Habitual Total Drinking Fluid Intake Did Not Affect Plasma Hydration Biomarkers among Young Male Athletes in Beijing, China: A Cross-Sectional Study
Abstract
:1. Background
2. Methods
2.1. Subjects
2.2. Sample Size Calculation
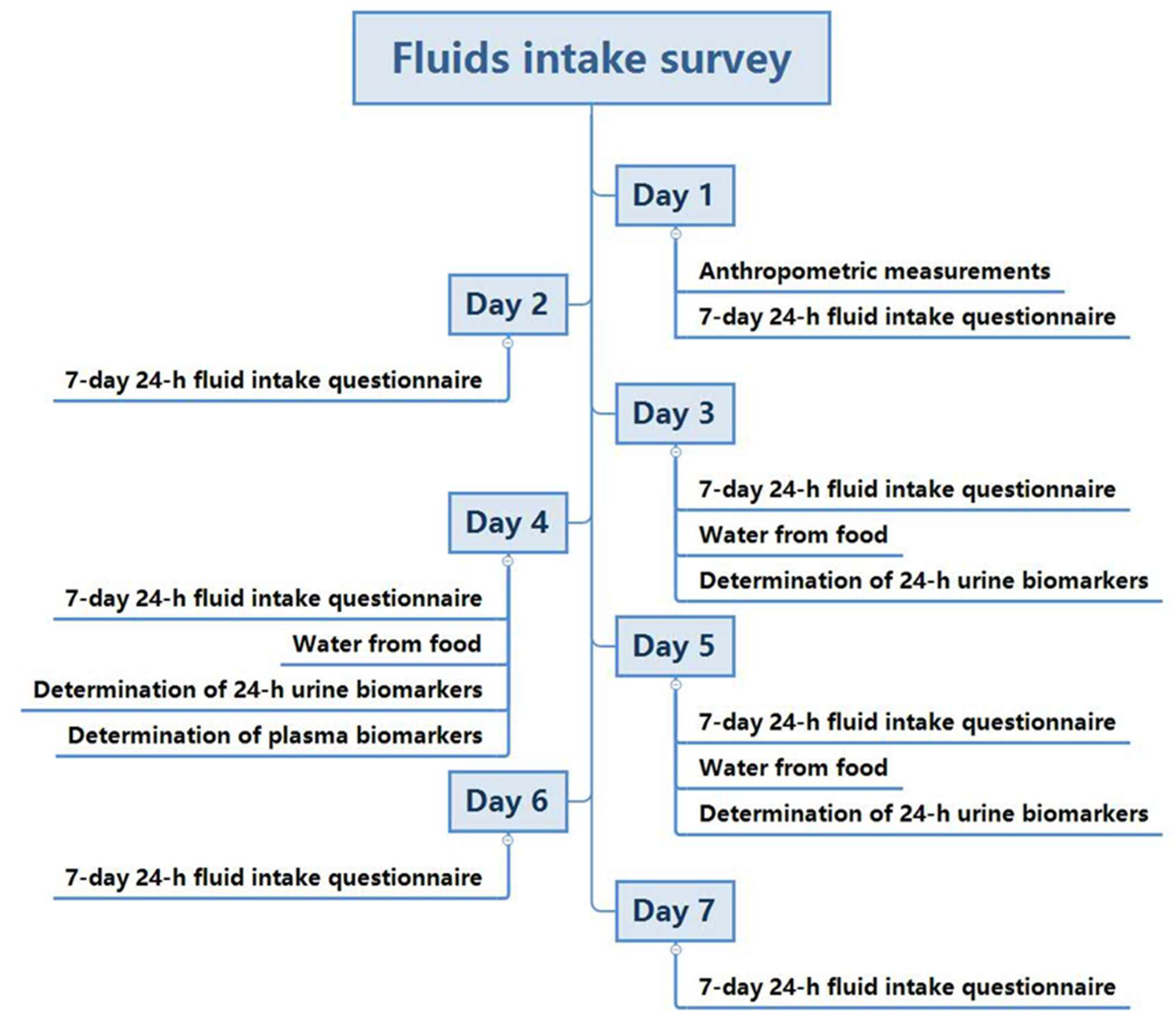
2.3. Study Procedure
2.4. Measurement of Total Water Intake (TWI)
2.5. Temperature and Humidity of the Environment
2.6. Anthropometric Measurements
2.7. Urine Biomarkers
2.8. Blood Biomarkers
2.9. Physical Activity
2.10. Statistics
3. Results
3.1. Measurement of TWI
3.2. Measurement of Urine Indexes
3.3. Measurement of Blood Indexes
4. Discussion
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| LD | Low drinker |
| HD | High drinker |
| TWI | Total water intake |
| EFSA | European Food Safety Authority |
References
- Watso, J.C.; Farquhar, W.B. Hydration Status and Cardiovascular Function. Nutrients 2019, 11, 1866. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wittbrodt, M.T.; Millard-Stafford, M. Dehydration Impairs Cognitive Performance. Med. Sci. Sports Exerc. 2018, 50, 2360–2368. [Google Scholar] [CrossRef] [PubMed]
- Sohn, W.; Heller, K.E.; Burt, B.A. Fluid Consumption Related to Climate among Children in the United States. J. Public Health Dent. 2001, 61, 99–106. [Google Scholar] [CrossRef]
- Westerterp, K.R.; Plasqui, G.; Goris, A.H.C. Water loss as a function of energy intake, physical activity and season. Br. J. Nutr. 2005, 93, 199–203. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galagan, D.J.; Vermillion, J.R.; Nevitt, G.A.; Stadt, Z.M.; Dart, R.E. Climate and Fluid Intake. Public Health Rep. 1957, 72, 484–490. [Google Scholar] [CrossRef] [PubMed]
- Carlson, L.A.; Lawrence, M.A.; Kenefick, R.W. Hydration Status and Thermoregulatory Responses in Drivers during Competitive Racing. J. Strength Cond. Res. 2018, 32, 2061–2065. [Google Scholar] [CrossRef]
- Ferreira-Pêgo, C.; Guelinckx, I.; Moreno, L.A.; Kavouras, S.A.; Gandy, J.; Martinez, H.; Bardosono, S.; Abdollahi, M.; Nasseri, E.; Jarosz, A.; et al. Total fluid intake and its determinants: Cross-sectional surveys among adults in 13 countries worldwide. Eur. J. Nutr. 2015, 54, 35–43. [Google Scholar] [CrossRef] [Green Version]
- Iglesia, I.; Guelinckx, I.; De Miguel-Etayo, P.M.; González-Gil, E.M.; Salas-Salvadó, J.; Kavouras, S.A.; Gandy, J.; Martínez, H.; Bardosono, S.; Abdollahi, M.; et al. Total fluid intake of children and adolescents: Cross-sectional surveys in 13 countries worldwide. Eur. J. Nutr. 2015, 54, 57–67. [Google Scholar] [CrossRef]
- Botigué, T.; Masot, O.; Miranda, J.; Nuin, C.; Viladrosa, M.; Lavedán, A.; Zwakhalen, S. Prevalence and Risk Factors Associated with Low Fluid Intake in Institutionalized Older Residents. J. Am. Med. Dir. Assoc. 2019, 20, 317–322. [Google Scholar] [CrossRef]
- Barnes, K.A.; Anderson, M.L.; Stofan, J.R.; Dalrymple, K.J.; Reimel, A.J.; Roberts, T.J.; Randell, R.K.; Ungaro, C.T.; Baker, L.B. Normative data for sweating rate, sweat sodium concentration, and sweat sodium loss in athletes: An update and analysis by sport. J. Sports Sci. 2019, 37, 2356–2366. [Google Scholar] [CrossRef]
- Kenefick, R.W. Drinking Strategies: Planned Drinking Versus Drinking to Thirst. Sports Med. 2018, 48, 31–37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lavoué, C.; Siracusa, J.; Chalchat, É.; Bourrilhon, C.; Charlot, K. Analysis of food and fluid intake in elite ultra-endurance runners during a 24-h world championship. J. Int. Soc. Sports Nutr. 2020, 17, 36. [Google Scholar] [CrossRef] [PubMed]
- McCartney, D.; Irwin, C.; Cox, G.R.; Desbrow, B. Fluid, energy, and nutrient recovery via ad libitum intake of different commercial beverages and food in female athletes. Appl. Physiol. Nutr. Metab. 2019, 44, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Stofan, J.R.; Osterberg, K.L.; Horswill, C.A.; Lacambra, M.; Eichner, E.R.; Anderson, S.A.; Murray, R. Daily fluid turnover during preseason training in U.S. college football. Int. J. Sport Nutr. Exerc. Metab. 2007, 17, 340–351. [Google Scholar] [CrossRef] [PubMed]
- Ma, G.; Zhang, Q.; Liu, A.; Zuo, J.; Zhang, W.; Zou, S.; Li, X.; Lu, L.; Pan, H.; Hu, X. Fluid intake of adults in four Chinese cities. Nutr. Rev. 2012, 70 (Suppl. 2), S105–S110. [Google Scholar] [CrossRef]
- Zhang, Q.; Hu, X.; Zhou, S.; Zuo, J.; Liu, Z.; Pan, Q.; Liu, C.; Pan, H.; Ma, G. Water intake of adults in four cities of China in summer. Chin. J. Prev. Med. 2011, 45, 677–682. (In Chinese) [Google Scholar]
- Zhang, J.; Zhang, N.; Wang, Y.; Liang, S.; Liu, S.; Du, S.; Xu, Y.; He, H.; Cai, H.; Ma, G. Drinking patterns and hydration biomarkers among young adults with different levels of habitual total drinking fluids intake in Baoding, Hebei Province, China: A cross-sectional study. BMC Public Health 2020, 20, 468. [Google Scholar] [CrossRef]
- Zhang, N.; Du, S.; Tang, Z.; Zheng, M.; Yan, R.; Zhu, Y.; Ma, G. Hydration, Fluid Intake, and Related Urine Biomarkers among Male College Students in Cangzhou, China: A Cross-Sectional Study—Applications for Assessing Fluid Intake and Adequate Water Intake. Int. J. Environ. Res. Public Health 2017, 14, 513. [Google Scholar] [CrossRef] [Green Version]
- Ozen, A.E.; Bibiloni, M.M.; Pons, A.; Tur, J.A. Fluid intake from beverages across age groups: A systematic review. J. Hum. Nutr. Diet. 2015, 28, 417–442. [Google Scholar] [CrossRef]
- Harris, P.R.; Keen, D.A.; Constantopoulos, E.; Weninger, S.N.; Hines, E.; Koppinger, M.P.; Khalpey, Z.I.; Konhilas, J.P. Fluid type influences acute hydration and muscle performance recovery in human subjects. J. Int. Soc. Sports Nutr. 2019, 16, 15. [Google Scholar] [CrossRef] [Green Version]
- Trangmar, S.J.; Gonzalez-Alonso, J. Heat, Hydration and the Human Brain, Heart and Skeletal Muscles. Sports Med. 2019, 49, 69–85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Millard-Stafford, M.; Snow, T.K.; Jones, M.L.; Suh, H. The Beverage Hydration Index: Influence of Electrolytes, Carbohydrate and Protein. Nutrients 2021, 13, 2933. [Google Scholar] [CrossRef] [PubMed]
- Merson, S.J.; Maughan, R.J.; Shirreffs, S.M. Rehydration with drinks differing in sodium concentration and recovery from moderate exercise-induced hypohydration in man. Eur. J. Appl. Physiol. 2008, 103, 585–594. [Google Scholar] [CrossRef] [PubMed]
- Castro-Sepulveda, M.; Johannsen, N.; Astudillo, S.; Jorquera, C.; Álvarez, C.; Zbinden-Foncea, H.; Ramírez-Campillo, R. Effects of Beer, Non-Alcoholic Beer and Water Consumption before Exercise on Fluid and Electrolyte Homeostasis in Athletes. Nutrients 2016, 8, 345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berry, C.W.; Wolf, S.T.; Cottle, R.M.; Kenney, W.L. Hydration Is More Important Than Exogenous Carbohydrate Intake during Push-to-the-Finish Cycle Exercise in the Heat. Front. Sports Act. Living 2021, 3, 742710. [Google Scholar] [CrossRef]
- Perrier, E.; Vergne, S.; Klein, A.; Poupin, M.; Rondeau, P.; Le-Bellego, L.; Armstrong, L.E.; Lang, F.; Stookey, J.; Tack, I. Hydration biomarkers in free-living adults with different levels of habitual fluid consumption. Br. J. Nutr. 2013, 109, 1678–1687. [Google Scholar] [CrossRef] [Green Version]
- Perrier, E.T.; Armstrong, L.E.; Bottin, J.H.; Clark, W.F.; Dolci, A.; Guelinckx, I.; Iroz, A.; Kavouras, S.A.; Lang, F.; Lieberman, H.R.; et al. Hydration for health hypothesis: A narrative review of supporting evidence. Eur. J. Nutr. 2021, 60, 1167–1180. [Google Scholar] [CrossRef]
- Barley, O.R.; Chapman, D.W.; Abbiss, C.R. Reviewing the current methods of assessing hydration in athletes. J. Int. Soc. Sports Nutr. 2020, 17, 52. [Google Scholar] [CrossRef]
- GB/T 1–789-2015; General Administration of Quality Supervision, Inspection and Quarantine of China. General Standard for Beverages. Standardization Administration of China: Beijing, China, 2015.
- Zhang, J.; Zhang, N.; Liang, S.; Wang, Y.; Liu, S.; Liu, S.; Du, S.; He, H.; Xu, Y.; Cai, H.; et al. The amounts and contributions of total drinking fluids and water from food to total water intake of young adults in Baoding, China. Eur. J. Nutr. 2019, 58, 2669–2677. [Google Scholar] [CrossRef]
- GB 5009.3–2016; National Food Safety Standard: Determination of Moisture in Foods. Standardization Administration of China: Beijing, China, 2016.
- Institute for Nutrition and Health, Chinese Center for Disease Control and Prevention. China Food Composition; Peking University Medicine Press: Beijing, China, 2016. [Google Scholar]
- WS/T 424-2013; Anthropometric measurements method in health surveillance. National Health Commission of the People’s Republic of China: Beijing, China, 2013.
- Working Group on Obesity in China. Guidelines for the prevention and control of overweight and obesity in Chinese adults. Acta Nutr. Sin. 2004, 26, 1–4. [Google Scholar]
- Santaliestra-Pasías, A.M.; Dios, J.E.L.; Sprengeler, O.; Hebestreit, A.; De-Henauw, S.; Eiben, G.; Felsö, R.; Lauria, F.; Tornaritis, M.; Veidebaum, T.; et al. Food and beverage intakes according to physical activity levels in European children: The IDEFICS (Identification and prevention of Dietary and lifestyle induced health Effects In Children and Infants) study. Public Health Nutr. 2018, 21, 1717–1725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, X.; Zhang, Q.; Zhang, W.; Wang, Y.; Zhang, D.; Nie, S.; Zuo, J.; Pan, H.; Ma, G. Water intake sources analysis of adults in four cities of China in summer. Chin. J. Prev. Med. 2011, 45, 688–691. (In Chinese) [Google Scholar]
- Hodge, A.M.; Bassett, J.K.; Milne, R.L.; English, D.R.; Giles, G.G. Consumption of sugar-sweetened and artificially sweetened soft drinks and risk of obesity-related cancers. Public Health Nutr. 2018, 21, 1618–1626. [Google Scholar] [CrossRef] [Green Version]
- Kwak, J.H.; Jo, G.; Chung, H.; Shin, M. Association between sugar-sweetened beverage consumption and incident hypertension in Korean adults: A prospective study. Eur. J. Nutr. 2019, 58, 1009–1017. [Google Scholar] [CrossRef] [PubMed]
- Papier, K.; D’Este, C.; Bain, C.; Banwell, C.; Seubsman, S.; Sleigh, A.; Jordan, S. Consumption of sugar-sweetened beverages and type 2 diabetes incidence in Thai adults: Results from an 8-year prospective study. Nutr. Diabetes 2017, 7, e283. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xi, Y.; Lin, Q.; Yang, Q.; Li, F.; Liu, H.; Luo, J.; Ouyang, Y.; Sun, M.; Yong, C.; Xiang, C.; et al. Association between Free Sugars Intake and Excessive Daytime Sleepiness among Chinese Adolescents. Nutrients 2021, 13, 3959. [Google Scholar] [CrossRef]
- Campbell, B.; Wilborn, C.; La, B.P.; Taylor, L.; Nelson, M.T.; Greenwood, M.; Ziegenfuss, T.N.; Lopez, H.L.; Hoffman, J.R.; Stout, J.R.; et al. International Society of Sports Nutrition position stand: Energy drinks. J. Int. Soc. Sports Nutr. 2013, 10, 1. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; Zhang, N.; Liu, S.; Du, S.; He, H.; Ma, G. The comparison of water intake patterns and hydration biomarkers among young adults with different hydration statuses in Hebei, China. Nutr. Metab. 2021, 18, 2. [Google Scholar] [CrossRef]
- Perales-Garcia, A.; Ortega, R.M.; Urrialde, R.; Lopez-Sobaler, A.M. Physical activity and sedentary behavior impacts on dietary water intake and hydration status in Spanish schoolchildren: A cross-sectional study. PLoS ONE 2018, 13, e208748. [Google Scholar] [CrossRef]
- Sekiguchi, Y.; Adams, W.M.; Curtis, R.M.; Benjamin, C.L.; Casa, D.J. Factors influencing hydration status during a National Collegiate Athletics Association division 1 soccer preseason. J. Sci. Med. Sport 2019, 22, 624–628. [Google Scholar] [CrossRef]
- Yeargin, S.; Torres-McGehee, T.M.; Emerson, D.; Koller, J.; Dickinson, J. Hydration, Eating Attitudes and Behaviors in Age and Weight-Restricted Youth American Football Players. Nutrients 2021, 13, 2565. [Google Scholar] [CrossRef]
- Belval, L.N.; Hosokawa, Y.; Casa, D.J.; Adams, W.M.; Armstrong, L.E.; Baker, L.B.; Burke, L.; Cheuvront, S.; Chiampas, G.; González-Alonso, J.; et al. Practical Hydration Solutions for Sports. Nutrients 2019, 11, 1550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Slimani, M.; Baker, J.S.; Cheour, F.; Taylor, L.; Bragazzi, N.L. Steroid hormones and psychological responses to soccer matches: Insights from a systematic review and meta-analysis. PLoS ONE 2017, 12, e186100. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.C.; Munoz, C.X.; Jimenez, L.; Le Bellego, L.; Kupchak, B.R.; Kraemer, W.J.; Casa, D.J.; Maresh, C.M.; Armstrong, L.E. Hormonal and Thirst Modulated Maintenance of Fluid Balance in Young Women with Different Levels of Habitual Fluid Consumption. Nutrients 2016, 8, 302. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Enhörning, S.; Struck, J.; Wirfält, E.; Hedblad, B.; Morgenthaler, N.G.; Melander, O. Plasma Copeptin, A Unifying Factor behind the Metabolic Syndrome. J. Clin. Endocrinol. Metab. 2011, 96, E1065–E1072. [Google Scholar] [CrossRef]
- Perrier, E.; Demazieres, A.; Girard, N.; Pross, N.; Osbild, D.; Metzger, D.; Guelinckx, I.; Klein, A. Circadian variation and responsiveness of hydration biomarkers to changes in daily water intake. Eur. J. Appl. Physiol. 2013, 113, 2143–2151. [Google Scholar] [CrossRef] [Green Version]
- Costello, J.T.; Rendell, R.A.; Furber, M.; Massey, H.C.; Tipton, M.J.; Young, J.S.; Corbett, J. Effects of acute or chronic heat exposure, exercise and dehydration on plasma cortisol, IL-6 and CRP levels in trained males. Cytokine 2018, 110, 277–283. [Google Scholar] [CrossRef] [Green Version]
- Carroll, H.A.; Templeman, I.; Chen, Y.; Edinburgh, R.M.; Burch, E.K.; Jewitt, J.T.; Povey, G.; Robinson, T.D.; Dooley, W.L.; Jones, R.; et al. Effect of acute hypohydration on glycemic regulation in healthy adults: A randomized crossover trial. J. Appl. Physiol. 2019, 126, 422–430. [Google Scholar] [CrossRef]
- Zaplatosch, M.E.; Adams, W.M. The Effect of Acute Hypohydration on Indicators of Glycemic Regulation, Appetite, Metabolism and Stress: A Systematic Review and Meta-Analysis. Nutrients 2020, 12, 2526. [Google Scholar] [CrossRef]
- Castro-Sepulveda, M.; Ramirez-Campillo, R.; Abad-Colil, F.; Monje, C.; Peñailillo, L.; Cancino, J.; Zbinden-Foncea, H. Basal Mild Dehydration Increase Salivary Cortisol after a Friendly Match in Young Elite Soccer Players. Front. Physiol. 2018, 9, 1347. [Google Scholar] [CrossRef] [Green Version]
- Judelson, D.A.; Maresh, C.M.; Yamamoto, L.M.; Farrell, M.J.; Armstrong, L.E.; Kraemer, W.J.; Volek, J.S.; Spiering, B.A.; Casa, D.J.; Anderson, J.M. Effect of hydration state on resistance exercise-induced endocrine markers of anabolism, catabolism, and metabolism. J. Appl. Physiol. 2008, 105, 816–824. [Google Scholar] [CrossRef] [PubMed]
- Baker, L.B.; Ungaro, C.T.; Sopeña, B.C.; Nuccio, R.P.; Reimel, A.J.; Carter, J.M.; Stofan, J.R.; Barnes, K.A. Body map of regional vs. whole body sweating rate and sweat electrolyte concentrations in men and women during moderate exercise-heat stress. J. Appl. Physiol. 2018, 124, 1304–1318. [Google Scholar] [CrossRef] [PubMed]
| LD1 (n = 27) | LD2 (n = 28) | HD1 (n = 27) | HD2 (n = 27) | Total (n = 109) | p | |
|---|---|---|---|---|---|---|
| Age (year) | 20.4 ± 1.1 | 20.6 ± 1.0 | 20.9 ± 1.2 | 20.8 ± 1.0 | 20.8 ± 1.0 | 0.308 |
| Height (cm) | 177.4 ± 4.1 | 178.4 ± 5.1 | 178.1 ± 5.2 | 180.8 ± 5.8 | 178.7 ± 5.2 | 0.082 |
| Weight (kg) | 71.2 ± 5.9 | 69.9 ± 6.4 | 69.5 ± 8.1 | 72.2 ± 8.6 | 70.7 ± 7.3 | 0.500 |
| BMI (Kg/m2) | 22.6 ± 1.5 | 22.0 ± 2.1 | 21.9 ± 2.2 | 22.1 ± 2.1 | 22.1 ± 2.0 | 0.545 |
| Skeletal muscle | 34.1 ± 2.5 | 34.7 ± 2.7 | 34.6 ± 3.2 | 36.2 ± 4.2 | 34.9 ± 3.3 | 0.116 |
| LD1 (n = 27) | LD2 (n = 28) | HD1 (n = 27) | HD2 (n = 27) | Total (n = 109) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| M | Q | % | M | Q | % | M | Q | % | M | Q | % | M | Q | % | |
| Total drinking fluids | 1252 a | 280 | 56.9 a | 1578 d,e | 140 | 63.3 d,e | 1952 b | 280 | 67.2 b | 2685 c,f | 687 | 72.7 c,f | 1789 | 863 | 65.0 |
| Water | 720 a | 414 | 64.0 | 951 d,e | 349 | 61.2 | 1370 b | 255 | 64.4 | 1934 c,f | 653 | 69.7 | 1181 | 666 | 64.8 |
| Tea | 0 | 0 | 1.0 | 0 | 0 | 0.5 | 0 | 0 | 0.7 | 0 | 0 | 0.3 | 0 | 0 | 0.6 |
| Milk and milk products | 31 | 107 | 7.4 | 33 | 104 | 3.8 | 50 | 99 | 4.4 | 50 | 196 | 4.3 | 40 | 111 | 5.0 |
| SSBs | 231 | 241 | 25.4 | 554 | 334 | 31.8 | 535 | 323 | 27.5 | 605 c | 422 | 24.0 | 469 | 424 | 27.2 |
| Sports drinks | 0 | 71 | 3.3 | 71 | 139 | 5.4 | 71 | 169 | 6.1 | 141 c | 193 | 6.8 | 65 | 154 | 5.4 |
| Other SSBs | 231 | 241 | 22.0 | 440 | 275 | 26.4 | 429 | 282 | 21.4 | 423 c | 427 | 17.2 | 383 | 365 | 21.8 |
| Alcohol | 0 | 0 | 2.2 | 0 | 45 | 2.7 | 0 | 86 | 3.0 | 0 | 0 | 1.2 | 0 | 0 | 2.3 |
| Others | 0 | 0 | 0.0 | 0 e | 0 | 0.0 | 0 | 0 | 0.1 | 0 c,f | 0 | 0.5 | 0 | 0 | 0.1 |
| Water from food | 894 | 363 | 43.1 a | 946 | 381 | 36.7 d,e | 1002 | 579 | 32.8 b | 960 | 421 | 27.3 c,f | 955 | 472 | 35.0 |
| Staple food | 330 | 213 | 39.4 | 322 | 92 | 35.0 | 324 | 99 | 35.6 | 363 | 113 | 36.2 | 330 | 107 | 36.5 |
| Dishes | 458 | 224 | 44.9 | 479 | 222 | 48.0 | 407 | 213 | 47.7 | 473 | 225 | 46.2 | 458 | 213 | 46.7 |
| Soup | 0 | 0 | 2.0 | 0 | 31 | 3.5 | 0 | 85 | 3.7 | 0 | 0 | 3.7 | 0 | 0 | 3.3 |
| Porridge | 56 | 179 | 10.3 | 48 | 130 | 8.5 | 43 | 146 | 8.1 | 83 | 171 | 11.1 | 60 | 151 | 9.5 |
| Snacks | 0 | 56 | 3.3 | 14 | 64 | 4.9 | 17 | 69 | 4.9 | 0 | 72 | 2.8 | 9 | 64 | 4.0 |
| Total water intake | 2133 a | 569 | _ | 2550 d | 333 | _ | 2900 b,f | 712 | _ | 3753 c,e | 864 | _ | 2701 | 973 | _ |
| LD1 (n = 27) | LD2 (n = 28) | HD1 (n = 27) | HD2 (n = 27) | Total (n = 109) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| M | Q | M | Q | M | Q | M | Q | M | Q | |
| Volume (mL) | 711 | 386 | 726 | 534 | 876 | 281 | 938 | 513 | 850 | 408 |
| Urine Osmolality (mOsm/kg) | 858 | 292 | 774 | 200 | 732 | 303 | 631 | 379 | 764 | 286 |
| (≤500 mOsm/kg, n, %) | 2 (11.8%) a,* | 1 (5.9%) a,b | 4 (23.5%) b | 10 (58.8%) b | 17 (15.6%) | |||||
| Void | 3.8 ± 1.2 | 3.8 ± 1.3 | 3.9 ± 1.1 | 4.2 ± 1.3 | 3.9 ± 1.2 | |||||
| Na (mmol/L) | 229 | 63 | 212 | 52 | 186 | 74 | 192 | 85 | 202 | 66 |
| K (mmol/L) | 45.45 | 14.59 | 48.25 | 9.76 | 42.14 | 13.99 | 41.29 | 15.00 | 45.21 | 12.93 |
| Cl (mmol/L) | 222 | 35 | 229 | 35 | 203 | 49 | 218 | 83 | 221 | 53 |
| USG | 1.020 | 0.010 | 1.022 | 0.007 | 1.020 | 0.007 | 1.020 | 0.008 | 1.020 | 0.007 |
| pH | 6.7 | 0.7 | 6.2 | 0.6 | 6.3 | 0.5 | 6.3 | 0.6 | 6.3 | 0.5 |
| LD1 (n = 27) | LD2 (n = 28) | HD1 (n = 27) | HD2 (n = 27) | Total (n = 109) | p | |
|---|---|---|---|---|---|---|
| Copeptin (ρmmol/L) | 1.68 ± 0.14 | 1.66 ± 0.15 | 1.65 ± 0.12 | 1.68 ± 0.10 | 1.67 ± 0.13 | 0.776 |
| Testosterone (nmol/L) | 17.4 ± 2.6 | 17.5 ± 2.6 | 16.4 ± 2.2 | 16.4 ± 2.2 | 16.9 ± 2.5 | 0.177 |
| Cortisol (ng/L) | 80.9 ± 11.4 | 81.4 ± 16.9 | 84.1 ± 14.9 | 96.9 ± 15.7 | 83.3 ± 14.9 | 0.425 |
| Creatinine (μmmol/L) | 65.5 ± 14.2 | 65.2 ± 16.8 | 65.5 ± 15.0 | 66.8 ± 14.1 | 65.7 ± 14.9 | 0.980 |
| Na (mmol/L) | 141 ± 3 | 142 ± 3 | 141 ± 3 | 140 ± 4 | 141 ± 4 | 0.327 |
| K (mmol/L) | 4.06 ± 0.51 | 4.20 ± 0.64 | 4.31 ± 0.63 | 4.63 ± 0.73 c | 4.30 ± 0.66 | 0.010 |
| Cl (mmol/L) | 104 ± 8 | 102 ± 6 | 104 ± 7 | 102 ± 7 | 103 ± 7 | 0.534 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Zhang, N.; Li, Y.; He, H.; Ma, G. Habitual Total Drinking Fluid Intake Did Not Affect Plasma Hydration Biomarkers among Young Male Athletes in Beijing, China: A Cross-Sectional Study. Nutrients 2022, 14, 2311. https://doi.org/10.3390/nu14112311
Zhang J, Zhang N, Li Y, He H, Ma G. Habitual Total Drinking Fluid Intake Did Not Affect Plasma Hydration Biomarkers among Young Male Athletes in Beijing, China: A Cross-Sectional Study. Nutrients. 2022; 14(11):2311. https://doi.org/10.3390/nu14112311
Chicago/Turabian StyleZhang, Jianfen, Na Zhang, Yibin Li, Hairong He, and Guansheng Ma. 2022. "Habitual Total Drinking Fluid Intake Did Not Affect Plasma Hydration Biomarkers among Young Male Athletes in Beijing, China: A Cross-Sectional Study" Nutrients 14, no. 11: 2311. https://doi.org/10.3390/nu14112311
APA StyleZhang, J., Zhang, N., Li, Y., He, H., & Ma, G. (2022). Habitual Total Drinking Fluid Intake Did Not Affect Plasma Hydration Biomarkers among Young Male Athletes in Beijing, China: A Cross-Sectional Study. Nutrients, 14(11), 2311. https://doi.org/10.3390/nu14112311







