An Investigation into the Effects of a Curcumin Extract (Curcugen®) on Osteoarthritis Pain of the Knee: A Randomised, Double-Blind, Placebo-Controlled Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Sample Size Calculation
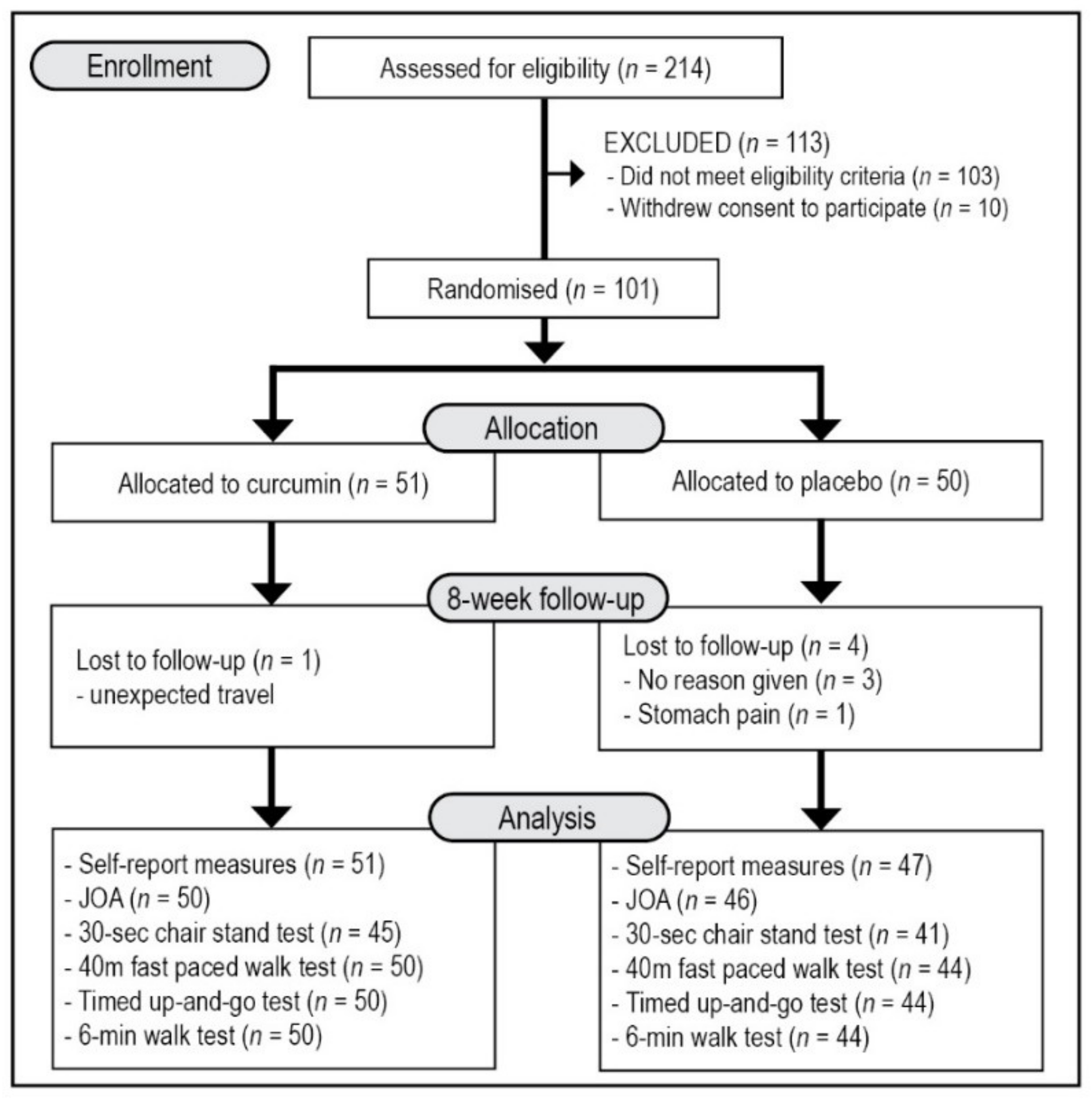
2.3. Recruitment and Randomisation
2.4. Participants
2.5. Interventions
2.6. Outcome Measures
2.6.1. Primary Outcome Measure:
2.6.2. Secondary Outcome Measures:
- 30-s chair stand test—from a sitting position, participants stood up completely so their hips and knees were fully extended, then completely back down, so that their bottom fully touched the seat. The chair had no arms, and from the seated position the chair height was 50 cm. The number of repetitions over 30 s was recorded.
- 40-m fast-paced walk test—the amount of time taken for participants to walk 4 by 10-m lengths where they were required to change direction 180 degrees and walk around a cone after every 10-m length was recorded.
- 6-min walk test—participants walked as quickly as possible for six minutes to cover as much ground as possible. The distance travelled was recorded. The distance between cones was 17 m.
- Timed up-and-go test—participants were asked to stand up, walk around a cone 3 m away, and return to sit back in the chair at their regular pace. The chair had no arms, and from the seated position the chair height was 50 cm. The time to complete this task was recorded.
2.7. Statistical Analysis
3. Results
3.1. Study Population
3.2. Outcome Measures
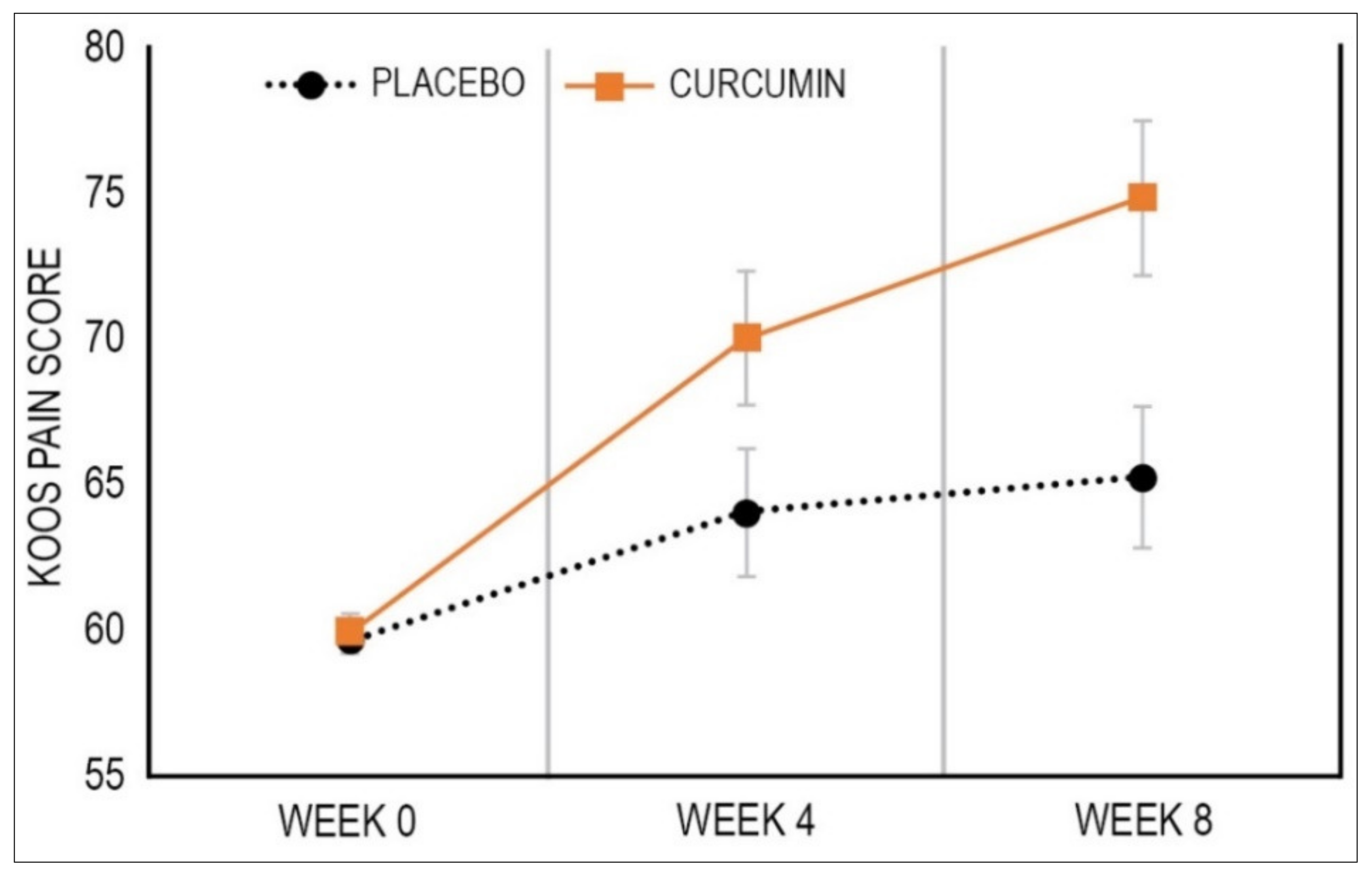
3.2.1. Primary Outcome Measure: KOOS Knee Pain Subscale Score
3.2.2. Secondary Outcome Measures: Remaining KOOS Subscale Scores
3.2.3. Secondary Outcome Measure: Knee Pain Rating
3.2.4. Secondary Outcome Measure: JOA
3.2.5. Secondary Outcome Measure: PROMIS-29 Subscale Scores
3.2.6. Secondary Outcome Measure: Performance Testing Scores
3.2.7. Pain Medication Use
3.2.8. Intake of Supplements
3.2.9. Efficacy of Participant Blinding
3.2.10. Adverse Events
4. Discussion
Study Limitations and Directions for Future Research
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brandt, K.D.; Radin, E.L.; Dieppe, P.A.; van de Putte, L. Yet more evidence that osteoarthritis is not a cartilage disease. Ann. Rheum. Dis. 2006, 65, 1261–1264. [Google Scholar] [CrossRef] [PubMed]
- Hunter, D.J.; Bierma-Zeinstra, S. Osteoarthritis. Lancet 2019, 393, 1745–1759. [Google Scholar] [CrossRef]
- Feigin, V.; GBD 2015 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: A systematic analysis for the global burden of disease study 2015. Lancet 2016, 388, 1545–1602. [Google Scholar] [CrossRef] [Green Version]
- Cui, A.; Li, H.; Wang, D.; Zhong, J.; Chen, Y.; Lu, H. Global, regional prevalence, incidence and risk factors of knee osteoarthritis in population-based studies. EClinicalMedicine 2020, 29–30, 100587. [Google Scholar] [CrossRef]
- Mora, J.C.; Przkora, R.; Cruz-Almeida, Y. Knee osteoarthritis: Pathophysiology and current treatment modalities. J. Pain Res. 2018, 11, 2189–2196. [Google Scholar] [CrossRef] [Green Version]
- da Costa, B.R.; Reichenbach, S.; Keller, N.; Nartey, L.; Wandel, S.; Juni, P.; Trelle, S. Effectiveness of non-steroidal anti-inflammatory drugs for the treatment of pain in knee and hip osteoarthritis: A network meta-analysis. Lancet 2017, 390, e21–e33. [Google Scholar] [CrossRef]
- Beaudart, C.; Lengele, L.; Leclercq, V.; Geerinck, A.; Sanchez-Rodriguez, D.; Bruyere, O.; Reginster, J.Y. Symptomatic efficacy of pharmacological treatments for knee osteoarthritis: A systematic review and a network meta-analysis with a 6-month time horizon. Drugs 2020, 80, 1947–1959. [Google Scholar] [CrossRef]
- Wongrakpanich, S.; Wongrakpanich, A.; Melhado, K.; Rangaswami, J. A comprehensive review of non-steroidal anti-inflammatory drug use in the elderly. Aging Dis. 2018, 9, 143–150. [Google Scholar] [CrossRef] [Green Version]
- Sharifi-Rad, J.; Rayess, Y.E.; Rizk, A.A.; Sadaka, C.; Zgheib, R.; Zam, W.; Sestito, S.; Rapposelli, S.; Neffe-Skocinska, K.; Zielinska, D.; et al. Turmeric and its major compound curcumin on health: Bioactive effects and safety profiles for food, pharmaceutical, biotechnological and medicinal applications. Front. Pharmacol. 2020, 11, 01021. [Google Scholar] [CrossRef]
- Shehzad, A.; Rehman, G.; Lee, Y.S. Curcumin in inflammatory diseases. BioFactors 2013, 39, 69–77. [Google Scholar] [CrossRef]
- Marton, L.T.; Barbalho, S.M.; Sloan, K.P.; Sloan, L.A.; Goulart, R.A.; Araujo, A.C.; Bechara, M.D. Curcumin, autoimmune and inflammatory diseases: Going beyond conventional therapy—A systematic review. Crit. Rev. Food Sci. Nutr. 2020, 1–19. [Google Scholar] [CrossRef]
- Paultre, K.; Cade, W.; Hernandez, D.; Reynolds, J.; Greif, D.; Best, T.M. Therapeutic effects of turmeric or curcumin extract on pain and function for individuals with knee osteoarthritis: A systematic review. BMJ Open Sport Exerc. Med. 2021, 7, e000935. [Google Scholar] [CrossRef]
- Wang, Z.; Singh, A.; Jones, G.; Winzenberg, T.; Ding, C.; Chopra, A.; Das, S.; Danda, D.; Laslett, L.; Antony, B. Efficacy and safety of turmeric extracts for the treatment of knee osteoarthritis: A systematic review and meta-analysis of randomised controlled trials. Curr. Rheumatol. Rep. 2021, 23, 11. [Google Scholar] [CrossRef]
- Hsiao, A.F.; Lien, Y.C.; Tzeng, I.S.; Liu, C.T.; Chou, S.H.; Horng, Y.S. The efficacy of high- and low-dose curcumin in knee osteoarthritis: A systematic review and meta-analysis. Complement. Ther. Med. 2021, 63, 102775. [Google Scholar] [CrossRef]
- Roos, E.M.; Toksvig-Larsen, S. Knee injury and Osteoarthritis Outcome Score (KOOS)—validation and comparison to the WOMAC in total knee replacement. Health Qual. Life Outcomes 2003, 1, 17. [Google Scholar] [CrossRef] [Green Version]
- Roos, E.M.; Roos, H.P.; Lohmander, L.S.; Ekdahl, C.; Beynnon, B.D. Knee injury and osteoarthritis outcome score (KOOS)—Development of a self-administered outcome measure. J. Orthop. Sports Phys. Ther. 1998, 28, 88–96. [Google Scholar] [CrossRef]
- Okuda, M.; Omokawa, S.; Okahashi, K.; Akahane, M.; Tanaka, Y. Validity and reliability of the Japanese Orthopaedic Association score for osteoarthritic knees. J. Orthop. Sci. 2012, 17, 750–756. [Google Scholar] [CrossRef]
- Bevans, M.; Ross, A.; Cella, D. Patient-Reported Outcomes Measurement Information System (PROMIS): Efficient, standardized tools to measure self-reported health and quality of life. Nurs. Outlook 2014, 62, 339–345. [Google Scholar] [CrossRef] [Green Version]
- Alghadir, A.H.; Anwer, S.; Iqbal, A.; Iqbal, Z.A. Test-retest reliability, validity, and minimum detectable change of visual analog, numerical rating, and verbal rating scales for measurement of osteoarthritic knee pain. J. Pain Res. 2018, 11, 851–856. [Google Scholar] [CrossRef] [Green Version]
- Ferreira-Valente, M.A.; Pais-Ribeiro, J.L.; Jensen, M.P. Validity of four pain intensity rating scales. Pain 2011, 152, 2399–2404. [Google Scholar] [CrossRef]
- Dobson, F.; Hinman, R.S.; Hall, M.; Terwee, C.B.; Roos, E.M.; Bennell, K.L. Measurement properties of performance-based measures to assess physical function in hip and knee osteoarthritis: A systematic review. Osteoarthr. Cartil. 2012, 20, 1548–1562. [Google Scholar] [CrossRef] [Green Version]
- Dobson, F.; Hinman, R.S.; Roos, E.M.; Abbott, J.H.; Stratford, P.; Davis, A.M.; Buchbinder, R.; Snyder-Mackler, L.; Henrotin, Y.; Thumboo, J.; et al. OARSI recommended performance-based tests to assess physical function in people diagnosed with hip or knee osteoarthritis. Osteoarthr. Cartil. 2013, 21, 1042–1052. [Google Scholar] [CrossRef] [Green Version]
- Dobson, F.; Bennell, K.L.; Hinman, R.S. Recommended Performance-Based Tests to Assess Physical Function in People Diagnosed with Hip or Knee Osteoarthritis. Osteoarthritis Research Society International. 2013. Available online: https://oarsi.org/sites/default/files/docs/2013/manual.pdf (accessed on 1 March 2020).
- Jacquet, C.; Pioger, C.; Khakha, R.; Steltzlen, C.; Kley, K.; Pujol, N.; Ollivier, M. Evaluation of the "Minimal Clinically Important Difference" (MCID) of the KOOS, KSS and SF-12 scores after open-wedge high tibial osteotomy. Knee Surg. Sports Traumatol. Arthrosc. 2021, 29, 820–826. [Google Scholar] [CrossRef]
- Roos, E.M.; Lohmander, L.S. The Knee injury and Osteoarthritis Outcome Score (KOOS): From joint injury to osteoarthritis. Health Qual. Life Outcomes 2003, 1, 64. [Google Scholar] [CrossRef] [Green Version]
- Alghadir, A.; Anwer, S.; Brismee, J.M. The reliability and minimal detectable change of Timed Up and Go test in individuals with grade 1-3 knee osteoarthritis. BMC Musculoskelet. Disord. 2015, 16, 174. [Google Scholar] [CrossRef] [Green Version]
- Shahtahmassebi, B.; Hebert, J.J.; Hecimovich, M.; Fairchild, T.J. Trunk exercise training improves muscle size, strength, and function in older adults: A randomized controlled trial. Scand. J. Med. Sci. Sports 2019, 29, 980–991. [Google Scholar] [CrossRef]
- Camarri, B.; Eastwood, P.R.; Cecins, N.M.; Thompson, P.J.; Jenkins, S. Six minute walk distance in healthy subjects aged 55–75 years. Respir. Med. 2006, 100, 658–665. [Google Scholar] [CrossRef] [Green Version]
- Deyle, G.D.; Henderson, N.E.; Matekel, R.L.; Ryder, M.G.; Garber, M.B.; Allison, S.C. Effectiveness of manual physical therapy and exercise in osteoarthritis of the knee. A randomized, controlled trial. Ann. Intern. Med. 2000, 132, 173–181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Z.; Jones, G.; Winzenberg, T.; Cai, G.; Laslett, L.L.; Aitken, D.; Hopper, I.; Singh, A.; Jones, R.; Fripp, J.; et al. Effectiveness of curcuma longa extract for the treatment of symptoms and effusion-synovitis of knee osteoarthritis: A randomized trial. Ann. Intern. Med. 2020, 173, 861–869. [Google Scholar] [CrossRef]
- Zeng, C.; Wei, J.; Persson, M.S.M.; Sarmanova, A.; Doherty, M.; Xie, D.; Wang, Y.; Li, X.; Li, J.; Long, H.; et al. Relative efficacy and safety of topical non-steroidal anti-inflammatory drugs for osteoarthritis: A systematic review and network meta-analysis of randomised controlled trials and observational studies. Br. J. Sports Med. 2018, 52, 642–650. [Google Scholar] [CrossRef] [PubMed]
- Kuptniratsaikul, V.; Thanakhumtorn, S.; Chinswangwatanakul, P.; Wattanamongkonsil, L.; Thamlikitkul, V. Efficacy and safety of Curcuma domestica extracts in patients with knee osteoarthritis. J. Altern. Complement. Med. 2009, 15, 891–897. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pinsornsak, P.; Niempoog, S. The efficacy of Curcuma Longa L. extract as an adjuvant therapy in primary knee osteoarthritis: A randomized control trial. J. Med. Assoc. Thail. 2012, 95 (Suppl. S1), S51–S58. [Google Scholar]
- Hewlings, S.J.; Kalman, D.S. Curcumin: A review of its’ effects on human health. Foods 2017, 6, 92. [Google Scholar] [CrossRef] [PubMed]
- Jurenka, J.S. Anti-inflammatory properties of curcumin, a major constituent of Curcuma longa: A review of preclinical and clinical research. Altern. Med. Rev. 2009, 14, 141–153. [Google Scholar]
- Chen, T.; Zhou, R.; Chen, Y.; Fu, W.; Wei, X.; Ma, G.; Hu, W.; Lu, C. Curcumin ameliorates IL-1beta-induced apoptosis by activating autophagy and inhibiting the NF-kappaB signaling pathway in rat primary articular chondrocytes. Cell Biol. Int. 2021, 45, 976–988. [Google Scholar] [CrossRef]
- Yabas, M.; Orhan, C.; Er, B.; Tuzcu, M.; Durmus, A.S.; Ozercan, I.H.; Sahin, N.; Bhanuse, P.; Morde, A.A.; Padigaru, M.; et al. A next generation formulation of curcumin ameliorates experimentally induced osteoarthritis in rats via regulation of inflammatory mediators. Front. Immunol. 2021, 12, 609629. [Google Scholar] [CrossRef]
- Atabaki, M.; Shariati-Sarabi, Z.; Tavakkol-Afshari, J.; Mohammadi, M. Significant immunomodulatory properties of curcumin in patients with osteoarthritis; a successful clinical trial in Iran. Int. Immunopharmacol. 2020, 85, 106607. [Google Scholar] [CrossRef]
- Kertia, N.; Asdie, A.H.; Marsetyawan, R.W. Ability of curcuminoid compared to diclofenac sodium in reducing the secretion of cycloxygenase-2 enzyme by synovial fluid’s monocytes of patients with osteoarthritis. Acta Med. Indones. 2012, 44, 105–113. [Google Scholar]
- Singhal, S.; Hasan, N.; Nirmal, K.; Chawla, R.; Chawla, S.; Kalra, B.S.; Dhal, A. Bioavailable turmeric extract for knee osteoarthritis: A randomized, non-inferiority trial versus paracetamol. Trials 2021, 22, 105. [Google Scholar] [CrossRef]
- Rahimnia, A.R.; Panahi, Y.; Alishiri, G.; Sharafi, M.; Sahebkar, A. Impact of supplementation with curcuminoids on systemic inflammation in patients with knee osteoarthritis: Findings from a randomized double-blind placebo-controlled trial. Drug Res. 2015, 65, 521–525. [Google Scholar] [CrossRef]
- Panahi, Y.; Alishiri, G.H.; Parvin, S.; Sahebkar, A. Mitigation of systemic oxidative stress by curcuminoids in osteoarthritis: Results of a randomized controlled trial. J. Diet. Suppl. 2016, 13, 209–220. [Google Scholar] [CrossRef]
- Srivastava, S.; Saksena, A.K.; Khattri, S.; Kumar, S.; Dagur, R.S. Curcuma longa extract reduces inflammatory and oxidative stress biomarkers in osteoarthritis of knee: A four-month, double-blind, randomized, placebo-controlled trial. Inflammopharmacology 2016, 24, 377–388. [Google Scholar] [CrossRef]
- Henrotin, Y.; Malaise, M.; Wittoek, R.; de Vlam, K.; Brasseur, J.P.; Luyten, F.P.; Jiangang, Q.; Van den Berghe, M.; Uhoda, R.; Bentin, J.; et al. Bio-optimized Curcuma longa extract is efficient on knee osteoarthritis pain: A double-blind multicenter randomized placebo controlled three-arm study. Arthritis Res. Ther. 2019, 21, 179. [Google Scholar] [CrossRef] [Green Version]
- Kellgren, J.H.; Lawrence, J.S. Radiological assessment of osteo-arthrosis. Ann. Rheum. Dis. 1957, 16, 494–502. [Google Scholar] [CrossRef] [Green Version]
- Oishi, K.; Tsuda, E.; Yamamoto, Y.; Maeda, S.; Sasaki, E.; Chiba, D.; Takahashi, I.; Nakaji, S.; Ishibashi, Y. The Knee injury and Osteoarthritis Outcome Score reflects the severity of knee osteoarthritis better than the revised Knee Society Score in a general Japanese population. Knee 2016, 23, 35–42. [Google Scholar] [CrossRef] [Green Version]
- Filosa, S.; Di Meo, F.; Crispi, S. Polyphenols-gut microbiota interplay and brain neuromodulation. Neural Regen. Res. 2018, 13, 2055–2059. [Google Scholar] [CrossRef]
- Correa, T.A.F.; Rogero, M.M.; Hassimotto, N.M.A.; Lajolo, F.M. The Two-Way Polyphenols-Microbiota Interactions and Their Effects on Obesity and Related Metabolic Diseases. Front. Nutr. 2019, 6, 188. [Google Scholar] [CrossRef] [Green Version]
| Placebo (n = 50) | Curcumin (n = 51) | p-Value | ||
|---|---|---|---|---|
| Age | Mean | 57.92 | 59.59 | 0.195 a |
| SE | 0.88 | 0.92 | ||
| Gender | Female (n) | 26 | 24 | 0.619 b |
| Male (n) | 24 | 27 | ||
| BMI | Mean | 28.82 | 28.93 | 0.898 a |
| SE | 0.60 | 0.65 | ||
| Systolic blood pressure (mmHg) | Mean | 141.50 | 138.16 | 0.312 a |
| SE | 2.54 | 2.09 | ||
| Diastolic blood pressure (mmHg) | Mean | 87.56 | 87.08 | 0.793 a |
| SE | 1.46 | 1.10 | ||
| Marital status | Single | 9 | 11 | 0.653 b |
| Married/defacto | 41 | 40 | ||
| Took pain-relieving medications during previous week | Yes (%) | 49 | 53 | 0.692 b |
| Educational level | Secondary | 28 | 23 | 0.149 b |
| Tertiary | 16 | 14 | ||
| Post-graduate | 6 | 14 | ||
| Exercise level | Never/rarely | 14 | 11 | 0.629 b |
| 1 to 2 times a week | 4 | 6 | ||
| 3 to 5 times a week | 16 | 13 | ||
| 6+ times a week | 16 | 21 | ||
| KOOS—Pain | Mean | 61.17 | 60.68 | 0.848 a |
| SE | 1.93 | 1.68 | ||
| KOOS—Symptoms | Mean | 59.29 | 59.00 | 0.922 a |
| SE | 2.07 | 2.09 | ||
| KOOS—Daily Living | Mean | 69.41 | 68.96 | 0.886 a |
| SE | 2.39 | 2.08 | ||
| KOOS—Sports and recreational activities | Mean | 41.00 | 43.77 | 0.559 a |
| SE | 3.36 | 3.33 | ||
| KOOS—Quality of life | Mean | 38.25 | 40.81 | 0.485 a |
| SE | 2.72 | 2.43 | ||
| Knee pain rating | Mean | 5.80 | 6.22 | 0.174 a |
| SE | 0.22 | 0.20 | ||
| JOA—Total Score | Mean | 70.90 | 69.90 | 0.753 a |
| SE | 2.23 | 2.24 | ||
| PROMIS-29—Physical function | Mean | 3.92 | 3.93 | 0.979 a |
| SE | 0.09 | 0.10 | ||
| PROMIS-29—Anxiety | Mean | 1.46 | 1.43 | 0.802 a |
| SE | 0.09 | 0.08 | ||
| PROMIS- 29—Depression | Mean | 1.35 | 1.36 | 0.954 a |
| SE | 0.08 | 0.09 | ||
| PROMIS-29—Fatigue | Mean | 2.16 | 2.12 | 0.820 a |
| SE | 0.13 | 0.12 | ||
| PROMIS-29—Sleep disturbance | Mean | 2.52 | 2.66 | 0.476 a |
| SE | 0.13 | 0.15 | ||
| PROMIS-29—Social roles | Mean | 3.55 | 3.63 | 0.660 a |
| SE | 0.14 | 0.13 | ||
| PROMIS-29—Pain interference | Mean | 2.44 | 2.45 | 0.933 a |
| SE | 0.11 | 0.14 | ||
| PROMIS-29—Pain intensity | Mean | 4.28 | 4.69 | 0.245 a |
| SE | 0.22 | 0.27 | ||
| 30-s chair stand test | Mean | 17.02 | 15.86 | 0.280 a |
| SE | 0.75 | 0.77 | ||
| 40 m fast paced walk test | Mean | 25.87 | 26.18 | 0.708 a |
| SE | 0.64 | 0.56 | ||
| Timed up-and-go test | Mean | 6.46 | 6.84 | 0.126 a |
| SE | 0.17 | 0.18 | ||
| 6-min walk test | Mean | 522.15 | 515.42 | 0.669 a |
| SE | 12.61 | 9.40 |
| Endpoints | Placebo (n = 47) | Curcumin (n = 51) | Mean Between-Group Difference in Change (95% CI) | p-value † | Cohen’s d |
|---|---|---|---|---|---|
| Mean Change (95% CI) | Mean Change (95% CI) | ||||
| Primary | |||||
| KOOS—Pain (Primary) | 5.52 (0.75 to 10.28) | 11.98 (7.38 to 16.59) | 6.47 (0.18 to 12.75) | 0.009 | 0.39 |
| Secondary | |||||
| Knee pain rating | −1.57 (−2.08 to −1.07) | −2.09 (−2.58 to −1.61) | −0.52 (−1.19 to 0.14) | 0.001 | 0.30 |
| KOOS—Symptoms | 5.21 (0.45 to 9.98) | 6.48 (1.86 to 11.09) | 1.26 (−5.02 to 7.55) | 0.473 | 0.08 |
| KOOS—Daily living | 5.42 (0.86 to 9.98) | 8.20 (3.79 to 12.61) | 2.78 (−3.23 to 8.79) | 0.106 | 0.18 |
| KOOS—Sports and recreation | 10.13 (3.63 to 16.63) | 8.79 (2.53 to 16.06) | −1.34 (−9.89 to 7.21) | 0.454 | 0.06 |
| KOOS—Quality of life | 9.83 (3.83 to 15.84) | 13.69 (7.92 to 19.47) | 3.86 (−4.00 to 11.72) | 0.624 | 0.19 |
| PROMIS-29 Physical function | −0.01 (−2.11 to 0.19) | 0.24 (0.05 to 0.43) | 0.25 (−0.01 to 0.52) | 0.082 | 0.36 |
| PROMIS-28 Anxiety | 0.07 (−0.10 to 0.24) | −0.14 (−0.03 to 0.03) | −0.21 (−0.43 to 0.01) | 0.168 | 0.36 |
| PROMIS-29 Depression | −0.10 (−0.25 to 0.04) | −0.14 (−0.28 to 0.00) | −0.04 (−0.23 to 0.15) | 0.103 | 0.08 |
| PROMIS-29 Fatigue | −0.20 (−0.40 to 0.01) | −0.06 (−0.25 to 0.14) | 0.14 (−0.13 to 0.41) | 0.370 | 0.20 |
| PROMIS-29 Sleep | −0.14 (−0.36 to 0.08) | −0.15 (−0.36 to 0.07) | 0.01 (−0.30 to 0.29) | 0.553 | 0.01 |
| PROMIS-29 Social roles | 0.15 (−0.11 to 0.41) | 0.08 (−0.16 to 0.33) | −0.07 (−0.41 to 0.27) | 0.501 | 0.08 |
| PROMIS-29 Pain interference | −0.45 (−0.68 to −0.23) | −0.47 (−0.68 to −0.25) | −0.01 (−0.31 to 0.28) | 0.872 | 0.03 |
| PROMIS-29 Pain intensity | −0.74 (−1.37 to −0.11) | −0.82 (−1.43 to −0.20) | −0.08 (−0.92 to 0.77) | 0.550 | 0.04 |
| JOA | 1.11 (−2.25 to 4.47) | 9.59 (6.34 to 12.84) | 8.48 (4.03 to 12.92) | <0.001 | 0.74 |
| Placebo (n = 47) | Curcumin (n = 51) | p-Value | ||
|---|---|---|---|---|
| KOOS—Symptoms | Yes | 11 | 14 | 0.646 |
| No | 36 | 37 | ||
| KOOS—Pain | Yes | 9 | 20 | 0.045 |
| No | 38 | 31 | ||
| KOOS—Daily living | Yes | 11 | 10 | 0.647 |
| No | 36 | 41 | ||
| KOOS—Sports and recreation | Yes | 18 | 18 | 0.758 |
| No | 29 | 33 | ||
| KOOS—Quality of life | Yes | 13 | 17 | 0.543 |
| No | 34 | 34 |
| Placebo | Curcumin | Between-Group, p-Value b | ||||||
|---|---|---|---|---|---|---|---|---|
| Week 0 | Week 8 | Within-Group, p-Value a | Week 0 | Week 8 | Within-Group, p-Value a | |||
| 30-s chair stand test (repetitions) | n | 41 | <0.001 a | 45 | <0.001 | 0.391 | ||
| Mean | 17.32 | 20.54 | 16.29 | 19.91 | ||||
| SE | 0.83 | 0.87 | 0.8 | 0.88 | ||||
| 40 m fast paced walk test (s) | n | 44 | 0.795 a | 50 | 0.699 | 0.604 | ||
| Mean | 25.6 | 25.68 | 26.28 | 26.11 | ||||
| SE | 0.64 | 0.6 | 0.57 | 0.61 | ||||
| Timed up-and-go test (s) | n | 44 | 0.061 a | 50 | <0.001 | 0.032 | ||
| Mean | 6.43 | 6.22 | 6.86 | 6.36 | ||||
| SE | 0.18 | 0.18 | 0.19 | 0.18 | ||||
| 6-min walk test (m) | n | 44 | 0.480 a | 50 | 0.005 | 0.013 | ||
| Mean | 525.81 | 521.07 | 512.3 | 530.18 | ||||
| SE | 13.91 | 13.54 | 9.05 | 11.21 | ||||
| Placebo (n = 47) | Curcumin (n = 51) | p-Value * | |
|---|---|---|---|
| No Change | 31 | 27 | 0.014 |
| Decreased | 6 | 19 | |
| Increased | 10 | 5 |
| Placebo | Curcumin | |
|---|---|---|
| Loose stools/diarrhoea | 1 | 3 |
| Heartburn/indigestion | 1 | |
| Stomach pain | 2 | 1 |
| Nausea | 1 | |
| Constipation | 1 | |
| Tiredness | 2 | |
| Irritability | 1 | |
| Mouth ulcer | 1 | |
| Pins and needles in foot | 1 | |
| Total number of adverse effects | 10 | 5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lopresti, A.L.; Smith, S.J.; Jackson-Michel, S.; Fairchild, T. An Investigation into the Effects of a Curcumin Extract (Curcugen®) on Osteoarthritis Pain of the Knee: A Randomised, Double-Blind, Placebo-Controlled Study. Nutrients 2022, 14, 41. https://doi.org/10.3390/nu14010041
Lopresti AL, Smith SJ, Jackson-Michel S, Fairchild T. An Investigation into the Effects of a Curcumin Extract (Curcugen®) on Osteoarthritis Pain of the Knee: A Randomised, Double-Blind, Placebo-Controlled Study. Nutrients. 2022; 14(1):41. https://doi.org/10.3390/nu14010041
Chicago/Turabian StyleLopresti, Adrian L., Stephen J. Smith, Shavon Jackson-Michel, and Timothy Fairchild. 2022. "An Investigation into the Effects of a Curcumin Extract (Curcugen®) on Osteoarthritis Pain of the Knee: A Randomised, Double-Blind, Placebo-Controlled Study" Nutrients 14, no. 1: 41. https://doi.org/10.3390/nu14010041
APA StyleLopresti, A. L., Smith, S. J., Jackson-Michel, S., & Fairchild, T. (2022). An Investigation into the Effects of a Curcumin Extract (Curcugen®) on Osteoarthritis Pain of the Knee: A Randomised, Double-Blind, Placebo-Controlled Study. Nutrients, 14(1), 41. https://doi.org/10.3390/nu14010041






