A Pilot Randomized Controlled Trial of a Partial Meal Replacement Preconception Weight Loss Program for Women with Overweight and Obesity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design
2.3. Study Visits
2.4. Partial Meal Replacement Protocol
2.5. Conventional Dietary Advice Protocol
2.6. Data Collection and Management
2.7. Sample Size
2.8. Statistical Treatment
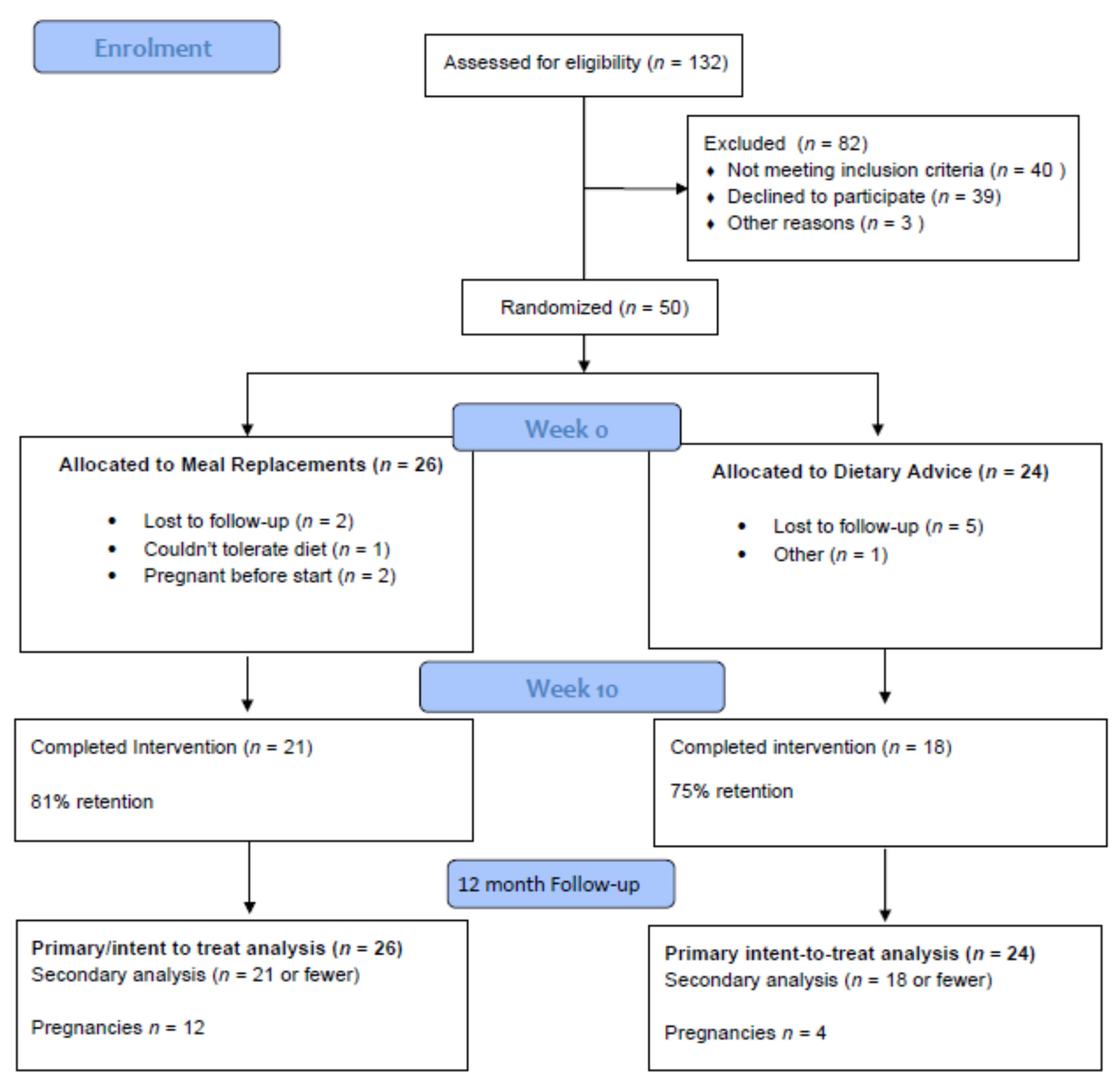
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- National Health Survey: First Results, 2017-18 (Cat. No. 4364.0.55.001). Available online: https://www.abs.gov.au/statistics/health/health-conditions-and-risks/overweight-and-obesity/2017-18 (accessed on 11 September 2021).
- Allman-Farinelli, M.A.; Chey, T.; Bauman, A.E.; Gill, T.; James, W.P.T. Age, period and birth cohort effects on prevalance of overweight and obesity in Australian adults from 1990 to 2000. Eur. J. Clin. Nutr. 2008, 62, 898–907. [Google Scholar] [CrossRef] [Green Version]
- Thrift, A.P.; Callaway, L.K. The effect of obesity on pregnancy outcomes among Australian Indigenous and non-Indigenous women. Med. J. Aust. 2014, 201, 592–595. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zaballa, K.; Liu, A.; Peek, M.J.; Mongelli, M.; Nanan, R. Association between World Health Organization categories of body mass index and relative risks for weight-related pregnancy outcomes: A retrospective cohort study. Obstet. Med. 2012, 5, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Godfrey, K.M.; Reynolds, R.M.; Prescott, S.L.; Nyirenda, M.; Jaddoe, V.W.; Eriksson, J.G.; Broekman, B.F. Influence of maternal obesity on the long-term health of offspring. Lancet Diabetes Endocrinol. 2017, 5, 53–64. [Google Scholar] [CrossRef] [Green Version]
- Astbury, N.M.; Piernas, C.; Hartmann-Boyce, J.; Lapworth, S.; Aveyard, P.; Jebb, S.A. A systematic review and meta-analysis of the effectiveness of meal replacements for weight loss. Obes. Rev. 2019, 20, 569–587. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ross, L.J.; Wallin, S.; Osland, E.J.; Memon, M.A. Commercial Very Low Energy Meal Replacements for Preoperative Weight Loss in Obese Patients: A Systematic Review. Obes. Surg. 2016, 26, 1343–1351. [Google Scholar] [CrossRef]
- Opray, N.; Grivell, R.M.; Deussen, A.R.; Dodd, J.M. Directed preconception health programs and interventions for improving pregnancy outcomes for women who are overweight or obese. Cochrane Database Syst. Rev. 2015, 7, CD010932. [Google Scholar] [CrossRef]
- Black, K.I.; Gordon, A. Obesity before pregnancy: New evidence and future strategies. Med. J. Aust. 2016, 205, 152–154. [Google Scholar] [CrossRef] [PubMed]
- Einarsson, S.; Bergh, C.; Friberg, B.; Pinborg, A.; Klajnbard, A.; Karlström, P.O.; Kluge, L.; Larsson, I.; Loft, A.; Mikkelsen-Englund, A.-L.; et al. Weight reduction intervention for obese infertile women prior to IVF: A randomized controlled trial. Hum. Reprod. 2017, 32, 1621–1630. [Google Scholar] [CrossRef] [Green Version]
- Moran, L.; Tsagareli, V.; Norman, R.; Noakes, M. Diet and IVF pilot study: Short-term weight loss improves pregnancy rates in overweight/obese women undertaking IVF. Aust. N. Z. J. Obstet. Gynaecol. 2011, 51, 455–459. [Google Scholar] [CrossRef]
- Sweeting, A.N.; Caterson, I.D. Approaches to obesity management. Intern. Med. J. 2017, 47, 734–739. [Google Scholar] [CrossRef] [Green Version]
- Sim, K.A.; Dezarnaulds, G.M.; Denyer, G.S.; Skilton, M.R.; Caterson, I.D. Weight loss improves reproductive outcomes in obese women undergoing fertility treatment: A randomized controlled trial. Clin. Obes. 2014, 4, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Sim, K.A.; Partridge, S.R.; Sainsbury, A. Does weight loss in overweight or obese women improve fertility treatment outcomes? A systematic review. Obes. Rev. 2014, 15, 839–850. [Google Scholar] [PubMed]
- Flexi-by-Impromy. 2020. Available online: https://impromy.com/flexi-by-impromy/ (accessed on 24 August 2020).
- NSW Get Healthy. 2020. Available online: https://www.gethealthynsw.com.au/ (accessed on 24 August 2020).
- Gloster, A.T.; Rhoades, H.M.; Novy, D.; Klotsche, J.; Senior, A.; Kunik, M.; Wilson, N.; Stanley, M.A. Psychometric properties of the Depression Anxiety and Stress Scale-21 in older primary care patients. J. Affec. Disord. 2008, 110, 248–259. [Google Scholar] [CrossRef] [Green Version]
- Collins, C.E.; Boggess, M.M.; Watson, J.F.; Guest, M.; Duncanson, K.; Pezdirc, K.; Rollo, R.; Hutchesson, M.J.; Burrows, T.L. Reproducibility and comparative validity of a food frequency questionnaire for Australian adults. Clin. Nutr. 2014, 33, 906–914. [Google Scholar] [CrossRef]
- Brindal, E.; Hendrie, G.A.; Taylor, P.; Freyne, J.; Noakes, M. Cohort Analysis of a 24-Week Randomized Controlled Trial to Assess the Efficacy of a Novel, Partial Meal Replacement Program Targeting Weight Loss and Risk Factor Reduction in Overweight/Obese Adults. Nutrients 2016, 8, 265. [Google Scholar] [CrossRef] [Green Version]
- Meike, A.Q.; Mutsaerts, M.D.; Anne, M.; van Oers, M.D.; Henk Groen, M.D.; Jan, M.; Burggraaff, M.D.; Walter, K.H.; Kuchenbecker, M.D.; Denise, A.M.; et al. Randomized Trial of a Lifestyle Program in Obese Infertile Women. N. Engl. J. Med. 2016, 374, 1942–1953. [Google Scholar]
- Price, S.A.; Sumithran, P.; Nankervis, A.; Permezel, M.; Proietto, J. Preconception management of women with obesity: A systematic review. Obes. Rev. 2019, 20, 510–526. [Google Scholar] [CrossRef]
- Best, D.; Avenell, A.; Bhattacharya, S. How effective are weight-loss interventions for improving fertility in women and men who are overweight or obese? A systematic review and meta-analysis of the evidence. Hum. Reprod. Update 2017, 23, 681–705. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dodd, J.M.; Turnbull, D.; McPhee, A.J.; Deussen, A.R.; Grivell, R.M.; Yelland, L.N.; Robinson, J.S. Antenatal lifestyle advice for women who are overweight or obese: LIMIT randomised trial. BMJ Br. Med. J. 2014, 348, g1285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Black, M.H.; Sacks, D.A.; Xiang, A.H.; Lawrence, J.M. The relative contribution of prepregnancy overweight and obesity, gestational weight gain, and IADPSG-defined gestational diabetes mellitus to fetal overgrowth. Diabetes Care 2013, 36, 56–62. [Google Scholar] [CrossRef] [Green Version]
- Muktabhant, B.; Lawrie, T.A.; Lumbiganon, P.; Laopaiboon, M. Diet or exercise, or both, for preventing excessive weight gain in pregnancy. Cochrane Database Syst. Rev. 2015, CD007145. [Google Scholar] [CrossRef]
- Poston, L.; Bell, R.; Croker, H.; Flynn, A.C.; Godfrey, K.M.; Goff, L.; Hayes, L.; Khazaezadeh, N.; Nelson, S.M.; Oteng-Ntim, E.; et al. Effect of a behavioural intervention in obese pregnant women (the UPBEAT study): A multicentre, randomised controlled trial. Lancet Diabetes Endocrinol. 2015, 3, 767–777. [Google Scholar] [CrossRef]
- The Royal Australian and New Zealand College of Obstetricians and Gynaecologists. Management of Obesity in Pregnancy; The Royal Australian and New Zealand College of Obstetricians and Gynaecologists: Melbourne, Austria, 2013. [Google Scholar]
- Eldridge, S.M.; Chan, C.L.; Campbell, M.J.; Bond, C.M.; Hopewell, S.; Thabane, L.; Lancaster, G.A. CONSORT 2010 statement: Extension to randomised pilot and feasibility trials. BMJ 2016, 355, i5239. [Google Scholar] [CrossRef] [Green Version]

| Screening | Week 1 Visit 1 | Week 5 Visit 2 | Week 10 Visit 3 | Week 10 +1 m | Week 10 to 12 Months | |
|---|---|---|---|---|---|---|
| Screen | X | |||||
| Consent | X | |||||
| Allocation | X | |||||
| Clinic visit | X | X | X | |||
| Anthropometry | X | X | ||||
| Venous blood | X | X | ||||
| Questionnaire | X | |||||
| Depression, Anxiety and Stress Scale 21DASS-21 | X | |||||
| Dietary survey | X | X | ||||
| Dietary advice | X | |||||
| Diet review | X | |||||
| Evaluation | X | |||||
| Conception | Quarterly text/email |
| Dietary Advice (n = 24) | Meal Replacements (n = 26) | |
|---|---|---|
| Age (years) | 33.7 ± 3.8 | 31.5 ± 6.9 |
| Weight (kg) | 92.0 ± 16.2 | 89.5 ± 17.1 |
| Height (cm) | 162.8 ± 5.9 | 164.9 ± 6.1 |
| Body Mass Index (kg/m2) | 34.7 ± 5.9 | 32.9 ± 5.9 |
| Waist circumference (cm) | 101.7 ± 11.7 | 97.9 ± 11.1 |
| Education: Secondary n (%) | 3 (13%) | 1 (4%) |
| Tertiary | 11 (46%) | 12 (46%) |
| Post graduate degree | 10 (42%) | 13 (50%) |
| Occupation: Do not have a job (n) | 0 | 2 |
| Part-time | 9 | 8 |
| Full time | 14 | 15 |
| Student or other | 1 | 1 |
| Medical history: Hypertension (n) | 2 | 0 |
| High cholesterol (n) | 2 | 1 |
| Pre-diabetes or gestational diabetes (n) | 3 | 2 |
| Obesity (n) | 7 | 4 |
| Polycystic ovary syndrome (n) | 1 | 2 |
| Other conditions * | 11 | 7 |
| Number of prior pregnancies: 0 | 12 | 10 |
| 1 | 7 | 13 |
| 2 or more | 5 | 3 |
| Number of children: 0 | 16 | 15 |
| 1 | 7 | 11 |
| 2 or more | 2 | 0 |
| Previous pregnancy loss:Yes (n) | 6 | 5 |
| No (n) | 18 | 21 |
| Depression (DASS-21) | 6.1 ± 6.4 | 6.8 ± 5.7 |
| Anxiety (DASS-21) | 6.8 ± 6.4 | 5.4 ± 5.6 |
| Stress (DASS-21) | 10.9 ± 8.3 | 10.5 ± 7.0 |
| n | Dietary Advice | Meal Replacements | p-Value 1 | ||
|---|---|---|---|---|---|
| Baseline | Week 10 | Baseline | Week 10 | ||
| 24 | 18 | 26 | 21 | ||
| Weight (kg) | 92.0 ± 16.2 | 86.3 ± 13.6 | 89.5 ± 17.1 | 84.9 ± 16.8 | 0.949 |
| Weight loss (kg) | 2.7 ± 3.8 | 4.7 ± 2.8 | 0.061 | ||
| Weight loss (%) | 2.8 ± 3.9 | 5.3 ± 3.0 | 0.029 | ||
| Waist (cm) | 101.7 ± 11.7 | 94.3 ± 9.0 | 97.9 ± 11.1 | 93.9 ± 11.7 | 0.900 |
| Waist loss (cm) | 5.4 ± 4.8 | 5.3 ± 4.4 | 0.941 | ||
| Waist loss (%) | 5.7 ± 5.2 | 5.6 ± 4.9 | 0.912 | ||
| Dietary Advice | Mean Difference | Meal Replacements | Mean Difference | |||
|---|---|---|---|---|---|---|
| Baseline | Week 10 | Baseline | Week 10 | |||
| HbA1c (mmol/mol) | 32.55 ± 4.17 (n = 20) | 33.07 ± 4.60 (n = 14) | 0 ± 1.8 | 30.89 ± 6.13 (n = 26) | 31.16 ± 2.39 (n = 19) | 0.15± 6.6 |
| Total cholesterol (mmol/L) | 4.84 ± 1.07 (n = 20) | 5.00 ± 0.99 (n = 15) | 0.04 ± 0.9 | 4.88 ± 0.89 (n = 26) | 4.51 ± 0.97 (n = 18) | 0.33 ± 0.6 |
| Triglycerides (mmol/L) | 1.84 ± 1.13 (n = 21) | 1.96 ± 1.58 (n = 15) | 0.14 ± 1.5 | 1.31 ± 0.63 (n = 25) | 1.17 ± 0.49 (n = 19) | 0.13 ± 0.5 |
| Iron (umol/L) | 14.70 ± 4.61 (n = 20) | 16.73 ± 5.74 (n = 15) | 0.92 ± 5.7 | 14.50 ± 4.53 (n = 26) | 14.95 ± 4.90 (n = 19) | 0.53 ± 4.1 |
| Ferritin (ug/L) | 77.40 ± 52.56 (n = 20) | 84.27 ± 54.58 (n = 15) | 5.85 ± 39.5 | 81.38 ± 59.97 (n = 26) | 75.84 ± 49.93 (n = 19) | 6.68 ± 26.1 |
| Transferrin (g/L) | 2.83 ± 0.36 (n = 20) | 2.80 ± 0.40 (n = 15) | 0.12 ± 0.31 | 2.77 ± 0.42 (n = 26) | 2.80 ± 0.34 (n = 19) | 0.053 ± 0.18 |
| Transferrin saturation (%) | 20.60 ± 5.77 (n = 20) | 24.53 ± 10.50 (n = 15) | 3.00 ± 10.2 | 21.42 ± 7.57 (n = 26) | 21.32 ± 7.16 (n = 19) | 0.74 ± 5.9 |
| Serum folate (nmol/L) | 36.12 ± 9.53 (n = 20) | 40.30 ± 7.62 (n = 15) | 3.33 ± 6.0 | 35.03 ± 9.11 (n = 26) | 35.93 ± 8.55 (n = 19) | 0.98 ± 9.7 |
| Vitamin D (nmol/L) | 61.76 ± 24.86 (n = 21) | 66.53 ± 24.91 (n = 15) | 9.0 ± 27.5 | 65.04 ± 25.24 (n = 24) | 70.00 ± 26.11 (n = 19) | 3.53 ± 19.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muirhead, R.; Kizirian, N.; Lal, R.; Black, K.; Prys-Davies, A.; Nassar, N.; Baur, L.; Sainsbury, A.; Sweeting, A.; Markovic, T.; et al. A Pilot Randomized Controlled Trial of a Partial Meal Replacement Preconception Weight Loss Program for Women with Overweight and Obesity. Nutrients 2021, 13, 3200. https://doi.org/10.3390/nu13093200
Muirhead R, Kizirian N, Lal R, Black K, Prys-Davies A, Nassar N, Baur L, Sainsbury A, Sweeting A, Markovic T, et al. A Pilot Randomized Controlled Trial of a Partial Meal Replacement Preconception Weight Loss Program for Women with Overweight and Obesity. Nutrients. 2021; 13(9):3200. https://doi.org/10.3390/nu13093200
Chicago/Turabian StyleMuirhead, Roslyn, Nathalie Kizirian, Ravin Lal, Kirsten Black, Ann Prys-Davies, Natasha Nassar, Louise Baur, Amanda Sainsbury, Arianne Sweeting, Tania Markovic, and et al. 2021. "A Pilot Randomized Controlled Trial of a Partial Meal Replacement Preconception Weight Loss Program for Women with Overweight and Obesity" Nutrients 13, no. 9: 3200. https://doi.org/10.3390/nu13093200
APA StyleMuirhead, R., Kizirian, N., Lal, R., Black, K., Prys-Davies, A., Nassar, N., Baur, L., Sainsbury, A., Sweeting, A., Markovic, T., Skilton, M., Hyett, J., de Vries, B., Tarnow-Mordi, W., Brand-Miller, J., & Gordon, A., on behalf of the PreBabe Investigator Team. (2021). A Pilot Randomized Controlled Trial of a Partial Meal Replacement Preconception Weight Loss Program for Women with Overweight and Obesity. Nutrients, 13(9), 3200. https://doi.org/10.3390/nu13093200








