Consumption of New Zealand Blackcurrant Extract Improves Recovery from Exercise-Induced Muscle Damage in Non-Resistance Trained Men and Women: A Double-Blind Randomised Trial
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design
2.3. Preliminary Measures and Familarisation Session
2.4. Supplement Dosing (Day 1–12)
2.5. Muscle Damage Exercise Protocol (Day 8)
2.6. Criterion Measures (Days 8–12)
2.7. Statistical Analysis
3. Results
3.1. Participants
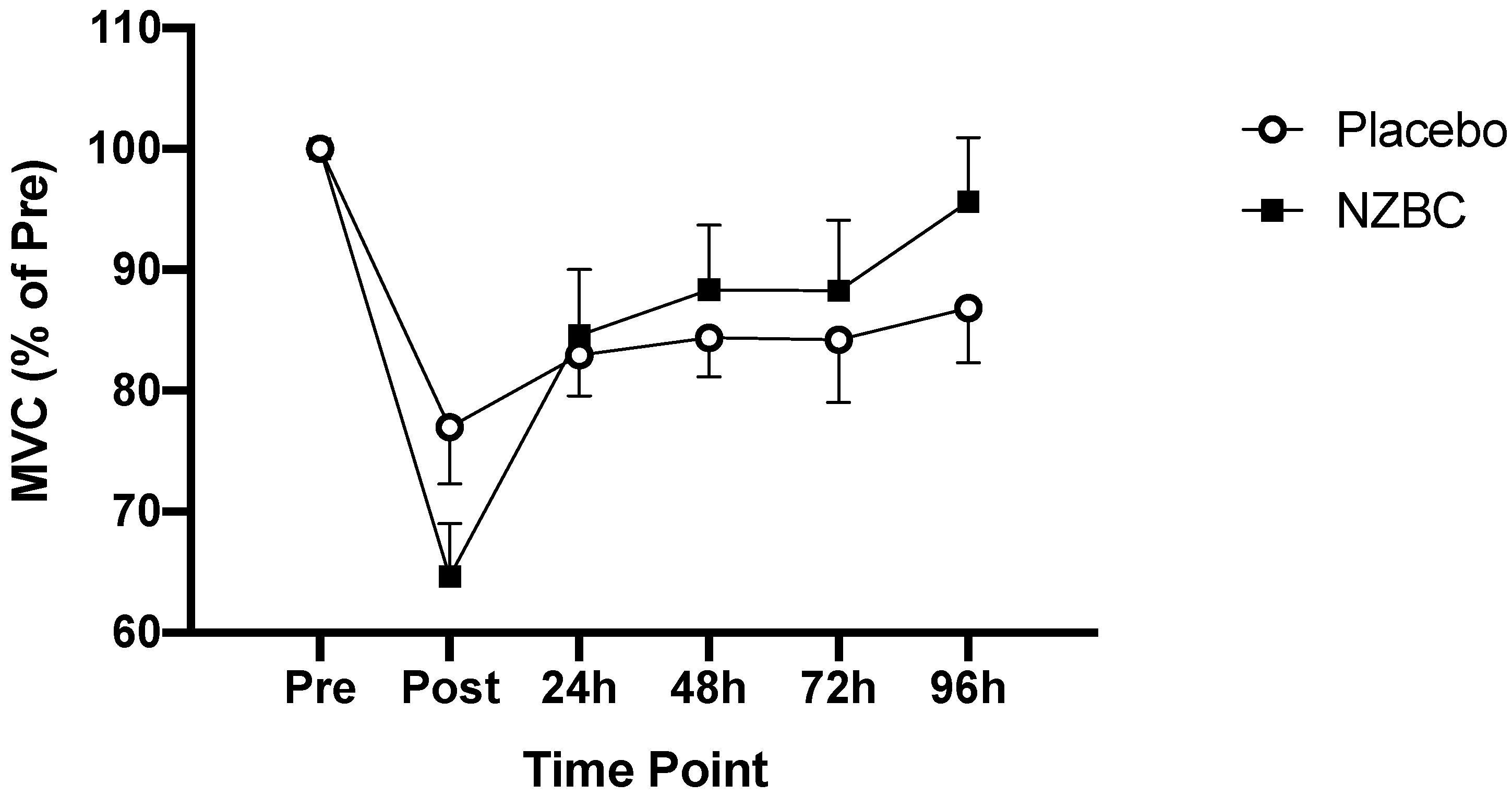
3.2. Muscle Function
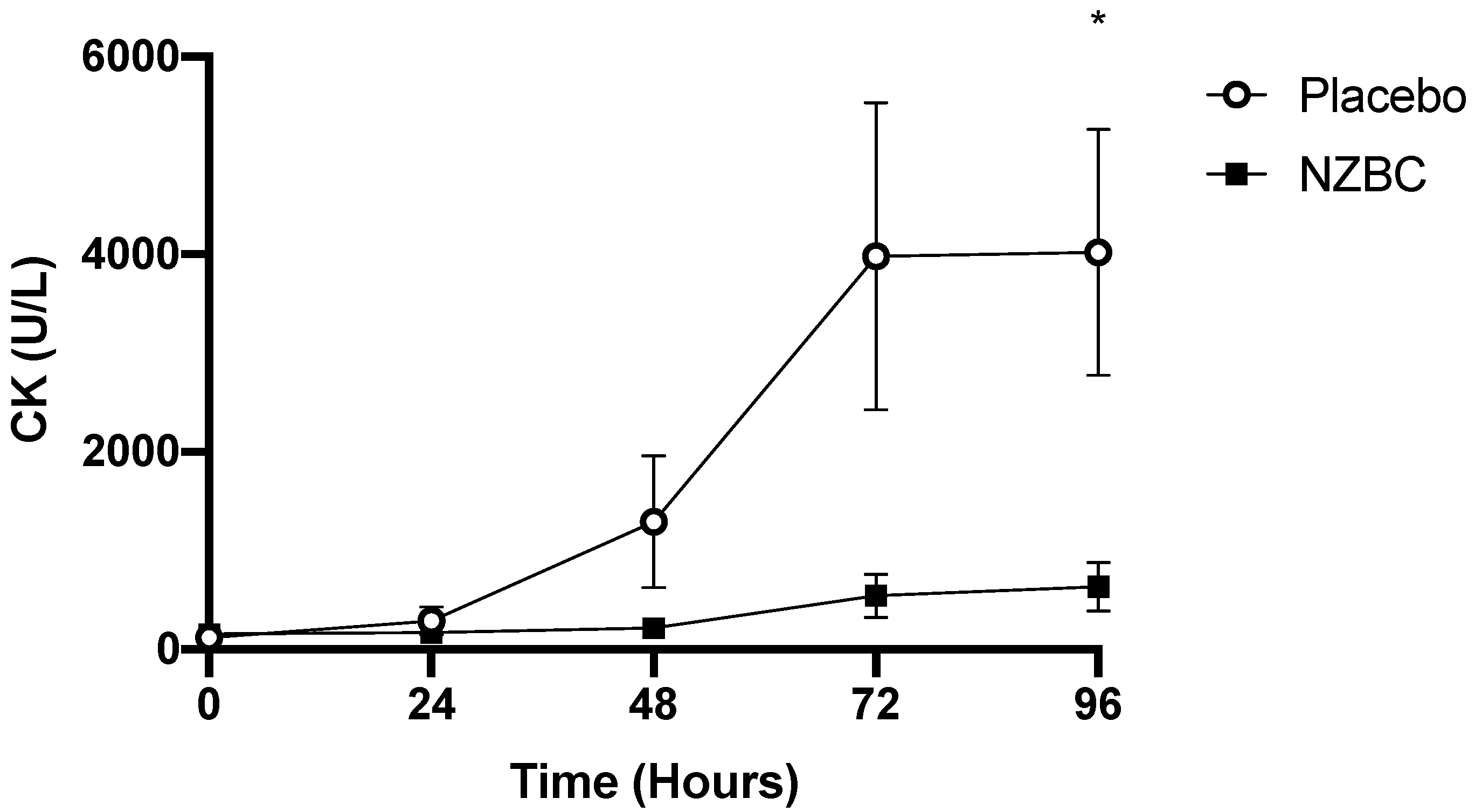
3.3. Creatine Kinase
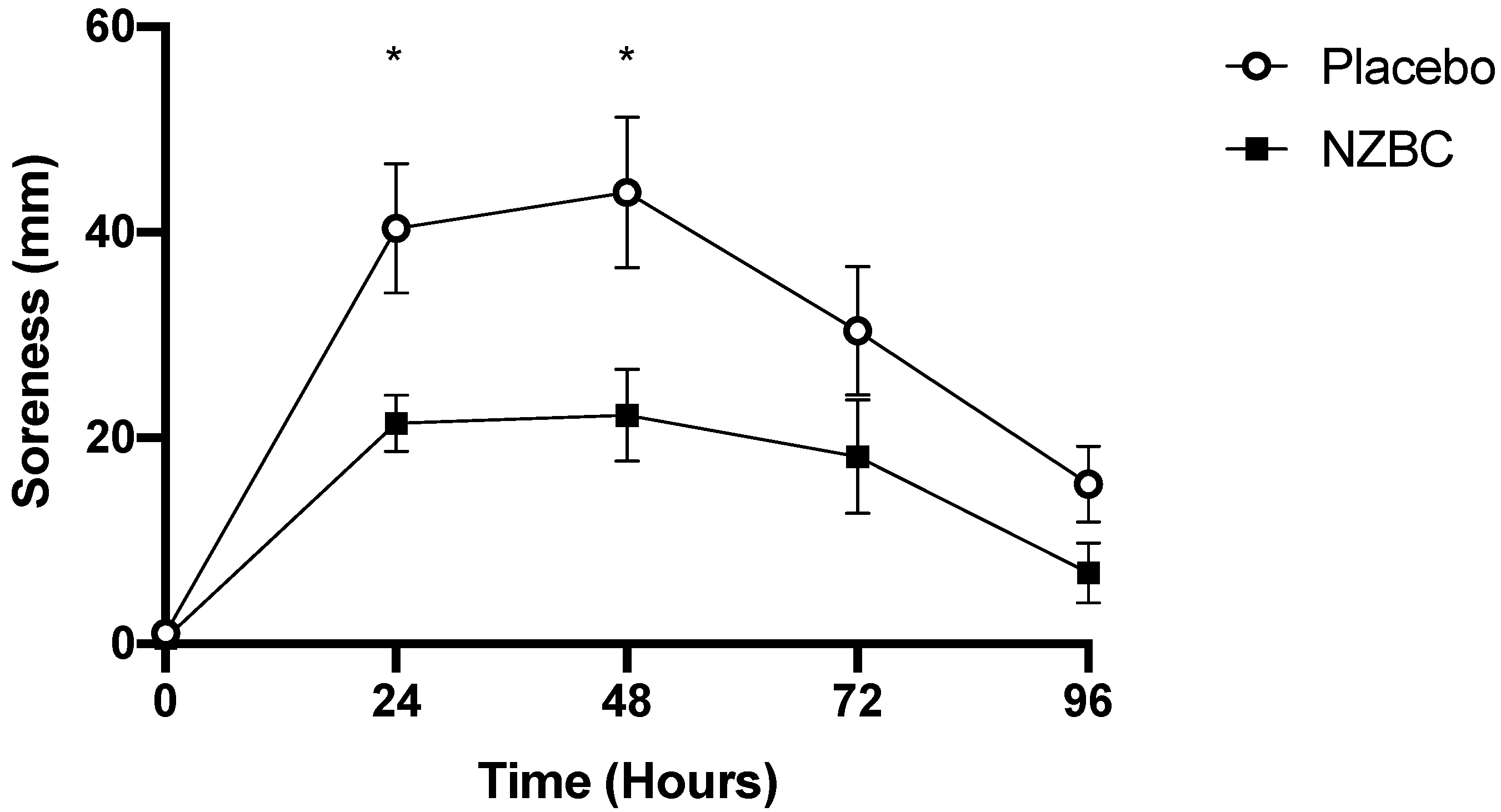
3.4. Muscle Soreness
3.5. Range of Motion
3.6. Mid-Arm Circumference
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Peake, J.M.; Neubauer, O.; Della Gatta, P.A.; Nosaka, K. Muscle damage and inflammation during recovery from exercise. J. Appl. Physiol. 2017, 122, 559–570. [Google Scholar] [CrossRef] [PubMed]
- Schoenfeld, B.J. Does Exercise-Induced Muscle Damage Play a Role in Skeletal Muscle Hypertrophy? J. Strength Cond. Res. 2012, 26, 1441–1453. [Google Scholar] [CrossRef] [Green Version]
- Toumi, H.; Best, T.M. The inflammatory response: Friend or enemy for muscle injury? Br. J. Sports Med. 2003, 37, 284–286. [Google Scholar] [CrossRef] [PubMed]
- Hody, S.; Croisier, J.L.; Bury, T.; Rogister, B.; Leprince, P. Eccentric muscle contractions: Risks and benefits. Front. Physiol. 2019, 10, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Csepregi, K.; Neugart, S.; Schreiner, M.; Hideg, É. Comparative evaluation of total antioxidant capacities of plant polyphenols. Molecules 2016, 21, 208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hussain, T.; Tan, B.; Yin, Y.; Blachier, F.; Tossou, M.C.B.; Rahu, N. Oxidative Stress and Inflammation: What Polyphenols Can Do for Us? Oxid. Med. Cell Longev. 2016, 2016, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yoon, J.H.; Baek, S.J. Molecular targets of dietary polyphenols with anti-inflammatory properties. Yonsei Med. J. 2005, 46, 585–596. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bazzucchi, I.; Patrizio, F.; Ceci, R.; Duranti, G.; Sgrò, P.; Sabatini, S.; Felici, F. The effects of quercetin supplementation on eccentric exercise-induced muscle damage. Nutrients 2019, 11, 205. [Google Scholar] [CrossRef] [Green Version]
- O’Fallon, K.S.; Kaushik, D.; Michniak-Kohn, B.; Dunne, C.P.; Zambraski, E.J.; Clarkson, P.M. Effects of Quercetin Supplementation on Markers of Muscle Damage and Inflammation after Eccentric Exercise. Int. J. Sport Nutr. Exerc. Metab. 2012, 22, 430–437. [Google Scholar] [CrossRef] [Green Version]
- Clifford, T.; Bell, O.; West, D.J.; Howatson, G.; Stevenson, E.J. The effects of beetroot juice supplementation on indices of muscle damage following eccentric exercise. Eur. J. Appl. Physiol. 2016, 116, 353–362. [Google Scholar] [CrossRef]
- McLeay, Y.; Barnes, M.J.; Mundel, T.; Hurst, S.M.; Hurst, R.D.; Stannard, S.R. Effect of New Zealand blueberry consumption on recovery from eccentric exercise-induced muscle damage. J. Int. Soc. Sports Nutr. 2012, 9, 19. [Google Scholar] [CrossRef] [Green Version]
- Bowtell, J.L.; Sumners, D.P.; Dyer, A.; Fox, P.; Mileva, K.N. Montmorency cherry juice reduces muscle damage caused by intensive strength exercise. Med. Sci. Sports Exerc. 2011, 43, 1544–1551. [Google Scholar] [CrossRef] [Green Version]
- Beals, K.; Allison, K.F.; Darnell, M.; Lovalekar, M.; Baker, R.; Nieman, D.C.; Vodovotz, Y.; Lephart, S.M. The effects of a tart cherry beverage on reducing exercise-induced muscle soreness. Isokinet. Exerc. Sci. 2017, 25, 53–63. [Google Scholar] [CrossRef]
- Connolly, D.A.J.; McHugh, M.P.; Padilla-Zakour, O.I. Efficacy of a tart cherry juice blend in preventing the symptoms of muscle damage. Br. J. Sports Med. 2006, 40, 679–683. [Google Scholar] [CrossRef] [PubMed]
- Trombold, J.R.; Reinfeld, A.S.; Casler, J.R.; Coyle, E.F. The Effect of Pomegranate Juice Supplementation on Strength and Soreness after Eccentric Exercise. J. Strength Cond. Res. 2011, 25, 782–788. [Google Scholar] [CrossRef] [PubMed]
- Lamb, K.L.; Ranchordas, M.K.; Johnson, E.; Denning, J.; Downing, F.; Lynn, A. No Effect of Tart Cherry Juice or Pomegranate Juice on Recovery from Exercise-Induced Muscle Damage in Non-Resistance Trained Men. Nutrients 2019, 11, 1593. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Howatson, G.; van Someren, K.A. Evidence of a contralateral repeated bout effect after maximal eccentric contractions. Eur. J. Appl. Physiol. 2007, 101, 7–14. [Google Scholar] [CrossRef]
- Tsuchiya, Y.; Nakazato, K.; Ochi, E. Contralateral repeated bout effect after eccentric exercise on muscular activation. Eur. J. Appl. Physiol. 2018, 118, 1997–2005. [Google Scholar] [CrossRef]
- Jamurtas, A.Z.; Theocharis, V.; Tofas, T.; Tsiokanos, A.; Yfanti, C.; Paschalis, V.; Koutedakis, Y.; Nosaka, K. Comparison between leg and arm eccentric exercises of the same relative intensity on indices of muscle damage. Eur. J. Appl. Physiol. 2005, 95, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.C.; Lin, K.Y.; Chen, H.L.; Lin, M.J.; Nosaka, K. Comparison in eccentric exercise-induced muscle damage among four limb muscles. Eur. J. Appl. Physiol. 2011, 111, 211–223. [Google Scholar] [CrossRef]
- Nosaka, K.; Newton, M.; Sacco, P. Muscle damage and soreness after endurance exercise of the elbow flexors. Med. Sci. Sport Exerc. 2002, 34, 920–927. [Google Scholar] [CrossRef] [PubMed]
- Nosaka, K.; Sakamoto, K. Effect of elbow joint angle on the magnitude of muscle damage to the elbow flexors. Med. Sci. Sports Exerc. 2001, 33, 22–29. [Google Scholar] [CrossRef]
- Hutchison, A.T.; Flieller, E.B.; Dillon, K.J.; Leverett, B.D. Black Currant Nectar Reduces Muscle Damage and Inflammation Following a Bout of High-Intensity Eccentric Contractions. J. Diet. Suppl. 2016, 13, 1–15. [Google Scholar] [CrossRef]
- Kevers, C.; Pincemail, J.; Defraigne, J.O.; Dommes, J. Antioxidant capacity of small dark fruits: Influence of cultivars and harvest time. J. Berry Res. 2014, 4, 97–105. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.G.; Vance, T.M.; Nam, T.G.; Kim, D.O.; Koo, S.I.; Chun, O.K. Contribution of Anthocyanin Composition to Total Antioxidant Capacity of Berries. Plant. Foods Hum. Nutr. 2015, 70, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Cook, M.D.; Myers, S.D.; Gault, M.L.; Willems, M.E.T. Blackcurrant Alters Physiological Responses and Femoral Artery Diameter during Sustained Isometric Contraction. Nutrients 2017, 9, 556. [Google Scholar] [CrossRef] [PubMed]
- Willems, M.E.T.; Myers, S.D.; Gault, M.L.; Cook, M.D. Beneficial physiological effects with blackcurrant intake in endurance athletes. Int. J. Sport Nutr. Exerc. Metab. 2015, 25, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Barnes, M.J.; Perry, B.G.; Hurst, R.D.; Lomiwes, D. Anthocyanin-Rich New Zealand Blackcurrant Extract Supports the Maintenance of Forearm Blood-Flow During Prolonged Sedentary Sitting. Front. Nutr. 2020, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Willems, M.E.T.; Silva, J.D.S.; Cook, M.D.; Blacker, S.D. Beneficial effects on fasting insulin and postprandial responses through 7-day intake of New Zealand blackcurrant powder. Funct. Foods Heath Dis. 2017, 7, 483. [Google Scholar] [CrossRef] [Green Version]
- Lyall, K.A.; Hurst, S.M.; Cooney, J.; Jensen, D.; Lo, K.; Hurst, R.D.; Stevenson, L.M. Short-term blackcurrant extract consumption modulates exercise-induced oxidative stress and lipopolysaccharide-stimulated inflammatory responses. Am. J. Physiol. Integr. Comp. Physiol. 2009, 297, 70–81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Møller, P.; Loft, S.; Alfthan, G.; Freese, R. Oxidative DNA damage in circulating mononuclear blood cells after ingestion of blackcurrant juice or anthocyanin-rich drink. Mutat. Res. Fundam Mol. Mech. Mutagen. 2004, 551, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, H.; Takenami, E.; Iwasaki-Kurashige, K.; Osada, T.; Katsumura, T.; Hamaoka, T. Effects of blackcurrant anthocyanin intake on peripheral muscle circulation during typing work in humans. Eur. J. Appl. Physiol. 2005, 94, 36–45. [Google Scholar] [CrossRef] [PubMed]
- De Ferrars, R.M.; Czank, C.; Zhang, Q.; Botting, N.P.; Kroon, P.A.; Cassidy, A.; Kay, C.D. The pharmacokinetics of anthocyanins and their metabolites in humans. Br. J. Pharmacol. 2014, 171, 3268–3282. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McGhie, T.K.; Walton, M.C. The bioavailability and absorption of anthocyanins: Towards a better understanding. Mol. Nutr. Food Res. 2007, 51, 702–713. [Google Scholar] [CrossRef] [PubMed]
- Hurst, R.D.; Lyall, K.A.; Roberts, J.M.; Perthaner, A.; Wells, R.W.; Cooney, J.M.; Jensen, D.J.; Burr, N.S.; Hurst, S.M. Consumption of an Anthocyanin-Rich Extract Made From New Zealand Blackcurrants Prior to Exercise May Assist Recovery From Oxidative Stress and Maintains Circulating Neutrophil Function: A Pilot Study. Front. Nutr. 2019, 6, 73. [Google Scholar] [CrossRef]
- Hurst, R.D.; Lyall, K.A.; Wells, R.W.; Sawyer, G.M.; Lomiwes, D.; Ngametua, N.; Hurst, S.M. Daily Consumption of an Anthocyanin-Rich Extract Made From New Zealand Blackcurrants for 5 Weeks Supports Exercise Recovery Through the Management of Oxidative Stress and Inflammation: A Randomized Placebo Controlled Pilot Study. Front. Nutr. 2020, 7, 16. [Google Scholar] [CrossRef] [Green Version]
- Neveu, V.; Perez-Jiménez, J.; Vos, F.; Crespy, V.; du Chaffaut, L.; Mennen, L. Phenol-Explorer: An online comprehensive database on polyphenol contents in foods. Database 2010, 2010, bap024. [Google Scholar] [CrossRef] [PubMed]
- Warren, G.L.; Lowe, D.A.; Armstrong, R.B. Measurement tools used in the study of eccentric contraction-induced injury. Sport Med. 1999, 7, 43–59. [Google Scholar] [CrossRef]
- Chen, T.C.; Huang, G.L.; Hsieh, C.C.; Tseng, K.W.; Tseng, W.C.; Chou, T.Y.; Nosaka, K. Comparison among three different intensities of eccentric contractions of the elbow flexors resulting in the same strength loss at one day post-exercise for changes in indirect muscle damage markers. Eur. J. Appl. Physiol. 2020, 120, 267–279. [Google Scholar] [CrossRef]
- Powers, S.K.; Jackson, M.J. Exercise-Induced Oxidative Stress: Cellular Mechanisms and Impact on Muscle Force Production. Physiol. Rev. 2008, 88, 1243–1276. [Google Scholar] [CrossRef] [Green Version]
- Crameri, R.M.; Aagaard, P.; Qvortrup, K.; Langberg, H.; Olesen, J.; Kjaer, M. Myofibre damage in human skeletal muscle: Effects of electrical stimulation versus voluntary contraction. J. Physiol. 2007, 583, 365–380. [Google Scholar] [CrossRef] [PubMed]
- Trombold, J.R.; Barnes, J.N.; Critchley, L.; Coyle, E.F. Ellagitannin consumption improves strength recovery 2–3 d after eccentric exercise. Med. Sci. Sports Exerc. 2010, 42, 493–498. [Google Scholar] [CrossRef] [Green Version]
- Koch, J.; Pereira, R.; Machado, M. The creatine kinase response to resistance exercise. J. Musculoskelet. Neuronal. Interact. 2014, 14, 68–77. [Google Scholar] [PubMed]
- Jia, N.; Xiong, Y.L.; Kong, B.; Liu, Q.; Xia, X. Radical scavenging activity of black currant (Ribes nigrum L.) extract and its inhibitory effect on gastric cancer cell proliferation via induction of apoptosis. J. Funct. Foods 2012, 4, 382–390. [Google Scholar] [CrossRef]
- Beaton, L.J.; Allan, D.A.; Tarnopolsky, M.A.; Tiidus, P.M.; Phillips, S.M. Contraction-induced muscle damage is unaffected by vitamin E supplementation. Med. Sci. Sports Exerc. 2002, 34, 798–805. [Google Scholar] [CrossRef]
- Tidball, J.G. Inflammatory processes in muscle injury and repair. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2005, 288, 345–353. [Google Scholar] [CrossRef] [Green Version]
- Close, G.L.; Ashton, T.; McArdle, A.; MacLaren, D.P.M. The emerging role of free radicals in delayed onset muscle soreness and contraction-induced muscle injury. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2005, 142, 257–266. [Google Scholar] [CrossRef]
- Paulsen, G.; Mikkelsen, U.R.; Raastad, T.; Peake, J.M. Leucocytes, cytokines and satellite cells: What role do they play in muscle damage and regeneration following eccentric exercise? Exerc. Immunol. Rev. 2012, 18, 42–97. [Google Scholar]
- Peake, J.M.; Della Gatta, P.; Suzuki, K.; Nieman, D.C. Cytokine expression and secretion by skeletal muscle cells: Regulatory mechanisms and exercise effects. Exerc. Immunol. Rev. 2015, 21, 8–25. [Google Scholar]
- Thoenes, M. Rhabdomyolysis: When Exercising Becomes a Risk. J. Pediatr. Health Care 2010, 24, 189–193. [Google Scholar] [CrossRef]
- Gibala, M.J.; MacDougall, J.D.; Tarnopolsky, M.A.; Stauber, W.T.; Elorriaga, A. Changes in human skeletal muscle ultrastructure and force production after acute resistance exercise. J. Appl. Physiol. 1995, 78, 702–708. [Google Scholar] [CrossRef]
- Hortobágyi, T.; Houmard, J.; Fraser, D.; Dudek, R.; Lambert, J.; Tracy, J. Normal forces and myofibrillar disruption after repeated eccentric exercise. J. Appl. Physiol. 1998, 84, 492–498. [Google Scholar] [CrossRef]
- Romero-Parra, N.; Cupeiro, R.; Alfaro-Magallanes, V.M.; Rael, B.; Rubio-Arias, J.Á.; Peinado, A.B.; Benito, P.J. Exercise-Induced Muscle Damage During the Menstrual Cycle: A Systematic Review and Meta-Analysis. J. Strength Cond. Res. 2021, 35, 549–561. [Google Scholar] [CrossRef]
| NZBC | Placebo | p Value | |
|---|---|---|---|
| Gender | 10 women, 4 men | 9 women, 4 men | |
| Age (yrs) | 24 ± 2 | 23 ± 2 | 0.272 |
| Stature (cm) | 170 ± 9 | 164 ± 11 | 0.175 |
| Body Mass (kg) | 67.9 ± 15.7 | 59.0 ± 7.9 | 0.120 * |
| BMI (kg/m2) | 23.4 ± 3.5 | 21.8 ± 2.3 | 0.204 |
| Baseline MVC (Nm) | 42.2 ± 18.3 | 35.8 ± 13.9 | 0.299 |
| Baseline CK (UL) | 118 ± 80 | 155 ± 99 | 0.487 |
| NZBC | Placebo | p Value | |
|---|---|---|---|
| Energy (kcal day−1) | 1880 ± 905 | 1617 ± 408 | 1.000 * |
| Carbohydrate (g day−1) | 221 ± 110 | 182 ± 53 | 0.305 |
| Protein (g day−1) | 78 ± 22 | 69 ± 22 | 0.484 |
| Fat (g day−1) | 68 ± 25 | 74 ± 46 | 0.784 |
| Anthocyanin (mg day−1) | 24 ± 28 | 38 ± 33 | 0.279 * |
| Variable | Group | 0 h | 24 h | 48 h | 72 h | 96 h |
|---|---|---|---|---|---|---|
| ROM (°) | NZBC | 150 ± 21 | 139 ± 34 * | 139 ± 31 * | 140 ± 25 * | 142 ± 26 |
| PLA | 147 ± 20 | 134 ± 26 * | 134 ± 26 * | 136 ± 22 * | 136 ± 20 * | |
| MAC (cm) | NZBC | 28.6 ± 4.2 | 29.0 ± 4.2 | 29.1 ± 4.2 | 28.6 ± 4.3 | 28.6 ± 4.1 |
| PLA | 27.9 ± 2.9 | 28.0 ± 2.9 | 28.3 ± 2.7 | 28.6 ± 2.6 | 28.6 ± 2.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hunt, J.E.A.; Coelho, M.O.C.; Buxton, S.; Butcher, R.; Foran, D.; Rowland, D.; Gurton, W.; Macrae, H.; Jones, L.; Gapper, K.S.; et al. Consumption of New Zealand Blackcurrant Extract Improves Recovery from Exercise-Induced Muscle Damage in Non-Resistance Trained Men and Women: A Double-Blind Randomised Trial. Nutrients 2021, 13, 2875. https://doi.org/10.3390/nu13082875
Hunt JEA, Coelho MOC, Buxton S, Butcher R, Foran D, Rowland D, Gurton W, Macrae H, Jones L, Gapper KS, et al. Consumption of New Zealand Blackcurrant Extract Improves Recovery from Exercise-Induced Muscle Damage in Non-Resistance Trained Men and Women: A Double-Blind Randomised Trial. Nutrients. 2021; 13(8):2875. https://doi.org/10.3390/nu13082875
Chicago/Turabian StyleHunt, Julie E. A., Mariana O. C. Coelho, Sean Buxton, Rachel Butcher, Daniel Foran, Daniel Rowland, William Gurton, Heather Macrae, Louise Jones, Kyle S. Gapper, and et al. 2021. "Consumption of New Zealand Blackcurrant Extract Improves Recovery from Exercise-Induced Muscle Damage in Non-Resistance Trained Men and Women: A Double-Blind Randomised Trial" Nutrients 13, no. 8: 2875. https://doi.org/10.3390/nu13082875
APA StyleHunt, J. E. A., Coelho, M. O. C., Buxton, S., Butcher, R., Foran, D., Rowland, D., Gurton, W., Macrae, H., Jones, L., Gapper, K. S., Manders, R. J. F., & King, D. G. (2021). Consumption of New Zealand Blackcurrant Extract Improves Recovery from Exercise-Induced Muscle Damage in Non-Resistance Trained Men and Women: A Double-Blind Randomised Trial. Nutrients, 13(8), 2875. https://doi.org/10.3390/nu13082875





