Reducing White Adipose Tissue Browning Using p38α MAPK Inhibitors Ameliorates Cancer-Associated Cachexia as Assessed by Magnetic Resonance Imaging
Abstract
:1. Introduction
2. Materials and Methods
2.1. Culture of Primary White Adipocytes and Pancreatic Cancer Cells
2.2. In Vitro Cell Culture Assay
2.3. In Vitro Assays for Evaluating p38 MAPK Inhibition
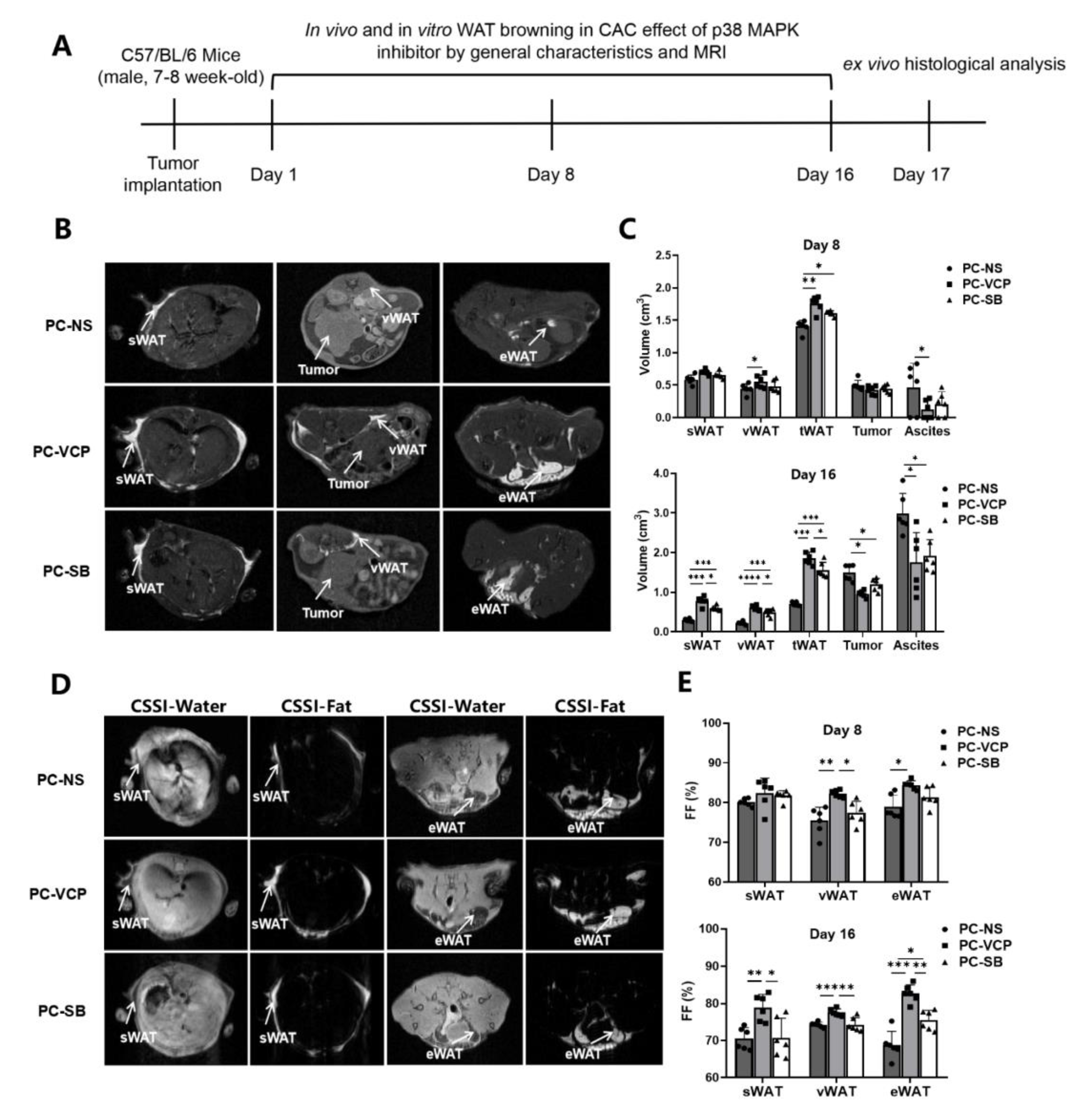
2.4. p38 MAPK Inhibitor Treatment for Mice with Pancreatic Cancer
2.5. MRI Scan and Image Analysis
2.6. Histopathologic Analysis and Western Blotting
2.7. Statistical Analysis
3. Results
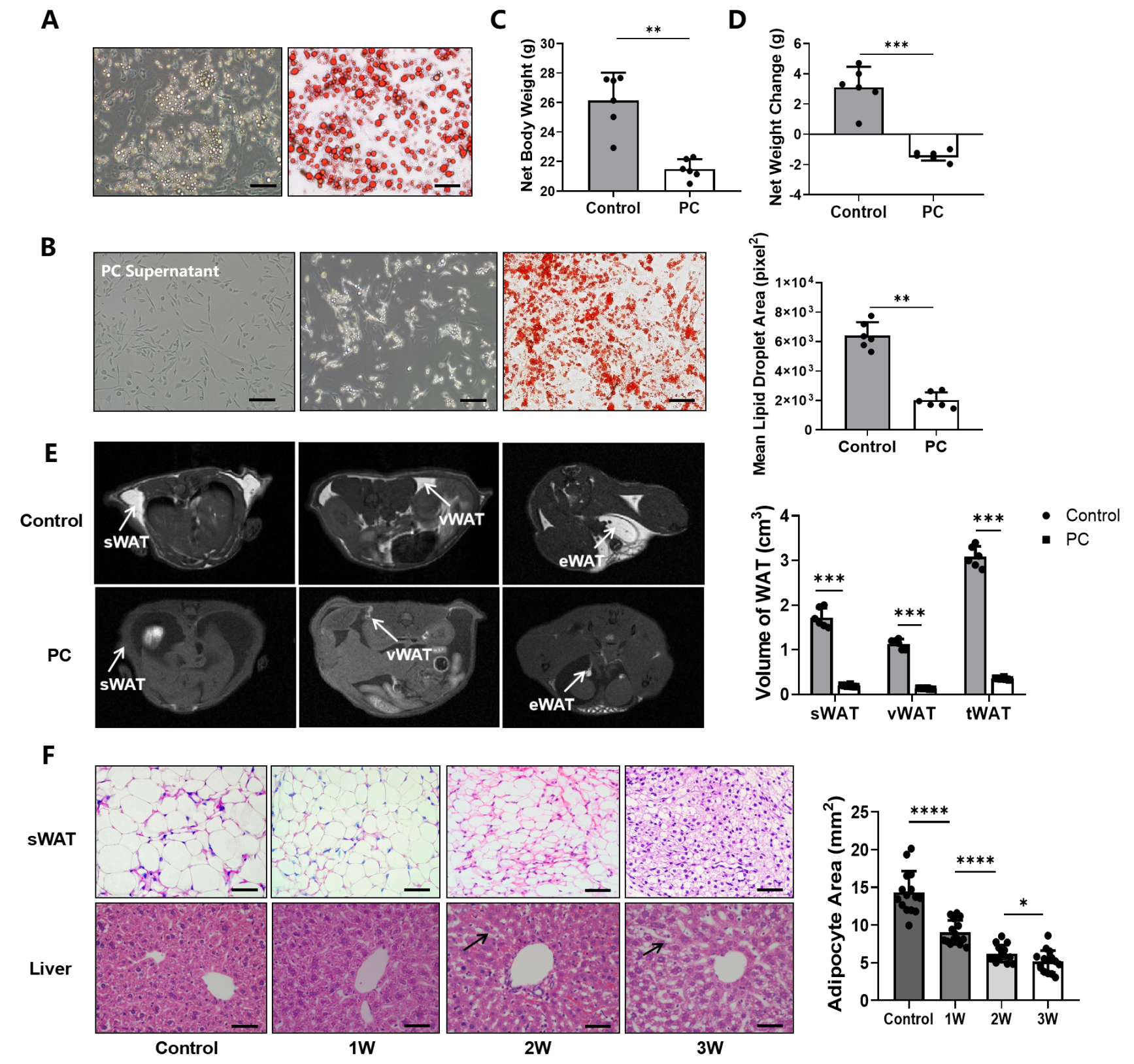
3.1. Pancreatic Cancer Promoting Lipid Metabolism That Leads to CAC
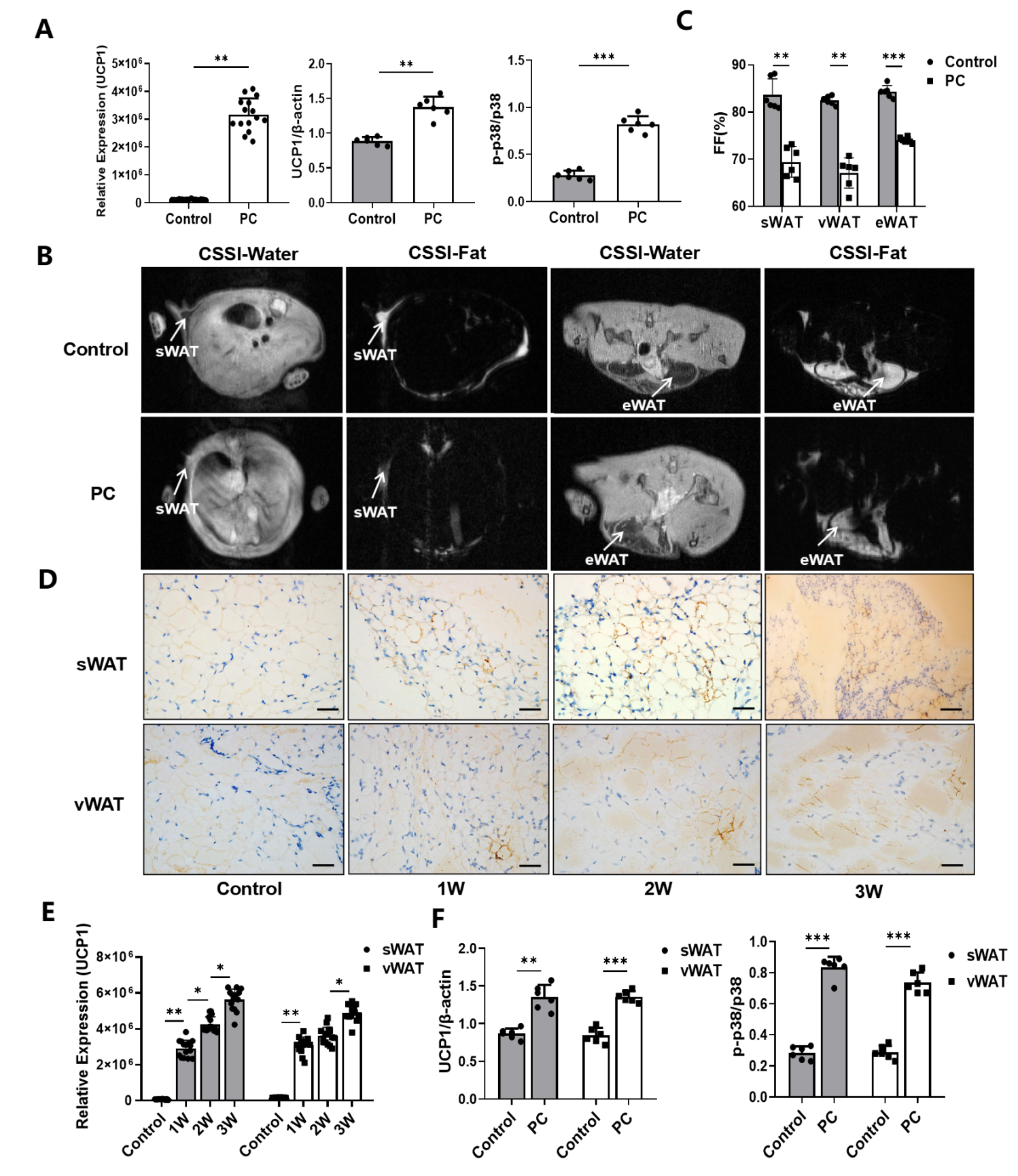
3.2. Increased WAT Browning in CAC
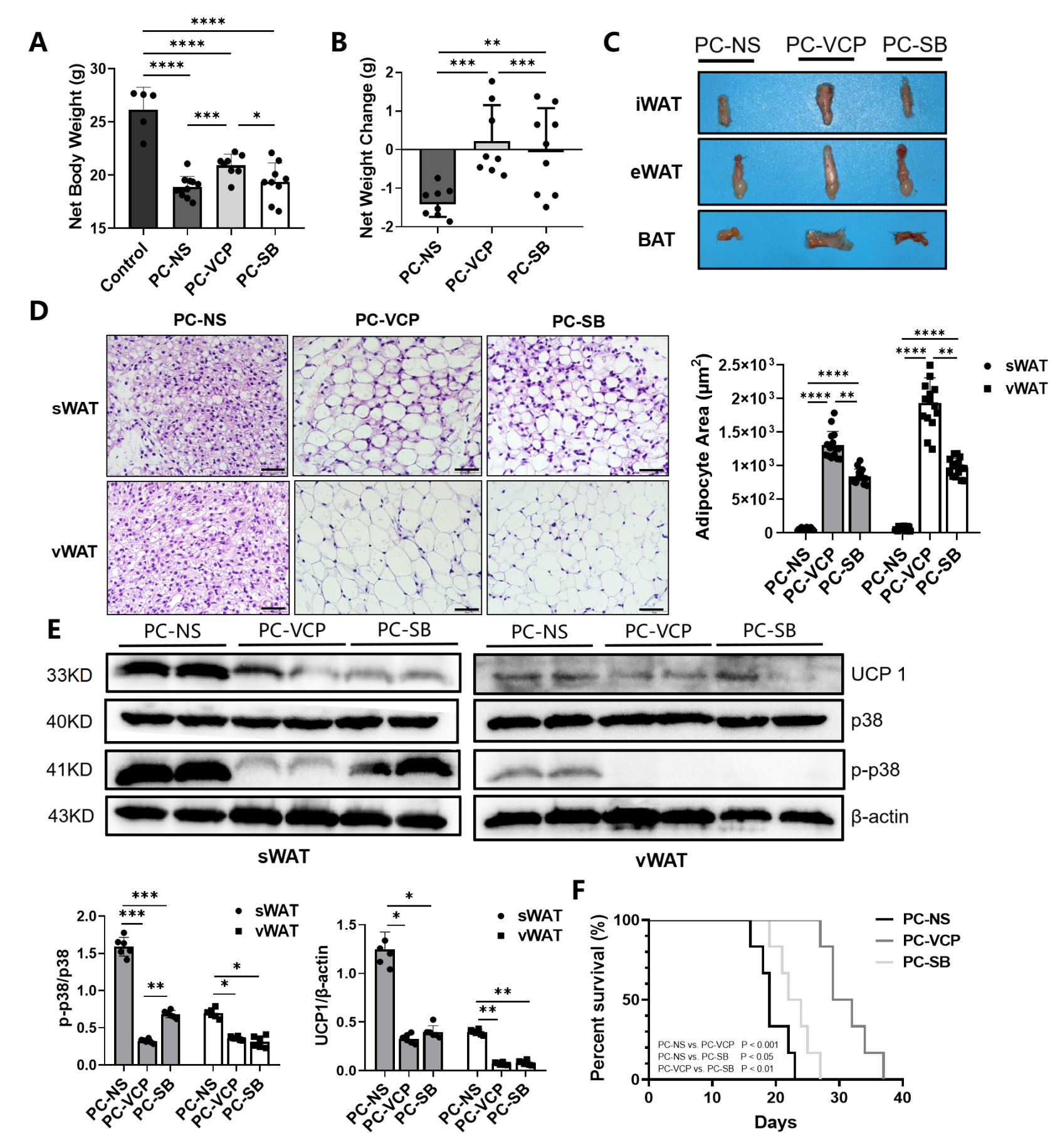
3.3. Effect of P38 MAPK Inhibitors on the Browning of White Adipocytes and CAC
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Baracos, V.E.; Martin, L.; Korc, M.; Guttridge, D.C.; Fearon, K.C.H. Cancer-associated cachexia. Nat. Rev. Dis. Primers 2018, 4, 17105. [Google Scholar] [CrossRef]
- Biswas, A.K.; Acharyya, S. Understanding cachexia in the context of metastatic progression. Nat. Rev. Cancer 2020, 20, 274–284. [Google Scholar] [CrossRef] [PubMed]
- Fearon, K.; Strasser, F.; Anker, S.D.; Bosaeus, I.; Bruera, E.; Fainsinger, R.L.; Jatoi, A.; Loprinzi, C.; MacDonald, N.; Mantovani, G.; et al. Definition and classification of cancer cachexia: An international consensus. Lancet Oncol. 2011, 12, 489–495. [Google Scholar] [CrossRef]
- Fearon, K.; Arends, J.; Baracos, V. Understanding the mechanisms and treatment options in cancer cachexia. Nat. Rev. Clin. Oncol. 2013, 10, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Petruzzelli, M.; Schweiger, M.; Schreiber, R.; Campos-Olivas, R.; Tsoli, M.; Allen, J.; Swarbrick, M.; Rose-John, S.; Rincon, M.; Robertson, G.; et al. A switch from white to brown fat increases energy expenditure in cancer-associated cachexia. Cell Metab. 2014, 20, 433–447. [Google Scholar] [CrossRef] [Green Version]
- Sah, R.P.; Sharma, A.; Nagpal, S.; Patlolla, S.H.; Sharma, A.; Kandlakunta, H.; Anani, V.; Angom, R.S.; Kamboj, A.K.; Ahmed, N.; et al. Phases of Metabolic and Soft Tissue Changes in Months Preceding a Diagnosis of Pancreatic Ductal Adenocarcinoma. Gastroenterology 2019, 156, 1742–1752. [Google Scholar] [CrossRef]
- Argilés, J.M.; Busquets, S.; Stemmler, B.; López-Soriano, F.J. Cancer cachexia: Understanding the molecular basis. Nat. Rev. Cancer 2014, 14, 754–762. [Google Scholar] [CrossRef]
- Kir, S.; White, J.P.; Kleiner, S.; Kazak, L.; Cohen, P.; Baracos, V.E.; Spiegelman, B.M. Tumour-derived PTH-related protein triggers adipose tissue browning and cancer cachexia. Nature 2014, 513, 100–104. [Google Scholar] [CrossRef]
- Kyriakis, J.M.; Avruch, J. Mammalian MAPK signal transduction pathways activated by stress and inflammation: A 10-year update. Physiol. Rev. 2012, 92, 689–737. [Google Scholar] [CrossRef] [Green Version]
- Nebreda, A.R.; Porras, A. p38 MAP kinases: Beyond the stress response. Trends Biochem. Sci. 2000, 25, 257–260. [Google Scholar] [CrossRef]
- Weston, C.R.; Davis, R.J. The JNK signal transduction pathway. Curr. Opin. Cell Biol. 2007, 19, 142–149. [Google Scholar] [CrossRef]
- Zarubin, T.; Han, J. Activation and signaling of the p38 MAP kinase pathway. Cell Res. 2005, 15, 11–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cao, W.; Daniel, K.W.; Robidoux, J.; Puigserver, P.; Medvedev, A.V.; Bai, X.; Floering, L.M.; Spiegelman, B.M.; Collins, S. p38 mitogen-activated protein kinase is the central regulator of cyclic AMP-dependent transcription of the brown fat uncoupling protein 1 gene. Mol. Cell. Biol. 2004, 24, 3057–3067. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Inagaki, T.; Sakai, J.; Kajimura, S. Transcriptional and epigenetic control of brown and beige adipose cell fate and function. Nat. Rev. Mol. Cell. Biol. 2016, 17, 480–495. [Google Scholar] [CrossRef] [Green Version]
- Xu, T.; Ding, J.; Ge, H.; Xu, X.; Scammells, P.; Wang, B.; Ju, S. Effects of VCP979 Novel p38 Mitogen Activated Protein Kinase Inhibitor on Progression of Pancreatic Cancer in Mouse Model with Diabetic Conditions. J. Biomed. Nanotechnol. 2019, 15, 1325–1333. [Google Scholar] [CrossRef]
- Huang, X.; He, C.; Hua, X.; Kan, A.; Mao, Y.; Sun, S.; Duan, F.; Wang, J.; Huang, P.; Li, S. Oxidative stress induces monocyte-to-myofibroblast transdifferentiation through p38 in pancreatic ductal adenocarcinoma. Clin. Transl. Med. 2020, 10, e41. [Google Scholar] [CrossRef] [PubMed]
- Tisdale, M.J. Mechanisms of cancer cachexia. Physiol. Rev. 2009, 89, 381–410. [Google Scholar] [CrossRef] [Green Version]
- Peng, X.-G.; Ju, S.; Fang, F.; Wang, Y.; Fang, K.; Cui, X.; Liu, G.; Li, P.; Mao, H.; Teng, G.-J. Comparison of brown and white adipose tissue fat fractions in ob, seipin, and Fsp27 gene knockout mice by chemical shift-selective imaging and (1)H-MR spectroscopy. Am. J. Physiol. Endocrinol. Metab. 2013, 304, E160–E167. [Google Scholar] [CrossRef] [Green Version]
- Orsso, C.E.; Mackenzie, M.; Alberga, A.S.; Sharma, A.M.; Richer, L.; Rubin, D.A.; Prado, C.M.; Haqq, A.M. The use of magnetic resonance imaging to characterize abnormal body composition phenotypes in youth with Prader-Willi syndrome. Metabolism 2017, 69, 67–75. [Google Scholar] [CrossRef]
- Yang, J.-Y.; Della-Fera, M.A.; Rayalam, S.; Park, H.J.; Ambati, S.; Hausman, D.B.; Hartzell, D.L.; Baile, C.A. Regulation of adipogenesis by medium-chain fatty acids in the absence of hormonal cocktail. J. Nutr. Biochem. 2009, 20, 537–543. [Google Scholar] [CrossRef]
- Ying, W.; Riopel, M.; Bandyopadhyay, G.; Dong, Y.; Birmingham, A.; Seo, J.B.; Ofrecio, J.M.; Wollam, J.; Hernandez-Carretero, A.; Fu, W.; et al. Adipose Tissue Macrophage-Derived Exosomal miRNAs Can Modulate In Vivo and In Vitro Insulin Sensitivity. Cell 2017, 171, 372–384. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lv, X.; Qiu, J.; Hao, T.; Zhang, H.; Jiang, H.; Tan, Y. HDAC inhibitor Trichostatin A suppresses adipogenesis in 3T3-L1 preadipocytes. Aging 2021, 13, 17489–17498. [Google Scholar] [CrossRef] [PubMed]
- Miao, C.; Lv, Y.; Zhang, W.; Chai, X.; Feng, L.; Fang, Y.; Liu, X.; Zhang, X. Pyrrolidine Dithiocarbamate (PDTC) Attenuates Cancer Cachexia by Affecting Muscle Atrophy and Fat Lipolysis. Front. Pharmacol. 2017, 8, 915. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, S.; Li, Y.; Shen, Q.; Zhang, W.; Gu, X.; Ma, M.; Li, Y.; Zhang, L.; Liu, X.; Zhang, X. Carnosol and its analogues attenuate muscle atrophy and fat lipolysis induced by cancer cachexia. J. Cachexia Sarcopenia Muscle 2021, 12, 779–795. [Google Scholar] [CrossRef] [PubMed]
- Kershaw, J.C.; Elzey, B.D.; Guo, X.-X.; Kim, K.-H. Piceatannol, a Dietary Polyphenol, Alleviates Adipose Tissue Loss in Pre-Clinical Model of Cancer-Associated Cachexia via Lipolysis Inhibition. Nutrients 2022, 14, 2306. [Google Scholar] [CrossRef]
- Shen, Q.; Kuang, J.-X.; Miao, C.-X.; Zhang, W.-L.; Li, Y.-W.; Zhang, X.-W.; Liu, X. Alantolactone ameliorates cancer cachexia-associated muscle atrophy mainly by inhibiting the STAT3 signaling pathway. Phytomedicine 2022, 95, 153858. [Google Scholar] [CrossRef]
- Lim, S.; Deaver, J.W.; Rosa-Caldwell, M.E.; Haynie, W.S.; Morena da Silva, F.; Cabrera, A.R.; Schrems, E.R.; Saling, L.W.; Jansen, L.T.; Dunlap, K.R.; et al. Development of metabolic and contractile alterations in development of cancer cachexia in female tumor-bearing mice. J. Appl. Physiol. 2022, 132, 58–72. [Google Scholar] [CrossRef]
- Banh, T.; Snoke, D.; Cole, R.M.; Angelotti, A.; Schnell, P.M.; Belury, M.A. Higher tumor mass and lower adipose mass are associated with colon-26 adenocarcinoma-induced cachexia in male, female and ovariectomized mice. Oncol. Rep. 2019, 41, 2909–2918. [Google Scholar] [CrossRef]
- Wang, M.; Li, Y.; Wang, M.; Liu, K.; Hoover, A.R.; Li, M.; Towner, R.A.; Mukherjee, P.; Zhou, F.; Qu, J.; et al. Synergistic interventional photothermal therapy and immunotherapy using an iron oxide nanoplatform for the treatment of pancreatic cancer. Acta Biomater. 2022, 138, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Li, J.; Wang, H.; Chiu, Y.; Kingsley, C.V.; Fry, D.; Delaney, S.N.; Wei, S.C.; Zhang, J.; Maitra, A.; et al. Combination of PD-1 Inhibitor and OX40 Agonist Induces Tumor Rejection and Immune Memory in Mouse Models of Pancreatic Cancer. Gastroenterology 2020, 159, 306–319. [Google Scholar] [CrossRef]
- Pan, L.; Shang, N.; Shangguan, J.; Figini, M.; Xing, W.; Wang, B.; Sun, C.; Yang, J.; Zhang, Y.; Hu, S.; et al. Magnetic resonance imaging monitoring therapeutic response to dendritic cell vaccine in murine orthotopic pancreatic cancer models. Am. J. Cancer Res. 2019, 9, 562–573. [Google Scholar] [PubMed]
- Xie, Y.; Zhu, S.; Zhong, M.; Yang, M.; Sun, X.; Liu, J.; Kroemer, G.; Lotze, M.; Zeh, H.J.; Kang, R.; et al. Inhibition of Aurora Kinase A Induces Necroptosis in Pancreatic Carcinoma. Gastroenterology 2017, 153, 1429–1443. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.; Ding, J.; Zhao, Y.; Ju, S.; Mao, H.; Peng, X.-G. Irisin induces white adipose tissue browning in mice as assessed by magnetic resonance imaging. Exp. Biol. Med. 2021, 246, 1597–1606. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.-G.; Ju, S.; Qin, Y.; Fang, F.; Cui, X.; Liu, G.; Ni, Y.; Teng, G.-J. Quantification of liver fat in mice: Comparing dual-echo Dixon imaging, chemical shift imaging, and 1H-MR spectroscopy. J. Lipid Res. 2011, 52, 1847–1855. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Auger, C.; Knuth, C.M.; Abdullahi, A.; Samadi, O.; Parousis, A.; Jeschke, M.G. Metformin prevents the pathological browning of subcutaneous white adipose tissue. Mol. Metab. 2019, 29, 12–23. [Google Scholar] [CrossRef]
- Kays, J.K.; Shahda, S.; Stanley, M.; Bell, T.M.; O’Neill, B.H.; Kohli, M.D.; Couch, M.E.; Koniaris, L.G.; Zimmers, T.A. Three cachexia phenotypes and the impact of fat-only loss on survival in FOLFIRINOX therapy for pancreatic cancer. J. Cachexia Sarcopenia Muscle 2018, 9, 673–684. [Google Scholar] [CrossRef]
- Qiu, Y.; Nguyen, K.D.; Odegaard, J.I.; Cui, X.; Tian, X.; Locksley, R.M.; Palmiter, R.D.; Chawla, A. Eosinophils and type 2 cytokine signaling in macrophages orchestrate development of functional beige fat. Cell 2014, 157, 1292–1308. [Google Scholar] [CrossRef] [Green Version]
- Abdullahi, A.; Jeschke, M.G. White Adipose Tissue Browning: A Double-edged Sword. Trends Endocrinol. Metab. 2016, 27, 542–552. [Google Scholar] [CrossRef] [Green Version]
- Carobbio, S.; Guenantin, A.C.; Samuelson, I.; Bahri, M.; Vidal-Puig, A. Brown and beige fat: From molecules to physiology and pathophysiology. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2019, 1864, 37–50. [Google Scholar] [CrossRef]
- Pfeifer, A.; Hoffmann, L.S. Brown, beige, and white: The new color code of fat and its pharmacological implications. Annu. Rev. Pharmacol. Toxicol. 2015, 55, 207–227. [Google Scholar] [CrossRef]
- Molfino, A.; Carletti, R.; Imbimbo, G.; Amabile, M.I.; Belli, R.; di Gioia, C.R.T.; Belloni, E.; Spinelli, F.; Rizzo, V.; Catalano, C.; et al. Histomorphological and inflammatory changes of white adipose tissue in gastrointestinal cancer patients with and without cachexia. J. Cachexia Sarcopenia Muscle 2022, 13, 333–342. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.J.; Lee, J.; Anderson, B.; Lee, B.; Migula, D.; Sauer, A.; Chong, N.; Liu, H.; Wu, P.C.; Dash, A.; et al. Whole-body and adipose tissue metabolic phenotype in cancer patients. J. Cachexia Sarcopenia Muscle 2022, 13, 1124–1133. [Google Scholar] [CrossRef] [PubMed]
- Kassouf, T.; Sumara, G. Impact of Conventional and Atypical MAPKs on the Development of Metabolic Diseases. Biomolecules 2020, 10, 1256. [Google Scholar] [CrossRef] [PubMed]
- Ronkina, N.; Gaestel, M. MAPK-Activated Protein Kinases: Servant or Partner? Annu. Rev. Biochem. 2022, 91, 505–540. [Google Scholar] [CrossRef] [PubMed]
- Luan, Y.; Zhang, Y.; Yu, S.-Y.; You, M.; Xu, P.C.; Chung, S.; Kurita, T.; Zhu, J.; Kim, S.-Y. Development of ovarian tumour causes significant loss of muscle and adipose tissue: A novel mouse model for cancer cachexia study. J. Cachexia Sarcopenia Muscle 2022, 13, 1289–1301. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.; Li, L.; Du, Y.; Zhu, H.; Li, Z.; Kong, X. Exosomal adrenomedullin derived from cancer-associated fibroblasts promotes lipolysis in adipose tissue. Gut 2018, 67, 2226–2227. [Google Scholar] [CrossRef]
- Manieri, E.; Sabio, G. Stress kinases in the modulation of metabolism and energy balance. J. Mol. Endocrinol. 2015, 55, R11–R22. [Google Scholar] [CrossRef]
- Matesanz, N.; Nikolic, I.; Leiva, M.; Pulgarín-Alfaro, M.; Santamans, A.M.; Bernardo, E.; Mora, A.; Herrera-Melle, L.; Rodríguez, E.; Beiroa, D.; et al. p38α blocks brown adipose tissue thermogenesis through p38δ inhibition. PLoS Biol. 2018, 16, e2004455. [Google Scholar] [CrossRef]
- Zhang, S.; Cao, H.; Li, Y.; Jing, Y.; Liu, S.; Ye, C.; Wang, H.; Yu, S.; Peng, C.; Hui, L.; et al. Metabolic benefits of inhibition of p38α in white adipose tissue in obesity. PLoS Biol. 2018, 16, e2004225. [Google Scholar] [CrossRef] [Green Version]
- Yang, L.; Sun, X.; Ye, Y.; Lu, Y.; Zuo, J.; Liu, W.; Elcock, A.; Zhu, S. p38alpha Mitogen-Activated Protein Kinase Is a Druggable Target in Pancreatic Adenocarcinoma. Front. Oncol. 2019, 9, 1294. [Google Scholar] [CrossRef]
- Vinh, N.B.; Devine, S.M.; Munoz, L.; Ryan, R.M.; Wang, B.H.; Krum, H.; Chalmers, D.K.; Simpson, J.S.; Scammells, P.J. Design, Synthesis, and Biological Evaluation of Tetra-Substituted Thiophenes as Inhibitors of p38alpha MAPK. ChemistryOpen 2015, 4, 56–64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Henriques, F.; Lopes, M.A.; Franco, F.O.; Knobl, P.; Santos, K.B.; Bueno, L.L.; Correa, V.A.; Bedard, A.H.; Guilherme, A.; Birbrair, A.; et al. Toll-Like Receptor-4 Disruption Suppresses Adipose Tissue Remodeling and Increases Survival in Cancer Cachexia Syndrome. Sci. Rep. 2018, 8, 18024. [Google Scholar] [CrossRef] [Green Version]
- Han, Y.-H.; Mun, J.-G.; Jeon, H.D.; Yoon, D.H.; Choi, B.-M.; Kee, J.-Y.; Hong, S.-H. The Extract of Arctium lappa L. Fruit (Arctii Fructus) Improves Cancer-Induced Cachexia by Inhibiting Weight Loss of Skeletal Muscle and Adipose Tissue. Nutrients 2020, 12, 3195. [Google Scholar] [CrossRef] [PubMed]
- Sarjeant, K.; Stephens, J.M. Adipogenesis. Cold Spring Harb. Perspect. Biol. 2012, 4, a008417. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chondronikola, M.; Beeman, S.C.; Wahl, R.L. Non-invasive methods for the assessment of brown adipose tissue in humans. J. Physiol. 2018, 596, 363–378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, J.; Meng, Q.; Liang, X.; Zhuang, R.; Yuan, D.; Ge, X.; Cao, H.; Lin, F.; Gong, X.; Fan, H.; et al. A novel small molecule compound VCP979 improves ventricular remodeling in murine models of myocardial ischemia/reperfusion injury. Int. J. Mol. Med. 2020, 45, 353–364. [Google Scholar] [CrossRef] [Green Version]
- Becker, A.S.; Zellweger, C.; Bacanovic, S.; Franckenberg, S.; Nagel, H.W.; Frick, L.; Schawkat, K.; Eberhard, M.; Blüthgen, C.; Volbracht, J.; et al. Brown fat does not cause cachexia in cancer patients: A large retrospective longitudinal FDG-PET/CT cohort study. PLoS ONE 2020, 15, e0239990. [Google Scholar] [CrossRef]
- Rohm, M.; Schäfer, M.; Laurent, V.; Üstünel, B.E.; Niopek, K.; Algire, C.; Hautzinger, O.; Sijmonsma, T.P.; Zota, A.; Medrikova, D.; et al. An AMP-activated protein kinase-stabilizing peptide ameliorates adipose tissue wasting in cancer cachexia in mice. Nat. Med. 2016, 22, 1120–1130. [Google Scholar] [CrossRef]

| Day 8 | Day 16 | |||||
|---|---|---|---|---|---|---|
| PC-NS | PC-VCP | PC-SB | PC-NS | PC-VCP | PC-SB | |
| sWAT (cm3) | 0.59 ± 0.07 | 0.70 ± 0.04 | 0.66 ± 0.05 | 0.30 ± 0.03 | 0.78 ± 0.11 *** | 0.60 ± 0.06 ###† |
| vWAT (cm3) | 0.45 ± 0.08 | 0.56 ± 0.10 * | 0.50 ± 0.08 | 0.22 ± 0.03 | 0.61 ± 0.07 **** | 0.49 ± 0.09 ###† |
| tWAT (cm3) | 1.41 ± 0.09 | 1.76 ± 0.12 ** | 1.58 ± 0.08 # | 0.70 ± 0.05 | 1.86 ± 0.16 *** | 1.56 ± 0.19 ###† |
| Tumor (cm3) | 0.50 ± 0.08 | 0.42 ± 0.07 | 0.44 ± 0.06 | 1.49 ± 0.20 | 0.96 ± 0.08 * | 1.19 ± 0.15 # |
| Ascites (cm3) | 0.40 ± 0.47 | 0.09 ± 0.18 * | 0.19 ± 0.19 | 3.00 ± 0.51 | 1.75 ± 0.76 * | 1.91 ± 0.41 # |
| Fat Fractions (%) | Day 8 | Day 16 | ||||
|---|---|---|---|---|---|---|
| PC-NS | PC-VCP | PC-SB | PC-NS | PC-VCP | PC-SB | |
| sWAT | 80.08 ± 0.92 | 82.43 ± 3.69 | 81.71 ± 1.30 | 69.67 ± 3.10 | 80.48 ± 0.92 ** | 72.26 ± 4.65 † |
| vWAT | 75.43 ± 3.41 | 82.22 ± 0.93 ** | 77.50 ± 2.88 † | 74.48 ± 0.61 | 77.44 ± 1.14 *** | 73.79 ± 1.66 †† |
| eWAT | 78.91 ± 2.96 | 84.37 ± 1.12 * | 81.39 ± 2.51 | 69.72 ± 3.51 | 82.54 ± 2.70 *** | 76.54 ± 1.43 #†† |
| PC-NS | PC-VCP | PC-SB | |
|---|---|---|---|
| Net weight (g) | 18.88 ± 1.00 | 20.92 ± 1.05 *** | 19.38 ± 1.78 † |
| Net weight change (g) | −1.61 ± 0.59 | 0.17 ± 0.99 *** | −0.01 ± 1.09 ##††† |
| Percentage of change (%) | −6.71 ± 1.18 | 0.11 ± 3.77 *** | −2.02 ± 3.36 ###††† |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, Y.; Dai, J.; Jiang, Y.; Wu, H.; Cui, Y.; Li, X.; Mao, H.; Wang, B.; Ju, S.; Peng, X.-G. Reducing White Adipose Tissue Browning Using p38α MAPK Inhibitors Ameliorates Cancer-Associated Cachexia as Assessed by Magnetic Resonance Imaging. Nutrients 2022, 14, 3013. https://doi.org/10.3390/nu14153013
Zhao Y, Dai J, Jiang Y, Wu H, Cui Y, Li X, Mao H, Wang B, Ju S, Peng X-G. Reducing White Adipose Tissue Browning Using p38α MAPK Inhibitors Ameliorates Cancer-Associated Cachexia as Assessed by Magnetic Resonance Imaging. Nutrients. 2022; 14(15):3013. https://doi.org/10.3390/nu14153013
Chicago/Turabian StyleZhao, Yufei, Jingyue Dai, Yang Jiang, Honghong Wu, Ying Cui, Xinxiang Li, Hui Mao, Binghui Wang, Shenghong Ju, and Xin-Gui Peng. 2022. "Reducing White Adipose Tissue Browning Using p38α MAPK Inhibitors Ameliorates Cancer-Associated Cachexia as Assessed by Magnetic Resonance Imaging" Nutrients 14, no. 15: 3013. https://doi.org/10.3390/nu14153013
APA StyleZhao, Y., Dai, J., Jiang, Y., Wu, H., Cui, Y., Li, X., Mao, H., Wang, B., Ju, S., & Peng, X.-G. (2022). Reducing White Adipose Tissue Browning Using p38α MAPK Inhibitors Ameliorates Cancer-Associated Cachexia as Assessed by Magnetic Resonance Imaging. Nutrients, 14(15), 3013. https://doi.org/10.3390/nu14153013





