Recovery Focused Nutritional Therapy across the Continuum of Care: Learning from COVID-19
Abstract
:1. Introduction
2. Focus on the Role of Nutrition in Recovery from the First Day of Hospital Admission
2.1. Multiple Nutritional Challenges Highlight the Need for Early Individualised Nutrition Intervention
2.1.1. Risk Factors
2.1.2. Nutritional Support
| Pre-Acute Illness | Acute Illness | Recovery Phase |
|---|---|---|
Pre-existing noncommunicable disease (NCD):
Pre-existing loss of body tissue/wasting:
| Effects of acute illness:
| Recovery phase complicated by persistent symptoms:
|
| Symptom, Metabolic and Nutritional Status Factors ↓ | ||
| MALNUTRITION, LOSS OF LEAN BODY MASS AND PHYSICAL FUNCTION CONTRIBUTING TO IMPAIRED/DELAYED RECOVERY | ||
| ↑ Nutritional Intake Factors | ||
| Suboptimal dietary quality may already be a concern before onset of acute illness | Nutrient deficits accumulate during hospital stay
| Ongoing nutritional needs frequently not addressed at discharge
|
2.2. Enabling Individualised Nutritional Care during Hospitalisation
2.3. Enabling Individualised Nutritional Care in ICU
3. Ensure Continuity of Nutritional Therapy after Discharge
3.1. Challenges in Nutritional Care at Discharge
3.2. Enabling Individualised Nutritional Care in Primary Care
3.3. The Role of Muscle-Targeted Nutrition in Recovery
4. Integrate Individualised, Multi-Modal Nutritional Care across the Patient Journey
5. Learning from COVID-19
6. Research Gaps and Solutions
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Anker, M.S.; Landmesser, U.; von Haehling, S.; Butler, J.; Coats, A.J.S.; Anker, S.D. Weight loss, malnutrition, and cachexia in COVID-19: Facts and numbers. J. Cachexia Sarcopenia Muscle 2021, 12, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Paneroni, M.; Simonelli, C.; Saleri, M.; Bertacchini, L.; Venturelli, M.; Troosters, T.; Ambrosino, N.; Vitacca, M. Muscle strength and physical performance in patients without previous disabilities recovering from COVID-19 pneumonia. Am. J. Phys. Med. Rehabil. 2021, 100, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Belli, S.; Balbi, B.; Prince, I.; Cattaneo, D.; Masocco, F.; Zaccaria, S.; Bertalli, L.; Cattini, F.; Lomazzo, A.; Dal Negro, F.; et al. Low physical functioning and impaired performance of activities of daily life in COVID-19 patients who survived hospitalisation. Eur. Respir. J. 2020, 56, 2002096. [Google Scholar] [CrossRef]
- Venturelli, S.; Benatti, S.V.; Casati, M.; Binda, F.; Zuglian, G.; Imeri, G.; Conti, C.; Biffi, A.M.; Spada, M.S.; Bondi, E.; et al. Surviving COVID-19 in Bergamo province: A post-acute outpatient re-evaluation. Epidemiol. Infect. 2021, 149, e32. [Google Scholar] [CrossRef]
- Huang, C.; Huang, L.; Wang, Y.; Li, X.; Ren, L.; Gu, X.; Kang, L.; Guo, L.; Liu, M.; Zhou, X.; et al. 6-month consequences of COVID-19 in patients discharged from hospital: A cohort study. Lancet 2021, 397, 220–232. [Google Scholar] [CrossRef]
- Carfi, A.; Bernabei, R.; Landi, F.; Gemelli Against COVID-19 Post-Acute Care Study Group. Persistent Symptoms in patients after acute COVID-19. JAMA 2020, 324, 603–605. [Google Scholar] [CrossRef]
- Vaes, A.W.; Machado, F.V.C.; Meys, R.; Delbressine, J.M.; Goertz, Y.M.J.; Van Herck, M.; Houben-Wilke, S.; Franssen, F.M.E.; Vijlbrief, H.; Spies, Y.; et al. Care dependency in non-hospitalized patients with COVID-19. J. Clin. Med. 2020, 9, 2946. [Google Scholar] [CrossRef]
- Singer, P.; Blaser, A.R.; Berger, M.M.; Alhazzani, W.; Calder, P.C.; Casaer, M.P.; Hiesmayr, M.; Mayer, K.; Montejo, J.C.; Pichard, C.; et al. ESPEN guideline on clinical nutrition in the intensive care unit. Clin. Nutr. 2019, 38, 48–79. [Google Scholar] [CrossRef] [Green Version]
- Virgens, I.P.A.; Santana, N.M.; Lima, S.; Fayh, A.P.T. Can COVID-19 be a risk for cachexia for patients during intensive care? Narrative review and nutritional recommendations. Br. J. Nutr. 2020, 126, 1–9. [Google Scholar] [CrossRef]
- Morley, J.E.; Kalantar-Zadeh, K.; Anker, S.D. COVID-19: A major cause of cachexia and sarcopenia? J. Cachexia Sarcopenia Muscle 2020, 11, 863–865. [Google Scholar] [CrossRef]
- Barazzoni, R.; Bischoff, S.C.; Breda, J.; Wickramasinghe, K.; Krznaric, Z.; Nitzan, D.; Pirlich, M.; Singer, P.; Endorsed by the ESPEN Council. ESPEN expert statements and practical guidance for nutritional management of individuals with SARS-CoV-2 infection. Clin. Nutr. 2020, 39, 1631–1638. [Google Scholar] [CrossRef]
- Cederholm, T.; Jensen, G.L.; Correia, M.; Gonzalez, M.C.; Fukushima, R.; Higashiguchi, T.; Baptista, G.; Barazzoni, R.; Blaauw, R.; Coats, A.; et al. GLIM criteria for the diagnosis of malnutrition—A consensus report from the global clinical nutrition community. Clin. Nutr. 2019, 38, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Ye, L.; Yang, Z.; Liu, J.; Liao, L.; Wang, F. Digestive system manifestations and clinical significance of coronavirus disease 2019: A systematic literature review. J. Gastroenterol. Hepatol. 2021, 36, 1414–1422. [Google Scholar] [CrossRef]
- Liu, R.; Paz, M.; Siraj, L.; Boyd, T.; Salamone, S.; Lite, T.V.; Leung, K.M.; Chirinos, J.D.; Shang, H.H.; Townsend, M.J.; et al. Feeding intolerance in critically ill patients with COVID-19. Clin. Nutr. 2021, S0261-5614. [Google Scholar] [CrossRef]
- Prado, C.M.; Purcell, S.A.; Alish, C.; Pereira, S.L.; Deutz, N.E.; Heyland, D.K.; Goodpaster, B.H.; Tappenden, K.A.; Heymsfield, S.B. Implications of low muscle mass across the continuum of care: A narrative review. Ann. Med. 2018, 50, 675–693. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, A.K.; Gillies, C.L.; Singh, R.; Singh, A.; Chudasama, Y.; Coles, B.; Seidu, S.; Zaccardi, F.; Davies, M.J.; Khunti, K. Prevalence of co-morbidities and their association with mortality in patients with COVID-19: A systematic review and meta-analysis. Diabetes Obes. Metab. 2020, 22, 1915–1924. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Lu, Y.; Huang, Y.M.; Wang, M.; Ling, W.; Sui, Y.; Zhao, H.L. Obesity in patients with COVID-19: A systematic review and meta-analysis. Metab. Clin. Exp. 2020, 113, 154378. [Google Scholar] [CrossRef] [PubMed]
- Gualtieri, P.; Falcone, C.; Romano, L.; Macheda, S.; Correale, P.; Arciello, P.; Polimeni, N.; Lorenzo, A. Body composition findings by computed tomography in SARS-CoV-2 patients: Increased risk of muscle wasting in obesity. Int. J. Mol. Sci. 2020, 21, 4670. [Google Scholar] [CrossRef]
- Caccialanza, R.; Formisano, E.; Klersy, C.; Ferretti, V.; Ferrari, A.; Demontis, S.; Mascheroni, A.; Masi, S.; Crotti, S.; Lobascio, F.; et al. Nutritional parameters associated with prognosis in non-critically ill hospitalized COVID-19 patients: The NUTRI-COVID19 study. Clin. Nutr. 2021, S0261-5614. [Google Scholar] [CrossRef]
- Martin–Martinez, A.; Ortega, O.; Viñas, P.; Arreola, V.; Nascimento, W.; Costa, A.; Riera, S.A.; Alarcón, C.; Clavé, P. COVID-19 is associated with oropharyngeal dysphagia and malnutrition in hospitalized patients during the spring 2020 wave of the pandemic. Clin. Nutr. 2021. [Google Scholar] [CrossRef]
- Xydakis, M.S.; Dehgani-Mobaraki, P.; Holbrook, E.H.; Geisthoff, U.W.; Bauer, C.; Hautefort, C.; Herman, P.; Manley, G.T.; Lyon, D.M.; Hopkins, C. Smell and taste dysfunction in patients with COVID-19. Lancet Infect. Dis. 2020, 20, 1015–1016. [Google Scholar] [CrossRef]
- Vergara, J.; Lirani-Silva, C.; Brodsky, M.B.; Miles, A.; Clave, P.; Nascimento, W.; Mourao, L.F. Potential influence of olfactory, gustatory, and pharyngolaryngeal sensory dysfunctions on swallowing physiology in COVID-19. Otolaryngol.-Head Neck Surg.. 2021, 164, 1134–1135. [Google Scholar] [CrossRef]
- Schindler, A.; Baijens, L.W.J.; Clave, P.; Degen, B.; Duchac, S.; Dziewas, R.; Farneti, D.; Hamdy, S.; Michou, E.; Pokieser, P.; et al. ESSD commentary on dysphagia management during COVID pandemia. Dysphagia 2021, 36, 764–767. [Google Scholar] [CrossRef]
- Vergara, J.; Skoretz, S.A.; Brodsky, M.B.; Miles, A.; Langmore, S.E.; Wallace, S.; Seedat, J.; Starmer, H.M.; Bolton, L.; Clave, P.; et al. Assessment, diagnosis, and treatment of dysphagia in patients infected With SARS-CoV-2: A review of the literature and international guidelines. Am. J. Speech Lang. Pathol. 2020, 29, 2242–2253. [Google Scholar] [CrossRef] [PubMed]
- Cereda, E.; Guzzardella, A.; Klersy, C.; Belliato, M.; Pellegrini, A.; Sciutti, F.; Mongodi, S.; Masi, S.; Crotti, S.; Savioli, M.; et al. Early caloric deficit is associated with a higher risk of death in invasive ventilated COVID-19 patients. Clin. Nutr. 2021, S0261-5614. [Google Scholar] [CrossRef]
- Im, J.H.; Je, Y.S.; Baek, J.; Chung, M.H.; Kwon, H.Y.; Lee, J.S. Nutritional status of patients with COVID-19. Int. J. Infect. Dis. 2020, 100, 390–393. [Google Scholar] [CrossRef]
- Cereda, E.; Bogliolo, L.; de Stefano, L.; Caccialanza, R. A brief discussion of the benefit and mechanism of vitamin D supplementation on coronavirus disease 2019. Curr. Opin. Clin. Nutr. Metab. Care 2021, 24, 102–107. [Google Scholar] [CrossRef]
- Murai, I.H.; Fernandes, A.L.; Sales, L.P.; Pinto, A.J.; Goessler, K.F.; Duran, C.S.C.; Silva, C.B.R.; Franco, A.S.; Macedo, M.B.; Dalmolin, H.H.H.; et al. Effect of a single high dose of vitamin D3 on hospital length of stay in patients with moderate to severe COVID-19: A randomized clinical trial. JAMA 2021, 325, 1053–1060. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.; Patel, D.; Bittel, B.; Wolski, K.; Wang, Q.; Kumar, A.; Il’Giovine, Z.J.; Mehra, R.; McWilliams, C.; Nissen, S.E.; et al. Effect of high-dose Zinc and ascorbic acid supplementation vs usual care on symptom length and reduction among ambulatory patients with SARS-CoV-2 infection: The COVID A to Z randomized clinical trial. JAMA Netw. Open 2021, 4, e210369. [Google Scholar] [CrossRef]
- Afshin, A.; Sur, P.J.; Fay, K.A.; Cornaby, L.; Ferrara, G.; Salama, J.S.; Mullany, E.C.; Abate, K.H.; Abbafati, C.; Abebe, Z.; et al. Health effects of dietary risks in 195 countries, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2019, 393, 1958–1972. [Google Scholar] [CrossRef] [Green Version]
- Hengeveld, L.M.; Wijnhoven, H.A.H.; Olthof, M.R.; Brouwer, I.A.; Simonsick, E.M.; Kritchevsky, S.B.; Houston, D.K.; Newman, A.B.; Visser, M. Prospective associations of diet quality with incident frailty in older adults: The health, aging, and body Composition study. J. Am. Geriatr. Soc. 2019, 67, 1835–1842. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, M.; Geirsdottir, O.G.; Launer, L.J.; Gudnasson, V.; Visser, M.; Gunnarsdottir, I. A poor appetite or ability to eat and its association with physical function amongst community-dwelling older adults: Age, gene/environment susceptibility-Reykjavik study. Eur. J. Ageing 2020, 18, 405–415. [Google Scholar] [CrossRef]
- Granic, A.; Mendonca, N.; Sayer, A.A.; Hill, T.R.; Davies, K.; Adamson, A.; Siervo, M.; Mathers, J.C.; Jagger, C. Low protein intake, muscle strength and physical performance in the very old: The Newcastle 85+ Study. Clin. Nutr. 2018, 37, 2260–2270. [Google Scholar] [CrossRef] [Green Version]
- Hiesmayr, M.; Schindler, K.; Pernicka, E.; Schuh, C.; Schoeniger-Hekele, A.; Bauer, P.; Laviano, A.; Lovell, A.D.; Mouhieddine, M.; Schuetz, T.; et al. Decreased food intake is a risk factor for mortality in hospitalised patients: The NutritionDay survey 2006. Clin. Nutr. 2009, 28, 484–491. [Google Scholar] [CrossRef]
- Lambell, K.J.; Tatucu-Babet, O.A.; Chapple, L.A.; Gantner, D.; Ridley, E.J. Nutrition therapy in critical illness: A review of the literature for clinicians. Crit. Care 2020, 24, 35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peterson, S.J.; Tsai, A.A.; Scala, C.M.; Sowa, D.C.; Sheean, P.M.; Braunschweig, C.L. Adequacy of oral intake in critically ill patients 1 week after extubation. J. Am. Diet. Assoc. 2010, 110, 427–433. [Google Scholar] [CrossRef]
- Ridley, E.J.; Parke, R.L.; Davies, A.R.; Bailey, M.; Hodgson, C.; Deane, A.M.; McGuinness, S.; Cooper, D.J. What happens to nutrition intake in the post-intensive care unit hospitalization period? An observational cohort study in critically ill adults. J. Parenter. Enter. Nutr. 2019, 43, 88–95. [Google Scholar] [CrossRef]
- Chapple, L.S.; Deane, A.M.; Heyland, D.K.; Lange, K.; Kranz, A.J.; Williams, L.T.; Chapman, M.J. Energy and protein deficits throughout hospitalization in patients admitted with a traumatic brain injury. Clin. Nutr. 2016, 35, 1315–1322. [Google Scholar] [CrossRef]
- Wright, L.; Cotter, D.; Hickson, M.; Frost, G. Comparison of energy and protein intakes of older people consuming a texture modified diet with a normal hospital diet. J. Hum. Nutr. Diet. 2005, 18, 213–219. [Google Scholar] [CrossRef]
- Brooks, M.; Vest, M.T.; Shapero, M.; Papas, M. Malnourished adults’ receipt of hospital discharge nutrition care instructions: A pilot study. J. Hum. Nutr. Diet. 2019, 32, 659–666. [Google Scholar] [CrossRef] [PubMed]
- Cawood, A.L.; Walters, E.R.; Stratton, R.J.; Smith, T. Malnutrition and Nutritional Care of COVID-19 Patients in Hospitals during the First Wave of the Pandemic: A Summary of the BAPEN Hospital COVID-19 Survey of Health Care Professionals; BAPEN: 2021. Available online: https://www.bapen.org.uk/news-and-media/news/869-a-summary-of-the-bapen-hospital-covid-19-survey-of-health-care-professionals (accessed on 23 March 2021).
- Reeves, A.; White, H.; Sosnowski, K.; Tran, K.; Jones, M.; Palmer, M. Energy and protein intakes of hospitalised patients with acute respiratory failure receiving non-invasive ventilation. Clin. Nutr. 2014, 33, 1068–1073. [Google Scholar] [CrossRef]
- Chapple, L.S.; Fetterplace, K.; Asrani, V.; Burrell, A.; Cheng, A.C.; Collins, P.; Doola, R.; Ferrie, S.; Marshall, A.P.; Ridley, E.J. Nutrition management for critically and acutely unwell hospitalised patients with coronavirus disease 2019 (COVID-19) in Australia and New Zealand. Aust. Crit. Care 2020, 33, 399–406. [Google Scholar] [CrossRef]
- Caccialanza, R.; Laviano, A.; Lobascio, F.; Montagna, E.; Bruno, R.; Ludovisi, S.; Corsico, A.G.; Di Sabatino, A.; Belliato, M.; Calvi, M.; et al. Early nutritional supplementation in non-critically ill patients hospitalized for the 2019 novel coronavirus disease (COVID-19): Rationale and feasibility of a shared pragmatic protocol. Nutrition 2020, 74, 110835. [Google Scholar] [CrossRef]
- British Association for Parenteral and Enteral Nutrition, British Dietetic Association. Route of Nutrition Support in Patients Requiring NIV & CPAP during the COVID-19 Response. 2020. Available online: https://www.bapen.org.uk/pdfs/covid-19/nutrition-in-niv-21-04-20.pdf (accessed on 26 March 2021).
- Turner, P.; Montgomery, H.; Stroud, M.; Grimble, G.; Smith, T. Malnutrition risk in hospitalised COVID-19 patients receiving CPAP. Lancet 2021, 397, 1261. [Google Scholar] [CrossRef]
- van Zanten, A.R.H.; De Waele, E.; Wischmeyer, P.E. Nutrition therapy and critical illness: Practical guidance for the ICU, post-ICU, and long-term convalescence phases. Crit. Care 2019, 23, 368. [Google Scholar] [CrossRef] [Green Version]
- Berlin, D.A.; Gulick, R.M.; Martinez, F.J. Severe Covid-19. N. Engl. J. Med. 2020, 383, 2451–2460. [Google Scholar] [CrossRef] [PubMed]
- Latronico, N.; Bolton, C.F. Critical illness polyneuropathy and myopathy: A major cause of muscle weakness and paralysis. Lancet Neurol. 2011, 10, 931–941. [Google Scholar] [CrossRef]
- Martindale, R.; Patel, J.J.; Taylor, B.; Arabi, Y.M.; Warren, M.; McClave, S.A. Nutrition therapy in critically ill patients with coronavirus disease 2019. J. Parenter. Enter. Nutr. 2020, 44, 1174–1184. [Google Scholar] [CrossRef]
- Thibault, R.; Seguin, P.; Tamion, F.; Pichard, C.; Singer, P. Nutrition of the COVID-19 patient in the intensive care unit (ICU): A practical guidance. Crit. Care 2020, 24, 447. [Google Scholar] [CrossRef]
- Singer, P.; Pichard, C.; De Waele, E. Practical guidance for the use of indirect calorimetry during COVID 19 pandemic. Clin. Nutr. Exp. 2020, 33, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Whittle, J.; Molinger, J.; MacLeod, D.; Haines, K.; Wischmeyer, P.E.; LEEP-COVID Study Group. Persistent hypermetabolism and longitudinal energy expenditure in critically ill patients with COVID-19. Crit. Care 2020, 24, 581. [Google Scholar] [CrossRef] [PubMed]
- Mitch, W.E.; Goldberg, A.L. Mechanisms of muscle wasting. The role of the ubiquitin-proteasome pathway. N. Engl. J. Med. 1996, 335, 1897–1905. [Google Scholar] [CrossRef]
- Cawood, A.L.; Walters, E.R.; Smith, T.R.; Sipaul, R.H.; Stratton, R.J. A Review of nutrition support guidelines for individuals with or recovering from COVID-19 in the community. Nutrients 2020, 12, 3230. [Google Scholar] [CrossRef]
- Holdoway, A. Nutritional management of patients during and after COVID-19 illness. Br. J. Community Nurs. 2020, 25, S6–S10. [Google Scholar] [CrossRef]
- Rooney, S.; Webster, A.; Paul, L. Systematic review of changes and recovery in physical function and fitness after severe acute respiratory syndrome-related coronavirus infection: Implications for COVID-19 rehabilitation. Phys. Ther. 2020, 100, 1717–1729. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyere, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef] [Green Version]
- Bauer, J.; Biolo, G.; Cederholm, T.; Cesari, M.; Cruz-Jentoft, A.J.; Morley, J.E.; Phillips, S.; Sieber, C.; Stehle, P.; Teta, D.; et al. Evidence-based recommendations for optimal dietary protein intake in older people: A position paper from the PROT-AGE Study Group. J. Am. Med. Dir. Assoc. 2013, 14, 542–559. [Google Scholar] [CrossRef]
- Deutz, N.E.; Bauer, J.M.; Barazzoni, R.; Biolo, G.; Boirie, Y.; Bosy-Westphal, A.; Cederholm, T.; Cruz-Jentoft, A.; Krznaric, Z.; Nair, K.S.; et al. Protein intake and exercise for optimal muscle function with aging: Recommendations from the ESPEN Expert Group. Clin. Nutr. 2014, 33, 929–936. [Google Scholar] [CrossRef] [Green Version]
- Rondanelli, M.; Cereda, E.; Klersy, C.; Faliva, M.A.; Peroni, G.; Nichetti, M.; Gasparri, C.; Iannello, G.; Spadaccini, D.; Infantino, V.; et al. Improving rehabilitation in sarcopenia: A randomized-controlled trial utilizing a muscle-targeted food for special medical purposes. J. Cachexia Sarcopenia Muscle 2020, 11, 1535–1547. [Google Scholar] [CrossRef]
- Sociedad Espanola de Endocrinologia y Nutrición. Abordaje de la Desnutrición Relacionada con la Enfermedad en Pacientes Hospitalizados con COVID-19. Available online: https://fesnad.org/docs/noticias/DRE%20COVID-19%20NutriSEEN.pdf (accessed on 7 April 2021).
- Dutch Malnutrition Steering Group. Guideline on Malnutrition: Recognising, Diagnosing and Treating Malnutrition in Adults. 2017. Available online: http://www.fightmalnutrition.eu/wp-content/uploads/2017/09/Guideline-malnutrition-DMSG-2017.pdf (accessed on 7 April 2021).
- Massanet, P.L.; Petit, L.; Louart, B.; Corne, P.; Richard, C.; Preiser, J.C. Nutrition rehabilitation in the intensive care unit. J. Parenter. Enter. Nutr. 2015, 39, 391–400. [Google Scholar] [CrossRef]
- Buchholz, A.C. Weaning patients with dysphagia from tube feeding to oral nutrition: A proposed algorithm. Can. J. Diet. Pract. Res. 1998, 59, 208–214. [Google Scholar]
- McClave, S.A.; Taylor, B.E.; Martindale, R.G.; Warren, M.M.; Johnson, D.R.; Braunschweig, C.; McCarthy, M.S.; Davanos, E.; Rice, T.W.; Cresci, G.A.; et al. Guidelines for the provision and assessment of nutrition support therapy in the adult critically ill patient: Society of Critical Care Medicine (SCCM) and American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.). J. Parenter. Enter. Nutr. 2016, 40, 159–211. [Google Scholar] [CrossRef]
- Dhaliwal, R.; Cahill, N.; Lemieux, M.; Heyland, D.K. The Canadian critical care nutrition guidelines in 2013: An update on current recommendations and implementation strategies. Nutr. Clin. Pract. 2014, 29, 29–43. [Google Scholar] [CrossRef] [PubMed]
- Sioson, M.S.; Martindale, R.; Abayadeera, A.; Abouchaleh, N.; Aditianingsih, D.; Bhurayanontachai, R.; Chiou, W.C.; Higashibeppu, N.; Mat Nor, M.B.; Osland, E.; et al. Nutrition therapy for critically ill patients across the Asia-Pacific and Middle East regions: A consensus statement. Clin. Nutr. ESPEN 2018, 24, 156–164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
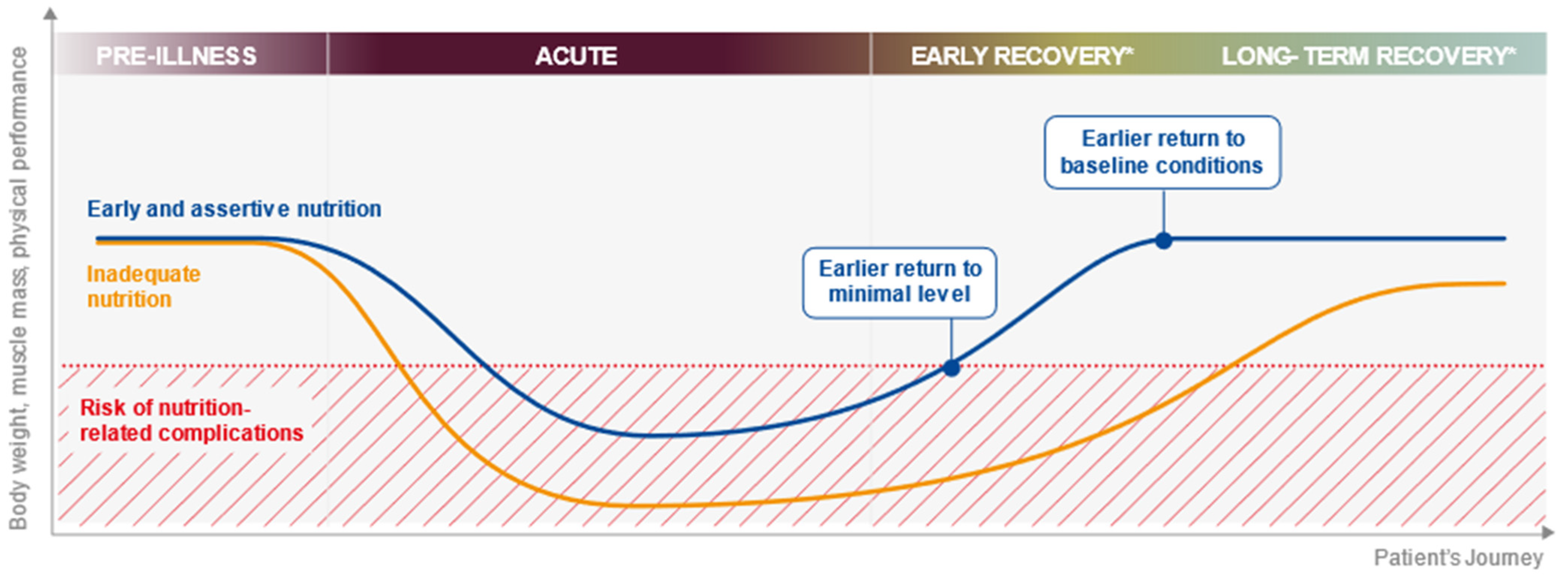
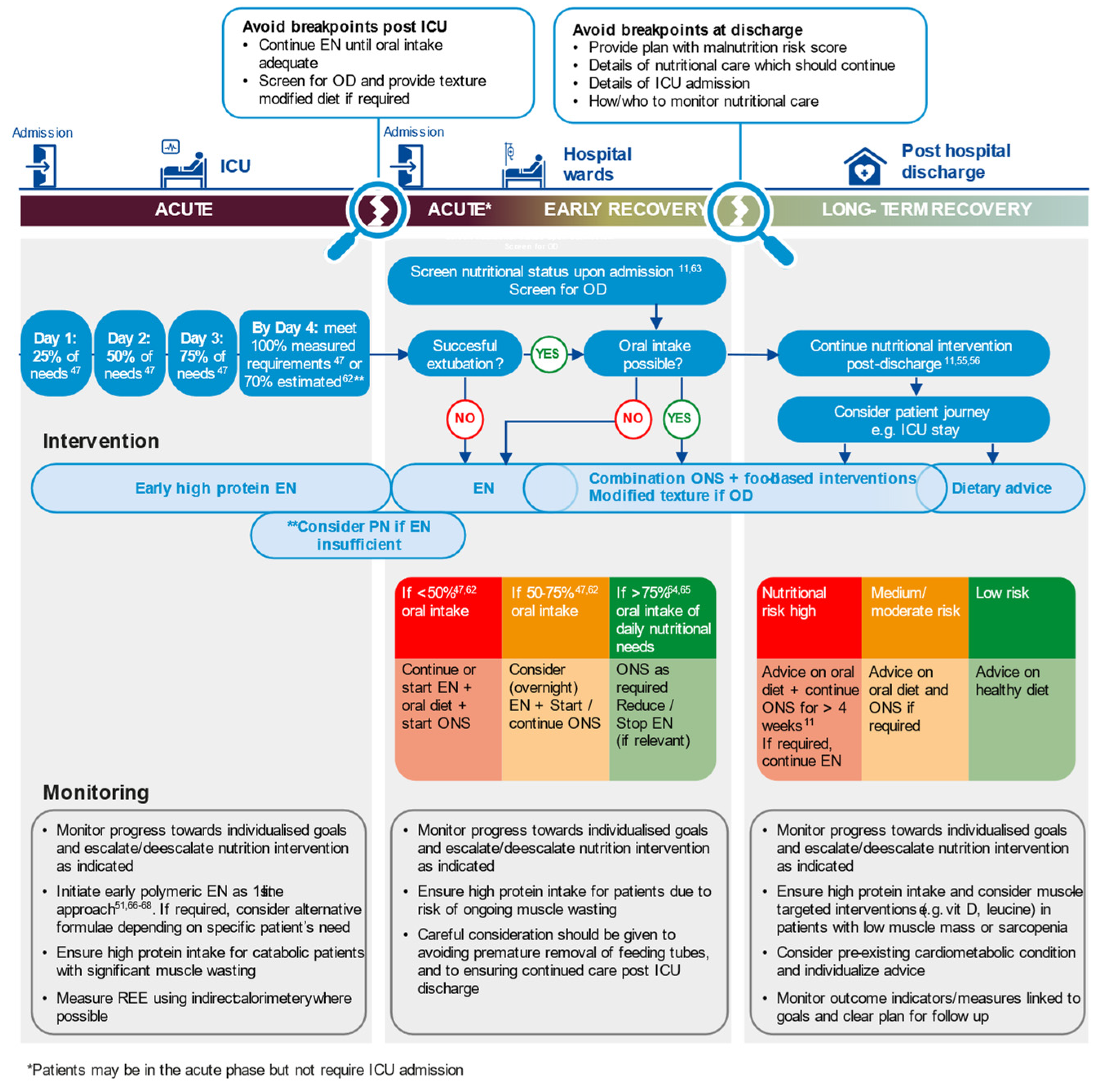
| Multi-Modal Nutritional Therapy |
|---|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cereda, E.; Clavé, P.; Collins, P.F.; Holdoway, A.; Wischmeyer, P.E. Recovery Focused Nutritional Therapy across the Continuum of Care: Learning from COVID-19. Nutrients 2021, 13, 3293. https://doi.org/10.3390/nu13093293
Cereda E, Clavé P, Collins PF, Holdoway A, Wischmeyer PE. Recovery Focused Nutritional Therapy across the Continuum of Care: Learning from COVID-19. Nutrients. 2021; 13(9):3293. https://doi.org/10.3390/nu13093293
Chicago/Turabian StyleCereda, Emanuele, Pere Clavé, Peter F. Collins, Anne Holdoway, and Paul E. Wischmeyer. 2021. "Recovery Focused Nutritional Therapy across the Continuum of Care: Learning from COVID-19" Nutrients 13, no. 9: 3293. https://doi.org/10.3390/nu13093293
APA StyleCereda, E., Clavé, P., Collins, P. F., Holdoway, A., & Wischmeyer, P. E. (2021). Recovery Focused Nutritional Therapy across the Continuum of Care: Learning from COVID-19. Nutrients, 13(9), 3293. https://doi.org/10.3390/nu13093293








