Use of 1-MNA to Improve Exercise Tolerance and Fatigue in Patients after COVID-19
Abstract
1. Introduction
2. Methods
2.1. Patients and Eligibility Criteria
- SARS-CoV-2 virus infection was confirmed by the RT-PCR test result in accordance with the current guidelines of the Ministry of Health of Poland with home isolation (no hospitalization);
- Patients expressing subjective feelings of limited tolerance to exercise, and above 50% greater fatigue, compared with their pre-COVID-19 levels (symptoms must have continued for at least four weeks since the last symptoms of infection);
- No myocardial infarction, no heart failure diagnosis before COVID-19;
- No pulmonary diseases;
- Age ≥ 18 years;
- Consent of the respondent to participate in the study.
- Cardiological or pulmonological complications after COVID-19;
- Anemia in blood test count;
- Hypothyroidism and/or hyperthyroidism;
- Renal insufficiency with eGFR < 60 mL/min.
2.2. Study Protocol
- Gr 1: NO-1-MNA—without supplementation;
- Gr 2: 1-MNA—with 1-MNA supplementation. 1-MNA supplements were taken once a day at a dose of 58 mg, in the morning after a meal.
- oxygen saturation assessment performed with a pulse oximeter;
- heart rate monitoring;
- dyspnea assessment according to the Borg scale;
- assessment of fatigue with the Fatigue Severity Scale (FSS) test questionnaire.
2.3. Fatigue Assessment
- distance, and the mean difference in distance, in 6MWT;
- number and percentage of pts with improved distance in 6MWT;
- FSS score;
- number and percentage of patients with FSS ≥ 4.
2.4. Bioethics Approval
2.5. Statistical Analysis
3. Results
3.1. Results after One Month with MNA-1 Supplementation
3.1.1. Six-Minute Walk Test
3.1.2. Fatigue
4. Discussion
5. Conclusions
- 1-MNA supplementation significantly improved physical performance in a 6-minute walk test and reduced the percentage of patients with severe fatigue after COVID-19;
- The comprehensive action of 1-MNA, including anti-inflammatory and anticoagulant effects, as well as activation of the SIRT1 enzyme, may be beneficial for the recovery of patients with persistent symptoms of fatigue and low tolerance to exercise after COVID-19.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Carfi, A.; Bernabei, R.; Landi, F.; Gemelli Against COVID-19 Post-Acute Care Study Group. Persistent symptoms in patients after acute COVID-19. JAMA 2020, 324, 603–605. [Google Scholar] [CrossRef] [PubMed]
- Moldofsky, H.; Patcai, J. Chronic widespread musculoskeletal pain, fatigue, depression and disordered sleep in chronic post-SARS syndrome; a case-controlled study. BMC Neurol. 2011, 11, 37. [Google Scholar] [CrossRef] [PubMed]
- Lam, M.H.-B.; Wing, Y.-K.; Yu, M.W.-M.; Leung, C.-M.; Ma, R.C.; Kong, A.P.; So, W.Y.; Fong, S.Y.; Lam, S.P. Mental morbidities and chronic fatigue in severe acute respiratory syndrome survivors: Long-term follow-up. Arch. Intern. Med. 2009, 169, 2142–2147. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Hu, B.; Hu, C.; Zhu, F.; Liu, X.; Zhang, J.; Wang, B.; Xiang, H.; Cheng, Z.; Xiong, Y.; et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus–infected pneumonia in Wuhan, China. JAMA 2020, 323, 1061–1069. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.-W.; Wu, X.; Jiang, X.-G.; Xu, K.-J.; Ying, L.-J.; Ma, C.-L.; Li, S.-B.; Wang, H.-Y.; Zhang, S.; Gao, H.-N.; et al. Clinical findings in a group of patients infected with the 2019 novel coronavirus (SARS-Cov-2) outside of Wuhan, China: Retrospective case series. BMJ 2020, 368, m606. [Google Scholar] [CrossRef]
- Tian, S.; Hu, N.; Lou, J.; Chen, K.; Kang, X.; Xiang, Z.; Chen, H.; Wang, D.; Liu, N.; Liu, D.; et al. Characteristics of COVID-19 infection in Beijing. J. Infect. 2020, 80, 401–406. [Google Scholar] [CrossRef]
- Afari, N.; Buchwald, D. Chronic fatigue syndrome: A review. Am. J. Psychiatry. 2003, 160, 221–236. [Google Scholar] [CrossRef]
- Bansal, A.; Bradley, A.; Bishop, K.; Kiani-Alikhan, S.; Ford, B. Chronic fatigue syndrome, the immune system and viral infection. Brain Behav. Immun. 2012, 26, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Klimas, N.G.; Broderick, G.; Fletcher, M.A. Biomarkers for chronic fatigue. Brain Behav. Immun. 2012, 26, 1202–1210. [Google Scholar] [CrossRef] [PubMed]
- Montoya, J.G.; Holmes, T.H.; Anderson, J.N.; Maecker, H.T.; Rosenberg-Hasson, Y.; Valencia, I.; Chu, L.; Younger, J.-W.; Tato, C.M.; Davis, M.M. Cytokine signature associated with disease severity in chronic fatigue syndrome patients. Proc. Natl. Acad. Sci. USA 2017, 114, E7150–E7158. [Google Scholar] [CrossRef] [PubMed]
- Natelson, B.H.; Haghighi, M.H.; Ponzio, N.M. Evidence for the presence of immune dysfunction in chronic fatigue syndrome. Clin. Diagn Lab. Immunol. 2002, 9, 747–752. [Google Scholar] [CrossRef] [PubMed]
- Gąsecka, A.; Filipiak, K.J.; Jaguszewski, M.J. Impaired microcirculation function in COVID-19 and implications for potential therapies. Cardiol. J. 2020, 27, 485–488. [Google Scholar] [CrossRef] [PubMed]
- Jimeno-Almazán, A.; Pallares, J.G.; Buendía-Romero, A.; Martinez-Cava, A.; Franco-Lopez, F.; Sanchez-Alcaraz Martinez, B.J.; Bernal-Morel, E.; Courel-Lbanez, J. Post-COVID-19 Syndrome and the Potential Benefits of Exercise. Int. J. Environ. Res. Public Health 2021, 18, 5329. [Google Scholar] [CrossRef]
- Chlopicki, S.; Swies, J.; Mogielnicki, A.; Buczko, W.; Bartus, M.; Lomnicka, M.; Adamus, J.; Gebicki, J. 1-Methylnicotinamide (MNA), a primary metabolite of nicotinamide, exerts anti-thrombotic activity mediated by a cyclooxygenase-2/prostacyclin pathway. Br. J. Pharmacol. 2007, 152, 230–239. [Google Scholar] [CrossRef]
- Gebicki, J.; Sysa-Jedrzejowska, A.; Adamus, J.; Woźniacka, A.; Rybak, M.; Zielonka, J. 1-Methylnicotinamide: A potent anti-inflammatory agent of vitamin origin. Pol. J. Pharmacol. 2003, 55, 109–112. [Google Scholar]
- Schmeisser, K.; Mansfeld, J.; Kuhlow, D.; Weimer, S.; Priebe, S.; Heiland, I.; Birringer, M.; Groth, M.; Segref, A.; Kanfi, Y.; et al. Role of sirtuins in lifespan regulation is linked to methylation of nicotinamide. Nat. Chem. Biol. 2013, 9, 693–700. [Google Scholar] [CrossRef]
- Domagala, T.B.; Szeffler, A.; Dobrucki, L.W.; Dropinski, J.; Polanski, S.; Leszczynska-Wiloch, M.; Kotula-Horowitz, K.; Wojciechowski, J.; Wojnowski, L.; Szczeklik, A.; et al. Nitric oxide production and endothelium-dependent vasorelaxation ameliorated by N1-methylnicotinamide in human blood vessels. Hypertension 2012, 59, 825–832. [Google Scholar] [CrossRef]
- Ström, K.; Morales-Alamo, D.; Ottosson, F.; Edlund, A.; Hjort, L.; Jorgensen, S.W.; Almgren, P.; Zhou, Y.; Martin-Rincon, M.; Ekman, C.; et al. N1-methylnicotinamide is a signalling molecule produced in skeletal muscle coordinating energy metabolism. Sci. Rep. 2018, 14, 3016. [Google Scholar] [CrossRef]
- Valko, P.O.; Bassetti, C.L.; Bloch, K.E.; Held, U.; Baumann, C.h.R. Validation of the Fatigue Severity Scale in a Swiss Cohort. Sleep 2008, 31, 1601–1607. [Google Scholar] [CrossRef]
- Armutlu, K.; Korkmaz, N.C.; Keser, I.; Sumbuloglu, V.; Akbiyik, D.I.; Guney, Z.; Karabudak, R. The validity and reliability of the Fatigue Severity Scale in Turkish multiple sclerosis patients. Int. J. Rehabil. Res. 2007, 30, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Kaynak, H.; Altintaş, A.; Kaynak, D.; Uyanik, O.; Saip, S.; Agaoglu, J.; Onger, G.; Siva, A. Fatigue and sleep disturbance in multiple sclerosis. Eur. J. Neurol. 2006, 13, 1333–1339. [Google Scholar] [CrossRef] [PubMed]
- ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories. ATS Statement: Guidelines for the six-minute walk test. Am. J. Respir. Crit. Care Med. 2002, 166, 111–117. [Google Scholar] [CrossRef]
- Liu, K.; Zhang, W.; Yang, Y.; Zhang, J.; Li, Y.; Chen, Y. Respiratory rehabilitation in elderly patients with COVID-19: A randomized controlled study. Complement. Ther. Clin. Pract. 2020, 39, 101166. [Google Scholar] [CrossRef] [PubMed]
- Ayman, I.; Kinza, I.; Shajeea, A.A.; Azim, D.; Farid, E.; Baig, M.; Arif, T.B.; Raza, M. The COVID-19 Sequelae: A Cross-Sectional Evaluation of Post-recovery Symptoms and the Need for Rehabilitation of COVID-19 Survivors. Cureus 2021, 13, e13080. [Google Scholar]
- Mandal, S.; Barnett, J.; Brill, S.E.; Brown, J.S.; Denneny, E.K.; Hare, S.S.; Heightman, M.; Hillman, T.E.; Jacob, J.; Jarvis, H.C.; et al. ‘Long-COVID’: A cross-sectional study of persisting symptoms, biomarker and imaging abnormalities following hospitalisation for COVID-19. Thorax 2021, 76, 396–398. [Google Scholar] [CrossRef]
- Halpin, S.J.; McIvor, C.; Whyatt, G.; Adams, A.; Harvey, O.; McLean, L.; Walshaw, C.; Kemp, S.; Corrado, J.; Singh, R.; et al. Postdischarge symptoms and rehabilitation needs in survivors of COVID-19 infection: A cross-sectional evaluation. J. Med. Virol. 2021, 93, 1013–1022. [Google Scholar] [CrossRef]
- Goërtz, Y.M.; Van Herck, M.; Delbressine, J.M.; Vaes, A.W.; Meys, R.; Machado, F.V.C.; Houben-Wilke, S.; Burtin, C.; Posthuma, R.; Franssen, F.M.; et al. Persistent symptoms 3 months after a SARS-CoV-2 infection: The post-COVID-19 syndrome? ERJ Open Res. 2020, 6, 00542–02020. [Google Scholar] [CrossRef]
- Bartus, M.; Lomnicka, M.; Kostogrys, R.B.; Kazmierczak, P.; Watala, C.; Slominska, E.M.; Smolenski, R.T.; Pisulewski, P.M.; Adamus, J.; Gebicki, J.; et al. 1-Methylnicotinamide (MNA) prevents endothelial dysfunction in hypertriglyceridemic and diabetic rats. Pharmacol. Rep. PR 2008, 60, 127–138. [Google Scholar]
- Shi, Y.; Wang, Y.; Shao Ch Huang, J.; Gan, J.; Huang, X.; Bucci, E.; Piacentini, M.; Ippolito, G.; Melino, G. COVID-19 infection: The perspectives on immune responses. Cell Death Differ. 2020, 27, 1451–1454. [Google Scholar] [CrossRef]
- Przyborowski, K.; Wojewoda, M.; Sitek, B.; Zakrzewska, A.; Kij, A.; Wandzel, K.; Zoladz, J.A.; Chlopicki, S. Effects of 1-methylnicotinamide (MNA) on exercise capacity and endothelial response in diabetic mice. PLoS ONE 2015, 10, e0130908. [Google Scholar] [CrossRef]
- Chlopicki, S.; Kurdziel, M.; Sternak, M.; Szafarz, M.; Szymura-Oleksiak, J.; Kaminski, K.; Żołądź, J.A. Single bout of endurance exercise increases NNMT activity in the liver and MNA concentration in plasma. Pharmacol. Rep. 2012, 64, 369–376. [Google Scholar] [CrossRef]
- Canner, K.; Berge, K.G.; Wenger, N.K.; Stamler, J.; Friedman, L.; Prineas, R.J.; Friedewald, W. Fifteen year mortality in Coronary Drug Project patients: Long-term benefit with niacin. J. Am. Coll. Cardiol. 1986, 8, 1245–1255. [Google Scholar] [CrossRef]
- Brown, A.W.; Nathan, S.D. The Value and Application of the 6-Minute-Walk Test in Idiopathic Pulmonary Fibrosis. Ann. Am. Thorac. Soc. 2018, 15, 3–10. [Google Scholar] [CrossRef]
- Chirico, D.; Davidson, T.W.; Terada, T.; Scott, K.; Keast, M.-L.; Reid, R.D.; Pipe, A.L.; Reed, J.L. Using the 6-min Walk Test to Monitor Peak Oxygen Uptake Response to Cardiac Rehabilitation in Patients With Heart Failure. J. Cardiopulm. Rehabil. Prev. 2020, 40, 378–382. [Google Scholar] [CrossRef]
- Huang, C.; Huang, L.; Wang, Y.; Li, X.; Ren, L.; Gu, X.; Kang, L.; Guo, L.; Liu, M.; Zhou, X.; et al. 6-month consequences of COVID-19 in patients discharged from hospital: A cohort study. Lancet 2021, 397, 220–232. [Google Scholar] [CrossRef]
- Fuglebjerg, N.J.U.; Jensen, T.O.; Hoyer, N.; Ryrsø, C.K.; Lindegaard, B.; Harboe, Z.B. Silent hypoxia in patients with SARS CoV-2 infection before hospital discharge. Int. J. Infect. Dis. 2020, 99, 100–101. [Google Scholar] [CrossRef]
- Zampogna, E.; Paneroni, M.; Belli, S.; Aliani, M.; Gandolfo, A.; Visca, D.; Bellanti, M.T.; Ambrosino, N.; Vitacca, M. Pulmonary Rehabilitation in Patients Recovering from COVID-19. Respiration 2021, 100, 416–422. [Google Scholar] [CrossRef]
- Hermann, M.; Pekacka-Egli, A.-M.; Witassek, F.; Baumgaertner, R.; Schoendorf, S.; Spielmanns, M. Feasibility and Efficacy of Cardiopulmonary Rehabilitation After COVID-19. Am. J. Phys. Med. Rehabil. 2020, 99, 865–869. [Google Scholar] [CrossRef]
- Bellan, M.; Soddu, D.; Balbo, P.E.; Baricich, A.; Zeppegno, P.; Avanzi, G.C.; Baldon, G.; Bartolomei, G.; Battaglia, M.; Battisini, S.; et al. Respiratory and Psychophysical Sequelae Among Patients With COVID-19 Four Months After Hospital Discharge. JAMA Netw. Open. 2021, 4, e2036142. [Google Scholar] [CrossRef]
- Abodonya, A.M.; Abdelbasset, W.K.; Awad, A.E.; Elalfy, I.E.; Salem, H.A.; Elsayed, S.H. Inspiratory muscle training for recovered COVID-19 patients after weaning from mechanical ventilation: A pilot control clinical study. Medicine 2021, 100, e25339. [Google Scholar] [CrossRef]
- Curci, C.; Negrini, F.; Ferrillo, M.; Bergonzi, R.; Bonacci, E.; Camozzi, D.M.; Ceravolo, C.; Franceschi, S.D.E.; Guarnieri, R.; Moro, P.; et al. Functional outcome after inpatient rehabilitation in post-intensive care unit COVID-19 patients: Findings and clinical implications from a real-practice retrospective study. Eur. J. Phys. Rehabil. Med. 2020, 56, 633–641. [Google Scholar]
- Wong, A.; López-Romero, S.; Figueroa-Hurtado, E.; Vazquez-Lopez, S.; Milne, K.; Ryerson, C.; Guenette, J.; Cortés-Telles, A. Predictors of reduced 6-minute walk distance after COVID-19: A cohort study in Mexico. Pulmonology 2021, 27, 563–565. [Google Scholar] [CrossRef]
- Spielmanns, M.; Pekacka-Egli, A.-M.; Schoendorf, S.; Windisch, W.; Hermann, M. Effects of a Comprehensive Pulmonary Rehabilitation in Severe Post-COVID-19 Patients. Int. J. Environ. Res. Public Health. 2021, 18, 2695. [Google Scholar] [CrossRef]
- Tozato, C.; Fernandes Costa Ferreira, B.; Pereira Dalavina, J.; Vitelli Molinari, C.; Lúcia dos Santos Alves, V. Cardiopulmonary rehabilitation in post-COVID-19 patients: Case series. Rev. Bras. Ter. Intensiva. 2021, 33, 167–171. [Google Scholar]
- Kamal, M.; Abo Omirah, M.; Hussein, A.; Saeed, H. Assessment and characterisation of post-COVID-19 manifestations. Int. J. Clin. Pract. 2021, 75, e13746. [Google Scholar] [CrossRef]
- Arnold, D.T.; Hamilton, F.; Milne, A.; Morley, A.J.; Viner, J.; Attwood, M.; Noel, A.; Gunning, S.; Hatrick, J.; Hamilton, S.; et al. Patient outcomes after hospitalisation with COVID-19 and implications for follow-up: Results from a prospective UK cohort. Thorax 2021, 76, 399–401. [Google Scholar] [CrossRef]
- White, P.D.; Thomas, J.M.; Amess, J.; Crawford, D.H.; Grover, S.A.; Kangro, H.O.; Clare, A.W. Incidence, risk and prognosis of acute and chronic fatigue syndromes and psychiatric disorders after glandular fever. Br. J. Psychiatry. 1998, 173, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Salamanna, F.; Veronesi, F.; Martini, L.; Landini, M.P.; Milena, F. Post-COVID-19 Syndrome: The Persistent Symptoms at the Post-viral Stage of the Disease. A Systematic Review of the Current Data. Front. Med. (Lausanne) 2021, 4, 653516. [Google Scholar] [CrossRef]
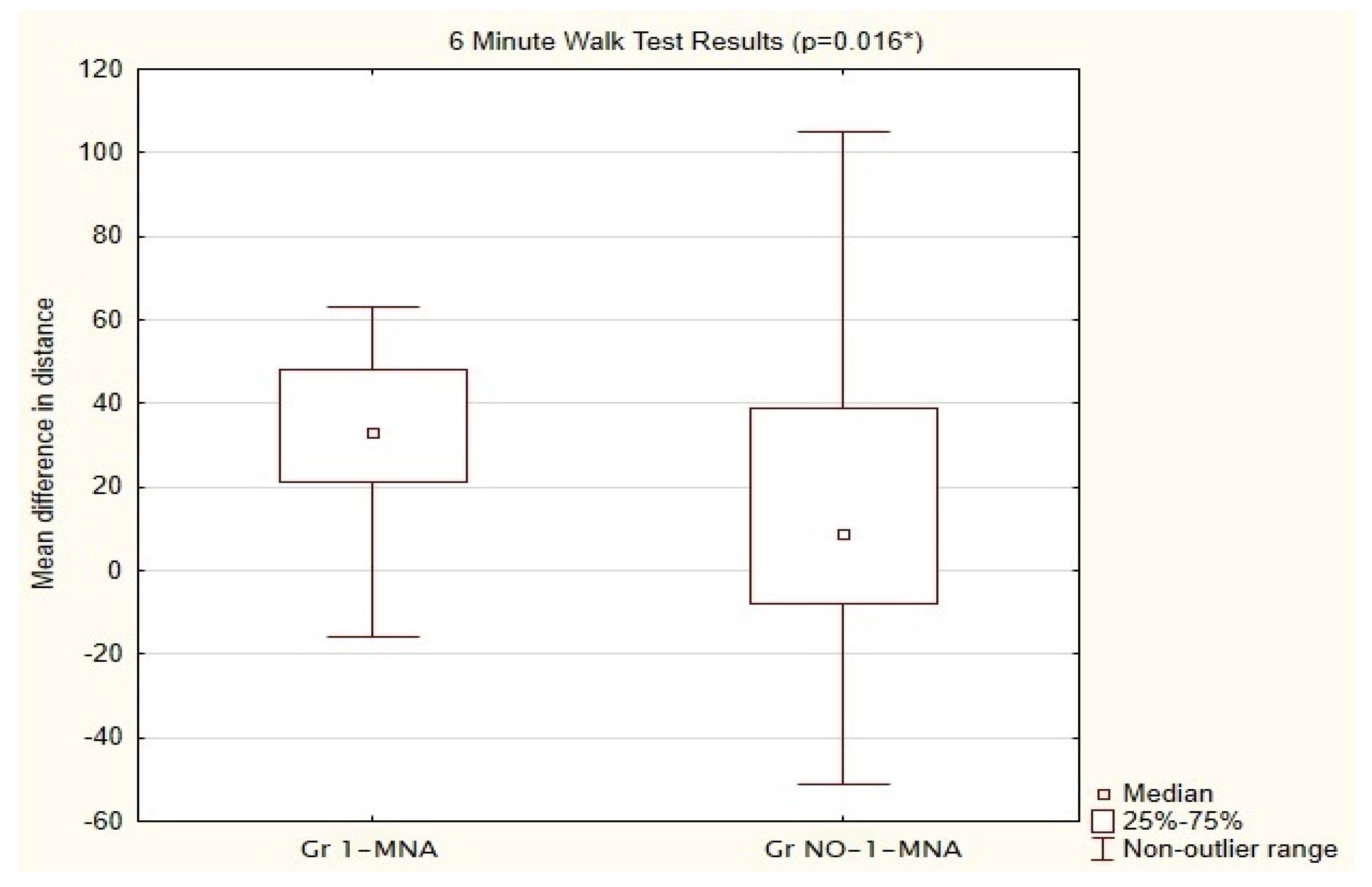
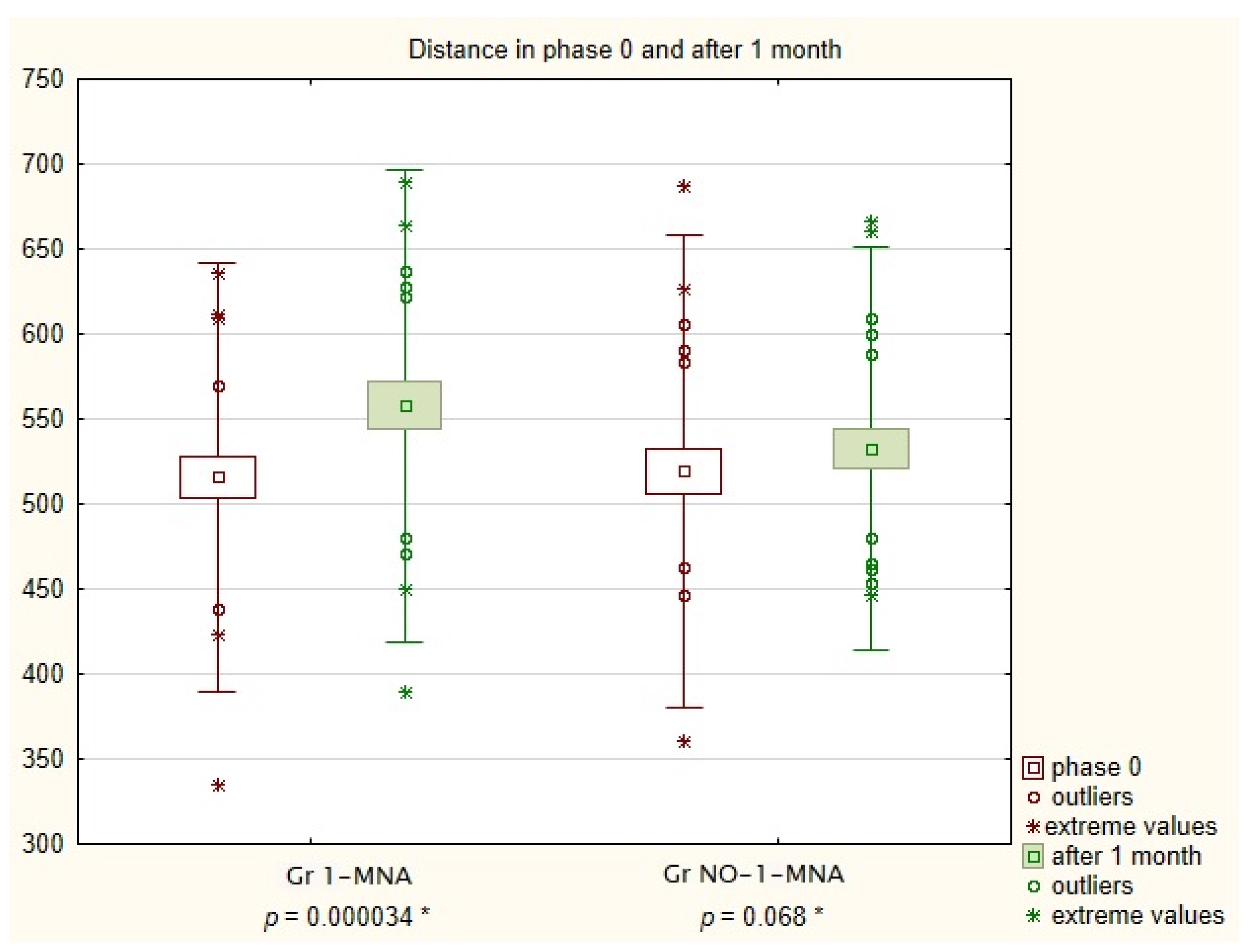
| Parameters | Gr 1-MNA n = 25 M ± SD 95% CI | Gr NO-1-MNA n = 25 M ± SD 95% CI | p-Value | |
|---|---|---|---|---|
| Age | 47.40 ± 9.73 43.38–51.42 | 49.60 ± 10.55 45.24–53.96 | 0.447 | |
| Median | 49 | 50 | ||
| Sex | Female | 17 (0.68) | 17 (0.68) | 1.000 |
| Male | 8 (0.32) | 8 (0.32) | ||
| BMI (kg/m2) | 26.88 ± 4.69 24.94–28.82 | 27.17 ± 4.92 25.14–29.20 | 0.834 | |
| Median | 25.97 | 26.42 | ||
| Heart rate at rest in Phase 0 | 72.40 ± 11.74 67.55–77.25 | 78.64 ± 1.32 78.10–79.18 | 0.352 | |
| Median | 76 | 79 | ||
| Oxygen saturation at rest in Phase 0 | 97.28 ± 2.03 96.44–98.12 | 96.96 ± 4.71 95.02–98.90 | 0.522 | |
| Median | 98 | 98 | ||
| Distance in 6 min walk test in Phase 0 | 515.8 ± 63 512.24–519.36 | 519.2 ± 69 490.72–547.68 | 0.850 | |
| Median | 516 | 504 | ||
| Fatigue assessment according to Fatigue Severity Score in Phase 0 | 4.28 ± 1.38 3.71–4.85 | 4.53 ± 1.16 4.05–5.01 | 0.490 | |
| Median | 4.44 | 4.54 | ||
| Dyspnoea on the Borg Scale at rest in Phase 0 | 0.128 | |||
| 0 | 8 (0.32) | 7 (0.28) | ||
| 1 | 3 (0.12) | 2 (0.08) | ||
| 2 | 9 (0.36) | 3 (0.12) | ||
| 3 | 2 (0.08) | 9 (0.36) | ||
| 4 | 2 (0.08) | 2 (0.08) | ||
| 5 | 1 (0.04) | 2 (0.08) | ||
| Parameters | Gr 1-MNA n = 25 | Gr NO-1-MNA n = 25 | Absolute Mean Difference Between Groups | p-Value |
|---|---|---|---|---|
| Mean distance in phase 0 | 515.80 | 519.24 | 3.44 | 0.000034 |
| Mean distance after 1 month | 557.80 | 532.52 | 25.28 | 0.068 |
| Mean difference between distances | 42.00 | 13.28 | 28.72 | 0.0016 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chudzik, M.; Burzyńska, M.; Kapusta, J. Use of 1-MNA to Improve Exercise Tolerance and Fatigue in Patients after COVID-19. Nutrients 2022, 14, 3004. https://doi.org/10.3390/nu14153004
Chudzik M, Burzyńska M, Kapusta J. Use of 1-MNA to Improve Exercise Tolerance and Fatigue in Patients after COVID-19. Nutrients. 2022; 14(15):3004. https://doi.org/10.3390/nu14153004
Chicago/Turabian StyleChudzik, Michał, Monika Burzyńska, and Joanna Kapusta. 2022. "Use of 1-MNA to Improve Exercise Tolerance and Fatigue in Patients after COVID-19" Nutrients 14, no. 15: 3004. https://doi.org/10.3390/nu14153004
APA StyleChudzik, M., Burzyńska, M., & Kapusta, J. (2022). Use of 1-MNA to Improve Exercise Tolerance and Fatigue in Patients after COVID-19. Nutrients, 14(15), 3004. https://doi.org/10.3390/nu14153004






