Gut Microbiota, Probiotic Interventions, and Cognitive Function in the Elderly: A Review of Current Knowledge
Abstract
:1. Introduction
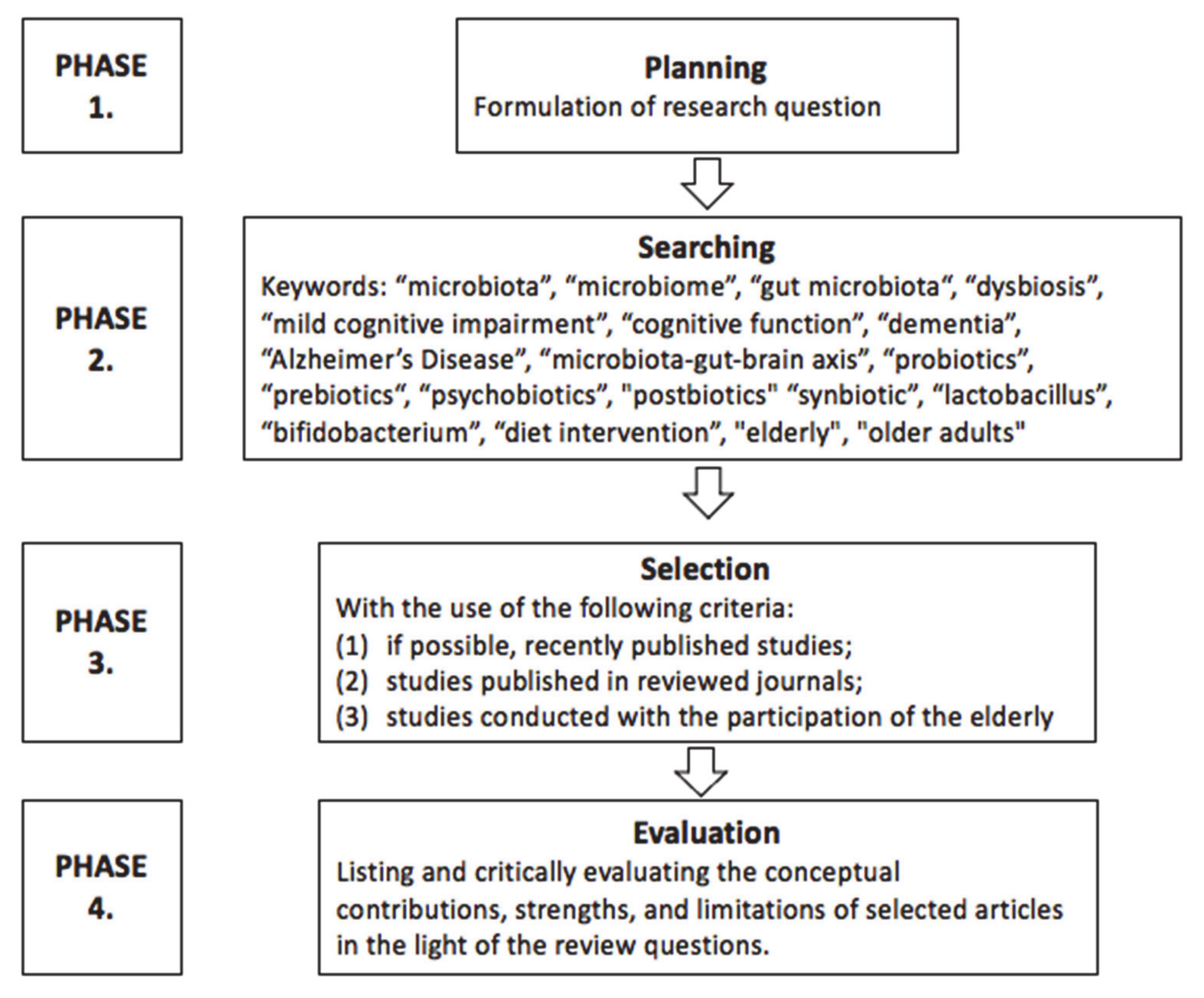
2. Review Methodology
3. How Does the Gut Microbiota Change with Aging?
4. Gut Microbiota in Cognitive Disorders—Is There a Difference?
5. Is There a Link Between Diet, Cognitive Function, and Gut Microbiota in the Elderly?
6. Can Administering Probiotic or Prebiotics Supplementation Improve Cognitive Functions in the Elderly?
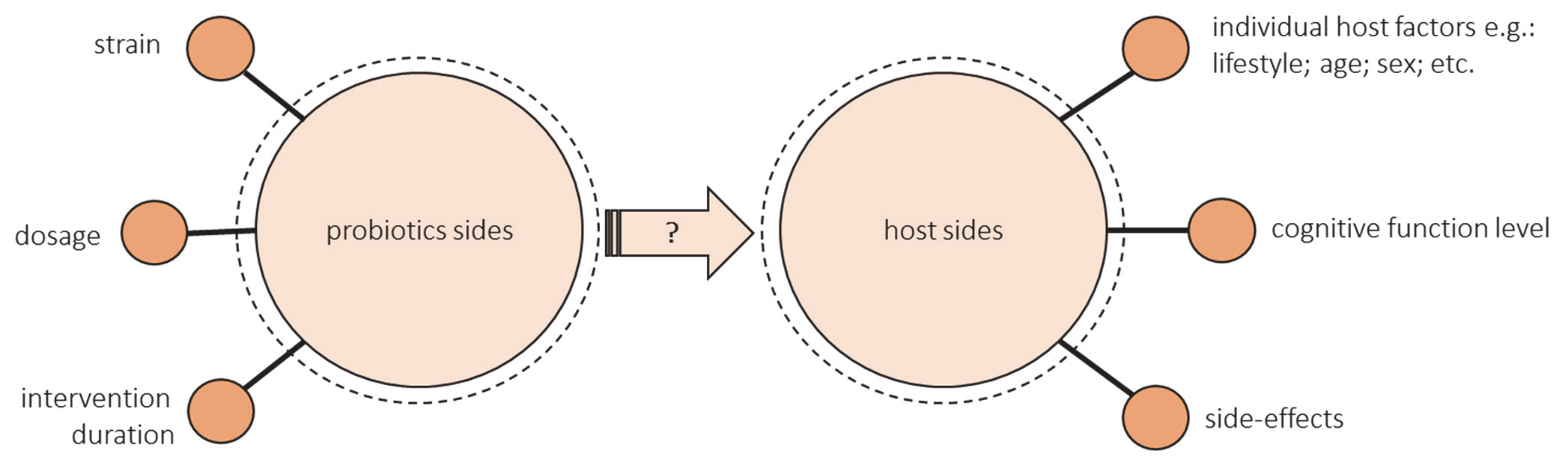
- The health-promoting effect of a probiotic depends on the strain. Some strains show a positive effect on cognitive function, and some do not. The research uses either single strain or multiple strain probiotics. Lv et al. observed that a single strain of probiotics was more effective in human studies [90]. However, as previously mentioned, diversity may be crucial for the cognitive health of the elderly, but most studies have not assessed changes in the microbiota after administering probiotic supplementation.
- There is not enough evidence to provide information on dose–response functions associated with probiotics. Most studies have not compared the different doses. Doses that ranged from 108 to 1011 were used most often, but more reliable evidence from various dosages is needed, especially outside these common doses [96].
- There are also many unknowns about the duration of probiotics ingestion. The most common intervention duration of the studies was 12 weeks. Some authors emphasize that the intervention could have been too short [94,99]. Lv et al. suggest that a duration of less than 12 weeks was more effective, but these studies included data from middle-aged adults [90]. There is a lack of long-term studies especially in the group of elderly.
- The success of the intervention could be modified by the hosts’ diet and lifestyle, age, sex, geographic region, concomitant disease, antibiotic exposure, and baseline microbiota composition [105,106,107]. All these factors should be controlled. Still, the studies did not assess the effectiveness of probiotic intake through gut microbiota composition. Most studies did not consider the baseline gut microbiota characteristics of included individuals. Despite the influence of diet on the gut microbiome, the majority of studies contain little or no analysis of dietary intake. There is a need of conducting more well-controlled longitudinal studies and randomized controlled trials that may isolate the impact of specific changes in dietary intake on gut microbiota [108].
- The level of cognitive functioning can make a difference. There are no studies that would compare people with different levels of cognitive functioning. Kobayashi found a beneficial effect of probiotics in the low-score subgroup but not in the high-score subgroup (indicating favorable cognitive performance), so they suggest comparing the results between MCI or early dementia and cognitively normal individuals [99]. Lv et al. showed that the effects of probiotics supplementation were greater in the case of cognitively impaired individuals than those on healthy ones, but they only included one study with cognitively healthy people [90]. What is more, the current RCT, including patients with cognitive impairment, are mainly focused on inflammatory and oxidative biomarkers rather than cognitive function, so they used MMSE or TYM, which are used for screening rather than cognitive assessment [100,101,102].
- The side-effects of probiotics intervention should be considered alongside the observed benefits [109]. Hibberd et al. reported that Lactobacillus rhamnosus GG ATCC 53,103 (LGG) (1 × 1010 CFU) is safe and well-tolerated in healthy adults aged 65 years and older [110]. Similarly, the results of Hwang et al. suggest that Lactobacillus plantarum C29-fermented soybean can be safely administered to enhance cognitive function in individuals with MCI. Adverse events observed were stomach aches, headaches, gastritis, erectile dysfunction, and seborrheic dermatitis, all of which were classified as a mild adverse event [98].
7. What Is the Link Between the Microbiota–Gut–Brain Axis and Cognitive Function?
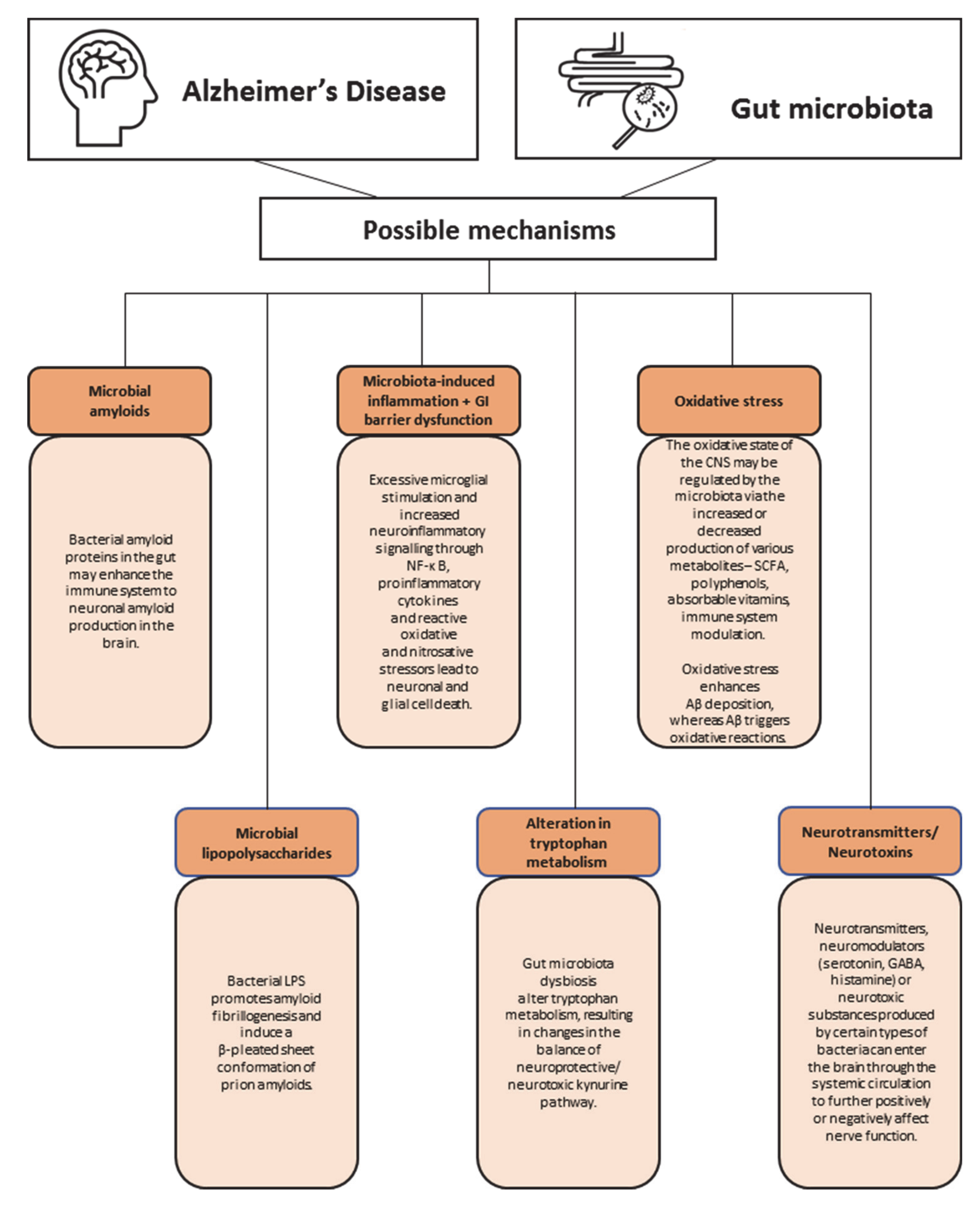
8. What Are the Possible Mechanisms behind Gut Microbiota and Dementia?
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jafari Nasabian, P.; Inglis, J.E.; Reilly, W.; Kelly, O.J.; Ilich, J.Z. Aging Human Body: Changes in Bone, Muscle and Body Fat with Consequent Changes in Nutrient Intake. J. Endocrinol. 2017, 234, R37–R51. [Google Scholar] [CrossRef] [Green Version]
- Müller, L.; Di Benedetto, S.; Pawelec, G. The Immune System and Its Dysregulation with Aging. In Biochemistry and Cell Biology of Ageing: Part II Clinical Science; Subcellular Biochemistry; Harris, J.R., Korolchuk, V.I., Eds.; Springer: Singapore, 2019; Volume 91, pp. 21–43. ISBN 9789811336805. [Google Scholar]
- Cai, L.; Chan, J.S.Y.; Yan, J.H.; Peng, K. Brain Plasticity and Motor Practice in Cognitive Aging. Front. Aging Neurosci. 2014, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crous-Bou, M.; Minguillón, C.; Gramunt, N.; Molinuevo, J.L. Alzheimer’s Disease Prevention: From Risk Factors to Early Intervention. Alzheimer’s Res. Ther. 2017, 9, 71. [Google Scholar] [CrossRef]
- Petersen, R.C. Mild Cognitive Impairment. Contin. Lifelong Learn. Neurol. 2016, 22, 404–418. [Google Scholar] [CrossRef] [PubMed]
- Alzheimer’s Disease International. Alzheimer’s Disease International World Alzheimer Report 2019: Attitudes to Dementia; Alzheimer’s Disease International: London, UK, 2019. [Google Scholar]
- Marchesi, J.R.; Ravel, J. The Vocabulary of Microbiome Research: A Proposal. Microbiome 2015, 3, 31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruan, W.; Engevik, M.A.; Spinler, J.K.; Versalovic, J. Healthy Human Gastrointestinal Microbiome: Composition and Function After a Decade of Exploration. Dig. Dis. Sci. 2020, 65, 695–705. [Google Scholar] [CrossRef] [Green Version]
- Mohajeri, M.H.; Brummer, R.J.M.; Rastall, R.A.; Weersma, R.K.; Harmsen, H.J.M.; Faas, M.; Eggersdorfer, M. The Role of the Microbiome for Human Health: From Basic Science to Clinical Applications. Eur. J. Nutr. 2018, 57, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Gaulke, C.A.; Sharpton, T.J. The Influence of Ethnicity and Geography on Human Gut Microbiome Composition. Nat. Med. 2018, 24, 1495–1496. [Google Scholar] [CrossRef]
- Thursby, E.; Juge, N. Introduction to the Human Gut Microbiota. Biochem. J. 2017, 474, 1823–1836. [Google Scholar] [CrossRef]
- Hillman, E.T.; Lu, H.; Yao, T.; Nakatsu, C.H. Microbial Ecology along the Gastrointestinal Tract. Microbes. Environ. 2017, 32, 300–313. [Google Scholar] [CrossRef] [Green Version]
- Arumugam, M.; Raes, J.; Pelletier, E.; Le Paslier, D.; Yamada, T.; Mende, D.R.; Fernandes, G.R.; Tap, J.; Bruls, T.; Batto, J.-M.; et al. Enterotypes of the Human Gut Microbiome. Nature 2011, 473, 174–180. [Google Scholar] [CrossRef]
- Ghaisas, S.; Maher, J.; Kanthasamy, A. Gut Microbiome in Health and Disease: Linking the Microbiome-Gut-Brain Axis and Environmental Factors in the Pathogenesis of Systemic and Neurodegenerative Diseases. Pharm. Ther. 2016, 158, 52–62. [Google Scholar] [CrossRef] [Green Version]
- An, R.; Wilms, E.; Masclee, A.A.M.; Smidt, H.; Zoetendal, E.G.; Jonkers, D. Age-Dependent Changes in GI Physiology and Microbiota: Time to Reconsider? Gut 2018, 67, 2213–2222. [Google Scholar] [CrossRef] [PubMed]
- Baumgart, M.; Snyder, H.M.; Carrillo, M.C.; Fazio, S.; Kim, H.; Johns, H. Summary of the Evidence on Modifiable Risk Factors for Cognitive Decline and Dementia: A Population-Based Perspective. Alzheimer’s Dement. 2015, 11, 718–726. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Legdeur, N.; Heymans, M.W.; Comijs, H.C.; Huisman, M.; Maier, A.B.; Visser, P.J. Age Dependency of Risk Factors for Cognitive Decline. BMC Geriatr 2018, 18, 187. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Tan, L.; Wang, H.-F.; Jiang, T.; Tan, M.-S.; Tan, L.; Zhao, Q.-F.; Li, J.-Q.; Wang, J.; Yu, J.-T. Meta-Analysis of Modifiable Risk Factors for Alzheimer’s Disease. J. Neurol. Neurosurg. Psychiatry 2015, 86, 1299–1306. [Google Scholar] [CrossRef] [PubMed]
- Lynch, S.V.; Pedersen, O. The Human Intestinal Microbiome in Health and Disease. N. Engl. J. Med. 2016, 375, 2369–2379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petersen, C.; Round, J.L. Defining Dysbiosis and Its Influence on Host Immunity and Disease. Cell Microbiol. 2014, 16, 1024–1033. [Google Scholar] [CrossRef] [PubMed]
- Berg, G.; Rybakova, D.; Fischer, D.; Cernava, T.; Vergès, M.-C.C.; Charles, T.; Chen, X.; Cocolin, L.; Eversole, K.; Corral, G.H.; et al. Microbiome Definition Re-Visited: Old Concepts and New Challenges. Microbiome 2020, 8, 103. [Google Scholar] [CrossRef]
- Salminen, S.; Collado, M.C.; Endo, A.; Hill, C.; Lebeer, S.; Quigley, E.M.M.; Sanders, M.E.; Shamir, R.; Swann, J.R.; Szajewska, H.; et al. The International Scientific Association of Probiotics and Prebiotics (ISAPP) Consensus Statement on the Definition and Scope of Postbiotics. Nat. Rev. Gastroenterol. Hepatol. 2021, 1–19. [Google Scholar] [CrossRef]
- de Almada, C.N.; Almada, C.N.; Martinez, R.C.R.; Sant’Ana, A.S. Paraprobiotics: Evidences on Their Ability to Modify Biological Responses, Inactivation Methods and Perspectives on Their Application in Foods. Trends Food Sci. Technol. 2016, 58, 96–114. [Google Scholar] [CrossRef]
- Gibson, G.R.; Probert, H.M.; Loo, J.V.; Rastall, R.A.; Roberfroid, M.B. Dietary Modulation of the Human Colonic Microbiota: Updating the Concept of Prebiotics. Nutr. Res. Rev. 2004, 17, 259–275. [Google Scholar] [CrossRef] [Green Version]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. The International Scientific Association for Probiotics and Prebiotics Consensus Statement on the Scope and Appropriate Use of the Term Probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dinan, T.G.; Stanton, C.; Cryan, J.F. Psychobiotics: A Novel Class of Psychotropic. Biol. Psychiatry 2013, 74, 720–726. [Google Scholar] [CrossRef] [PubMed]
- Pandey, K.R.; Naik, S.R.; Vakil, B.V. Probiotics, Prebiotics and Synbiotics—A Review. J. Food Sci. Technol. 2015, 52, 7577–7587. [Google Scholar] [CrossRef]
- Bratburd, J.R.; Keller, C.; Vivas, E.; Gemperline, E.; Li, L.; Rey, F.E.; Currie, C.R. Gut Microbial and Metabolic Responses to Salmonella Enterica Serovar Typhimurium and Candida albicans. mBio 2018, 9, e02032-18. [Google Scholar] [CrossRef] [Green Version]
- Vaiserman, A.M.; Koliada, A.K.; Marotta, F. Gut Microbiota: A Player in Aging and a Target for Anti-Aging Intervention. Ageing Res. Rev. 2017, 35, 36–45. [Google Scholar] [CrossRef]
- Claesson, M.J.; Jeffery, I.B.; Conde, S.; Power, S.E.; O’Connor, E.M.; Cusack, S.; Harris, H.M.B.; Coakley, M.; Lakshminarayanan, B.; O’Sullivan, O.; et al. Gut Microbiota Composition Correlates with Diet and Health in the Elderly. Nature 2012, 488, 178–184. [Google Scholar] [CrossRef]
- Alkasir, R.; Li, J.; Li, X.; Jin, M.; Zhu, B. Human Gut Microbiota: The Links with Dementia Development. Protein Cell 2017, 8, 90–102. [Google Scholar] [CrossRef] [Green Version]
- Hopkins, M.J.; Macfarlane, G.T. Changes in Predominant Bacterial Populations in Human Faeces with Age and with Clostridium Difficile Infection. J. Med. Microbiol. 2002, 51, 448–454. [Google Scholar] [CrossRef]
- Biagi, E.; Nylund, L.; Candela, M.; Ostan, R.; Bucci, L.; Pini, E.; Nikkïla, J.; Monti, D.; Satokari, R.; Franceschi, C.; et al. Through Ageing, and beyond: Gut Microbiota and Inflammatory Status in Seniors and Centenarians. PLoS ONE 2010, 5, e10667. [Google Scholar] [CrossRef]
- Drago, L.; Toscano, M.; Rodighiero, V.; De Vecchi, E.; Mogna, G. Cultivable and Pyrosequenced Fecal Microflora in Centenarians and Young Subjects. J. Clin. Gastroenterol. 2012, 46, S81–S84. [Google Scholar] [CrossRef]
- Askarova, S.; Umbayev, B.; Masoud, A.-R.; Kaiyrlykyzy, A.; Safarova, Y.; Tsoy, A.; Olzhayev, F.; Kushugulova, A. The Links Between the Gut Microbiome, Aging, Modern Lifestyle and Alzheimer’s Disease. Front. Cell. Infect. Microbiol. 2020, 10. [Google Scholar] [CrossRef] [Green Version]
- Senghor, B.; Sokhna, C.; Ruimy, R.; Lagier, J.-C. Gut Microbiota Diversity According to Dietary Habits and Geographical Provenance. Hum. Microbiome J. 2018, 7–8, 1–9. [Google Scholar] [CrossRef]
- Angelucci, F.; Cechova, K.; Amlerova, J.; Hort, J. Antibiotics, Gut Microbiota, and Alzheimer’s Disease. J. Neuroinflamm. 2019, 16. [Google Scholar] [CrossRef]
- Whitelock, E.; Ensaff, H. On Your Own: Older Adults’ Food Choice and Dietary Habits. Nutrients 2018, 10. [Google Scholar] [CrossRef] [Green Version]
- Lindroos, E.K.; Saarela, R.K.T.; Suominen, M.H.; Muurinen, S.; Soini, H.; Kautiainen, H.; Pitkälä, K.H. Burden of Oral Symptoms and Its Associations with Nutrition, Well-Being, and Survival Among Nursing Home Residents. J. Am. Med. Dir. Assoc. 2019, 20, 537–543. [Google Scholar] [CrossRef] [Green Version]
- Agarwal, E.; Miller, M.; Yaxley, A.; Isenring, E. Malnutrition in the Elderly: A Narrative Review. Maturitas 2013, 76, 296–302. [Google Scholar] [CrossRef] [Green Version]
- Corish, C.A.; Bardon, L.A. Malnutrition in Older Adults: Screening and Determinants. Proc. Nutr. Soc. 2019, 78, 372–379. [Google Scholar] [CrossRef]
- M Silva, F.; Petrica, J.; Serrano, J.; Paulo, R.; Ramalho, A.; Lucas, D.; Ferreira, J.P.; Duarte-Mendes, P. The Sedentary Time and Physical Activity Levels on Physical Fitness in the Elderly: A Comparative Cross Sectional Study. Int. J. Environ. Res. Public Health 2019, 16. [Google Scholar] [CrossRef] [Green Version]
- Park, S.; Castaneda-Gameros, D.; Park, S.-Y.; Oh, I.-H. Factors of Physical Activity and Sedentary Behavior in Elderly Koreans. Am. J. Health Behav. 2019, 43, 1040–1049. [Google Scholar] [CrossRef]
- Codella, R.; Luzi, L.; Terruzzi, I. Exercise Has the Guts: How Physical Activity May Positively Modulate Gut Microbiota in Chronic and Immune-Based Diseases. Dig. Liver Dis. 2018, 50, 331–341. [Google Scholar] [CrossRef] [Green Version]
- Rinninella, E.; Cintoni, M.; Raoul, P.; Lopetuso, L.R.; Scaldaferri, F.; Pulcini, G.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. Food Components and Dietary Habits: Keys for a Healthy Gut Microbiota Composition. Nutrients 2019, 11, 2393. [Google Scholar] [CrossRef] [Green Version]
- Schlegel, P.; Novotny, M.; Klimova, B.; Valis, M. “Muscle-Gut-Brain Axis”: Can Physical Activity Help Patients with Alzheimer’s Disease Due to Microbiome Modulation? J. Alzheimer’s Dis. 2019, 71, 861–878. [Google Scholar] [CrossRef]
- Heeney, D.D.; Gareau, M.G.; Marco, M.L. Intestinal Lactobacillus in Health and Disease, a Driver or Just along for the Ride? Curr. Opin. Biotechnol. 2018, 49, 140–147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hidalgo-Cantabrana, C.; Delgado, S.; Ruiz, L.; Ruas-Madiedo, P.; Sánchez, B.; Margolles, A. Bifidobacteria and Their Health-Promoting Effects. Microbiol. Spectr. 2017, 5. [Google Scholar] [CrossRef]
- Saji, N.; Niida, S.; Murotani, K.; Hisada, T.; Tsuduki, T.; Sugimoto, T.; Kimura, A.; Toba, K.; Sakurai, T. Analysis of the Relationship between the Gut Microbiome and Dementia: A Cross-Sectional Study Conducted in Japan. Sci. Rep. 2019, 9, 1008. [Google Scholar] [CrossRef]
- Saji, N.; Murotani, K.; Hisada, T.; Tsuduki, T.; Sugimoto, T.; Kimura, A.; Niida, S.; Toba, K.; Sakurai, T. The Relationship between the Gut Microbiome and Mild Cognitive Impairment in Patients without Dementia: A Cross-Sectional Study Conducted in Japan. Sci. Rep. 2019, 9, 19227. [Google Scholar] [CrossRef] [Green Version]
- Vogt, N.M.; Kerby, R.L.; Dill-McFarland, K.A.; Harding, S.J.; Merluzzi, A.P.; Johnson, S.C.; Carlsson, C.M.; Asthana, S.; Zetterberg, H.; Blennow, K.; et al. Gut Microbiome Alterations in Alzheimer’s Disease. Sci. Rep. 2017, 7, 13537. [Google Scholar] [CrossRef]
- Cattaneo, A.; Cattane, N.; Galluzzi, S.; Provasi, S.; Lopizzo, N.; Festari, C.; Ferrari, C.; Guerra, U.P.; Paghera, B.; Muscio, C.; et al. Association of Brain Amyloidosis with Pro-Inflammatory Gut Bacterial Taxa and Peripheral Inflammation Markers in Cognitively Impaired Elderly. Neurobiol. Aging 2017, 49, 60–68. [Google Scholar] [CrossRef] [Green Version]
- Stadlbauer, V.; Engertsberger, L.; Komarova, I.; Feldbacher, N.; Leber, B.; Pichler, G.; Fink, N.; Scarpatetti, M.; Schippinger, W.; Schmidt, R.; et al. Dysbiosis, Gut Barrier Dysfunction and Inflammation in Dementia: A Pilot Study. BMC Geriatr. 2020, 20, 248. [Google Scholar] [CrossRef]
- Liu, P.; Wu, L.; Peng, G.; Han, Y.; Tang, R.; Ge, J.; Zhang, L.; Jia, L.; Yue, S.; Zhou, K.; et al. Altered Microbiomes Distinguish Alzheimer’s Disease from Amnestic Mild Cognitive Impairment and Health in a Chinese Cohort. Brain Behav. Immun. 2019, 80, 633–643. [Google Scholar] [CrossRef]
- Nagpal, R.; Neth, B.J.; Wang, S.; Craft, S.; Yadav, H. Modified Mediterranean-Ketogenic Diet Modulates Gut Microbiome and Short-Chain Fatty Acids in Association with Alzheimer’s Disease Markers in Subjects with Mild Cognitive Impairment. EBioMedicine 2019, 47, 529–542. [Google Scholar] [CrossRef] [Green Version]
- Cerajewska, T.L.; Davies, M.; West, N.X. Periodontitis: A Potential Risk Factor for Alzheimer’s Disease. Br. Dent. J. 2015, 218, 29–34. [Google Scholar] [CrossRef]
- Holmer, J.; Aho, V.; Eriksdotter, M.; Paulin, L.; Pietiäinen, M.; Auvinen, P.; Schultzberg, M.; Pussinen, P.J.; Buhlin, K. Subgingival Microbiota in a Population with and without Cognitive Dysfunction. J. Oral Microbiol. 2021, 13. [Google Scholar] [CrossRef]
- Wang, R.P.-H.; Ho, Y.-S.; Leung, W.K.; Goto, T.; Chang, R.C.-C. Systemic Inflammation Linking Chronic Periodontitis to Cognitive Decline. Brain Behav. Immun. 2019, 81, 63–73. [Google Scholar] [CrossRef]
- Olsen, I.; Singhrao, S.K. Is There a Link between Genetic Defects in the Complement Cascade and Porphyromonas Gingivalis in Alzheimer’s Disease? J. Oral Microbiol. 2020, 12, 1676486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zmora, N.; Suez, J.; Elinav, E. You Are What You Eat: Diet, Health and the Gut Microbiota. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 35–56. [Google Scholar] [CrossRef] [PubMed]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet Rapidly and Reproducibly Alters the Human Gut Microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef] [Green Version]
- Wu, G.D.; Chen, J.; Hoffmann, C.; Bittinger, K.; Chen, Y.-Y.; Keilbaugh, S.A.; Bewtra, M.; Knights, D.; Walters, W.A.; Knight, R.; et al. Linking Long-Term Dietary Patterns with Gut Microbial Enterotypes. Science 2011, 334, 105–108. [Google Scholar] [CrossRef] [Green Version]
- Leeming, E.R.; Johnson, A.J.; Spector, T.D.; Le Roy, C.I. Effect of Diet on the Gut Microbiota: Rethinking Intervention Duration. Nutrients 2019, 11, 2862. [Google Scholar] [CrossRef] [Green Version]
- Singh, R.K.; Chang, H.-W.; Yan, D.; Lee, K.M.; Ucmak, D.; Wong, K.; Abrouk, M.; Farahnik, B.; Nakamura, M.; Zhu, T.H.; et al. Influence of Diet on the Gut Microbiome and Implications for Human Health. J. Transl. Med. 2017, 15, 73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soltani, S.; Jayedi, A.; Shab-Bidar, S.; Becerra-Tomás, N.; Salas-Salvadó, J. Adherence to the Mediterranean Diet in Relation to All-Cause Mortality: A Systematic Review and Dose-Response Meta-Analysis of Prospective Cohort Studies. Adv. Nutr. 2019, 10, 1029–1039. [Google Scholar] [CrossRef] [PubMed]
- Psaltopoulou, T.; Sergentanis, T.N.; Panagiotakos, D.B.; Sergentanis, I.N.; Kosti, R.; Scarmeas, N. Mediterranean Diet, Stroke, Cognitive Impairment, and Depression: A Meta-Analysis: Annals of Neurology. Ann. Neurol. 2013, 74, 580–591. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Parsaik, A.K.; Mielke, M.M.; Erwin, P.J.; Knopman, D.S.; Petersen, R.C.; Roberts, R.O. Association of Mediterranean Diet with Mild Cognitive Impairment and Alzheimer’s Disease: A Systematic Review and Meta-Analysis. J. Alzheimer’s Dis. 2014, 39, 271–282. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loughrey, D.G.; Lavecchia, S.; Brennan, S.; Lawlor, B.A.; Kelly, M.E. The Impact of the Mediterranean Diet on the Cognitive Functioning of Healthy Older Adults: A Systematic Review and Meta-Analysis. Adv. Nutr. 2017, 8, 571–586. [Google Scholar] [CrossRef] [PubMed]
- De Filippis, F.; Pellegrini, N.; Vannini, L.; Jeffery, I.B.; La Storia, A.; Laghi, L.; Serrazanetti, D.I.; Di Cagno, R.; Ferrocino, I.; Lazzi, C.; et al. High-Level Adherence to a Mediterranean Diet Beneficially Impacts the Gut Microbiota and Associated Metabolome. Gut 2016, 65, 1812–1821. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Mantrana, I.; Selma-Royo, M.; Alcantara, C.; Collado, M.C. Shifts on Gut Microbiota Associated to Mediterranean Diet Adherence and Specific Dietary Intakes on General Adult Population. Front. Microbiol. 2018, 9, 890. [Google Scholar] [CrossRef]
- Mitsou, E.K.; Kakali, A.; Antonopoulou, S.; Mountzouris, K.C.; Yannakoulia, M.; Panagiotakos, D.B.; Kyriacou, A. Adherence to the Mediterranean Diet Is Associated with the Gut Microbiota Pattern and Gastrointestinal Characteristics in an Adult Population. Br. J. Nutr. 2017, 117, 1645–1655. [Google Scholar] [CrossRef] [Green Version]
- Ghosh, T.S.; Rampelli, S.; Jeffery, I.B.; Santoro, A.; Neto, M.; Capri, M.; Giampieri, E.; Jennings, A.; Candela, M.; Turroni, S.; et al. Mediterranean Diet Intervention Alters the Gut Microbiome in Older People Reducing Frailty and Improving Health Status: The NU-AGE 1-Year Dietary Intervention across Five European Countries. Gut 2020, 69, 1218–1228. [Google Scholar] [CrossRef] [Green Version]
- Doorduijn, A.S.; van de Rest, O.; van der Flier, W.M.; Visser, M.; de van der Schueren, M.A.E. Energy and Protein Intake of Alzheimer’s Disease Patients Compared to Cognitively Normal Controls: Systematic Review. J. Am. Med. Dir. Assoc. 2019, 20, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Cao, G.-Y.; Li, M.; Han, L.; Tayie, F.; Yao, S.-S.; Huang, Z.; Ai, P.; Liu, Y.-Z.; Hu, Y.-H.; Xu, B. Dietary Fat Intake and Cognitive Function among Older Populations: A Systematic Review and Meta-Analysis. J. Prev. Alzheimer’s Dis. 2019, 204–211. [Google Scholar] [CrossRef]
- Ruan, Y.; Tang, J.; Guo, X.; Li, K.; Li, D. Dietary Fat Intake and Risk of Alzheimer’s Disease and Dementia: A Meta-Analysis of Cohort Studies. Curr. Alzheimer’s Res. 2018, 15, 869–876. [Google Scholar] [CrossRef]
- Brainard, J.S.; Jimoh, O.F.; Deane, K.H.O.; Biswas, P.; Donaldson, D.; Maas, K.; Abdelhamid, A.S.; Hooper, L.; Ajabnoor, S.; Alabdulghafoor, F.; et al. Omega-3, Omega-6, and Polyunsaturated Fat for Cognition: Systematic Review and Meta-Analysis of Randomized Trials. J. Am. Med. Dir. Assoc. 2020, 21, 1439.e21–1450.e21. [Google Scholar] [CrossRef] [PubMed]
- Wolters, M.; Ahrens, J.; Romaní-Pérez, M.; Watkins, C.; Sanz, Y.; Benítez-Páez, A.; Stanton, C.; Günther, K. Dietary Fat, the Gut Microbiota, and Metabolic Health—A Systematic Review Conducted within the MyNewGut Project. Clin. Nutr. 2019, 38, 2504–2520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- So, D.; Whelan, K.; Rossi, M.; Morrison, M.; Holtmann, G.; Kelly, J.T.; Shanahan, E.R.; Staudacher, H.M.; Campbell, K.L. Dietary Fiber Intervention on Gut Microbiota Composition in Healthy Adults: A Systematic Review and Meta-Analysis. Am. J. Clin. Nutr. 2018, 107, 965–983. [Google Scholar] [CrossRef] [Green Version]
- Potì, F.; Santi, D.; Spaggiari, G.; Zimetti, F.; Zanotti, I. Polyphenol Health Effects on Cardiovascular and Neurodegenerative Disorders: A Review and Meta-Analysis. Int. J. Mol. Sci. 2019, 20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ammar, A.; Trabelsi, K.; Müller, P.; Bouaziz, B.; Boukhris, O.; Glenn, J.M.; Bott, N.; Driss, T.; Chtourou, H.; Müller, N.; et al. The Effect of (Poly)Phenol-Rich Interventions on Cognitive Functions and Neuroprotective Measures in Healthy Aging Adults: A Systematic Review and Meta-Analysis. J. Clin. Med. 2020, 9. [Google Scholar] [CrossRef] [Green Version]
- Ford, A.H.; Almeida, O.P. Effect of Vitamin B Supplementation on Cognitive Function in the Elderly: A Systematic Review and Meta-Analysis. Drugs Aging 2019, 36, 419–434. [Google Scholar] [CrossRef] [PubMed]
- Goodwill, A.M.; Szoeke, C. A Systematic Review and Meta-Analysis of The Effect of Low Vitamin D on Cognition. J. Am. Geriatr. Soc. 2017, 65, 2161–2168. [Google Scholar] [CrossRef]
- Etgen, T.; Sander, D.; Bickel, H.; Sander, K.; Förstl, H. Vitamin D Deficiency, Cognitive Impairment and Dementia: A Systematic Review and Meta-Analysis. Dement. Geriatr. Cogn. Disord. 2012, 33, 297–305. [Google Scholar] [CrossRef]
- Waterhouse, M.; Hope, B.; Krause, L.; Morrison, M.; Protani, M.M.; Zakrzewski, M.; Neale, R.E. Vitamin D and the Gut Microbiome: A Systematic Review of in Vivo Studies. Eur. J. Nutr. 2019, 58, 2895–2910. [Google Scholar] [CrossRef]
- Mullan, K.; Cardwell, C.R.; McGuinness, B.; Woodside, J.V.; McKay, G.J. Plasma Antioxidant Status in Patients with Alzheimer’s Disease and Cognitively Intact Elderly: A Meta-Analysis of Case-Control Studies. J. Alzheimer’s Dis. 2018, 62, 305–317. [Google Scholar] [CrossRef] [Green Version]
- Rutjes, A.W.; Denton, D.A.; Di Nisio, M.; Chong, L.-Y.; Abraham, R.P.; Al-Assaf, A.S.; Anderson, J.L.; Malik, M.A.; Vernooij, R.W.; Martínez, G.; et al. Vitamin and Mineral Supplementation for Maintaining Cognitive Function in Cognitively Healthy People in Mid and Late Life. Cochrane Database Syst. Rev. 2018, 12. [Google Scholar] [CrossRef]
- Coutts, L.; Ibrahim, K.; Tan, Q.Y.; Lim, S.E.R.; Cox, N.J.; Roberts, H.C. Can Probiotics, Prebiotics and Synbiotics Improve Functional Outcomes for Older People: A Systematic Review. Eur. Geriatr. Med. 2020, 11, 975–993. [Google Scholar] [CrossRef]
- Romijn, A.R.; Rucklidge, J.J. Systematic Review of Evidence to Support the Theory of Psychobiotics. Nutr. Rev. 2015, 73, 675–693. [Google Scholar] [CrossRef] [Green Version]
- Bedani, R.; Isay Saad, S.M.; Sivieri, K. Potential Benefits of Probiotics, Prebiotics, and Synbiotics on the Intestinal Microbiota of the Elderly. In Probiotics, Prebiotics, and Synbiotics; Elsevier: Amsterdam, The Netherlands, 2016; pp. 525–538. ISBN 978-0-12-802189-7. [Google Scholar]
- Lv, T.; Ye, M.; Luo, F.; Hu, B.; Wang, A.; Chen, J.; Yan, J.; He, Z.; Chen, F.; Qian, C.; et al. Probiotics Treatment Improves Cognitive Impairment in Patients and Animals: A Systematic Review and Meta-Analysis. Neurosci. Biobehav. Rev. 2020, S014976342030631X. [Google Scholar] [CrossRef] [PubMed]
- Benton, D.; Williams, C.; Brown, A. Impact of Consuming a Milk Drink Containing a Probiotic on Mood and Cognition. Eur. J. Clin. Nutr. 2007, 61, 355–361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ohsawa, K.; Nakamura, F.; Uchida, N.; Mizuno, S.; Yokogoshi, H. Lactobacillus Helveticus -Fermented Milk Containing Lactononadecapeptide (NIPPLTQTPVVVPPFLQPE) Improves Cognitive Function in Healthy Middle-Aged Adults: A Randomised, Double-Blind, Placebo-Controlled Trial. Int. J. Food Sci. Nutr. 2018, 69, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.-C.; Jin, H.-M.; Cui, Y.; Kim, D.S.; Jung, J.M.; Park, J.-I.; Jung, E.-S.; Choi, E.-K.; Chae, S.-W. Fermented Milk of Lactobacillus Helveticus IDCC3801 Improves Cognitive Functioning during Cognitive Fatigue Tests in Healthy Older Adults. J. Funct. Foods 2014, 10, 465–474. [Google Scholar] [CrossRef]
- Kim, C.-S.; Cha, L.; Sim, M.; Jung, S.; Chun, W.Y.; Baik, H.W.; Shin, D.-M. Probiotic Supplementation Improves Cognitive Function and Mood with Changes in Gut Microbiota in Community-Dwelling Older Adults: A Randomized, Double-Blind, Placebo-Controlled, Multicenter Trial. J. Gerontol. Ser. A 2020, glaa090. [Google Scholar] [CrossRef]
- Inoue, T.; Kobayashi, Y.; Mori, N.; Sakagawa, M.; Xiao, J.-Z.; Moritani, T.; Sakane, N.; Nagai, N. Effect of Combined Bifidobacteria Supplementation and Resistance Training on Cognitive Function, Body Composition and Bowel Habits of Healthy Elderly Subjects. Benef. Microbes 2018, 9, 843–853. [Google Scholar] [CrossRef] [PubMed]
- Den, H.; Dong, X.; Chen, M.; Zou, Z. Efficacy of Probiotics on Cognition, and Biomarkers of Inflammation and Oxidative Stress in Adults with Alzheimer’s Disease or Mild Cognitive Impairment—A Meta-Analysis of Randomized Controlled Trials. Aging 2020, 12, 4010–4039. [Google Scholar] [CrossRef]
- Krüger, J.F.; Hillesheim, E.; Pereira, A.C.S.N.; Camargo, C.Q.; Rabito, E.I. Probiotics for Dementia: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutr. Rev. 2020, nuaa037. [Google Scholar] [CrossRef]
- Hwang, Y.-H.; Park, S.; Paik, J.-W.; Chae, S.-W.; Kim, D.-H.; Jeong, D.-G.; Ha, E.; Kim, M.; Hong, G.; Park, S.-H.; et al. Efficacy and Safety of Lactobacillus Plantarum C29-Fermented Soybean (DW2009) in Individuals with Mild Cognitive Impairment: A 12-Week, Multi-Center, Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Nutrients 2019, 11, 305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kobayashi, Y.; Kuhara, T.; Oki, M.; Xiao, J.-Z. Effects of Bifidobacterium Breve A1 on the Cognitive Function of Older Adults with Memory Complaints: A Randomised, Double-Blind, Placebo-Controlled Trial. Benef. Microbes 2019, 10, 511–520. [Google Scholar] [CrossRef] [PubMed]
- Agahi, A.; Hamidi, G.A.; Daneshvar, R.; Hamdieh, M.; Soheili, M.; Alinaghipour, A.; Esmaeili Taba, S.M.; Salami, M. Does Severity of Alzheimer’s Disease Contribute to Its Responsiveness to Modifying Gut Microbiota? A Double Blind Clinical Trial. Front. Neurol. 2018, 9, 662. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akbari, E.; Asemi, Z.; Daneshvar Kakhaki, R.; Bahmani, F.; Kouchaki, E.; Tamtaji, O.R.; Hamidi, G.A.; Salami, M. Effect of Probiotic Supplementation on Cognitive Function and Metabolic Status in Alzheimer’s Disease: A Randomized, Double-Blind and Controlled Trial. Front. Aging Neurosci. 2016, 8. [Google Scholar] [CrossRef] [Green Version]
- Tamtaji, O.R.; Heidari-soureshjani, R.; Mirhosseini, N.; Kouchaki, E.; Bahmani, F.; Aghadavod, E.; Tajabadi-Ebrahimi, M.; Asemi, Z. Probiotic and Selenium Co-Supplementation, and the Effects on Clinical, Metabolic and Genetic Status in Alzheimer’s Disease: A Randomized, Double-Blind, Controlled Trial. Clin. Nutr. 2019, 38, 2569–2575. [Google Scholar] [CrossRef] [PubMed]
- Buigues, C.; Fernández-Garrido, J.; Pruimboom, L.; Hoogland, A.; Navarro-Martínez, R.; Martínez-Martínez, M.; Verdejo, Y.; Mascarós, M.; Peris, C.; Cauli, O. Effect of a Prebiotic Formulation on Frailty Syndrome: A Randomized, Double-Blind Clinical Trial. Int. J. Mol. Sci. 2016, 17, 932. [Google Scholar] [CrossRef]
- Louzada, E.R.; Ribeiro, S.M.L. Synbiotic Supplementation, Systemic Inflammation, and Symptoms of Brain Disorders in Elders: A Secondary Study from a Randomized Clinical Trial. Nutr. Neurosci. 2020, 23, 93–100. [Google Scholar] [CrossRef]
- Bowyer, R.C.E.; Jackson, M.A.; Pallister, T.; Skinner, J.; Spector, T.D.; Welch, A.A.; Steves, C.J. Use of Dietary Indices to Control for Diet in Human Gut Microbiota Studies. Microbiome 2018, 6, 77. [Google Scholar] [CrossRef] [PubMed]
- Mueller, S.; Saunier, K.; Hanisch, C.; Norin, E.; Alm, L.; Midtvedt, T.; Cresci, A.; Silvi, S.; Orpianesi, C.; Verdenelli, M.C.; et al. Differences in Fecal Microbiota in Different European Study Populations in Relation to Age, Gender, and Country: A Cross-Sectional Study. Appl. Environ. Microbiol. 2006, 72, 1027–1033. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suez, J.; Zmora, N.; Elinav, E. Probiotics in the Next-Generation Sequencing Era. Gut Microbes 2020, 11, 77–93. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.J.; Zheng, J.J.; Kang, J.W.; Saboe, A.; Knights, D.; Zivkovic, A.M. A Guide to Diet-Microbiome Study Design. Front. Nutr. 2020, 7, 79. [Google Scholar] [CrossRef]
- Sanders, M.E.; Akkermans, L.M.A.; Haller, D.; Hammerman, C.; Heimbach, J.T.; Hörmannsperger, G.; Huys, G. Safety Assessment of Probiotics for Human Use. Gut Microbes 2010, 1, 164–185. [Google Scholar] [CrossRef]
- Hibberd, P.L.; Kleimola, L.; Fiorino, A.-M.; Botelho, C.; Haverkamp, M.; Andreyeva, I.; Poutsiaka, D.; Fraser, C.; Solano-Aguilar, G.; Snydman, D.R. No Evidence of Harms of Probiotic Lactobacillus Rhamnosus GG ATCC 53103 in Healthy Elderly—A Phase I Open Label Study to Assess Safety, Tolerability and Cytokine Responses. PLoS ONE 2014, 9, e113456. [Google Scholar] [CrossRef]
- Liu, S.; Gao, J.; Zhu, M.; Liu, K.; Zhang, H.-L. Gut Microbiota and Dysbiosis in Alzheimer’s Disease: Implications for Pathogenesis and Treatment. Mol. Neurobiol. 2020, 57, 5026–5043. [Google Scholar] [CrossRef] [PubMed]
- Martin, C.R.; Osadchiy, V.; Kalani, A.; Mayer, E.A. The Brain-Gut-Microbiome Axis. Cell Mol. Gastroenterol. Hepatol. 2018, 6, 133–148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quigley, E.M.M. Microbiota-Brain-Gut Axis and Neurodegenerative Diseases. Curr. Neurol. Neurosci. Rep. 2017, 17, 94. [Google Scholar] [CrossRef]
- Gao, W.; Baumgartel, K.L.; Alexander, S.A. The Gut Microbiome as a Component of the Gut–Brain Axis in Cognitive Health. Biol. Res. Nurs. 2020, 22, 485–494. [Google Scholar] [CrossRef]
- Zhu, F.; Li, C.; Chu, F.; Tian, X.; Zhu, J. Target Dysbiosis of Gut Microbes as a Future Therapeutic Manipulation in Alzheimer’s Disease. Front. Aging Neurosci. 2020, 12. [Google Scholar] [CrossRef]
- Ticinesi, A.; Tana, C.; Nouvenne, A.; Prati, B.; Lauretani, F.; Meschi, T. Gut Microbiota, Cognitive Frailty and Dementia in Older Individuals: A Systematic Review. Clin. Interv. Aging 2018, 13, 1497–1511. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferrucci, L.; Fabbri, E. Inflammageing: Chronic Inflammation in Ageing, Cardiovascular Disease, and Frailty. Nat. Rev. Cardiol. 2018, 15, 505–522. [Google Scholar] [CrossRef]
- Saji, N.; Murotani, K.; Hisada, T.; Kunihiro, T.; Tsuduki, T.; Sugimoto, T.; Kimura, A.; Niida, S.; Toba, K.; Sakurai, T. Relationship between Dementia and Gut Microbiome-Associated Metabolites: A Cross-Sectional Study in Japan. Sci. Rep. 2020, 10, 8088. [Google Scholar] [CrossRef] [PubMed]
- Long, J.M.; Holtzman, D.M. Alzheimer Disease: An Update on Pathobiology and Treatment Strategies. Cell 2019, 179, 312–339. [Google Scholar] [CrossRef] [PubMed]
- Bostanciklioğlu, M. The Role of Gut Microbiota in Pathogenesis of Alzheimer’s Disease. J. Appl. Microbiol. 2019, 127, 954–967. [Google Scholar] [CrossRef] [PubMed]
- Jagust, W. Imaging the Evolution and Pathophysiology of Alzheimer Disease. Nat. Rev. Neurosci. 2018, 19, 687–700. [Google Scholar] [CrossRef] [PubMed]
- Friedland, R.P.; Chapman, M.R. The Role of Microbial Amyloid in Neurodegeneration. PLoS Pathog. 2017, 13. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.M.; Lukiw, W.J. Microbial-Generated Amyloids and Alzheimer’s Disease (AD). Front. Aging Neurosci. 2015, 7. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Y.; Dua, P.; Lukiw, W. Microbial Sources of Amyloid and Relevance to Amyloidogenesis and Alzheimer’s Disease (AD). J. Alzheimer’s Dis. Parkinsonism 2015, 5, 177. [Google Scholar] [CrossRef] [Green Version]
- Friedland, R.P. Mechanisms of Molecular Mimicry Involving the Microbiota in Neurodegeneration. J. Alzheimer’s Dis. 2015, 45, 349–362. [Google Scholar] [CrossRef] [Green Version]
- Hufnagel, D.A.; Tükel, Ç.; Chapman, M.R. Disease to Dirt: The Biology of Microbial Amyloids. PLoS Pathog. 2013, 9, e1003740. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schwartz, R.H. Marijuana: A Decade and a Half Later, Still a Crude Drug with Underappreciated Toxicity. Pediatrics 2002, 109, 284–289. [Google Scholar] [CrossRef]
- Kowalski, K.; Mulak, A. Brain-Gut-Microbiota Axis in Alzheimer’s Disease. J. Neurogastroenterol. Motil. 2019, 25, 48–60. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Y.; Jaber, V.; Lukiw, W.J. Secretory Products of the Human GI Tract Microbiome and Their Potential Impact on Alzheimer’s Disease (AD): Detection of Lipopolysaccharide (LPS) in AD Hippocampus. Front. Cell Infect. Microbiol. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Zhan, X.; Stamova, B.; Sharp, F.R. Lipopolysaccharide Associates with Amyloid Plaques, Neurons and Oligodendrocytes in Alzheimer’s Disease Brain: A Review. Front. Aging Neurosci. 2018, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Derkacz, A.; Olczyk, P.; Komosinska-Vassev, K. Diagnostic Markers for Nonspecific Inflammatory Bowel Diseases. Dis. Markers 2018, 2018. [Google Scholar] [CrossRef] [PubMed]
- Vatn, M.H.; Sandvik, A.K. Inflammatory Bowel Disease. Scand. J. Gastroenterol. 2015, 50, 748–762. [Google Scholar] [CrossRef]
- Leblhuber, F.; Geisler, S.; Steiner, K.; Fuchs, D.; Schütz, B. Elevated Fecal Calprotectin in Patients with Alzheimer’s Dementia Indicates Leaky Gut. J. Neural. Transm. 2015, 122, 1319–1322. [Google Scholar] [CrossRef]
- Chang, K.-A.; Kim, H.J.; Suh, Y.-H. The Role of S100a9 in the Pathogenesis of Alzheimer’s Disease: The Therapeutic Effects of S100a9 Knockdown or Knockout. Neurodegener. Dis. 2012, 10, 27–29. [Google Scholar] [CrossRef] [PubMed]
- Horvath, I.; Jia, X.; Johansson, P.; Wang, C.; Moskalenko, R.; Steinau, A.; Forsgren, L.; Wågberg, T.; Svensson, J.; Zetterberg, H.; et al. Pro-Inflammatory S100A9 Protein as a Robust Biomarker Differentiating Early Stages of Cognitive Impairment in Alzheimer’s Disease. ACS Chem. Neurosci. 2016, 7, 34–39. [Google Scholar] [CrossRef]
- Minter, M.R.; Taylor, J.M.; Crack, P.J. The Contribution of Neuroinflammation to Amyloid Toxicity in Alzheimer’s Disease. J. Neurochem. 2016, 136, 457–474. [Google Scholar] [CrossRef] [PubMed]
- Rapsinski, G.J.; Wynosky-Dolfi, M.A.; Oppong, G.O.; Tursi, S.A.; Wilson, R.P.; Brodsky, I.E.; Tükel, Ç. Toll-like Receptor 2 and NLRP3 Cooperate to Recognize a Functional Bacterial Amyloid, Curli. Infect. Immun. 2015, 83, 693–701. [Google Scholar] [CrossRef] [Green Version]
- Nishimori, J.H.; Newman, T.N.; Oppong, G.O.; Rapsinski, G.J.; Yen, J.-H.; Biesecker, S.G.; Wilson, R.P.; Butler, B.P.; Winter, M.G.; Tsolis, R.M.; et al. Microbial Amyloids Induce Interleukin 17A (IL-17A) and IL-22 Responses via Toll-Like Receptor 2 Activation in the Intestinal Mucosa. Infect. Immun. 2012, 80, 4398–4408. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Erickson, M.A.; Dohi, K.; Banks, W.A. Neuroinflammation: A Common Pathway in CNS Diseases as Mediated at the Blood-Brain Barrier. Neuroimmunomodulation 2012, 19, 121–130. [Google Scholar] [CrossRef] [Green Version]
- Garcez, M.L.; Jacobs, K.R.; Guillemin, G.J. Microbiota Alterations in Alzheimer’s Disease: Involvement of the Kynurenine Pathway and Inflammation. Neurotox. Res. 2019, 36, 424–436. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, M.J.; Guillemin, G.J.; Teipel, S.J.; Buerger, K.; Hampel, H. Increased 3-Hydroxykynurenine Serum Concentrations Differentiate Alzheimer’s Disease Patients from Controls. Eur. Arch. Psychiatry Clin. Neurosci. 2013, 263, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Smoller, J.W.R.S.; Lee, P.H.; Neale, B.; Nurnberger, J.I.; Santangelo, S.; Sullivan, P.F.; Schulze, T.G.; Thapar, A.; Anney, R.; Buitelaar, J.K.; et al. Identification of Risk Loci with Shared Effects on Five Major Psychiatric Disorders: A Genome-Wide Analysis. Lancet 2013, 381, 1371–1379. [Google Scholar] [CrossRef]
- Giil, L.M.; Midttun, Ø.; Refsum, H.; Ulvik, A.; Advani, R.; Smith, A.D.; Ueland, P.M. Kynurenine Pathway Metabolites in Alzheimer’s Disease. J. Alzheimer’s Dis. 2017, 60, 495–504. [Google Scholar] [CrossRef]
- Guillemin, G.J.; Williams, K.R.; Smith, D.G.; Smythe, G.A.; Croitoru-Lamoury, J.; Brew, B.J. Quinolinic Acid in the Pathogenesis of Alzheimer’s Disease. Adv. Exp. Med. Biol. 2003, 527, 167–176. [Google Scholar] [PubMed]
- Widner, B.; Leblhuber, F.; Walli, J.; Tilz, G.P.; Demel, U.; Fuchs, D. Tryptophan Degradation and Immune Activation in Alzheimer’s Disease. J. Neural. Transm. 2000, 107, 343–353. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.-J.; Zhang, X.; Chen, W.-W. Role of Oxidative Stress in Alzheimer’s Disease. Biomed. Rep. 2016, 4, 519–522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharon, G.; Garg, N.; Debelius, J.; Knight, R.; Dorrestein, P.C.; Mazmanian, S.K. Specialized Metabolites from the Microbiome in Health and Disease. Cell Metab. 2014, 20, 719–730. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rowland, I.; Gibson, G.; Heinken, A.; Scott, K.; Swann, J.; Thiele, I.; Tuohy, K. Gut Microbiota Functions: Metabolism of Nutrients and Other Food Components. Eur. J. Nutr. 2018, 57, 1–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kullisaar, T.; Zilmer, M.; Mikelsaar, M.; Vihalemm, T.; Annuk, H.; Kairane, C.; Kilk, A. Two Antioxidative Lactobacilli Strains as Promising Probiotics. Int. J. Food Microbiol. 2002, 72, 215–224. [Google Scholar] [CrossRef]
- Dumitrescu, L.; Popescu-Olaru, I.; Cozma, L.; Tulbă, D.; Hinescu, M.E.; Ceafalan, L.C.; Gherghiceanu, M.; Popescu, B.O. Oxidative Stress and the Microbiota-Gut-Brain Axis. Oxid. Med. Cell. Longev. 2018, 2018. [Google Scholar] [CrossRef] [Green Version]
- Shao, C.; Xiong, S.; Li, G.-M.; Gu, L.; Mao, G.; Markesbery, W.R.; Lovell, M.A. Altered 8-Oxoguanine Glycosylase in Mild Cognitive Impairment and Late-Stage Alzheimer’s Disease Brain. Free. Radic. Biol. Med. 2008, 45, 813–819. [Google Scholar] [CrossRef] [Green Version]
- Pocernich, C.B.; Butterfield, D.A. Elevation of Glutathione as a Therapeutic Strategy in Alzheimer Disease. Biochim. Biophys. Acta 2012, 1822, 625–630. [Google Scholar] [CrossRef] [Green Version]
- Marcus, D.L.; Thomas, C.; Rodriguez, C.; Simberkoff, K.; Tsai, J.S.; Strafaci, J.A.; Freedman, M.L. Increased Peroxidation and Reduced Antioxidant Enzyme Activity in Alzheimer’s Disease. Exp. Neurol. 1998, 150, 40–44. [Google Scholar] [CrossRef]
- Oniszczuk, A.; Oniszczuk, T.; Gancarz, M.; Szymańska, J. Role of Gut Microbiota, Probiotics and Prebiotics in the Cardiovascular Diseases. Molecules 2021, 26, 1172. [Google Scholar] [CrossRef] [PubMed]
- Arora, K.; Green, M.; Prakash, S. The Microbiome and Alzheimer’s Disease: Potential and Limitations of Prebiotic, Synbiotic, and Probiotic Formulations. Front. Bioeng. Biotechnol. 2020, 8, 537847. [Google Scholar] [CrossRef] [PubMed]
- Ton, A.M.M.; Campagnaro, B.P.; Alves, G.A.; Aires, R.; Côco, L.Z.; Arpini, C.M.; Guerra e Oliveira, T.; Campos-Toimil, M.; Meyrelles, S.S.; Pereira, T.M.C.; et al. Oxidative Stress and Dementia in Alzheimer’s Patients: Effects of Synbiotic Supplementation. Oxid. Med. Cell. Longev. 2020, 2020, e2638703. [Google Scholar] [CrossRef] [PubMed]
| Terms | Definitions |
|---|---|
| “Dysbiosis” | is defined as a loss of beneficial microbes, expansion of pathobionts or potentially harmful microorganisms, and a loss of microbial diversity [20]. |
| “Microbiota” | is defined as the microbial taxon (bacteria, archaea, or lower eukaryotes) associated with humans health and disease [7]. |
| “Microbiome” | is defined as the catalog of microorganisms, their genomes, and the surrounding environmental condition [7], but we are lacking a clear commonly agreed definition [21]. |
| “Postbiotics” | are defined as “preparation of inanimate microorganisms and/or their components that confers a health benefit on the host” [22] they are also known as “non-viable probiotics”, “ghost probiotics”, “paraprobiotics”, and “inactivated probiotics”, they have also ability to deliver health benefits if consumed with prebiotics or foods throughout their shelf life [23]. |
| “Prebiotics” | defined as “a selectively fermented ingredient that allows specific changes, both in the composition and/or activity in the gastrointestinal microflora that confers benefits upon host wellbeing and health” [24]. |
| “Probiotics” | are defined as “live microorganisms which when administered in adequate amounts confer a health benefit on the host” [25]. |
| “Psychobiotics” | are defined as live bacteria which, when ingested in adequate amounts, produces a mental health benefits [26]. |
| “Synbiotic” | contains combined both a probiotics and prebiotics that work synergistically and have beneficially affect the host, and this effect is higher than that of the probiotic alone [27]. |
| Phyla | Class | Order | Family | Genus | Species | Cognition | References | ||
|---|---|---|---|---|---|---|---|---|---|
| MCI | D | AD | |||||||
| Firmicutes | Clostridia | Clostridiales | ↓ | [52] | |||||
| Clostridiaceae | ↓ | ↓ | [52] | ||||||
| Clostridium | ↓ | [49] | |||||||
| Eubacteriales | Ruminococcaceae | ↓ ↓ | [52] [49] | ||||||
| Ruminococcus | ↓ | [52] | |||||||
| Lachnospiraceae | - unclassified | Lachnospiraceae bacterium NK4A136 | ↓ | ↓ ↓ | ↓ | [52] [51] | |||
| Blautia | ↓ | ↓ ↑ | [52] [49] | ||||||
| Eubacteriaceae | Eubacterium | E. rectale | ↓ ↓ | ↑ ↑ | [51] [50] | ||||
| Erysipelotrichia | Erysipelotrichales | Turicibacteraceae | Turicibacter | ↓ | [49] | ||||
| Negativicutes | Veillonellales | Veillonellaceae | ↑ | [52] | |||||
| Dialister | ↓ | [49] | |||||||
| Selenomonadales | Acidaminococcaceae | Phascolarctobacterium | ↑ | [49] | |||||
| Bacilli | Lactibacillales | Lactobacillaceae | Lactobacillus | ↑ | [48] | ||||
| Staphylococcales | Gemellaceae | Gemella | ↑ | [49] | |||||
| Bacteroidetes | Bacteroidia | Bacteroidales | Bacteroidaceae | ↑ ↑ | [52] [49] | ||||
| Bacteroides | ↑ ↑ | ↓ | ↑ ↑ ↑ | [47] [48] [49] [52] | |||||
| B. fragilis | ↓ | [50] | |||||||
| Rikenellaceae | Alistipes | ↑ | [49] | ||||||
| Actinobacteria | Actinomycetia | Bifidobacteriales | Bifidobacteriaceae | Bifidobacterium | ↑ | ↓ | [47] [49] | ||
| Coriobacteriia | Eggerthellales | Eggerthellaceae | Adlercreutzia | ↓ | [49] | ||||
| Proteobacteria | Gammaproteobacteria | Enterobacteriales | Enterobacteriaceae | ↑ | [52] | ||||
| Escherichia/Shigella | ↑ | [50] | |||||||
| Deltaproteobacteria | Desulfovibrionales | Desulfovibrionaceae | Bilophila | ↑ | [49] | ||||
| Country [References] | N (% Women) Cases | N (% Women) Controls | Age Cases | Age Controls | Cognitive Function Assessment/Diagnosis | Cognitive Impairment | Microbiome Assessment |
|---|---|---|---|---|---|---|---|
| Japan [49] | 94 (85) | 34 (49) | 77, 74–82 | 76, 68–80 | MMSE/CDR | Dementia | Fecal samples, bacterial 16 s rRNA Sequencing |
| Japan [50] | 61 (54) | 21 (48) | 77, 73–81 | 69, 61–76 | MMSE/CDR | MCI | Fecal samples, bacterial 16 s rRNA Sequencing |
| USA [51] | 25 (72) | 25 (68) | 69 ± 7 | 71 ± 7 | CDR | AD | Fecal samples, bacterial 16 s rRNA Sequencing |
| Austria [53] | 23 (65) | 18 (61) | 88 | 75 | MMSE | Dementia | Fecal samples, bacterial 16 s rRNA Sequencing |
| China [54] | 32 (56) aMCI 33 (42) AD | 32 (50) | 70 ± 11 aMCI 75 ± 11 AD | 77 ± 9 | MMSE, CDR, MoCA | aMCI AD | Fecal samples, bacterial 16 s rRNA Sequencing |
| Italy [52] | 40 (50) Amy+ 33 (52) Amy- | 10 (60) | 71 ± 7 Amy+ 70 ± 7 Amy- | 68 ± 8 | MMSE | AD (Amy+ Amy-) | Fecal samples, qPCR Assay, StepOnePlus instrument |
| Nutrients | Type of Study/Impact on Cognitive Function | Type of Study/Impact on Gut Microbiota |
|---|---|---|
| proteins | Observational studies/no statistically significant differences in protein intake levels in AD patients and healthy controls [73] | No meta-analysis and systematic reviews were identified. |
| fats | Prospective studies/significantly higher risk of AD and dementia development in patients with higher SFA, while the results regarding unsaturated fatty acids intake were not statistically significant [74,75] RCT/no protective effects of omega-3 supplements in the prevention of cognitive decline, which is contradictory with previous findings [76] | RCT, observational studies (cross-sectional)/higher intake of fat and SFA was associated with poorer gut microbiota diversity, diets high in MUFA were associated with decreased total bacterial number, while whereas high PUFA intake had no effect on gut microbiota diversity and richness [77] |
| fiber | No meta-analysis and systematic reviews were identified. | RCT/dietary fiber resulted in higher abundance of Bifidobacterium and Lactobacillus spp. but does not affect α-diversity [78] |
| polyphenols | RCT/no definitive confirmation of the preventive effect of polyphenols on cognitive disorders [79] nor of neuroprotective effect of polyphenols supplementation in aging adults [80]. | No meta-analysis and systematic reviews were identified.. |
| vitamin B | RCT/no effect of B vitamins on cognitive function in older adults with or without cognitive impairment[81] | No meta-analysis and systematic reviews were identified. |
| vitamin D | Observational (cross-sectional and longitudinal cohort) studies/low vitamin D status is related to poorer cognition [82] and increased risk of cognitive impairment [83]. Interventional studies with control group/no significant effect of vitamin D supplementation on cognition [82] | RCT, interventional and observational studies/vitamin D influences the composition of the gastrointestinal microbiome (varied and limited studies) [84] |
| antioxidant vitamin | Case–control studies/significantly lower plasma levels of α-carotene, β-carotene, lycopene, lutein, vitamin A, C, and E in AD patients [85] RCT/no good evidence that supplementation can preserve cognitive function, prevent dementia [86] | No meta-analysis and systematic reviews were identified. |
| Intervention | Cognitive Impairment | Control Group | Study Group | Cognitive Function Assessment/Diagnosis | Microbiome Assessment | Other Assessed Parameters | Country [References] | ||
|---|---|---|---|---|---|---|---|---|---|
| N | Age | N | Age | ||||||
| PROBIOTICS | |||||||||
| L. casei Shirota 65 mL milk drink 6.5 × 109 CFU 3 wk. | healthy | 66 | 61.8 | 66 | 61.8 | Memory (Wechsler Memory Scale) Retrieval from long-term memory Verbal fluency | no | eating-associated behaviour NART | UK [91] |
| L. helveticus-fermented milk drink 190 g/day 8 wk. | healthy | 29 | 57.8 ± 5.9 | 31 | 58.5 ± 6.5 | RBANS | no | POMS | Japan [92] |
| L. helveticus-fermented milk 500 mg, 1000 mg, 200 mg 12 wk. | healthy | 10 | 64.5 ±4.8 | 10 7 9 | 64.5 ± 2.2 64.4 ± 4.5 66.6 ± 5.0 | neuropsychological test battery (DST, VLT, SRT) | no | cognitive fatigue PSS GDS-SF BDNF WBV | Korea [93] |
| B. bifidum, B. longum 1 × 109 CFU 12 wk. | healthy | 26 | 72.0 | 27 | 71.1 | CERAD-K | yes | BDNF | Korea [94] |
| B. longum, B. infantis, B. breve, B. breve 1.25 × 1010 CFU 12 wk. | healthy | 18 | 70.9 ± 3.2 | 20 | 69.9 ± 3.0 | MoCA-J Flanker task | no | PHQ-9, GAD-7, MNA, energy intake, BMI, height, weight, blood pressure, bowel movement characteristics | Japan [95] |
| L. plantarum ≥ 1.25 × 1010 CFU/day fermented soybean 2 capsules, once a day 12 wk. | MCI | 50 | 69.2 ± 7.0 | 50 | 68.0 ± 5.1 | CNT VLT ACPT DST | yes | BDNF, height, weight, blood pressure and pulse rate, complete blood cell count and blood parameters | South Korea [98] |
| B. breve > 1.0 × 1010 CFU/day 2 capsules 12 wk. | MCI | 60 | 61.6 ± 6.4 | 61 | 61.5 ± 6.8 | MMSE RBANS | no | hs-CRP haematological and biological blood parameters | Japan [99] |
| L. fermentum, L. plantarum, L. acidophilus, B. lactis, B. longum, B. bifidum 3 × 109 CFU/g of each 2 capsules, once a day 12 wk. | AD | 23 | 80.6 ± 1.8 | 25 | 79.7 ± 1.7 | TYM | no | TAC, GSH, MDA, Il-6, Il-10, TNF-a, NO, 8-OHdG, weight, BMI | Iran [100] |
| L. acidophilus, L. casei, L. fermentum B. bifidum, 2 × 109 CFU/g of each 200 mL probiotic milk/day 12 wk. | AD | 30 | 82 ± 1.7 | 30 | 77.7 ± 2.6 | MMSE | no | TAC, GSH, hs-CRP, MDA, NO, HOMA-B, HOMA-IR, QUICK, FPG, TG, TC, LDL, HDL, VLDL, TC/HDL, insulin, weight, BMI dietary intakes (3-day food records) | Iran [101] |
| L. acidophilus, B. bifidum, B. longum 2 × 109 CFU/g each for probiotic capsule + selenium 200 ng/day 12 wk. | AD | 52 | 78.8 ± 10.2 | 27 | 76.2 ± 8.1 | MMSE | no | TAC, GSH, hsCRP, insulin, HOMA-IR, QUICKI, TG, NO, FPG, MDA, TC, LDL, VLDL, HDL, TC/HDL, Gene expression related to inflammation, insulin and lipid metabolism BMI, weight, dietary intakes (3-day food records) | Iran [102] |
| PREBIOTICS | |||||||||
| Darmocare Pre® (inulin + fructooligosaccharides) 13 wk. | non-demented | 22 | 73.4 ± 1.8 | 28 | 74.2 ± 1.6 | MMSE | no | Frailty Functional Impairment Sleep quality Blood analysis and haemogram | Spain [103] |
| SYNBIOTIC | |||||||||
| L. paracasei, L. ramnosus, L. acidophilus, B. lactis (108–109 CFU of each) + fructooligosaccharide twice a day 24 wk. | apparently healthy | 24 | 77.0 ± 1.3 | 25 | 67.9 ± 4.5 | MMSE | no | GDS-15, % of body fat, IL-6, TNF-α, IL-10, DAO, IFABP, LPS | Brazil [104] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Białecka-Dębek, A.; Granda, D.; Szmidt, M.K.; Zielińska, D. Gut Microbiota, Probiotic Interventions, and Cognitive Function in the Elderly: A Review of Current Knowledge. Nutrients 2021, 13, 2514. https://doi.org/10.3390/nu13082514
Białecka-Dębek A, Granda D, Szmidt MK, Zielińska D. Gut Microbiota, Probiotic Interventions, and Cognitive Function in the Elderly: A Review of Current Knowledge. Nutrients. 2021; 13(8):2514. https://doi.org/10.3390/nu13082514
Chicago/Turabian StyleBiałecka-Dębek, Agata, Dominika Granda, Maria Karolina Szmidt, and Dorota Zielińska. 2021. "Gut Microbiota, Probiotic Interventions, and Cognitive Function in the Elderly: A Review of Current Knowledge" Nutrients 13, no. 8: 2514. https://doi.org/10.3390/nu13082514
APA StyleBiałecka-Dębek, A., Granda, D., Szmidt, M. K., & Zielińska, D. (2021). Gut Microbiota, Probiotic Interventions, and Cognitive Function in the Elderly: A Review of Current Knowledge. Nutrients, 13(8), 2514. https://doi.org/10.3390/nu13082514








