Effect of Docosahexaenoic Acid and Eicosapentaenoic Acid Supplementation on Sleep Quality in Healthy Subjects: A Randomized, Double-Blinded, Placebo-Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Ethics Statements
2.3. Participants
2.4. Randomization and Blinding
2.5. Treatment
2.6. Subjective Evaluation
2.6.1. OSA-MA
2.6.2. POMS2
2.7. Objective Evaluation
Sleep State Test
2.8. Blood Sampling
2.9. Dietary Survey
2.10. Safety Evaluation
2.11. Sample Size
2.12. Statistical Analysis
3. Results
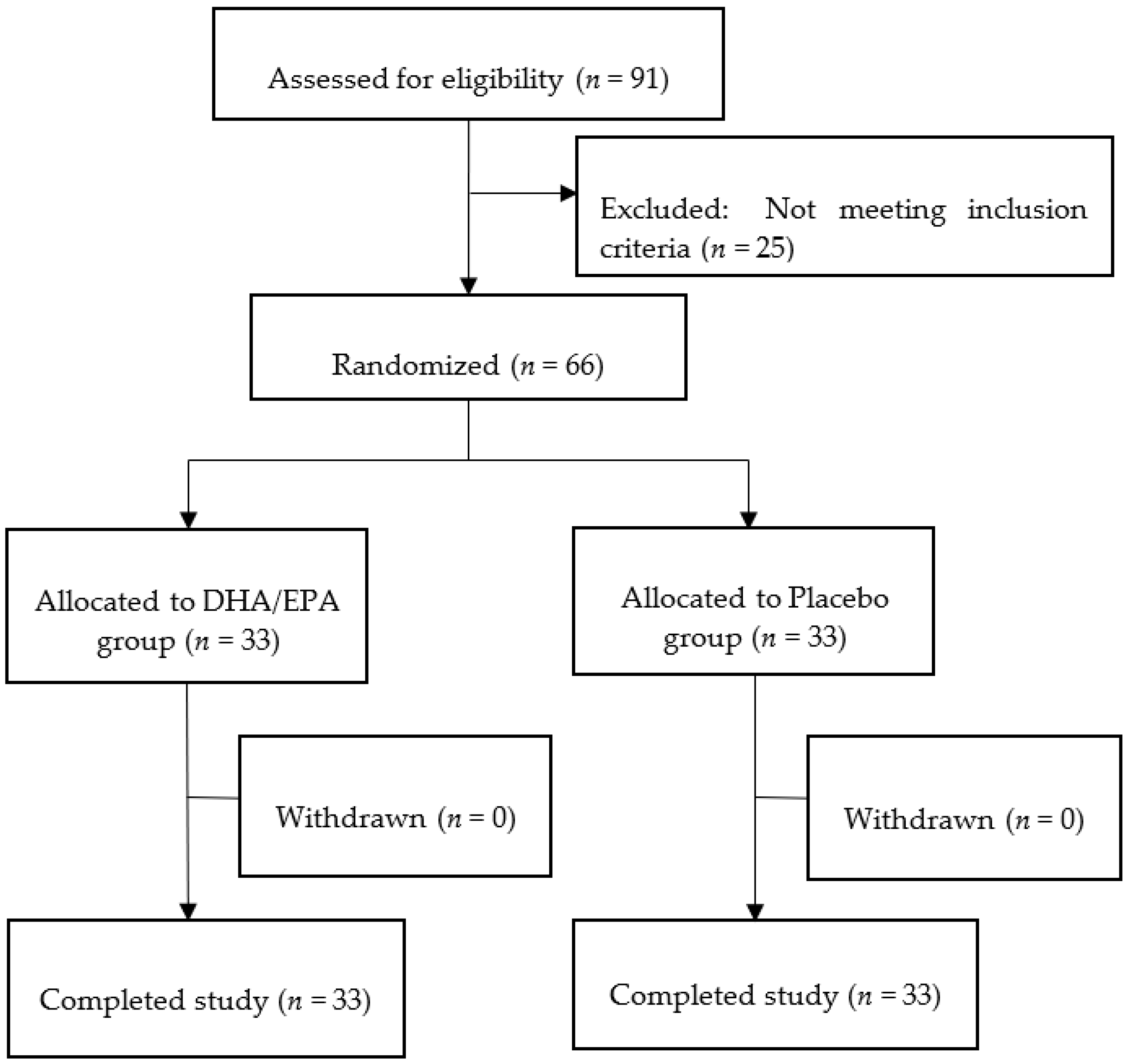
3.1. Participants
3.2. Subjective Evaluation
3.3. Objective Evaluation
3.4. Adverse Events
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- National Sleep Foundation. What Is Sleep Quality? Available online: https://www.thensf.org/what-is-sleep-quality/ (accessed on 29 September 2022).
- Nelson, K.L.; Davis, J.E.; Corbett, C.F. Sleep quality: An evolutionary concept analysis. Nurs. Forum. 2022, 57, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kim, J.H.; Chung, J.H. The association between sleep quality and quality of life: A population-based study. Sleep Med. 2021, 84, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Zammit, G.K.; Weiner, J.; Damato, N.; Sillup, G.P.; McMillan, C.A. Quality of life in people with insomnia. Sleep 1999, 22, S379–S385. [Google Scholar] [PubMed]
- Sella, E.; Miola, L.; Toffalini, E.; Borella, E. The relationship between sleep quality and quality of life in aging: A systematic review and meta-analysis. Health Psychol. Rev. 2021, 1–23. [Google Scholar] [CrossRef]
- Cappuccio, F.P.; D’Elia, L.; Strazzullo, P.; Miller, M.A. Quantity and quality of sleep and incidence of type 2 diabetes: A systematic review and meta-analysis. Diabetes Care 2010, 33, 414–420. [Google Scholar] [CrossRef]
- Hayashino, Y.; Fukuhara, S.; Suzukamo, Y.; Okamura, T.; Tanaka, T.; Ueshima, H.; the HIPOP-OHP Research group. Relation between sleep quality and quantity, quality of life, and risk of developing diabetes in healthy workers in Japan: The High-risk and Population Strategy for Occupational Health Promotion (HIPOP-OHP) Study. BMC Public Health. 2007, 7, 129. [Google Scholar] [CrossRef]
- Baglioni, C.; Battagliese, G.; Feige, B.; Spiegelhalder, K.; Nissen, C.; Voderholzer, U.; Lombardo, C.; Riemann, D. Insomnia as a predictor of depression: A meta-analytic evaluation of longitudinal epidemiological studies. J. Affect. Disord. 2011, 135, 10–19. [Google Scholar] [CrossRef]
- Yokoyama, E.; Kaneita, Y.; Saito, Y.; Uchiyama, M.; Matsuzaki, Y.; Tamaki, T.; Munezawa, T.; Ohida, T. Association between depression and insomnia subtypes: A longitudinal study on the elderly in Japan. Sleep 2010, 33, 1693–1702. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Health People 2020. Available online: https://www.cdc.gov/nchs/healthy_people/hp2020.htm (accessed on 26 September 2022).
- Ministry of Health, Labour and Welfare, Japan. Sleep Guidelines for Health Promotion 2014. Available online: https://www.mhlw.go.jp/stf/seisakunitsuite/bunya/kenkou_iryou/kenkou/suimin/index.html (accessed on 26 September 2022).
- Hattori, A. Melatonin and aging. Comp. Physiol. Biochem. 2017, 34, 2–11. [Google Scholar] [CrossRef]
- Garfinkel, D.; Laudon, M.; Nof, D.; Zisapel, N. Improvement of sleep quality in elderly people by controlled-release melatonin. Lancet 1995, 346, 541–544. [Google Scholar] [CrossRef]
- Espiritu, J.R.D. Aging-related sleep changes. Clin. Geriatr. Med. 2008, 24, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Alia, K.; Cathy, A. Sleep and quality of life in older people. In Sleep and Quality of Life in Clinical Medicine; Humana Press: Totowa, NJ, USA, 2008; pp. 131–138. [Google Scholar] [CrossRef]
- Yuzo, F.; Tomoko, T.; Hideoki, O.; Hiroshige, I. Effects of a beverage containing eicosapentaenoic acid and docosahexaenoic acid on the reduction of blood triglyceride levels and safety in adult male and female subjects with elevated blood triglyceride levels after 12 weeks of continuous intake. J. Jpn. Soc. Clin. Nutr. 2011, 33, 120–135. [Google Scholar]
- Yokoyama, M.; Origasa, H.; Matsuzaki, M.; Matsuzawa, Y.; Saito, Y.; Ishikawa, Y.; Oikawa, S.; Sasaki, J.; Hishida, H.; Itakura, H.; et al. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): A randomised open-label, blinded endpoint analysis. Lancet 2007, 369, 1090–1098. [Google Scholar] [CrossRef]
- Yurko-Mauro, K.; McCarthy, D.; Rom, D.; Nelson, E.B.; Ryan, A.S.; Blackwell, A.; Salem, N.; Stedman, M.; MIDAS Investigators. Beneficial effects of docosahexaenoic acid on cognition in age-related cognitive decline. Alzheimer’s Dement. 2010, 6, 456–464. [Google Scholar] [CrossRef]
- Nilsson, A.; Radeborg, K.; Salo, I.; Björck, I. Effects of supplementation with n-3 polyunsaturated fatty acids on cognitive performance and cardiometabolic risk markers in healthy 51 to 72 years old subjects: A randomized controlled cross-over study. Nutr. J. 2012, 11, 99. [Google Scholar] [CrossRef]
- Bhatt, D.L.; Steg, P.G.; Miller, M.; Brinton, E.A.; Jacobson, T.A.; Ketchum, S.B.; Doyle, R.T.; Juliano, R.A.; Jiao, L.; Granowitz, C.; et al. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N. Engl. J. Med. 2019, 380, 11–22. [Google Scholar] [CrossRef]
- Hu, Y.; Hu, F.B.; Manson, J.E. Marine Omega-3 supplementation and cardiovascular disease: An updated meta-analysis of 13 randomized controlled trials involving 127 477 participants. J. Am. Heart Assoc. 2019, 8, e013543. [Google Scholar] [CrossRef]
- Delgado-Lista, J.; Perez-Martinez, P.; Lopez-Miranda, J.; Perez-Jimenez, F. Long chain omega-3 fatty acids and cardiovascular disease: A systematic review. Br. J. Nutr. 2012, 107, S201–S213. [Google Scholar] [CrossRef]
- Liao, Y.; Xie, B.; Zhang, H.; He, Q.; Guo, L.; Subramanieapillai, M.; Fan, B.; Lu, C.; McIntyre, R.S. Efficacy of omega-3 PUFAs in depression: A meta-analysis. Transl. Psychiatry 2019, 9, 190. [Google Scholar] [CrossRef]
- Hashimoto, M.; Hossain, M.S.; Yamasaki, H.; Yazawa, K.; Masumura, S. Effects of eicosapentaenoic acid and docosahexaenoic acid on plasma membrane fluidity of aortic endothelial cells. Lipids 1999, 34, 1297–1304. [Google Scholar] [CrossRef]
- Sherratt, S.C.R.; Mason, R.P. Eicosapentaenoic Acid and Docosahexaenoic Acid Have Distinct Membrane Locations and Lipid Interactions as Determined by X-ray Diffraction. Chem. Phys. Lipids 2018, 212, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Sherratt, S.C.R.; Juliano, R.A.; Copland, C.; Bhatt, D.L.; Libby, P.; Mason, R.P. EPA and DHA containing phospholipids have contrasting effects on membrane structure. J. Lipid Res. 2021, 62, 100106. [Google Scholar] [CrossRef] [PubMed]
- Stillwell, W.; Wassall, S.R. Docosahexaenoic acid: Membrane properties of a unique fatty acid. Chem. Phys. Lipids 2003, 126, 1–27. [Google Scholar] [CrossRef]
- Serhan, C.N.; Gotlinger, K.; Hong, S.; Arita, M. Resolvins, docosatrienes, and neuroprotectins, novel omega-3-derived mediators, and their aspirin-triggered endogenous epimers: An overview of their protective roles in catabasis. Prostaglandins Other Lipid Mediat. 2004, 73, 155–172. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Lu, Y.; Yang, R.; Gotlinger, K.H.; Petasis, N.A.; Serhan, C.N. Resolvin D1, protectin D1, and related docosahexaenoic acid-derived products: Analysis via electrospray/low energy tandem mass spectrometry based on spectra and fragmentation mechanisms. J. Am. Soc. Mass Spectrom. 2007, 18, 128–144. [Google Scholar] [CrossRef]
- Schwarz, D.; Kisselev, P.; Ericksen, S.S.; Szklarz, G.D.; Chernogolov, A.; Honeck, H.; Schunck, W.-H.; Roots, I. Arachidonic and eicosapentaenoic acid metabolism by human CYP1A1: Highly stereoselective formation of 17(R),18(S)-epoxyeicosatetraenoic acid. Biochem. Pharmacol. 2004, 67, 1445–1457. [Google Scholar] [CrossRef]
- Lavialle, M.; Champeil-Potokar, G.; Alessandri, J.M.; Balasse, L.; Guesnet, P.; Papillon, C.; Pévet, P.; Vancassel, S.; Vivien-Roels, B.; Denis, I. An (n-3) polyunsaturated fatty acid-deficient diet disturbs daily locomotor activity, melatonin rhythm, and striatal dopamine in Syrian hamsters. J. Nutr. 2008, 138, 1719–1724. [Google Scholar] [CrossRef]
- Sarda, N.; Gazzah, N.; Gharib, A.; Moliere, P.; Durand, G.; Lagarde, M. Dietary n-3 fatty acids modulate the melatonin in the rat pineal gland. In Proceedings of the 8th International Conference on Prostaglandins and Related Compounds, Montreal, QC, Canada, 26–31 July 1992; Volume 38, p. 12. [Google Scholar]
- Del Brutto, O.H.; Mera, R.M.; Ha, J.-E.; Gillman, J.; Zambrano, M.; Castillo, P.R. Dietary fish intake and sleep quality: A population-based study. Sleep Med. 2016, 17, 126–128. [Google Scholar] [CrossRef]
- Katagiri, R.; Asakura, K.; Kobayashi, S.; Suga, H.; Sasaki, S.; the Three-generation Study of Women on Diets and Health Study Group. Low intake of vegetables, high intake of confectionary, and unhealthy eating habits are associated with poor sleep quality among middle-aged female Japanese workers. J. Occup. Health 2014, 56, 359–368. [Google Scholar] [CrossRef]
- Judge, M.P.; Cong, X.; Harel, O.; Courville, A.B.; Lammi-Keefe, C.J. Maternal consumption of a DHA-containing functional food benefits infant sleep patterning: An early neurodevelopmental measure. Early Hum. Dev. 2012, 88, 531–537. [Google Scholar] [CrossRef]
- Patan, M.J.; Kennedy, D.O.; Husberg, C.; Hustvedt, S.O.; Calder, P.C.; Middleton, B.; Khan, J.; Forster, J.; Jackson, P.A. Differential effects of DHA- and EPA-rich oils on sleep in healthy young adults: A randomized controlled trial. Nutrients 2021, 13, 248. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, Y.; Tanaka, H.; Takase, M.M.; Yamazaki, K.; Azumi, K.; Shirakawa, S. Standardization of revised version of OSA sleep inventory for middle age and aged. Brain Sci. Ment. Disord. 1999, 10, 401–409. [Google Scholar]
- Heuchert, J.P.; McNair, D.M. Profile of Mood States, 2nd ed.; Multi-Health Systems Inc.: North Tonawanda, NY, USA, 2012. [Google Scholar]
- Yokoyama, K.; Watanabe, K. Japanese Translation of POMS 2: Profile of Mood States, 2nd ed.; Kaneko Shobo: Tokyo, Japan, 2015. (In Japanese) [Google Scholar]
- Yamamoto, M.; Iga, T.; Shimizu, M.; Kohara, S.; Yoshii, F.; Urano, T.; Aoki, T.; Abe, T. Evaluation of the usefulness of mat-type sleep monitor. Sleep Med. Jpn. 2012, 6, 473–480. [Google Scholar]
- Yoshii, F.; Shimizu, M.; Kohara, S.; Yamamoto, M.; Iga, T.; Sasaki, T.; Honda, Y.; Yamaya, C. Development of mat-type sleep monitor and evaluation of sleep status by using sleep score. Sleep Med. Jpn. 2012, 6, 361–365. [Google Scholar]
- Suzuki, N.; Baba, A.; Kakinuma, T.; Sano, Y.; Tanaka, M.; Ouchi, S.; Watanabe, T.; Yamamoto, K. A novel dietary questionnaire: The Calorie and Nutrition Diary (CAND). New Food Indust. 2019, 61, 721–732. [Google Scholar]
- Montgomery, P.; Burton, J.R.; Sewell, R.P.; Spreckelsen, T.F.; Richardson, A.J. Fatty acids and sleep in UK children: Subjective and pilot objective sleep results from the DOLAB study—A randomized controlled trial. J. Sleep Res. 2014, 23, 364–388. [Google Scholar] [CrossRef]
- Cohen, J. A power primer. Psychol. Bull. 1992, 112, 155–159. [Google Scholar] [CrossRef]
- Boone, K.M.; Rausch, J.; Pelak, G.; Li, R.; Turner, A.N.; Klebanoff, M.A.; Keim, S.A. Docosahexaenoic acid and arachidonic acid supplementation and sleep in toddlers born preterm: Secondary analysis of a randomized clinical trial. J. Clin. Sleep Med. 2019, 15, 1197–1208. [Google Scholar] [CrossRef]
- Hysing, M.; Kvestad, I.; Kjellevold, M.; Kolden Midtbø, L.; Graff, I.E.; Lie, Ø.; Hurum, H.; Stormark, K.M.; Øyen, J. Fatty fish intake and the effect on mental health and sleep in preschool children in FINS-KIDS, a randomized controlled trial. Nutrients 2018, 10, 1478. [Google Scholar] [CrossRef]
- Dai, Y.; Liu, J. Omega-3 long-chain polyunsaturated fatty acid and sleep: A systematic review and meta-analysis of randomized controlled trials and longitudinal studies. Nutr. Rev. 2021, 79, 847–868. [Google Scholar] [CrossRef]
- Hansen, A.L.; Dahl, L.; Olson, G.; Thornton, D.; Graff, I.E.; Frøyland, L.; Thayer, J.F.; Pallesen, S. Fish consumption, sleep, daily functioning, and heart rate variability. J. Clin. Sleep Med. 2014, 10, 567–575. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, N.; Matsuoka, Y.; Kumachi, M.; Hamazaki, K.; Horikoshi, M.; Furukawa, T.A. Omega-3 fatty acids for a better mental state in working populations—Happy Nurse Project: A 52-week randomized controlled trial. J. Psychiatr. Res. 2018, 102, 72–80. [Google Scholar] [CrossRef]
- Doornbos, B.; van Goor, S.A.; Dijck-Brouwer, D.A.J.; Schaafsma, A.; Korf, J.; Muskiet, F.A.J. Supplementation of a low dose of DHA or DHA AA does not prevent peripartum depressive symptoms in a small population based sample. Prog. Neuropsychopharmacol. Biol. Psychiatry 2009, 33, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Judge, M.P.; Beck, C.T.; Durham, H.; McKelvey, M.M.; Lammi-Keefe, C.J. Pilot trial evaluating maternal docosahexaenoic acid consumption during pregnancy: Decreased postpartum depressive symptomatology. Int. J. Nurs. Sci. 2014, 1, 339–345. [Google Scholar] [CrossRef][Green Version]
- Cohen, L.S.; Joffe, H.; Guthrie, K.A.; Ensrud, K.E.; Freeman, M.; Carpenter, J.S.; Learman, L.A.; Newton, K.M.; Reed, S.D.; Manson, J.E.; et al. Efficacy of omega-3 for vasomotor symptoms treatment: A randomized controlled trial. Menopause 2014, 21, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Ladesich, J.B.; Pottala, J.V.; Romaker, A.; Harris, W.S. Membrane level of omega-3 docosahexaenoic acid is associated with severity of obstructive sleep apnea. J. Clin. Sleep Med. 2011, 7, 391–396. [Google Scholar] [CrossRef]
- Tan, Z.S.; Harris, W.S.; Beiser, A.S.; Au, R.; Himali, J.J.; Debette, S.; Pikula, A.; Decarli, C.; Wolf, P.A.; Vasan, R.S.; et al. Red blood cell omega-3 fatty acid levels and markers of accelerated brain aging. Neurology 2012, 78, 658–664. [Google Scholar] [CrossRef]
- Hamazaki-Fujita, N.; Hamazaki, K.; Tohno, H.; Itomura, M.; Terashima, Y.; Hamazaki, T.; Nakamura, N.; Yomoda, S. Polyunsaturated fatty acids and blood circulation in the forebrain during a mental arithmetic task. Brain Res. 2011, 1397, 38–45. [Google Scholar] [CrossRef]
- Shirakawa, S. How to Use OSA Sleep Questionnaire MA Version. Available online: https://www.jobs.gr.jp/osa_ma.html (accessed on 25 September 2022). (In Japanese).
- Weller, L.; Avinir, O. Hassles, uplifts, and quality of sleep. Percept. Mot. Ski. 1993, 76, 571–576. [Google Scholar] [CrossRef]
- Fruhstorfer, B.; Fruhstorfer, H.; Grass, P. Daytime noise and subsequent night sleep in man. Eur. J. Appl. Physiol. Occup. Physiol. 1984, 53, 159–163. [Google Scholar] [CrossRef]
- Pasternak, R.E.; Reynolds, C.F.; Hoch, C.C.; Buysse, D.S.; Schlernitzauer, M.; Machen, M.; Kupfer, D.J. Sleep in spousally bereaved elders with subsyndromal depressive symptoms. Psychiatry Res. 1992, 43, 43–53. [Google Scholar] [CrossRef]
- Reynolds, C.F.; Hoch, C.C.; Buysse, D.J.; Houck, P.R.; Schlernitzauer, M.; Frank, E.; Mazumdar, S.; Kupfer, D.J. Electroencephalographic sleep in spousal bereavement and bereavement related depression of late life. Biol. Psychiatry 1992, 31, 69–82. [Google Scholar] [CrossRef]
- Cartwright, R.D.; Wood, E. Adjustment disorders of sleep: The sleep effects of a major stressful event and its resolution. Psychiatry Res. 1991, 39, 199–209. [Google Scholar] [CrossRef]
- Owens, J.F.; Matthews, K.A. Sleep disturbance in healthy middle aged women. Maturitas 1998, 30, 41–50. [Google Scholar] [CrossRef]
- Kim, K.; Uchiyama, M.; Okawa, M.; Liu, X.; Ogihara, R. An epidemiological study of insomnia among the Japanese general population. Sleep 2000, 23, 41–47. [Google Scholar] [CrossRef]
- Konishi, Y.; Nakanishi, J.; Nishida, K. The usefulness of Mat-Type Sleeping Monitor in elderly people’s Automatic sleep/wake identification judging―From comparison with a Actigraph. Bull. Ehime Prefect. Univ. Health Sci. 2012, 9, 5–9. (In Japanese) [Google Scholar]
- Kawai, H.; Togashi, Y.; Ishibashi, T.; Iwadate, R.; Mitsumoto, A. The Efficacy of a Mattress Type Sleep Measuring Device in Analyzing Sleep in Healthy University Students: Comparison with Actigraphy. BPB Rep. 2019, 2, 125–129. [Google Scholar] [CrossRef]
- Zaouali-Ajina, M.; Gharib, A.; Durand, G.; Gazzah, N.; Claustrat, B.; Gharib, C.; Sarda, N. Dietary docosahexaenoic acid-enriched phospholipids normalize urinary melatonin excretion in adult (n-3) polyunsaturated fatty acid-deficient rats. J. Nutr. 1999, 129, 2074–2080. [Google Scholar] [CrossRef]
- Xin, W.; Wei, W.; Li, X.-Y. Short-term effects of fish-oil supplementation on heart rate variability in humans: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2013, 97, 926–935. [Google Scholar] [CrossRef]
- Baharav, A.; Kotagal, S.; Gibbons, V.; Rubin, B.K.; Pratt, G.; Karin, J.; Akselrod, S. Fluctuations in autonomic nervous activity during sleep displayed by power spectrum analysis of heart rate variability. Neurology 1995, 45, 1183–1187. [Google Scholar] [CrossRef]
- Matsumura, H.; Honda, K.; Goh, Y.; Ueno, R.; Sakai, T.; Inoué, S.; Hayaishi, O. Awaking effect of prostaglandin E2 in freely moving rats. Brain Res. 1989, 481, 242–249. [Google Scholar] [CrossRef]
- Moonen, H.J.J.; Dommels, Y.E.M.; van Zwam, M.; van Herwijnen, M.H.M.; Kleinjans, J.C.S.; Alink, G.M.; de Kok, T.M.C.M. Effects of polyunsaturated fatty acids on prostaglandin synthesis and cyclooxygenase-mediated DNA adduct formation by heterocyclic aromatic amines in human adenocarcinoma colon cells. Mol. Carcinog. 2004, 40, 180–188. [Google Scholar] [CrossRef] [PubMed]
| Variable | DHA/EPA Group (n = 33) | Placebo Group (n = 33) | p Value |
|---|---|---|---|
| n (males/females) | 11/22 | 11/22 | 1.000 |
| Age (years) | 52.8 ± 5.9 | 52.8 ± 5.0 | 1.000 |
| Weight (kg) | 59.8 ± 11.5 | 58.3 ± 10.5 | 0.579 |
| Height (cm) | 162.6 ± 7.5 | 163.3 ± 6.4 | 0.689 |
| BMI (kg/m2) | 22.5 ± 3.3 | 21.8 ± 3.3 | 0.393 |
| Variable | Baseline | Week 12 | ||||
|---|---|---|---|---|---|---|
| DHA/EPA Group (n = 33) | Placebo Group (n = 33) | p Value | DHA/EPA Group (n = 33) | Placebo Group (n = 33) | p Value | |
| Arachidonic acid (µg/mL) | 228.8 ± 51.3 | 234.2 ± 57.9 | 0.687 | 204.0 ± 59.1 | 211.6 ± 53.2 | 0.588 |
| Eicosapentaenoic acid (µg/mL) | 52.0 ± 35.3 | 54.0 ± 38.4 | 0.824 | 79.2 ± 40.5 * | 56.3 ± 39.1 | 0.023 |
| Docosahexaenoic acid (µg/mL) | 114.0 ± 32.2 | 117.2 ± 31.3 | 0.680 | 137.5 ± 32.6 ** | 111.0 ± 35.4 | 0.002 |
| Variable | Baseline | Week 12 | ||||
|---|---|---|---|---|---|---|
| DHA/EPA Group (n = 33) | Placebo Group (n = 33) | p Value | DHA/EPA Group (n = 32) | Placebo Group (n = 32) | p Value | |
| Energy (kcal) | 2391.7 ± 714.6 | 2261.8 ± 459.5 | 0.383 | 2505.9 ± 745.9 | 2245.7 ± 453.4 | 0.143 |
| Protein (g) | 98.7 ± 35.1 | 95.6 ± 19.7 | 0.655 | 103.5 ± 34.1 | 95.3 ± 19.3 | 0.226 |
| Fat (g) | 86.2 ± 33.7 | 84.5 ± 22.2 | 0.811 | 90.8 ± 33.6 | 82.7 ± 20.3 | 0.165 |
| Carbohydrate (g) | 294.3 ± 77.4 | 268.5 ± 58.2 | 0.131 | 307.1 ± 90.4 | 269.2 ± 55.0 | 0.172 |
| Omega-3 fatty acid (g) | 2.6 ± 1.1 | 2.5 ± 0.6 | 0.831 | 2.7 ± 1.0 | 2.6 ± 0.7 | 0.545 |
| Eicosapentaenoic acid (mg) | 163.5 ± 97.2 | 150.9 ± 66.2 | 0.542 | 163.2 ± 94.3 | 164.1 ± 86.1 | 0.523 |
| Docosahexaenoic acid (mg) | 347.1 ± 180.9 | 337.1 ± 124.0 | 0.795 | 352.3 ± 175.7 | 357.9 ± 147.2 | 0.659 |
| Variable | Baseline | Week 12 | ||||
|---|---|---|---|---|---|---|
| DHA/EPA Group (n = 33) | Placebo Group (n = 33) | p Value | DHA/EPA Group (n = 33) | Placebo Group (n = 33) | p Value | |
| Sleepiness on rising | 14.3 ± 5.1 | 14.3 ± 5.1 | 0.998 | 17.7 ± 5.4 | 18.5 ± 5.5 | 0.488 |
| Initiation and maintenance of sleep | 12.9 ± 5.3 | 13.3 ± 4.3 | 0.792 | 16.7 ± 4.9 | 17.9 ± 5.0 | 0.351 |
| Frequent dreaming | 20.1 ± 8.0 | 21.0 ± 7.5 | 0.647 | 24.4 ± 4.1 * | 22.2 ± 6.4 | 0.043 |
| Refreshing on rising | 14.9 ± 3.9 | 15.0 ± 4.9 | 0.974 | 19.2 ± 5.5 | 20.0 ± 4.8 | 0.451 |
| Sleep length | 16.5 ± 4.7 | 15.1 ± 4.1 | 0.194 | 18.6 ± 4.6 | 19.2 ± 5.4 | 0.125 |
| Variable | Baseline | Week 12 | ||||
|---|---|---|---|---|---|---|
| DHA/EPA Group (n = 33) | Placebo Group (n = 33) | p Value | DHA/EPA Group (n = 33) | Placebo Group (n = 33) | p Value | |
| Anger–hostility | 46.8 ± 9.0 | 48.1 ± 12.3 | 0.955 | 47.1 ± 10.4 | 46.9 ± 8.5 | 0.996 |
| Confusion–bewilderment | 47.1 ± 8.9 | 47.4 ± 8.7 | 0.633 | 47.0 ± 9.4 | 47.5 ± 7.6 | 0.481 |
| Depression–dejection | 49.3 ± 9.5 | 48.3 ± 8.1 | 0.889 | 48.8 ± 8.4 | 48.0 ± 6.2 | 0.832 |
| Fatigue–inertia | 47.9 ± 8.4 | 46.7 ± 7.4 | 0.656 | 46.2 ± 9.2 | 46.3 ± 7.7 | 0.856 |
| Tension–anxiety | 50.4 ± 9.0 | 48.3 ± 9.0 | 0.537 | 48.7 ± 9.1 | 48.8 ± 8.5 | 0.677 |
| Vigor–activity | 52.8 ± 10.7 | 50.9 ± 9.7 | 0.356 | 54.4 ± 10.5 | 54.5 ± 9.6 | 0.332 |
| Friendliness | 53.0 ± 11.2 | 52.7 ± 10.0 | 0.458 | 53.9 ± 10.9 | 53.3 ± 9.7 | 0.414 |
| Total mood disturbance | 47.7 ± 8.6 | 47.6 ± 8.9 | 0.908 | 46.8 ± 9.2 | 46.7 ± 6.9 | 0.853 |
| Variable | Baseline | Week 12 | ||||
|---|---|---|---|---|---|---|
| DHA/EPA Group (n = 33) | Placebo Group (n = 33) | p Value | DHA/EPA Group (n = 32) | Placebo Group (n = 32) | p Value | |
| Sleep efficiency (%) | 95.6 ± 2.9 | 96.3 ± 3.1 | 0.401 | 96.0 ± 2.6 * | 93.7 ± 9.4 | 0.018 |
| Sleep latency (min) | 21.7 ± 12.1 | 20.9 ± 15.6 | 0.827 | 27.8 ± 13.5 | 21.5 ± 12.6 | 0.062 |
| Total sleep time (min) | 365.9 ± 63.9 | 384.0 ± 60.9 | 0.244 | 378.8 ± 90.4 | 400.6 ± 89.3 | 0.867 |
| Actual sleep time (min) | 351.7 ± 62.3 | 370.9 ± 58.4 | 0.202 | 364.2 ± 82.4 | 374.1 ± 68.2 | 0.468 |
| Average awakening length (min) | 14.2 ± 10.5 | 13.2 ± 12.8 | 0.715 | 14.6 ± 12.5 | 26.6 ± 53.9 | 0.134 |
| Average ratio of awakening (%) | 3.9 ± 2.9 | 3.2 ± 3.1 | 0.369 | 3.5 ± 2.5 * | 5.8 ± 9.4 | 0.018 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yokoi-Shimizu, K.; Yanagimoto, K.; Hayamizu, K. Effect of Docosahexaenoic Acid and Eicosapentaenoic Acid Supplementation on Sleep Quality in Healthy Subjects: A Randomized, Double-Blinded, Placebo-Controlled Trial. Nutrients 2022, 14, 4136. https://doi.org/10.3390/nu14194136
Yokoi-Shimizu K, Yanagimoto K, Hayamizu K. Effect of Docosahexaenoic Acid and Eicosapentaenoic Acid Supplementation on Sleep Quality in Healthy Subjects: A Randomized, Double-Blinded, Placebo-Controlled Trial. Nutrients. 2022; 14(19):4136. https://doi.org/10.3390/nu14194136
Chicago/Turabian StyleYokoi-Shimizu, Kaori, Kenichi Yanagimoto, and Kohsuke Hayamizu. 2022. "Effect of Docosahexaenoic Acid and Eicosapentaenoic Acid Supplementation on Sleep Quality in Healthy Subjects: A Randomized, Double-Blinded, Placebo-Controlled Trial" Nutrients 14, no. 19: 4136. https://doi.org/10.3390/nu14194136
APA StyleYokoi-Shimizu, K., Yanagimoto, K., & Hayamizu, K. (2022). Effect of Docosahexaenoic Acid and Eicosapentaenoic Acid Supplementation on Sleep Quality in Healthy Subjects: A Randomized, Double-Blinded, Placebo-Controlled Trial. Nutrients, 14(19), 4136. https://doi.org/10.3390/nu14194136




