Development and Validation of Global Leadership Initiative on Malnutrition for Prognostic Prediction in Patients Who Underwent Cardiac Surgery
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Data Collection
2.3. Follow Up
2.4. Muscle Mass Measurements
2.5. Assessment of Nutritional Status
2.6. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. Relationship between GLIM-Defined Malnutrition and Short-Term Outcomes
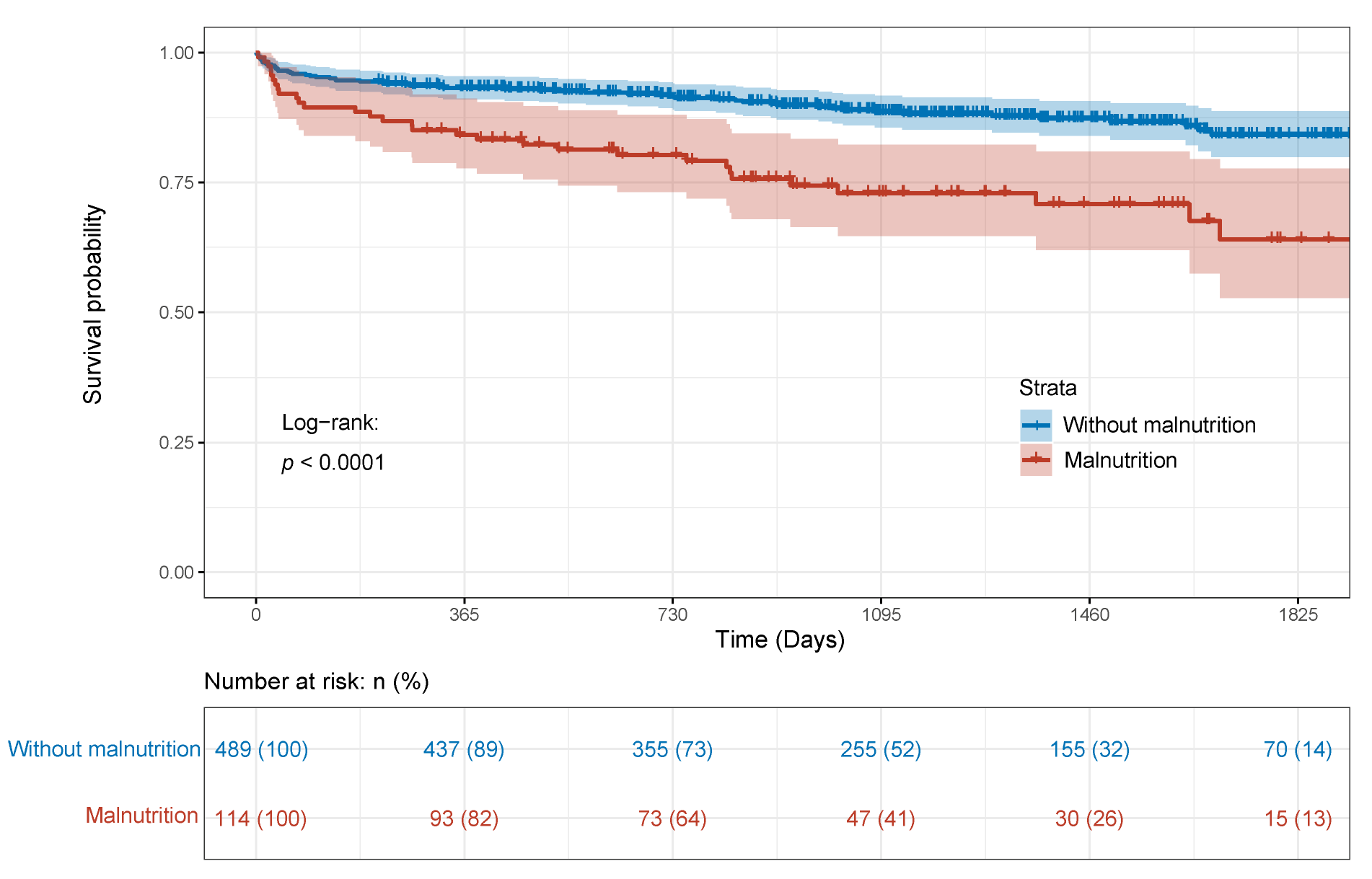
3.3. Relationship between GLIM-Defined Malnutrition and OS
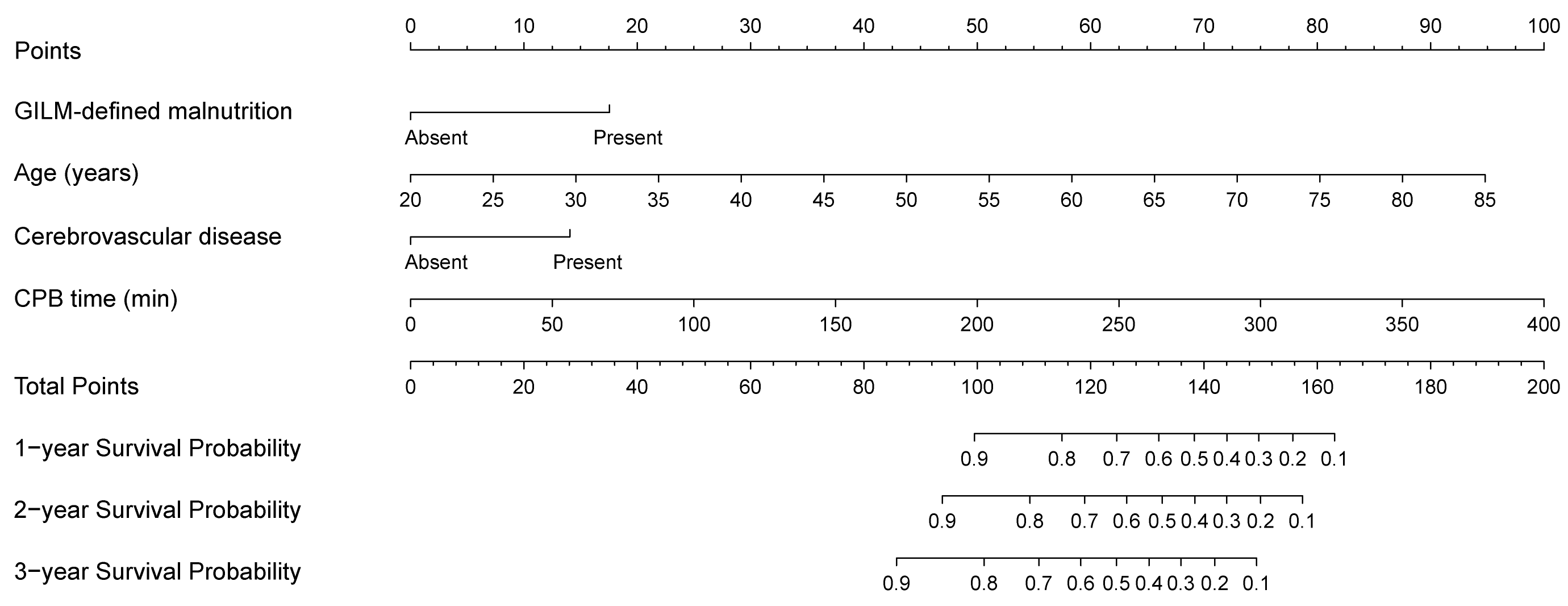
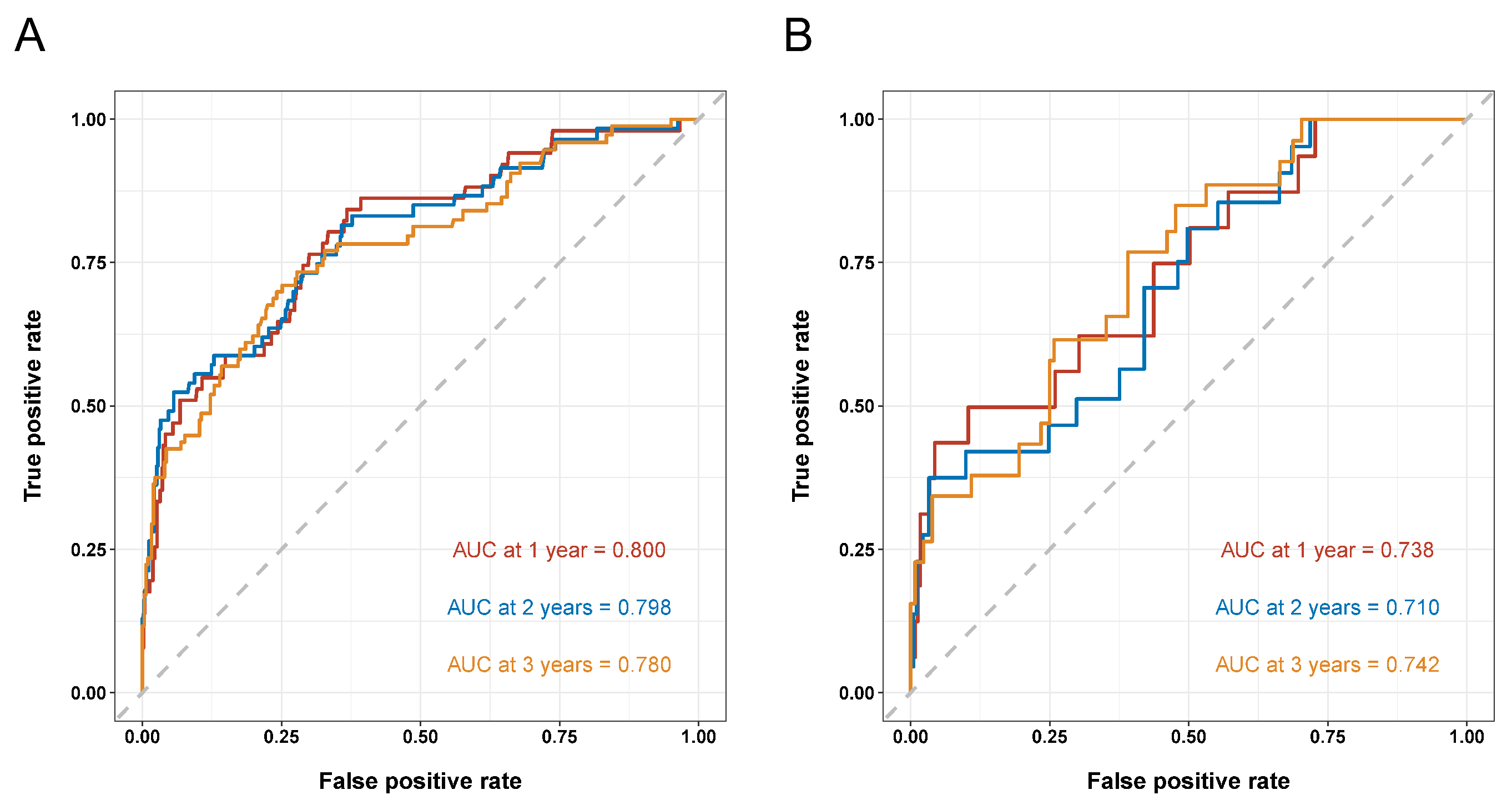
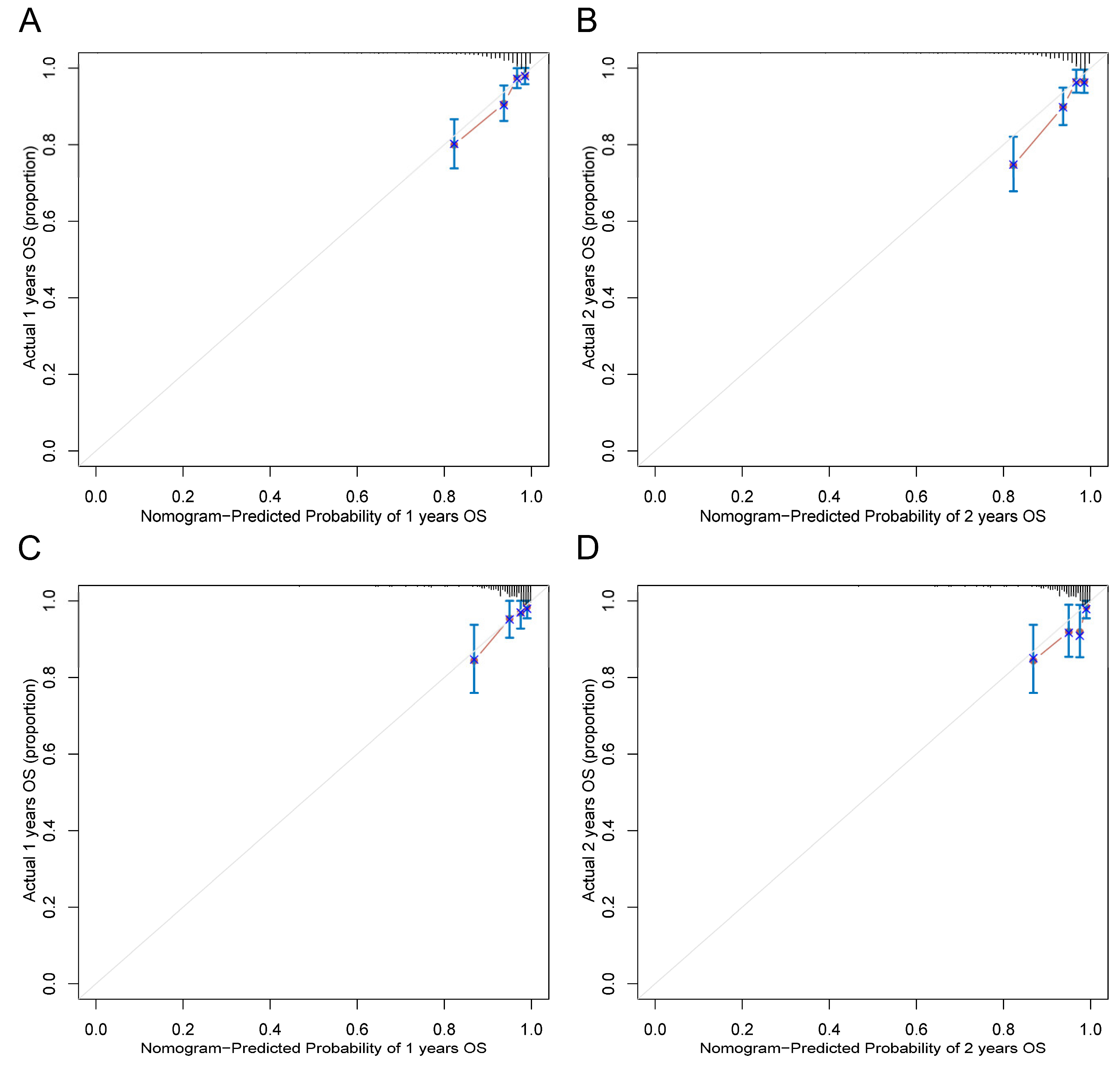
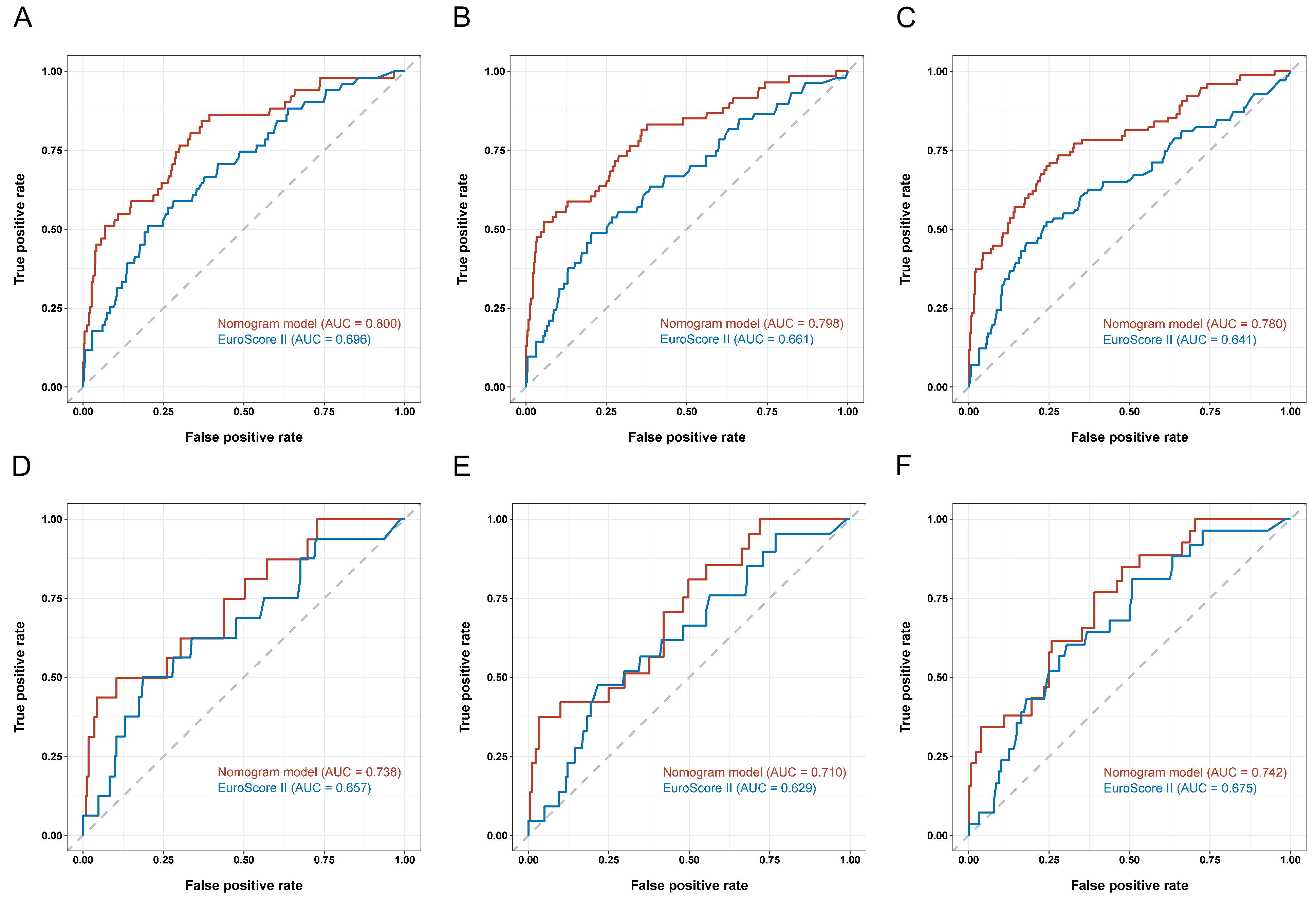
3.4. Development and Validation of a Prognostic Nomogram
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lomivorotov, V.V.; Efremov, S.M.; Boboshko, V.A.; Nikolaev, D.A.; Vedernikov, P.E.; Lomivorotov, V.N.; Karaskov, A.M. Evaluation of nutritional screening tools for patients scheduled for cardiac surgery. Nutrition 2013, 29, 436–442. [Google Scholar] [CrossRef] [PubMed]
- Goldfarb, M.; Lauck, S.; Webb, J.G.; Asgar, A.W.; Perrault, L.P.; Piazza, N.; Martucci, G.; Lachapelle, K.; Noiseux, N.; Kim, D.H.; et al. Malnutrition and Mortality in Frail and Non-Frail Older Adults Undergoing Aortic Valve Replacement. Circulation 2018, 138, 2202–2211. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.S.; Shim, J.K.; Kim, K.S.; Lee, S.; Kwak, Y.L. Impact of preoperative nutritional scores on 1-year postoperative mortality in patients undergoing valvular heart surgery. J. Thorac. Cardiovasc. Surg. 2021; in press. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.; Nesterova, E.; Lomivorotov, V.; Efremov, S.; Goetzenich, A.; Benstoem, C.; Zamyatin, M.; Chourdakis, M.; Heyland, D.; Stoppe, C. Current Evidence about Nutrition Support in Cardiac Surgery Patients-What Do We Know? Nutrients 2018, 10, 597. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nashef, S.A.; Roques, F.; Sharples, L.D.; Nilsson, J.; Smith, C.; Goldstone, A.R.; Lockowandt, U. EuroSCORE II. Eur. J. Cardiothorac. Surg. 2012, 41, 734–744; discussion 744–735. [Google Scholar] [CrossRef] [Green Version]
- Cederholm, T.; Jensen, G.L.; Correia, M.; Gonzalez, M.C.; Fukushima, R.; Higashiguchi, T.; Baptista, G.; Barazzoni, R.; Blaauw, R.; Coats, A.; et al. GLIM criteria for the diagnosis of malnutrition—A consensus report from the global clinical nutrition community. Clin. Nutr. 2019, 38, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Oguri, M.; Ishii, H.; Yasuda, K.; Sumi, T.; Takahashi, H.; Murohara, T. Combined prognostic value of malnutrition using GLIM criteria and renal insufficiency in elderly heart failure. ESC Heart Fail. 2022, 9, 704–711. [Google Scholar] [CrossRef]
- Huang, D.D.; Yu, D.Y.; Wang, W.B.; Song, H.N.; Luo, X.; Wu, G.F.; Chen, X.L.; Yu, Z.; Yan, J.Y. Global leadership initiative in malnutrition (GLIM) criteria using hand-grip strength adequately predicts postoperative complications and long-term survival in patients underwent radical gastrectomy for gastric cancer. Eur. J. Clin. Nutr. 2022; in press. [Google Scholar] [CrossRef]
- Przekop, Z.; Szostak-Wegierek, D.; Milewska, M.; Panczyk, M.; Zaczek, Z.; Sobocki, J. Efficacy of the Nutritional Risk Index, Geriatric Nutritional Risk Index, BMI, and GLIM-Defined Malnutrition in Predicting Survival of Patients with Head and Neck Cancer Patients Qualified for Home Enteral Nutrition. Nutrients 2022, 14, 1268. [Google Scholar] [CrossRef]
- Yin, L.; Lin, X.; Li, N.; Zhang, M.; He, X.; Liu, J.; Kang, J.; Chen, X.; Wang, C.; Wang, X.; et al. Evaluation of the Global Leadership Initiative on Malnutrition Criteria Using Different Muscle Mass Indices for Diagnosing Malnutrition and Predicting Survival in Lung Cancer Patients. JPEN J. Parenter Enter. Nutr. 2021, 45, 607–617. [Google Scholar] [CrossRef]
- Zhang, X.; Tang, M.; Zhang, Q.; Zhang, K.P.; Guo, Z.Q.; Xu, H.X.; Yuan, K.T.; Yu, M.; Braga, M.; Cederholm, T.; et al. The GLIM criteria as an effective tool for nutrition assessment and survival prediction in older adult cancer patients. Clin. Nutr. 2021, 40, 1224–1232. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wan, Z.; Zhu, Y.; Wan, H. Predictive validity of the GLIM criteria in treatment outcomes in cancer patients with radiotherapy. Clin. Nutr. 2022, 41, 855–861. [Google Scholar] [CrossRef] [PubMed]
- Derstine, B.A.; Holcombe, S.A.; Ross, B.E.; Wang, N.C.; Su, G.L.; Wang, S.C. Skeletal muscle cutoff values for sarcopenia diagnosis using T10 to L5 measurements in a healthy US population. Sci. Rep. 2018, 8, 11369. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shen, Z.L.; Liu, Z.; Zang, W.F.; Zhang, P.; Zou, H.B.; Dong, W.X.; Chen, W.H.; Yan, X.L.; Yu, Z. Thoracic sarcopenia predicts clinical outcomes in patients underwent coronary artery bypass grafting: A 6-year cohort study. Asian J. Surg. 2022; in press. [Google Scholar] [CrossRef] [PubMed]
- Weber, C.; Noels, H. Atherosclerosis: Current pathogenesis and therapeutic options. Nat. Med. 2011, 17, 1410–1422. [Google Scholar] [CrossRef]
- Schoen, F.J. Mechanisms of function and disease of natural and replacement heart valves. Annu. Rev. Pathol. 2012, 7, 161–183. [Google Scholar] [CrossRef]
- Gillis, C.; Wischmeyer, P.E. Pre-operative nutrition and the elective surgical patient: Why, how and what? Anaesthesia 2019, 74 (Suppl. S1), 27–35. [Google Scholar] [CrossRef] [Green Version]
- Weimann, A.; Braga, M.; Carli, F.; Higashiguchi, T.; Hubner, M.; Klek, S.; Laviano, A.; Ljungqvist, O.; Lobo, D.N.; Martindale, R.; et al. ESPEN guideline: Clinical nutrition in surgery. Clin. Nutr. 2017, 36, 623–650. [Google Scholar] [CrossRef] [Green Version]
- Wolfe, R.R. The underappreciated role of muscle in health and disease. Am. J. Clin. Nutr. 2006, 84, 475–482. [Google Scholar] [CrossRef]
- Wischmeyer, P.E.; Carli, F.; Evans, D.C.; Guilbert, S.; Kozar, R.; Pryor, A.; Thiele, R.H.; Everett, S.; Grocott, M.; Gan, T.J.; et al. American Society for Enhanced Recovery and Perioperative Quality Initiative Joint Consensus Statement on Nutrition Screening and Therapy Within a Surgical Enhanced Recovery Pathway. Anesth. Analg. 2018, 126, 1883–1895. [Google Scholar] [CrossRef]
- Stoppe, C.; Goetzenich, A.; Whitman, G.; Ohkuma, R.; Brown, T.; Hatzakorzian, R.; Kristof, A.; Meybohm, P.; Mechanick, J.; Evans, A.; et al. Role of nutrition support in adult cardiac surgery: A consensus statement from an International Multidisciplinary Expert Group on Nutrition in Cardiac Surgery. Crit. Care 2017, 21, 131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singer, P.; Blaser, A.R.; Berger, M.M.; Alhazzani, W.; Calder, P.C.; Casaer, M.P.; Hiesmayr, M.; Mayer, K.; Montejo, J.C.; Pichard, C.; et al. ESPEN guideline on clinical nutrition in the intensive care unit. Clin. Nutr. 2019, 38, 48–79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hill, A.; Arora, R.C.; Engelman, D.T.; Stoppe, C. Preoperative Treatment of Malnutrition and Sarcopenia in Cardiac Surgery: New Frontiers. Crit Care Clin. 2020, 36, 593–616. [Google Scholar] [CrossRef] [PubMed]
- Pisano, C.; Polisano, D.; Balistreri, C.R.; Altieri, C.; Nardi, P.; Bertoldo, F.; Trombetti, D.; Asta, L.; Ferrante, M.S.; Buioni, D.; et al. Role of Cachexia and Fragility in the Patient Candidate for Cardiac Surgery. Nutrients 2021, 13, 517. [Google Scholar] [CrossRef] [PubMed]
- Unosawa, S.; Taoka, M.; Osaka, S.; Yuji, D.; Kitazumi, Y.; Suzuki, K.; Kamata, K.; Sezai, A.; Tanaka, M. Is malnutrition associated with postoperative complications after cardiac surgery? J. Card Surg. 2019, 34, 908–912. [Google Scholar] [CrossRef] [PubMed]
- Chermesh, I.; Hajos, J.; Mashiach, T.; Bozhko, M.; Shani, L.; Nir, R.R.; Bolotin, G. Malnutrition in cardiac surgery: Food for thought. Eur. J. Prev. Cardiol. 2014, 21, 475–483. [Google Scholar] [CrossRef]
- Lee, K.; Ahn, J.M.; Kang, D.Y.; Ko, E.; Kwon, O.; Lee, P.H.; Lee, S.W.; Kim, D.H.; Kim, H.J.; Kim, J.B.; et al. Nutritional status and risk of all-cause mortality in patients undergoing transcatheter aortic valve replacement assessment using the geriatric nutritional risk index and the controlling nutritional status score. Clin. Res. Cardiol. 2020, 109, 161–171. [Google Scholar] [CrossRef]
- Chen, C.C.; Chen, T.H.; Tu, P.H.; Wu, V.C.; Yang, C.H.; Wang, A.Y.; Lee, S.T.; Tsai, F.C.; Chen, S.W. Long-Term Outcomes for Patients With Stroke After Coronary and Valve Surgery. Ann. Thorac. Surg. 2018, 106, 85–91. [Google Scholar] [CrossRef] [Green Version]
- Onorati, F.; De Feo, M.; Mastroroberto, P.; Cristodoro, L.; Pezzo, F.; Renzulli, A.; Cotrufo, M. Determinants and prognosis of myocardial damage after coronary artery bypass grafting. Ann. Thorac. Surg. 2005, 79, 837–845. [Google Scholar] [CrossRef]
- Karim, M.N.; Reid, C.M.; Huq, M.; Brilleman, S.L.; Cochrane, A.; Tran, L.; Billah, B. Predicting long-term survival after coronary artery bypass graft surgery. Interact. Cardiovasc. Thorac. Surg. 2018, 26, 257–263. [Google Scholar] [CrossRef] [Green Version]
- Efremov, S.M.; Ionova, T.I.; Nikitina, T.P.; Vedernikov, P.E.; Dzhumatov, T.A.; Ovchinnikov, T.S.; Rashidov, A.A.; Stoppe, C.; Heyland, D.K.; Lomivorotov, V.V. Effects of malnutrition on long-term survival in adult patients after elective cardiac surgery. Nutrition 2021, 83, 111057. [Google Scholar] [CrossRef] [PubMed]
- Madhavan, S.; Chan, S.P.; Tan, W.C.; Eng, J.; Li, B.; Luo, H.D.; Teoh, L.K. Cardiopulmonary bypass time: Every minute counts. J. Cardiovasc. Surg. 2018, 59, 274–281. [Google Scholar] [CrossRef]
- Thourani, V.H.; Keeling, W.B.; Kilgo, P.D.; Puskas, J.D.; Lattouf, O.M.; Chen, E.P.; Guyton, R.A. The impact of body mass index on morbidity and short-and long-term mortality in cardiac valvular surgery. J. Thorac. Cardiovasc. Surg. 2011, 142, 1052–1061. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhamidipati, C.M.; LaPar, D.J.; Mehta, G.S.; Kern, J.A.; Upchurch, G.R., Jr.; Kron, I.L.; Ailawadi, G. Albumin is a better predictor of outcomes than body mass index following coronary artery bypass grafting. Surgery 2011, 150, 626–634. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, F.; Shan, L.; Wang, C.; Meng, X.; Chen, J.; Han, L.; Zhang, Y.; Li, Z. Predictive Ability of European Heart Surgery Risk Assessment System II (EuroSCORE II) and the Society of Thoracic Surgeons (STS) Score for in-Hospital and Medium-Term Mortality of Patients Undergoing Coronary Artery Bypass Grafting. Int. J. Gen. Med. 2021, 14, 8509–8519. [Google Scholar] [CrossRef]
- Barili, F.; Pacini, D.; D’Ovidio, M.; Dang, N.C.; Alamanni, F.; Di Bartolomeo, R.; Grossi, C.; Davoli, M.; Fusco, D.; Parolari, A. The Impact of EuroSCORE II Risk Factors on Prediction of Long-Term Mortality. Ann. Thorac. Surg. 2016, 102, 1296–1303. [Google Scholar] [CrossRef] [Green Version]
| Malnutrition, n = 162 (18.8%) | Phenotypic Criteria | |||
|---|---|---|---|---|
| Weight Loss, n = 74 (8.6%) | Low BMI, n = 29 (3.4%) | Reduced Muscle Mass, n = 135 (15.7%) | ||
| Training cohort | 114(18.9%) | 46(7.6%) | 18(3.0%) | 98(16.3%) |
| Validation cohort | 48 (18.6%) | 28 (10.9%) | 11 (4.3%) | 37 (14.3%) |
| Training Cohort (n = 603) | Validation Cohort (n = 258) | |||||
|---|---|---|---|---|---|---|
| GLIM Criteria | GLIM Criteria | |||||
| Without Malnutrition (n = 489) | Malnutrition (n = 114) | p Value | Without Malnutrition (n = 210) | Malnutrition (n = 48) | p Value | |
| Age, years | 63 (11) | 65 (11) | 0.122 | 63 (12) | 68 (16) | 0.020 * |
| Sex, male | 319 (65.2%) | 90 (78.9%) | 0.005 * | 130 (61.9%) | 30 (62.5%) | 0.939 |
| BMI, kg/m2 | 24.74 (3.66) | 21.09 (3.82) | <0.001 * | 24.99 (4.24) | 22.15 (5.12) | <0.001 * |
| Tobacco use | 179 (36.6%) | 41 (36.0%) | 0.898 | 62 (29.5%) | 13 (27.1%) | 0.737 |
| Alcohol use | 67 (13.7%) | 15 (13.2%) | 0.879 | 34 (16.2%) | 5 (10.4%) | 0.314 |
| LVEF, % | 60 (8) | 60 (9) | 0.880 | 60 (9) | 62 (10) | 0.636 |
| NYHA class 3 or 4 | 426 (87.1%) | 100 (87.7%) | 0.862 | 184 (87.6%) | 44 (91.7%) | 0.430 |
| CCI | 3 (2) | 3 (2) | 0.254 | 3 (2) | 3 (2) | 0.208 |
| EuroSCORE II | 1.64 (1.09) | 1.83 (1.50) | 0.097 | 1.65 (0.97) | 1.81 (1.27) | 0.065 |
| Comorbidities (%) | ||||||
| Hypertension | 338 (69.1%) | 67 (58.8%) | 0.034 * | 136 (64.8%) | 30 (62.5%) | 0.768 |
| Diabetes | 154 (31.5%) | 33 (28.9%) | 0.597 | 72 (34.3%) | 8 (16.7%) | 0.017 * |
| Chronic heart failure | 39 (8.0%) | 10 (8.8%) | 0.779 | 18 (8.6%) | 6 (12.5%) | 0.569 |
| Atrial fibrillation | 97 (19.8%) | 20 (17.5%) | 0.577 | 38 (18.1%) | 8 (16.7%) | 0.816 |
| Previous myocardial infarction | 21 (4.3%) | 6 (5.3%) | 0.652 | 9 (4.3%) | 2 (4.2%) | 1.000 |
| COPD | 15 (3.1%) | 5 (4.4%) | 0.676 | 4 (1.9%) | 2 (4.2%) | 0.684 |
| Recent pneumonia | 25 (5.1%) | 4 (3.5%) | 0.471 | 9 (4.3%) | 3 (6.3%) | 0.839 |
| Cerebrovascular disease | 61 (12.5%) | 16 (14.0%) | 0.653 | 35 (16.7%) | 9 (18.8%) | 0.729 |
| Laboratory data | ||||||
| C-reactive protein, mg/L | 3.17 (1.44) | 3.23 (5.26) | 0.020 * | 3.17 (1.83) | 3.3 (14.66) | 0.020 * |
| White blood cells, ×109/L | 6.36 (2.48) | 6.43 (2.66) | 0.927 | 6.31 (2.66) | 5.67 (3.03) | 0.389 |
| Red blood cells, ×1012/L | 4.37 ± 0.54 | 4.17 ± 0.58 | <0.001 * | 4.39 ± 0.56 | 4.17 ± 0.47 | 0.013 * |
| Hemoglobin, g/L | 131 (23) | 127 (24) | 0.023 * | 132 (23) | 125.5 (16.75) | 0.037 * |
| Platelets, ×109/L | 203 (80.5) | 199.5 (82.5) | 0.939 | 204.5 (82.75) | 202.0 (102.5) | 0.626 |
| Neutrophil percentage, % | 61.77 ± 9.74 | 64.41 ± 10.68 | 0.011 * | 62.02 ± 9.62 | 64.64 ± 11.75 | 0.155 |
| Lymphocytes, ×109/L | 1.73 (0.77) | 1.58 (0.69) | 0.004 * | 1.71 (0.80) | 1.49 (0.73) | 0.003 * |
| NLR | 2.23 (1.46) | 2.44 (2.30) | 0.021 * | 2.26 (1.46) | 2.76 (3.34) | 0.064 |
| PLR | 113.77 (58.62) | 131.12 (81.16) | 0.010 * | 118.47 (62.81) | 143.38 (77.83) | 0.025 * |
| Total protein, g/L | 68.76 (7.35) | 68.14 (7.03) | 0.879 | 69.00 (7.05) | 68.14 (10.13) | 0.360 |
| Albumin, g/L | 41.1 (5.0) | 40.25 (5.0) | 0.047 * | 41.0 (5.0) | 41.0 (6.75) | 0.058 |
| BUN, μmol/L | 5.9 (2.5) | 5.9 (3.0) | 0.351 | 6.1 (2.6) | 5.6(2.5) | 0.144 |
| Creatinine, μmol/L | 76.0 (25.0) | 74.1 (28.2) | 0.396 | 76.0 (26.3) | 74.8 (28.2) | 0.662 |
| Training Cohort (n = 603) | Validation Cohort (n = 258) | |||||
|---|---|---|---|---|---|---|
| GLIM Criteria | GLIM Criteria | |||||
| Without Malnutrition (n = 489) | Malnutrition (n = 114) | p Value | Without Malnutrition (n = 210) | Malnutrition (n = 48) | p Value | |
| Surgical Type | 0.760 | 0.887 | ||||
| Isolated CABG | 277 (56.6%) | 65 (57.0%) | 110 (52.4%) | 27 (56.3%) | ||
| Isolated valve surgery | 177 (36.2%) | 43 (37.7%) | 91 (43.3%) | 19 (39.6%) | ||
| CABG + valve surgery | 35 (7.2%) | 6 (5.3%) | 9 (4.3%) | 2 (4.2%) | ||
| Operative time, min | 215 (61) | 211 (57) | 0.335 | 213 (56) | 214 (60) | 0.790 |
| CPB time, min | 81 (54) | 80.5 (52) | 0.857 | 82 (41) | 76 (52) | 0.853 |
| Aortic cross-clamp time, min | 59 (31) | 61 (25) | 0.917 | 54.5 (28) | 60 (35) | 0.476 |
| Type of involved valves | 0.710 | 0.712 | ||||
| aortic valve | 56 (26.4%) | 15 (30.6%) | 25 (25.0%) | 3 (14.3%) | ||
| mitral valve | 55 (25.9%) | 14 (28.6%) | 21 (21.0%) | 3 (14.3%) | ||
| tricuspid | 12 (5.7%) | 5 (10.2%) | 10 (10.0%) | 4 (19.0%) | ||
| aortic valve + mitral valve | 15 (7.1%) | 3 (6.1%) | 9 (9.0%) | 2 (9.5%) | ||
| aortic valve + tricuspid | 5 (2.4%) | 1 (2.0%) | 0 (0.0%) | 0 (0.0%) | ||
| mitral valve + tricuspid | 54 (25.5%) | 10 (20.4%) | 29 (29.0%) | 7 (33.3%) | ||
| aortic valve + mitral valve + tricuspid | 15 (7.1%) | 1 (2.0%) | 6 (6.0%) | 2 (9.5%) | ||
| CABG details | ||||||
| CABG type: on pump | 202 (64.7%) | 52 (73.2%) | 0.172 | 87 (73.1%) | 21 (72.4%) | 0.940 |
| Use of LIMA | 160 (51.3%) | 24 (33.8%) | 0.008 * | 63 (52.9%) | 9 (31.0%) | 0.034 * |
| Number of bypassed vessels | 0.377 | 0.320 | ||||
| 1 | 33 (10.6%) | 4 (5.6%) | 7 (5.9%) | 0 (0.0%) | ||
| 2 | 39 (12.5%) | 7 (9.9%) | 8 (6.7%) | 4 (13.8%) | ||
| 3 | 87 (27.9%) | 18 (25.4%) | 43 (36.1%) | 12 (41.4%) | ||
| 4 or more | 153 (49.0%) | 42 (59.2%) | 61 (51.3%) | 13 (44.8%) | ||
| All Patients (n = 603) | GLIM Criteria | p Value | ||
|---|---|---|---|---|
| Without Malnutrition (n = 489) | Malnutrition (n = 114) | |||
| Postoperative hospital stay, day | 10 (5) | 10 (4) | 10 (7) | 0.126 |
| Prolonged intensive care stay (>5 d) | 92 (15.3%) | 67 (13.7%) | 25 (21.9%) | 0.028 * |
| Indwelling drainage tube time, day | 3 (1) | 3 (1) | 3 (1) | 0.503 |
| Cost, CNY | 130,926 (44,393) | 130,661 (42,484) | 131,751 (56,718) | 0.084 |
| 30 days readmission | 35 (5.8%) | 26 (5.3%) | 9 (7.9%) | 0.289 |
| Total Complications | 268 (44.4%) | 207 (42.3%) | 61 (53.5%) | 0.031 * |
| Pneumonia | 18 (3.0%) | 13 (2.7%) | 5 (4.4%) | |
| Delirium | 16 (2.7%) | 12 (2.5%) | 4 (3.5%) | |
| Poor wound healing (no debridement) | 15 (2.5%) | 13 (2.7%) | 2 (1.8%) | |
| Poor wound healing need debridement | 15 (2.5%) | 11 (2.2%) | 4 (3.5%) | |
| Pleural effusion | 93 (15.4%) | 74 (15.1%) | 19 (16.7%) | |
| Reoperation | 6 (1.0%) | 5 (1.0%) | 1 (0.9%) | |
| Stroke | 4 (0.7%) | 3 (0.6%) | 1 (0.9%) | |
| Low cardiac output syndrome | 22 (3.6%) | 16 (3.3%) | 6 (5.3%) | |
| Respiratory failure | 43 (7.1%) | 35 (7.2%) | 8 (7.0%) | |
| MODS | 6 (1.0%) | 5 (1.0%) | 1 (0.9%) | |
| In-hospital mortality | 30 (5.0%) | 20 (4.1%) | 10 (8.8%) | |
| Factors | Univariate Analysis | Multivariate Analysis | ||||
|---|---|---|---|---|---|---|
| OR | 95% CI | p Value | OR | 95% CI | p Value | |
| GLIM-defined malnutrition | 1.568 | 1.041–2.361 | 0.031 * | 1.661 | 1.063–2.594 | 0.026 * |
| Age | 1.043 | 1.024–1.063 | <0.001 * | 1.044 | 1.024–1.065 | <0.001 * |
| Sex (male) | 0.741 | 0.526–1.044 | 0.087 | 0.587 | 0.402–0.858 | 0.006 * |
| BMI < 18.5 kg/m2 | 2.031 | 0.657–6.281 | 0.219 | |||
| Tobacco use | 0.978 | 0.700–1.365 | 0.895 | |||
| Alcohol use | 1.450 | 0.909–2.313 | 0.118 | |||
| LVEF ≤ 50% | 2.332 | 1.506–3.612 | <0.001 * | 2.197 | 1.359–3.552 | 0.001 * |
| NYHA class 3 or 4 | 1.378 | 0.843–2.254 | 0.201 | |||
| CCI ≥ 2 | 2.901 | 1.788–4.707 | <0.001 * | |||
| EuroSCORE II ≥ 4% | 5.043 | 2.453–10.367 | <0.001 * | 2.642 | 1.231–5.670 | 0.013 * |
| Hypertension | 1.541 | 1.088–2.181 | 0.015 * | |||
| Diabetes | 1.450 | 1.025–2.051 | 0.036 * | |||
| Chronic heart failure | 2.536 | 1.376–4.677 | 0.003 * | |||
| Atrial fibrillation | 1.187 | 0.792–1.778 | 0.407 | |||
| Previous myocardial infarction | 2.201 | 0.991–4.890 | 0.053 | |||
| COPD | 1.024 | 0.418–2.507 | 0.959 | |||
| Recent pneumonia | 1.359 | 0.644–2.869 | 0.420 | |||
| Cerebrovascular disease | 1.798 | 1.109–2.915 | 0.017 * | 1.711 | 1.027–2.851 | 0.039 * |
| Hypoproteinemia | 1.651 | 0.805–3.384 | 0.171 | |||
| Surgical Type | 0.005 * | |||||
| Isolated CABG | 1.000 | Reference | ||||
| Isolated valve surgery | 0.647 | 0.458–0.916 | 0.014 * | |||
| CABG + valve surgery | 1.736 | 0.895–3.367 | 0.103 | |||
| Operative time, min | 1.008 | 1.005–1.012 | <0.001 * | 1.008 | 1.005–1.012 | <0.001 * |
| CPB time, min | 1.003 | 1.000–1.006 | 0.080 | |||
| C-reactive protein > 10 mg/L | 1.582 | 0.988–2.532 | 0.056 | |||
| Hemoglobin, g/L | 0.986 | 0.977–0.995 | 0.003 * | |||
| NLR ≥ 3.5 | 1.281 | 0.870–1.886 | 0.210 | |||
| PLR ≥ 133 | 1.207 | 0.864–1.687 | 0.270 | |||
| Factors | Univariate Analysis | Multivariate Analysis | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p Value | HR | 95% CI | p Value | |
| GLIM-defined malnutrition | 2.602 | 1.687–4.014 | <0.001 * | 2.339 | 1.504–3.637 | <0.001 * |
| Age | 1.077 | 1.049–1.105 | <0.001 * | 1.073 | 1.046–1.101 | <0.001 * |
| Sex (male) | 1.216 | 0.770–1.921 | 0.401 | |||
| BMI < 18.5 kg/m2 | 1.037 | 0.255–4.216 | 0.960 | |||
| Tobacco use | 0.987 | 0.642–1.517 | 0.951 | |||
| Alcohol use | 1.273 | 0.730–2.220 | 0.395 | |||
| LVEF ≤ 50% | 1.378 | 0.820–2.314 | 0.226 | |||
| NYHA class 3 or 4 | 1.444 | 0.723–2.885 | 0.298 | |||
| CCI ≥ 2 | 1.709 | 0.885–3.299 | 0.110 | |||
| EuroSCORE II ≥ 4% | 2.679 | 1.513–4.745 | 0.001 * | |||
| Hypertension | 0.853 | 0.553–1.315 | 0.470 | |||
| Diabetes | 1.405 | 0.916–2.155 | 0.119 | |||
| Chronic heart failure | 1.795 | 0.928–3.473 | 0.082 | |||
| Atrial fibrillation | 1.464 | 0.911–2.354 | 0.116 | |||
| Previous myocardial infarction | 1.064 | 0.390–2.903 | 0.903 | |||
| COPD | 0.296 | 0.041–2.126 | 0.226 | |||
| Recent pneumonia | 0.452 | 0.111–1.836 | 0.267 | |||
| Cerebrovascular disease | 1.906 | 1.148–3.166 | 0.013 * | 1.980 | 1.188–3.298 | 0.009 * |
| Hypoproteinemia | 1.493 | 0.651–3.420 | 0.344 | |||
| Surgical Type | 0.382 | |||||
| Isolated CABG | 1.000 | Reference | ||||
| Isolated valve surgery | 1.357 | 0.881–2.090 | 0.166 | |||
| CABG + valve surgery | 1.112 | 0.473–2.612 | 0.808 | |||
| Operative time, min | 1.006 | 1.003–1.009 | <0.001 * | |||
| CPB time, min | 1.013 | 1.009–1.017 | <0.001 * | 1.012 | 1.009–1.015 | <0.001 * |
| C-reactive protein > 10 mg/L | 1.172 | 0.650–2.112 | 0.598 | |||
| Hemoglobin, g/L | 0.979 | 0.967–0.991 | <0.001 * | |||
| NLR ≥ 3.5 | 1.154 | 0.707–1.883 | 0.567 | |||
| PLR ≥ 133 | 0.889 | 0.570–1.384 | 0.602 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Z.; Shen, Z.; Zang, W.; Zhou, J.; Yu, Z.; Zhang, P.; Yan, X. Development and Validation of Global Leadership Initiative on Malnutrition for Prognostic Prediction in Patients Who Underwent Cardiac Surgery. Nutrients 2022, 14, 2409. https://doi.org/10.3390/nu14122409
Liu Z, Shen Z, Zang W, Zhou J, Yu Z, Zhang P, Yan X. Development and Validation of Global Leadership Initiative on Malnutrition for Prognostic Prediction in Patients Who Underwent Cardiac Surgery. Nutrients. 2022; 14(12):2409. https://doi.org/10.3390/nu14122409
Chicago/Turabian StyleLiu, Zhang, Zile Shen, Wangfu Zang, Jian Zhou, Zhen Yu, Peng Zhang, and Xialin Yan. 2022. "Development and Validation of Global Leadership Initiative on Malnutrition for Prognostic Prediction in Patients Who Underwent Cardiac Surgery" Nutrients 14, no. 12: 2409. https://doi.org/10.3390/nu14122409
APA StyleLiu, Z., Shen, Z., Zang, W., Zhou, J., Yu, Z., Zhang, P., & Yan, X. (2022). Development and Validation of Global Leadership Initiative on Malnutrition for Prognostic Prediction in Patients Who Underwent Cardiac Surgery. Nutrients, 14(12), 2409. https://doi.org/10.3390/nu14122409






