Potential Health Benefits of Curcumin on Female Reproductive Disorders: A Review
Abstract
:1. Introduction
2. Methods of Review
3. Effect of Curcumin on Polycystic Ovary Syndrome (PCOS)
4. Effect of Curcumin on Ovarian Diseases
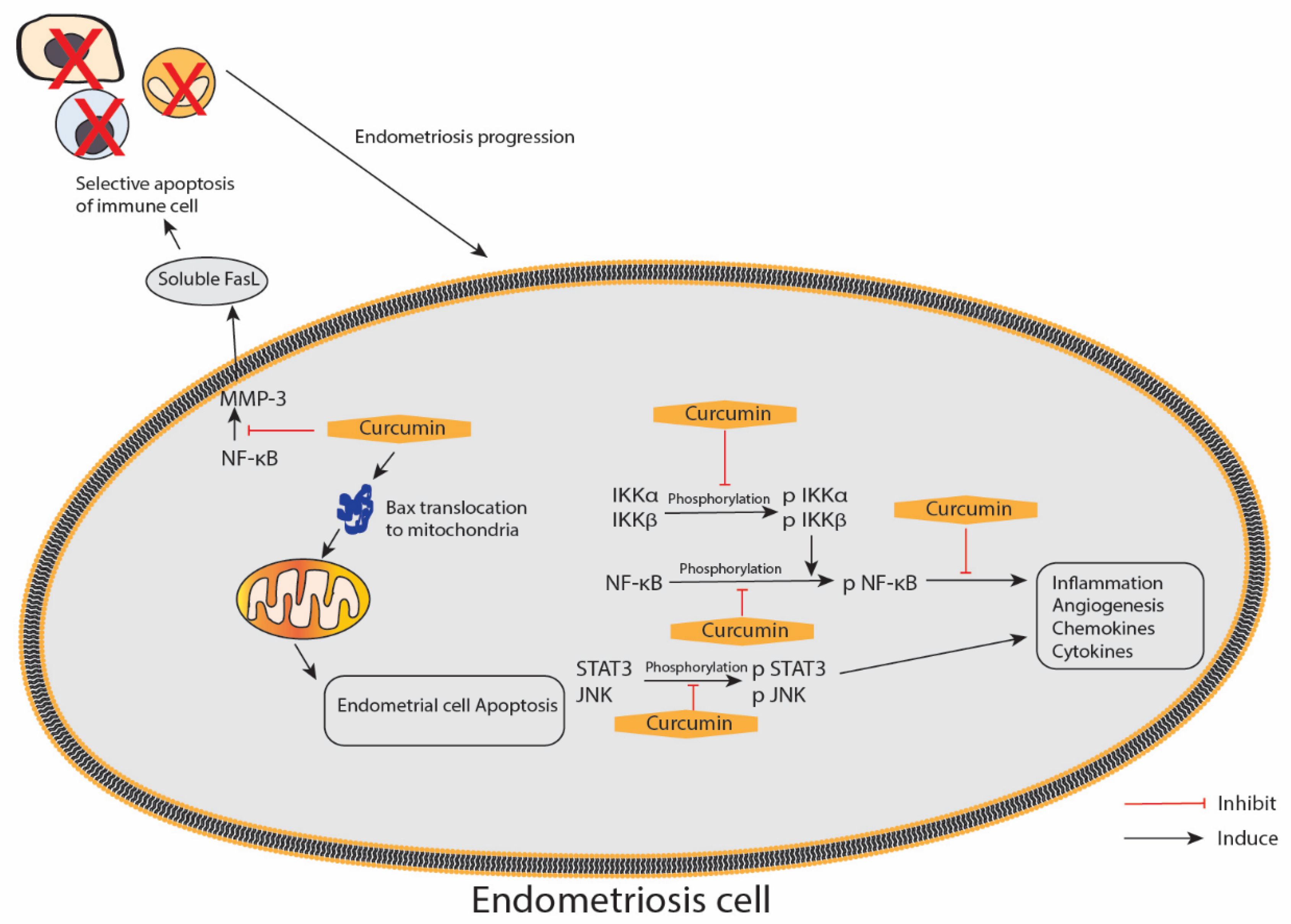
5. Effect of Curcumin on Endometriosis
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dosoky, N.S.; Setzer, W.N. Chemical Composition and Biological Activities of Essential Oils of Curcuma Species. Nutrients 2018, 10, 1196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharifi-Rad, J.; Rayess, Y.E.; Rizk, A.A.; Sadaka, C.; Zgheib, R.; Zam, W.; Sestito, S.; Rapposelli, S.; Neffe-Skocińska, K.; Zielińska, D.; et al. Turmeric and Its Major Compound Curcumin on Health: Bioactive Effects and Safety Profiles for Food, Pharmaceutical, Biotechnological and Medicinal Applications. Front. Pharmacol. 2020, 11, 01021. [Google Scholar] [CrossRef] [PubMed]
- Prasad, S.; Aggarwal, B.B. Turmeric, the Golden Spice: From Traditional Medicine to Modern Medicine. In Herbal Medicine: Biomolecular and Clinical Aspects; Benzie, I.F.F., Wachtel-Galor, S., Eds.; CRC Press: Boca Raton, FL, USA; Taylor and Francis Group, LLC.: Boca Raton, FL, USA, 2011. [Google Scholar]
- Priyadarsini, K.I. The chemistry of curcumin: From extraction to therapeutic agent. Molecules 2014, 19, 20091–20112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akram, M.; Afzal, A.; Khan, U.; Abdul, H.; Mohiuddin, E.; Asif, M. Curcuma longa and Curcumin: A review article. Rom. J. Biol.-Plant Biol. 2010, 55, 65–70. [Google Scholar]
- Tabrizi, R.; Vakili, S.; Akbari, M.; Mirhosseini, N.; Lankarani, K.B.; Rahimi, M.; Mobini, M.; Jafarnejad, S.; Vahedpoor, Z.; Asemi, Z. The effects of curcumin-containing supplements on biomarkers of inflammation and oxidative stress: A systematic review and meta-analysis of randomized controlled trials. Phytother. Res. 2019, 33, 253–262. [Google Scholar] [CrossRef]
- Gómez-Estaca, J.; Balaguer, M.; López-Carballo, G.; Gavara, R.; Hernández-Muñoz, P. Improving antioxidant and antimicrobial properties of curcumin by means of encapsulation in gelatin through electrohydrodynamic atomization. Food Hydrocoll. 2017, 70, 313–320. [Google Scholar] [CrossRef]
- Amalraj, A.; Varma, K.; Jacob, J.; Divya, C.; Kunnumakkara, A.B.; Stohs, S.J.; Gopi, S. A Novel Highly Bioavailable Curcumin Formulation Improves Symptoms and Diagnostic Indicators in Rheumatoid Arthritis Patients: A Randomized, Double-Blind, Placebo-Controlled, Two-Dose, Three-Arm, and Parallel-Group Study. J. Med. Food 2017, 20, 1022–1030. [Google Scholar] [CrossRef]
- Wang, M.; Jiang, S.; Zhou, L.; Yu, F.; Ding, H.; Li, P.; Zhou, M.; Wang, K. Potential Mechanisms of Action of Curcumin for Cancer Prevention: Focus on Cellular Signaling Pathways and miRNAs. Int. J. Biol. Sci. 2019, 15, 1200–1214. [Google Scholar] [CrossRef] [Green Version]
- Ng, Z.Y.; Wong, J.Y.; Panneerselvam, J.; Madheswaran, T.; Kumar, P.; Pillay, V.; Hsu, A.; Hansbro, N.; Bebawy, M.; Wark, P.; et al. Assessing the potential of liposomes loaded with curcumin as a therapeutic intervention in asthma. Colloids Surf. B Biointerfaces 2018, 172, 51–59. [Google Scholar] [CrossRef]
- Yang, Q.-Q.; Farha, A.K.; Kim, G.; Gul, K.; Gan, R.-Y.; Corke, H. Antimicrobial and anticancer applications and related mechanisms of curcumin-mediated photodynamic treatments. Trends Food Sci. Technol. 2020, 97, 341–354. [Google Scholar] [CrossRef]
- Song, L.; Zhang, F.; Yu, J.; Wei, C.; Han, Q.; Meng, X. Antifungal effect and possible mechanism of curcumin mediated photodynamic technology against Penicillium expansum. Postharvest Biol. Technol. 2020, 167, 111234. [Google Scholar] [CrossRef]
- Thimmulappa, R.K.; Mudnakudu-Nagaraju, K.K.; Shivamallu, C.; Subramaniam, K.J.T.; Radhakrishnan, A.; Bhojraj, S.; Kuppusamy, G. Antiviral and immunomodulatory activity of curcumin: A case for prophylactic therapy for COVID-19. Heliyon 2021, 7, e06350. [Google Scholar] [CrossRef]
- Ak, T.; Gülçin, I. Antioxidant and radical scavenging properties of curcumin. Chem. Biol. Interact. 2008, 174, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Sahebkar, A.; Serban, M.-C.; Ursoniu, S.; Banach, M. Effect of curcuminoids on oxidative stress: A systematic review and meta-analysis of randomized controlled trials. J. Funct. Foods 2015, 18, 898–909. [Google Scholar] [CrossRef]
- Anthwal, A.; Thakur, B.K.; Rawat, M.S.; Rawat, D.S.; Tyagi, A.K.; Aggarwal, B.B. Synthesis, characterization and in vitro anticancer activity of C-5 curcumin analogues with potential to inhibit TNF-α-induced NF-κB activation. BioMed Res. Int. 2014, 2014, 524161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rathore, S.; Siddiqui, M.; Sharma, P.; Devi, S.; Nagar, J.; Khalid, M. Curcumin: A Review for Health Benefits. Int. J. Sci. Res. (IJSR) 2020, 7, 273–290. [Google Scholar]
- González-Ramos, R.; Van Langendonckt, A.; Defrère, S.; Lousse, J.C.; Colette, S.; Devoto, L.; Donnez, J. Involvement of the nuclear factor-κB pathway in the pathogenesis of endometriosis. Fertil. Steril. 2010, 94, 1985–1994. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.C.; Patchva, S.; Aggarwal, B.B. Therapeutic roles of curcumin: Lessons learned from clinical trials. AAPS J. 2013, 15, 195–218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hewlings, S.J.; Kalman, D.S. Curcumin: A Review of Its Effects on Human Health. Foods 2017, 6, 92. [Google Scholar] [CrossRef] [PubMed]
- Kocaadam, B.; Şanlier, N. Curcumin, an active component of turmeric (Curcuma longa), and its effects on health. Crit. Rev. Food Sci. Nutr. 2017, 57, 2889–2895. [Google Scholar] [CrossRef] [PubMed]
- Basnet, P.; Skalko-Basnet, N. Curcumin: An anti-inflammatory molecule from a curry spice on the path to cancer treatment. Molecules 2011, 16, 4567–4598. [Google Scholar] [CrossRef] [Green Version]
- Vallée, A.; Lecarpentier, Y. Curcumin and Endometriosis. Int. J. Mol. Sci. 2020, 21, 2440. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prasad, S.; Tyagi, A.K.; Aggarwal, B.B. Recent developments in delivery, bioavailability, absorption and metabolism of curcumin: The golden pigment from golden spice. Cancer Res. Treat. 2014, 46, 2–18. [Google Scholar] [CrossRef] [Green Version]
- Shehzad, A.; Wahid, F.; Lee, Y.S. Curcumin in cancer chemoprevention: Molecular targets, pharmacokinetics, bioavailability, and clinical trials. Arch. Pharm. 2010, 343, 489–499. [Google Scholar] [CrossRef]
- Karthikeyan, A.; Senthil, N.; Min, T. Nanocurcumin: A Promising Candidate for Therapeutic Applications. Front. Pharmacol. 2020, 11, 487. [Google Scholar] [CrossRef]
- Stohs, S.J.; Chen, O.; Ray, S.D.; Ji, J.; Bucci, L.R.; Preuss, H.G. Highly Bioavailable Forms of Curcumin and Promising Avenues for Curcumin-Based Research and Application: A Review. Molecules 2020, 25, 1397. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shoba, G.; Joy, D.; Joseph, T.; Majeed, M.; Rajendran, R.; Srinivas, P.S. Influence of piperine on the pharmacokinetics of curcumin in animals and human volunteers. Planta Med. 1998, 64, 353–356. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kanai, M.; Imaizumi, A.; Otsuka, Y.; Sasaki, H.; Hashiguchi, M.; Tsujiko, K.; Matsumoto, S.; Ishiguro, H.; Chiba, T. Dose-escalation and pharmacokinetic study of nanoparticle curcumin, a potential anticancer agent with improved bioavailability, in healthy human volunteers. Cancer Chemother. Pharmacol. 2012, 69, 65–70. [Google Scholar] [CrossRef] [Green Version]
- Xu, H.; Gong, Z.; Zhou, S.; Yang, S.; Wang, D.; Chen, X.; Wu, J.; Liu, L.; Zhong, S.; Zhao, J.; et al. Liposomal Curcumin Targeting Endometrial Cancer Through the NF-κB Pathway. Cell. Physiol. Biochem. 2018, 48, 569–582. [Google Scholar] [CrossRef] [PubMed]
- Teede, H.; Deeks, A.; Moran, L. Polycystic ovary syndrome: A complex condition with psychological, reproductive and metabolic manifestations that impacts on health across the lifespan. BMC Med. 2010, 8, 41. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Escobar-Morreale, H.F. Polycystic ovary syndrome: Definition, aetiology, diagnosis and treatment. Nat. Rev. Endocrinol. 2018, 14, 270. [Google Scholar] [CrossRef]
- Khadilkar, S.S. Can Polycystic Ovarian Syndrome be cured? Unfolding the Concept of Secondary Polycystic Ovarian Syndrome! J. Obstet. Gynaecol. India 2019, 69, 297–302. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dennett, C.C.; Simon, J. The role of polycystic ovary syndrome in reproductive and metabolic health: Overview and approaches for treatment. Diabetes Spectr. 2015, 28, 116–120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kwon, C.-Y.; Cho, I.-H.; Park, K.S. Therapeutic Effects and Mechanisms of Herbal Medicines for Treating Polycystic Ovary Syndrome: A Review. Front. Pharmacol. 2020, 11, 1192. [Google Scholar] [CrossRef]
- Arentz, S.; Smith, C.A.; Abbott, J.; Bensoussan, A. Nutritional supplements and herbal medicines for women with polycystic ovary syndrome; a systematic review and meta-analysis. BMC Complement. Altern. Med. 2017, 17, 500. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moini Jazani, A.; Nasimi Doost Azgomi, H.; Nasimi Doost Azgomi, A.; Nasimi Doost Azgomi, R. A comprehensive review of clinical studies with herbal medicine on polycystic ovary syndrome (PCOS). Daru 2019, 27, 863–877. [Google Scholar] [CrossRef]
- Kamal, D.A.M.; Salamt, N.; Zaid, S.S.M.; Mokhtar, M.H. Beneficial Effects of Green Tea Catechins on Female Reproductive Disorders: A Review. Molecules 2021, 26, 2675. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; Yang, H.; Lee, S.R.; Kwon, S.W.; Hong, E.J.; Lee, H.W. Welsh Onion Root (Allium fistulosum) Restores Ovarian Functions from Letrozole Induced-Polycystic Ovary Syndrome. Nutrients 2018, 10, 1430. [Google Scholar] [CrossRef] [Green Version]
- Ong, M.; Peng, J.; Jin, X.; Qu, X. Chinese Herbal Medicine for the Optimal Management of Polycystic Ovary Syndrome. Am. J. Chin. Med. 2017, 45, 405–422. [Google Scholar] [CrossRef] [PubMed]
- Jamilian, M.; Foroozanfard, F.; Kavossian, E.; Aghadavod, E.; Shafabakhsh, R.; Hoseini, A.; Asemi, Z. Effects of curcumin on body weight, glycemic control and serum lipids in women with polycystic ovary syndrome: A randomized, double-blind, placebo-controlled trial. Clin. Nutr. ESPEN 2020, 36, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Heshmati, J.; Moini, A.; Sepidarkish, M.; Morvaridzadeh, M.; Salehi, M.; Palmowski, A.; Mojtahedi, M.F.; Shidfar, F. Effects of curcumin supplementation on blood glucose, insulin resistance and androgens in patients with polycystic ovary syndrome: A randomized double-blind placebo-controlled clinical trial. Phytomed. Int. J. Phytother. Phytopharm. 2021, 80, 153395. [Google Scholar] [CrossRef] [PubMed]
- Sohaei, S.; Amani, R.; Tarrahi, M.J.; Ghasemi-Tehrani, H. The effects of curcumin supplementation on glycemic status, lipid profile and hs-CRP levels in overweight/obese women with polycystic ovary syndrome: A randomized, double-blind, placebo-controlled clinical trial. Complementary Ther. Med. 2019, 47, 102201. [Google Scholar] [CrossRef] [PubMed]
- Heshmati, J.; Golab, F.; Morvaridzadeh, M.; Potter, E.; Akbari-Fakhrabadi, M.; Farsi, F.; Tanbakooei, S.; Shidfar, F. The effects of curcumin supplementation on oxidative stress, Sirtuin-1 and peroxisome proliferator activated receptor γ coactivator 1α gene expression in polycystic ovarian syndrome (PCOS) patients: A randomized placebo-controlled clinical trial. Diabetes Metab. Syndr. 2020, 14, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, S.; Karimzadeh Bardei, L.; Hojati, V.; Ghorbani, A.G.; Nabiuni, M. Anti-Inflammatory Effects of Curcumin on Insulin Resistance Index, Levels of Interleukin-6, C-Reactive Protein, and Liver Histology in Polycystic Ovary Syndrome-Induced Rats. Cell J. 2017, 19, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Fatemi Abhari, S.M.; Khanbabaei, R.; Hayati Roodbari, N.; Parivar, K.; Yaghmaei, P. Curcumin-loaded super-paramagnetic iron oxide nanoparticle affects on apoptotic factors expression and histological changes in a prepubertal mouse model of polycystic ovary syndrome-induced by dehydroepiandrosterone—A molecular and stereological study. Life Sci. 2020, 249, 117515. [Google Scholar] [CrossRef] [PubMed]
- Abuelezz, N.Z.; Shabana, M.E.; Abdel-Mageed, H.M.; Rashed, L.; Morcos, G.N.B. Nanocurcumin alleviates insulin resistance and pancreatic deficits in polycystic ovary syndrome rats: Insights on PI3K/AkT/mTOR and TNF-α modulations. Life Sci. 2020, 256, 118003. [Google Scholar] [CrossRef]
- Mohammadi, S.; Kayedpoor, P.; Karimzadeh-Bardei, L.; Nabiuni, M. The Effect of Curcumin on TNF-α, IL-6 and CRP Expression in a Model of Polycystic Ovary Syndrome as an Inflammation State. J. Reprod. Infertil. 2017, 18, 352–360. [Google Scholar] [PubMed]
- Reddy, P.S.; Begum, N.; Mutha, S.; Bakshi, V. Beneficial effect of Curcumin in Letrozole induced polycystic ovary syndrome. Asian Pac. J. Reprod. 2016, 5, 116–122. [Google Scholar] [CrossRef]
- Rosenfield, R.L.; Ehrmann, D.A. The Pathogenesis of Polycystic Ovary Syndrome (PCOS): The Hypothesis of PCOS as Functional Ovarian Hyperandrogenism Revisited. Endocr. Rev. 2016, 37, 467–520. [Google Scholar] [CrossRef] [PubMed]
- Burt Solorzano, C.M.; Beller, J.P.; Abshire, M.Y.; Collins, J.S.; McCartney, C.R.; Marshall, J.C. Neuroendocrine dysfunction in polycystic ovary syndrome. Steroids 2012, 77, 332–337. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bachmeier, B.E.; Mirisola, V.; Romeo, F.; Generoso, L.; Esposito, A.; Dell’Eva, R.; Blengio, F.; Killian, P.H.; Albini, A.; Pfeffer, U. Reference Profile Correlation Reveals Estrogen-like Trancriptional Activity of Curcumin. Cell. Physiol. Biochem. 2010, 26, 471–482. [Google Scholar] [CrossRef] [Green Version]
- Dumesic, D.A.; Oberfield, S.E.; Stener-Victorin, E.; Marshall, J.C.; Laven, J.S.; Legro, R.S. Scientific Statement on the Diagnostic Criteria, Epidemiology, Pathophysiology, and Molecular Genetics of Polycystic Ovary Syndrome. Endocr. Rev. 2015, 36, 487–525. [Google Scholar] [CrossRef] [Green Version]
- Wang, F.; Wang, S.; Zhang, Z.; Lin, Q.; Liu, Y.; Xiao, Y.; Xiao, K.; Wang, Z. Defective insulin signaling and the protective effects of dimethyldiguanide during follicular development in the ovaries of polycystic ovary syndrome. Mol. Med. Rep. 2017, 16, 8164–8170. [Google Scholar] [CrossRef] [Green Version]
- Barber, T.M.; Hanson, P.; Weickert, M.O.; Franks, S. Obesity and Polycystic Ovary Syndrome: Implications for Pathogenesis and Novel Management Strategies. Clin. Med. Insights Reprod. Health 2019, 13, 1179558119874042. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McCartney, C.R.; Marshall, J.C. Polycystic Ovary Syndrome. N. Engl. J. Med. 2016, 375, 54–64. [Google Scholar] [CrossRef] [Green Version]
- Farhood, B.; Mortezaee, K.; Goradel, N.H.; Khanlarkhani, N.; Salehi, E.; Nashtaei, M.S.; Najafi, M.; Sahebkar, A. Curcumin as an anti-inflammatory agent: Implications to radiotherapy and chemotherapy. J. Cell. Physiol. 2019, 234, 5728–5740. [Google Scholar] [CrossRef] [PubMed]
- Khashchenko, E.; Vysokikh, M.; Uvarova, E.; Krechetova, L.; Vtorushina, V.; Ivanets, T.; Volodina, M.; Tarasova, N.; Sukhanova, I.; Sukhikh, G. Activation of Systemic Inflammation and Oxidative Stress in Adolescent Girls with Polycystic Ovary Syndrome in Combination with Metabolic Disorders and Excessive Body Weight. J. Clin. Med. 2020, 9, 1399. [Google Scholar] [CrossRef]
- Murri, M.; Luque-Ramírez, M.; Insenser, M.; Ojeda-Ojeda, M.; Escobar-Morreale, H.F. Circulating markers of oxidative stress and polycystic ovary syndrome (PCOS): A systematic review and meta-analysis. Hum. Reprod. Update 2013, 19, 268–288. [Google Scholar] [CrossRef]
- Jakubczyk, K.; Drużga, A.; Katarzyna, J.; Skonieczna-Żydecka, K. Antioxidant Potential of Curcumin—A Meta-Analysis of Randomized Clinical Trials. Antioxidants 2020, 9, 1092. [Google Scholar] [CrossRef]
- Tanvir, E.M.; Hossen, M.S.; Hossain, M.F.; Afroz, R.; Gan, S.H.; Khalil, M.I.; Karim, N. Antioxidant Properties of Popular Turmeric (Curcuma longa) Varieties from Bangladesh. J. Food Qual. 2017, 2017, 8471785. [Google Scholar] [CrossRef] [Green Version]
- Guo, J.; Cao, X.; Hu, X.; Li, S.; Wang, J. The anti-apoptotic, antioxidant and anti-inflammatory effects of curcumin on acrylamide-induced neurotoxicity in rats. BMC Pharmacol. Toxicol. 2020, 21, 62. [Google Scholar] [CrossRef]
- Loganes, C.; Lega, S.; Bramuzzo, M.; Vecchi Brumatti, L.; Piscianz, E.; Valencic, E.; Tommasini, A.; Marcuzzi, A. Curcumin Anti-Apoptotic Action in a Model of Intestinal Epithelial Inflammatory Damage. Nutrients 2017, 9, 578. [Google Scholar] [CrossRef] [Green Version]
- Tomeh, M.A.; Hadianamrei, R.; Zhao, X. A Review of Curcumin and Its Derivatives as Anticancer Agents. Int. J. Mol. Sci. 2019, 20, 1033. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yan, Z.; Dai, Y.; Fu, H.; Zheng, Y.; Bao, D.; Yin, Y.; Chen, Q.; Nie, X.; Hao, Q.; Hou, D.; et al. Curcumin exerts a protective effect against premature ovarian failure in mice. J. Mol. Endocrinol. 2018, 60, 261–271. [Google Scholar] [CrossRef] [Green Version]
- Gozzelino, R.; Jeney, V.; Soares, M.P. Mechanisms of Cell Protection by Heme Oxygenase-1. Annu. Rev. Pharmacol. Toxicol. 2010, 50, 323–354. [Google Scholar] [CrossRef] [Green Version]
- Kádasi, A.; Maruniaková, N.; Štochmaľová, A.; Bauer, M.; Grossmann, R.; Harrath, A.H.; Kolesárová, A.; Sirotkin, A.V. Direct effect of curcumin on porcine ovarian cell functions. Anim. Reprod. Sci. 2017, 182, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Eser, A.; Hizli, D.; Haltas, H.; Namuslu, M.; Kosus, A.; Kosus, N.; Kafali, H. Effects of curcumin on ovarian ischemia-reperfusion injury in a rat model. Biomed. Rep. 2015, 3, 807–813. [Google Scholar] [CrossRef] [Green Version]
- Behroozi-Lak, T.; Ebrahimpour, M.; Zarei, L.; Pourjabali, M.; Farhad, N.; Mohaddesi, H. Systemic administration of curcumin nanoparticles protects ischemia-reperfusion injury in ovaries: An animal model study. Rev. Assoc. Med. Bras. 2018, 64, 22–31. [Google Scholar] [CrossRef] [Green Version]
- Alekseyeva, I.N.; Makogon, N.V.; Bryzgina, T.M.; Voznesenskaya, T.Y.; Sukhina, V.S. Effects of NF-κB blocker curcumin on oogenesis and immunocompetent organ cells in immune ovarian injury in mice. Bull. Exp. Biol. Med. 2011, 151, 432–435. [Google Scholar] [CrossRef]
- Wang, X.N.; Zhang, C.J.; Diao, H.L.; Zhang, Y. Protective Effects of Curcumin against Sodium Arsenite-induced Ovarian Oxidative Injury in a Mouse Model. Chin. Med. J. 2017, 130, 1026–1032. [Google Scholar] [CrossRef]
- Betts, D.H.; Bain, N.T.; Madan, P. The p66Shc Adaptor Protein Controls Oxidative Stress Response in Early Bovine Embryos. PLoS ONE 2014, 9, e86978. [Google Scholar] [CrossRef] [Green Version]
- Kevenaar, M.E.; Meerasahib, M.F.; Kramer, P.; van de Lang-Born, B.M.N.; de Jong, F.H.; Groome, N.P.; Themmen, A.P.N.; Visser, J.A. Serum Anti-Müllerian Hormone Levels Reflect the Size of the Primordial Follicle Pool in Mice. Endocrinology 2006, 147, 3228–3234. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jana, S.; Paul, S.; Swarnakar, S. Curcumin as anti-endometriotic agent: Implication of MMP-3 and intrinsic apoptotic pathway. Biochem. Pharmacol. 2012, 83, 797–804. [Google Scholar] [CrossRef]
- Culley, L.; Law, C.; Hudson, N.; Denny, E.; Mitchell, H.; Baumgarten, M.; Raine-Fenning, N. The social and psychological impact of endometriosis on women’s lives: A critical narrative review. Hum. Reprod. Update 2013, 19, 625–639. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferrero, S.; Evangelisti, G.; Barra, F. Current and emerging treatment options for endometriosis. Expert Opin. Pharm. 2018, 19, 1109–1125. [Google Scholar] [CrossRef]
- Jelodar, G.; Azimifar, A. Evaluation of serum cancer antigen 125, resistin, leptin, homocysteine, and total antioxidant capacity in rat model of endometriosis treated with Curcumin. Physiol. Rep. 2019, 7, e14016. [Google Scholar] [CrossRef] [Green Version]
- Swarnakar, S.; Paul, S. Curcumin arrests endometriosis by downregulation of matrix metalloproteinase-9 activity. Indian J. Biochem. Biophys. 2009, 46, 59–65. [Google Scholar] [PubMed]
- Zhang, Y.; Cao, H.; Hu, Y.Y.; Wang, H.; Zhang, C.J. Inhibitory effect of curcumin on angiogenesis in ectopic endometrium of rats with experimental endometriosis. Int. J. Mol. Med. 2011, 27, 87–94. [Google Scholar] [CrossRef] [Green Version]
- Kizilay, G.; Uz, Y.H.; Seren, G.; Ulucam, E.; Yilmaz, A.; Cukur, Z.; Kayisli, U.A. In vivo effects of curcumin and deferoxamine in experimental endometriosis. Adv. Clin. Exp. Med. 2017, 26, 207–213. [Google Scholar] [CrossRef]
- Jana, S.; Rudra, D.S.; Paul, S.; Snehasikta, S. Curcumin delays endometriosis development by inhibiting MMP-2 activity. Indian J. Biochem. Biophys. 2012, 49, 342–348. [Google Scholar]
- Kim, K.H.; Lee, E.N.; Park, J.K.; Lee, J.R.; Kim, J.H.; Choi, H.J.; Kim, B.S.; Lee, H.W.; Lee, K.S.; Yoon, S. Curcumin attenuates TNF-α-induced expression of intercellular adhesion molecule-1, vascular cell adhesion molecule-1 and proinflammatory cytokines in human endometriotic stromal cells. Phytother. Res. 2012, 26, 1037–1047. [Google Scholar] [CrossRef]
- Chowdhury, I.; Banerjee, S.; Driss, A.; Xu, W.; Mehrabi, S.; Nezhat, C.; Sidell, N.; Taylor, R.N.; Thompson, W.E. Curcumin attenuates proangiogenic and proinflammatory factors in human eutopic endometrial stromal cells through the NF-κB signaling pathway. J. Cell. Physiol. 2019, 234, 6298–6312. [Google Scholar] [CrossRef] [Green Version]
- Hendarto, H.; Yohanes Ardianta Widyanugraha, M.; Widjiati, W. Curcumin improves growth factors expression of bovine cumulus-oocyte complexes cultured in peritoneal fluid of women with endometriosis. Int. J. Reprod. Biomed. 2018, 16, 775–782. [Google Scholar] [CrossRef]
- Zhang, Y.; Cao, H.; Yu, Z.; Peng, H.Y.; Zhang, C.J. Curcumin inhibits endometriosis endometrial cells by reducing estradiol production. Iran J. Reprod. Med. 2013, 11, 415–422. [Google Scholar]
- Cao, H.; Wei, Y.X.; Zhou, Q.; Zhang, Y.; Guo, X.P.; Zhang, J. Inhibitory effect of curcumin in human endometriosis endometrial cells via downregulation of vascular endothelial growth factor. Mol. Med. Rep. 2017, 16, 5611–5617. [Google Scholar] [CrossRef] [Green Version]
- Bharti, A.C.; Donato, N.; Singh, S.; Aggarwal, B.B. Curcumin (diferuloylmethane) down-regulates the constitutive activation of nuclear factor-kappa B and IkappaBalpha kinase in human multiple myeloma cells, leading to suppression of proliferation and induction of apoptosis. Blood 2003, 101, 1053–1062. [Google Scholar] [CrossRef]
- Liu, H.; Lang, J.H. Is abnormal eutopic endometrium the cause of endometriosis? The role of eutopic endometrium in pathogenesis of endometriosis. Med. Sci. Monit. 2011, 17, Ra92–Ra99. [Google Scholar] [CrossRef] [Green Version]
- Laschke, M.W.; Menger, M.D. Basic mechanisms of vascularization in endometriosis and their clinical implications. Hum. Reprod. Update 2018, 24, 207–224. [Google Scholar] [CrossRef]
- Meyer, M.; Clauss, M.; Lepple-Wienhues, A.; Waltenberger, J.; Augustin, H.G.; Ziche, M.; Lanz, C.; Büttner, M.; Rziha, H.J.; Dehio, C. A novel vascular endothelial growth factor encoded by Orf virus, VEGF-E, mediates angiogenesis via signalling through VEGFR-2 (KDR) but not VEGFR-1 (Flt-1) receptor tyrosine kinases. EMBO J 1999, 18, 363–374. [Google Scholar] [CrossRef] [Green Version]
- McLaren, J.; Prentice, A.; Charnock-Jones, D.S.; Millican, S.A.; Müller, K.H.; Sharkey, A.M.; Smith, S.K. Vascular endothelial growth factor is produced by peritoneal fluid macrophages in endometriosis and is regulated by ovarian steroids. J. Clin. Investig. 1996, 98, 482–489. [Google Scholar] [CrossRef]
- Kaczanowski, S. Apoptosis: Its origin, history, maintenance and the medical implications for cancer and aging. Phys Biol. 2016, 13, 031001. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kutscher, L.M.; Shaham, S. Non-apoptotic cell death in animal development. Cell Death Differ. 2017, 24, 1326–1336. [Google Scholar] [CrossRef] [PubMed]
- Ravindran, J.; Prasad, S.; Aggarwal, B.B. Curcumin and cancer cells: How many ways can curry kill tumor cells selectively? AAPS J. 2009, 11, 495–510. [Google Scholar] [CrossRef] [PubMed]
- Taylor, H.S.; Giudice, L.C.; Lessey, B.A.; Abrao, M.S.; Kotarski, J.; Archer, D.F.; Diamond, M.P.; Surrey, E.; Johnson, N.P.; Watts, N.B.; et al. Treatment of Endometriosis-Associated Pain with Elagolix, an Oral GnRH Antagonist. N. Engl. J. Med. 2017, 377, 28–40. [Google Scholar] [CrossRef]
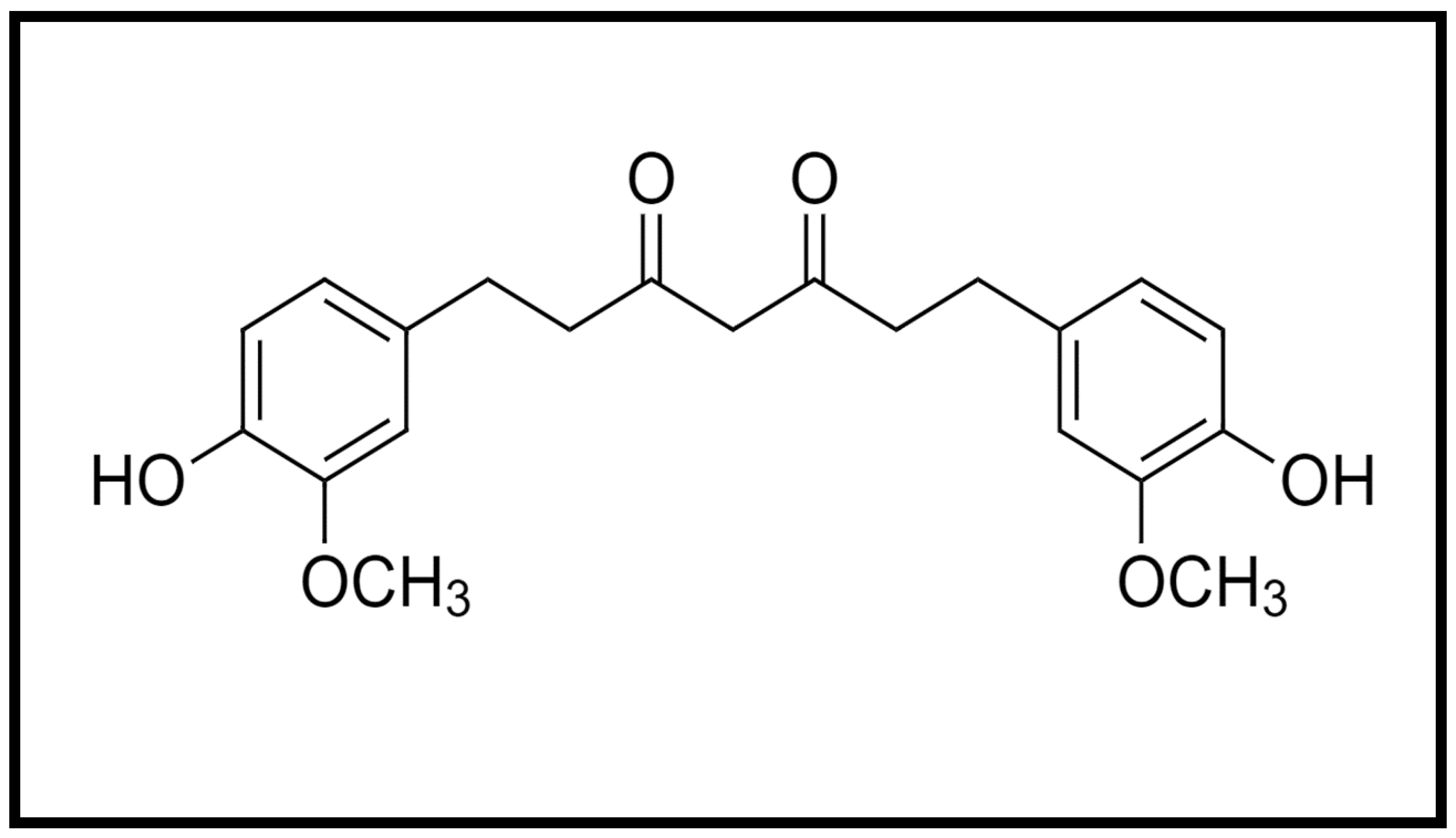
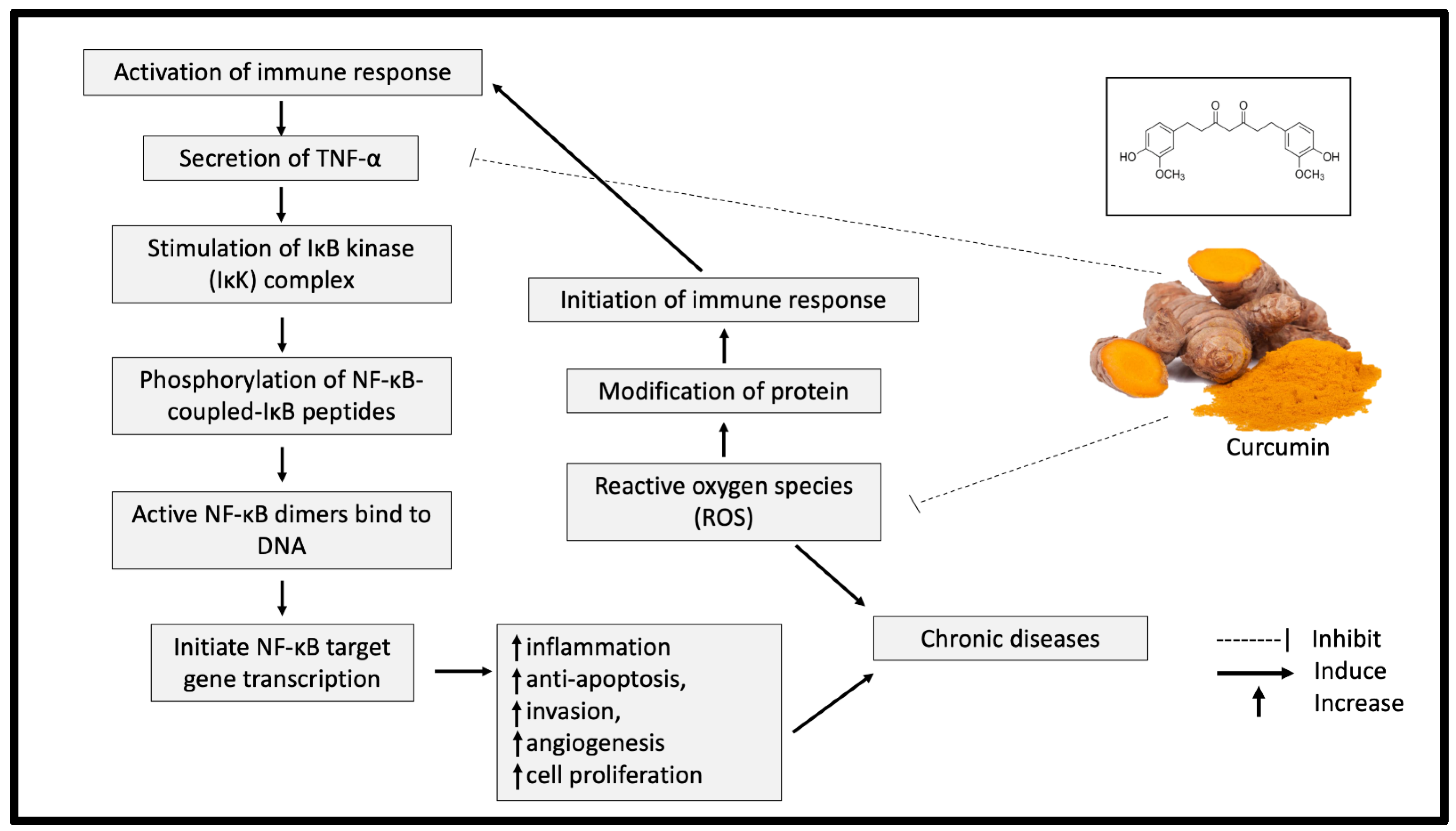
| Type of Model | Treatment and Treatment Duration | Findings | References |
|---|---|---|---|
| A randomised, double-blind, placebo-controlled trial on 60 women with PCOS aged from 18 to 40 years old | 1. 500 mg/day curcumin 2. Placebo Orally 12 weeks | Curcumin significantly reduced the following parameters compared with the placebo: 1. weight and BMI; 2. fasting glucose; 3. serum insulin; 4. HOMA-IR (insulin resistance); 5. total cholesterol; 6. LDL-cholesterol; 7. total-/HDL cholesterol ratio Curcumin significantly increased the following parameter compared with the placebo: 1. HDL-cholesterol levels; 2. QUICKI (insulin sensitivity); 3. gene expression of peroxisome proliferator-activated receptor-gamma (PPAR-γ); 4. gene expression of low-density lipoprotein receptor (LDLR) | [41] |
| A randomised, double-blind, placebo-controlled clinical trial on 67 women with PCOS aged from 18 to 49 years old | 1. 500 mg curcumin powder in a capsule 2. Placebo (maltodextrin) capsules Orally 3 times daily for 12 weeks | -Decreased fasting plasma glucose (FPG) and dehydroepiandrosterone levels in the curcumin-treated group compared with the placebo -Statistically non-significant increase in oestradiol levels -No changes in fasting insulin, LH and FSH, homeostatic model assessment of insulin resistance (HOMA-IR), quantitative insulin sensitivity check index (QUICKI), BMI and waist circumference among different groups | [42] |
| A randomised, double-blind, placebo-controlled clinical trial on 60 overweight or obese women with PCOS | 1. 500 mg curcumin twice daily 2. Placebo Orally 6 weeks | -Intragroup analysis of serum insulin decreased, whereas QUICKI increases significantly in the curcumin-treated group -No significant changes were recorded in all parameters between the curcumin-treated and placebo groups, including FBS, insulin, HOMA-IR, QUICKI, total cholesterol, triglyceride, LDL and HDL and high-sensitivity C-reactive protein (hs-CRP) levels | [43] |
| A randomised, double-blind, placebo-controlled clinical trial on 72 women with PCOS | 1. 1500 mg curcumin 3 times daily 2. Placebo Orally 3 months | Significantly increased gene expression of peroxisome proliferator-activated receptor γ coactivator 1α and the glutathione peroxidase enzyme activity -Non-significantly increased gene expression of sirtuin-1 and activity of the superoxide dismutase (SOD) enzyme | [44] |
| Type of Model | Treatment and Treatment Duration | Findings | References |
|---|---|---|---|
| Adult female Wistar rats treated with oestradiol valerate to induce PCOS | 1. 100 mg/kg curcumin 2. 300 mg/kg curcumin Method of administration not specified 14 days | -Reduced number of insulin resistance index (HOMA-IR decreased, QUICKI increased) -Reduced interleukin-6 and C-reactive protein -Reduced necrotic liver cells | [45] |
| Prepuberal BALB/c female mice treated with DHEA to induce PCOS | 5.4 mg/100 g curcumin in the form of curcumin-loaded super-paramagnetic iron oxide (Fe3O4) nanoparticles Intraperitoneally 20 days | -Reduced ovarian volume and total number of primary, secondary, antral and primordial follicles compared to the PCOS and vehicle groups -Significantly decreased Bcl-2-associated X protein (BAX) and levels of expression of Caspase3 (CASP3) protein; increased levels of B-cell lymphoma 2 (Bcl2) expression and moderated apoptosis in granulosa cells compared with PCOS group | [46] |
| Adult female Wistar rats treated with letrozole to induce PCOS | 1. Nanocurcumin (50, 100 and 200 mg/kg) 2. Clomiphene citrate (1 mg/kg) Orally 15 days | -Nanocurcumin (50 mg/kg) and clomiphene citrate attenuated the PCOS-induced reduction in oestradiol and progesterone levels -Nanocurcumin (50 and 100 mg/kg) and clomiphene citrate attenuated the PCOS-induced testosterone increment -Nanocurcumin (50 mg/kg) and clomiphene citrate improved triglyceride, total cholesterol, and LDL and HDL cholesterol levels -All doses of curcumin and clomiphene citrate reduced the PCOS-induced increment of fasting blood glucose and insulin -Clomiphene citrate and curcumin alleviated insulin resistance -Clomiphene citrate and curcumin decreased the MDA level and increase GSH and SOD activity -Clomiphene citrate and curcumin decreased TNF-α levels -Clomiphene citrate and curcumin increased the protein expression of PI3K/AKT/mTOR levels -Treatment with nanocurcumin showed thickened granulosa cells and the appearance of oocytes in a dose-dependent manner -Nano curcumin treatment and clomiphene retained the pancreatic tissue integrity and caused a gradual increase in the area of the islet and count of β-cells | [47] |
| Adult female Wistar rats treated with oestradiol valerate to induce PCOS | Curcumin (100, 200, 300 and 400 mg/kg) Intraperitoneally 14 days | -Significant reduction in thickness of theca layer and increased corpus luteum diameter in the curcumin-treated group compared with the PCOS group -Curcumin decreased the IL-6 and CRP levels -Curcumin decreased TNF-α in the granulosa layer and follicular fluid | [48] |
| Adult female Wistar rats treated with letrozole to induce PCOS | 1. Curcumin (100 and 200 mg/kg) 2. 1 mg/kg clomiphene citrate Orally 15 days | -Curcumin significantly inhibited the decrease in uterine weight -Clomiphene citrate and 100 and 200 mg/kg curcumin reversed the disturbance in testosterone and progesterone levels in PCOS-induced rats, whereas only clomiphene and 200 mg/kg curcumin effectively normalised the oestrogen level -Both doses of curcumin reduced fasting blood glucose and HbA1c levels -Clomiphene and 100 and 200 mg/kg curcumin decreased triglyceride, total cholesterol and LDL levels, whereas only 200 mg/kg curcumin increased HDL level -100 and 200 mg/kg curcumin increased SOD and CAT activity, and only 200 mg/kg curcumin decreased the TBARS level and increased GSH level -Clomiphene increased catalase activity, reduced TBARS and showed no effect on GSH and SOD -Clomiphene citrate and both curcumin doses resulted in the disappearance of cysts and the appearance of healthy follicles and corpora lutea | [49] |
| Type of Model | Treatment and Treatment Duration | Findings | References |
|---|---|---|---|
| D-galactose-induced premature ovarian failure (POF) in mice | 100 mg/kg/day curcumin Intraperitoneally 42 days | -Increased progesterone and oestrogen levels while decreasing FSH and LH levels -Increased SOD and decreased the MDA and SOD2 levels and CAT mRNA expression -Increased primordial follicles -Decreased 8-OhdG, 4-HNE, NTY and senescence-associated protein P16 expression levels -Increased AMH expression levels -Reduced apoptosis in granulosa cells -Increased p-Akt, Nrf2 and HO-1 protein expression levels -Reduced cleaved caspase-3 and -9 protein expression levels | [65] |
| Porcine ovarian granulosa cells | Curcumin medium at 0, 1, 10 and 100 μg/mL 2 days | -Reduced PCNA and its mRNA, increased Bax and its mRNA, reduced cell viability and stimulated progesterone and testosterone release | [67] |
| Rat model of ovarian ischemia–reperfusion injury | Curcumin at 200 mg/kg Administered intraperitoneally with reperfusion Group 1: 2 h ischemia and 2 h reperfusion Group 2: 4 h ischemia and 4 h reperfusion Subgroup: (1) Sham: abdominal incision with no ischemia/perfusion (2) Control: abdominal incision with ischemia/perfusion (3) Curcumin: abdominal incision with ischemia/perfusion and curcumin at 200 mg/kg | Group 1: -No significant differences were observed between nitric oxide (NO), NO synthase (NOS), xanthine oxidase (XO), total antioxidant status (TAS) and total oxidant status (TOS) -Significantly higher ovary histological grade in the control and curcumin subgroups compared with the sham subgroup Group 2: -Significantly higher TOS and TAS in the control group than in the sham and curcumin groups -Significantly higher histological grade in the control and curcumin subgroups compared with the sham subgroup -No change in NO, NOS or XO levels | [68] |
| Rat model of ovarian ischemia–reperfusion injury | 1. 100 mg/kg curcumin 2. 1 mg/kg nano curcumin Intraperitoneal Group SSG: laparotomy only Group I: 3 h ischemia only Group I/R: 3 h ischemia and 3 h reperfusion Group I/C: 3 h ischemia only and 100 mg/kg curcumin Group I/R/C: 3 h ischemia, 3 h reperfusion and 100 mg/kg curcumin Group I/NC: 3 h ischemia only and 1 mg/kg nano curcumin Group I/R/NC: 3 h ischemia, 3 h reperfusion and 1 mg/kg nano curcumin | -Nanocurcumin-treated animals showed significantly improved development of ischemia and reperfusion tissue injury compared with other groups -I/R/NC group showed significantly higher superoxide dismutase values, total glutathione, glutathione peroxidase, glutathione reductase and glutathione S-transferase than other groups (p < 0.05) -I/R/NC group showed a significantly lower value of nitric oxide synthase, malondialdehyde, myeloperoxidase and 8-hydroxy-2 deoxyguanine levels compared with other Groups -No difference in all biochemical parameters in other groups | [69] |
| CBA female mice immunised with an extract from the ovaries of outbred albino mice to induce immune disease of the ovaries (model of autoimmune disease in women) | Curcumin at 100 μg/g (four times a week) Intragastric | -The percentage of oocytes in metaphases I and II: Immunised mice: the numbers of oocytes with dissolved germinal vesicle (metaphase I) and the formed polar body (metaphase II) decreased significantly Curcumin-treated mice: the level of oocytes in metaphases I and II increased significantly compared with the immunised group -The number of apoptotic and necrotic cells in the thymus, spleen and lymph node: No changes were recorded in apoptosis cell levels in all groups Immunised mice: significantly increased necrotic cell number in thymus, spleen and lymph node Curcumin-treated mice: Decreased necrotic cell number in thymus, spleen and lymph node compared with the immunised group -Percentage of blood stab neutrophils: Immunised group—increased Curcumin-treated group—decreased -Correlation between parameters of death of immunocompetent cells and number of oocytes resuming meiosis: Significantly negative correlation in immunocompetent cell necrosis and percentage of oocytes in metaphase I for spleen and lymph nodes only | [70] |
| Female Kunming mice injected with 8 mg/kg sodium arsenite to induce ovarian oxidative stress | 0, 100, 150 or 200 mg/kg curcumin once a day Intragastric 21 days | -All doses of curcumin reduced the sodium arsenite-induced increment in ROS value -All doses of curcumin reduced the sodium arsenite-induced increment in MDA value -All doses of curcumin increased the sodium arsenite-induced decrease in SOD value -Decreased sodium arsenite treatment; no change in comparison with curcumin treatment -All doses of curcumin reduced the sodium arsenite-induced increment in the number of atretic follicles. No changes were recorded in primordial, primary and secondary follicles -Curcumin prevented the inhibition of proliferation of granular cells in the sodium arsenite group -p66Shc expression upregulated under oxidative stress was lowered by curcumin | [71] |
| Type of Model | Treatment and Treatment Duration | Findings | References |
|---|---|---|---|
| Hetero-transplantation of endometrium tissue of ovariectomised donor mice into the peritoneal cavity containing subcutaneous implant of oestradiol-17β of recipient BALB/c mice | Model: 12, 24 and 48 mg/kg/day via intraperitoneal injection Therapeutic: 48 mg/kg/day curcumin 5 mg/kg, twice/day celecoxib Intraperitoneal injection Mice model: Administered once daily for 1 day prior to the inoculation of the endometrium and continued for another 3 days Therapeutic model (no pre-treatment given prior to inoculation): Curcumin once daily and celecoxib twice daily for the next 5 days | -Pre-treatment with curcumin showed obliteration of glandular regions with sparse infiltration inflammatory cells in the intestinal mucosal layer Curcumin treatment: -Downregulated MMP-3 activity during early regression of endometriosis in a dose-dependent manner -Decreased activity of MMP-3 and increased IκB-α expression leads to reduced NF-κB translocation within the nucleus -Increased in TUNEL positive cells in pre-treated endometriotic tissues as compared with untreated tissue -Induced Cyt-c release and caspase-9 expression suggested the involvement of mitochondrial pathway in stimulating apoptosis -Increased the size of mitochondria, decreased the expression of Cyt-c and increased the expression of Bax within the mitochondria -Distinct lesion of endometriosis was significantly reduced in number and volume by curcumin and moderately by celecoxib -P53 protein expression was upregulated during curcumin treatment but not during celecoxib treatment -Both curcumin and celecoxib activated phosphorylated p38 MAP kinase -The apoptotic response was triggered by increased caspase-9 expression by curcumin and celecoxib compared with normal samples | [74] |
| Autotransplantation of endometrium tissue of the left horn of the uterus to the abdominal wall and intestinal mesentery of Sprague-Dawley rats | 48 mg/kg/day of curcumin 7.2 mg/kg/day of danazol 0.3 mL of ethanol 50% (vehicle) Intraperitoneal injection 4 weeks | -Serum level of leptin in the curcumin treatment group was significantly higher than in all the study groups except the danazol-treated group -No significant difference in serum levels of resistin, homocysteine and CA 125 in the danazol and curcumin treatment groups -Significantly lower serum level TAC in the curcumin-treated group compared with control and danazol-treated group | [77] |
| Endometriosis with varying severity developed in mice by peritoneal implantation of uterine fragments | (1) 16, 32 and 48 mg/kg/day curcumin (2) 48 mg/kg/day curcumin for therapeutic model (3) PBS (vehicle) Intraperitoneal injection Daily for 3 days Therapeutic model: -Daily for 10 days -Daily for 20 days from day 15 post-endometriosis | At doses of 16, 32 and 48 mg/kg: -Curcumin decreased the secreted matrix metalloproteinase (MMP)-9 activity by 50%, 70% and 80% and the synthesis of MMP-9 decrease significantly by 60%, 70% and 90%, respectively In the therapeutic model: -Curcumin decreased the secreted MMP-9 activity by 45% and 85%, inhibited TNF-α and elevated TIMP-1 -Curcumin decreased lipid peroxidation and protein carbonylation | [78] |
| Autotransplantation of endometrium tissue during oestrus stage in Wistar rats | 50, 100 and 150 mg/kg/d curcumin 0.5% sodium carboxymethyl cellulose solution (vehicle) Intragastric injection Daily for 4 weeks | -Hyperplasia in ectopic tissue was halted -Disappearance of the ectopic gland tissue with the narrowed lumen and sparse cells -Microvessel density (MVD) was higher in ectopic endometrium -In eutopic endometrium, all treated groups showed no significant difference compared with the normal group -Eutopic and ectopic endometrium expression of VEGF protein in the model rat group was higher than in the normal group | [79] |
| Dimethyl sulfoxide (DMSO) autotransplantation of endometrium tissue from the right horn of the uterus and placed be-tween the peritoneum and muscle of ovariectomised albino Wistar rats | DMSO as vehicle 100 mg/kg of curcumin 100 mg/kg of deferoxamine + curcumin Intragastric Group A: Water for injection daily for 3 days and DMSO Group B: Curcumin Group C: Deferoxamine at 6 h interval for 3 days and curcumin Daily for 20 days | -Endometriotic-like implant sizes were significantly reduced in groups B and C compared with group A (control) -Stroma and glands with surrounding fibrous connective tissue were present and well vascularised in the ectopic endometrium -Cytokeratin-7 antibodies displayed an immunoreactivity in glandular epithelial cells of both eutopic and ectopic endometrium -Blood iron levels were not significantly different between the treatment groups according to atomic absorption spectrophotometry results | [80] |
| Hetero-transplant of the endometrium of the uterine horn of donor mice into the peritoneal cavity containing subcutaneous implant of oestradiol-17β pellet of recipient BALB/c mice | 48 mg/kg/day of curcumin Intraperitoneal injection Daily for 3 days | -No changes were seen in MMP-2 activity for the first 24 h -Both synthesised and secreted proMMP-2 activity were increased after 48 h -Curcumin suppressed the pro-MMP-2 activity, and progression of endometriosis was delayed -Activation of pro-MMP-2 was inhibited; hence, MMP-2 activity was halted by curcumin during the early stage of endometriosis -Upregulation of TIMP-2 inhibited MMP-2 activity during regression of endometriosis by curcumin -MT1MMP regulated MMP-2 activity, as confirmed by immunoblotting. Curcumin downregulated MT1MMP expression by 40% compared with the normal sample -Complex formation between MMP-2 and TIMP-2 was significantly increased in the curcumin-treated group, which inhibited MMP-2 activity and endometriosis progression | [81] |
| Type of Model | Treatment and Treatment Duration | Findings | References |
|---|---|---|---|
| Cell culture of human endometriotic stromal cells from women with stage III/IV endometriosis | (1) 1, 10, 30 and 50 μM of curcumin (2) 30 μM of curcumin + 15 ng/mL of TNF-α primary (1) 24 h (2) Curcumin for 1 h and stimulated with TNF-α for 15 h for ELISA | -Curcumin did not affect the viability of endometriotic stromal cells at doses up to 50 μM -Pre-treatment with curcumin significantly suppressed TNF-α-induced ICAM-1 and VCAM-1 mRNA expression in a dose-dependent manner -Curcumin significantly suppressed TNF-α-induced ICAM-1 VCAM-1 cell surface expression in a dose-dependent manner -Pre-treatment with curcumin (30 μM) markedly suppressed TNF-α-induced total cellular protein expression of ICAM-1 and VCAM-1 -Curcumin (30 μM) suppressed the TNF-α-induced IL-6, IL-8 and MCP-1 from endometriotic stromal cells -Pre-treatment with curcumin (30 μM) blocked the TNF-α-induced nuclear translocation of NF-κB p65 from the cytosol into the nucleus and strongly inhibited TNF-α-induced phosphorylation and degradation of IκB -Curcumin (30 μM) significantly reduced the density of the NF-κB shifted band that was induced by TNF-α | [82] |
| Primary cell culture of human endometriotic stromal cells from women with endometriosis (EESCs) and normal endometrial stromal cells (NESCs) | (1) 1, 5, 10, 20 and 40 μg/mL of curcumin (2) DMSO as vehicle 24, 48 and 72 h | -100% apoptotic cell deaths at 40 μg/mL of curcumin but < 20 μg/mL had no significant apoptotic effects on endometrial stromal cells (ESCs) -IL-6, IL-8, IP-10, G-CSF, MCP-1 and RANTES were highly expressed in EESCs -IL-10 and IL-12 expressions were not different in both EESCs and NESCs -Curcumin treatment significantly inhibited secretion of IL-6, IL-8, IP-10, G-CSF, MCP-1 and RANTES in EESCs after 48 h -Curcumin significantly promoted IL-10 and IL-12 secretion in EESCs after 48 h -IL-17 was completely absent in the media after treatment with curcumin for 24 and 48 h -Curcumin inhibited the phosphorylation of IKKα, IKKβ and NF-κB in EESCs -Curcumin treatment significantly inhibited the phosphorylation of JNK and STAT3 in EESCs -JNK expression significantly decreased after curcumin treatment | [83] |
| Bovine cumulus–oocyte complex cultured in a TCM 199 plus peritoneal fluid of infertile women with endometriosis | (1) TCM199 (control) (2) TCM199 + peritoneal fluid (PF) (3) TCM199 + PF + 0.2 mL curcumin 24 h | -GDF-9 and Kit Ligand expressions were higher in the treated group than in the non-treated group but decreased compared with the control group -TNFα expression in bovine COC cultured in PF from infertile women with endometriosis was reduced compared with those in the non-treated group -TNF-α was absent in the control group | [84] |
| Endometriotic stromal cells, normal endometrial stromal cells, endometriotic epithelial cells and normal endometrial epithelial cells | 10, 30 and 50 μM of curcumin (2) 10 μL of WST-8 0, 24, 48, 72 and 96 h of curcumin treatment Oestradiol (E2) assay: 24 h incubation period ELISA: WST-8 for 4 h | -E2 value of endometriotic epithelial cells was higher than the endometriotic stromal cells -Expression of E2 in normal endometrial stromal and epithelial cells was extremely low -WST-8 result showed that compared with endometrial stromal cells, ectopic endometriotic stromal cells had a higher growth rate -The number of endometriotic stromal cells was reduced, and cell growth slowed compared with the 0 μmol/L group after curcumin treatment -Compared with the 0 μmol/L group, E2 level was lower after treatment with curcumin, especially in the 30 and 50 μmol/L groups | [85] |
| Primary cell culture of human endometriotic stromal cells from women with endometriosis and eutopic endometrial stromal cells with no endometriosis | 0, 20 and 50 μmol/l of curcumin 48 h 72 h (for immunostaining assay) | -Cell morphology between ectopic and eutopic stromal cells was similar (spindle-shaped, abundant cytoplasm and oval-shaped nucleus); positive vimentin biomarker -Altered morphology, reduced permeability and increased cell suspension at 20 μmol/l curcumin after 48 h -Adherent cell decreased; a significant increase in cell suspension and presence of cell debris at 50 μmol/l curcumin after 48 h -Curcumin decreased the eutopic and ectopic cell growth -At 20 and 50 μmol/l curcumin after 48 h, endometriotic and endometrial stromal cells demonstrated increased percentages of G1-phase cells and decreased percentages of S-phase cells -Treatment with 20 and 50 μmol/l curcumin decreased the proportion of VEGF positive expression compared with the 0 μmol/l group -More in late apoptosis than early apoptosis in both endometriotic and endometrial cells | [86] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kamal, D.A.M.; Salamt, N.; Yusuf, A.N.M.; Kashim, M.I.A.M.; Mokhtar, M.H. Potential Health Benefits of Curcumin on Female Reproductive Disorders: A Review. Nutrients 2021, 13, 3126. https://doi.org/10.3390/nu13093126
Kamal DAM, Salamt N, Yusuf ANM, Kashim MIAM, Mokhtar MH. Potential Health Benefits of Curcumin on Female Reproductive Disorders: A Review. Nutrients. 2021; 13(9):3126. https://doi.org/10.3390/nu13093126
Chicago/Turabian StyleKamal, Datu Agasi Mohd, Norizam Salamt, Allia Najmie Muhammad Yusuf, Mohd Izhar Ariff Mohd Kashim, and Mohd Helmy Mokhtar. 2021. "Potential Health Benefits of Curcumin on Female Reproductive Disorders: A Review" Nutrients 13, no. 9: 3126. https://doi.org/10.3390/nu13093126
APA StyleKamal, D. A. M., Salamt, N., Yusuf, A. N. M., Kashim, M. I. A. M., & Mokhtar, M. H. (2021). Potential Health Benefits of Curcumin on Female Reproductive Disorders: A Review. Nutrients, 13(9), 3126. https://doi.org/10.3390/nu13093126





