Interplay between Intestinal Bacterial Communities and Unicellular Parasites in a Morbidly Obese Population: A Neglected Trinomial
Abstract
1. Introduction
2. Materials and Methods
2.1. Design of the Study
2.2. Study Population
2.3. Clinical Interview
2.4. Stool Sampling, DNA Extraction and Parasitological Analysis
2.5. Microbiota Analysis by Next Generation Sequencing and Interpretation
2.6. Statistical Analysis
2.7. Ethical Statement
3. Results
3.1. Studied Population Description
3.2. Metagenomics Analysis of the Intestinal Bacterial Community
3.2.1. Reads by Sample
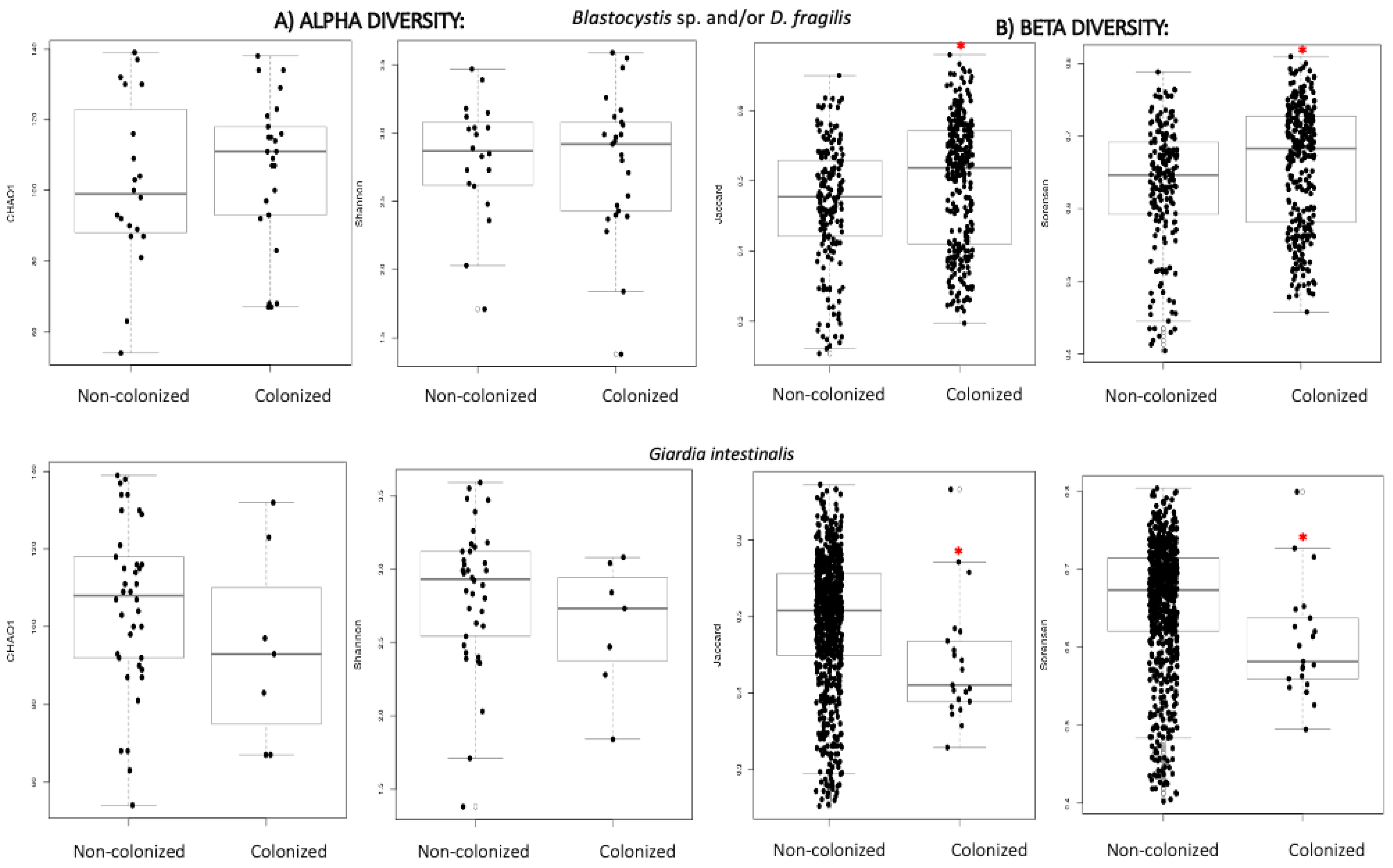
3.2.2. Alpha and Beta Diversity
3.2.3. Relative Abundance of Taxa
3.3. Dietary Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yatsunenko, T.; Rey, F.E.; Manary, M.J.; Trehan, I.; Dominguez-Bello, M.G.; Contreras, M.; Magris, M.; Hidalgo, G.; Baldassano, R.N.; Anokhin, A.P.; et al. Human gut microbiome viewed across age and geography. Nature 2012, 486, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Delzenne, N.M. The role of the gut microbiota in energy metabolism and metabolic disease. Curr. Pharm. Des. 2009, 15, 1546–1558. [Google Scholar] [CrossRef] [PubMed]
- Heiss, C.N.; Olofsson, L.E. Gut microbiota-dependent modulation of energy metabolism. J. Innate Immun. 2018, 10, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Zhang, J. Role of intestinal microbiota and metabolites on gut homeostasis and human diseases. BMC Immunol. 2017, 18, 2. [Google Scholar] [CrossRef]
- Ratajczak, W.; Rył, A.; Mizerski, A.; Walczakiewicz, K.; Sipak, O.; Laszczyńska, M. Immunomodulatory potential of gut microbiome-derived short-chain fatty acids (SCFAs). Acta Biochim. Pol. 2019, 66, 1–12. [Google Scholar] [CrossRef]
- Burgess, S.L.; Gilchrist, C.A.; Lynn, T.C.; Petri, W.A. Parasitic protozoa and interactions with the host intestinal microbiota. Infect. Immun. 2017, 85, e00101–e00117. [Google Scholar] [CrossRef]
- Bär, A.; Phukan, N.; Pinheiro, J.; Simoes-Barbosa, A. The interplay of host microbiota and parasitic protozoans at mucosal interfaces: Implications for the outcomes of infections and diseases. PLoS Negl. Trop. Dis. 2015, 9, e0004176. [Google Scholar] [CrossRef]
- Stensvold, C.R.; van der Giezen, M. Associations between gut microbiota and common luminal intestinal parasites. Trends Parasitol. 2018, 34, 369–377. [Google Scholar] [CrossRef]
- Ley, R.E.; Bäckhed, F.; Turnbaugh, P.; Lozupone, C.A.; Knight, R.D.; Gordon, J.I. Obesity alters gut microbial ecology. Proc. Natl. Acad. Sci. USA 2005, 102, 11070–11075. [Google Scholar] [CrossRef]
- Turnbaugh, P.J.; Hamady, M.; Yatsunenko, T.; Cantarel, B.L.; Duncan, A.; Ley, R.E.; Sogin, M.L.; Jones, W.J.; Roe, B.A.; Affourtit, J.P.; et al. A core gut microbiome in obese and lean twins. Nature 2009, 457, 480–484. [Google Scholar] [CrossRef]
- Clemente, J.C.; Ursell, L.K.; Parfrey, L.W.; Knight, R. The impact of the gut microbiota on human health: An integrative view. Cell 2012, 148, 1258–1270. [Google Scholar] [CrossRef]
- Joyce, S.A.; Gahan, C.G.M. The gut microbiota and the metabolic health of the host. Curr. Opin. Gastroenterol. 2014, 30, 120–127. [Google Scholar] [CrossRef]
- Allain, T.; Amat, C.B.; Motta, J.-P.; Motta, J.-P.; Manko, A.; Buret, A.G. Interactions of Giardia sp. with the intestinal barrier: Epithelium, mucus, and microbiota. Tissue Barriers 2017, 5, e1274354. [Google Scholar] [CrossRef]
- Iebba, V.; Santangelo, F.; Totino, V.; Pantanella, F.; Monsia, A.; Di Cristanziano, V.; Di Cave, D.; Schippa, S.; Berrilli, F.; D’Alfonso, R. Gut microbiota related to Giardia duodenalis, Entamoeba spp. and Blastocystis hominis infections in humans from Côte d’Ivoire. J. Infect. Dev. Ctries. 2016, 10, 1035–1041. [Google Scholar] [CrossRef]
- Beatty, J.K.; Akierman, S.V.; Motta, J.-P.; Muise, S.; Workentine, M.L.; Harrison, J.J.; Bhargava, A.; Beck, P.L.; Rioux, K.P.; McKnight, G.W.; et al. Giardia duodenalis induces pathogenic dysbiosis of human intestinal microbiota biofilms. Int. J. Parasitol. 2017, 47, 311–326. [Google Scholar] [CrossRef]
- Fekete, E.; Allain, T.; Siddiq, A.; Sosnowski, O.; Buret, A.G. Giardia spp. and the gut microbiota: Dangerous liaisons. Front. Microbiol. 2021, 11, 618106. [Google Scholar] [CrossRef]
- Scanlan, P.D.; Stensvold, C.R.; Rajilić-Stojanović, M.; Heilig, H.G.; De Vos, W.M.; O’Toole, P.W.; Cotter, P.D. The microbial eukaryote Blastocystis is a prevalent and diverse member of the healthy human gut microbiota. FEMS Microbiol. Ecol. 2014, 90, 326–330. [Google Scholar] [CrossRef]
- Beghini, F.; Pasolli, E.; Truong, T.D.; Putignani, L.; Cacciò, S.M.; Segata, N. Large-scale comparative metagenomics of Blastocystis, a common member of the human gut microbiome. ISME J. 2017, 11, 2848–2863. [Google Scholar] [CrossRef]
- Tan, K.S.; Mirza, H.; Teo, J.D.; Wu, B.; Macary, P.A. Current views on the clinical relevance of Blastocystis spp. Curr. Infect. Dis. Rep. 2010, 12, 28–35. [Google Scholar] [CrossRef]
- Bäckhed, F.; Fraser, C.M.; Ringel, Y.; Sanders, M.E.; Sartor, R.B.; Sherman, P.M.; Versalovic, J.; Young, V.; Finlay, B.B. Defining a healthy human gut microbiome: Current concepts, future directions, and clinical applications. Cell Host Microbe 2012, 12, 611–622. [Google Scholar] [CrossRef]
- Le Chatelier, E.; Nielsen, T.; Qin, J.; Prifti, E.; Hildebrand, F.; Falony, G.; Almeida, M.; Arumugam, M.; Batto, J.M.; Kennedy, S.; et al. Richness of human gut microbiome correlates with metabolic markers. Nature 2013, 500, 541–546. [Google Scholar] [CrossRef]
- Audebert, C.; Even, G.; Cian, A.; Blastocystis Investigation Group; Loywick, A.; Merlin, S.; Viscogliosi, E.; Chabe, M. Colonization with the enteric protozoa Blastocystis is associated with increased diversity of human gut bacterial microbiota. Sci. Rep. 2016, 6, 25255. [Google Scholar] [CrossRef]
- Andersen, L.O.; Bonde, I.; Nielsen, H.B.; Stensvold, C.R. A retrospective metagenomics approach to studying Blastocystis. FEMS Microbiol. Ecol. 2015, 91, fiv072. [Google Scholar] [CrossRef]
- Garcia, L.S. Dientamoeba fragilis, one of the neglected intestinal protozoa. J. Clin. Microbiol. 2016, 54, 2243–2250. [Google Scholar] [CrossRef]
- Knoll, E.W.; Howell, K.M. Studies on Dientamoeba fragilis: Its incidence and possible pathogenicity. Am. J. Clin. Pathol. 1945, 15, 178–183. [Google Scholar] [CrossRef][Green Version]
- Wong, Z.; Faulder, K.; Robinson, J.L. Does Dientamoeba fragilis cause diarrhea? A systematic review. Parasitol. Res. 2018, 117, 971–980. [Google Scholar] [CrossRef]
- van Kalleveen, M.W.; Budding, A.E.; Benninga, M.A.; Savelkoul, P.H.M.; van Gool, T.; van Maldeghem, I.; Dorigo-Zetsma, J.W.; Bart, A.; Plötz, F.B.; de Meij, T.G. Intestinal microbiota in children with symptomatic Dientamoeba fragilis infection: A case-control study. Pediatr. Infect. Dis. J. 2020, 40, 279–283. [Google Scholar] [CrossRef]
- Garvey, W.T.; Mechanick, J.I.; Brett, E.M.; Garber, A.J.; Hurley, D.L.; Jastreboff, A.M.; Nadolsky, K.; Pessah-Pollack, R.; Plodkowski, R. American association of clinical endocrinologists and American college of endocrinology comprehensive clinical practice guidelines for medical care of patients with obesity. Endocr. Pract. 2016, 22 (Suppl. S3), 1–203. [Google Scholar] [CrossRef]
- Martínez-González, M.A.; García-Arellano, A.; Toledo, E.; Salas-Salvadó, J.; Buil-Cosiales, P.; Corella, D.; Covas, M.I.; Schröder, H.; Arós, F.; Gómez-Gracia, E.; et al. A 14-Item Mediterranean diet assessment tool and obesity indexes among high-risk subjects: The PREDIMED trial. PLoS ONE 2012, 7, e43134. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean diet and survival in a Greek population. N. Engl. J. Med. 2003, 348, 2599–2608. [Google Scholar] [CrossRef] [PubMed]
- Martin-Moreno, J.M.; Boyle, P.; Gorgojo, L.; Maisonneuve, P.; Fernandez-Rodriguez, J.C.; Salvini, S.; Willett, W.C. Development and validation of a food frequency questionnaire in Spain. Int. J. Epidemiol. 1993, 22, 512–519. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority. General principles for the collection of national food consumption data in the view of a Pan-European dietary survey. EFSA J. 2009, 7, 1435. [Google Scholar] [CrossRef]
- Brussaard, J.H.; Löwik, M.R.; Steingrímsdóttir, L.; Møller, A.; Kearney, J.; De Henauw, S. A European food consumption survey method—Conclusions and recommendations. Eur. J. Clin. Nutr. 2002, 56, S89–S94. [Google Scholar] [CrossRef]
- Ortega, R.M.; Pérez-Rodrigo, C.; López-Sobaler, A.M. Dietary assessment methods: Dietary records. Nutr. Hosp. 2015, 31 (Suppl. S3), 38–45. [Google Scholar] [CrossRef]
- Trumbo, P.; Schlicker, S.; Yates, A.A.; Poos, M. Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein, and amino acids. J. Am. Diet Assoc. 2002, 102, 1621–1630. [Google Scholar] [CrossRef]
- Huang, P.L. A comprehensive definition for metabolic syndrome. Dis. Model. Mech. 2009, 2, 231–237. [Google Scholar] [CrossRef]
- Ascaso, J.F.; Romero, P.; Real, J.T.; Priego, A.; Valdecabres, C.; Carmena, R. Cuantificación de insulinorresistencia con los valores de insulina basal e índice HOMA en una población no diabética [Insulin resistance quantification by fasting insulin plasma values and HOMA index in a non-diabetic population]. Med. Clin. 2001, 117, 530–533. [Google Scholar] [CrossRef]
- Brewer, G.J.; Blue, M.N.M.; Hirsch, K.R.; Saylor, H.E.; Gould, L.M.; Nelson, A.G.; Smith-Ryan, A.E. Validation of InBody 770 bioelectrical impedance analysis compared to a four-compartment model criterion in young adults. Clin. Physiol. Funct. Imaging 2021, 41, 317–325. [Google Scholar] [CrossRef]
- Autier, B.; Gangneux, J.; Robert-Gangneux, F. Evaluation of the AllplexTM gastrointestinal panel-parasite assay for protozoa detection in stool samples: A retrospective and prospective study. Microorganisms 2020, 8, 569. [Google Scholar] [CrossRef]
- Kim, B.; Kim, H.S.; Kim, J.J.; Park, Y.J.; Kim, D.; Yong, D. Detection of intestinal protozoa in Korean patients using BD Max enteric parasite panel and Seegene Allplex gastrointestinal parasite assay. Lab. Med. 2020, 10, 221–226. [Google Scholar] [CrossRef]
- Galán-Puchades, M.T.; Trelis, M.; Sáez-Durán, S.; Cifre, S.; Gosálvez, C.; Sanxis-Furió, J.; Pascual, J.; Bueno-Marí, R.; Franco, S.; Peracho, V.; et al. One Health approach to zoonotic parasites: Molecular detection of intestinal protozoans in an urban population of Norway rats, Rattus norvegicus, in Barcelona, Spain. Pathogens 2021, 10, 311. [Google Scholar] [CrossRef]
- Klindworth, A.; Pruesse, E.; Schweer, T.; Peplies, J.; Quast, C.; Horn, M.; Glöckner, F.O. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res. 2013, 41, e1. [Google Scholar] [CrossRef]
- Schmieder, R.; Edwards, R. Quality control and preprocessing of metagenomic datasets. Bioinformatics 2011, 27, 863–864. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Peña, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef]
- Dao, M.C.; Everard, A.; Aron-Wisnewsky, J.; Sokolovska, N.; Prifti, E.; Verger, E.O.; Kayser, B.D.; Levenez, F.; Chilloux, J.; Hoyles, L.; et al. Akkermansia muciniphila and improved metabolic health during a dietary intervention in obesity: Relationship with gut microbiome richness and ecology. Gut 2016, 65, 426–436. [Google Scholar] [CrossRef]
- Sokol, H.; Pigneur, B.; Watterlot, L.; Lakhdari, O.; Bermúdez-Humarán, L.G.; Gratadoux, J.J.; Blugeon, S.; Bridonneau, C.; Furet, J.P.; Corthier, G.; et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc. Natl. Acad. Sci. USA 2008, 105, 16731–16736. [Google Scholar] [CrossRef]
- Tamanai-Shacoori, Z.; Smida, I.; Bousarghin, L.; Loreal, O.; Meuric, V.; Fong, S.B.; Bonnaure-Mallet, M.; Jolivet-Gougeon, A. Roseburia spp.: A marker of health? Future Microbiol 2017, 12, 157–170. [Google Scholar] [CrossRef]
- Russell, D.A.; Ross, R.P.; Fitzgerald, G.F.; Stanton, C. Metabolic activities and probiotic potential of bifidobacteria. Int. J. Food Microbiol. 2011, 149, 88–105. [Google Scholar] [CrossRef]
- Crovesy, L.; Ostrowski, M.; Ferreira, D.; Rosado, E.L.; Soares-Mota, M. Effect of Lactobacillus on body weight and body fat in overweight subjects: A systematic review of randomized controlled clinical trials. Int. J. Obes. 2017, 41, 1607–1614. [Google Scholar] [CrossRef] [PubMed]
- Ley, R.E. Gut microbiota in 2015: Prevotella in the gut: Choose carefully. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 69–70. [Google Scholar] [CrossRef] [PubMed]
- Christensen, L.; Roager, H.M.; Astrup, A.; Hjorth, M.F. Microbial enterotypes in personalized nutrition and obesity management. Am. J. Clin. Nutr. 2018, 108, 645–651. [Google Scholar] [CrossRef] [PubMed]
- RC Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- National Heart, Lung, and Blood Institute; The National Institute of Diabetes and Digestive and Kidney Diseases. Clinical Guidelines on the Identification, Evaluation, and Treatment of Overweight and Obesity in Adults: The Evidence Report; National Institutes of Health: Bethesda, MD, USA, 1998. [Google Scholar]
- Caudet, J.; Trelis, M.; Cifre, S.; Soriano, J.M.; Merino-Torres, J.F. Presence and significance of intestinal unicellular parasites in a morbidly obese population. Int. J. Obes. 2022, 46, 220–227. [Google Scholar] [CrossRef]
- Lim, M.Y.; You, H.J.; Yoon, H.S.; Kwon, B.; Lee, J.Y.; Lee, S.; Song, Y.M.; Lee, K.; Sung, J.; Ko, G. The effect of heritability and host genetics on the gut microbiota and metabolic syndrome. Gut 2017, 66, 1031–1038. [Google Scholar] [CrossRef]
- Kodio, A.; Menu, E.; Ranque, S. Eukaryotic and prokaryotic microbiota interactions. Microorganisms 2020, 8, 2018. [Google Scholar] [CrossRef]
- Kodio, A.; Coulibaly, D.; Koné, A.K.; Konaté, S.; Doumbo, S.; Guindo, A.; Bittar, F.; Gouriet, F.; Raoult, D.; Thera, M.A.; et al. Blastocystis colonization is associated with increased diversity and altered gut bacterial communities in healthy Malian children. Microorganisms 2019, 7, 649. [Google Scholar] [CrossRef]
- O’Brien Andersen, L.; Karim, A.B.; Roager, H.M.; Vigsnæs, L.K.; Krogfelt, K.A.; Licht, T.R.; Stensvold, C.R. Associations between common intestinal parasites bacteria in humans as revealed by qPCR. Eur. J. Clin. Microbiol. Infect Dis. 2016, 35, 1427–1431. [Google Scholar] [CrossRef]
- Forsell, J.; Bengtsson-Palme, J.; Angelin, M.; Johansson, A.; Evengård, B.; Granlund, M. The relation between Blastocystis and the intestinal microbiota in Swedish travellers. BMC Microbiol. 2017, 17, 231. [Google Scholar] [CrossRef]
- Krogsgaard, L.R.; Andersen La Johannesen, T.B.; Engsbro, A.L.; Stensvold, C.R.; Nielsen, H.V.; Bytzer, P. Characteristics of the bacterial microbiome in association with common intestinal parasites in irritable bowel syndrome. Clin. Transl. Gastroenterol. 2018, 9, 161. [Google Scholar] [CrossRef]
- Mejia, R.; Damania, A.; Jeun, R.; Bryan, P.E.; Vargas, P.; Juarez, M.; Cajal, P.S.; Nasser, J.; Krolewiecki, A.; Lefoulon, E.; et al. Impact of intestinal parasites on microbiota and cobalamin gene sequences: A pilot study. Parasites Vectors 2020, 13, 200. [Google Scholar] [CrossRef]
- Chen, T.L.; Chen, S.; Wu, H.W.; Lee, T.C.; Lu, Y.Z.; Wu, L.L.; Ni, Y.H.; Sun, C.H.; Yu, W.H.; Buret, A.G.; et al. Persistent gut barrier damage and commensal bacterial influx following eradication of Giardia infection in mice. Gut Pathog. 2013, 5, 26. [Google Scholar] [CrossRef]
- Billy, V.; Lhotská, Z.; Jirků, M.; Kadlecová, O.; Frgelecová, L.; Parfrey, L.W.; Pomajbíková, K.J. Blastocystis colonization alters the gut microbiome and, in some cases, promotes faster recovery from induced colitis. Front. Microbiol. 2021, 12, 641483. [Google Scholar] [CrossRef]
- Barash, N.R.; Maloney, J.G.; Singer, S.M.; Dawson, S.C. Giardia alters commensal microbial diversity throughout the murine gut. Infect. Immun. 2017, 85, e00948-e16. [Google Scholar] [CrossRef]
- Calderón de la Barca, A.M.; Castillo-Fimbres, R.S.; Mejía-León, M.E.; Quihui-Cota, L.; Ochoa-Leyva, A.; Aguayo-Patrón, S.V. Enteric parasitic infection disturbs bacterial structure in Mexican children with autoantibodies for type 1 diabetes and/or celiac disease. Gut Pathog. 2020, 12, 37. [Google Scholar] [CrossRef]
- Santacruz, A.; Collado, M.C.; Garcia-Valdes, L.; Segura, M.T.; Martin-Lagos, J.A.; Anjos, T.; Martí-Romero, M.; Lopez, R.M.; Florido, J.; Campoy, C.; et al. Gut microbiota composition is associated with body weight, weight gain and biochemical parameters in pregnant women. Br. J. Nutr. 2010, 104, 83–92. [Google Scholar] [CrossRef]
- Karlsson, C.L.; Önnerfält, J.; Xu, J.; Molin, G.; Ahrné, S.; Thorngren-Jerneck, K. The microbiota of the gut in preschool children with normal and excessive body weight. Obesity 2012, 20, 2257–2261. [Google Scholar] [CrossRef]
- Everard, A.; Belzer, C.; Geurts, L.; Ouwerkerk, J.P.; Druart, C.; Bindels, L.B.; Guiot, Y.; Derrien, M.; Muccioli, G.G.; Delzenne, N.M.; et al. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc. Natl. Acad. Sci. USA 2013, 110, 9066–9071. [Google Scholar] [CrossRef]
- Anhê, F.F.; Roy, D.; Pilon, G.; Dudonné, S.; Matamoros, S.; Varin, T.V.; Garofalo, C.; Moine, Q.; Desjardins, Y.; Levy, E.; et al. A polyphenol-rich cranberry extract protects from diet-induced obesity, insulin resistance and intestinal inflammation in association with increased Akkermansia spp. population in the gut microbiota of mice. Gut 2015, 64, 872–883. [Google Scholar] [CrossRef]
- Depommier, C.; Everard, A.; Druart, C.; Plovier, H.; Van Hul, M.; Vieira-Silva, S.; Falony, G.; Raes, J.; Maiter, D.; Delzenne, N.M.; et al. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: A proof-of-concept exploratory study. Nat. Med. 2019, 25, 1096–1103. [Google Scholar] [CrossRef] [PubMed]
- Machado, M.V.; Cortez-Pinto, H. Diet, microbiota, obesity, and NAFLD: A dangerous quartet. Int. J. Mol. Sci. 2016, 17, 481. [Google Scholar] [CrossRef] [PubMed]
- Mouzaki, M.; Comelli, E.M.; Arendt, B.M.; Bonengel, J.; Fung, S.K.; Fischer, S.E.; McGilvray, I.D.; Allard, J.P. Intestinal microbiota in patients with nonalcoholic fatty liver disease. Hepatology 2013, 58, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Wong, V.W.S.; Tse, C.H.; Lam, T.T.Y.; Wong, G.L.H.; Chim, A.M.L.; Chu, W.C.W.; Yeung, D.K.W.; Law, P.T.W.; Kwan, H.S.; Yu, J.; et al. Molecular characterization of the fecal microbiota in patients with nonalcoholic steatohepatitis—A longitudinal study. PLoS ONE 2013, 8, e62885. [Google Scholar] [CrossRef]
- Boursier, J.; Mueller, O.; Barret, M.; Machado, M.; Fizanne, L.; Araujo-Perez, F.; Guy, C.D.; Seed, P.C.; Rawls, J.F.; David, L.A.; et al. The severity of nonalcoholic fatty liver disease is associated with gut dysbiosis and shift in the metabolic function of the gut microbiota. Hepatology 2016, 63, 764–775. [Google Scholar] [CrossRef]
- Yoon, W.J.; Kim, H.; Park, E.; Ryu, S.; Chang, Y.; Shin, H.; Kim, H.L.; Yi, S.Y. The impact of cholecystectomy on the gut microbiota: A case-control study. J. Clin. Med. 2019, 8, 79. [Google Scholar] [CrossRef]

| Colonized | Non-Colonized | |
|---|---|---|
| Female n (%) | 19 (61.3) | 17 (68.0) |
| Male n (%) | 12 (38.7) | 8 (32.0) |
| Age (years) mean (SD) | 47.8 (10.3) | 48.6 (9.3) |
| BMI (kg/m2) mean (SD) | 47.3 (6.7) | 43.4 (5.8) |
| Abdominal circumference (cm) mean (SD) | 132.4 (11.6) | 126.9 (15.4) |
| Type II obesity n (%) | 3 (9.7) | 6 (24.0) |
| Type III obesity n (%) | 28 (90.3) | 19 (76.0) |
| Smokers n (%) | 11 (35.5) | 4 (16.0) |
| Meal replacement consumption n (%) | 11 (35.5) | 11 (44.0) |
| PPI regular use n (%) | 3 (9.7) | 5 (20.0) |
| History of a cholecystectomy n (%) | 1 (3.2) | 4 (16.0) |
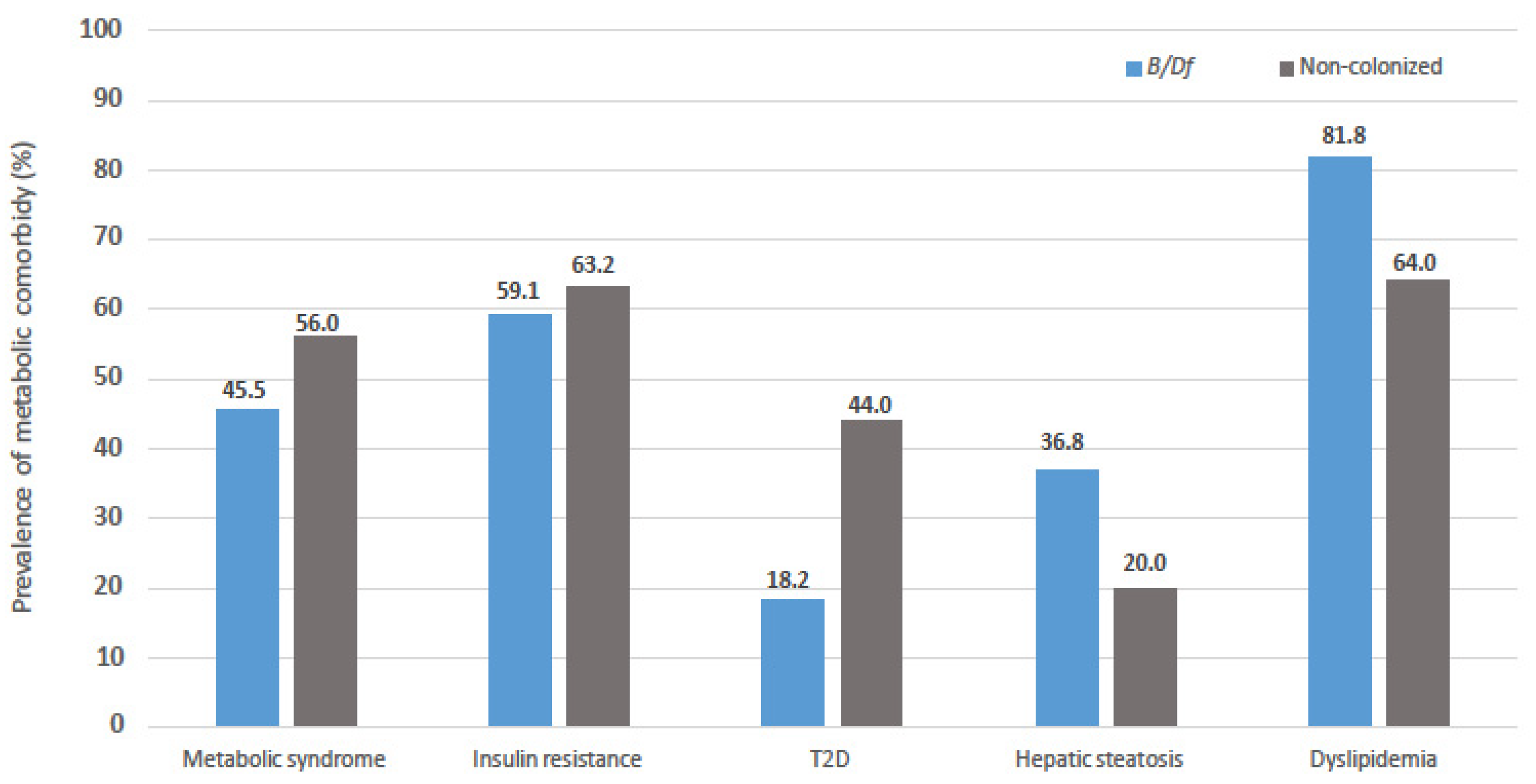
| Hypertension n (%) | 14 (45.2) | 16 (64.0) |
| Dyslipidaemia n (%) | 25 (80.6) | 16 (64.0) |
| T2D n (%) | 8 (25.8) | 11 (44.0) |
| Hepatic steatosis n (%) | 10 + (37.0) | 5 (20.0) |
| Hyperuricemia n (%) | 15 (48.4) | 11(44.0) |
| Metabolic syndrome n (%) | 15 (48.4) | 14 (56.0) |
| Insulin resistance n (%) | 17 (56.7) | 12 (63.2) |
| Non-Colonized (n = 25) | B/DF (n = 22) | G. intestinalis (n = 9) | |
|---|---|---|---|
| Chao 1 | 109.8 | 119.1 | 103.3 |
| Shannon | 2.89 | 2.94 | 2.72 |
| Jaccard | 0.443 | 0.477 | 0.376 |
| Sorensen | 0.599 | 0.632 | 0.520 |
| Metabolic syndrome | Metabolically healthy | |||
| Non-colonized | B/DF | Non-colonized | B/DF | |
| Chao 1 | 104.9 | 112.5 | 116.3 | 124.5 |
| Shannon | 2.86 | 2.74 | 2.94 | 3.09 |
| NAFLD positives | NAFLD negatives | |||
| Non-colonized | B/DF | Non-colonized | B/DF | |
| Chao 1 | 107.4 | 110.9 | 120.0 | 129.6 |
| Shannon | 2.90 | 2.75 | 2.89 | 3.09 |
| Jaccard | Sorensen | |||||
|---|---|---|---|---|---|---|
| Nc | B/DF | G. intestinalis | Nc | B/DF | G. intestinalis | |
| Metabolic syndrome | 0.429 | 0.400 | 0.320 * | 0.582 | 0.542 | 0.445 * |
| Dyslipidaemia | 0.459 | 0.475 * | 0.384 * | 0.610 | 0.622 * | 0.522 * |
| Hepatic steatosis | 0.430 | 0.425 | 0.331 * | 0.579 | 0.567 | 0.459 * |
| Insulin resistance | 0.444 | 0.481 * | 0.320 * | 0.598 | 0.627 * | 0.445 * |
| Type II diabetes mellitus | 0.412 | 0.341 | 0.279 * | 0.559 | 0.451 | 0.393 * |
| Non-Colonized | B/DF | G. intestinalis | |
|---|---|---|---|
| Phyla | |||
| Firmicutes | 40.1 | 42.2 | 35.0 |
| Bacteroidetes | 49.8 | 48.6 | 53.8 |
| Proteobacteria | 6.2 | 6.5 | 8.1 |
| Actinobacteria | 1.6 | 0.9 | 0.8 |
| Verrucomicrobia | 1.4 | 1.0 | 1.1 |
| Ratio (mean ± S.D.) | |||
| Firmicutes/Bacteroidetes | 0.97 ± 0.8 | 1.0 ± 0.7 | 0.68 ± 0.2 |
| Non-Colonized | B/DF | G. intestinalis | |
|---|---|---|---|
| Faecalibacterium spp. | 3.6 | 3.8 | 2.0 |
| Roseburia spp. | 2.9 | 3.6 | 2.2 |
| Akkermansia spp. | 1.3 | 0.9 | 1.1 |
| Bacteroides spp. | 21.3 | 20.5 | 27.1 |
| Prevotella spp. | 11.5 | 11.0 | 10.5 |
| Lactobacillus spp. | 0.09 | 0.82 | 0.03 |
| Bifidobacterium spp. | 0.68 | 0.28 | 0.51 |
| Escherichia spp. | 0.68 | 0.25 | 0.67 |
| Non-Colonized | B/DF | G. intestinalis | |
|---|---|---|---|
| Median [Q1, Q2] | Median [Q1, Q2] | Median [Q1, Q2] | |
| Bacteroides spp./Prevotella spp. | 460 [0.3, 11.300] | 200 [0.3, 4.681] | 1.625.0 [0.9, 3.687.1] |
| Faecalibacterium spp./Escherichia spp. | 40.6 [9.0, 107.6] | 63.5 [1.9, 277.8] | 84.6 [0.9, 345.0] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caudet, J.; Trelis, M.; Cifre, S.; Soriano, J.M.; Rico, H.; Merino-Torres, J.F. Interplay between Intestinal Bacterial Communities and Unicellular Parasites in a Morbidly Obese Population: A Neglected Trinomial. Nutrients 2022, 14, 3211. https://doi.org/10.3390/nu14153211
Caudet J, Trelis M, Cifre S, Soriano JM, Rico H, Merino-Torres JF. Interplay between Intestinal Bacterial Communities and Unicellular Parasites in a Morbidly Obese Population: A Neglected Trinomial. Nutrients. 2022; 14(15):3211. https://doi.org/10.3390/nu14153211
Chicago/Turabian StyleCaudet, Jana, María Trelis, Susana Cifre, José M. Soriano, Hortensia Rico, and Juan F. Merino-Torres. 2022. "Interplay between Intestinal Bacterial Communities and Unicellular Parasites in a Morbidly Obese Population: A Neglected Trinomial" Nutrients 14, no. 15: 3211. https://doi.org/10.3390/nu14153211
APA StyleCaudet, J., Trelis, M., Cifre, S., Soriano, J. M., Rico, H., & Merino-Torres, J. F. (2022). Interplay between Intestinal Bacterial Communities and Unicellular Parasites in a Morbidly Obese Population: A Neglected Trinomial. Nutrients, 14(15), 3211. https://doi.org/10.3390/nu14153211








