Nutrient Intake and Status in Adults Consuming Plant-Based Diets Compared to Meat-Eaters: A Systematic Review
Abstract
1. Introduction
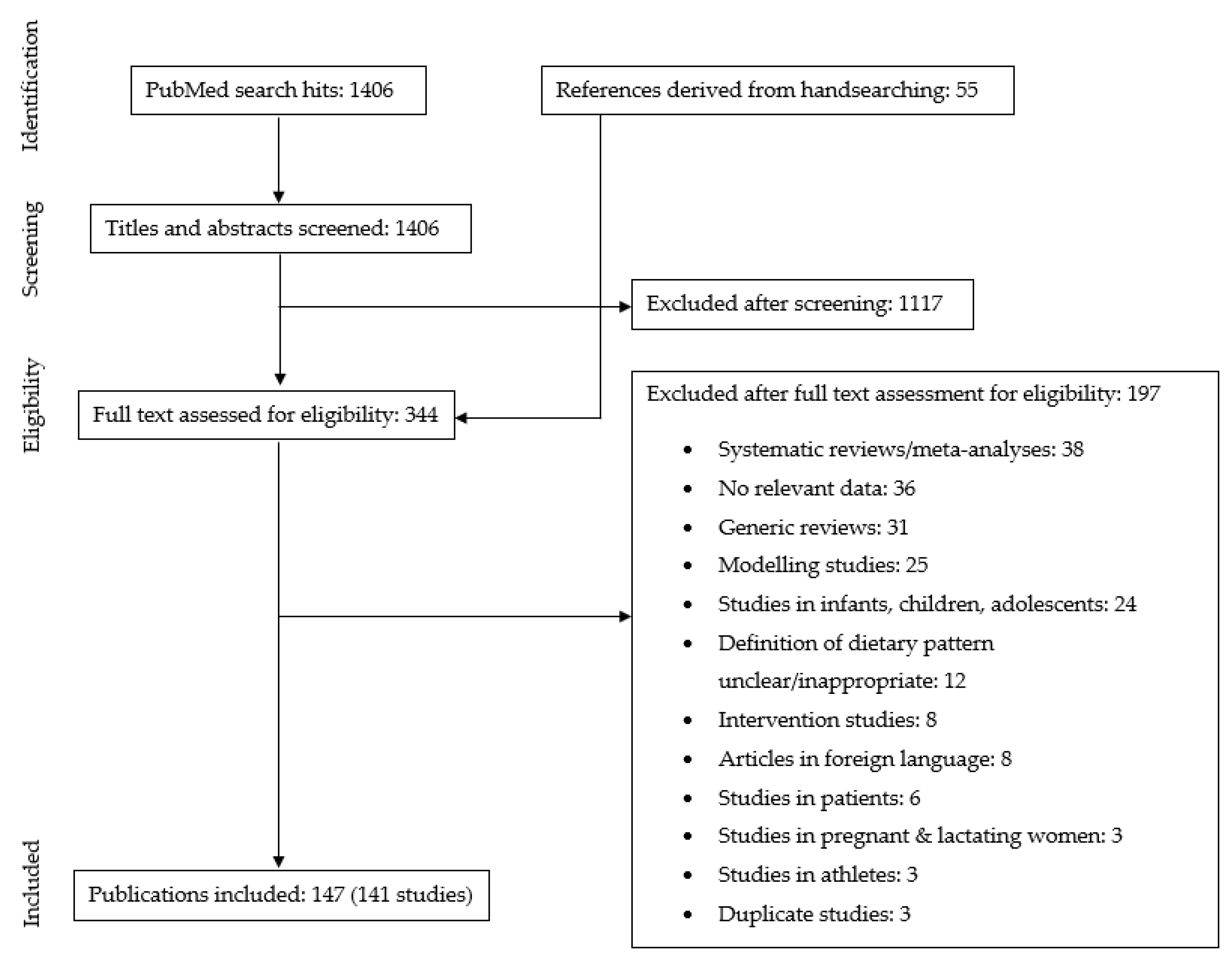
2. Materials and Methods
2.1. Search Strategy
2.2. In- and Exclusion Criteria
- Type of studies: Observational studies and intervention studies (baseline data only), that compared nutrient intake and/or status of subjects following a predominantly plant-based diet with subjects following a conventional diet with meat were included. In addition, studies that reported only on subjects following a predominantly plant-based diet were also included. Generic reviews, case studies, and articles not published in English language were excluded;
- Diets: To be included in our review, studies had to report on voluntary self-selected diets with a primary focus on reducing animal food intake. Studies reporting on imposed or predesigned plant-based diets (e.g., marginal plant-based staple diets in developing countries, a prescribed vegetarian diet intervention, or modelled vegetarian diet scenario) were excluded, as well as articles on overly restrictive plant-based diets (e.g., raw food diet, macrobiotic diet), or healthy diets designed to lower non-communicable diseases (e.g., DASH diet, Mediterranean diet);
- Outcome parameters: Included studies provided data on either one or more of the following parameters: dietary intake of energy, protein, poly-unsaturated fatty acids (PUFA), α-linolenic acid (ALA), eicosapentanoic acid (EPA); docosahexanoic acid (DHA); dietary intake or nutritional status of micronutrients; bone markers;
- Study population: Generally healthy adult populations of 18 years and older. We excluded studies conducted in pregnant and lactating women, populations with specific diseases or in athletes;
2.3. Data Extraction
2.4. Data Handling
- Vegan: consuming meat, fish, dairy and eggs not at all/not during the days of dietary assessment OR ≤ once per month OR self-defined vegans;
- Vegetarian:consuming meat and fish not at all/not during the days of dietary assessment OR ≤ once per month OR self-defined vegetarians;
- Pesco-vegetarian: consuming meat not at all/not during the days of dietary assessment OR ≤ once per month OR self-defined;
- Semi-vegetarian: consuming meat (and fish) ≤ once per week but > once per month OR consuming meat (and fish) “seldom”/”occasionally”;
- Meat eating: consuming meat > once per week OR self-defined.
2.5. Data Analysis
3. Results
3.1. Energy, Protein, Fiber and Fatty Acids
3.1.1. Energy
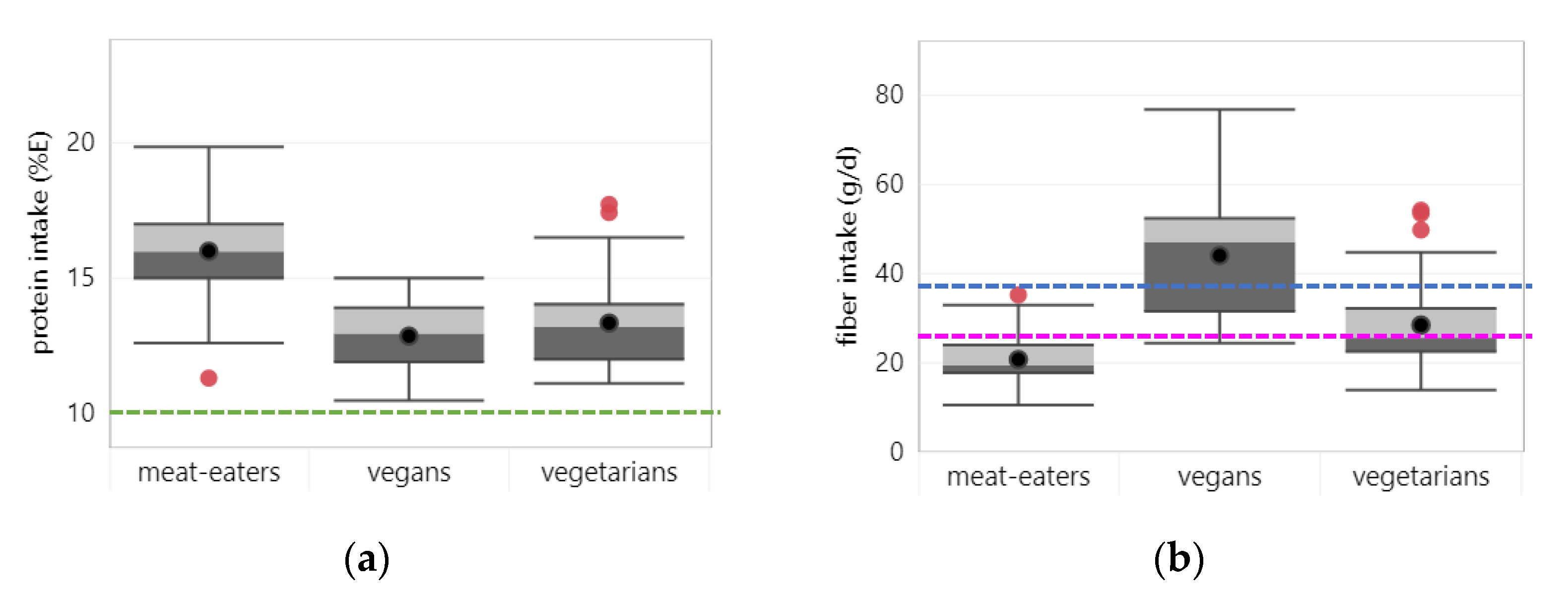
3.1.2. Protein
3.1.3. Fiber
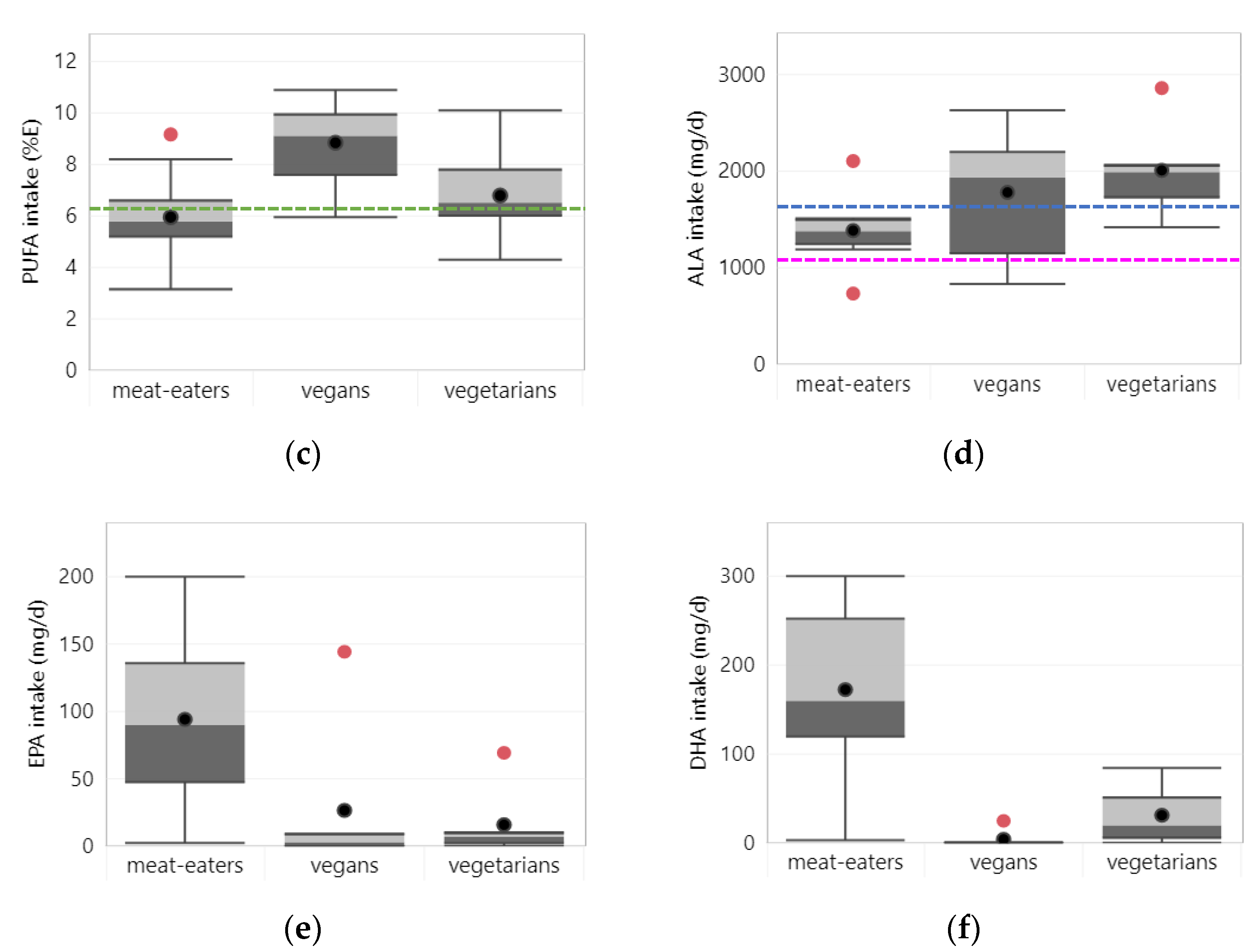
3.1.4. PUFA
3.1.5. N-3 Fatty Acids
3.2. Micronutrients
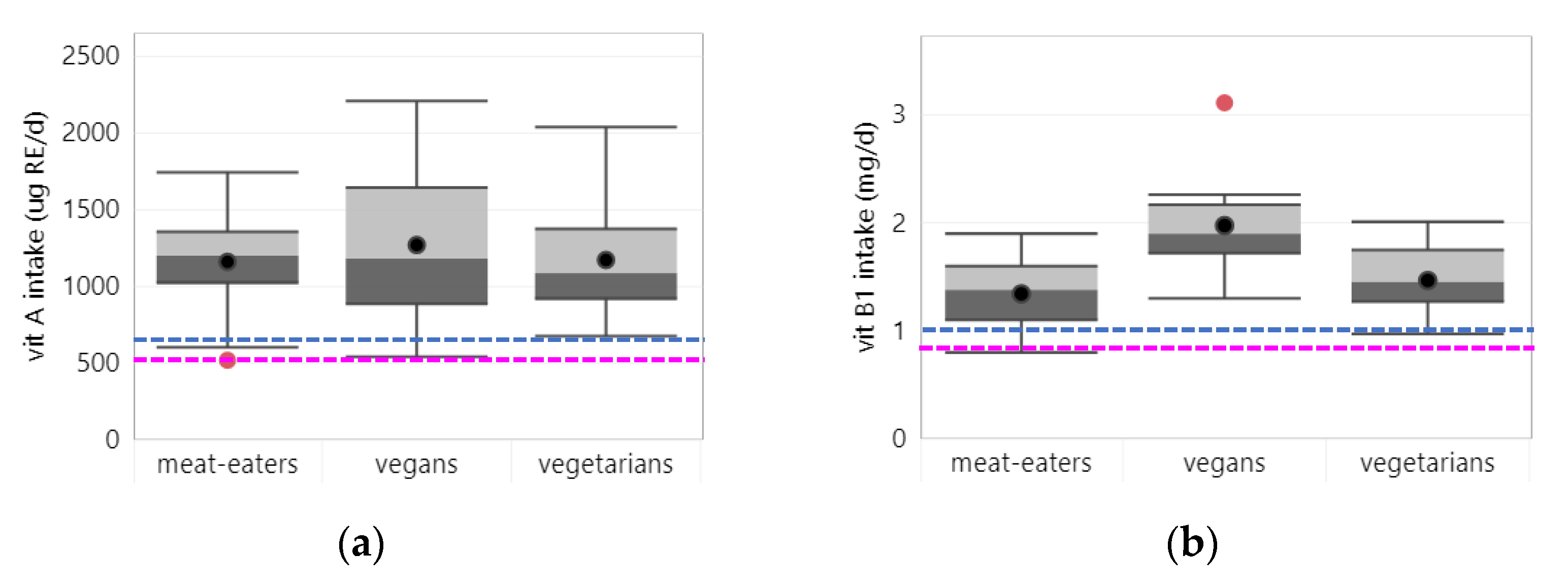
3.2.1. Vitamin A
3.2.2. Vitamin B1
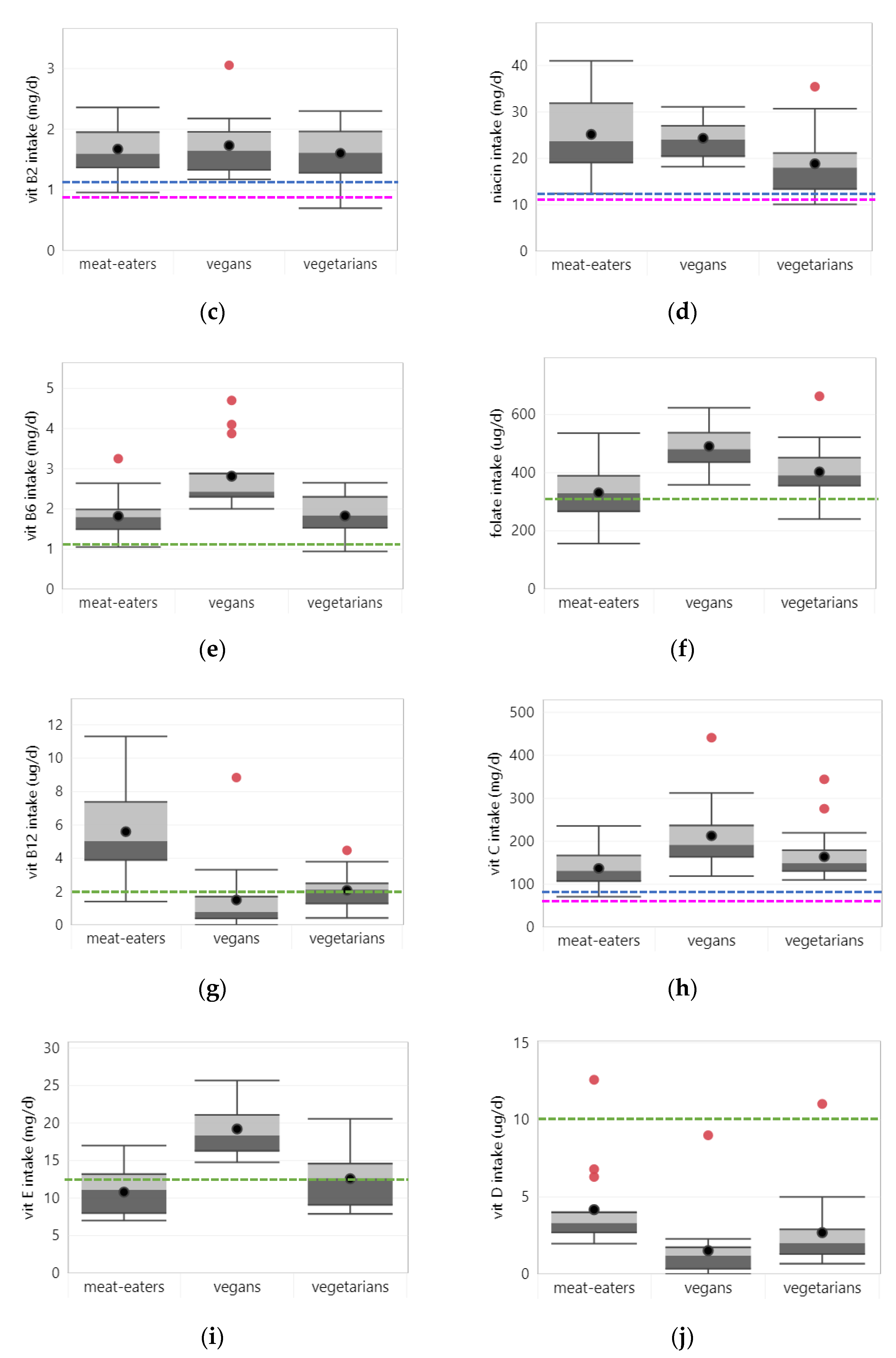
3.2.3. Vitamin B2
3.2.4. Niacin
3.2.5. Vitamin B6
3.2.6. Folate
3.2.7. Vitamin B12
3.2.8. Vitamin C
3.2.9. Vitamin D
3.2.10. Vitamin E
3.3. Minerals
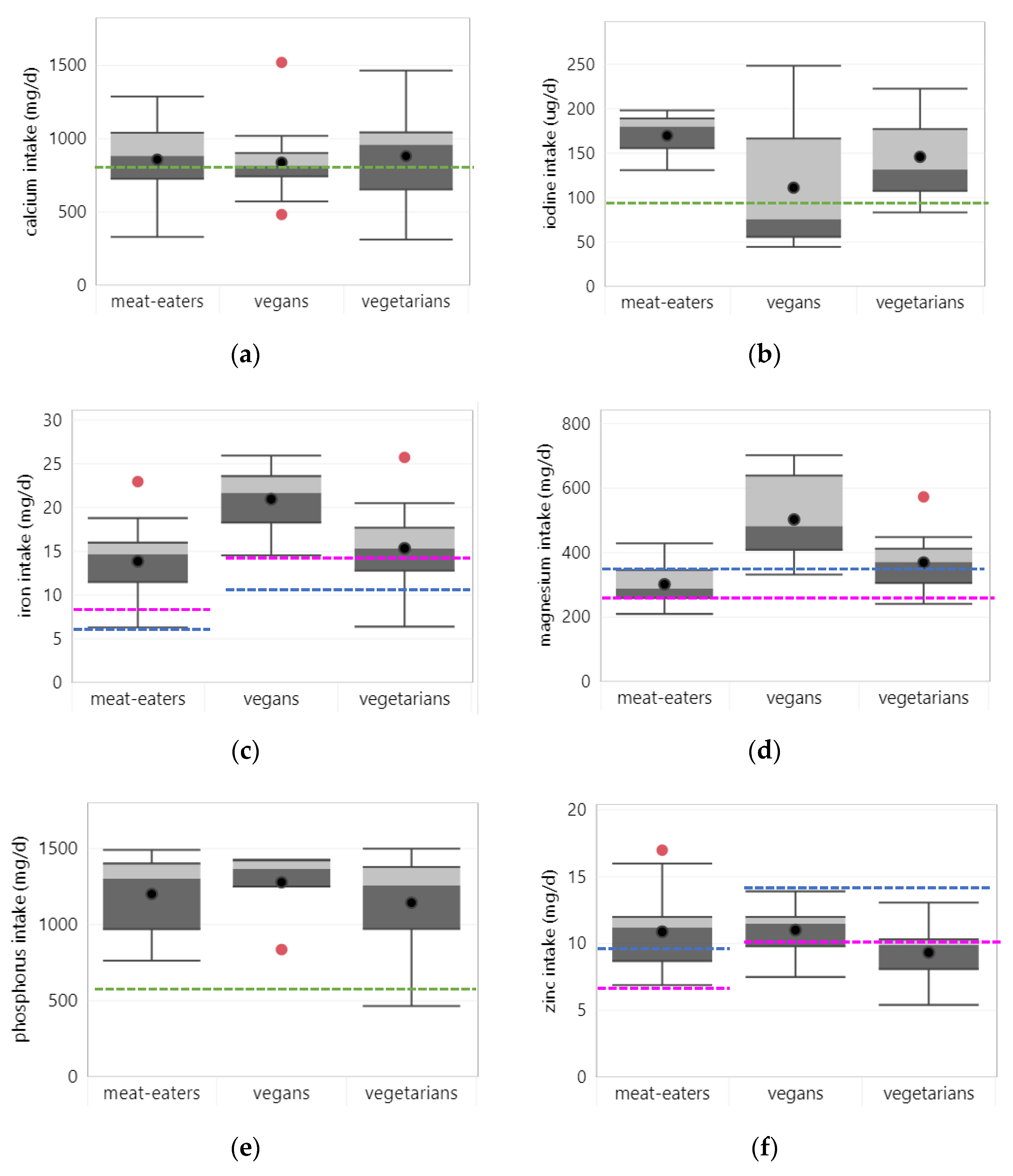
3.3.1. Calcium
3.3.2. Iodine
3.3.3. Iron
3.3.4. Magnesium
3.3.5. Phosphorus
3.3.6. Zinc
4. Discussion
4.1. Main Findings and Their Significance
4.2. Strenghts and Limitations of This Review
4.3. Findings on Energy and Macronutrients
4.4. Findings on Micronutrients
4.5. Implications for Public Health and Recommendations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Initiatives, D. Global Nutrition Report: Action on Equity to End Malnutrition; Development Initiatives Poverty Research Ltd.: Bristol, UK, 2020. [Google Scholar]
- GBD 2019 Risk Factors Collaborators. Global burden of 87 risk factors in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1223–1249. [Google Scholar] [CrossRef]
- United Nations. Sustainable Development—The 17 Goals. Available online: https://sdgs.un.org/goals (accessed on 24 August 2021).
- Willett, W.; Rockström, J.; Loken, B.; Springmann, M.; Lang, T.; Vermeulen, S.; Garnett, T.; Tilman, D.; DeClerck, F.; Wood, A.; et al. Food in the Anthropocene: The EAT–Lancet Commission on healthy diets from sustainable food systems. Lancet 2019, 393, 447–492. [Google Scholar] [CrossRef]
- FAO/WHO. Sustainable Healthy Diets—Guiding Principles; World Health Organization and Food Agriculture Organization of the United Nations: Rome, Italy, 2019. [Google Scholar]
- Springmann, M.; Wiebe, K.; Mason-D’Croz, D.; Sulser, T.B.; Rayner, M.; Scarborough, P. Health and nutritional aspects of sustainable diet strategies and their association with environmental impacts: A global modelling analysis with country-level detail. Lancet Planet Health 2018, 2, e451–e461. [Google Scholar] [CrossRef]
- GBD 2017 Diet Collaborators. Health effects of dietary risks in 195 countries, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2019, 393, 1958–1972. [Google Scholar] [CrossRef]
- Agnoli, C.; Baroni, L.; Bertini, I.; Ciappellano, S.; Fabbri, A.; Papa, M.; Pellegrini, N.; Sbarbati, R.; Scarino, M.L.; Siani, V.; et al. Position paper on vegetarian diets from the working group of the Italian Society of Human Nutrition. Nutr. Metab. Cardiovasc. Dis. NMCD 2017, 27, 1037–1052. [Google Scholar] [CrossRef]
- Haider, L.M.; Schwingshackl, L.; Hoffmann, G.; Ekmekcioglu, C. The effect of vegetarian diets on iron status in adults: A systematic review and meta-analysis. Crit. Rev. Food Sci. Nutr. 2018, 58, 1359–1374. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, G.; Laganà, A.S.; Rapisarda, A.M.C.; La Ferrera, G.M.G.; Buscema, M.; Rossetti, P.; Nigro, A.; Muscia, V.; Valenti, G.; Sapia, F.; et al. Vitamin B12 among Vegetarians: Status, Assessment and Supplementation. Nutrients 2016, 8, 767. [Google Scholar] [CrossRef] [PubMed]
- Pawlak, R. Is vitamin B12 deficiency a risk factor for cardiovascular disease in vegetarians? Am. J. Prev. Med. 2015, 48, e11–e26. [Google Scholar] [CrossRef]
- Pawlak, R.; Parrott, S.J.; Raj, S.; Cullum-Dugan, D.; Lucus, D. How prevalent is vitamin B(12) deficiency among vegetarians? Nutr. Rev. 2013, 71, 110–117. [Google Scholar] [CrossRef]
- Saunders, A.V.; Craig, W.J.; Baines, S.K. Zinc and vegetarian diets. Med. J. Aust. 2013, 199, S17–S21. [Google Scholar] [CrossRef]
- Foster, M.; Samman, S. Vegetarian diets across the lifecycle: Impact on zinc intake and status. Adv. Food Nutr. Res. 2015, 74, 93–131. [Google Scholar] [CrossRef] [PubMed]
- Saunders, A.V.; Davis, B.C.; Garg, M.L. Omega-3 polyunsaturated fatty acids and vegetarian diets. Med. J. Aust. 2013, 199, S22–S26. [Google Scholar] [CrossRef]
- Sanders, T.A.B. Plant compared with marine n-3 fatty acid effects on cardiovascular risk factors and outcomes: What is the verdict? Am. J. Clin. Nutr. 2014, 100 (Suppl. 1), 453S–458S. [Google Scholar] [CrossRef]
- Wan, X.; Wang, W.; Jiming, L.; Tiejun, T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med. Res. Methodol. 2014, 14, 135. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Li, T.; Deeks, J.J. Obtaining standard deviations from standard errors and confidence intervals for group means. In Cochrane Handbook for Systematic Reviews of Interventions; Version 6.1th (updated September 2020); Higgins, J.P.T., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M.J., Welch, V.A., Eds.; Cochrane, 2020; Available online: www.training.cochrane.org/handbook (accessed on 11 November 2021).
- IOM. Dietary Reference Intakes. The Essential Guide to Nutrient Requirements; Instititute of Medicine: Washington, DC, USA, 2006. [Google Scholar]
- IOM. Dietary Reference Intakes for Calcium and Vitamin D; Institute of Medicine: Washington, DC, USA, 2011. [Google Scholar]
- Rizzo, N.S.; Jaceldo-Siegl, K.; Sabate, J.; Fraser, G.E. Nutrient profiles of vegetarian and nonvegetarian dietary patterns. J. Acad. Nutr. Diet. 2013, 113, 1610–1619. [Google Scholar] [CrossRef]
- Bedford, J.L.; Barr, S.I. Diets and selected lifestyle practices of self-defined adult vegetarians from a population-based sample suggest they are more ‘health conscious’. Int. J. Behav. Nutr. Phys. Act. 2005, 2, 4. [Google Scholar] [CrossRef] [PubMed]
- Nebl, J.; Schuchardt, J.P.; Ströhle, A.; Wasserfurth, P.; Haufe, S.; Eigendorf, J.; Tegtbur, U.; Hahn, A. Micronutrient Status of Recreational Runners with Vegetarian or Non-Vegetarian Dietary Patterns. Nutrients 2019, 11, 1146. [Google Scholar] [CrossRef]
- Johnston, C.S.; Bliss, C.; Knurick, J.R.; Scholtz, C. Rapid Eating Assessment for Participants [shortened version] scores are associated with Healthy Eating Index-2010 scores and other indices of diet quality in healthy adult omnivores and vegetarians. Nutr. J. 2018, 17, 89. [Google Scholar] [CrossRef]
- Lightowler, H.J.; Davies, G.J. Micronutrient intakes in a group of UK vegans and the contribution of self-selected dietary supplements. J. R. Soc. Promot. Health 2000, 120, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Elorinne, A.-L.; Alfthan, G.; Erlund, I.; Kivimäki, H.; Paju, A.; Salminen, I.; Turpeinen, U.; Voutilainen, S.; Laakso, J. Food and Nutrient Intake and Nutritional Status of Finnish Vegans and Non-Vegetarians. PLoS ONE 2016, 11, e0148235. [Google Scholar] [CrossRef]
- Kazimírová, A.; Barancoková, M.; Krajcovicová-Kudlácková, M.; Volkovová, K.; Staruchová, M.; Valachovicová, M.; Pauková, V.; Blazícek, P.; Wsólová, L.; Dusinská, M. The relationship between micronuclei in human lymphocytes and selected micronutrients in vegetarians and non-vegetarians. Mutat. Res. 2006, 611, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Krajcovicova-Kudlackova, M.; Babinska, K.; Blazicek, P.; Valachovicova, M.; Spustova, V.; Mislanova, C.; Paukova, V. Selected biomarkers of age-related diseases in older subjects with different nutrition. Bratisl. Lek. Listy 2011, 112, 610–613. [Google Scholar]
- Li, D.; Sinclair, A.J.; Mann, N.J.; Turner, A.; Ball, M.J. Selected micronutrient intake and status in men with differing meat intakes, vegetarians and vegans. Asian Pac. J. Clin. Nutr. 2000, 9, 18–23. [Google Scholar] [CrossRef]
- Schüpbach, R.; Wegmüller, R.; Berguerand, C.; Bui, M.; Herter-Aeberli, I. Micronutrient status and intake in omnivores, vegetarians and vegans in Switzerland. Eur. J. Nutr. 2017, 56, 283–293. [Google Scholar] [CrossRef]
- Somannavar, M.S.; Kodliwadmath, M.V. Correlation between oxidative stress and antioxidant defence in South Indian urban vegetarians and non-vegetarians. Eur. Rev. Med. Pharmacol. Sci. 2012, 16, 351–354. [Google Scholar]
- Hung, C.-J.; Huang, P.-C.; Lu, S.-C.; Li, Y.-H.; Huang, H.-B.; Lin, B.-F.; Chang, S.-J.; Chou, H.-F. Plasma homocysteine levels in Taiwanese vegetarians are higher than those of omnivores. J. Nutr. 2002, 132, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Nakamoto, K.; Watanabe, S.; Kudo, H.; Tanaka, A. Nutritional characteristics of middle-aged Japanese vegetarians. J. Atheroscler. Thromb. 2008, 15, 122–129. [Google Scholar] [CrossRef]
- Karabudak, E.; Kiziltan, G.; Cigerim, N. A comparison of some of the cardiovascular risk factors in vegetarian and omnivorous Turkish females. J. Hum. Nutr. Diet. Off. J. Br. Diet. Assoc. 2008, 21, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Majchrzak, D.; Singer, I.; Männer, M.; Rust, P.; Genser, D.; Wagner, K.H.; Elmadfa, I. B-vitamin status and concentrations of homocysteine in Austrian omnivores, vegetarians and vegans. Ann. Nutr. Metab. 2006, 50, 485–491. [Google Scholar] [CrossRef]
- Yen, C.-E.; Yen, C.-H.; Huang, M.-C.; Cheng, C.-H.; Huang, Y.-C. Dietary intake and nutritional status of vegetarian and omnivorous preschool children and their parents in Taiwan. Nutr. Res. 2008, 28, 430–436. [Google Scholar] [CrossRef]
- Huang, Y.-C.; Chang, S.-J.; Chiu, Y.-T.; Chang, H.-H.; Cheng, C.-H. The status of plasma homocysteine and related B-vitamins in healthy young vegetarians and nonvegetarians. Eur. J. Nutr. 2003, 42, 84–90. [Google Scholar] [CrossRef]
- Herrmann, W.; Schorr, H.; Obeid, R.; Geisel, J. Vitamin B-12 status, particularly holotranscobalamin II and methylmalonic acid concentrations, and hyperhomocysteinemia in vegetarians. Am. J. Clin. Nutr. 2003, 78, 131–136. [Google Scholar] [CrossRef]
- Krajcovicova-Kudlackova, M.; Blazicek, P.; Mislanova, C.; Valachovicova, M.; Paukova, V.; Spustova, V. Nutritional determinants of plasma homocysteine. Bratisl. Lek. Listy 2007, 108, 510–515. [Google Scholar] [PubMed]
- Allen, L.H. Causes of vitamin B12 and folate deficiency. Food Nutr. Bull. 2008, 29, S20–S33. [Google Scholar] [CrossRef] [PubMed]
- Kwok, T.; Cheng, G.; Woo, J.; Lai, W.K.; Pang, C.P. Independent effect of vitamin B12 deficiency on hematological status in older Chinese vegetarian women. Am. J. Hematol. 2002, 70, 186–190. [Google Scholar] [CrossRef] [PubMed]
- Gallego-Narbón, A.; Zapatera, B.; Barrios, L.; Vaquero, M.P. Vitamin B(12) and folate status in Spanish lacto-ovo vegetarians and vegans. J. Nutr. Sci. 2019, 8, e7. [Google Scholar] [CrossRef] [PubMed]
- Gilsing, A.M.J.; Crowe, F.L.; Lloyd-Wright, Z.; Sanders, T.A.B.; Appleby, P.N.; Allen, N.E.; Key, T.J. Serum concentrations of vitamin B12 and folate in British male omnivores, vegetarians and vegans: Results from a cross-sectional analysis of the EPIC-Oxford cohort study. Eur. J. Clin. Nutr. 2010, 64, 933–939. [Google Scholar] [CrossRef] [PubMed]
- Kwok, T.; Chook, P.; Qiao, M.; Tam, L.; Poon, Y.K.P.; Ahuja, A.T.; Woo, J.; Celermajer, D.S.; Woo, K.S. Vitamin B-12 supplementation improves arterial function in vegetarians with subnormal vitamin B-12 status. J. Nutr. Health Aging 2012, 16, 569–573. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Donoso, C.; Martínez-González, M.Á.; Martínez, J.A.; Gea, A.; Sanz-Serrano, J.; Perez-Cueto, F.J.A.; Bes-Rastrollo, M. A Provegetarian Food Pattern Emphasizing Preference for Healthy Plant-Derived Foods Reduces the Risk of Overweight/Obesity in the SUN Cohort. Nutrients 2019, 11, 1553. [Google Scholar] [CrossRef]
- Outila, T.A.; Karkkainen, M.U.; Seppanen, R.H.; Lamberg-Allardt, C.J. Dietary intake of vitamin D in premenopausal, healthy vegans was insufficient to maintain concentrations of serum 25-hydroxyvitamin D and intact parathyroid hormone within normal ranges during the winter in Finland. J. Am. Diet. Assoc. 2000, 100, 434–441. [Google Scholar] [CrossRef]
- Chan, J.; Jaceldo-Siegl, K.; Fraser, G.E. Serum 25-hydroxyvitamin D status of vegetarians, partial vegetarians, and nonvegetarians: The Adventist Health Study-2. Am. J. Clin. Nutr. 2009, 89, 1686S–1692S. [Google Scholar] [CrossRef]
- Krajcovicova-Kudlackova, M.; Simoncic, R.; Bederova, A.; Klvanova, J.; Magalova, T.; Grancicova, E.; Brtkova, A. Nutritional risk factors of a vegetarian diet in adult lacto-ovo vegetarians. Bratisl. Lek. Listy 2000, 101, 38–43. [Google Scholar] [PubMed]
- Krivosíková, Z.; Krajcovicová-Kudlácková, M.; Spustová, V.; Stefíková, K.; Valachovicová, M.; Blazícek, P.; Nĕmcová, T. The association between high plasma homocysteine levels and lower bone mineral density in Slovak women: The impact of vegetarian diet. Eur. J. Nutr. 2010, 49, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, W.; Obeid, R.; Schorr, H.; Hubner, U.; Geisel, J.; Sand-Hill, M.; Ali, N.; Herrmann, M. Enhanced bone metabolism in vegetarians--the role of vitamin B12 deficiency. Clin. Chem. Lab. Med. 2009, 47, 1381–1387. [Google Scholar] [CrossRef]
- Hansen, T.H.; Madsen, M.T.B.; Jørgensen, N.R.; Cohen, A.S.; Hansen, T.; Vestergaard, H.; Pedersen, O.; Allin, K.H. Bone turnover, calcium homeostasis, and vitamin D status in Danish vegans. Eur. J. Clin. Nutr. 2018, 72, 1046–1054. [Google Scholar] [CrossRef] [PubMed]
- Waldmann, A.; Koschizke, J.W.; Leitzmann, C.; Hahn, A. Dietary intakes and lifestyle factors of a vegan population in Germany: Results from the German Vegan Study. Eur. J. Clin. Nutr. 2003, 57, 947–955. [Google Scholar] [CrossRef]
- Brantsæter, A.L.; Knutsen, H.K.; Johansen, N.C.; Nyheim, K.A.; Erlund, I.; Meltzer, H.M.; Henjum, S. Inadequate Iodine Intake in Population Groups Defined by Age, Life Stage and Vegetarian Dietary Practice in a Norwegian Convenience Sample. Nutrients 2018, 10, 230. [Google Scholar] [CrossRef]
- Kristensen, N.B.; Madsen, M.L.; Hansen, T.H.; Allin, K.H.; Hoppe, C.; Fagt, S.; Lausten, M.S.; Gøbel, R.J.; Vestergaard, H.; Hansen, T.; et al. Intake of macro- and micronutrients in Danish vegans. Nutr. J. 2015, 14, 115. [Google Scholar] [CrossRef]
- He, J.; Shen, X.; Fang, A.; Song, J.; Li, H.; Guo, M.; Li, K. Association between predominantly plant-based diets and iron status in Chinese adults: A cross-sectional analysis. Br. J. Nutr. 2016, 116, 1621–1632. [Google Scholar] [CrossRef]
- Ho-Pham, L.T.; Nguyen, P.L.T.; Le, T.T.T.; Doan, T.A.T.; Tran, N.T.; Le, T.A.; Nguyen, T.V. Veganism, bone mineral density, and body composition: A study in Buddhist nuns. Osteoporos. Int. 2009, 20, 2087–2093. [Google Scholar] [CrossRef]
- Deriemaeker, P.; Aerenhouts, D.; De Ridder, D.; Hebbelinck, M.; Clarys, P. Health aspects, nutrition and physical characteristics in matched samples of institutionalized vegetarian and non-vegetarian elderly (>65 yrs). Nutr. Metab. 2011, 8, 37. [Google Scholar] [CrossRef] [PubMed]
- Dyett, P.A.; Sabaté, J.; Haddad, E.; Rajaram, S.; Shavlik, D. Vegan lifestyle behaviors: An exploration of congruence with health-related beliefs and assessed health indices. Appetite 2013, 67, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Murphy, S.P.; Poos, M.I. Dietary Reference Intakes: Summary of applications in dietary assessment. Public Health Nutr. 2002, 5, 843–849. [Google Scholar] [CrossRef] [PubMed]
- WHO/FAO/UNU. Protein and Amino Acid Requirements in Human Nutrition. Report of a Joint WHO/FAO/UNU Expert Consultation; WHO: Geneva, Switzerland, 2007. [Google Scholar]
- Venti, C.A.; Johnston, C.S. Modified Food Guide Pyramid for Lactovegetarians and Vegans. J. Nutr. 2002, 132, 1050–1054. [Google Scholar] [CrossRef] [PubMed]
- Kniskern, M.A.; Johnston, C.S. Protein dietary reference intakes may be inadequate for vegetarians if low amounts of animal protein are consumed. Nutrition 2011, 27, 727–730. [Google Scholar] [CrossRef]
- IOM. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids (Macronutrients); Institute of Medicine: Washington, DC, USA, 2005. [Google Scholar]
- Millward, D.J.; Jackson, A.A. Protein/energy ratios of current diets in developed and developing countries compared with a safe protein/energy ratio: Implications for recommended protein and amino acid intakes. Public Health Nutr. 2004, 7, 387–405. [Google Scholar] [CrossRef]
- Paddon-Jones, D.; Campbell, W.W.; Jacques, P.F.; Kritchevsky, S.B.; Moore, L.L.; Rodriguez, N.R.; van Loon, L.J. Protein and healthy aging. Am. J. Clin. Nutr. 2015, 101, 1339S–1345S. [Google Scholar] [CrossRef] [PubMed]
- Association, A.D. Position of the American Dietetic Association: Vegetarian Diets. J. Am. Diet. Assoc. 2009, 109, 1266–1282. [Google Scholar] [CrossRef]
- Dawczynski, C.; Schubert, R.; Jahreis, G. Amino acids, fatty acids, and dietary fibre in edible seaweed products. Food Chem. 2007, 103, 891–899. [Google Scholar] [CrossRef]
- TOKUS¸OGLU, Ö.; ÜNAL, M.K. Biomass Nutrient Profiles of Three Microalgae: Spirulina platensis, Chlorella vulgaris, and Isochrisis galbana. Food Chem. Toxicol. 2003, 68, 1144–1148. [Google Scholar]
- Pawlak, R.; Lester, S.E.; Babatunde, T. The prevalence of cobalamin deficiency among vegetarians assessed by serum vitamin B12: A review of literature. Eur. J. Clin. Nutr. 2014, 68, 541–548. [Google Scholar] [CrossRef]
- Phillips, F. Briefing Paper Vegetarian nutrition. British Nutrition Foundation, London, UK. Nutr. Bull. 2005, 30, 132–167. [Google Scholar] [CrossRef]
- Messina, V.; Melina, V.; Mangels, A.R. A new food guide for North American vegetarians. J. Am. Diet. Assoc. 2003, 103, 771–775. [Google Scholar] [CrossRef] [PubMed]
- Spiro, A.; Buttriss, J.L. Vitamin D: An overview of vitamin D status and intake in Europe. Nutr. Bull. 2014, 39, 322–350. [Google Scholar] [CrossRef] [PubMed]
- Bonjour, J.P.; Kohrt, W.; Levasseur, R.; Warren, M.; Whiting, S.; Kraenzlin, M. Biochemical markers for assessment of calcium economy and bone metabolism: Application in clinical trials from pharmaceutical agents to nutritional products. Nutr. Res. Rev. 2014, 27, 252–267. [Google Scholar] [CrossRef]
- Iguacel, I.; Miguel-Berges, M.L.; Gomez-Bruton, A.; Moreno, L.A.; Julian, C. Veganism, vegetarianism, bone mineral density, and fracture risk: A systematic review and meta-analysis. Nutr. Rev. 2019, 77, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Li, Y.; Wu, S. Comparison of human bone mineral densities in subjects on plant-based and omnivorous diets: A systematic review and meta-analysis. Arch Osteoporos. 2021, 16, 95. [Google Scholar] [CrossRef]
- Pawlak, R.; Bell, K. Iron Status of Vegetarian Children: A Review of Literature. Ann. Nutr. Metab. 2017, 70, 88–99. [Google Scholar] [CrossRef]
- Hurrell, R.; Egli, I. Iron bioavailability and dietary reference values. Am. J. Clin. Nutr. 2010, 91, 1461S–1467S. [Google Scholar] [CrossRef]
- Foster, M.; Chu, A.; Petocz, P.; Samman, S. Effect of vegetarian diets on zinc status: A systematic review and meta-analysis of studies in humans. J. Sci. Food Agric. 2013, 93, 2362–2371. [Google Scholar] [CrossRef]
- Richter, M.; Boeing, H.; Grunewald-Funk, D.; Heseker, H.; Kroke, A.; Leschik-Bonnet, E.; Oberritter, H.; Strohm, D.; Watzl, B. Vegan diet. Position of the German Nutrition Society (DGE). Ernahr. Umsch. 2016, 63, 92–102. [Google Scholar] [CrossRef]
- Teas, J.; Pino, S.; Critchley, A.; Braverman, L.E. Variability of iodine content in common commercially available edible seaweeds. Thyroid 2004, 14, 836–841. [Google Scholar] [CrossRef]
- Yeh, T.S.; Hung, N.H.; Lin, T.C. Analysis of iodine content in seaweed by GC-ECD and estimation of iodine intake. J. Food Drug Anal. 2014, 22, 189–196. [Google Scholar] [CrossRef]
- Haldimann, M.; Alt, A.; Blanc, A.; Blondeau, K. Iodine content of food groups. J. Food Compos. Anal. 2005, 18, 461–471. [Google Scholar] [CrossRef]
- FAO/WHO. Vitamin and Mineral Requirements in Human Nutrition. Report of a joint FAO/WHO Expert Consultation, Bangkok, Thailand, 21–30 September 1998; World Health Organization and Food Agriculture Organization of the United Nations: Rome, Italy, 2004. [Google Scholar]
- Sobiecki, J.G.; Appleby, P.N.; Bradbury, K.E.; Key, T.J. High compliance with dietary recommendations in a cohort of meat eaters, fish eaters, vegetarians, and vegans: Results from the European Prospective Investigation into Cancer and Nutrition-Oxford study. Nutr. Res. 2016, 36, 464–477. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Number of Studies (n) |
|---|---|
| Europe | 74 (mostly Germany, UK) |
| South/East Asia | 33 (mostly Taiwan, India, China) |
| North America | 22 (mostly US) |
| Australasia | 8 |
| South America | 2 (Brazil) |
| West Asia | 2 (Israel, Jordan) |
| Women only | 27 |
| Men only | 9 |
| Older adults 1 | 11 |
| Nutrient intake, assessed from foods only | 66 |
| Nutrient intake, assessed from foods and supplements | 17 |
| Nutrient status in non-users of supplements | 44 |
| Nutrient status in users and non-users of supplements | 55 |
| Meat-eating | 101 |
| Vegetarian | 118 |
| Vegan | 63 |
| Semi-vegetarian | 7 |
| Pesco-vegetarian | 6 |
| High/medium/low animal protein intake | 2 |
| High/medium/low meat intake | 1 |
| Dietary Pattern | Nutrients at Risk of Inadequacy | Nutrients of Favorably High Intake |
|---|---|---|
| Vegans | EPA, DHA, | fiber, PUFA, ALA, |
| vitamins B12, D, | vitamins B1, B6, C, E, folate, | |
| calcium, iodine, iron (in women), zinc | magnesium | |
| Vegetarians | fiber, EPA, DHA, | PUFA, ALA, |
| vitamins B12, D, E, | vitamin C, folate, | |
| calcium, iodine, iron (in women), zinc | magnesium | |
| Meat-eaters | fiber, PUFA, ALA (in men), | protein, |
| vitamins D, E, folate, | niacin, vitamin B12, | |
| calcium, magnesium | zinc |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Neufingerl, N.; Eilander, A. Nutrient Intake and Status in Adults Consuming Plant-Based Diets Compared to Meat-Eaters: A Systematic Review. Nutrients 2022, 14, 29. https://doi.org/10.3390/nu14010029
Neufingerl N, Eilander A. Nutrient Intake and Status in Adults Consuming Plant-Based Diets Compared to Meat-Eaters: A Systematic Review. Nutrients. 2022; 14(1):29. https://doi.org/10.3390/nu14010029
Chicago/Turabian StyleNeufingerl, Nicole, and Ans Eilander. 2022. "Nutrient Intake and Status in Adults Consuming Plant-Based Diets Compared to Meat-Eaters: A Systematic Review" Nutrients 14, no. 1: 29. https://doi.org/10.3390/nu14010029
APA StyleNeufingerl, N., & Eilander, A. (2022). Nutrient Intake and Status in Adults Consuming Plant-Based Diets Compared to Meat-Eaters: A Systematic Review. Nutrients, 14(1), 29. https://doi.org/10.3390/nu14010029






