The Effects of Korean Red Ginseng on Biological Aging and Antioxidant Capacity in Postmenopausal Women: A Double-Blind Randomized Controlled Study
Abstract
:1. Introduction
2. Materials and Methods
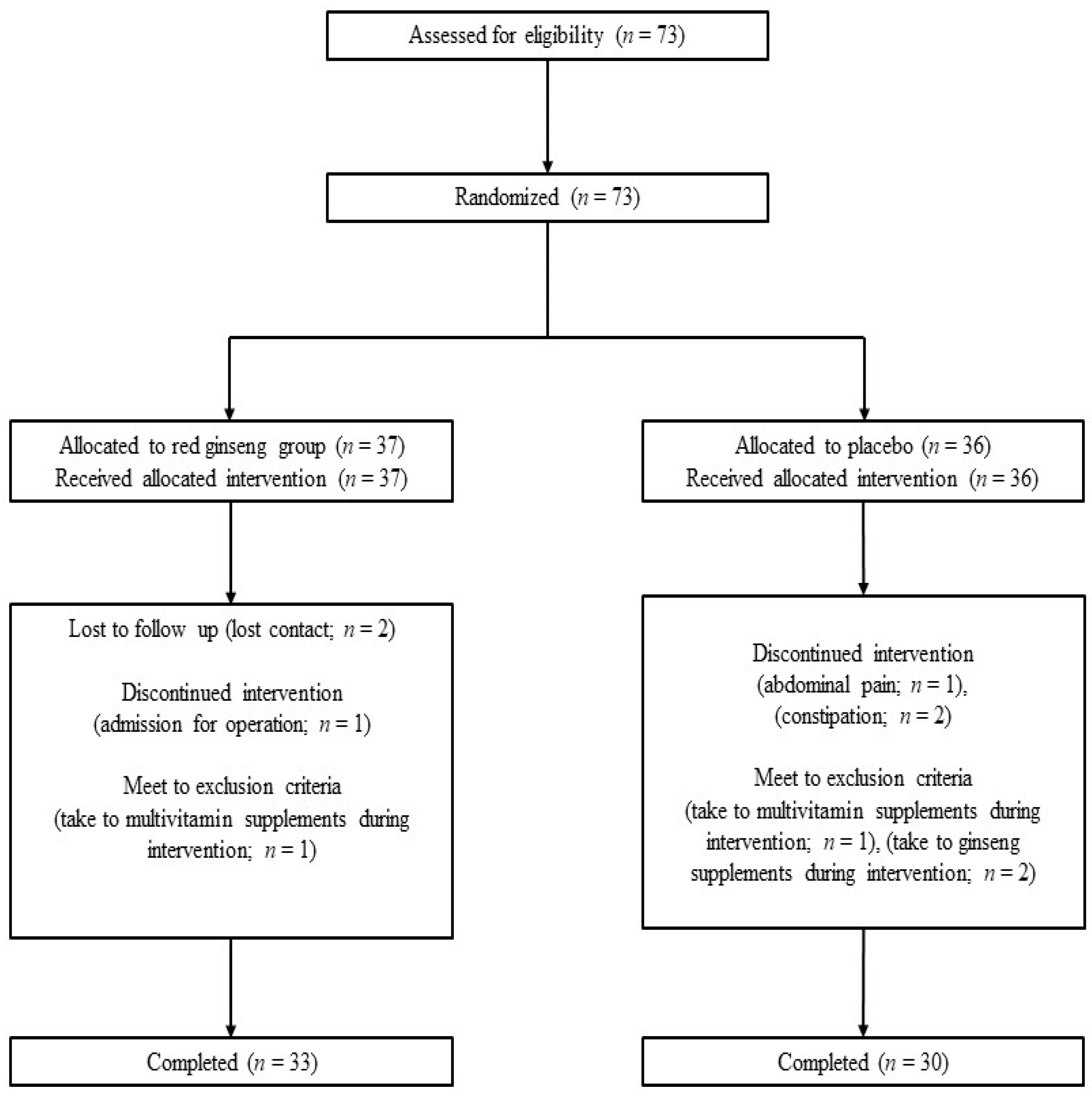
2.1. Study Design and Participants
2.2. Randomization
2.3. Korean Red Ginseng and Placebo Supplements
2.4. Data Collection
2.5. Dependent Variable Assessment
2.5.1. Total Antioxidant Status
2.5.2. Mitochondria DNA Copy Number
2.5.3. Fatigue Severity Scale
2.6. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Baeg, I.H.; So, S.H. The world ginseng market and the ginseng (Korea). J. Ginseng Res. 2013, 37, 1–7. [Google Scholar] [CrossRef] [Green Version]
- So, S.H.; Lee, J.W.; Kim, Y.S.; Hyun, S.H.; Han, C.K. Red ginseng monograph. J. Ginseng Res. 2018, 42, 549–561. [Google Scholar] [CrossRef] [PubMed]
- Ossewaarde, M.E.; Bots, M.L.; Verbeek, A.L.; Peeters, P.H.; van der Graaf, Y.; Grobbee, D.E.; van der Schouw, Y.T. Age at menopause, cause-specific mortality and total life expectancy. Epidemiology 2005, 16, 556–562. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, H.M.; Park, J.; Ryu, S.Y.; Kim, J. The effect of menopause on the metabolic syndrome among Korean women: The Korean national health and nutrition examination survey, 2001. Diabetes Care 2007, 30, 701–706. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Auro, K.; Joensuu, A.; Fischer, K.; Kettunen, J.; Salo, P.; Mattsson, H.; Niironen, M.; Kaprio, J.; Eriksson, J.G.; Lehtimaki, T.; et al. A metabolic view on menopause and ageing. Nat. Commun. 2014, 5, 4708. [Google Scholar] [CrossRef] [Green Version]
- Balaban, R.S.; Nemoto, S.; Finkel, T. Mitochondria, oxidants, and aging. Cell 2005, 120, 483–495. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, R.; Wang, Y.; Ye, K.; Picard, M.; Gu, Z. Independent impacts of aging on mitochondrial DNA quantity and quality in humans. BMC Genom. 2017, 18, 890. [Google Scholar] [CrossRef] [Green Version]
- Lopez-Otin, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The hallmarks of aging. Cell 2013, 153, 1194–1217. [Google Scholar] [CrossRef] [Green Version]
- Liguori, I.; Russo, G.; Curcio, F.; Bulli, G.; Aran, L.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; et al. Oxidative stress, aging, and diseases. Clin. Interv. Aging 2018, 13, 757–772. [Google Scholar] [CrossRef] [Green Version]
- Pandey, K.B.; Rizvi, S.I. Markers of oxidative stress in erythrocytes and plasma during aging in humans. Oxid. Med. Cell. Longev. 2010, 3, 2–12. [Google Scholar] [CrossRef]
- Seo, S.K.; Hong, Y.; Yun, B.H.; Chon, S.J.; Jung, Y.S.; Park, J.H.; Cho, S.; Choi, Y.S.; Lee, B.S. Antioxidative effects of Korean red ginseng in postmenopausal women: A double-blind randomized controlled trial. J. Ethnopharmacol. 2014, 154, 753–757. [Google Scholar] [CrossRef]
- Erel, O. A novel automated direct measurement method for total antioxidant capacity using a new generation, more stable abts radical cation. Clin. Biochem. 2004, 37, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Krupp, L.B.; LaRocca, N.G.; Muir-Nash, J.; Steinberg, A.D. The fatigue severity scale. Application to patients with multiple sclerosis and systemic lupus erythematosus. Arch. Neurol. 1989, 46, 1121–1123. [Google Scholar] [CrossRef] [PubMed]
- Mills, R.; Young, C.; Nicholas, R.; Pallant, J.; Tennant, A. Rasch analysis of the fatigue severity scale in multiple sclerosis. Mult. Scler. 2009, 15, 81–87. [Google Scholar] [CrossRef]
- Mattsson, M.; Moller, B.; Lundberg, I.; Gard, G.; Bostrom, C. Reliability and validity of the fatigue severity scale in swedish for patients with systemic lupus erythematosus. Scand. J. Rheumatol. 2008, 37, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Herlofson, K.; Larsen, J.P. Measuring fatigue in patients with parkinson’s disease—The fatigue severity scale. Eur. J. Neurol. 2002, 9, 595–600. [Google Scholar] [CrossRef] [PubMed]
- Shin, E.J.; Jo, S.; Choi, S.; Cho, C.W.; Lim, W.C.; Hong, H.D.; Lim, T.G.; Jang, Y.J.; Jang, M.; Byun, S.; et al. Red ginseng improves exercise endurance by promoting mitochondrial biogenesis and myoblast differentiation. Molecules 2020, 25, 865. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, J.K.; Shim, J.Y.; Cho, A.R.; Cho, M.R.; Lee, Y.J. Korean red ginseng protects against mitochondrial damage and intracellular inflammation in an animal model of type 2 diabetes mellitus. J. Med. Food 2018, 21, 544–550. [Google Scholar] [CrossRef]
- Jung, D.H.; Lee, Y.J.; Kim, C.B.; Kim, J.Y.; Shin, S.H.; Park, J.K. Effects of ginseng on peripheral blood mitochondrial DNA copy number and hormones in men with metabolic syndrome: A randomized clinical and pilot study. Complement. Ther. Med. 2016, 24, 40–46. [Google Scholar] [CrossRef]
- Taherzadeh-Fard, E.; Saft, C.; Akkad, D.A.; Wieczorek, S.; Haghikia, A.; Chan, A.; Epplen, J.T.; Arning, L. Pgc-1alpha downstream transcription factors nrf-1 and tfam are genetic modifiers of huntington disease. Mol. Neurodegener. 2011, 6, 32. [Google Scholar] [CrossRef] [Green Version]
- Gureev, A.P.; Shaforostova, E.A.; Popov, V.N. Regulation of mitochondrial biogenesis as a way for active longevity: Interaction between the nrf2 and pgc-1alpha signaling pathways. Front. Genet. 2019, 10, 435. [Google Scholar] [CrossRef] [Green Version]
- Kim, M.J.; Koo, Y.D.; Kim, M.; Lim, S.; Park, Y.J.; Chung, S.S.; Jang, H.C.; Park, K.S. Rg3 improves mitochondrial function and the expression of key genes involved in mitochondrial biogenesis in c2c12 myotubes. Diabetes Metab. J. 2016, 40, 406–413. [Google Scholar] [CrossRef]
- Wallace, D.C. A mitochondrial paradigm of metabolic and degenerative diseases, aging, and cancer: A dawn for evolutionary medicine. Annu. Rev. Genet. 2005, 39, 359–407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, Y.S.; Kim, Y.H.; Noh, J.R.; Cho, E.S.; Park, J.H.; Son, H.Y. Protective effect of Korean red ginseng against aflatoxin b1-induced hepatotoxicity in rat. J. Ginseng Res. 2011, 35, 243–249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramesh, T.; Kim, S.W.; Hwang, S.Y.; Sohn, S.H.; Yoo, S.K.; Kim, S.K. Panax ginseng reduces oxidative stress and restores antioxidant capacity in aged rats. Nutr. Res. 2012, 32, 718–726. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.Y.; Yoon, B.R.; Lee, Y.J.; Lee, J.S.; Hong, H.D.; Lee, Y.C.; Kim, Y.C.; Cho, C.W.; Kim, K.T.; Lee, O.H. Inhibitory effect of high temperature- and high pressure-treated red ginseng on exercise-induced oxidative stress in icr mouse. Nutrients 2014, 6, 1003–1015. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.Y.; Park, J.Y.; Kang, H.J.; Kim, O.Y.; Lee, J.H. Beneficial effects of Korean red ginseng on lymphocyte DNA damage, antioxidant enzyme activity, and ldl oxidation in healthy participants: A randomized, double-blind, placebo-controlled trial. Nutr. J. 2012, 11, 47. [Google Scholar] [CrossRef]
- Bak, M.J.; Jun, M.; Jeong, W.S. Antioxidant and hepatoprotective effects of the red ginseng essential oil in h(2)o(2)-treated hepg2 cells and ccl(4)-treated mice. Int. J. Mol. Sci. 2012, 13, 2314–2330. [Google Scholar] [CrossRef] [Green Version]
- Park, H.M.; Kim, S.J.; Mun, A.R.; Go, H.K.; Kim, G.B.; Kim, S.Z.; Jang, S.I.; Lee, S.J.; Kim, J.S.; Kang, H.S. Korean red ginseng and its primary ginsenosides inhibit ethanol-induced oxidative injury by suppression of the mapk pathway in tib-73 cells. J. Ethnopharmacol. 2012, 141, 1071–1076. [Google Scholar] [CrossRef]
- Park, S.H.; Jang, J.H.; Chen, C.Y.; Na, H.K.; Surh, Y.J. A formulated red ginseng extract rescues pc12 cells from pcb-induced oxidative cell death through nrf2-mediated upregulation of heme oxygenase-1 and glutamate cysteine ligase. Toxicology 2010, 278, 131–139. [Google Scholar] [CrossRef]
- Dong, G.Z.; Jang, E.J.; Kang, S.H.; Cho, I.J.; Park, S.D.; Kim, S.C.; Kim, Y.W. Red ginseng abrogates oxidative stress via mitochondria protection mediated by lkb1-ampk pathway. BMC Complement. Altern. Med. 2013, 13, 64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bach, H.V.; Kim, J.; Myung, S.K.; Cho, Y.A. Efficacy of ginseng supplements on fatigue and physical performance: A meta-analysis. J. Korean Med. Sci. 2016, 31, 1879–1886. [Google Scholar] [CrossRef] [PubMed]
- Sung, W.S.; Kang, H.R.; Jung, C.Y.; Park, S.S.; Lee, S.H.; Kim, E.J. Efficacy of Korean red ginseng (panax ginseng) for middle-aged and moderate level of chronic fatigue patients: A randomized, double-blind, placebo-controlled trial. Complement. Ther. Med. 2020, 48, 102246. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Park, K.S. Effects of panax ginseng extract on lipid metabolism in humans. Pharmacol. Res. 2003, 48, 511–513. [Google Scholar] [CrossRef]
- Delui, M.H.; Fatehi, H.; Manavifar, M.; Amini, M.; Ghayour-Mobarhan, M.; Zahedi, M.; Ferns, G. The effects of panax ginseng on lipid profile, pro-oxidant: Antioxidant status and high-sensitivity c reactive protein levels in hyperlipidemic patients in iran. Int. J. Prev. Med. 2013, 4, 1045–1051. [Google Scholar]
- Borras, C.; Gambini, J.; Lopez-Grueso, R.; Pallardo, F.V.; Vina, J. Direct antioxidant and protective effect of estradiol on isolated mitochondria. Biochim. Biophys. Acta 2010, 1802, 205–211. [Google Scholar] [CrossRef] [Green Version]
| KRG | Placebo | p-Value | |
|---|---|---|---|
| N | 33 | 30 | |
| Age, years | 58.7 ± 4.2 | 59.7 ± 4.2 | 0.330 |
| SBP (mmHg) | 120.7 ± 15.3 | 125.0 ± 11.1 | 0.207 |
| DBP (mmHg) | 73.0 ± 11.6 | 77.2 ± 7.9 | 0.104 |
| Waist circumference (cm) | 79.3 ± 6.9 | 80.9 ± 7.2 | 0.371 |
| BMI (kg/m2) | 23.7 ± 2.4 | 23.8 ± 3.1 | 0.844 |
| Luekocyte count (cells/μL) | 4890 ± 931 | 5553 ± 1361 | 0.027 |
| Fasting plasma glucose (mg/dL) | 95 ± 11 | 96 ± 17 | 0.831 |
| Total cholesterol (mg/dL) | 230 ± 31 | 210 ± 41 | 0.034 |
| Triglyceride (mg/dL) | 129 ± 66 | 116 ± 52 | 0.392 |
| HDL cholesterol (mg/dL) | 68 ± 16 | 65 ± 16 | 0.373 |
| LDL cholesterol (mg/dL) | 136 ± 38 | 124 ± 36 | 0.205 |
| AST (U/L) | 27 ± 8 | 25 ± 4 | 0.360 |
| ALT (U/L) | 23 ± 9 | 23 ± 7 | 0.873 |
| Gamma-GT (U/L) | 17 ± 11 | 16 ± 7 | 0.606 |
| Alcohol consumption, n (%) | 3 (9.1) | 5 (16.7) | 0.367 |
| Physical activity, n (%) | 17 (51.5) | 10 (33.3) | 0.145 |
| HTN, n (%) | 4 (12.1) | 4 (13.3) | 0.885 |
| Diabetes, n (%) | 1 (3.0) | 2 (6.7) | 0.498 |
| Dyslipidemia, n (%) | 5 (15.2) | 7 (23.3) | 0.409 |
| TAS (mmol/L) | 1.42 ± 0.16 | 1.44 ± 0.14 | 0.690 |
| mtDNA copy number | 4.35 ± 2.54 | 4.70 ± 3.94 | 0.678 |
| FSS | 34 ± 16 | 32 ± 15 | 0.619 |
| KRG | Placebo | ||||||
|---|---|---|---|---|---|---|---|
| Baseline | 8 Weeks | p-Value | Baseline | 8 Weeks | p-Value | Changed p-Value | |
| BMI (kg/m2) | 23.7 ± 2.4 | 23.7 ± 2.40 | 0.755 | 23.8 ± 3.1 | 23.9 ± 3.00 | 0.811 | |
| Change | 0 ± 0.5 | 0.1 ± 0.1 | 0.972 | ||||
| SBP (mmHg) | 120.7 ± 15.3 | 120.3 ± 14.9 | 0.822 | 125.0 ± 11.1 | 124.5 ± 12.7 | 0.819 | |
| Change | −0.4 ± 9.9 | −0.5 ± 11 | 0.973 | ||||
| DBP (mmHg) | 73.3 ± 11.6 | 74.4 ± 9.3 | 0.255 | 77.2 ± 9.6 | 77.3 ± 7.9 | 0.948 | |
| Change | 1.4 ± 6.7 | 0.1 ± 8 | 0.511 | ||||
| Luekocyte count (cells/μL) | 4890 ± 931 | 4886 ± 1385 | 0.800 | 5553 ± 1361 | 5354 ± 1380 | 0.414 | |
| Change | −4 ± 970 | −199 ± 1317 | 0.505 | ||||
| Fasting plasma glucose (mg/dL) | 95 ± 11 | 95 ± 8 | 0.969 | 96 ± 17 | 96 ± 16 | 0.736 | |
| Change | 0 ± 9 | 0 ± 8 | 0.847 | ||||
| Total cholesterol (mg/dL) | 230 ± 31 | 223 ± 31 | 0.172 | 210 ± 41 | 209 ± 44 | 0.773 | |
| Change | −7 ± 30 | −1 ± 31 | 0.465 | ||||
| Triglyceride (mg/dL) | 129 ± 66 | 137 ± 124 | 0.560 | 116 ± 52 | 107 ± 44 | 0.258 | |
| Change | 8 ± 80 | −9 ± 41 | 0.305 | ||||
| HDLcholesterol (mg/dL) | 68 ± 16 | 67 ± 16 | 0.549 | 65 ± 16 | 63 ± 15 | 0.240 | |
| Change | −1 ± 7 | −2 ± 8 | 0.619 | ||||
| LDL cholesterol (mg/dL) | 136 ± 38 | 127 ± 34 | 0.067 | 124 ± 36 | 128 ± 42 | 0.414 | |
| Change | −9 ± 24 | 4 ± 26 | 0.064 | ||||
| AST (U/L) | 27 ± 8 | 25 ± 4 | 0.334 | 25 ± 4 | 27 ± 6 | 0.103 | |
| Change | −2 ± 8 | 2 ± 6 | 0.082 | ||||
| ALT (U/L) | 23 ± 9 | 22 ± 9 | 0.608 | 23 ± 7 | 24 ± 9 | 0.536 | |
| Change | −1 ± 7 | 1 ± 10 | 0.414 | ||||
| Gamma-GT (U/L) | 17 ± 11 | 19 ± 12 | 0.395 | 16 ± 7 | 16 ± 5 | 0.346 | |
| Change | −2 ± 7 | 0 ± 4 | 0.243 | ||||
| KRG | Placebo | ||||||
|---|---|---|---|---|---|---|---|
| Baseline | 8 Weeks | p-Value | Baseline | 8 Weeks | p-Value | Changed p-Value | |
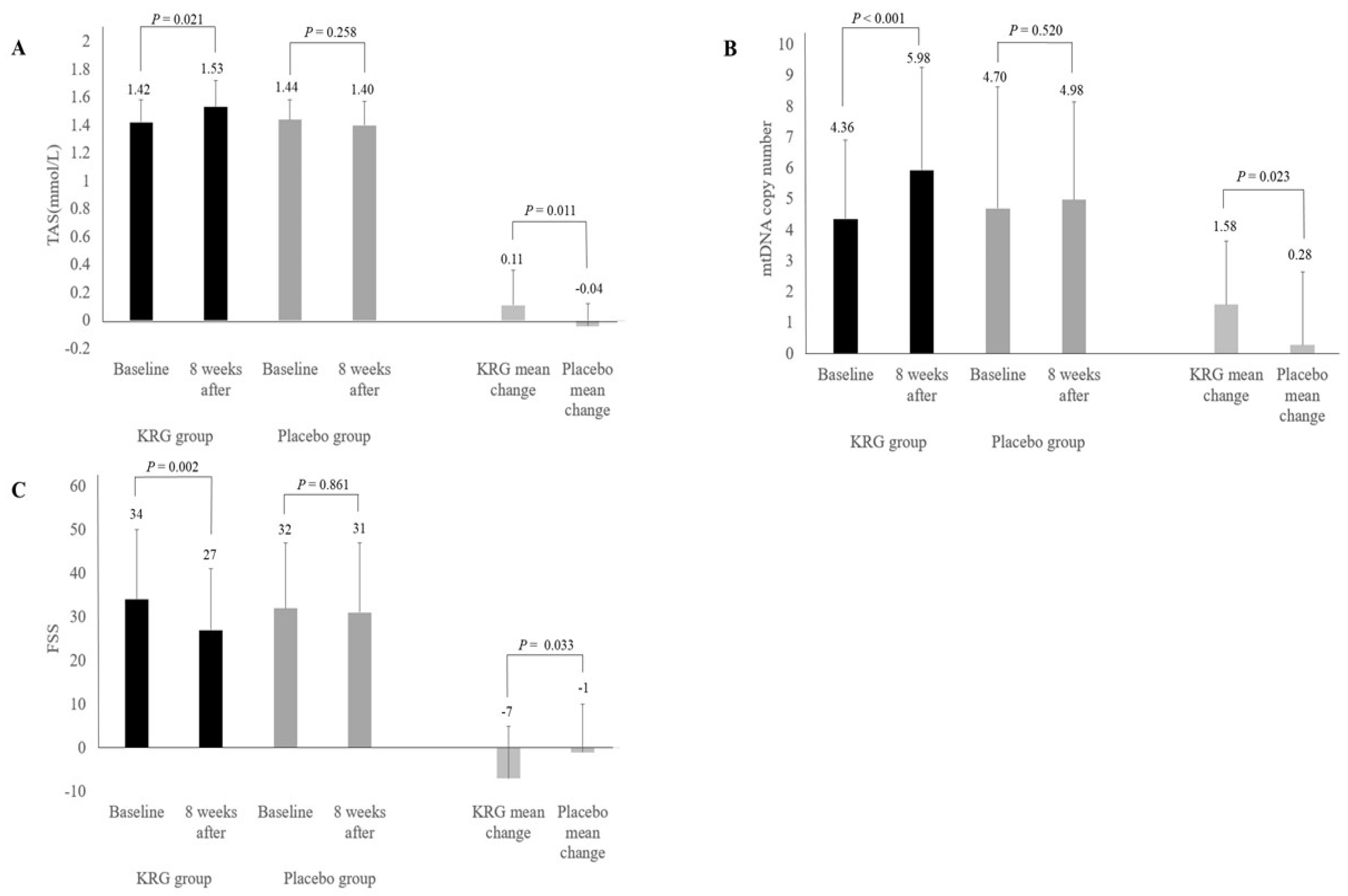
| TAS (mmol/L) | 1.42 ± 0.16 | 1.53 ± 0.19 | 0.021 | 1.44 ± 0.14 | 1.40 ± 0.17 | 0.258 | |
| Change | 0.11 ± 0.25 | −0.04 ± 0.16 | 0.011 | ||||
| mtDNA copy number | 4.36 ± 2.54 | 5.93 ± 3.33 | <0.001 | 4.70 ± 3.94 | 4.98 ± 3.17 | 0.520 | |
| Change | 1.58 ± 2.05 | 0.28 ± 2.36 | 0.023 | ||||
| Fold change | 1.44 ± 0.46 | 1.15 ± 0.40 | 0.012 | ||||
| KRG | Placebo | ||||||
|---|---|---|---|---|---|---|---|
| Baseline | 8 Weeks | p-Value | Baseline | 8 Weeks | p-Value | Changed p-Value | |
| FSS | 34 ± 16 | 27 ± 14 | 0.002 | 32 ± 15 | 31 ± 16 | 0.861 | |
| Change | −7 ± 12 | −1 ± 11 | 0.033 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chung, T.-H.; Kim, J.-H.; Seol, S.-Y.; Kim, Y.-J.; Lee, Y.-J. The Effects of Korean Red Ginseng on Biological Aging and Antioxidant Capacity in Postmenopausal Women: A Double-Blind Randomized Controlled Study. Nutrients 2021, 13, 3090. https://doi.org/10.3390/nu13093090
Chung T-H, Kim J-H, Seol S-Y, Kim Y-J, Lee Y-J. The Effects of Korean Red Ginseng on Biological Aging and Antioxidant Capacity in Postmenopausal Women: A Double-Blind Randomized Controlled Study. Nutrients. 2021; 13(9):3090. https://doi.org/10.3390/nu13093090
Chicago/Turabian StyleChung, Tae-Ha, Ji-Hye Kim, So-Young Seol, Yon-Ji Kim, and Yong-Jae Lee. 2021. "The Effects of Korean Red Ginseng on Biological Aging and Antioxidant Capacity in Postmenopausal Women: A Double-Blind Randomized Controlled Study" Nutrients 13, no. 9: 3090. https://doi.org/10.3390/nu13093090
APA StyleChung, T.-H., Kim, J.-H., Seol, S.-Y., Kim, Y.-J., & Lee, Y.-J. (2021). The Effects of Korean Red Ginseng on Biological Aging and Antioxidant Capacity in Postmenopausal Women: A Double-Blind Randomized Controlled Study. Nutrients, 13(9), 3090. https://doi.org/10.3390/nu13093090






