The Impact of Nutrition-Based Interventions on Nutritional Status and Metabolic Health in Small Island Developing States: A Systematic Review and Narrative Synthesis
Abstract
1. Introduction
2. Methods
2.1. Eligibility Criteria
2.2. Search Strategy
2.3. Study Selection
2.4. Data Extraction
2.5. Risk of Bias
2.6. Results Synthesis
3. Results
3.1. Study Characteristics
3.1.1. Overview
3.1.2. Location and Design
| Ref. | Region/Country | Study Design | Nutrition/Metabolic | Measured Outcome | Evidence for Effectiveness | Risk of Bias |
|---|---|---|---|---|---|---|
| Specific Food | ||||||
| [42] | Trinidad and Tobago | Non-randomised controlled before/after study | Metabolic | Blood pressure | + | |
| [48] | Singapore | Individually-randomised parallel-group trial | Metabolic | Insulin sensitivity | − | |
| [65] | Seychelles | Individually-randomised cross over trial | Metabolic | TAGS | +/− | |
| [58] | Singapore | Individually-randomised cross over trial | Metabolic | Fasting blood glucose | + | |
| Supplements/Fortified Foods | ||||||
| [25] | Haiti | Cluster-randomised parallel group trial | Nutrition | Haemoglobin concentration | + | |
| [50] | Singapore | Individually-randomised parallel-group trial | Nutrition | Vitamin D deficiency | + | |
| [51] | Singapore | Individually-randomised parallel-group trial | Metabolic | Endothelial function | + | |
| [41] | Jamaica | Individually-randomised parallel-group trial | Nutrition | Weight for age/height z scores | − | |
| [27] | Haiti | Individually-randomised cross over trial | Nutrition | Iron absorption | +/− | |
| [28] | Haiti | Individually-randomised parallel-group trial | Nutrition | Stunting | + | |
| [29] | Haiti | Cluster-randomised parallel group trial | Nutrition | Weight for age/height z scores | +/− | |
| [30] | Haiti | Cluster-randomised parallel group trial | Nutrition | Anaemia | +/− | |
| [37] | Dominican Republic | Non-randomised uncontrolled before and after study (pre/post-test study) | Nutrition | Weight for age/height z scores | − | |
| [31] | Haiti | Cluster-randomised parallel group trial | Nutrition | Anaemia | + | |
| [64] | Mauritius | Individually-randomised parallel-group trial | Metabolic | HbA1C | +/− | |
| Nutrition Education | ||||||
| [49] | Singapore | Individually-randomised parallel-group trial | Metabolic | BMI | + | |
| [63] | Mauritius | Non-randomised uncontrolled before and after study (pre/post-test study) | Metabolic | BMI | − | |
| [70] | Singapore | Individually-randomised parallel-group trial | Metabolic | Weight change | − | |
| [54] | Singapore | Non-randomised uncontrolled before and after study (pre/post-test study) | Metabolic | Weight change | + | |
| [38] | Dominican Republic | Non-randomised controlled before/after study | Nutrition | Weight change for age/height z scores | + | |
| [72] | Kiribati | Cluster-randomised parallel group trial | Metabolic | Change in mean blood glucose | − | |
| Multifaceted Intervention | ||||||
| [68] | American Samoa | Cluster-randomised parallel group trial | Metabolic | HbA1C | + | |
| [69] | Tonga | Non-randomised controlled before/after study | Metabolic | Weight change | +/− | |
| [70] | French Polynesia | Non-randomised controlled before/after study | Metabolic | Weight change | +/− | |
| [45] | Puerto Rico | Individually-randomised parallel-group trial | Metabolic | BMI | + | |
| [26] | Haiti | Non-randomised controlled before/after study | Nutrition | Weight change for age/height z scores | +/− | |
| [52] | Singapore | Non-randomised uncontrolled before and after study (pre/post test study) | Metabolic | HbA1C | + | |
| [66] | Fiji | Non-randomised controlled before/after study | Metabolic | BMI for age | − | |
| [71] | Papua New Guinea | Non-randomised uncontrolled before and after study (pre/post test study) | Nutrition | Weight change for age/height z scores | + | |
| [55] | Singapore | Individually-randomised parallel-group trial | Metabolic | Weight Change | − | |
| [44] | Trinidad and Tobago | Individually-randomised parallel-group trial | Metabolic | BMI | − | |
| [67] | Fiji | Non-randomised uncontrolled before and after study (pre/post test study) | Metabolic | Blood pressure | + | |
| [33] | Haiti | Cluster-randomised parallel group trial | Nutrition | Weight change for age/height z scores | +/− | |
| [57] | Singapore | Non-randomised uncontrolled before and after study (pre/post test study) | Metabolic | Weight change | + | |
| [73] | Samoa | Non-randomised interrupted time series studies and repeated measures study | Metabolic | Weight change | − | |
| [59] | Singapore | Non-randomised uncontrolled before and after study (pre/post test study) | Metabolic | BMI | + | |
| [60] | Singapore | Non-randomised controlled before/after study | Metabolic | BMI | + | |
| [35] | Cuba | Individually-randomised parallel-group trial | Metabolic | BMI | +/− | |
| [36] | Cuba | Non-randomised uncontrolled before and after study (pre/post test study) | Metabolic | Weight change | +/− | |
| [39] | Dominican Republic | Non-randomised uncontrolled before and after study (pre/post test study) | Metabolic | HbA1C | + | |
| [61] | Singapore | Cluster-randomised parallel group trial | Metabolic | Blood pressure | + | |
| [62] | Singapore | Non-randomised uncontrolled before and after study (pre/post test study) | Metabolic | HbA1C | + | |
| Policy | ||||||
| [32] | Haiti | Non-randomised uncontrolled before and after study (pre/post test study) | Nutrition | Severe childhood malnutrition | − | |
| Dietary Change | ||||||
| [43] | Trinidad and Tobago | Individually-randomised parallel-group trial | Metabolic | BMI | + | |
| [40] | Jamaica | Individually-randomised parallel-group trial | Metabolic | HbA1C | + | |
| [47] | Barbados | Non-randomised uncontrolled before and after study (pre/post test study) | Metabolic | Fasting blood glucose | + | |
| [24] | Jamaica and Nigeria | Individually-randomised cross over trial | Metabolic | Blood pressure | + | |
| [46] | Puerto Rico | Individually-randomised parallel-group trial | Metabolic | BMI | − | |
| [34] | Cuba | Non-randomised uncontrolled before and after study (pre/post test study) | Metabolic | Fasting blood glucose | +/− | |
| [56] | Singapore | Non-randomised uncontrolled before and after study (pre/post test study) | Metabolic | HbA1C | + | |
3.1.3. Intervention Types
3.2. Intervention Effectiveness
3.2.1. Effectiveness by Intervention Type
3.2.2. Effectiveness by Intervention Level and Setting
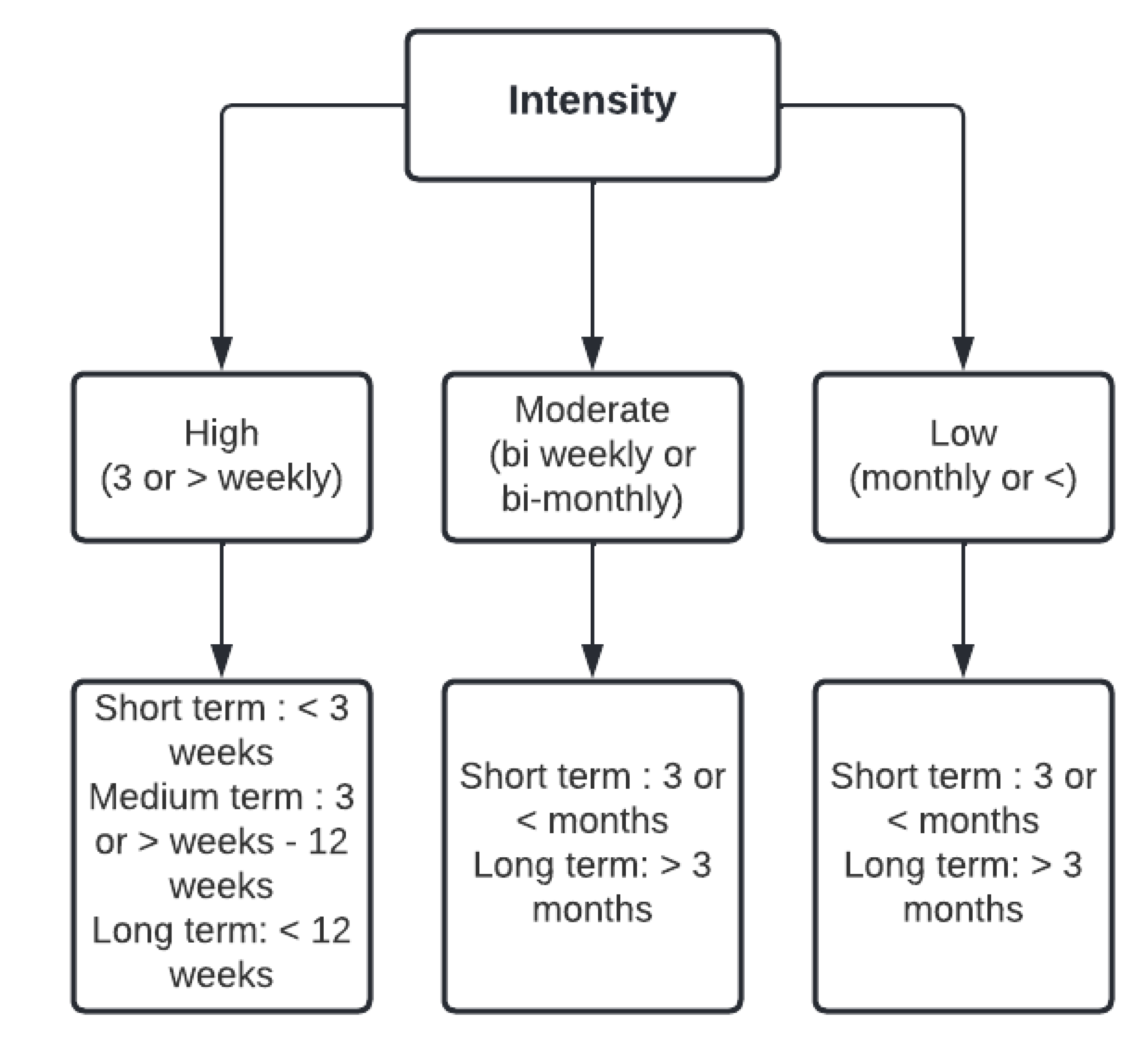
3.2.3. Effectiveness by Intensity
3.3. Risk of Bias
4. Discussion
4.1. Feasibility of Population-Level Interventions in SIDS
4.2. Interventions Addressing Both ‘under and over’ Nutrition
5. Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Haynes, E.; Bhagtani, D.; Iese, V.; Brown, C.R.; Fesaitu, J.; Hambleton, I.; Badrie, N.; Kroll, F.; Guell, C.; Brugulat-Panes, A.; et al. Food sources and dietary quality in small island developing states: Development of methods and policy relevant novel survey data from the Pacific and Caribbean. Nutrients 2020, 12, 3350. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.T.; Tseng, Y.C.; Iwaki, Y.; Huang, M.C. Sustainable food security in Small Island Developing States (SIDS): A case of Horticulture project in Marshall Islands. Mar. Policy 2021, 128, 104378. [Google Scholar] [CrossRef]
- Foley, A.M.; Moncada, S.; Mycoo, M.; Nunn, P.; Tandrayen-Ragoobur, V.; Evans, C. Small Island Developing States in a post-pandemic world: Challenges and opportunities for climate action. Wiley Interdiscip. Rev. Clim. Chang. 2022, 13, e769. [Google Scholar] [CrossRef]
- Von Tigerstrom, B. Small island developing states and international trade: Special challenges in the global partnership for development. Melb. J. Int. Law 2005, 6, 402–436. [Google Scholar]
- Food and Agriculture Organization. Global Action Programme on Food Security and Nutrition in Small Island Developing States: Supporting the Implementation of the SAMOA Pathway; FAO: Rome, Italy, 2017; Available online: http://www.fao.org/3/a-i7135e.pdf (accessed on 7 May 2022).
- Food and Agriculture Organization. The State of Food Security and Nutrition in Small Island Developing States; Contract No.: I5327E/1/01.16; FAO: Rome, Italy, 2016; Available online: https://www.fao.org/3/i5327e/i5327e.pdf (accessed on 7 May 2022).
- Hickey, G.M.; Unwin, N. Addressing the triple burden of malnutrition in the time of COVID-19 and climate change in Small Island Developing States: What role for improved local food production? Food Secur. 2020, 12, 831–835. [Google Scholar] [CrossRef]
- Popkin, B.M.; Adair, L.S.; Ng, S.W. Global nutrition transition and the pandemic of obesity in developing countries. Nutr. Rev. 2012, 70, 3–21. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Small island developing states: Health and WHO. In Country Presence Profile; World Health Organization: Geneva, Switzerland, 2017; Available online: https://apps.who.int/iris/bitstream/handle/10665/255804/WHO-CCU-17.08-eng.pdf?sequence=1&isAllowed=y (accessed on 7 May 2022).
- Pan American Health Organization. NCDs in Caribbean. Available online: www.paho.org/hq/index.phpoption=com_content&view=article&id=771&Itemid=40030&lang=en (accessed on 7 May 2022).
- Amoroso, L. The Second International Conference on Nutrition: Implications for Hidden Hunger. World Rev. Nutr. Diet. 2016, 115, 142–152. [Google Scholar]
- Join the E-Consultation on the Global Action Programme on Food Security and Nutrition in Pacific Small Island Developing States (PSIDS). Sustainable Development Knowledge Platform. 2017. Available online: https://sustainabledevelopment.un.org/?page=view&nr=1417&type=230&menu=2059 (accessed on 14 March 2022).
- Development Initiatives. Shining a Light to Spur Action on Nutrition; Global Nutrition Report; Development Initiatives: Bristol, UK, 2018. [Google Scholar]
- World Health Organization. Health in 2015: From MDGs, Millennium Development Goals to SDGs, Sustainable Development Goals; World Health Organization: Geneva, Switherland, 2015. [Google Scholar]
- Assembly, U.G. SIDS Accelerated Modalities of Action (SAMOA) Pathway; United Nations: New York, NY, USA, 2014; Volume Res. A/RES/69/15. [Google Scholar]
- Food and Agriculture Organization. Global Action Programme on Food Security and Nutrition in Small Island Developing States. 2017. Available online: https://www.fao.org/3/i7297e/i7297e.pdf (accessed on 14 March 2022).
- Haynes, E.; Brown, C.R.; Wou, C.; Vogliano, C.; Guell, C.; Unwin, N.; Community Food and Health Project. Health and other impacts of community food production in Small Island Developing States: A systematic scoping review. Revista Panamericana de Salud Pública 2018, 42, e176. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Syst. Rev. 2021, 10, 89. [Google Scholar] [CrossRef]
- United Nations, Small Island Developing States: List of SIDS. Available online: https://www.un.org/ohrlls/content/list-sids (accessed on 2 April 2022).
- Ouzzani, M.; Hammady, H.; Fedorowicz, Z.; Elmagarmid, A. Rayyan—A web and mobile app for systematic reviews. Syst. Rev. 2016, 5, 210. [Google Scholar] [CrossRef]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef]
- Sterne, J.A.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef]
- Sterne, J.A.; Hernán, M.A.; Reeves, B.C.; Savović, J.; Berkman, N.D.; Viswanathan, M.; Henry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I.; et al. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef] [PubMed]
- Forrester, T.; Adeyemo, A.; Soarres-Wynter, S.; Sargent, L.; Bennett, F.; Wilks, R.; Luke, A.; Prewitt, E.; Kramer, H.; Cooper, R.S. A randomized trial on sodium reduction in two developing countries. J. Hum. Hypertens 2005, 19, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Baum, A.; Elize, W.; Jean-Louis, F. Microfinance Institutions’ Successful Delivery Of Micronutrient Powders: A Randomized Trial In Rural Haiti. Health Aff. 2017, 36, 1938–1946. [Google Scholar] [CrossRef]
- Heidkamp, R.A.; Stoltzfus, R.J.; Fitzgerald, D.W.; Pape, J.W. Growth in late infancy among HIV-exposed children in urban Haiti is associated with participation in a clinic-based infant feeding support intervention. J. Nutr. 2012, 142, 774–780. [Google Scholar] [CrossRef]
- Herter-Aeberli, I.; Eliancy, K.; Rathon, Y.; Loechl, C.U.; Marhône Pierre, J.; Zimmermann, M.B. In Haitian women and preschool children, iron absorption from wheat flour-based meals fortified with sodium iron EDTA is higher than that from meals fortified with ferrous fumarate, and is not affected by Helicobacter pylori infection in children. Br. J. Nutr. 2017, 118, 273–279. [Google Scholar] [CrossRef]
- Iannotti, L.L.; Dulience, S.J.; Green, J.; Joseph, S.; François, J.; Anténor, M.L.; Lesorogol, C.; Mounce, J.; Nickerson, N.M. Linear growth increased in young children in an urban slum of Haiti: A randomized controlled trial of a lipid-based nutrient supplement. Am. J. Clin. Nutr. 2014, 99, 198–208. [Google Scholar] [CrossRef]
- Iannotti, L.L.; Henretty, N.M.; Delnatus, J.R.; Previl, W.; Stehl, T.; Vorkoper, S.; Bodden, J.; Maust, A.; Smidt, R.; Nash, M.L.; et al. Ready-to-use supplementary food increases fat mass and BMI in Haitian school-aged children. J. Nutr. 2015, 145, 813–822. [Google Scholar] [CrossRef] [PubMed]
- Iannotti, L.; Dulience, S.J.; Joseph, S.; Cooley, C.; Tufte, T.; Cox, K.; Eaton, J.; Delnatus, J.R.; Wolff, P.B. Fortified Snack Reduced Anemia in Rural School-Aged Children of Haiti: A Cluster-Randomized, Controlled Trial. PLoS ONE 2016, 11, e0168121. [Google Scholar] [CrossRef] [PubMed]
- Menon, P.; Ruel, M.T.; Loechl, C.U.; Arimond, M.; Habicht, J.P.; Pelto, G.; Michaud, L. Micronutrient Sprinkles reduce anemia among 9- to 24-mo-old children when delivered through an integrated health and nutrition program in rural Haiti. J. Nutr. 2007, 137, 1023–1030. [Google Scholar] [CrossRef]
- Reid, B.C.; Psoter, W.J.; Gebrian, B.; Wang, M.Q. The effect of an international embargo on malnutrition and childhood mortality in rural Haiti. Int. J. Health Serv. 2007, 37, 501–513. [Google Scholar] [CrossRef]
- Ruel, M.T.; Menon, P.; Habicht, J.P.; Loechl, C.; Bergeron, G.; Pelto, G.; Arimond, M.; Maluccio, J.; Michaud, L.; Hankebo, B. Age-based preventive targeting of food assistance and behaviour change and communication for reduction of childhood undernutrition in Haiti: A cluster randomised trial. Lancet 2008, 371, 588–595. [Google Scholar] [CrossRef]
- Porrata-Maury, C.; Hernández-Triana, M.; Rodríguez-Sotero, E.; Vilá-Dacosta-Calheiros, R.; Hernández-Hernández, H.; Mirabal-Sosa, M.; Campa-Huergo, C.; Pianesi, M. Medium- and short-term interventions with ma-pi 2 macrobiotic diet in type 2 diabetic adults of bauta, havana. J. Nutr. Metab. 2012, 2012, 856342. [Google Scholar] [CrossRef]
- Vilar Gomez, E.; Rodriguez De Miranda, A.; Gra Oramas, B.; Arus Soler, E.; Llanio Navarro, R.; Calzadilla Bertot, L.; Yasells Garcia, A.; Del Rosario Abreu Vazquez, M. Clinical trial: A nutritional supplement Viusid, in combination with diet and exercise, in patients with nonalcoholic fatty liver disease. Aliment. Pharmacol. Ther. 2009, 30, 999–1009. [Google Scholar] [CrossRef]
- Vilar-Gomez, E.; Martinez-Perez, Y.; Calzadilla-Bertot, L.; Torres-Gonzalez, A.; Gra-Oramas, B.; Gonzalez-Fabian, L.; Friedman, S.L.; Diago, M.; Romero-Gomez, M. Weight Loss Through Lifestyle Modification Significantly Reduces Features of Nonalcoholic Steatohepatitis. Gastroenterology 2015, 149, 367–378.e5. [Google Scholar] [CrossRef]
- McLennan, J.D.; Mills, R.M. Evaluation of a partial day treatment realimentation program for malnourished children in the Dominican Republic. J. Trop. Pediatr. 2006, 52, 394–398. [Google Scholar] [CrossRef]
- Navarro, J.I.; Sigulem, D.M.; Ferraro, A.A.; Polanco, J.J.; Barros, A.J. The double task of preventing malnutrition and overweight: A quasi-experimental community-based trial. BMC Public Health 2013, 13, 212. [Google Scholar] [CrossRef]
- West-Pollak, A.; Then, E.P.; Podesta, C.; Hedelt, A.; Perry, M.L.; Izarnotegui, W.V.; Perez, M.; Villegas, A.; Baez, N.I.; Bassa, R.; et al. Impact of a novel community-based lifestyle intervention program on type 2 diabetes and cardiovascular risk in a resource-poor setting in the Dominican Republic. Int. Health 2014, 6, 118–124. [Google Scholar] [CrossRef]
- Bahado-Singh, P.S.; Riley, C.K.; Wheatley, A.O.; Boyne, M.S.; Morrison, E.Y.; Asemota, H.N. High Fiber Caribbean Diets with Low-Intermediate GI Improve Glycemic Control, Cardiovascular and Inflammatory Indicators in Overweight Persons with Type 2 Diabetes: A Randomized Control Study. Curr. Res. Nutr. Food. Sci. 2015, 3, 36. [Google Scholar] [CrossRef]
- Gardner, J.M.; Powell, C.A.; Baker-Henningham, H.; Walker, S.P.; Cole, T.J.; Grantham-McGregor, S.M. Zinc supplementation and psychosocial stimulation: Effects on the development of undernourished Jamaican children. Am. J. Clin. Nutr. 2005, 82, 399–405. [Google Scholar] [CrossRef]
- Alleyne, T.; Alleyne, A.; Arrindell, D.; Balleram, N.; Cozier, D.; Haywood, R.; Humphrey, C.; Pran, L.; Rampersad, K.; Reyes, D.; et al. Short term effects of cocoa consumption on blood pressure. West Indian Med. J. 2014, 63, 312–317. [Google Scholar]
- Augustus, E.; Granderson, I.; Rocke, K.D. The Impact of a Ketogenic Dietary Intervention on the Quality of Life of Stage II and III Cancer Patients: A Randomized Controlled Trial in the Caribbean. Nutr. Cancer 2021, 73, 1590–1600. [Google Scholar] [CrossRef]
- Nichols, S.D.; Francis, M.P.; Dalrymple, N. Sustainability of a Curriculum-based Intervention on Dietary Behaviours and Physical Activity among Primary School Children in Trinidad and Tobago. West Indian Med. J. 2014, 63, 68–77. [Google Scholar] [CrossRef]
- Halperin, D.T.; Laux, J.; LeFranc-García, C.; Araujo, C.; Palacios, C. Findings From a Randomized Trial of Weight Gain Prevention Among Overweight Puerto Rican Young Adults. J. Nutr. Educ. Behav. 2019, 51, 205–216. [Google Scholar] [CrossRef]
- Palacios, C.; Bertrán, J.J.; Ríos, R.E.; Soltero, S. No effects of low and high consumption of dairy products and calcium supplements on body composition and serum lipids in Puerto Rican obese adults. Nutrition 2011, 27, 520–525. [Google Scholar] [CrossRef]
- Bynoe, K.; Unwin, N.; Taylor, C.; Murphy, M.M.; Bartholomew, L.; Greenidge, A.; Abed, M.; Jeyaseelan, S.; Cobelli, C.; Dalla Man, C.; et al. Inducing remission of Type 2 diabetes in the Caribbean: Findings from a mixed methods feasibility study of a low-calorie liquid diet-based intervention in Barbados. Diabet. Med. 2020, 37, 1816–1824. [Google Scholar] [CrossRef]
- Alperet, D.J.; Rebello, S.A.; Khoo, E.Y.; Tay, Z.; Seah, S.S.; Tai, B.C.; Tai, E.S.; Emady-Azar, S.; Chou, C.J.; Darimont, C.; et al. The effect of coffee consumption on insulin sensitivity and other biological risk factors for type 2 diabetes: A randomized placebo-controlled trial. Am. J. Clin. Nutr. 2020, 111, 448–458. [Google Scholar] [CrossRef] [PubMed]
- Ng, L.B.; Yang, Y.; Koh, G.C. The effect of a brief low-intensity therapeutic lifestyle counselling intervention on weight loss among overweight patients in primary care: A pilot randomised controlled trial. Proc. Singap. Healthc. 2014, 23, 118–125. [Google Scholar] [CrossRef]
- Chew, S.T.H.; Tan, N.C.; Cheong, M.; Oliver, J.; Baggs, G.; Choe, Y.; How, C.H.; Chow, W.L.; Tan, C.Y.L.; Kwan, S.C.; et al. Impact of specialized oral nutritional supplement on clinical, nutritional, and functional outcomes: A randomized, placebo-controlled trial in community-dwelling older adults at risk of malnutrition. Clin. Nutr. 2021, 40, 1879–1892. [Google Scholar] [CrossRef] [PubMed]
- Dalan, R.; Liew, H.; Assam, P.N.; Chan, E.S.; Siddiqui, F.J.; Tan, A.W.; Chew, D.E.; Boehm, B.O.; Leow, M.K. A randomised controlled trial evaluating the impact of targeted vitamin D supplementation on endothelial function in type 2 diabetes mellitus: The DIMENSION trial. Diab. Vasc. Dis. Res. 2016, 13, 192–200. [Google Scholar] [CrossRef]
- Koot, D.; Goh, P.S.C.; Lim, R.S.M.; Tian, Y.; Yau, T.Y.; Tan, N.C.; Finkelstein, E.A. A Mobile Lifestyle Management Program (GlycoLeap) for People With Type 2 Diabetes: Single-Arm Feasibility Study. JMIR Mhealth Uhealth 2019, 7, e12965. [Google Scholar]
- Li, L.J.; Aris, I.M.; Han, W.M.; Tan, K.H. A Promising Food-Coaching Intervention Program to Achieve Optimal Gestational Weight Gain in Overweight and Obese Pregnant Women: Pilot Randomized Controlled Trial of a Smartphone App. JMIR Form. Res. 2019, 3, e13013. [Google Scholar] [CrossRef]
- Lim, S.L.; Lin, X.; Chan, Y.H.; Ferguson, M.; Daniels, L. A Pre-post Evaluation of an Ambulatory Nutrition Support Service for Malnourished Patients Post Hospital Discharge: A Pilot Study. Ann. Acad. Med. Singap. 2013, 42, 507–513. [Google Scholar]
- Lim, K.; Chan, S.Y.; Lim, S.L.; Tai, B.C.; Tsai, C.; Wong, S.R.; Ang, S.M.; Yew, T.W.; Tai, E.S.; Yong, E.L. A Smartphone App to Restore Optimal Weight (SPAROW) in Women With Recent Gestational Diabetes Mellitus: Randomized Controlled Trial. JMIR Mhealth Uhealth 2021, 9, e22147. [Google Scholar] [CrossRef]
- Siaw, M.Y.; Chew, D.E.; Toh, M.P.; Seah, D.E.; Chua, R.; Tan, J.; Lee, E.Y.; Chan, S.Y.; Lee, J.Y. Metabolic parameters in type 2 diabetic patients with varying degrees of glycemic control during Ramadan: An observational study. J. Diabetes Investig. 2016, 7, 70–75. [Google Scholar] [CrossRef]
- Tan, W.J.; Wong, T.K. Demographic profile, clinical characteristics, motivations and weight loss outcomes of patients in a nonsurgical weight management programme. Singap. Med. J. 2014, 55, 150–154. [Google Scholar]
- Tey, S.L.; Lee, D.E.M.; Henry, C.J. Fruit form Influences Postprandial Glycemic Response in Elderly and Young Adults. J. Nutr. Health Aging 2017, 21, 887–891. [Google Scholar]
- Vasquez, K.; Malhotra, R.; Østbye, T.; Chan, M.F.; Amin, H.; Khoo, G.; Choo, L.; Chew, L.; Thilagaratnam, S. Extent and correlates of change in anthropometric and fitness outcomes among participants in a corporate team-based weight loss challenge in Singapore: Lose to win 2009. Asia Pac. J. Public Health 2015, 27, NP425–NP436. [Google Scholar] [CrossRef]
- Vasquez, K.; Malhotra, R.; Østbye, T.; Low, W.; Chan, M.F.; Chew, L.; Ling, A. Lessons from Singapore’s national weight management program, Lose To Win. Health Promot. Int. 2018, 33, 834–845. [Google Scholar] [CrossRef]
- Wong, E.Y.; James, A.P.; Lee, A.H.; Jancey, J. Effectiveness of a Singaporean Community-Based Physical Activity and Nutrition Intervention: A Cluster Randomized Controlled Trial. Asia Pac. J. Public Health 2021, 33, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Yeoh, E.C.; Zainudin, S.B.; Loh, W.N.; Chua, C.L.; Fun, S.; Subramaniam, T.; Sum, C.F.; Lim, S.C. Fasting during Ramadan and Associated Changes in Glycaemia, Caloric Intake and Body Composition with Gender Differences in Singapore. Ann. Acad. Med. Singap. 2015, 44, 202–206. [Google Scholar] [CrossRef] [PubMed]
- Cannoosamy, K.; Pem, D.; Bhagwant, S.; Jeewon, R. Is a Nutrition Education Intervention Associated with a Higher Intake of Fruit and Vegetables and Improved Nutritional Knowledge among Housewives in Mauritius? Nutrients 2016, 8, 723. [Google Scholar] [CrossRef] [PubMed]
- Somanah, J.; Aruoma, O.I.; Gunness, T.K.; Kowelssur, S.; Dambala, V.; Murad, F.; Googoolye, K.; Daus, D.; Indelicato, J.; Bourdon, E.; et al. Effects of a short term supplementation of a fermented papaya preparation on biomarkers of diabetes mellitus in a randomized Mauritian population. Prev. Med. 2012, 54, S90–S97. [Google Scholar] [CrossRef]
- Bovet, P.; Faeh, D.; Madeleine, G.; Viswanathan, B.; Paccaud, F. Decrease in blood triglycerides associated with the consumption of eggs of hens fed with food supplemented with fish oil. Nutr. Metab. Cardiovasc. Dis. 2007, 17, 280–287. [Google Scholar] [CrossRef]
- Kremer, P.; Waqa, G.; Vanualailai, N.; Schultz, J.T.; Roberts, G.; Moodie, M.; Mavoa, H.; Malakellis, M.; McCabe, M.P.; Swinburn, B.A. Reducing unhealthy weight gain in Fijian adolescents: Results of the Healthy Youth Healthy Communities study. Obes. Rev. 2011, 12 (Suppl. S2), 29–40. [Google Scholar] [CrossRef]
- Pillay, A.; Trieu, K.; Santos, J.A.; Sukhu, A.; Schultz, J.; Wate, J.; Bell, C.; Moodie, M.; Snowdon, W.; Ma, G.; et al. Assessment of a Salt Reduction Intervention on Adult Population Salt Intake in Fiji. Nutrients 2017, 9, 1350. [Google Scholar] [CrossRef]
- DePue, J.D.; Dunsiger, S.; Seiden, A.D.; Blume, J.; Rosen, R.K.; Goldstein, M.G.; Nu’usolia, O.; Tuitele, J.; McGarvey, S.T. Nurse-community health worker team improves diabetes care in American Samoa: Results of a randomized controlled trial. Diabetes Care 2013, 36, 1947–1953. [Google Scholar] [CrossRef]
- Fotu, K.F.; Millar, L.; Mavoa, H.; Kremer, P.; Moodie, M.; Snowdon, W.; Utter, J.; Vivili, P.; Schultz, J.T.; Malakellis, M.; et al. Outcome results for the Ma’alahi Youth Project, a Tongan community-based obesity prevention programme for adolescents. Obes. Rev. 2011, 12 (Suppl. S2), 41–50. [Google Scholar] [CrossRef]
- Gatti, C.; Suhas, E.; Côté, S.; Anassour Laouan-Sidi, E.; Dewailly, É.; Lucas, M. Obesity and metabolic parameters in adolescents: A school-based intervention program in French Polynesia. J. Adolesc. Health 2015, 56, 174–180. [Google Scholar] [CrossRef]
- Landi, M.; Swakin, E.; Minijihau, C.; Welch, H.; Tefuarani, N.; Duke, T. Severe malnutrition in children in Papua New Guinea: Effect of a multi-faceted intervention to improve quality of care and nutritional outcomes. Paediatr. Int. Child Health 2017, 37, 21–28. [Google Scholar]
- Plows, J.F.; Rios, C.; Berger, P.K.; Jones, R.B.; Alderete, T.L.; Steinberg, S.; Bangao, T.; Beale, E.; Goran, M.I. Effects of a sugar-reduction intervention on diet and continuous blood glucose in adolescents on the remote Pacific island of Kiritimati: A pilot randomized controlled trial. Res. Square 2020, 1–19. [Google Scholar] [CrossRef]
- Trieu, K.; Ieremia, M.; Santos, J.; Neal, B.; Woodward, M.; Moodie, M.; Bell, C.; Snowdon, W.; Faumuina, T.; Webster, J. Effects of a nationwide strategy to reduce salt intake in Samoa. J. Hypertens 2018, 36, 188–198. [Google Scholar]
- Nava, L.T.; Zambrano, J.M.; Arviso, K.P.; Brochetti, D.; Becker, K.L. Nutrition-based interventions to address metabolic syndrome in the N avajo: A systematic review. J. Clin. Nurs. 2015, 24, 3024–3045. [Google Scholar]
- Liu, Z.; Gao, P.; Gao, A.Y.; Lin, Y.; Feng, X.X.; Zhang, F.; Xu, L.; Niu, W.; Fang, H.; Zhou, S.; et al. Effectiveness of a multifaceted intervention for prevention of obesity in primary school children in China: A cluster randomized clinical trial. JAMA Pediatrics 2022, 176, e214375. [Google Scholar]
- Chuang, H.H.; Lin, R.H.; Chen, J.Y.; Yeh, W.C.; Lin, H.F.; Ueng SW, N.; Hsu, K.H. Effectiveness of a multi-faceted intervention among elementary school children. Medicine 2019, 98, e15079. [Google Scholar]
- Straznicky, N.E.; Grima, M.T.; Lambert, E.A.; Eikelis, N.; Dawood, T.; Lambert, G.W.; Nestel, P.J.; Masuo, K.; Sari, C.I.; Chopra, R.; et al. Exercise augments weight loss induced improvement in renal function in obese metabolic syndrome individuals. J. Hypertens. 2011, 29, 553–564. [Google Scholar]
- Tran, V.D.; James, A.P.; Lee, A.H.; Jancey, J.; Howat, P.A.; Thi Phuong Mai, L. Effectiveness of a community-based physical activity and nutrition behavior intervention on features of the metabolic syndrome: A cluster-randomized controlled trial. Metab. Syndr. Relat. Disord. 2017, 15, 63–71. [Google Scholar]
- Pettman, T.L.; Buckley, J.D.; Misan, G.M.; Coates, A.M.; Howe, P.R. Health benefits of a 4-month group-based diet and lifestyle modification program for individuals with metabolic syndrome. Obes. Res. Clin. Pract. 2009, 3, 221–235. [Google Scholar]
- Diabetes Prevention Program Research Group. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N. Engl. J. Med. 2002, 346, 393–403. [Google Scholar]
- van Namen, M.; Prendergast, L.; Peiris, C. Supervised lifestyle intervention for people with metabolic syndrome improves outcomes and reduces individual risk factors of metabolic syndrome: A systematic review and meta-analysis. Metabolism 2019, 101, 153988. [Google Scholar]
- Huybregts, L.; Houngbé, F.; Salpéteur, C.; Brown, R.; Roberfroid, D.; Ait-Aissa, M.; Kolsteren, P. The effect of adding ready-to-use supplementary food to a general food distribution on child nutritional status and morbidity: A cluster-randomized controlled trial. PloS Med. 2012, 9, e1001313. [Google Scholar]
- Olsen, M.F.; Iuel-Brockdorff, A.-S.; Yaméogo, C.W.; Cichon, B.; Fabiansen, C.; Filteau, S.; Phelan, K.; Ouédraogo, A.; Michaelsen, K.F.; Gladstone, M.; et al. Impact of food supplements on early child development in children with moderate acute malnutrition: A randomised 2 × 2 × 3 factorial trial in Burkina Faso. PLoS Med. 2020, 17, e1003442. [Google Scholar]
- Larsson, J.; Unosson, M.; Ek, A.C.; Nilsson, L.; Thorslund, S.; Bjurulf, P. Effect of dietary supplement on nutritional status and clinical outcome in 501 geriatric patients—A randomised study. Clin. Nutr. 1990, 9, 179–184. [Google Scholar] [PubMed]
- Sauer, A.C.; Li, J.; Partridge, J.; Sulo, S. Assessing the impact of nutrition interventions on health and nutrition outcomes of community-dwelling adults: A systematic review. Nutr. Diet. Suppl. 2018, 10, 45–57. [Google Scholar]
- Zhang, H.; Qiu, Y.; Zhang, J.; Ma, Z.; Amoah, A.N.; Cao, Y.; Wang, X.; Fu, P.; Lyu, Q. The effect of oral nutritional supplements on the nutritional status of community elderly people with malnutrition or risk of malnutrition. Asia Pac. J. Clin. Nutr. 2021, 30, 415–423. [Google Scholar] [PubMed]
- Shintani, T.T.; Hughes, C.K.; Beckham, S.; O’Connor, H.K. Obesity and cardiovascular risk intervention through the ad libitum feeding of traditional Hawaiian diet. Am. J. Clin. Nutr. 1991, 53, 1647s–1651s. [Google Scholar] [PubMed]
- Horta, P.M.; Dos Santos, L.C. Effectiveness of high-intensity nutritional interventions for overweight Brazilian women. Public Health 2016, 139, 231–233. [Google Scholar]
- Verheijden, M.W.; Kok, F.J. Public health impact of community-based nutrition and lifestyle interventions. Eur. J. Clin. Nutr. 2005, 59, S66–S76. [Google Scholar]
- Mahadzir MD, A.; Quek, K.F.; Ramadas, A. Group-Based Lifestyle Intervention Strategies for Metabolic Syndrome: A Scoping Review and Strategic Framework for Future Research. Medicina 2021, 57, 1169. [Google Scholar]
- Odgers-Jewell, K.; Ball, L.E.; Kelly, J.T.; Isenring, E.A.; Reidlinger, D.P.; Thomas, R. Effectiveness of group-based self-management education for individuals with Type 2 diabetes: A systematic review with meta-analyses and meta-regression. Diabet. Med. 2017, 34, 1027–1039. [Google Scholar] [CrossRef]
- Borek, A.J.; Abraham, C.; Greaves, C.J.; Tarrant, M. Group-based diet and physical activity weight-loss interventions: A systematic review and meta-analysis of randomised controlled trials. Appl. Psychol. Health Well-Being 2018, 10, 62–86. [Google Scholar] [CrossRef]
- Webel, A.R.; Okonsky, J.; Trompeta, J.; Holzemer, W.L. A systematic review of the effectiveness of peer-based interventions on health-related behaviors in adults. Am. J. Public Health 2010, 100, 247–253. [Google Scholar] [CrossRef]
- O’Brien, K.M.; Barnes, C.; Yoong, S.; Campbell, E.; Wyse, R.; Delaney, T.; Brown, A.; Stacey, F.; Davies, L.; Lorien, S.; et al. School-based nutrition interventions in children aged 6 to 18 Years: An umbrella review of systematic reviews. Nutrients 2021, 13, 4113. [Google Scholar] [CrossRef]
- Perdew, M.; Liu, S.; Naylor, P.J. Family-based nutrition interventions for obesity prevention among school-aged children: A systematic review. Transl. Behav. Med. 2021, 11, 709–723. [Google Scholar] [CrossRef]


| Criteria | Include | Exclude |
|---|---|---|
| Study design | Any experimental, quasi, or natural experimental designs (i.e., randomised controlled trials, non-randomised trials) | Non-intervention studies (i.e., cross-sectional studies, reviews, case reports, editorials) |
| Outcome | Quantitative outcomes—impact on any aspect of nutrition status or metabolic health | Dietary intake or nutrition knowledge as only outcome |
| Study setting | 58 UN SIDS [19] | Any country that is not a SIDS |
| Publication status | Published and unpublished literature | Not applicable |
| Language | No language barriers: search terms were in English | Not applicable |
| Time | Articles from 1 January 2000 to 1 August 2020 | All articles before and after the dates specified |
| Participant characteristics | Any ethnicity, age, gender, SIDS location, and socio-economic status. | Not applicable |
| Ref. | Intervention Description | Setting | Intervention Level | Participant Intensity | Main Outcome Assessed |
|---|---|---|---|---|---|
| Specific food | |||||
| [42] | Impact of cocoa on blood pressure | No info | Individual | Low | Metabolic—blood pressure |
| [58] | Effect of consuming different forms (bite size, puree) and two fruit types (guava, papaya) on glycaemic response | General population | Individual | High | Metabolic—fasting blood glucose |
| Supplements/Fortified Foods | |||||
| [25] | Impact of micronutrient powders on haemoglobin concentration | Community | Community | High | Nutrition—haemoglobin concentration |
| [50] | Impact of nutritional supplement on vitamin D | Combination (general public, community centers, senior activity centers, polyclinics, hospitals, and by referrals from healthcare professionals in Singapore) | Individual | High | Nutrition—vitamin D deficiency |
| [51] | The effect of cholecalciferol supplementation on endothelial function | Clinic | Individual | High | Metabolic—endothelial function |
| [28] | Lipid supplement impact on childhood stunting | Community | Individual | High | Nutrition—stunting |
| [31] | Effectiveness of micronutrient sprinkles on anaemia | Community | Individual | High | Nutrition—anaemia |
| Nutrition Education | |||||
| [49] | To pilot test therapeutic lifestyle counselling on weight reduction | Clinic | Individual | Low | Metabolic—BMI |
| [54] | To evaluate nutritional outcomes after using an ANS service | Clinic | Individual | Low | Metabolic—weight Change |
| [38] | Lifestyle counselling to improve childhood malnutrition and overweight | Community | Individual | Moderate | Nutrition—weight change for age/height z scores |
| Multifaceted Intervention | |||||
| [68] | Impact of multifaceted intervention to support diabetic self-care | Clinic | Household | Low | Metabolic—HbA1C |
| [45] | To assess impact of multimodality weight gain prevention intervention | School | Institutional | Moderate | Metabolic—BMI |
| [52] | To assess the potential effectiveness and feasibility of Glycoleap (food coaching app) | Clinic | Individual | High | Metabolic—HbA1C |
| [67] | Intervention aimed at reducing population level salt intake. | General population | National/policy | High | Metabolic—blood pressure |
| [57] | Impact of non-surgical weight loss program | Clinic | Individual | Low | Metabolic—weight change |
| [59] | To assess the effectiveness of a national corporate team-based weight loss intervention | Workplace | Institutional | Moderate | Metabolic—BMI |
| [60] | To promote individuals to lose weight in a healthy way through a multi-component intervention. | Workplace | Institutional | Moderate | Metabolic—BMI |
| [39] | Lifestyle management of prediabetes and diabetes | General population | Community | Low | Metabolic—HbA1C |
| [61] | To improve the health behaviours and outcomes among women aged 50 years and older | Community | Community | High | Metabolic—blood pressure |
| [62] | To study the changes in body composition and metabolic profile in Muslim patients | Clinic | Individual | Low | Metabolic—HbA1C |
| Dietary Change | |||||
| [43] | Impact of ketogenic diet on nutritional status | Clinic | Individual | High | Metabolic—BMI |
| [40] | Impact of traditional Caribbean foods, with pronounced differences in GI on metabolic parameters | Clinic | Individual | High | Metabolic—HbA1C |
| [47] | Eight-week liquid (760 calorie) diet. | Combination (persons with type 2 diabetes recruited through a combination of publicity and contacts with local government and private healthcare providers) | Individual | High | Metabolic—fasting blood glucose |
| [24] | Impact of low-salt or high-salt diet on BP | General population | Individual | High | Metabolic—blood pressure |
| [56] | Impact of fasting and other nutrition related intervention on blood glucose | Clinic | Individual | NA | Metabolic—HbA1C |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Augustus, E.; Haynes, E.; Guell, C.; Morrissey, K.; Murphy, M.M.; Halliday, C.; Jia, L.; Iese, V.; Anderson, S.G.; Unwin, N. The Impact of Nutrition-Based Interventions on Nutritional Status and Metabolic Health in Small Island Developing States: A Systematic Review and Narrative Synthesis. Nutrients 2022, 14, 3529. https://doi.org/10.3390/nu14173529
Augustus E, Haynes E, Guell C, Morrissey K, Murphy MM, Halliday C, Jia L, Iese V, Anderson SG, Unwin N. The Impact of Nutrition-Based Interventions on Nutritional Status and Metabolic Health in Small Island Developing States: A Systematic Review and Narrative Synthesis. Nutrients. 2022; 14(17):3529. https://doi.org/10.3390/nu14173529
Chicago/Turabian StyleAugustus, Eden, Emily Haynes, Cornelia Guell, Karyn Morrissey, Madhuvanti M. Murphy, Cassandra Halliday, Lili Jia, Viliamu Iese, Simon G. Anderson, and Nigel Unwin. 2022. "The Impact of Nutrition-Based Interventions on Nutritional Status and Metabolic Health in Small Island Developing States: A Systematic Review and Narrative Synthesis" Nutrients 14, no. 17: 3529. https://doi.org/10.3390/nu14173529
APA StyleAugustus, E., Haynes, E., Guell, C., Morrissey, K., Murphy, M. M., Halliday, C., Jia, L., Iese, V., Anderson, S. G., & Unwin, N. (2022). The Impact of Nutrition-Based Interventions on Nutritional Status and Metabolic Health in Small Island Developing States: A Systematic Review and Narrative Synthesis. Nutrients, 14(17), 3529. https://doi.org/10.3390/nu14173529








