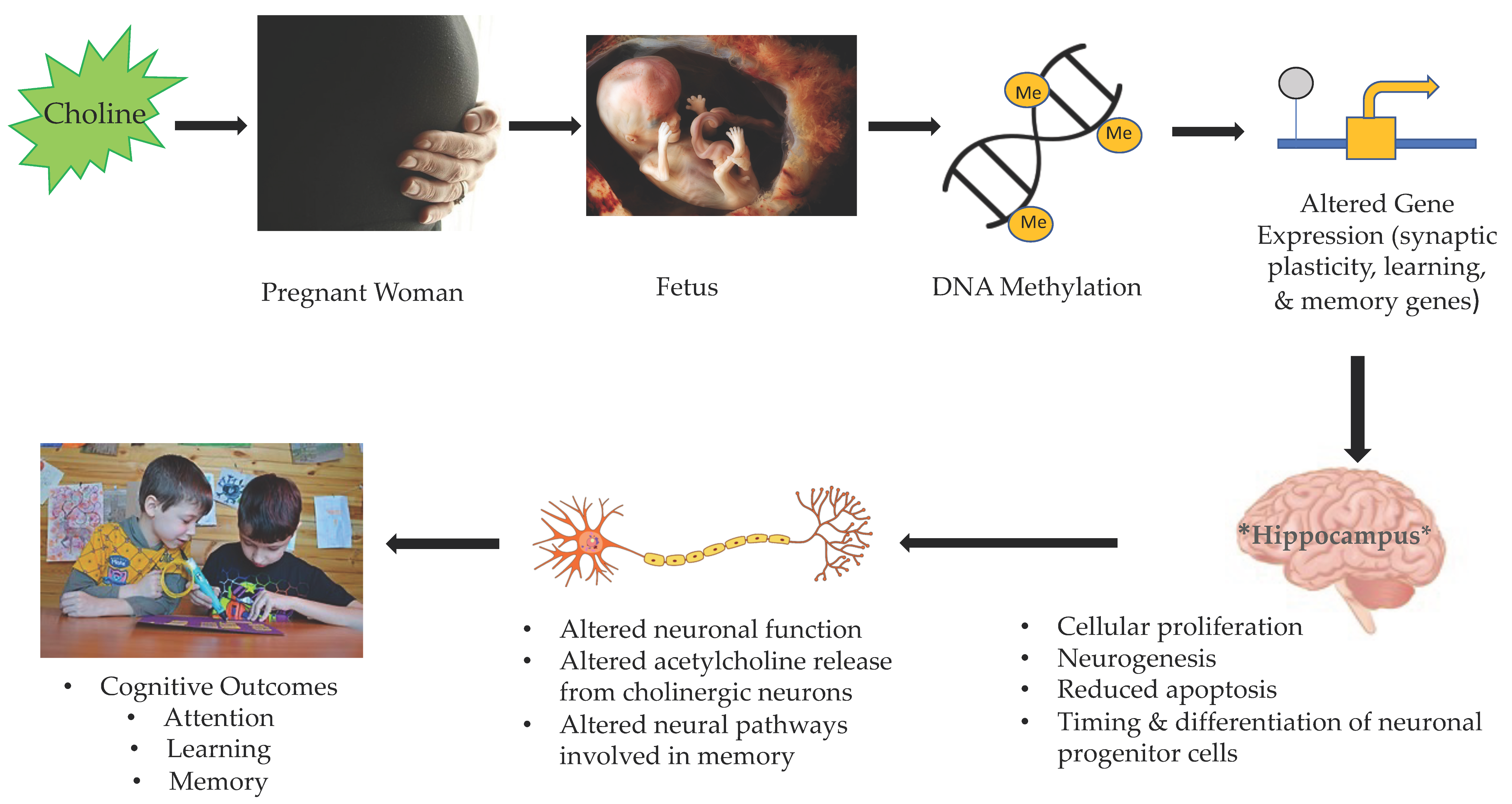
3. Gestational Choline and Fetal Brain Development
4. Potential Interactions between Gestational Folate and Choline
5. The Current State of Knowledge on the Association between Maternal Folate and Offspring Cognitive Outcomes
6. The Current State of Knowledge on the Associations between Maternal Choline and Offspring Cognitive Outcomes
7. Gaps in the Current State of Knowledge and Directions for Future Research
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mousa, A.; Naqash, A.; Lim, S. Macronutrient and Micronutrient Intake during Pregnancy: An Overview of Recent Evidence. Nutrients 2019, 11, 443. [Google Scholar] [CrossRef] [Green Version]
- Kominiarek, M.A.; Rajan, P. Nutrition Recommendations in Pregnancy and Lactation. Med. Clin. N. Am. 2016, 100, 1199–1215. [Google Scholar] [CrossRef] [Green Version]
- Darnton-Hill, I.; Mkparu, U.C. Micronutrients in Pregnancy in Low- and Middle-Income Countries. Nutrients 2015, 7, 1744–1768. [Google Scholar] [CrossRef] [Green Version]
- Baker, H.; DeAngelis, B.; Holland, B.; Gittens-Williams, L.; Barrett, T. Vitamin Profile of 563 Gravidas during Trimesters of Pregnancy. J. Am. Coll. Nutr. 2002, 21, 33–37. [Google Scholar] [CrossRef]
- Couperus, J.W.; Nelson, C.A. Early Brain Development and Plasticity. In Blackwell Handbook of Early Childhood Development; John Wiley & Sons Ltd.: New York, NY, USA, 2006; pp. 85–105. ISBN 978-0-470-75770-3. [Google Scholar]
- Raybaud, C.; Ahmad, T.; Rastegar, N.; Shroff, M.; Al Nassar, M. The Premature Brain: Developmental and Lesional Anatomy. Neuroradiology 2013, 55, 23–40. [Google Scholar] [CrossRef] [PubMed]
- Levitt, P. Structural and Functional Maturation of the Developing Primate Brain. J. Pediatr. 2003, 143, 35–45. [Google Scholar] [CrossRef]
- Huttenlocher, P.R.; Dabholkar, A.S. Regional Differences in Synaptogenesis in Human Cerebral Cortex. J. Comp. Neurol. 1997, 387, 167–178. [Google Scholar] [CrossRef]
- Nyaradi, A.; Li, J.; Hickling, S.; Foster, J.; Oddy, W.H. The Role of Nutrition in Children’s Neurocognitive Development, from Pregnancy through Childhood. Front. Hum. Neurosci. 2013, 7, 97. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.-M.; Huang, G.-W.; Tian, Z.-H.; Ren, D.-L.; Wilson, J.X. Folate Stimulates ERK1/2 Phosphorylation and Cell Proliferation in Fetal Neural Stem Cells. Nutr. Neurosci. 2009, 12, 226–232. [Google Scholar] [CrossRef]
- Zhu, J.; Thompson, C.B. Metabolic Regulation of Cell Growth and Proliferation. Nat. Rev. Mol. Cell Biol. 2019, 20, 436–450. [Google Scholar] [CrossRef] [PubMed]
- Georgieff, M.K. Nutrition and the Developing Brain: Nutrient Priorities and Measurement. Am. J. Clin. Nutr. 2007, 85, 614S–620S. [Google Scholar] [CrossRef]
- Ardila, A. Development of Metacognitive and Emotional Executive Functions in Children. Appl. Neuropsychol. Child 2013, 2, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Wilks, T.; Gerber, R.J.; Erdie-Lalena, C. Developmental Milestones: Cognitive Development. Pediatr. Rev. 2010, 31, 364–367. [Google Scholar] [CrossRef]
- Campbell, J.M.; Brown, R.T.; Cavanagh, S.E.; Vess, S.F.; Segall, M.J. Evidence-Based Assessment of Cognitive Functioning in Pediatric Psychology. J. Pediatr. Psychol. 2008, 33, 999–1014. [Google Scholar] [CrossRef] [Green Version]
- Hughes, D.; Bryan, J. The Assessment of Cognitive Performance in Children: Considerations for Detecting Nutritional Influences. Nutr. Rev. 2003, 61, 413–422. [Google Scholar] [CrossRef] [Green Version]
- Tikhomirova, T.; Malykh, A.; Malykh, S. Predicting Academic Achievement with Cognitive Abilities: Cross-Sectional Study across School Education. Behav. Sci. 2020, 10, 158. [Google Scholar] [CrossRef]
- Nesayan, A.; Amani, M.; Asadi Gandomani, R. Cognitive Profile of Children and Its Relationship with Academic Performance. Basic Clin. Neurosci. 2019, 10, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Strenze, T. Intelligence and Socioeconomic Success: A Meta-Analytic Review of Longitudinal Research. Intelligence 2007, 35, 401–426. [Google Scholar] [CrossRef]
- Li, Q.; Yan, H.; Zeng, L.; Cheng, Y.; Liang, W.; Dang, S.; Wang, Q.; Tsuji, I. Effects of Maternal Multimicronutrient Supplementation on the Mental Development of Infants in Rural Western China: Follow-up Evaluation of a Double-Blind, Randomized, Controlled Trial. Pediatrics 2009, 123, e685–e692. [Google Scholar] [CrossRef]
- He, Y.; Gao, J.; Wang, T.; Liu, C.; Luo, R. The Association between Prenatal Micronutrient Supplementation and Early Development of Children under Age Two: Evidence from Rural Guizhou, China. Child. Youth Serv. Rev. 2020, 112, 104929. [Google Scholar] [CrossRef]
- Shaw, G.M.; Finnell, R.H.; Blom, H.J.; Carmichael, S.L.; Vollset, S.E.; Yang, W.; Ueland, P.M. Choline and Risk of Neural Tube Defects in a Folate-Fortified Population. Epidemiol. Camb. Mass 2009, 20, 714–719. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bestwick, J.P.; Huttly, W.J.; Morris, J.K.; Wald, N.J. Prevention of Neural Tube Defects: A Cross-Sectional Study of the Uptake of Folic Acid Supplementation in Nearly Half a Million Women. PLoS ONE 2014, 9, e89354. [Google Scholar] [CrossRef] [PubMed]
- Scholl, T.O.; Hediger, M.L.; Schall, J.I.; Khoo, C.S.; Fischer, R.L. Dietary and Serum Folate: Their Influence on the Outcome of Pregnancy. Am. J. Clin. Nutr. 1996, 63, 520–525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Czeizel, A.E. Reduction of Urinary Tract and Cardiovascular Defects by Periconceptional Multivitamin Supplementation. Am. J. Med. Genet. 1996, 62, 179–183. [Google Scholar] [CrossRef]
- Tolarova, M. Periconceptional Supplementation with Vitamins and Folic Acid to Prevent Recurrence of Cleft Lip. Lancet Lond. Engl. 1982, 2, 217. [Google Scholar] [CrossRef]
- McKee, S.E.; Reyes, T.M. Effect of Supplementation with Methyl-Donor Nutrients on Neurodevelopment and Cognition: Considerations for Future Research. Nutr. Rev. 2018, 76, 497–511. [Google Scholar] [CrossRef] [Green Version]
- Naninck, E.F.G.; Stijger, P.C.; Brouwer-Brolsma, E.M. The Importance of Maternal Folate Status for Brain Development and Function of Offspring. Adv. Nutr. 2019, 10, 502–519. [Google Scholar] [CrossRef]
- Chan, Y.-M.; Bailey, R.; O’Connor, D.L. Folate. Adv. Nutr. 2013, 4, 123–125. [Google Scholar] [CrossRef]
- Partearroyo, T.; de Samaniego-Vaesken, M.L.; Ruiz, E.; Olza, J.; Aranceta-Bartrina, J.; Gil, Á.; González-Gross, M.; Ortega, R.M.; Serra-Majem, L.; Varela-Moreiras, G. Dietary Sources and Intakes of Folates and Vitamin B12 in the Spanish Population: Findings from the ANIBES Study. PLoS ONE 2017, 12, e0189230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ray, J.G. Folic Acid Food Fortification in Canada. Nutr. Rev. 2004, 62, S35–S39. [Google Scholar] [CrossRef]
- Government of Canada, C.F.I.A. Nutrient Content Claims: Reference Information. Available online: https://inspection.canada.ca/food-label-requirements/labelling/industry/nutrient-content/reference-information/eng/1389908857542/1389908896254?chap=1%20%20https://www.inspection.gc.ca/food-label-requirements/labelling/industry/nutrient-content/reference-information/eng/1389908857542/1389908896254?chap=1 (accessed on 3 October 2021).
- Health Canada. Regulations Amending the Food and Drug Regulations (1066). Can. Gaz. Part 1 1997, 131, 3702–3737. [Google Scholar]
- Institute of Medicine (US) Standing Committee on the Scientific Evaluation of Dietary Reference Intakes and its Panel on Folate, Other B Vitamins, and Choline. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline; The National Academies Collection. Reports funded by National Institutes of Health; National Academies Press (US): Washington, DC, USA, 1998; ISBN 978-0-309-06411-8.
- Antony, A.C. In Utero Physiology: Role of Folic Acid in Nutrient Delivery and Fetal Development. Am. J. Clin. Nutr. 2007, 85, 598S–603S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Viswanathan, M.; Treiman, K.A.; Kish-Doto, J.; Middleton, J.C.; Coker-Schwimmer, E.J.L.; Nicholson, W.K. Folic Acid Supplementation for the Prevention of Neural Tube Defects: An Updated Evidence Report and Systematic Review for the US Preventive Services Task Force. JAMA 2017, 317, 190–203. [Google Scholar] [CrossRef]
- World Health Organization. Guideline: Optimal Serum and Red Blood Cell Folate Concentrations in Women of Reproductive Age for Prevention of Neural Tube Defects. World Health Organization: Geneva, Switzerland. Available online: https://apps.who.int/iris/handle/10665/161988 (accessed on 10 January 2022).
- Bisseling, T.M.; Steegers, E.a.P.; van den Heuvel, J.J.M.; Siero, H.L.M.; van de Water, F.M.; Walker, A.J.; Steegers-Theunissen, R.P.M.; Smits, P.; Russel, F.G.M. Placental Folate Transport and Binding Are Not Impaired in Pregnancies Complicated by Fetal Growth Restriction. Placenta 2004, 25, 588–593. [Google Scholar] [CrossRef] [PubMed]
- Giugliani, E.R.; Jorge, S.M.; Gonçalves, A.L. Serum and Red Blood Cell Folate Levels in Parturients, in the Intervillous Space of the Placenta and in Full-Term Newborns. J. Perinat. Med. 1985, 13, 55–59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stark, K.D.; Pawlosky, R.J.; Sokol, R.J.; Hannigan, J.H.; Salem, N., Jr. Maternal Smoking Is Associated with Decreased 5-Methyltetrahydrofolate in Cord Plasma. Am. J. Clin. Nutr. 2007, 85, 796–802. [Google Scholar] [CrossRef] [Green Version]
- Economides, D.L.; Ferguson, J.; Mackenzie, I.Z.; Darley, J.; Ware, I.I.; Holmes-Siedle, M. Folate and Vitamin B12 Concentrations in Maternal and Fetal Blood, and Amniotic Fluid in Second Trimester Pregnancies Complicated by Neural Tube Defects. Br. J. Obstet. Gynaecol. 1992, 99, 23–25. [Google Scholar] [CrossRef]
- Guerra-Shinohara, E.M.; Morita, O.E.; Peres, S.; Pagliusi, R.A.; Sampaio Neto, L.F.; D’Almeida, V.; Irazusta, S.P.; Allen, R.H.; Stabler, S.P. Low Ratio of S-Adenosylmethionine to S-Adenosylhomocysteine Is Associated with Vitamin Deficiency in Brazilian Pregnant Women and Newborns. Am. J. Clin. Nutr. 2004, 80, 1312–1321. [Google Scholar] [CrossRef] [Green Version]
- Obeid, R.; Munz, W.; Jäger, M.; Schmidt, W.; Herrmann, W. Biochemical Indexes of the B Vitamins in Cord Serum Are Predicted by Maternal B Vitamin Status. Am. J. Clin. Nutr. 2005, 82, 133–139. [Google Scholar] [CrossRef]
- Molloy, A.M.; Mills, J.L.; Cox, C.; Daly, S.F.; Conley, M.; Brody, L.C.; Kirke, P.N.; Scott, J.M.; Ueland, P.M. Choline and Homocysteine Interrelations in Umbilical Cord and Maternal Plasma at Delivery. Am. J. Clin. Nutr. 2005, 82, 836–842. [Google Scholar] [CrossRef]
- Thorand, B.; Pietrzik, K.; Prinz-Langenohl, R.; Hages, M.; Holzgreve, W. Maternal and Fetal Serum and Red Blood Cell Folate and Vitamin B12 Concentrations in Pregnancies Affected by Neural Tube Defects. Z. Geburtshilfe Neonatol. 1996, 200, 176–180. [Google Scholar] [PubMed]
- De Wals, P.; Tairou, F.; Van Allen, M.I.; Lowry, R.B.; Evans, J.A.; Van den Hof, M.C.; Crowley, M.; Uh, S.-H.; Zimmer, P.; Sibbald, B.; et al. Spina Bifida before and after Folic Acid Fortification in Canada. Birth Defects Res. Part A Clin. Mol. Teratol. 2008, 82, 622–626. [Google Scholar] [CrossRef] [PubMed]
- Craciunescu, C.N.; Brown, E.C.; Mar, M.-H.; Albright, C.D.; Nadeau, M.R.; Zeisel, S.H. Folic Acid Deficiency during Late Gestation Decreases Progenitor Cell Proliferation and Increases Apoptosis in Fetal Mouse Brain. J. Nutr. 2004, 134, 162–166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Craciunescu, C.N.; Johnson, A.R.; Zeisel, S.H. Dietary Choline Reverses Some, but Not All, Effects of Folate Deficiency on Neurogenesis and Apoptosis in Fetal Mouse Brain. J. Nutr. 2010, 140, 1162–1166. [Google Scholar] [CrossRef] [Green Version]
- Zeisel, S.H.; da Costa, K.-A. Choline: An Essential Nutrient for Public Health. Nutr. Rev. 2009, 67, 615–623. [Google Scholar] [CrossRef] [Green Version]
- Blusztajn, J.K.; Slack, B.E.; Mellott, T.J. Neuroprotective Actions of Dietary Choline. Nutrients 2017, 9, 815. [Google Scholar] [CrossRef] [Green Version]
- Meck, W.H.; Williams, C.L. Metabolic Imprinting of Choline by Its Availability during Gestation: Implications for Memory and Attentional Processing across the Lifespan. Neurosci. Biobehav. Rev. 2003, 27, 385–399. [Google Scholar] [CrossRef]
- Wiedeman, A.M.; Barr, S.I.; Green, T.J.; Xu, Z.; Innis, S.M.; Kitts, D.D. Dietary Choline Intake: Current State of Knowledge Across the Life Cycle. Nutrients 2018, 10, 1513. [Google Scholar] [CrossRef] [Green Version]
- Korsmo, H.W.; Jiang, X.; Caudill, M.A. Choline: Exploring the Growing Science on Its Benefits for Moms and Babies. Nutrients 2019, 11, 1823. [Google Scholar] [CrossRef] [Green Version]
- Office of Dietary Supplements—Choline. Available online: https://ods.od.nih.gov/factsheets/Choline-HealthProfessional/ (accessed on 3 October 2021).
- Zeisel, S.H. Choline: Critical Role during Fetal Development and Dietary Requirements in Adults. Annu. Rev. Nutr. 2006, 26, 229–250. [Google Scholar] [CrossRef] [Green Version]
- Craciunescu, C.N.; Albright, C.D.; Mar, M.-H.; Song, J.; Zeisel, S.H. Choline Availability during Embryonic Development Alters Progenitor Cell Mitosis in Developing Mouse Hippocampus. J. Nutr. 2003, 133, 3614–3618. [Google Scholar] [CrossRef] [PubMed]
- Braun, L.D.; Cornford, E.M.; Oldendorf, W.H. Newborn Rabbit Blood-Brain Barrier Is Selectively Permeable and Differs Substantially from the Adult. J. Neurochem. 1980, 34, 147–152. [Google Scholar] [CrossRef]
- Shaw, G.M.; Carmichael, S.L.; Yang, W.; Selvin, S.; Schaffer, D.M. Periconceptional Dietary Intake of Choline and Betaine and Neural Tube Defects in Offspring. Am. J. Epidemiol. 2004, 160, 102–109. [Google Scholar] [CrossRef] [Green Version]
- Zeisel, S.H. The Fetal Origins of Memory: The Role of Dietary Choline in Optimal Brain Development. J. Pediatr. 2006, 149, S131–S136. [Google Scholar] [CrossRef] [Green Version]
- Sarter, M.; Parikh, V. Choline Transporters, Cholinergic Transmission and Cognition. Nat. Rev. Neurosci. 2005, 6, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Napoli, I.; Blusztajn, J.K.; Mellott, T.J. Prenatal Choline Supplementation in Rats Increases the Expression of IGF2 and Its Receptor IGF2R and Enhances IGF2-Induced Acetylcholine Release in Hippocampus and Frontal Cortex. Brain Res. 2008, 1237, 124–135. [Google Scholar] [CrossRef]
- Albright, C.D.; Friedrich, C.B.; Brown, E.C.; Mar, M.H.; Zeisel, S.H. Maternal Dietary Choline Availability Alters Mitosis, Apoptosis and the Localization of TOAD-64 Protein in the Developing Fetal Rat Septum. Dev. Brain Res. 1999, 115, 123–129. [Google Scholar] [CrossRef]
- Mehedint, M.G.; Craciunescu, C.N.; Zeisel, S.H. Maternal Dietary Choline Deficiency Alters Angiogenesis in Fetal Mouse Hippocampus. Proc. Natl. Acad. Sci. USA 2010, 107, 12834–12839. [Google Scholar] [CrossRef] [Green Version]
- Glenn, M.J.; Gibson, E.M.; Kirby, E.D.; Mellott, T.J.; Blusztajn, J.K.; Williams, C.L. Prenatal Choline Availability Modulates Hippocampal Neurogenesis and Neurogenic Responses to Enriching Experiences in Adult Female Rats. Eur. J. Neurosci. 2007, 25, 2473–2482. [Google Scholar] [CrossRef] [Green Version]
- Albright, C.D.; Tsai, A.Y.; Mar, M.H.; Zeisel, S.H. Choline Availability Modulates the Expression of TGFbeta1 and Cytoskeletal Proteins in the Hippocampus of Developing Rat Brain. Neurochem. Res. 1998, 23, 751–758. [Google Scholar] [CrossRef] [PubMed]
- Niculescu, M.D.; Craciunescu, C.N.; Zeisel, S.H. Dietary Choline Deficiency Alters Global and Gene-specific DNA Methylation in the Developing Hippocampus of Mouse Fetal Brains. FASEB J. 2006, 20, 43–49. [Google Scholar] [CrossRef] [Green Version]
- Mehedint, M.G.; Niculescu, M.D.; Craciunescu, C.N.; Zeisel, S.H. Choline Deficiency Alters Global Histone Methylation and Epigenetic Marking at the Rel Site of the Calbindin 1 Gene. FASEB J. 2010, 24, 184–195. [Google Scholar] [CrossRef] [Green Version]
- Jadavji, N.M.; Deng, L.; Malysheva, O.; Caudill, M.A.; Rozen, R. MTHFR Deficiency or Reduced Intake of Folate or Choline in Pregnant Mice Results in Impaired Short-Term Memory and Increased Apoptosis in the Hippocampus of Wild-Type Offspring. Neuroscience 2015, 300, 1–9. [Google Scholar] [CrossRef]
- Ferguson, S.A.; Berry, K.J.; Hansen, D.K.; Wall, K.S.; White, G.; Antony, A.C. Behavioral Effects of Prenatal Folate Deficiency in Mice. Birth Defects Res. Part A Clin. Mol. Teratol. 2005, 73, 249–252. [Google Scholar] [CrossRef]
- Julvez, J.; Fortuny, J.; Mendez, M.; Torrent, M.; Ribas-Fitó, N.; Sunyer, J. Maternal Use of Folic Acid Supplements during Pregnancy and Four-Year-Old Neurodevelopment in a Population-Based Birth Cohort. Paediatr. Perinat. Epidemiol. 2009, 23, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Wehby, G.L.; Murray, J.C. The Effects of Prenatal Use of Folic Acid and Other Dietary Supplements on Early Child Development. Matern. Child Health J. 2008, 12, 180–187. [Google Scholar] [CrossRef] [PubMed]
- McNulty, H.; Rollins, M.; Cassidy, T.; Caffrey, A.; Marshall, B.; Dornan, J.; McLaughlin, M.; McNulty, B.A.; Ward, M.; Strain, J.J.; et al. Effect of Continued Folic Acid Supplementation beyond the First Trimester of Pregnancy on Cognitive Performance in the Child: A Follow-up Study from a Randomized Controlled Trial (FASSTT Offspring Trial). BMC Med. 2019, 17, 196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- US Preventive Services Task Force; Bibbins-Domingo, K.; Grossman, D.C.; Curry, S.J.; Davidson, K.W.; Epling, J.W.; García, F.A.R.; Kemper, A.R.; Krist, A.H.; Kurth, A.E.; et al. Folic Acid Supplementation for the Prevention of Neural Tube Defects: US Preventive Services Task Force Recommendation Statement. JAMA 2017, 317, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Chatzi, L.; Papadopoulou, E.; Koutra, K.; Roumeliotaki, T.; Georgiou, V.; Stratakis, N.; Lebentakou, V.; Karachaliou, M.; Vassilaki, M.; Kogevinas, M. Effect of High Doses of Folic Acid Supplementation in Early Pregnancy on Child Neurodevelopment at 18 Months of Age: The Mother-Child Cohort “Rhea” Study in Crete, Greece. Public Health Nutr. 2012, 15, 1728–1736. [Google Scholar] [CrossRef] [Green Version]
- Chen, H.; Qin, L.; Gao, R.; Jin, X.; Cheng, K.; Zhang, S.; Hu, X.; Xu, W.; Wang, H. Neurodevelopmental Effects of Maternal Folic Acid Supplementation: A Systematic Review and Meta-analysis. Crit. Rev. Food Sci. Nutr. 2021, 1–17. [Google Scholar] [CrossRef]
- Villamor, E.; Rifas-Shiman, S.L.; Gillman, M.W.; Oken, E. Maternal Intake of Methyl-Donor Nutrients and Child Cognition at 3 Years of Age. Paediatr. Perinat. Epidemiol. 2012, 26, 328–335. [Google Scholar] [CrossRef] [Green Version]
- Boeke, C.E.; Gillman, M.W.; Hughes, M.D.; Rifas-Shiman, S.L.; Villamor, E.; Oken, E. Choline Intake during Pregnancy and Child Cognition at Age 7 Years. Am. J. Epidemiol. 2013, 177, 1338–1347. [Google Scholar] [CrossRef] [Green Version]
- Bouckaert, K.P.; Slimani, N.; Nicolas, G.; Vignat, J.; Wright, A.J.A.; Roe, M.; Witthöft, C.M.; Finglas, P.M. Critical Evaluation of Folate Data in European and International Databases: Recommendations for Standardization in International Nutritional Studies. Mol. Nutr. Food Res. 2011, 55, 166–180. [Google Scholar] [CrossRef]
- Messerer, M.; Johansson, S.-E.; Wolk, A. The Validity of Questionnaire-Based Micronutrient Intake Estimates Is Increased by Including Dietary Supplement Use in Swedish Men. J. Nutr. 2004, 134, 1800–1805. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Cogswell, M.E.; Hamner, H.C.; Carriquiry, A.; Bailey, L.B.; Pfeiffer, C.M.; Berry, R.J. Folic Acid Source, Usual Intake, and Folate and Vitamin B-12 Status in US Adults: National Health and Nutrition Examination Survey (NHANES) 2003-2006. Am. J. Clin. Nutr. 2010, 91, 64–72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hopkins, S.M.; Gibney, M.J.; Nugent, A.P.; McNulty, H.; Molloy, A.M.; Scott, J.M.; Flynn, A.; Strain, J.J.; Ward, M.; Walton, J.; et al. Impact of Voluntary Fortification and Supplement Use on Dietary Intakes and Biomarker Status of Folate and Vitamin B-12 in Irish Adults. Am. J. Clin. Nutr. 2015, 101, 1163–1172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, A.; Chanarin, I.; Slavin, G.; Levi, A.J. Folate Deficiency in the Alcoholic—Its Relationship to Clinical and Haematological Abnormalities, Liver Disease and Folate Stores. Br. J. Haematol. 1975, 29, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.-Y.; Rose, C.E.; Qi, Y.P.; Williams, J.L.; Yeung, L.F.; Berry, R.J.; Hao, L.; Cannon, M.J.; Crider, K.S. Defining the Plasma Folate Concentration Associated with the Red Blood Cell Folate Concentration Threshold for Optimal Neural Tube Defects Prevention: A Population-Based, Randomized Trial of Folic Acid Supplementation. Am. J. Clin. Nutr. 2019, 109, 1452–1461. [Google Scholar] [CrossRef] [Green Version]
- Cordero, A.M.; Crider, K.S.; Rogers, L.M.; Cannon, M.J.; Berry, R.J. Optimal Serum and Red Blood Cell Folate Concentrations in Women of Reproductive Age for Prevention of Neural Tube Defects: World Health Organization Guidelines. MMWR Morb. Mortal. Wkly. Rep. 2015, 64, 421–423. [Google Scholar] [PubMed]
- Veena, S.R.; Krishnaveni, G.V.; Srinivasan, K.; Wills, A.K.; Muthayya, S.; Kurpad, A.V.; Yajnik, C.S.; Fall, C.H. Higher Maternal Plasma Folate but Not Vitamin B-12 Concentrations during Pregnancy Are Associated with Better Cognitive Function Scores in 9-10 Year Old Children in South-India-. J. Nutr. 2010, 140, 1014–1022. [Google Scholar] [CrossRef] [Green Version]
- Fayyaz, F.; Wang, F.; Jacobs, R.L.; O’Connor, D.L.; Bell, R.C.; Field, C.J. APrON Study Team Folate, Vitamin B12, and Vitamin B6 Status of a Group of High Socioeconomic Status Women in the Alberta Pregnancy Outcomes and Nutrition (APrON) Cohort. Appl. Physiol. Nutr. Metab. 2014, 39, 1402–1408. [Google Scholar] [CrossRef]
- Pfeiffer, C.M.; Johnson, C.L.; Jain, R.B.; Yetley, E.A.; Picciano, M.F.; Rader, J.I.; Fisher, K.D.; Mulinare, J.; Osterloh, J.D. Trends in Blood Folate and Vitamin B-12 Concentrations in the United States, 1988 2004. Am. J. Clin. Nutr. 2007, 86, 718–727. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ars, C.L.; Nijs, I.M.; Marroun, H.E.; Muetzel, R.; Schmidt, M.; Steenweg-de Graaff, J.; van der Lugt, A.; Jaddoe, V.W.; Hofman, A.; Steegers, E.A.; et al. Prenatal Folate, Homocysteine and Vitamin B12 Levels and Child Brain Volumes, Cognitive Development and Psychological Functioning: The Generation R Study. Br. J. Nutr. 2019, 122, S1–S9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, B.T.F.; Dyer, R.A.; King, D.J.; Richardson, K.J.; Innis, S.M. Early Second Trimester Maternal Plasma Choline and Betaine Are Related to Measures of Early Cognitive Development in Term Infants. PLoS ONE 2012, 7, e43448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tamura, T.; Goldenberg, R.L.; Chapman, V.R.; Johnston, K.E.; Ramey, S.L.; Nelson, K.G. Folate Status of Mothers during Pregnancy and Mental and Psychomotor Development of Their Children at Five Years of Age. Pediatrics 2005, 116, 703–708. [Google Scholar] [CrossRef]
- Han, Y.H.; Yon, M.; Hyun, T.H. Folate Intake Estimated with an Updated Database and Its Association to Blood Folate and Homocysteine in Korean College Students. Eur. J. Clin. Nutr. 2005, 59, 246–254. [Google Scholar] [CrossRef] [Green Version]
- Houghton, L.A.; Sherwood, K.L.; O’Connor, D.L. How Well Do Blood Folate Concentrations Predict Dietary Folate Intakes in a Sample of Canadian Lactating Women Exposed to High Levels of Folate? An Observational Study. BMC Pregnancy Childbirth 2007, 7, 25. [Google Scholar] [CrossRef] [PubMed]
- Marshall, J.R. Methodologic and Statistical Considerations Regarding Use of Biomarkers of Nutritional Exposure in Epidemiology. J. Nutr. 2003, 133, 881S–887S. [Google Scholar] [CrossRef] [Green Version]
- Meck, W.H.; Smith, R.A.; Williams, C.L. Pre- and Postnatal Choline Supplementation Produces Long-Term Facilitation of Spatial Memory. Dev. Psychobiol. 1988, 21, 339–353. [Google Scholar] [CrossRef]
- Glenn, M.J.; Kirby, E.D.; Gibson, E.M.; Wong-Goodrich, S.; Mellott, T.J.; Blusztajn, J.K.; Williams, C.L. Age-Related Declines in Exploratory Behavior and Markers of Hippocampal Plasticity Are Attenuated by Prenatal Choline Supplementation in Rats. Brain Res. 2008, 1237, 110–123. [Google Scholar] [CrossRef] [Green Version]
- Meck, W.H.; Williams, C.L. Choline Supplementation during Prenatal Development Reduces Proactive Interference in Spatial Memory. Dev. Brain Res. 1999, 118, 51–59. [Google Scholar] [CrossRef]
- Meck, W.H.; Williams, C.L. Perinatal Choline Supplementation Increases the Threshold for Chunking in Spatial Memory. Neuroreport 1997, 8, 3053–3059. [Google Scholar] [CrossRef]
- Meck, W.H.; Williams, C.L. Simultaneous Temporal Processing Is Sensitive to Prenatal Choline Availability in Mature and Aged Rats. Neuroreport 1997, 8, 3045–3051. [Google Scholar] [CrossRef]
- Caudill, M.A.; Strupp, B.J.; Muscalu, L.; Nevins, J.E.H.; Canfield, R.L. Maternal Choline Supplementation during the Third Trimester of Pregnancy Improves Infant Information Processing Speed: A Randomized, Double-Blind, Controlled Feeding Study. FASEB J. 2018, 32, 2172–2180. [Google Scholar] [CrossRef] [Green Version]
- Signore, C.; Ueland, P.M.; Troendle, J.; Mills, J.L. Choline Concentrations in Human Maternal and Cord Blood and Intelligence at 5 y of Age. Am. J. Clin. Nutr. 2008, 87, 896–902. [Google Scholar] [CrossRef] [Green Version]
- Mujica, M.; Lewis, E.; Jacobs, R.; Letourneau, N.; Bell, R.; Field, C.; Lamers, Y. Plasma Free Choline Concentration Did Not Reflect Dietary Choline Intake in Early and Late Pregnancy: Findings from the APrON Study. Curr. Dev. Nutr. 2020, 4, 1825. [Google Scholar] [CrossRef]
- Lewis, E.D.; Subhan, F.B.; Bell, R.C.; McCargar, L.J.; Curtis, J.M.; Jacobs, R.L.; Field, C.J. APrON team Estimation of Choline Intake from 24 h Dietary Intake Recalls and Contribution of Egg and Milk Consumption to Intake among Pregnant and Lactating Women in Alberta. Br. J. Nutr. 2014, 112, 112–121. [Google Scholar] [CrossRef] [Green Version]
- de Neubourg, E.; Borghans, L.; Coppens, K.; Jansen, M. Explaining Children’s Life Outcomes: Parental Socioeconomic Status, Intelligence and Neurocognitive Factors in a Dynamic Life Cycle Model. Child Indic. Res. 2018, 11, 1495–1513. [Google Scholar] [CrossRef]
- Arkkola, T.; Uusitalo, U.; Pietikäinen, M.; Metsälä, J.; Kronberg-Kippilä, C.; Erkkola, M.; Veijola, R.; Knip, M.; Virtanen, S.M.; Ovaskainen, M.-L. Dietary Intake and Use of Dietary Supplements in Relation to Demographic Variables among Pregnant Finnish Women. Br. J. Nutr. 2006, 96, 913–920. [Google Scholar] [CrossRef] [Green Version]
- Ardila, A.; Rosselli, M.; Matute, E.; Inozemtseva, O. Gender Differences in Cognitive Development. Dev. Psychol. 2011, 47, 984–990. [Google Scholar] [CrossRef] [Green Version]
- Duncan, G.J.; Lee, K.T.H.; Rosales-Rueda, M.; Kalil, A. Maternal Age and Child Development. Demography 2018, 55, 2229–2255. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taylor, R.M.; Fealy, S.M.; Bisquera, A.; Smith, R.; Collins, C.E.; Evans, T.-J.; Hure, A.J. Effects of Nutritional Interventions during Pregnancy on Infant and Child Cognitive Outcomes: A Systematic Review and Meta-Analysis. Nutrients 2017, 9, 1265. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhatnagar, S.; Taneja, S. Zinc and Cognitive Development. Br. J. Nutr. 2001, 85, S139–S145. [Google Scholar] [CrossRef] [Green Version]
- Alvarez, S.; Urrechaga, E.; Llorente-Ballesteros, M.; Escanero, J. The Role of Iron and Other Trace Elements on Mental Development and Cognitive Function. In Psychiatry and Neuroscience Update; Springer: Cham, Switzerland; New York, NY, USA, 2015; pp. 157–179. ISBN 978-3-319-17102-9. [Google Scholar]
- Amorós, R.; Murcia, M.; Ballester, F.; Broberg, K.; Iñiguez, C.; Rebagliato, M.; Skröder, H.; González, L.; Lopez-Espinosa, M.-J.; Llop, S. Selenium Status during Pregnancy: Influential Factors and Effects on Neuropsychological Development among Spanish Infants. Sci. Total Environ. 2018, 610–611, 741–749. [Google Scholar] [CrossRef] [PubMed]
- Amorós, R.; Murcia, M.; González, L.; Soler-Blasco, R.; Rebagliato, M.; Iñiguez, C.; Carrasco, P.; Vioque, J.; Broberg, K.; Levi, M.; et al. Maternal Copper Status and Neuropsychological Development in Infants and Preschool Children. Int. J. Hyg. Environ. Health 2019, 222, 503–512. [Google Scholar] [CrossRef] [Green Version]
- Zeeshan, F.; Bari, A.; Farhan, S.; Jabeen, U.; Rathore, A.W. Correlation between Maternal and Childhood VitB12, Folic Acid and Ferritin Levels. Pak. J. Med. Sci. 2017, 33, 162–166. [Google Scholar] [CrossRef]
- Tran, P.V.; Kennedy, B.C.; Pisansky, M.T.; Won, K.-J.; Gewirtz, J.C.; Simmons, R.A.; Georgieff, M.K. Prenatal Choline Supplementation Diminishes Early-Life Iron Deficiency–Induced Reprogramming of Molecular Networks Associated with Behavioral Abnormalities in the Adult Rat Hippocampus123. J. Nutr. 2016, 146, 484–493. [Google Scholar] [CrossRef] [Green Version]
- Min, J.-Y.; Min, K.-B. The Folate-Vitamin B12 Interaction, Low Hemoglobin, and the Mortality Risk from Alzheimer’s Disease. J. Alzheimers Dis. JAD 2016, 52, 705–712. [Google Scholar] [CrossRef] [PubMed]
- Fuller, N.J.; Evans, P.H.; Howlett, M.; Bates, C.J. The Effects of Dietary Folate and Zinc on the Outcome of Pregnancy and Early Growth in Rats. Br. J. Nutr. 1988, 59, 251–259. [Google Scholar] [CrossRef] [Green Version]
- Huot, P.S.P.; Dodington, D.W.; Mollard, R.C.; Reza-López, S.A.; Sánchez-Hernández, D.; Cho, C.E.; Kuk, J.; Ward, W.E.; Anderson, G.H. High Folic Acid Intake during Pregnancy Lowers Body Weight and Reduces Femoral Area and Strength in Female Rat Offspring. J. Osteoporos. 2013, 2013, 154109. [Google Scholar] [CrossRef]
- Raghavan, R.; Riley, A.W.; Volk, H.; Caruso, D.; Hironaka, L.; Sices, L.; Hong, X.; Wang, G.; Ji, Y.; Brucato, M.; et al. Maternal Multivitamin Intake, Plasma Folate and Vitamin B12 Levels and Autism Spectrum Disorder Risk in Offspring. Paediatr. Perinat. Epidemiol. 2018, 32, 100–111. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Xu, B.; Cao, Y.; Shao, Y.; Wu, W.; Zhou, J.; Tan, X.; Wu, X.; Kong, J.; Hu, C.; et al. Association of Maternal Folate Intake during Pregnancy with Infant Asthma Risk. Sci. Rep. 2019, 9, 8347. [Google Scholar] [CrossRef]
- Schaible, T.D.; Harris, R.A.; Dowd, S.E.; Smith, C.W.; Kellermayer, R. Maternal Methyl-Donor Supplementation Induces Prolonged Murine Offspring Colitis Susceptibility in Association with Mucosal Epigenetic and Microbiomic Changes. Hum. Mol. Genet. 2011, 20, 1687–1696. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ross, R.G.; Hunter, S.K.; McCarthy, L.; Beuler, J.; Hutchison, A.K.; Wagner, B.D.; Leonard, S.; Stevens, K.E.; Freedman, R. Perinatal Choline Effects on Neonatal Pathophysiology Related to Later Schizophrenia Risk. Am. J. Psychiatry 2013, 170, 290–298. [Google Scholar] [CrossRef] [PubMed]
- Cheatham, C.L.; Goldman, B.D.; Fischer, L.M.; da Costa, K.-A.; Reznick, J.S.; Zeisel, S.H. Phosphatidylcholine Supplementation in Pregnant Women Consuming Moderate-Choline Diets Does Not Enhance Infant Cognitive Function: A Randomized, Double-Blind, Placebo-Controlled Trial. Am. J. Clin. Nutr. 2012, 96, 1465–1472. [Google Scholar] [CrossRef]
| Identification | Location | Sample | Method Used to Determine Prenatal Folate Level | Maternal Folate Assessment | Dose of Folic Acid Intake or Mean Levels in Blood | Offspring’s/Children’s Age at the Time of Assessment | Offspring’s/Children’s Assessment | Main Results |
|---|---|---|---|---|---|---|---|---|
| Animal Studies | ||||||||
| [48] Craciunescu et al., 2010 | United States | Fetal mice | Diet supplementation | Embryonic days 11–17 | Control diet: 2 mg folic acid/kg diet; 1.1 g choline chloride/kg diet | Embryonic day 17 | Histological and immune-histochemical assays of fetal brain regions of interest | Folate-deficient mice had fewer neural progenitor cells undergoing mitosis in the septum and greater apoptosis rates in the septum and hippocampus compared to the control mice |
| Folate-deficient diet: 10.0 mg folic acid/kg diet, 1 g choline chloride/kg diet | ||||||||
| Folate-deficient choline-supplemented diet: 0.0 mg folic acid/kg diet, 4.95 g choline chloride/kg diet | ||||||||
| [68] Jadavji et al., 2015 | Canada | Methylenetetrahydrofolate reductase (MTHFR)-deficient male mice | Diet supplementation | 6 weeks prior to breeding until the end of lactation | Folate-deficient diet: 0.3 mg folic acid/kg diet | 3-week-old male mice | Novel object recognition task, Y-maze task | Maternal folate deficiency in mice was associated with short-term memory impairment in offspring |
| Folate sufficient diet: 2 mg folic acid/kg diet | ||||||||
| [69] Ferguson et al., 2005 | United States | 20 female mice per group. 4 males and 4 females/litter were retained for initial behavioral testing. 1 male and 1 female pup/litter were used in postweaning behavioral testing | Diet supplementation | 8 weeks prior to breeding until the end of gestation | Folate-deficient diets: (1) 400 nmol folic acid/kg diet and (2) 600 nmol folic acid/kg diet | Postnatal days 4–83 | Righting reflex, negative geotaxis, forelimb hanging, motor coordination using Rotarod apparatus, open field activity, elevated plus maze | Maternal folate deficiency in mice produced offspring who exhibited more anxiety-related behavior in the elevated plus maze |
| Folate sufficient diet: 1200 nmol folic acid/kg diet | ||||||||
| Human Studies | ||||||||
| [70] Julvez et al., 2009 | Spain | 420 mother-child pairs | Maternal self-report questionnaire used to determine whether supplements containing folic acid were taken | First trimester | No or yes | 4 years of age | McCarthy Scales of Children’s Abilities, California Preschool Social Competence Scale | Verbal, motor-executive function, verbal-executive function, social competence and inattention symptom scores were positively associated with maternal use of folic acid supplements |
| [71] Wehby and Murray 2008 | United States | 6774 mother-child pairs | National survey that included data as to whether supplements containing folic acid had been taken | First trimester | No or yes | 3 years of age | 16 items from the Denver Developmental Screening Test | Prenatal folic acid supplementation had a positive effect on children’s overall cognitive and gross motor development |
| [72] McNulty et al., 2019 | United Kingdom | 37 mother-child pairs in the treatment group and 33 mother-child pairs in the placebo group | Supplements or placebos distributed to the women in 7-day pillboxes | 14 weeks gestation until the birth of the child | 400 ug/day supplement containing folic acid | 7 years of age | Wechsler Preschool and Primary Scale of Intelligence, Third Edition, UK Edition (WPPSI-III) | Children of mothers who were in the treatment group had higher scores on the WPPSI-III compared to the children of mothers who were given a placebo |
| Placebo containing no folic acid | ||||||||
| [74] Chatzi et al., 2012 | Greece | 553 mother-child pairs | A questionnaire was administered by a trained research nurse that asked whether the women had taken a folic acid supplement since they became pregnant. Supplement users reported the brand name, the dose, and the frequency of intake, which was converted into a measure of daily intake. | 14–18 weeks gestation | No folic acid intake from supplements | 18 months of age | Bayley Scales of Infant and Toddler Development, Third Edition (Bayley-III) | Children of mothers who reported taking a daily supplement of 5 mg of folic acid or more had a 5 unit increase on receptive communication and a 3.5 unit increase in expressive communication |
| Daily intake of 5 mg of folic acid from supplements | ||||||||
| Daily intake of folic acid from supplements higher than 5 mg | ||||||||
| [75] Chen et al., 2021 | China | 32 cohort studies and 7 case–control studies | Systematic review and meta-analysis of research articles that examined the association between prenatal folic acid supplementation and postnatal neurodevelopmental outcomes. All studies discussed folic acid supplementation only | During pregnancy | Varied by study; some reported whether or not women were supplemented; others reported specific supplementation levels. | 18 months to 17 years | Varied by study; different measures used to assess intelligence, risk of autistic traits, ADHD, behavior, language, and psychomotor problems | Appropriate maternal folic acid supplementation may have positive effects on children’s intelligence and development, and reduce the risk of autism traits, ADHD, and behavioral and language problems |
| [76] Villamor et al., 2012 | United States | 1210 mother-child pairs | Food frequency questionnaires | First and second trimester | Mean estimated folate intake of 949 ± 390 ug/day | 3 years of age | Peabody Picture Vocabulary Test, Third Edition (PPVT-III), Wide Range Assessment of Visual Motor Abilities | For each 600 μg per day increase in total folate intake during the first trimester of pregnancy there was a 1.6-point increase in scores on the PPVT-III in the children |
| [77] Boeke et al., 2013 | United States | 895 mother-child pairs | Food frequency questionnaires | First and second trimesters | Mean daily estimated folate intake of 972 ug/day in the first trimester and 1268 ug/day in the second trimesters | 7 years of age | Wide Range Assessment of Memory and Learning, Second Edition (WRAML2), Kaufman Brief Intelligence Test, Second Edition (KBIT-2) | No associations were found between maternal folate intake and child cognitive outcomes |
| [85] Veena et al., 2010 | India | 536 mother-child pairs | Maternal plasma/serum folate concentrations from blood samples | 28–32 weeks gestation | Mean plasma/serum folate concentrations of 34.7 nmol/L | 9 or 10 years of age | Kaufman Assessment Battery for Children | Positive association between maternal plasma/serum folate concentrations and children’s performance on the Kaufman Assessment Battery for Children |
| [88] Ars et al., 2019 | Netherlands | 256 mother-child pairs (62 in the low folate group and 194 in the normal folate group) | Maternal plasma/serum folate concentrations from venous blood samples | First trimester | Low folate group: plasma/serum folate levels below 8 nmol/L | 6 years of age | Neuroimaging and NEPSY-II-NL | Low maternal plasma/serum folate concentrations below 8 nmol/L were associated with smaller total brain volume and poorer language and visuospatial skills in children |
| Normal folate group: plasma/serum folate levels above 8 nmol/L | ||||||||
| [89] Wu et al., 2012 | United States | 154 mother-child pairs | Food frequency questionnaires and maternal plasma/serum folate concentrations from blood samples | 16 weeks gestation | Mean plasma/serum folate concentrations of 36.4 nmol/L | 18 months of age | Bayley Scales of Infant Development, Third Edition (Bayley-III) | No association found between maternal plasma/serum folate concentrations and child cognitive function |
| [90] Tamura et al., 2005 | United States | 355 mother-child pairs | Maternal red blood cell folate concentrations from blood samples | Second and third trimesters | Mean red blood cell folate concentrations were 873 nmol/L, 1070 nmol/L, and 1096 nmol/L at 19, 26, and 37 weeks gestation, respectively | 5 years of age | Differential Ability Scales, Visual and Auditory Sequential Memory Tests, Knox Cube Test, Gross Motor Scale, and Grooved Pegboard Test | No association found between maternal red blood cell folate concentrations and child cognitive function |
| Identification | Location | Sample | Method Used to Determine Prenatal Choline Level | Maternal Choline Assessment | Dose of Choline Intake or Mean Levels in Blood | Offspring’s/Children’s Age at the Time of Assessment | Offspring’s/Children’s Assessment | Main Results |
|---|---|---|---|---|---|---|---|---|
| Animal Studies | ||||||||
| [94] Meck et al., 1988 | United States | 16 male albino rats from 8 pregnant female rats | Diet supplementation | 2 days prior to conception until birth of offspring | Control diet: 50 mM saccharin | 60 days of age | 12 and 18 arm radial maze task | Offspring of choline-supplemented rats had enhanced visuospatial memory skills as compared to offspring of non-choline-supplemented rats |
| Choline supplemented diet: 50 mM saccharin containing 5 mL/L choline chloride | ||||||||
| [95] Glenn et al., 2008 | United States | 20 rats (10 supplemented, 10 not supplemented) | Diet supplementation | Embryonic days 12–17 | Control diet: 1.1 g/kg choline chloride | 1 and 24 months of age | Open field exploration, novel object exploration, BrdU immuno-histochemistry and unbiased stereology to assess hippocampal plasticity markers | Prenatal choline supplementation was positively associated with exploratory behavior in rats and preserved some features of hippocampal plasticity in offspring over a 2-year time span |
| 8 male and 8 female pups from each experimental group were used in the behavioral tests | Choline-supplemented diet: 5 g/kg choline chloride | |||||||
| [96] Meck and Williams 1999 | United States | 30 offspring of 18 pregnant rats | Supplementation in drinking water | Embryonic days 11–18 | Choline-deficient diet: no choline or saccharin | 120 days of age | Twelve arm radial maze | Choline-supplemented and choline-deficient rats performed more accurately than control rats during spaced trials. Choline-supplemented rats displayed less proactive interference during massed trials compared to control and choline-deficient rats |
| Control diet: 50 mM saccharin | ||||||||
| Choline-supplemented diet: 50 mM saccharin and 25 mM choline chloride | ||||||||
| [97]. Meck and Williams 1997 | United States | 34 pregnant dams that produced 128 adult female rats | Supplementation in drinking water | Embryonic days 12–17 | Control diet: 50 mM saccharin | 60 days of age | 6, 12, 18, 24 radial arm mazes | Rats treated perinatally with choline had a higher threshold for implementing a chunking strategy in the radial arm maze tasks |
| Supplemented diet: 50 mM saccharin and 25 mM choline chloride | ||||||||
| [98] Meck and Williams 1997 | United States | 30 offspring of 18 pregnant rats | Supplementation in drinking water | Embryonic days 11–18 | Choline-deficient diet: no choline or saccharin | 4–6 months of age and 24–26 months of age | Peak-interval timing procedure | Prenatal choline supplementation was positively associated with cognitive function in offspring and choline deficiency was positively associated with impaired divided attention and accelerated age-related declines in temporal processing |
| Control diet: 50 mM saccharin | ||||||||
| Choline-supplemented diet: 50 mM saccharin and 25 mM choline chloride | ||||||||
| Human Studies | ||||||||
| [77] Boeke et al., 2013 | United States | 895 mother-child pairs | Food frequency questionnaires | First and second trimesters | Mean estimated daily choline intake was 328 mg/day | 7 years of age | Wide Range Assessment of Memory and Learning, Second Edition (WRAML2), Kaufman Brief Intelligence Test, Second Edition (KBIT-2) | Prenatal choline intake was positively associated with memory scores, but not intelligence scores |
| [89] Wu et al., 2012 | United States | 154 mother-child pairs | Food frequency questionnaires; measured maternal plasma-free choline concentrations from blood samples | 16 weeks gestation | Mean plasma-free choline concentration of the women was 7.07 umol/L; mean estimated daily choline intake was 383 mg/day | 18 months of age | Bayley Scales of Infant Development, Third Edition (Bayley-III) | Positive associations were found between maternal plasma free choline concentrations during pregnancy and infant cognitive development |
| [99] Caudill et al., 2018 | United States | 12 mother-child pairs in the control group and 12 mother-child pairs in the treatment group | Supplement mixed in juice consumed at the study facility | 27 weeks gestation until the birth of the offspring | 480 mg choline/day or 930 mg choline/day | ∼4, 7, 10, and 13 months of age | Visual attention task | Infants born to women supplemented with 930 mg of choline chloride per day compared to infants of women, who received 480 mg of choline per day, displayed higher information processing speed |
| [100] Signore et al., 2008 | United States | 404 mother-child pairs | Maternal free and total serum choline concentrations from blood samples | 16 to 18 weeks, 24 to 26 weeks, 30 to 32 weeks, and 36 to 38 weeks gestation | Median free choline concentrations increased from 9.34 to 10.10 umol/L over the gestational period; median total choline concentrations increased from 2.57 to 2.75 mmol/L over the gestational period | 5 years of age | Wechsler Preschool and Primary Scales of Intelligence-Revised (WPPSI-R) | No associations found between maternal prenatal choline concentrations and offspring WISC-R Full-Scale IQ, visuospatial processing, or memory |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Irvine, N.; England-Mason, G.; Field, C.J.; Dewey, D.; Aghajafari, F. Prenatal Folate and Choline Levels and Brain and Cognitive Development in Children: A Critical Narrative Review. Nutrients 2022, 14, 364. https://doi.org/10.3390/nu14020364
Irvine N, England-Mason G, Field CJ, Dewey D, Aghajafari F. Prenatal Folate and Choline Levels and Brain and Cognitive Development in Children: A Critical Narrative Review. Nutrients. 2022; 14(2):364. https://doi.org/10.3390/nu14020364
Chicago/Turabian StyleIrvine, Nathalie, Gillian England-Mason, Catherine J. Field, Deborah Dewey, and Fariba Aghajafari. 2022. "Prenatal Folate and Choline Levels and Brain and Cognitive Development in Children: A Critical Narrative Review" Nutrients 14, no. 2: 364. https://doi.org/10.3390/nu14020364
APA StyleIrvine, N., England-Mason, G., Field, C. J., Dewey, D., & Aghajafari, F. (2022). Prenatal Folate and Choline Levels and Brain and Cognitive Development in Children: A Critical Narrative Review. Nutrients, 14(2), 364. https://doi.org/10.3390/nu14020364







